Collagen-Based Nanofibers for Skin Regeneration and Wound Dressing Applications
Abstract
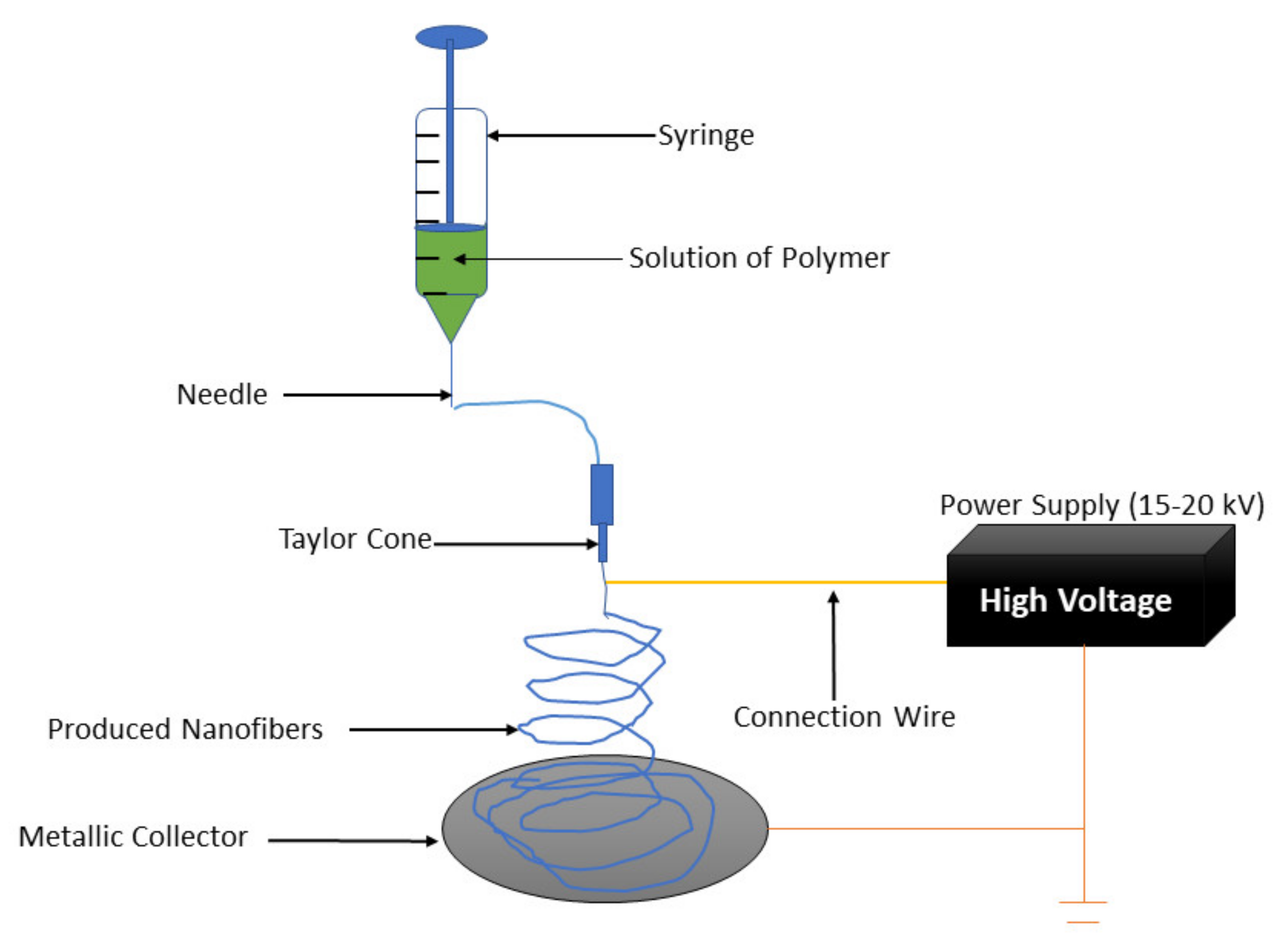
:1. Introduction
2. Phases of Wound Healing
3. Properties of Collagen in Biomedical Applications
| Form of Dressing | Composition | Product Name | Advantage | Limitations | Wounds Suitable for | Ref |
|---|---|---|---|---|---|---|
| Gel | Collagen | CelleraRX | Maintain moisture for wound bed | Bovine sources, and require secondary wound dressing | Partial and Full-thickness injuries including traumatic wounds, surgical wounds, diabetic ulcers, and burns | [52] |
| Gel | Collagen Polypeptides | Stimulen | Provide moisture for wound bed | Bovine source, and require secondary wound dressing | Full- and partial-thickness wounds including pressure ulcers, partial-thickness burns, abrasions | [53] |
| Pad | Collagen fleece, gentamicin salts | Septocoll E | Activate platelets | skin responses | Full and partial thickness injuries including infected wounds, and bleeding lesion | [54] |
| Pad | Collagen, carboxymethylcellulose, sodium alginate, AgCl | ColActive Plus Ag | Hinders the function of MMPs | Bovine sources, require secondary wound dressing | Full and partial thickness wounds including burns, dehisced surgical incisions abrasions, diabetic, venous, or pressure ulcers | [55] |
| Pad | Collagen and Ca alginate | Fibracol Plus | Maintain moisture for wound bed | Require secondary wound dressing | Full and partial thickness wounds including burns, dehisced surgical incisions abrasions, diabetic, venous, or pressure ulcers | [56] |
| Pad | Bovine collagen, and Manuka Honey | Puracol | No extra debridement Required | Bovine source and expensive. | Full and partial thickness injuries including dehisced surgical incisions abrasions, burns, diabetic, venous, or pressure ulcers | [57] |
| Pad | Type I equine Collagen | Biopad | Free from collagen degradation products | Equine source, time consuming, high cost | Full and partial thickness wounds including dehisced surgical incisions abrasions, diabetic, venous, or pressure ulcers | [58] |
| Pad | Bovine collagen and oxidized cellulose | Promogran | Hemostatic activity | Bovine source, not to be utilized in third-degree burns. | Full and partial thickness wounds including abrasions, bleeding wounds, venous or diabetic ulcers, pressure wounds | [59] |
| Powder | Collagen | Catrix | Decrease bleeding, Biodegradable | Bovine source, require secondary wound dressing | Full and Partial-thickness wounds including cuts, abrasions, irritations, pressure, diabetic ulcers radiation dermatitis, burns | [60] |
| Membrane | porcine dermal collagen, nylon, silicon | Biobrane | Flexibility | Bovine source, require secondary wound dressing | Partial-thickness burn wounds | [61] |
| Cellular matrix | Collagen, polycarbonate membrane | Apligraf | Resorbable | Not suitable for infected injuries, bovine source, and expensive. | Full and partial thickness injuries including venous leg ulcers, diabetic foot ulcers | [62] |
| Cellular matrix | Type I collagen | Orcel | Full resorbable | Not suitable for infected wounds, bovine source, high cost | Full-thickness injuries including burns | [63] |
4. Collagen-Based Nanofibers in Skin Regeneration and Wound Dressing
4.1. Advantages and Disadvantages of the Electrospun for Skin Regeneration and Wound Dressing
4.2. Application of Electrospun Collagen-Based Nanofibers in Skin Regeneration and Wound Dressing
4.3. In Vivo and In Vitro Studies of Collagen-Based Nanofibers
4.3.1. Plain Collagen-Based Nanofibers
4.3.2. Collagen-Based Nanofibers Loaded with Bioactive Agents
4.3.3. Other Collagen-Based Nanofibrous Scaffolds
5. Integrity of Collagen during the Fabrication of Nanofibers and Standardization of Raw Collagen
6. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Forbes, S.J.; Rosenthal, N. Preparing the ground for tissue regeneration: From mechanism to therapy. Nat. Med. 2014, 20, 857–869. [Google Scholar] [CrossRef] [PubMed]
- Muthukumar, T.; Sreekumar, G.; Sastry, T.P.; Chamundeeswari, M. Collagen as a potential biomaterial in biomedical applications. Rev. Adv. Mater. Sci. 2018, 53, 19–39. [Google Scholar] [CrossRef]
- Lan, W.; Xu, M.; Zhang, X.; Zhao, L.; Huang, D.; Wei, X.; Chen, W. Biomimetic polyvinyl alcohol/type II collagen hydrogels for cartilage tissue engineering. J. Biomater. Sci. Polym. Ed. 2020, 31, 1179–1198. [Google Scholar] [CrossRef] [PubMed]
- Facchin, F.; Bianconi, E.; Canaider, S.; Basoli, V.; Biava, P.M.; Ventura, C. Review Article Tissue Regeneration without Stem Cell Transplantation: Self- Healing Potential from Ancestral Chemistry and Physical Energies. Stem Cells Int. 2018, 2018, 7412035. [Google Scholar] [CrossRef] [PubMed]
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin Wound Healing Process and New Emerging Technologies for Skin Wound Care and Regeneration. Pharmaceutics 2020, 17, 735. [Google Scholar] [CrossRef]
- Fahimirad, S.; Ajalloueian, F. Naturally-derived electrospun wound dressings for target delivery of bio-active agents. Int. J. Pharm. 2019, 566, 307–328. [Google Scholar] [CrossRef]
- El Ashram, S.; El-Samad, L.M.; Basha, A.A.; El Wakil, A. Naturally-derived targeted therapy for wound healing: Beyond classical strategies. Pharmacol. Res. 2021, 170, 105749. [Google Scholar] [CrossRef]
- Alven, S.; Aderibigbe, B.A. Fabrication of Hybrid Nanofibers from Biopolymers and Poly (Vinyl Alcohol)/Poly (ε-Caprolactone) for Wound Dressing Applications. Polymers 2021, 13, 2104. [Google Scholar] [CrossRef] [PubMed]
- Graça, M.F.P.; Miguel, S.P.; Cabral, C.S.D.; Correia, I.J. Hyaluronic acid—Based wound dressings: A review. Carbohydr. Polym. 2020, 241, 116364. [Google Scholar] [CrossRef] [PubMed]
- Sudarsan, S.; Franklin, D.S.; Guhanathan, S. Imbibed salts and pH-responsive behaviours of sodium-alginate based eco-friendly biopolymeric hydrogels-A solventless approach. MMAIJ 2015, 11, 24–29. [Google Scholar]
- Karmanov, A.P.; Kanarsky, A.V.; Kocheva, L.S.; Belyy, V.A.; Semenov, E.I.; Rachkova, N.G.; Bogdanovich, N.I.; Pokryshkin, S.A. Chemical structure and polymer properties of wheat and cabbage lignins—Valuable biopolymers for biomedical applications. Polymer 2021, 220, 123571. [Google Scholar] [CrossRef]
- Chalitangkoon, J.; Monvisade, P. Synthesis of chitosan-based polymeric dyes as colorimetric pH-sensing materials: Potential for food and biomedical applications. Carbohydr. Polym. 2021, 260, 117836. [Google Scholar] [CrossRef]
- Alven, S.; Aderibigbe, B.A. Chitosan and Cellulose-Based Hydrogels for Wound Management. Int. J. Mol. Sci. 2020, 21, 9656. [Google Scholar] [CrossRef]
- Reddy, M.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A comparative review of natural and synthetic biopolymer composite scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef]
- Hussain, Z.; Thu, H.E.; Shuid, A.N.; Katas, H.; Hussain, F. Recent Advances in Polymer-based Wound Dressings for the Treatment of Diabetic Foot Ulcer: An Overview of State-of-the-art. Curr. Drug Targets 2017, 18, 527–550. [Google Scholar] [CrossRef]
- Alven, S.; Nqoro, X.; Aderibigbe, B.A. Polymer-Based Materials Loaded with Curcumin for Wound Healing Application. Polymers 2020, 12, 2286. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Ahmad, M.M.; Alruwaili, N.K.; Alrowaili, Z.A.; Alomar, F.A.; Akhtar, S.; Alsaidan, O.A.; Alhakamy, N.A.; Zafar, A.; Elmowafy, M.; et al. Antibiotic-loaded psyllium husk hemicellulose and gelatin-based polymeric films for wound dressing application. Pharmaceutics 2021, 13, 236. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.; Yao, Z.; Feng, L.; Liu, T.; Wei, S.; Xu, P.; Guo, R.; Cheng, B.; Wang, X. Antibiotic-loaded chitosan-gelatin scaffolds for infected seawater immersion wound healing. Int. J. Biol. Macromol. 2020, 159, 1140–1155. [Google Scholar] [CrossRef]
- Lee, C.H.; Singla, A.; Lee, Y. Biomedical applications of collagen. Int. J. Pharm. 2001, 221, 1–22. [Google Scholar] [CrossRef]
- Zhang, L.; Webster, T.J. Nanotechnology and nanomaterials: Promises for improved tissue regeneration. Nano Today 2009, 4, 66–80. [Google Scholar] [CrossRef]
- Krafts, K.P. Tissue repair The hidden drama. Organogenesis 2012, 6, 225–233. [Google Scholar] [CrossRef] [Green Version]
- Sorg, H.; Tilkorn, D.J.; Hager, S.; Hauser, J.; Mirastschijski, U. Skin Wound Healing: An Update on the Current Knowledge and Concepts. Eur. Surg. Res. 2017, 58, 81–94. [Google Scholar] [CrossRef]
- Catanzano, O.; Esposito, V.D.; Acierno, S.; Ambrosio, M.R.; Caro, C.D.; Avagliano, C.; Russo, P.; Russo, R.; Miro, A.; Ungaro, F.; et al. Alginate—Hyaluronan composite hydrogels accelerate wound healing process. Carbohydr. Polym. 2015, 131, 407–414. [Google Scholar] [CrossRef]
- Ambekar, R.S.; Kandasubramanian, B. Advancements in nanofibers for wound dressing: A review. Eur. Polym. J. 2019, 117, 304–336. [Google Scholar] [CrossRef]
- Kus, K.J.B.; Ruiz, E.S. Wound Dressings—A Practical Review. Curr. Dermatol. Rep. 2020, 9, 298–308. [Google Scholar] [CrossRef]
- Debele, T.A.; Su, W. Polysaccharide and protein-based functional wound dressing materials and applications. Int. J. Polym. Mater. Polym. Biomater. 2020, 1–22. [Google Scholar] [CrossRef]
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound healing: A cellular perspective. Physiol. Rev. 2018, 99, 665–706. [Google Scholar] [CrossRef]
- Pereira, R.; Carvalho, A.; Vaz, D.C.; Gil, M.H.; Mendes, A.; Bártolo, P. Development of novel alginate based hydrogel films for wound healing applications. Int. J. Biol. Macromol. 2013, 52, 221–230. [Google Scholar] [CrossRef]
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.E.; Eccleston, G.M. Wound Healing Dressings and Drug Delivery Systems: A Review. J. Pharm. Sci. 2008, 97, 2892–2923. [Google Scholar] [CrossRef]
- Fredric, S.; Gowda, D.V.; Yashashwini, M. Wafers for wound healing. J. Chem. Pharm. Res. 2015, 7, 450–468. [Google Scholar]
- Aderibigbe, B.A.; Buyana, B. Alginate in Wound Dressings. Pharmaceutics 2018, 10, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, S.; Srivastava, S.; Singh, M.R.; Singh, D. Mechanistic insight into diabetic wounds: Pathogenesis, molecular targets and treatment strategies to pace wound healing. Biomed. Pharmacother. 2019, 112, 108615. [Google Scholar] [CrossRef] [PubMed]
- Alven, S.; Khwaza, V.; Oyedeji, O.O.; Aderibigbe, B.A. Polymer-Based Scaffolds Loaded with Aloe vera Extract for the Treatment of Wounds. Pharmaceutics 2021, 13, 961. [Google Scholar] [CrossRef] [PubMed]
- Irastorza, A.; Zarandona, I.; Andonegi, M.; Guerrero, P.; de la Caba, K. Food Hydrocolloids The versatility of collagen and chitosan: From food to biomedical applications. Food Hydrocoll. 2021, 116, 106633. [Google Scholar] [CrossRef]
- Majid, Q.A.; Fricker, A.T.R.; Gregory, D.A.; Davidenko, N.; Hernandez, C.O.; Jabbour, R.J.; Owen, T.J.; Basnett, P.; Lukasiewicz, B.; Stevens, M.; et al. Natural Biomaterials for Cardiac Tissue Engineering: A Highly Biocompatible Solution. Front. Cardiovasc. Med. 2020, 7, 192. [Google Scholar] [CrossRef]
- Grabska-Zielińska, S.; Sionkowska, A.; Coelho, C.C.; Monteiro, F.J. Silk Fibroin/collagen/chitosan scaffolds cross-linked by a glyoxal solution as biomaterials toward bone tissue regeneration. Materials 2020, 15, 3433. [Google Scholar] [CrossRef]
- Abdulghani, S.; Mitchell, G.R. Biomaterials for in situ tissue regeneration: A review. Biomolecules 2019, 9, 750. [Google Scholar] [CrossRef] [Green Version]
- Ricard-Blum, S. The collagen family. Cold Spring Harb Perspect. Biol. 2011, 3, 004978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, F.M.; Liu, X. Advancing biomaterials of human origin for tissue engineering. Prog. Polym. Sci. 2016, 53, 86–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-based biomaterials for tissue engineering applications. Materials 2010, 3, 1863–1887. [Google Scholar] [CrossRef] [Green Version]
- Marina, A.; Gentile, P.; Chiono, V.; Ciardelli, G. Collagen for bone tissue regeneration. Acta Biomater. 2012, 8, 3191–3200. [Google Scholar]
- Amirrah, I.N.; Farhanulhakim, M.; Razip, M.; Tabata, Y.; Bt, R.; Idrus, H.; Nordin, A.; Fauzi, M.B. Antibacterial-Integrated Collagen Wound Dressing for Diabetes-Related Foot Ulcers: An Evidence-Based Review of Clinical Studies. Polymers 2020, 12, 2168. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, P.; Soni, S.; Mittal, G.; Bhatnagar, A. Role of polymeric biomaterials as wound healing agents. Int. J. Low Extrem. Wounds 2014, 13, 180–190. [Google Scholar] [CrossRef]
- Gaspar-pintiliescu, A.; Stanciuc, A.; Craciunescu, O. Natural composite dressings based on collagen, gelatin and plant bioactive compounds for wound healing: A review. Int. J. Biol. Macromol. 2019, 138, 854–865. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, S.; Raines, R.T. Review collagen-based biomaterials for wound healing. Biopolymers 2014, 101, 821–833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gould, L.J. Topical Collagen-Based Biomaterials for Chronic Wounds: Rationale and Clinical Application. Adv. Wound Care 2016, 5, 19–31. [Google Scholar] [CrossRef] [Green Version]
- Brett, D. A Review of Collagen and Collagen-based Wound Dressings. Wound Repair Regen. 2018, 20, 347–356. [Google Scholar]
- Dong, C.; Lv, Y. Application of collagen scaffold in tissue engineering: Recent advances and new perspectives. Polymers 2016, 8, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, N.; Wang, Y. Drug Delivery Systems for Wound Healing. Curr. Pharm. Biotechnol. 2015, 16, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Okur, M.E.; Karantas, I.D.; Şenyiğit, Z.; Üstündağ Okur, N.; Siafaka, P.I. Recent trends on wound management: New therapeutic choices based on polymeric carriers. Asian J. Pharm. Sci. 2020, 15, 661–684. [Google Scholar] [CrossRef]
- Saghazadeh, S.; Rinoldi, C.; Schot, M.; Kashaf, S.S.; Sharifi, F.; Jalilian, E.; Nuutila, K.; Giatsidis, G.; Mostafalu, P.; Derakhshandeh, H.; et al. Drug Delivery Systems and Materials for Wound Healing Applications. Adv. Drug Deliv. Rev. 2019, 127, 138–166. [Google Scholar] [CrossRef]
- Newman, M.I.; Baratta, L.G.; Swartz, K. Activated, type I collagen (CellerateRx) and its effectiveness in healing recalcitrant diabetic wounds: A case presentation. Adv. Ski. Wound Care 2008, 21, 370–374. [Google Scholar] [CrossRef]
- Elgharably, H.; Roy, S.; Khanna, S.; Abas, M.; Das, G.P.; Das, A.; Mohammed, K.; Sen, C.K. A modified collagen gel enhances healing outcome in a pre-clinical swine model of excisional wounds. Wound Repair Regen. 2013, 21, 473–481. [Google Scholar] [CrossRef] [Green Version]
- Gruessner, U.; Clemens, M.; Pahlplatz, P.V.; Sperling, P.; Witte, J.; Rosen, H.R. Improvement of perineal wound healing by local administration of gentamicin-impregnated collagen fleeces after abdominoperineal excision of rectal cancer. Am. J. Surg. 2001, 182, 502–509. [Google Scholar] [CrossRef]
- ColActive® Plus Ag (Collagen Matrix Dressing with Silver). Available online: https://www.woundsource.com/product/colactive-plus-ag-collagen-matrix-dressing-silver#:~:text=ColActive%C2%AE%20Plus%20Ag%20(Collagen%20Matrix%20Dressing%20with%20Silver),for%20the%20management%20of%20full-%20and%20partial-thickness%20wounds (accessed on 12 July 2021).
- Colenci, R.; Abbade, L.P.F. Fundamental aspects of the local approach to cutaneous ulcers. An. Bras. Dermatol. 2018, 93, 859–870. [Google Scholar] [CrossRef]
- Capella-Monsonís, H.; Tilbury, M.A.; Wall, J.G.; Zeugolis, D.I. Porcine mesothelium matrix as a biomaterial for wound healing applications. Mater. Today Bio. 2020, 7, 100057. [Google Scholar] [CrossRef]
- Turner, N.J.; Badylak, S.F. The use of biologic scaffolds in the treatment of chronic nonhealing wounds. Adv. Wound Care 2015, 4, 490–500. [Google Scholar] [CrossRef] [Green Version]
- da Silva, C.V.; Pereira, V.J.; Costa, G.M.N.; Cabral-Albuquerque, E.C.M.; Vieira de Melo, S.A.B.; de Sousa, H.C.; Dias, A.M.A.; Braga, M.E.M. Supercritical solvent impregnation/deposition of spilanthol-enriched extracts into a commercial collagen/cellulose-based wound dressing. J. Supercrit. Fluids 2018, 133, 503–511. [Google Scholar] [CrossRef]
- Kalin, M.; Kuru, S.; Kismet, K.; Barlas, A.M.; Akgun, Y.A.; Astarci, H.M.; Ustun, H.; Ertas, E. The effectiveness of porcine dermal collagen (Permacol®) on wound healing in the rat model. Indian J. Surg. 2015, 77, 407–411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harish, V.; Li, Z.; Maitz, P.K.M. The optimal timing of outpatient BiobraneTM application for superficial and mid dermal partial thickness burns: Evidence for the ‘12-hour rule. Burns 2019, 45, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Vlassova, N.; Lazarus, G. Use of Apligraf for a devascularized ulcer secondary to mastectomy and radiation. J. Am. Acad. Dermatol. 2011, 65, e132–e134. [Google Scholar] [CrossRef]
- Still, J.; Glat, P.; Silverstein, P.; Griswold, J.; Mozingo, D. The use of a collagen sponge/ living cell compositematerial to treat donor sites in burn patients. Burns 2003, 29, 837–841. [Google Scholar] [CrossRef]
- Azimi, B.; Maleki, H.; Zavagna, L.; De la Ossa, J.G.; Linari, S.; Lazzeri, A.; Danti, S. Bio-based electrospun fibers for wound healing. J. Funct. Biomater. 2020, 11, 67. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Boccaccini, A.R. Antibacterial biohybrid nanofibers for wound dressings. Acta Biomater. 2020, 107, 25–49. [Google Scholar] [CrossRef] [PubMed]
- Lannutti, J.; Reneker, D.; Ma, T.; Tomasko, D.; Farson, D. Electrospinning for tissue engineering scaffolds. Mater. Sci. Eng. C 2007, 27, 504–509. [Google Scholar] [CrossRef]
- Liu, H.; Ding, X.; Zhou, G.; Li, P.; Wei, X.; Fan, Y. Electrospinning of nanofibers for tissue engineering applications. J. Nanomater. 2013, 2013, 3. [Google Scholar] [CrossRef] [Green Version]
- Wen, Y.; Kok, M.D.R.; Tafoya, J.P.V.; Sobrido, A.B.J.; Bell, E.; Gostick, J.T.; Herou, S.; Schlee, P.; Titirici, M.M.; Brett, D.J.L.; et al. Electrospinning as a route to advanced carbon fibre materials for selected low-temperature electrochemical devices: A review. J. Energy Chem. 2020, 59, 492–529. [Google Scholar] [CrossRef]
- Bavatharani, C.; Muthusankar, E.; Wabaidur, S.M.; Alothman, Z.A.; Alsheetan, K.M.; AL-Anazy, M.; Ragupathy, D. Electrospinning technique for production of polyaniline nanocomposites/nanofibres for multi-functional applications: A review. Synth. Met. 2021, 271, 116609. [Google Scholar] [CrossRef]
- Alven, S.; Buyana, B.; Feketshane, Z.; Aderibige, B.A. Electrospun Nanofibers / Nanofibrous Scaffolds Loaded with Silver Nanoparticles as Effective Antibacterial Wound Dressing Materials. Pharmaceutics 2021, 13, 964. [Google Scholar] [CrossRef] [PubMed]
- Alturki, A.M. Rationally design of electrospun polysaccharides polymeric nanofiber webs by various tools for biomedical applications: A review. Int. J. Biol. Macromol. 2021, 184, 648–665. [Google Scholar] [CrossRef]
- Kamsani, N.H.; Haris, M.S.; Pandey, M.; Taher, M.; Rullah, K. Biomedical application of responsive ‘smart’ electrospun nanofibers in drug delivery system: A minireview. Arab. J. Chem. 2021, 14, 103199. [Google Scholar] [CrossRef]
- Sabra, S.; Ragab, D.M.; Agwa, M.M.; Rohani, S. Recent advances in electrospun nanofibers for some biomedical applications. Eur. J. Pharm. Sci. 2020, 144, 105224. [Google Scholar] [CrossRef]
- Kurakula, M.; Koteswara Rao, G.S.N. Moving polyvinyl pyrrolidone electrospun nanofibers and bioprinted scaffolds toward multidisciplinary biomedical applications. Eur. Polym. J. 2020, 136, 109919. [Google Scholar] [CrossRef]
- Taemeh, M.A.; Shiravandi, A.; Korayem, M.A.; Daemi, H. Fabrication challenges and trends in biomedical applications of alginate electrospun nanofibers. Carbohydr. Polym. 2020, 228, 115419. [Google Scholar] [CrossRef]
- Cheng, G.; Dai, J.; Dai, J.; Wang, H.; Chen, S.; Liu, Y.; Liu, X.; Li, X.; Zhou, X.; Deng, H. Extracellular matrix imitation utilizing nanofibers-embedded biomimetic scaffolds for facilitating cartilage regeneration. Chem. Eng. J. 2021, 410, 128379. [Google Scholar] [CrossRef]
- Arida, I.A.; Ali, I.H.; Nasr, M.; El-Sherbiny, I.M. Electrospun polymer-based nanofiber scaffolds for skin regeneration. J. Drug Deliv. Sci. Technol. 2021, 64, 102623. [Google Scholar] [CrossRef]
- Zhang, X.; Li, L.; Ouyang, J.; Zhang, L.; Xue, J.; Zhang, H. Electroactive electrospun nanofibers for tissue engineering. Nano Today 2021, 39, 101196. [Google Scholar] [CrossRef]
- Dias, J.R.; Granja, P.L.; Bártolo, P.J. Advances in electrospun skin substitutes. Prog. Mater. Sci. 2016, 84, 314–334. [Google Scholar] [CrossRef]
- Kalantari, K.; Afifi, A.M.; Jahangirian, H.; Webster, T.J. Biomedical applications of chitosan electrospun nanofibers as a green polymer—Review. Carbohydr. Polym. 2019, 207, 588–600. [Google Scholar] [CrossRef]
- Ghorbani, S.; Eyni, H.; Tiraihi, T.; Asl, L.S.; Soleimani, M.; Atashi, A.; Beiranvand, S.P.; Warkiani, M.E. Combined effects of 3D bone marrow stem cell-seeded wet-electrospun poly lactic acid scaffolds on full-thickness skin wound healing. Int. J. Polym. Mater. Polym. Biomater. 2018, 67, 905–912. [Google Scholar] [CrossRef]
- Dart, A.; Bhave, M.; Kingshott, P. Antimicrobial Peptide-Based Electrospun Fibers for Wound Healing Applications. Macromol. Biosci. 2019, 19, e1800488. [Google Scholar] [CrossRef]
- Akhmetova, A.; Heinz, A. Electrospinning proteins for wound healing purposes: Opportunities and challenges. Pharmaceutics 2021, 13, 4. [Google Scholar] [CrossRef]
- Dahlin, R.L.; Kasper, F.K.; Mikos, A.G. Polymeric nanofibers in tissue engineering. Tissue Eng. Part. B Rev. 2011, 17, 349–364. [Google Scholar] [CrossRef] [Green Version]
- Xue, J.; Wu, T.; Dai, Y.; Xia, Y. Electrospinning and electrospun nanofibers: Methods, materials, and applications. Chem. Rev. 2019, 119, 5298–5415. [Google Scholar] [CrossRef]
- Pant, B.; Park, M.; Park, S.-J. Drug delivery applications of core-sheath nanofibers prepared by coaxial electrospinning: A review. Pharmaceutics 2019, 11, 305. [Google Scholar] [CrossRef] [Green Version]
- Law, J.X.; Liau, L.L.; Saim, A.; Yang, Y.; Idrus, R. Electrospun collagen nanofibers and their applications in skin tissue engineering. J. Tissue. Eng. Regen. Med. 2017, 14, 699–718. [Google Scholar] [CrossRef]
- Wang, J.; Windbergs, M. Functional electrospun fibers for the treatment of human skin wounds. Eur. J. Pharm. Biopharm. 2017, 119, 283–299. [Google Scholar] [CrossRef]
- Blackstone, B.N.; Gallentine, S.C.; Powell, H.M. Collagen-based electrospun materials for tissue engineering: A systematic review. Bioengineering 2021, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Deng, A.; Yang, Y.; Du, S.; Yang, S. Electrospinning of in situ crosslinked recombinant human collagen peptide/chitosan nanofibers for wound healing. Biomater. Sci. 2018, 6, 2197–2208. [Google Scholar] [CrossRef] [PubMed]
- Pilehvar-Soltanahmadi, Y.; Akbarzadeh, A.; Moazzez-Lalaklo, N.; Zarghami, N. An update on clinical applications of electrospun nanofibers for skin bioengineering. Artif. Cells. Nanomed. Biotechnol. 2016, 44, 1350–1364. [Google Scholar] [CrossRef] [PubMed]
- Drexler, J.W.; Powell, H.M. Dehydrothermal crosslinking of electrospun collagen. Tissue Eng. Part. C. Methods 2011, 17, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Augustine, R.; Rehman, S.R.U.; Ahmed, R.; Zahid, A.A.; Sharifi, M.; Falahati, M.; Hasan, A. Electrospun chitosan membranes containing bioactive and therapeutic agents for enhanced wound healing. Int. J. Biol. Macromol. 2020, 156, 153–170. [Google Scholar] [CrossRef]
- Maleki, H.; Mathur, S.; Klein, A. Antibacterial Ag containing core-shell polyvinyl alcohol-poly (lactic acid) nanofibers for biomedical applications. Polym. Eng. Sci. 2020, 60, 1221–1230. [Google Scholar] [CrossRef]
- Zhou, T.; Wang, N.; Xue, Y.; Ding, T.; Liu, X.; Mo, X. Electrospun tilapia collagen nanofibers accelerating wound healing via inducing keratinocytes proliferation and differentiation. Colloids Surf. B Biointerfaces 2016, 143, 415–422. [Google Scholar] [CrossRef]
- Zhou, T.; Wang, N.; Xue, Y.; Ding, T.; Liu, X.; Mo, X.; Sun, J. Development of Biomimetic Tilapia Collagen Nanofibers for Skin Regeneration through Inducing Keratinocytes Differentiation and Collagen Synthesis of Dermal Fibroblasts. ACS Appl. Mater. Interfaces 2015, 7, 3253–3262. [Google Scholar] [CrossRef]
- Fu, X.; Wang, H. Spatial arrangement of polycaprolactone/collagen nanofiber scaffolds regulates the wound healing related behaviors of human adipose stromal cells. Tissue Eng. Part. A 2012, 18, 631–642. [Google Scholar] [CrossRef] [Green Version]
- Sobhanian, P.; Khorram, M.; Hashemi, S.; Mohammadi, A. Development of nanofibrous collagen-grafted poly (vinyl alcohol)/ gelatin/alginate scaffolds as potential skin substitute. Int. J. Biol. Macromol. 2019, 130, 977–987. [Google Scholar] [CrossRef] [PubMed]
- Su, K.; Jeong, L.; Lee, G.; Seo, B.; Jeong, Y.; Hong, S.; Roh, S.; Jin, J.; Ho, W.; Min, B. Electrospinning of collagen nanofibers: Effects on the behavior of normal human keratinocytes and early-stage wound healing. Biomaterials 2006, 27, 1452–1461. [Google Scholar]
- Powell, H.M.; Supp, D.M.; Boyce, S.T. Influence of electrospun collagen on wound contraction of engineered skin substitutes. Biomaterials 2008, 29, 834–843. [Google Scholar] [CrossRef]
- Elibol, E.; Fuat, Y.; Adnan, Y.; Müge, Ü.; Nurcan, O.; Kum, N.Y.; Kum, R.O.; Kulacoglu, S. Effects of hyaluronic acid-collagen nanofibers on early wound healing in vocal cord trauma. Eur. Arch. Oto Rhino Laryngol. 2021, 278, 1537–1544. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.J.; Guo, J.A.; Li, J.J.; Shi, X.; Wang, L.P.; Chen, W.; Mo, X.M. Repair of Skin Defects with Electrospun Collagen/Chitosan and Fibroin/Chitosan Compound Nanofiber Scaffolds Compared with Gauze Dressing. J. Biomater. Tissue Eng. 2017, 7, 386–392. [Google Scholar] [CrossRef]
- Vigneswari, S.; Murugaiyah, V.; Kaur, G.; Khalil, H.P.S.A.; Amirul, A.A. Simultaneous dual syringe electrospinning system using benign solvent to fabricate nanofibrous P (3HB-co-4HB)/collagen peptides construct as potential leave-on wound dressing. Mater. Sci. Eng. C 2016, 66, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Yong, T.; Chan, K.C.; Ramakrishna, S. Collagen-blended biodegradable polymer nanofibers: Potential substrates for wound healing in skin tissue engineering. In Proceedings of the Fifth IASTED International Conference, Innsbruck, Austria, 14–16 February 2007; pp. 262–266. [Google Scholar]
- Huang, C.; Fu, X.; Liu, J.; Qi, Y.; Li, S.; Wang, H. The involvement of integrin b 1 signaling in the migration and myofibroblastic differentiation of skin fibroblasts on anisotropic collagen-containing nanofibers. Biomaterials 2012, 33, 1791–1800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hadisi, Z.; Nourmohammadi, J.; Nassiri, S.M. The antibacterial and anti-inflammatory investigation of Lawsonia Inermis -gelatin-starch nano-fibrous dressing in burn wound. Int. J. Biol. Macromol. 2018, 107, 2008–2019. [Google Scholar] [CrossRef]
- Kheradvar, S.A.; Nourmohammadi, J.; Tabesh, H.; Bagheri, B. Starch nanoparticle as a vitamin E-TPGS carrier loaded in silk fibroin-poly(vinyl alcohol)-Aloe vera nanofibrous dressing. Colloids Surf. B Biointerfaces 2018, 166, 9–16. [Google Scholar] [CrossRef]
- Ghorbani, M.; Nezhad-mokhtari, P.; Ramazani, S. Aloe vera-loaded nanofibrous scaffold based on Zein / Polycaprolactone / Collagen for wound healing. Int. J. Biol. Macromol. 2020, 153, 921–930. [Google Scholar] [CrossRef]
- Rath, G.; Hussain, T.; Chauhan, G.; Tarun, G.; Amit, K. Collagen nanofiber containing silver nanoparticles for improved wound-healing applications. J. Drug Target. 2015, 24, 520–529. [Google Scholar] [CrossRef]
- Khartini, W.; Abdul, W.; Hakim, A.; Razak, A.; Ng, M.H.; Guarino, V.; Susanti, D. Encapsulation and Characterization of Gentamicin Sulfate in the Collagen Added Electrospun Nanofibers for Skin Regeneration. J. Funct. Biomater. 2018, 9, 36. [Google Scholar]
- Tort, S.; Acartürk, F.; Be, A. Evaluation of three-layered doxycycline-collagen loaded nanofiber wound dressing. Int. J. Pharm. 2017, 529, 642–653. [Google Scholar] [CrossRef]
- Selvaraj, S.; Duraipandy, N.; Syamala, M.; Fathima, N.N. Ant-oxidant enriched hybrid nanofibers: Effect on mechanical stability and biocompatibility. Int. J. Biol. Macromol. 2018, 117, 209–217. [Google Scholar] [CrossRef]
- Yao, C.; Chen, K.; Chen, Y.; Li, S.; Huang, C. Lithospermi radix extract-containing bilayer nano fi ber sca ff old for promoting wound healing in a rat model. Mater. Sci. Eng. C 2019, 96, 850–858. [Google Scholar] [CrossRef]
- Ribeiro, N.; Sousa, A.; Cunha-reis, C.; Oliveira, A.L.; Granja, P.L.; Monteiro, F.J.; Sousa, S.R. New prospects in skin regeneration and repair using nanophased hydroxyapatite embedded in collagen nanofibers. Nanomed. Nanotech. Biol. Med. 2021, 33, 102353. [Google Scholar] [CrossRef]
- Hou, J.; Chen, L.; Zhou, M.; Li, J.; Liu, J.; Fang, H.; Zeng, Y.; Sun, J.; Wang, Z. Multi-Layered Polyamide/Collagen Scaffolds with Topical Sustained Release of N-Acetylcysteine for Promoting Wound Healing. Int. J. Nanomed. 2020, 15, 1349–1361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, T.; Sui, B.; Mo, X.; Sun, J. Multifunctional and biomimetic fish collagen/bioactive glass nanofibers: Fabrication, antibacterial activity and inducing skin regeneration in vitro and in vivo. Int. J. Nanomed. 2017, 12, 3495–3507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kandhasamy, S.; Perumal, S.; Madhan, B.; Umamaheswari, N.; Banday, J.A.; Perumal, P.T.; Santhanakrishnan, V.P. Synthesis and Fabrication of Collagen-Coated Ostholamide Electrospun Nanofiber Scaffold for Wound Healing. ACS Appl. Mater. interfaces 2017, 9, 8556–8568. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.; Kuan, C.; Wu, H.; Tsai, J.; Chen, T.; Hsieh, D.; Wang, T. Tailored design of electrospun composite nanofibers with staged release of multiple angiogenic growth factors for chronic wound healing. Acta Biomater. 2014, 10, 4156–4166. [Google Scholar] [CrossRef]
- Lee, C.; Chang, S.; Chen, W.; Hung, K.; Lin, Y.; Liu, S.; Hsieh, M.; Pang, J.S.; Juang, J. Augmentation of diabetic wound healing and enhancement of collagen content using nanofibrous glucophage-loaded collagen/PLGA scaffold membranes. J. Colloid Interface Sci. 2015, 439, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Kau, Y.; Chou, C.; Chen, J.; Wu, R.; Yeh, W. Electrospun PLGA / collagen nanofibrous membrane as early-stage wound dressing. J. Memb. Sci. 2010, 355, 53–59. [Google Scholar] [CrossRef]
- Lin, J.; Li, C.; Zhao, Y.; Hu, J.; Zhang, L.-M. Co-electrospun Nanofibrous Membranes of Collagen and Zein for Wound Healing. ACS Appl. Mater. Interfaces 2012, 4, 1050–1057. [Google Scholar] [CrossRef]
- Chen, J.; Chang, G.; Chen, J. Electrospun collagen/chitosan nanofibrous membrane as wound dressing. Colloids Surf. A 2008, 314, 183–188. [Google Scholar] [CrossRef]
- Venugopal, J.R.; Zhang, Y.; Ramakrishna, S. In Vitro Culture of Human Dermal Fibroblasts on Electrospun Polycaprolactone Collagen Nanofibrous Membrane. Artif. Organs 2006, 30, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Ahmadian, S.; Ghorbani, M.; Mahmoodzadeh, F. Silver sulfadiazine-loaded electrospun ethyl cellulose / polylactic acid / collagen nano fi brous mats with antibacterial properties for wound healing. Int. J. Biol. Macromol. 2020, 162, 1555–1565. [Google Scholar] [CrossRef] [PubMed]
- Angele, P.; Abke, J.; Kujat, R.; Faltermeier, H.; Schumann, D.; Nerlich, M.; Kinner, B.; Englert, C.; Ruszczak, Z.; Mehrl, R.; et al. Influence of different collagen species on physico-chemical properties of crosslinked collagen matrices. Biomaterials 2004, 25, 2831–2841. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.K.; Liu, D.C. Comparison of physical–chemical properties of type I collagen from different species. Food Chem. 2006, 99, 244–251. [Google Scholar] [CrossRef]
- Lin, K.; Liu, D.C. Effect of pepsin digestion at different temperatures and time on properties of telopeptide-poor collagen from bird feet. Food Chem. 2006, 94, 621–625. [Google Scholar] [CrossRef]
- Li, H.; Liu, B.L.; Gao, L.Z.; Chen, H.L. Studies on bullfrog skin collagen. Food Chem. 2004, 84, 65–69. [Google Scholar] [CrossRef]
- Wang, T.; Lew, J.; Premkumar, J.; Poh, C.L.; Naing, M.W. Production of recombinant collagen: State of the art and challenges. Eng. Biol. 2017, 1, 18–23. [Google Scholar] [CrossRef] [Green Version]
- Willard, J.J.; Drexler, J.W.; Das, A.; Roy, S.; Shilo, S.; Shoseyov, O.; Powell, M.H. Plant-derived human collagen scaffolds for skin tissue engineering. Tissue Eng. Part. A 2013, 19, 1507–1518. [Google Scholar] [CrossRef]
- Hofman, K.; Tucker, N.; Stanger, J.; Staiger, M.; Marshall, S.; Hall, B. Effects of the molecular format of collagen on characteristics of electrospun fibres. J. Mater. Sci. 2012, 47, 1148–1155. [Google Scholar] [CrossRef]
- Zeugolis, D.; Li, B.; Lareu, R.R.; Chan, C.; Raghunath, M. Collagen solubility testing, a quality assurance step for reproducible electro-spun nano-fibre fabrication. A technical note. J. Biomater. Sci. Polym. Ed. 2008, 19, 1307–1317. [Google Scholar] [CrossRef] [Green Version]
- Matthews, J.A.; Wnek, G.E.; Simpson, D.G.; Bowlin, G.L. Electrospinning of collagen nanofibers. Biomacromolecules 2002, 3, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Zeugolis, D.I.; Khew, S.T.; Yew, E.S.; Ekaputra, A.K.; Tong, Y.W.; Yung, L.Y.L.; Hutmacher, D.W.; Sheppard, C.; Raghunath, M. Electro-spinning of pure collagen nano-fibres—just an expensive way to make gelatin? Biomaterials 2008, 29, 2293–2305. [Google Scholar] [CrossRef]
- Yang, L.; Fitie, C.F.; van der Werf, K.O.; Bennink, M.L.; Dijkstra, P.J.; Feijen, J. Mechanical properties of single electrospun collagen type I fibers. Biomaterials 2008, 29, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Elamparithi, A.; Punnoose, A.M.; Kuruvilla, S. Electrospun type 1 collagen matrices preserving native ultrastructure using benign binary solvent for cardiac tissue engineering. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1318–1325. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mbese, Z.; Alven, S.; Aderibigbe, B.A. Collagen-Based Nanofibers for Skin Regeneration and Wound Dressing Applications. Polymers 2021, 13, 4368. https://doi.org/10.3390/polym13244368
Mbese Z, Alven S, Aderibigbe BA. Collagen-Based Nanofibers for Skin Regeneration and Wound Dressing Applications. Polymers. 2021; 13(24):4368. https://doi.org/10.3390/polym13244368
Chicago/Turabian StyleMbese, Zintle, Sibusiso Alven, and Blessing Atim Aderibigbe. 2021. "Collagen-Based Nanofibers for Skin Regeneration and Wound Dressing Applications" Polymers 13, no. 24: 4368. https://doi.org/10.3390/polym13244368
APA StyleMbese, Z., Alven, S., & Aderibigbe, B. A. (2021). Collagen-Based Nanofibers for Skin Regeneration and Wound Dressing Applications. Polymers, 13(24), 4368. https://doi.org/10.3390/polym13244368








