Antibacterial Chitosan Nanofiber Thin Films with Bacitracin Zinc Salt
Abstract
1. Introduction
2. Materials and Methods
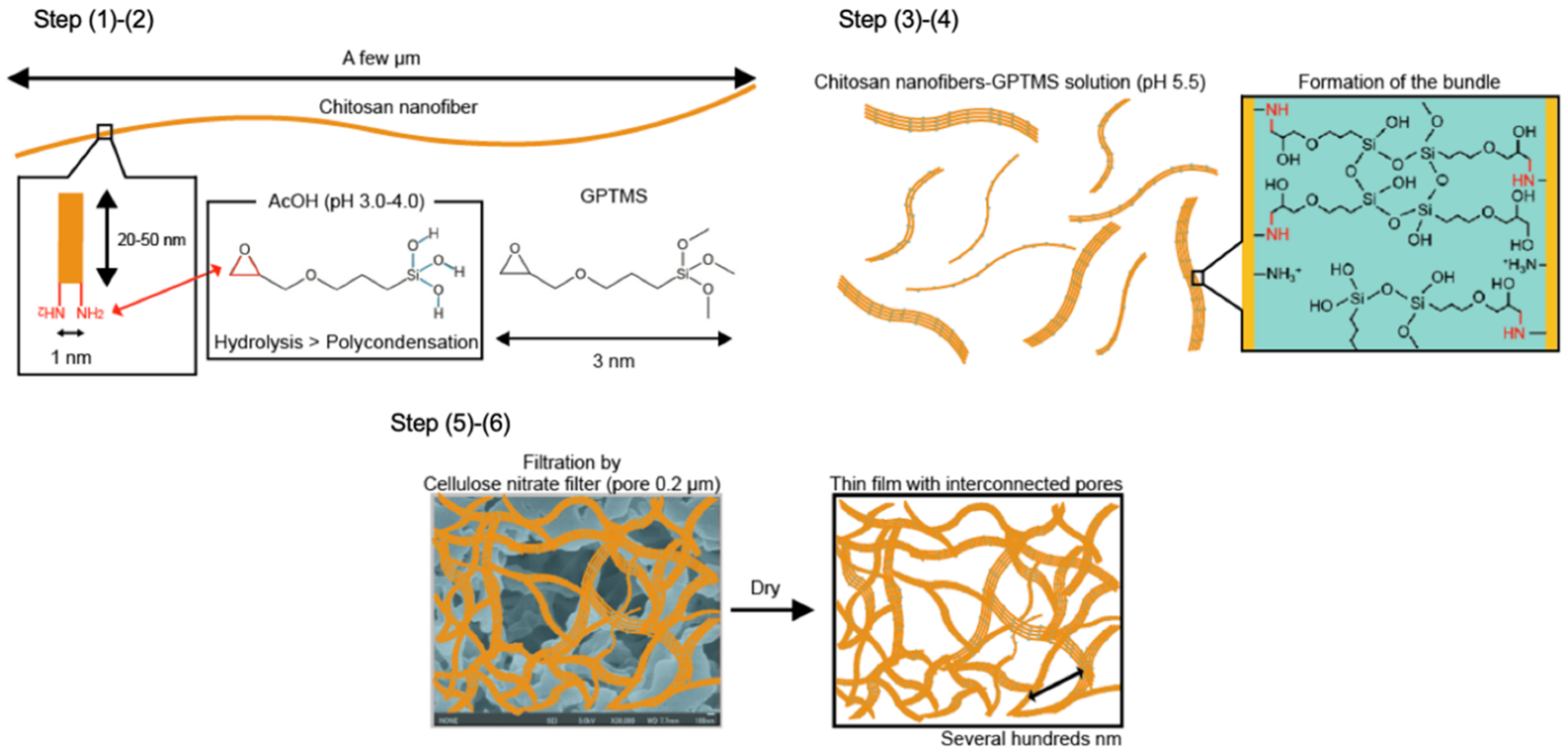
2.1. Preparation of the Thin Films
2.2. Thickness and Surface Morphology of Thin Films
2.3. Surface Wettability and Water Vapor Adsorption of Thin Films
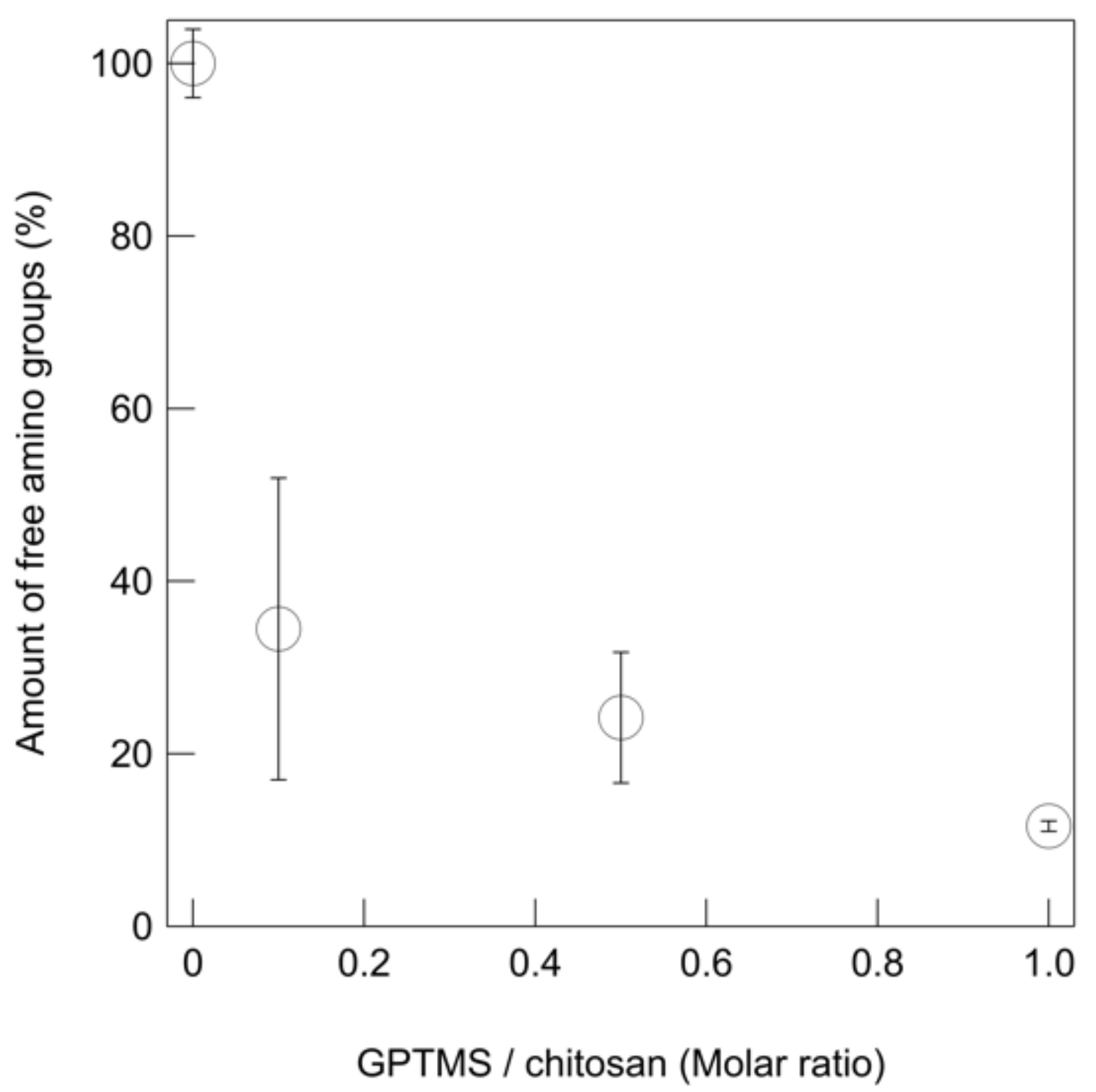
2.4. Residual Amino Groups of Thin Films
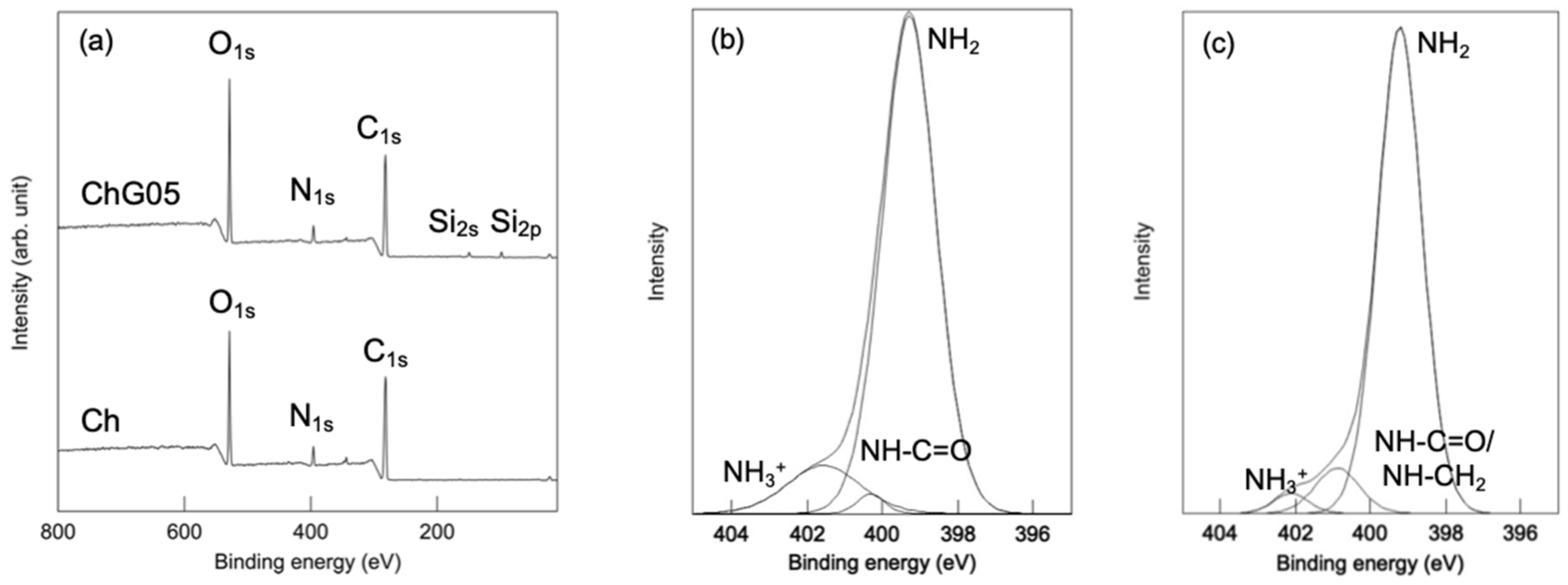
2.5. Microstructure of Thin Films
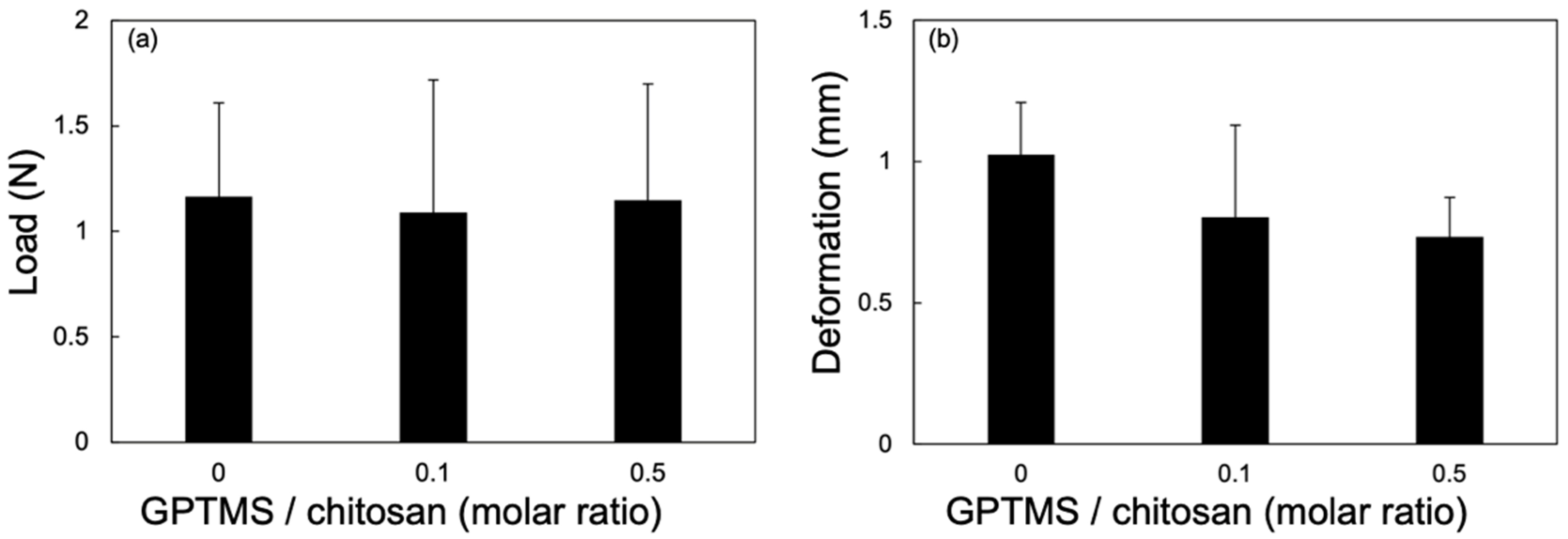
2.6. Mechanical Properties of the Thin Films
2.7. Release of Amount of BZ from the Thin Films
2.8. In Vitro Antibacterial Properties Test
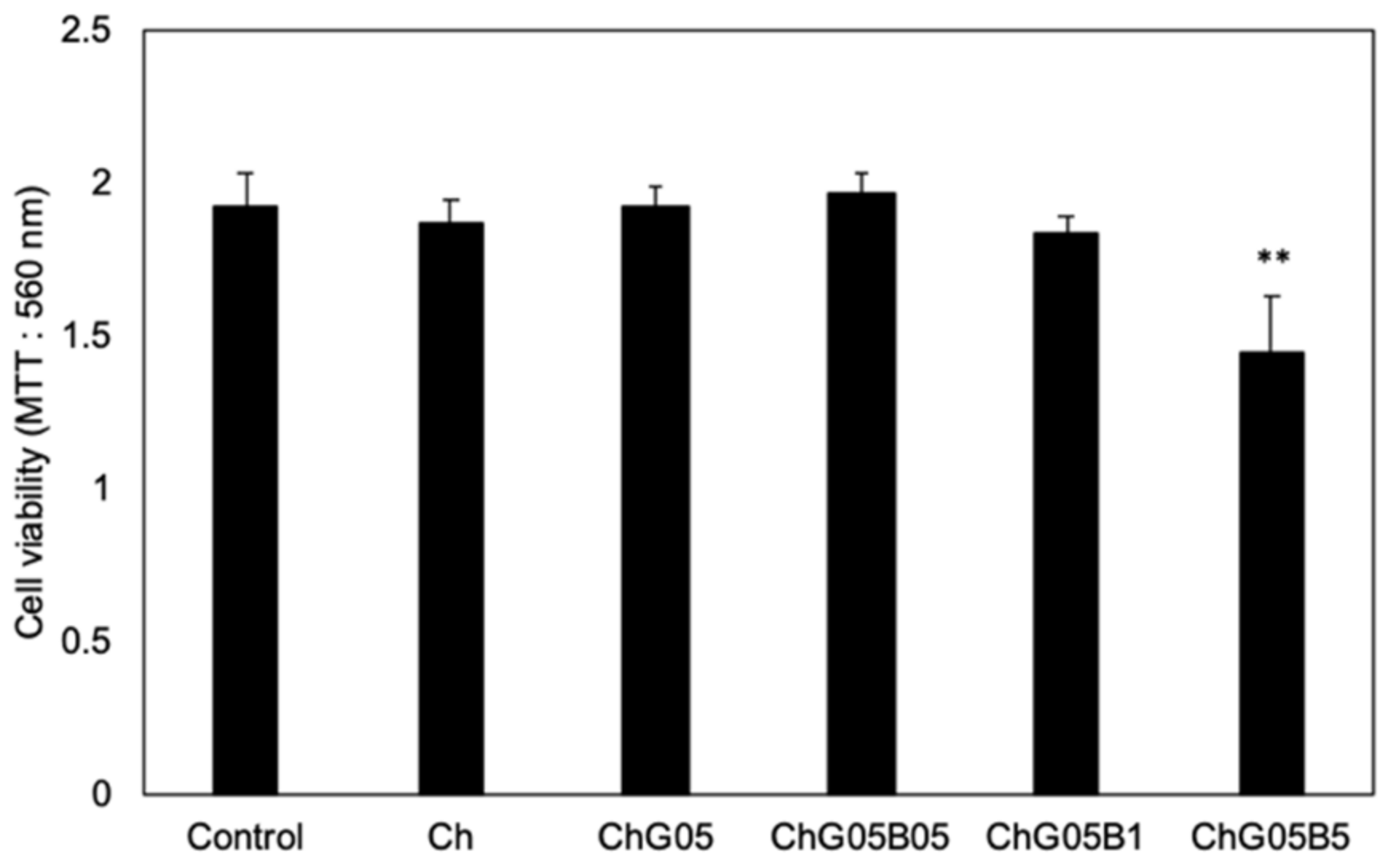
2.9. In Vitro Cytotoxicity Test
2.10. Statistical Analysis
3. Results and Discussion
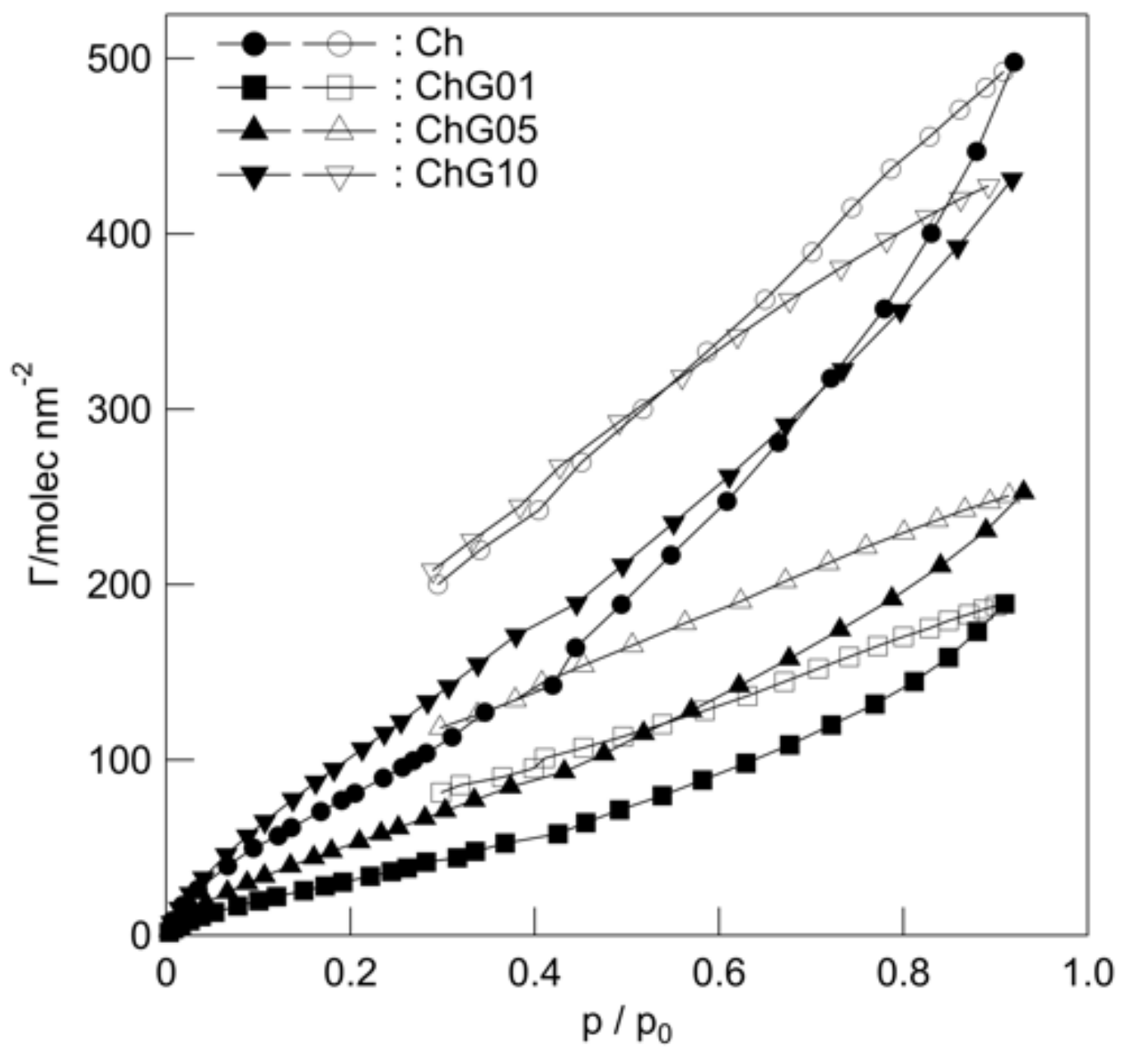
3.1. Surface Morphology and Hydrophilicity of the Thin Films
3.2. Microstructure and Mechanical Properties of the Thin Films
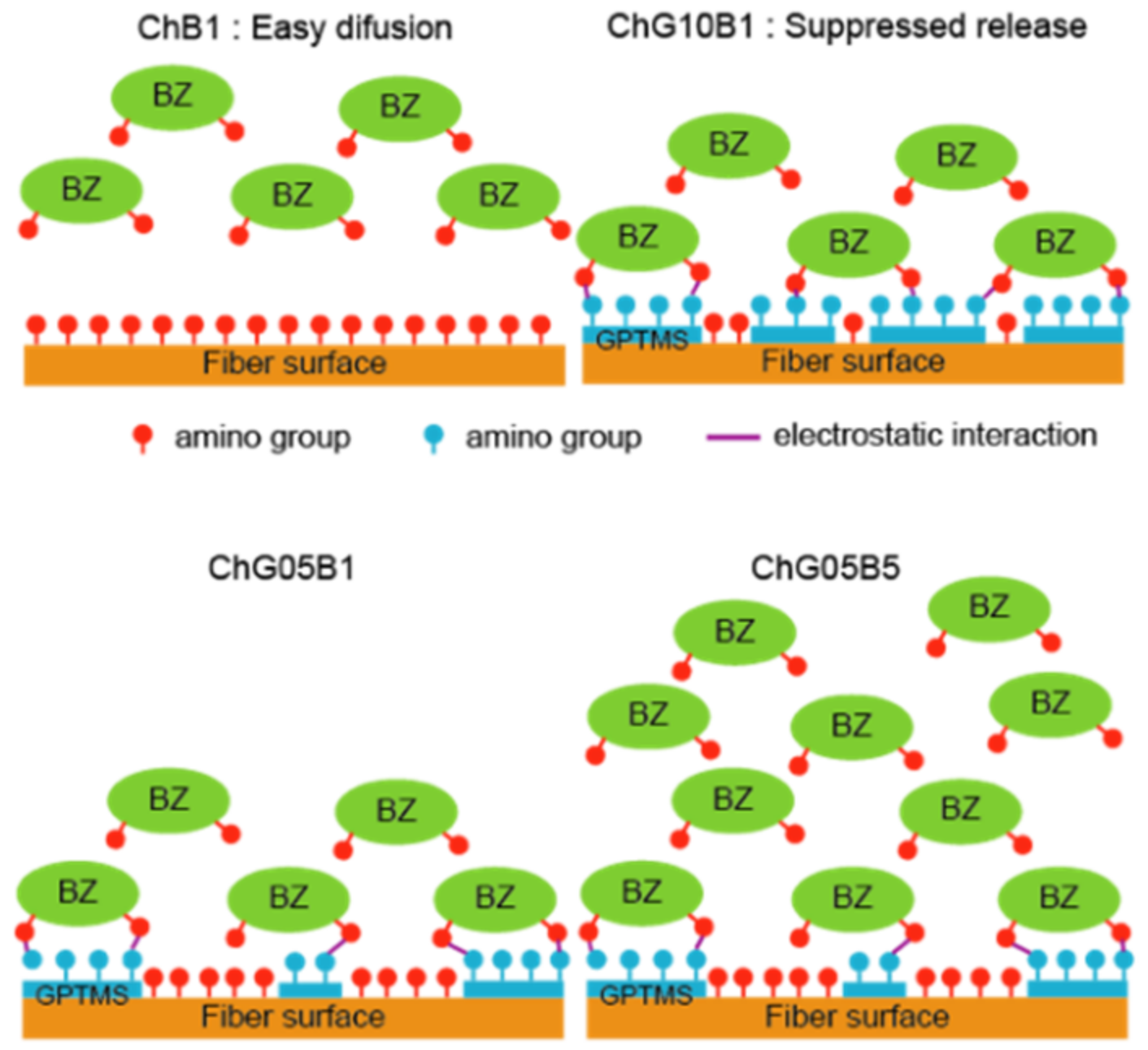
3.3. Release of BZ and In Vitro Biological Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taylor, T.A.; Unakal, C.G. Staphylococcus aureus. In StatPearls [Internet]; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2020. [Google Scholar]
- Stevens, L.D.; Bryant, E.A. Impetigo, Erysipelas and Cellulitis. In Staphylococcus pyogenes. Basic Biology to Clinical Manifestations; Ferretti, J.J., Stevens, L.D., Vincent, A.F., Eds.; University of Oklahoma Health Sciences Center: Oklahoma City, OK, USA, 2016. [Google Scholar]
- McCaig, F.L.; McDonald, C.L.; Mandal, S.; Jernigan, B.D. Staphylococcus aureus-associated skin and soft tissue infections in ambulatory care. Emerg. Infect. Dis. 2006, 12, 1715–1723. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, H.K.; Knop, K.F.; Frost, M.; Hallas, J.; Pottegård, A. Use of antibiotics and risk of type 2 diabetes: A population-based case-control study. J. Clin. Endocrinol. Metab. 2015, 100, 3633–3640. [Google Scholar] [CrossRef]
- Saga, T.; Yamaguchi, K. History of antimicrobial agents and resistant bacteria. JMAJ 2009, 52, 103–108. [Google Scholar]
- Barnhill, E.A.; Brewer, T.M.; Carlson, A.S. Adverse effects of antimicrobials via predictable or idiosyncratic inhibition of host mitochondrial components. Antimicrob. Agents Chemother. 2021, 56, 4046–4051. [Google Scholar] [CrossRef]
- Azuma, K.; Ifuku, S.; Osaki, T.; Okamoto, Y.; Minami, S. Preparation and biomedical applications of chitin and chitosan nanofibers. J. Biomed. Nanotechnol. 2014, 10, 2891–2920. [Google Scholar] [CrossRef]
- Ifuku, S. Chitin and chitosan nanofibers: Preparation and chemical modifications. Molecules 2014, 19, 18367–18380. [Google Scholar] [CrossRef] [PubMed]
- Ifuku, S.; Yamada, K.; Morimoto, M.; Saimoto, H. Nanofibrillation of dry chitin powder by star burst system. J. Nanomater. 2012, 2012, 645625. [Google Scholar] [CrossRef]
- Dutta, K.A.; Kawamoto, N.; Sugino, G.; Izawa, H.; Morimoto, M.; Saimoto, H.; Ifuku, S. Simple preparation of chitosan nanofibers from dry chitosan powder by the star burst system. Carbohydr. Polym. 2013, 97, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Zeiger, E.; Gollapudi, B.; Spencer, P. Genetic toxicity and carcinogenicity studies of glutaraldehyde—A review. Mutat. Res. 2005, 589, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Jameela, S.R.; Jayakrishnan, A. Glutaraldehyde crosslinked chitosan microspheres as a long acting biodegradable drug delivery vehicle: Studies on the in vitro release of mitoxantrone and in vivo degradation of microspheres in rat muscle. Biomaterials 1995, 16, 769–775. [Google Scholar] [CrossRef]
- Speer, D.P.; Chvapil, M.; Eskeson, C.D.; Ulreich, J. Biological effects of residual glutaraldehyde in glutaraldehyde-tanned collagen biomaterials. J. Biomed. Mater. Res. 1980, 14, 753–764. [Google Scholar] [CrossRef]
- Shirosaki, Y.; Tsuru, K.; Hayakawa, S.; Osaka, A.; Lopes, M.A.; Santos, J.D.; Fernandes, M.H. Physical, chemical and in vitro biological profile of chitosan hybrid membrane as a function of organosiloxane concentration. Acta Biomater. 2009, 5, 346–355. [Google Scholar] [CrossRef]
- Shirosaki, Y. Preparation of organic-inorganic hybrids with silicate network for the medical applications. J. Ceram. Soc. Jpn. 2012, 120, 555–559. [Google Scholar] [CrossRef]
- Shirosaki, Y.; Tsuru, K.; Moribayashi, H.; Hayakawa, S.; Nakamura, Y.; Gibson, I.R.; Osaka, A. Preparation of osteocompatible Si(IV)-enriched chitosan-silicate hybrids. J. Ceram. Soc. Jpn. 2010, 118, 989–992. [Google Scholar] [CrossRef]
- Shirosaki, Y.; Okayama, T.; Tsuru, K.; Hayakawa, S.; Osaka, A. In vitro bioactivity and MG63 cytocompatibility of chitosan-silicate hybrids. Int. J. Mater. Chem. 2013, 3, 1–7. [Google Scholar]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Dutta, P.K.; Tripathi, S.; Mehrotra, G.K.; Dutta, J. Perspectives for chitosan based antimicrobial films in food applications. Food Chem. 2009, 114, 1173–1182. [Google Scholar] [CrossRef]
- Proksch, E. pH in nature, humans and skin. J. Dermatol. 2018, 45, 1044–1052. [Google Scholar] [CrossRef]
- Smith, J.L.; Weinberg, E.D. Mechanisms of antibacterial action of bacitracin. J. Gen. Microbiol. 1962, 28, 559–569. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Abunowara, M.; Bustam, A.M.; Sufian, S.; Eldemerdash, U. Description of carbon dioxide adsorption and desorption onto Malaysian coals under subcritical condition. Procedia Eng. 2016, 148, 600–608. [Google Scholar] [CrossRef][Green Version]
- Shirosaki, Y.; Kubo, M.; Tsuru, K.; Hayakawa, S.; Osaka, A. In vitro apatite formation on organic polymers modified with a silane coupling reagent. J. R. Soc. Interface 2005, 2, 335–340. [Google Scholar] [CrossRef][Green Version]
- Kostov, L.K.; Belamie, E.; Alonso, B.; Mineva, T. Surface chemical states of cellulose, chitin and chitosan studied by density functional theory high-resolution photoelectron spectroscopy. Bulg. Chem. Commun. 2018, 50, 135–146. [Google Scholar]
- Zhang, M.; Hisamori, H.; Yamada, T.; Hirano, S. 13C CP/MAS NMR spectral-analysis of 6-o-tosyl, 6-deoxy-6-iodo, and 6-deoxy derivatives of N-acetylchitosan in solid-state. Biosci. Biotech. Biochem. 1994, 58, 1906–1908. [Google Scholar] [CrossRef]
- Zhang, M.; Haga, A.; Sekiguchi, H.; Hirano, S. Structure of insect chitin isolated from beetle larva cuticle and silkworm (Bombyx mori) pupa exuvia. Int. J. Biol. Macromol. 2000, 27, 99–105. [Google Scholar] [CrossRef]
- Sánchez-Fernández, A.; Peña-Parás, L.; Mendoza, M.E.; Leyva, A.; Bautista, A.L.; Bulach, F.-X.; Monsivais-Barrsón, A.; Bonilla-Ríos, J.; Elizalde, E.L. Spectroscopic and thermal studies of polyalkoxysilanes and silica-chitosan hybrid materials. J. Mater. Sci. Res. 2016, 5, 1–11. [Google Scholar] [CrossRef][Green Version]
- Lyerla, R.J., Jr.; Freedman, H.M. Special assignment and conformational analysis of cyclic peptides by carbon-13 nuclear magnetic resonance. J. Biol. Chem. 1972, 247, 8183–8192. [Google Scholar] [CrossRef]
- Raabe, D.; Al-Sawalmih, A.; Yi, B.S.; Fabritius, H. Preferred crystallographic texture of α-chitin as a microscopic and macroscopic design principle of the exoskeleton of the lobster Homarus americanus. Acta Biomater. 2007, 3, 882–895. [Google Scholar] [CrossRef]
- Kamra, T.; Chaudhary, S.; Xu, C.; Johansson, N.; Montelius, L.; Schnadt, J.; Ye, L. Covalent immobilization of molecularly imprinted polymer nanoparticles using an epoxy silane. J. Colloid Interface Sci. 2015, 445, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Plueddemann, E.P. Silane Coupling Agents, 2nd ed.; Springer: New York, NY, USA, 1991. [Google Scholar]






| Sample | GPTMS/Chitosan Amino Groups (Molar Ratio) | BZ Concentration (g L−1) |
|---|---|---|
| Ch | 1:0 | 0 |
| ChG01 | 1:0.1 | 0 |
| ChG025 | 1:0.25 | 0 |
| ChG05 | 1:0.5 | 0 |
| ChG05B05 | 1:0.5 | 0.77 |
| ChB1 | 1:0 | 1.54 |
| ChG05B1 | 1:0.5 | 1.54 |
| ChG10B1 | 1:1 | 1.54 |
| ChG05B5 | 1:0.5 | 7.7 |
| Ch | ChG01 | ChG05 | ChG10 | ChB1 | ChG05B05 | ChG05B1 | ChG05B5 | ChG10B1 |
|---|---|---|---|---|---|---|---|---|
| 91.9 ± 9.8 | 80.8 ± 4.9 | 76.4 ± 2.0 | 71.1 ± 3.6 | 70.0 ± 10.6 | 66.1 ± 0.7 | 52.5 ± 4.3 | 42.6 ± 5.0 | 55.7 ± 5.1 |
| Sample | SSA (m2 g−1) |
|---|---|
| Ch | 17.8 |
| ChG01 | 47.8 |
| ChG05 | 28.1 |
| ChG10 | 11.0 |
| Sample | –NH3+ | –NH–CH2–/–NH–C = O | –NH2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| BE a | FWHM b | I c | BE a | FWHM b | I c | BE a | FWHM b | I c | |
| Ch | 401.570 | 2.330 | 11.5 | 400.314 | 0.848 | 1.7 | 399.292 | 1.695 | 86.8 |
| ChG05 | 402.115 | 1.102 | 2.9 | 400.889 | 1.398 | 8.3 | 399.218 | 1.399 | 88.8 |
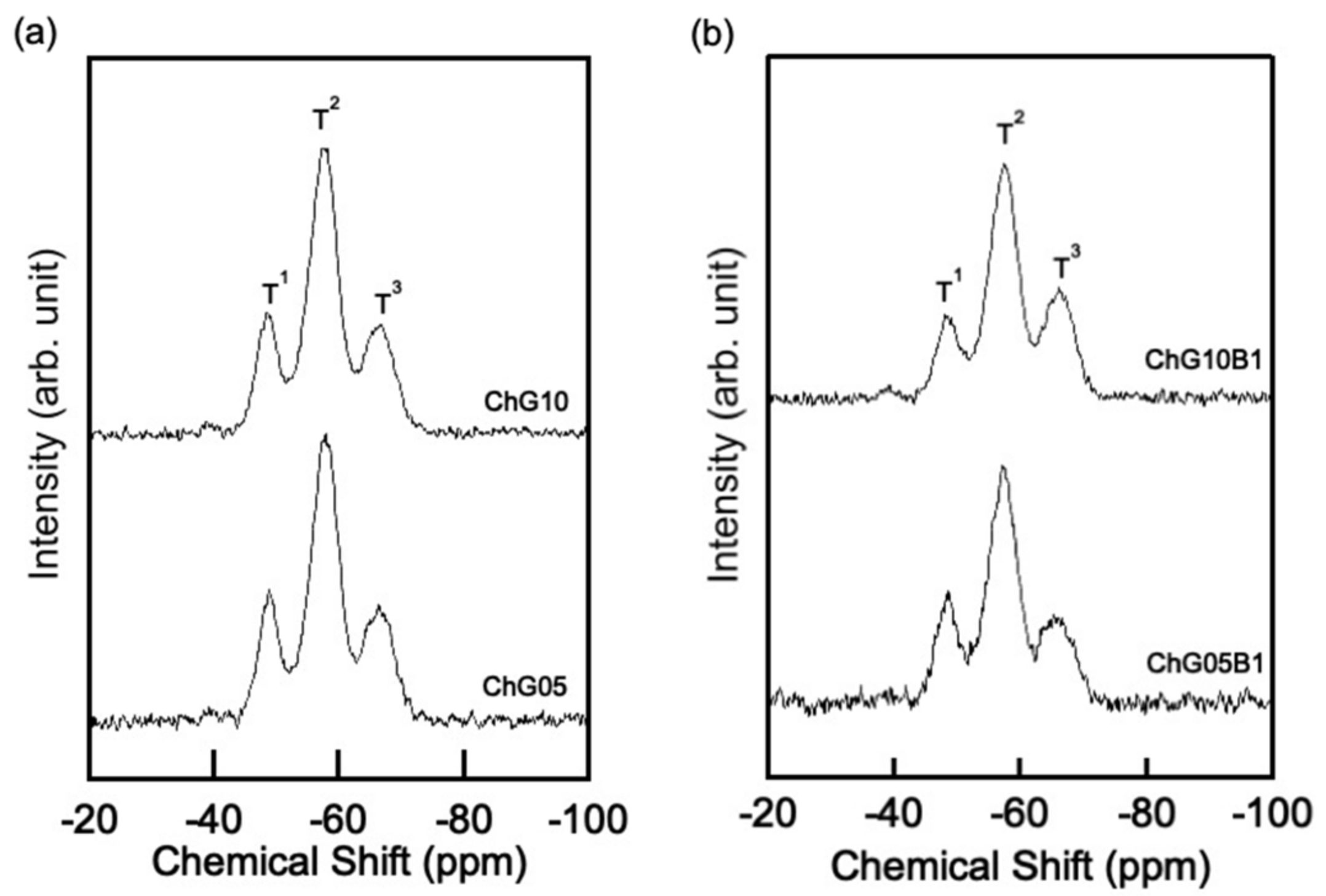
| Sample | T1 | T2 | T3 | N bo/Si | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| δ a | FWHM b | I c | δ a | FWHM b | I c | δ a | FWHM b | I c | ||
| ChG10 | −49.02 | 4.69 | 19.69 | −57.87 | 5.48 | 57.15 | −66.76 | 5.80 | 23.16 | 1.96 |
| ChG05 | −48.92 | 4.48 | 19.90 | −57.75 | 5.35 | 56.26 | −66.40 | 5.64 | 23.84 | 1.96 |
| ChG10B1 | −48.72 | 4.90 | 17.08 | −57.40 | 5.25 | 55.19 | −66.10 | 5.68 | 27.73 | 2.11 |
| ChG05B1 | −48.54 | 4.74 | 21.18 | −57.25 | 5.43 | 55.38 | −65.84 | 6.07 | 23.44 | 2.02 |
| Sample | BZ Release (µg/Sample) and Ratio (%) | Inhibition Zone (mm) |
|---|---|---|
| Ch | 0 | 0 |
| ChB1 | 13.0 (3.0%) | 17.5 ± 2.3 |
| ChG05 | 0 | 0 |
| ChG05B05 | 3.2 (1.5%) | 14.9 ± 4.8 |
| ChG05B1 | 11.9 (2.8%) | 14.1 ± 0.6 |
| ChG05B5 | 49.4 (2.3%) | 21.3 ± 0.1 * |
| ChG10B1 | 8.4 (1.9%) | 11.5 ± 0.3 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumamoto, K.; Maeda, T.; Hayakawa, S.; Mustapha, N.A.B.; Wang, M.-J.; Shirosaki, Y. Antibacterial Chitosan Nanofiber Thin Films with Bacitracin Zinc Salt. Polymers 2021, 13, 1104. https://doi.org/10.3390/polym13071104
Kumamoto K, Maeda T, Hayakawa S, Mustapha NAB, Wang M-J, Shirosaki Y. Antibacterial Chitosan Nanofiber Thin Films with Bacitracin Zinc Salt. Polymers. 2021; 13(7):1104. https://doi.org/10.3390/polym13071104
Chicago/Turabian StyleKumamoto, Kazutaka, Toshinari Maeda, Satoshi Hayakawa, Nurul Asyifah Binti Mustapha, Meng-Jiy Wang, and Yuki Shirosaki. 2021. "Antibacterial Chitosan Nanofiber Thin Films with Bacitracin Zinc Salt" Polymers 13, no. 7: 1104. https://doi.org/10.3390/polym13071104
APA StyleKumamoto, K., Maeda, T., Hayakawa, S., Mustapha, N. A. B., Wang, M.-J., & Shirosaki, Y. (2021). Antibacterial Chitosan Nanofiber Thin Films with Bacitracin Zinc Salt. Polymers, 13(7), 1104. https://doi.org/10.3390/polym13071104






