Photocatalytic Activity of Cellulose Acetate Nanoceria/Pt Hybrid Mats Driven by Visible Light Irradiation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Photocatalytic Mats Preparation
2.3. Characterization of Fibrous Mats
2.4. Photocatalytic Tests
2.5. Reactive Oxygen Species Identification
3. Results and Discussion
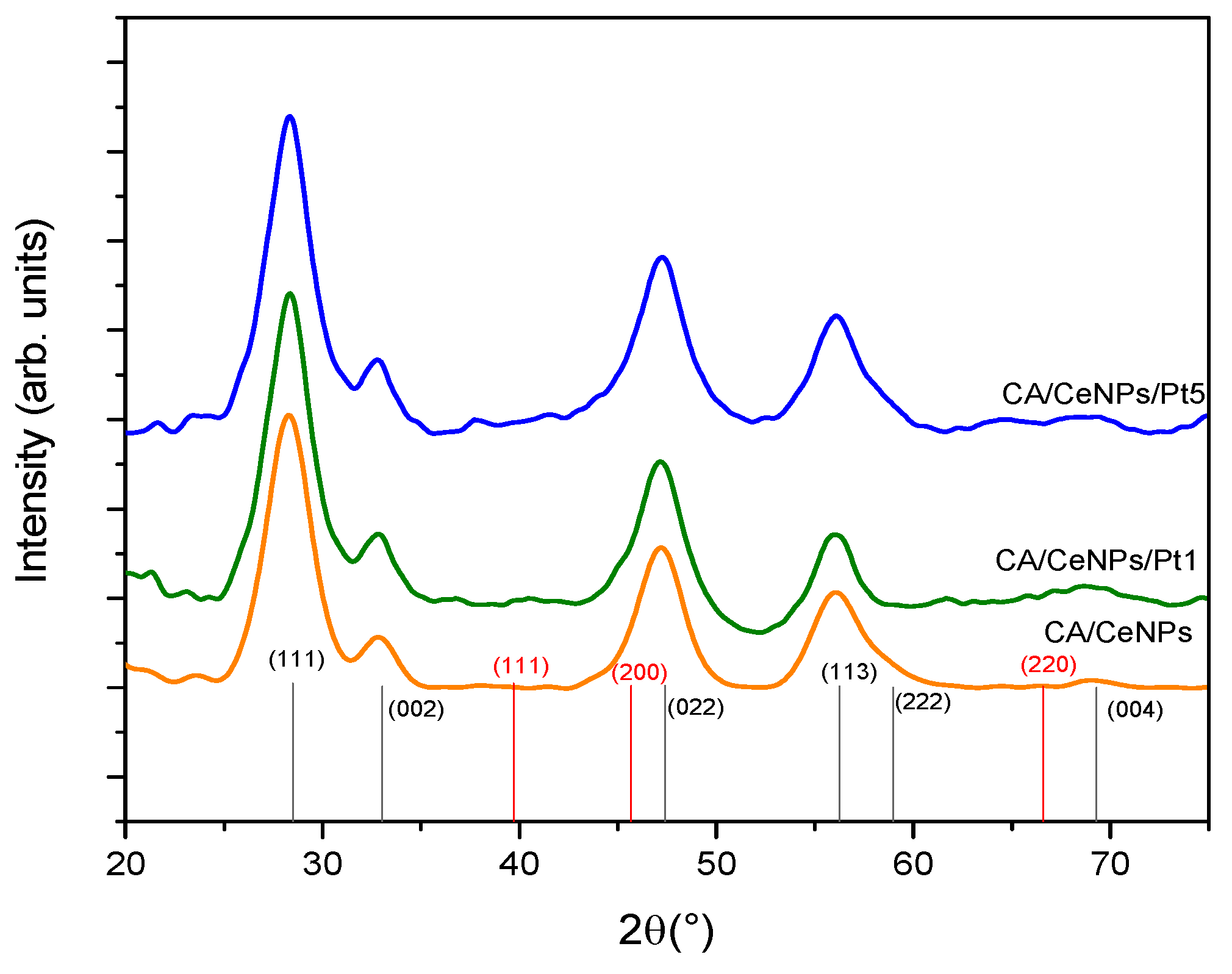
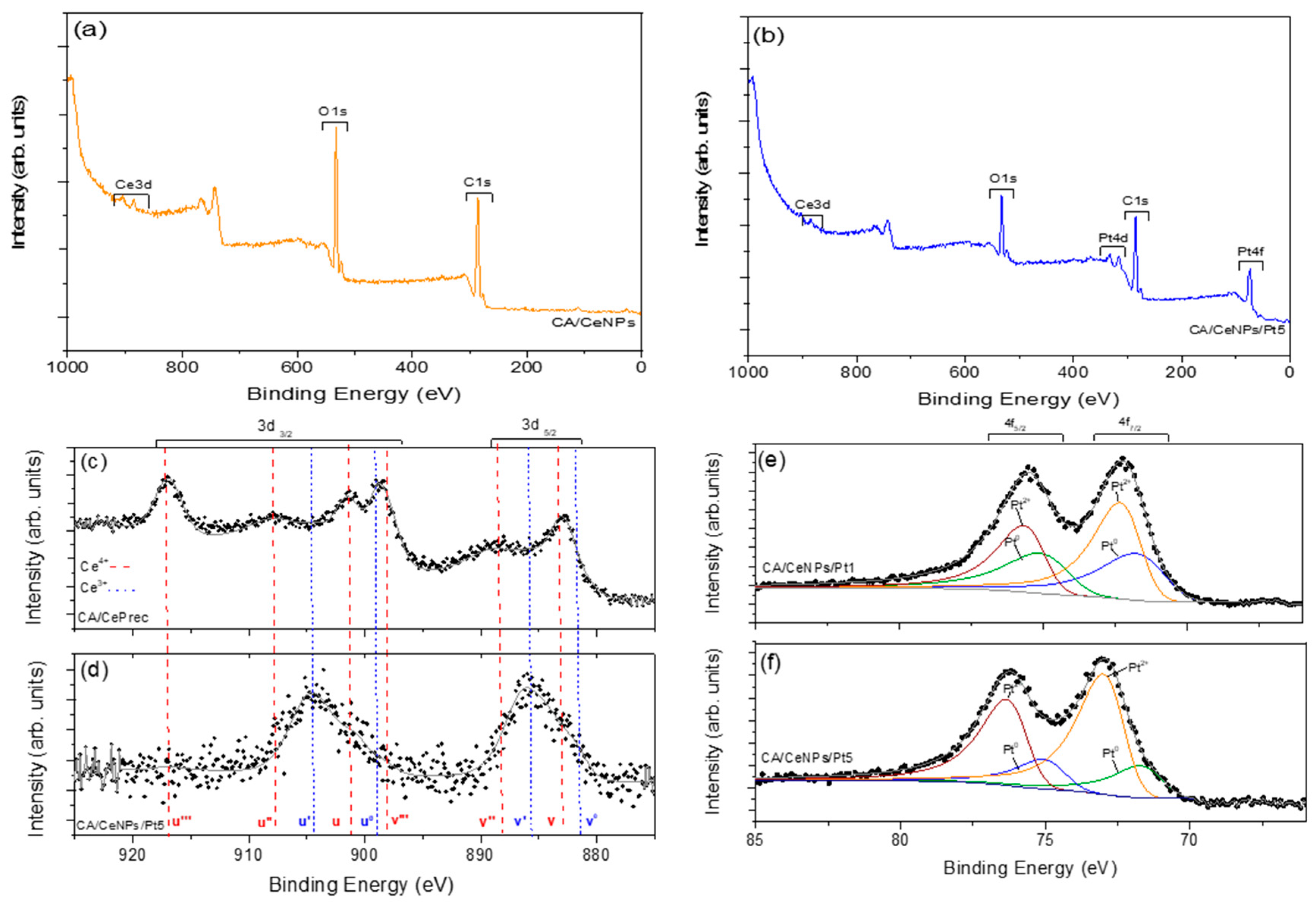
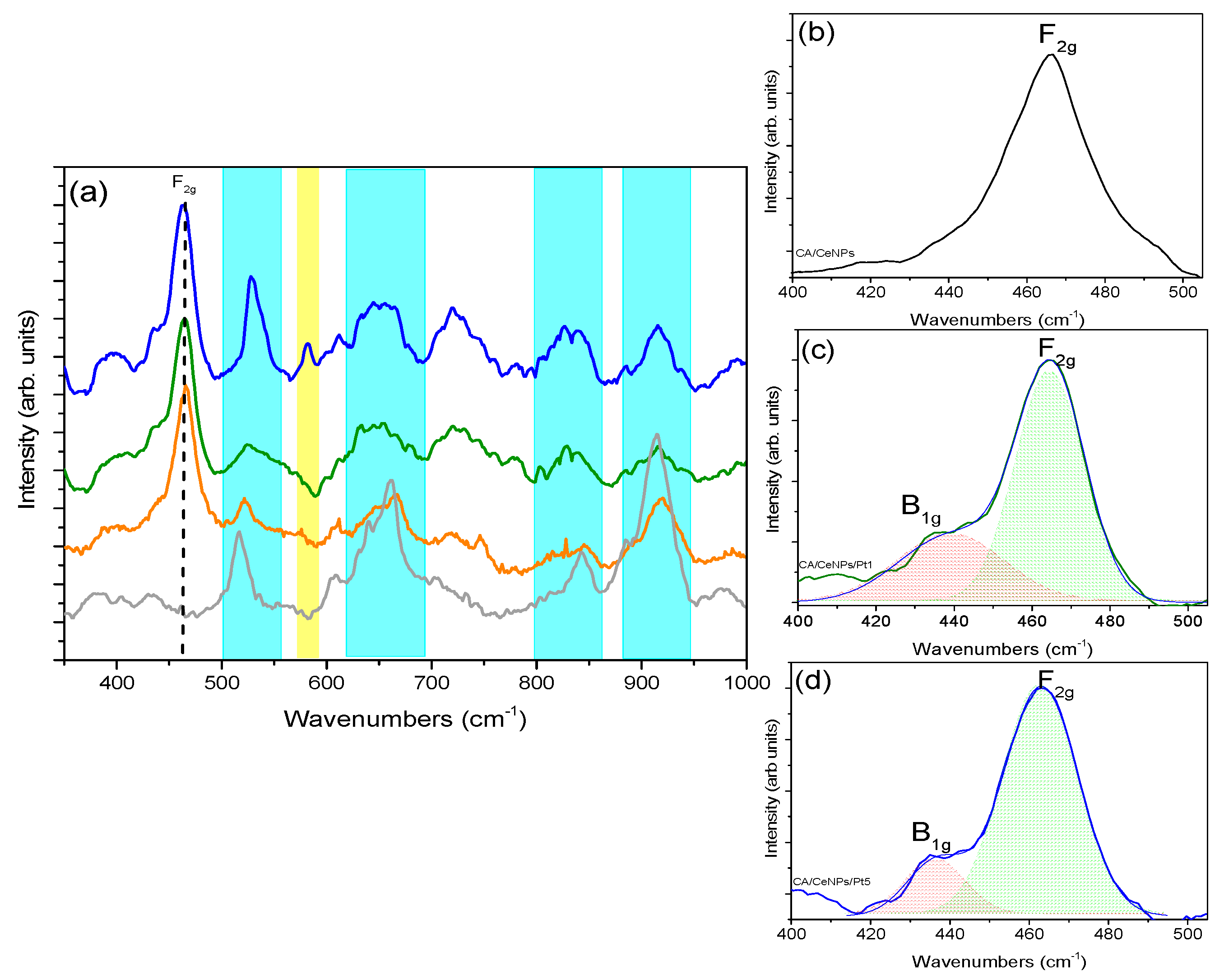
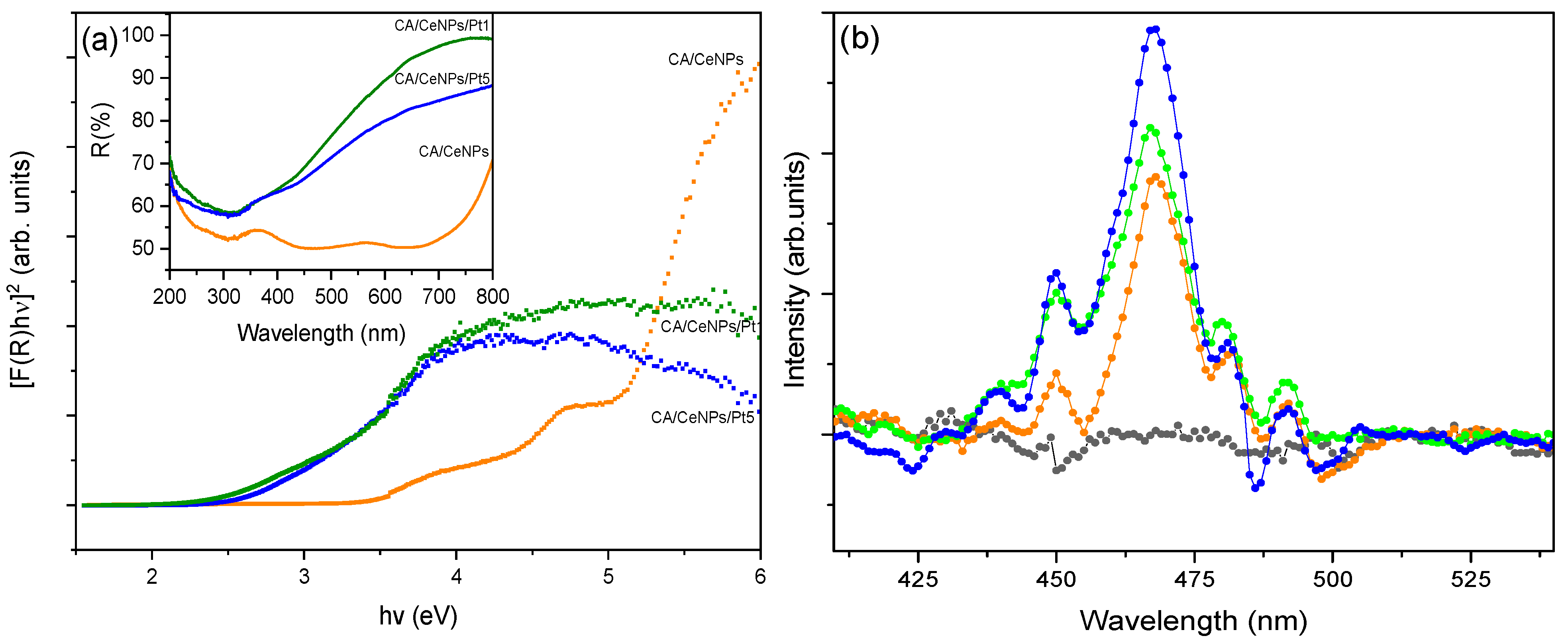
3.1. Morphological Characterization
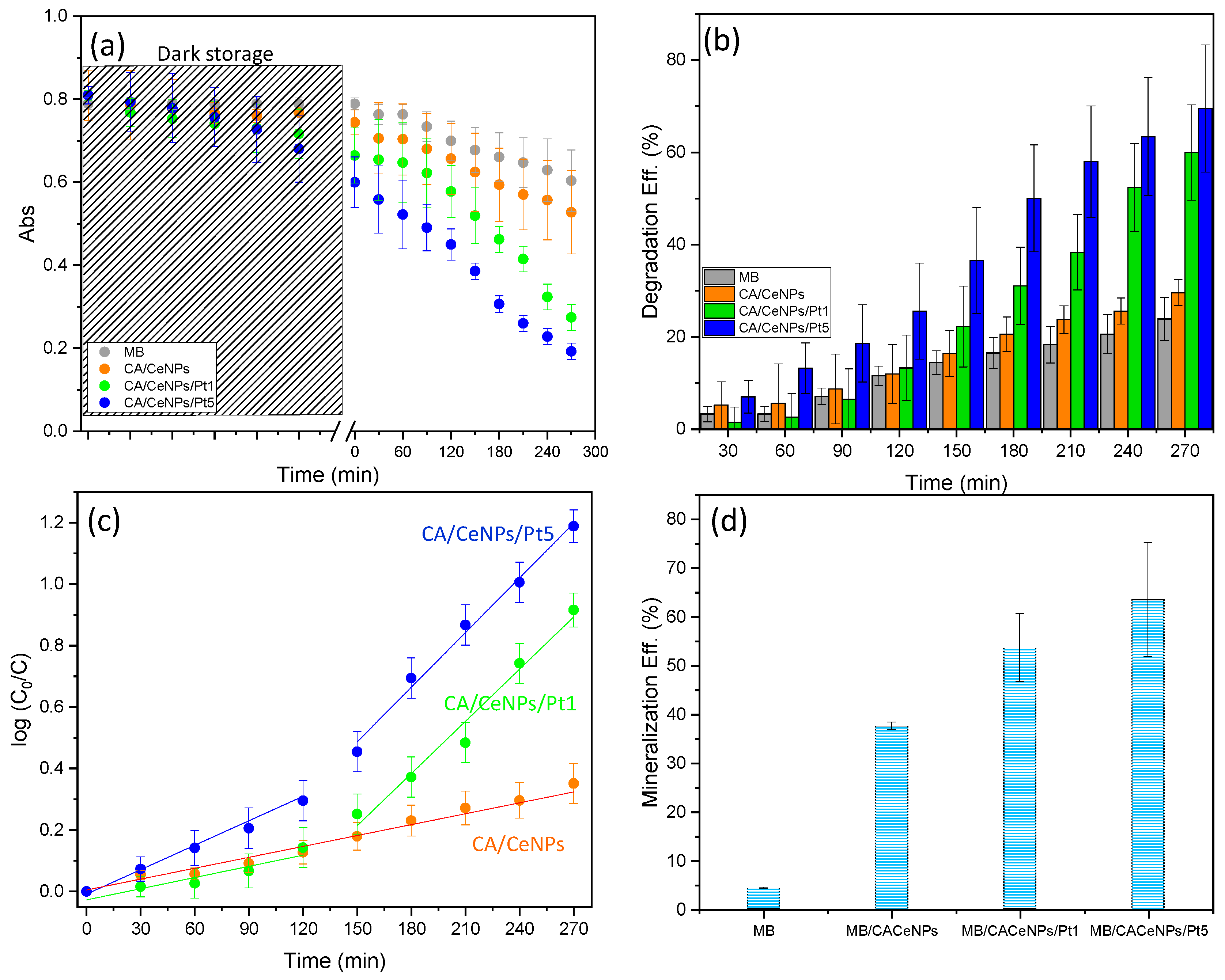
3.2. Photocatalytic Degradation Performance under Visible Light Irradiation
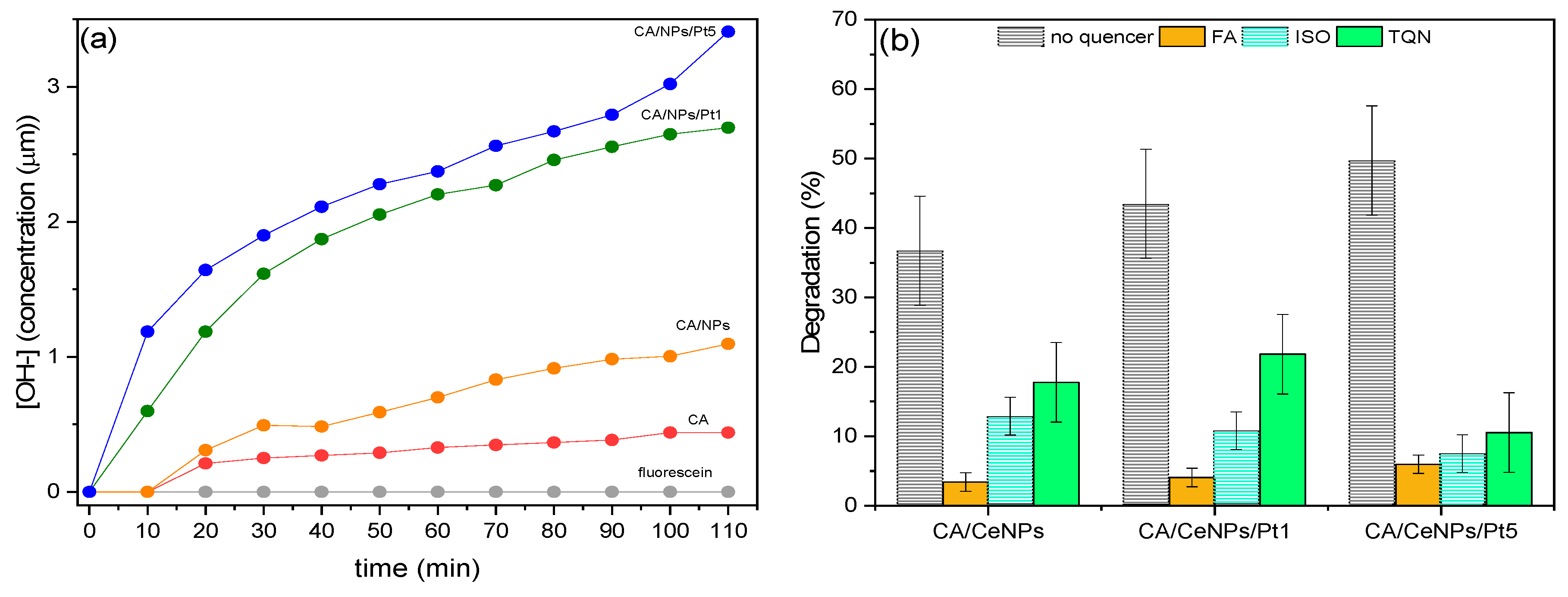
3.3. Reactive Oxygen Species (ROS) Identification
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chan, S.H.S.; Wu, T.Y.; Juan, J.C.; Teh, C.Y. Recent Developments of Metal Oxide Semiconductors as Photocatalysts in Advanced Oxidation Processes (AOPs) for Treatment of Dye Waste-Water. J. Chem. Technol. Biotechnol. 2011, 86, 1130–1158. [Google Scholar] [CrossRef]
- Ibhadon, A.; Fitzpatrick, P. Heterogeneous Photocatalysis: Recent Advances and Applications. Catalysts 2013, 3, 189–218. [Google Scholar] [CrossRef]
- Anastasescu, C.; Negrila, C.; Angelescu, D.G.; Atkinson, I.; Anastasescu, M.; Spataru, N.; Zaharescu, M.; Balint, I. Particularities of Photocatalysis and Formation of Reactive Oxygen Species on Insulators and Semiconductors: Cases of SiO2, TiO2 and Their Composite SiO2-TiO2. Catal. Sci. Technol. 2018, 8, 5657–5668. [Google Scholar] [CrossRef]
- Chatterjee, D.; Dasgupta, S. Visible Light Induced Photocatalytic Degradation of Organic Pollutants. J. Photochem. Photobiol. C Photochem. Rev. 2005, 6, 186–205. [Google Scholar] [CrossRef]
- Kamat, P.V. Photophysical, Photochemical and Photocatalytic Aspects of Metal Nanoparticles. J. Phys. Chem. B 2002, 106, 7729–7744. [Google Scholar] [CrossRef]
- Wang, P.; Huang, B.; Dai, Y.; Whangbo, M.-H. Plasmonic Photocatalysts: Harvesting Visible Light with Noble Metal Nanoparticles. Phys. Chem. Chem. Phys. 2012, 14, 9813–9825. [Google Scholar] [CrossRef] [PubMed]
- Bellardita, M.; Fiorenza, R.; Palmisano, L.; Scirè, S. Photocatalytic and Photothermocatalytic Applications of Cerium Oxide-Based Materials. In Cerium Oxide (CeO₂): Synthesis, Properties and Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 109–167. ISBN 978-0-12-815661-2. [Google Scholar]
- Trovarelli, A.; Llorca, J. Ceria Catalysts at Nanoscale: How Do Crystal Shapes Shape Catalysis? ACS Catal. 2017, 7, 4716–4735. [Google Scholar] [CrossRef]
- Song, S.; Wang, X.; Zhang, H. CeO2-Encapsulated Noble Metal Nanocatalysts: Enhanced Activity and Stability for Catalytic Application. NPG Asia Mater. 2015, 7, e179. [Google Scholar] [CrossRef]
- Morselli, D.; Valentini, P.; Perotto, G.; Scarpellini, A.; Pompa, P.P.; Athanassiou, A.; Fragouli, D. Thermally-Induced in Situ Growth of ZnO Nanoparticles in Polymeric Fibrous Membranes. Compos. Sci. Technol. 2017, 149, 11–19. [Google Scholar] [CrossRef]
- Hoque, M.A.; Guzman, M.I. Photocatalytic Activity: Experimental Features to Report in Heterogeneous Photocatalysis. Materials 2018, 11, 1990. [Google Scholar] [CrossRef]
- Jia, H.; Zhu, X.-M.; Jiang, R.; Wang, J. Aerosol-Sprayed Gold/Ceria Photocatalyst with Superior Plasmonic Hot Electron-Enabled Visible-Light Activity. ACS Appl. Mater. Interfaces 2017, 9, 2560–2571. [Google Scholar] [CrossRef]
- Fandi, Z.; Ameur, N.; Brahimi, F.T.; Bedrane, S.; Bachir, R. Photocatalytic and Corrosion Inhibitor Performances of CeO2 Nanoparticles Decorated by Noble Metals_ Au, Ag, Pt. J. Environ. Chem. Eng. 2020, 8, 104346. [Google Scholar] [CrossRef]
- Thamer, B.M.; Aldalbahi, A.; Moydeen, A.M.; Rahaman, M.; El-Newehy, M.H. Modified Electrospun Polymeric Nanofibers and Their Nanocomposites as Nanoadsorbents for Toxic Dye Removal from Contaminated Waters: A Review. Polymers 2021, 13, 20. [Google Scholar] [CrossRef]
- Li, C.; Chen, R.; Zhang, X.; Shu, S.; Xiong, J.; Zheng, Y.; Dong, W. Electrospinning of CeO2-ZnO Composite Nanofibers and Their Photocatalytic Property. Mater. Lett. 2011, 65, 1327–1330. [Google Scholar] [CrossRef]
- Sharma, R.; Singh, N.; Gupta, A.; Tiwari, S.; Tiwari, S.K.; Dhakate, S.R. Electrospun Chitosan–Polyvinyl Alcohol Composite Nanofibers Loaded with Cerium for Efficient Removal of Arsenic from Contaminated Water. J. Mater. Chem. A 2014, 2, 16669–16677. [Google Scholar] [CrossRef]
- Morselli, D.; Campagnolo, L.; Prato, M.; Papadopoulou, E.L.; Scarpellini, A.; Athanassiou, A.; Fragouli, D. Ceria/Gold Nanoparticles in Situ Synthesized on Polymeric Membranes with Enhanced Photocatalytic and Radical Scavenging Activity. ACS Appl. Nano Mater. 2018, 1, 5601–5611. [Google Scholar] [CrossRef]
- Costantino, F.; Armirotti, A.; Carzino, R.; Gavioli, L.; Athanassiou, A.; Fragouli, D. In Situ Formation of SnO2 Nanoparticles on Cellulose Acetate Fibrous Membranes for the Photocatalytic Degradation of Organic Dyes. J. Photochem. Photobiol. A Chem. 2020, 398, 112599. [Google Scholar] [CrossRef]
- Campagnolo, L.; Lauciello, S.; Athanassiou, A.; Fragouli, D. Au/ZnO Hybrid Nanostructures on Electrospun Polymeric Mats for Improved Photocatalytic Degradation of Organic Pollutants. Water 2019, 11, 1787. [Google Scholar] [CrossRef]
- Peli, S.; Cavaliere, E.; Benetti, G.; Gandolfi, M.; Chiodi, M.; Cancellieri, C.; Giannetti, C.; Ferrini, G.; Gavioli, L.; Banfi, F. Mechanical Properties of Ag Nanoparticle Thin Films Synthesized by Supersonic Cluster Beam Deposition. J. Phys. Chem. C 2016, 120, 4673–4681. [Google Scholar] [CrossRef]
- Fraters, B.D.; Cavaliere, E.; Mul, G.; Gavioli, L. Synthesis of Photocatalytic TiO2 Nano-Coatings by Supersonic Cluster Beam Deposition. J. Alloy. Compd. 2014, 615, S467–S471. [Google Scholar] [CrossRef]
- Benetti, G.; Cavaliere, E.; Brescia, R.; Salassi, S.; Ferrando, R.; Vantomme, A.; Pallecchi, L.; Pollini, S.; Boncompagni, S.; Fortuni, B.; et al. Tailored Ag-Cu-Mg Multielemental Nanoparticles for Wide-Spectrum Antibacterial Coating. Nanoscale 2019, 11, 1626–1635. [Google Scholar] [CrossRef]
- Nair, V.; Deepthi, A. Cerium(IV) Ammonium NitrateA Versatile Single-Electron Oxidant. Chem. Rev. 2007, 107, 1862–1891. [Google Scholar] [CrossRef] [PubMed]
- Benetti, G.; Cavaliere, E.; Banfi, F.; Gavioli, L. Antimicrobial Nanostructured Coatings: A Gas Phase Deposition and Magnetron Sputtering Perspective. Materials 2020, 13, 784. [Google Scholar] [CrossRef] [PubMed]
- Chiodi, M.; Cheney, C.P.; Vilmercati, P.; Cavaliere, E.; Mannella, N.; Weitering, H.H.; Gavioli, L. Enhanced Dopant Solubility and Visible-Light Absorption in Cr-N Codoped TiO2 Nanoclusters. J. Phys. Chem. C 2012, 116, 311–318. [Google Scholar] [CrossRef]
- Rueden, C.T.; Schindelin, J.; Hiner, M.C.; DeZonia, B.E.; Walter, A.E.; Arena, E.T.; Eliceiri, K.W. ImageJ2: ImageJ for the next Generation of Scientific Image Data. BMC Bioinform. 2017, 18, 1–26. [Google Scholar] [CrossRef]
- Mitchell, D.R.G. DiffTools: Electron Diffraction Software Tools for DigitalMicrographTM. Microsc. Res. Tech. 2008, 71, 588–593. [Google Scholar] [CrossRef]
- Serrà, A.; Zhang, Y.; Sepúlveda, B.; Gómez, E.; Nogués, J.; Michler, J.; Philippe, L. Highly Active ZnO-Based Biomimetic Fern-like Microleaves for Photocatalytic Water Decontamination Using Sunlight. Appl. Catal. B Environ. 2019, 248, 129–146. [Google Scholar] [CrossRef]
- Nosaka, Y.; Nosaka, A.Y. Generation and Detection of Reactive Oxygen Species in Photocatalysis. Chem. Rev. 2017, 117, 11302–11336. [Google Scholar] [CrossRef] [PubMed]
- Manwar, N.R.; Chilkalwar, A.A.; Nanda, K.K.; Chaudhary, Y.S.; Subrt, J.; Rayalu, S.S.; Labhsetwar, N.K. Ceria Supported Pt/PtO-Nanostructures: Efficient Photocatalyst for Sacrificial Donor Assisted Hydrogen Generation under Visible-NIR Light Irradiation. ACS Sustain. Chem. Eng. 2016, 4, 2323–2332. [Google Scholar] [CrossRef]
- Daelman, N.; Capdevila-Cortada, M.; López, N. Dynamic Charge and Oxidation State of Pt/CeO2 Single-Atom Catalysts. Nat. Mater. 2019, 18, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Calvache-Muñoz, J.; Prado, F.A.; Tirado, L.; Daza-Gomez, L.C.; Cuervo-Ochoa, G.; Calambas, H.L.; Rodríguez-Páez, J.E. Structural and Optical Properties of CeO2 Nanoparticles Synthesized by Modified Polymer Complex Method. J. Inorg. Organomet. Polym. Mater. 2019, 29, 813–826. [Google Scholar] [CrossRef]
- Ma, R.; Jahurul Islam, M.; Amaranatha Reddy, D.; Kim, T.K. Transformation of CeO2 into a Mixed Phase CeO2/Ce2O3 Nanohybrid by Liquid Phase Pulsed Laser Ablation for Enhanced Photocatalytic Activity through Z-Scheme Pattern. Ceram. Int. 2016, 42, 18495–18502. [Google Scholar] [CrossRef]
- Paparazzo, E. Use and Mis-Use of X-ray Photoemission Spectroscopy Ce3d Spectra of C2O3 and CeO2. J. Phys. Condens. Matter 2018, 30, 343003. [Google Scholar] [CrossRef]
- Ansari, S.A.; Khan, M.M.; Ansari, M.O.; Kalathil, S.; Lee, J.; Cho, M.H. Band Gap Engineering of CeO2 Nanostructure Using an Electrochemically Active Biofilm for Visible Light Applications. RSC Adv. 2014, 4, 16782–16791. [Google Scholar] [CrossRef]
- Qi, L.; Yu, Q.; Dai, Y.; Tang, C.; Liu, L.; Zhang, H.; Gao, F.; Dong, L.; Chen, Y. Influence of Cerium Precursors on the Structure and Reducibility of Mesoporous CuO-CeO2 Catalysts for CO Oxidation. Appl. Catal. B Environ. 2012, 119–120, 308–320. [Google Scholar] [CrossRef]
- Romanchenko, A.; Likhatski, M.; Mikhlin, Y. X-ray Photoelectron Spectroscopy (XPS) Study of the Products Formed on Sulfide Minerals Upon the Interaction with Aqueous Platinum (IV) Chloride Complexes. Minerals 2018, 8, 578. [Google Scholar] [CrossRef]
- Lee, J.; Ryou, Y.; Chan, X.; Kim, T.J.; Kim, D.H. How Pt Interacts with CeO2 under the Reducing and Oxidizing Environments at Elevated Temperature: The Origin of Improved Thermal Stability of Pt/CeO2 Compared to CeO2. J. Phys. Chem. C 2016, 120, 25870–25879. [Google Scholar] [CrossRef]
- Mao, M.; Lv, H.; Li, Y.; Yang, Y.; Zeng, M.; Li, N.; Zhao, X. Metal Support Interaction in Pt Nanoparticles Partially Confined in the Mesopores of Microsized Mesoporous CeO2 for Highly Efficient Purification of Volatile Organic Compounds. ACS Catal. 2016, 6, 418–427. [Google Scholar] [CrossRef]
- Filtschew, A.; Hofmann, K.; Hess, C. Ceria and Its Defect Structure: New Insights from a Combined Spectroscopic Approach. J. Phys. Chem. C 2016, 120, 6694–6703. [Google Scholar] [CrossRef]
- Agarwal, U.P. Analysis of Cellulose and Lignocellulose Materials by Raman Spectroscopy: A Review of the Current Status. Molecules 2019, 24, 1659. [Google Scholar] [CrossRef]
- Fischer, S.; Schenzel, K.; Fischer, K.; Diepenbrock, W. Applications of FT Raman Spectroscopy and Micro Spectroscopy Characterizing Cellulose and Cellulosic Biomaterials. Macromol. Symp. 2005, 223, 41–56. [Google Scholar] [CrossRef]
- Zhang, K.; Feldner, A.; Fischer, S. FT Raman Spectroscopic Investigation of Cellulose Acetate. Cellulose 2011, 18, 995–1003. [Google Scholar] [CrossRef]
- Sartoretti, E.; Novara, C.; Giorgis, F.; Piumetti, M.; Bensaid, S.; Russo, N.; Fino, D. In Situ Raman Analyses of the Soot Oxidation Reaction over Nanostructured Ceria-Based Catalysts. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef]
- Kosacki, I.; Suzuki, T.; Anderson, H.U.; Colomban, P. Raman Scattering and Lattice Defects in Nanocrystalline CeO2 Thin Films. Solid State Ion. 2002, 149, 99–105. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, F.; Liu, X.; Liu, Y.; Luo, M.; Teng, B.; Fan, M.; Liu, X. Resolving a Decade-Long Question of Oxygen Defects in Raman Spectra of Ceria-Based Catalysts at Atomic Level. J. Phys. Chem. C 2019, 123, 18889–18894. [Google Scholar] [CrossRef]
- McBride, J.R.; Graham, G.W.; Peters, C.R.; Weber, W.H. Growth and Characterization of Reactively Sputtered Thin-film Platinum Oxides. J. Appl. Phys. 1991, 69, 1596–1604. [Google Scholar] [CrossRef]
- Derevyannikova, E.A.; Kardash, T.Y.; Stadnichenko, A.I.; Stonkus, O.A.; Slavinskaya, E.M.; Svetlichnyi, V.A.; Boronin, A.I. Structural Insight into Strong Pt-CeO2 Interaction: From Single Pt Atoms to PtOx Clusters. J. Phys. Chem. C 2019, 123, 1320–1334. [Google Scholar] [CrossRef]
- Lin, W.; Herzing, A.A.; Kiely, C.J.; Wachs, I.E. Probing Metal−Support Interactions under Oxidizing and Reducing Conditions: In Situ Raman and Infrared Spectroscopic and Scanning Transmission Electron Microscopic−X-ray Energy-Dispersive Spectroscopic Investigation of Supported Platinum Catalysts. J. Phys. Chem. C 2008, 112, 5942–5951. [Google Scholar] [CrossRef]
- López, R.; Gómez, R. Band-Gap Energy Estimation from Diffuse Reflectance Measurements on Sol–Gel and Commercial TiO2: A Comparative Study. J. Sol. Gel Sci. Technol. 2012, 61, 1–7. [Google Scholar] [CrossRef]
- Bruix, A.; Migani, A.; Vayssilov, G.N.; Neyman, K.M.; Libuda, J.; Illas, F. Effects of Deposited Pt Particles on the Reducibility of CeO2 (111). Phys. Chem. Chem. Phys. 2011, 13, 11384–11392. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Shi, Y.; Huang, Y.-C.; Chen, R.; Zhang, Y.; Huo, J.; Zou, Y.; Yu, G.; Luo, J.; Dong, C.-L.; et al. Interface Engineering of Pt and CeO2 Nanorods with Unique Interaction for Methanol Oxidation. Nano Energy 2018, 53, 604–612. [Google Scholar] [CrossRef]
- Khan, M.M.; Ansari, S.A.; Pradhan, D.; Han, D.H.; Lee, J.; Cho, M.H. Defect-Induced Band Gap Narrowed CeO2 Nanostructures for Visible Light Activities. Ind. Eng. Chem. Res. 2014, 53, 9754–9763. [Google Scholar] [CrossRef]
- Chiu, Y.-H.; Chang, T.-F.M.; Chen, C.-Y.; Sone, M.; Hsu, Y.-J. Mechanistic Insights into Photodegradation of Organic Dyes Using Heterostructure Photocatalysts. Catalysts 2019, 9, 430. [Google Scholar] [CrossRef]
- Andronic, L.; Isac, L.; Cazan, C.; Enesca, A. Simultaneous Adsorption and Photocatalysis Processes Based on Ternary TiO2-CuxS-Fly Ash Hetero-Structures. Appl. Sci. 2020, 10, 8070. [Google Scholar] [CrossRef]
- Ranjith, K.S.; Satilmis, B.; Huh, Y.S.; Han, Y.-K.; Uyar, T. Highly Selective Surface Adsorption-Induced Efficient Photodegradation of Cationic Dyes on Hierarchical ZnO Nanorod-Decorated Hydrolyzed PIM-1 Nanofibrous Webs. J. Colloid Interface Sci. 2020, 562, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, Y.; Nosaka, A. Introduction to Photocatalysis: From Basic Science to Applications; The Royal Society of Chemistry: London, UK, 2016; ISBN 978-1-78262-320-5. [Google Scholar]
- Ribao, P.; Corredor, J.; Rivero, M.J.; Ortiz, I. Role of Reactive Oxygen Species on the Activity of Noble Metal-Doped TiO2 Photocatalysts. J. Hazard. Mater. 2019, 372, 45–51. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costantino, F.; Cavaliere, E.; Gavioli, L.; Carzino, R.; Leoncino, L.; Brescia, R.; Athanassiou, A.; Fragouli, D. Photocatalytic Activity of Cellulose Acetate Nanoceria/Pt Hybrid Mats Driven by Visible Light Irradiation. Polymers 2021, 13, 912. https://doi.org/10.3390/polym13060912
Costantino F, Cavaliere E, Gavioli L, Carzino R, Leoncino L, Brescia R, Athanassiou A, Fragouli D. Photocatalytic Activity of Cellulose Acetate Nanoceria/Pt Hybrid Mats Driven by Visible Light Irradiation. Polymers. 2021; 13(6):912. https://doi.org/10.3390/polym13060912
Chicago/Turabian StyleCostantino, Federica, Emanuele Cavaliere, Luca Gavioli, Riccardo Carzino, Luca Leoncino, Rosaria Brescia, Athanassia Athanassiou, and Despina Fragouli. 2021. "Photocatalytic Activity of Cellulose Acetate Nanoceria/Pt Hybrid Mats Driven by Visible Light Irradiation" Polymers 13, no. 6: 912. https://doi.org/10.3390/polym13060912
APA StyleCostantino, F., Cavaliere, E., Gavioli, L., Carzino, R., Leoncino, L., Brescia, R., Athanassiou, A., & Fragouli, D. (2021). Photocatalytic Activity of Cellulose Acetate Nanoceria/Pt Hybrid Mats Driven by Visible Light Irradiation. Polymers, 13(6), 912. https://doi.org/10.3390/polym13060912









