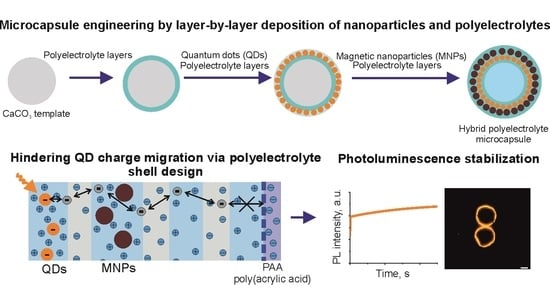
Nanoparticle-Doped Hybrid Polyelectrolyte Microcapsules with Controlled Photoluminescence for Potential Bioimaging Applications
Abstract
:1. Introduction
2. Materials and Methods
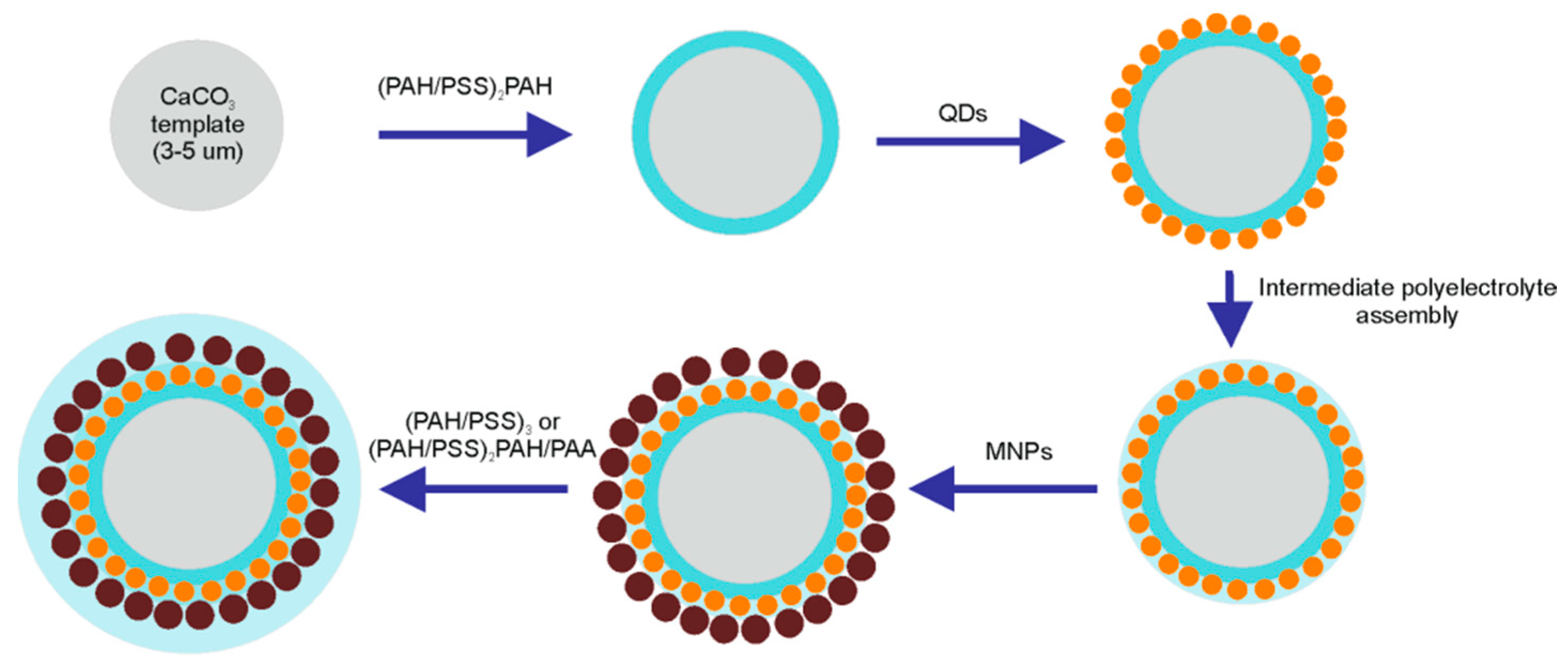
2.1. Fabrication of Nanoparticle-Doped Hybrid Polyelectrolyte Microcapsules
2.2. Measurements of the Zeta-Potential and Size
2.3. Analysis of Microcapsule Photoluminescence
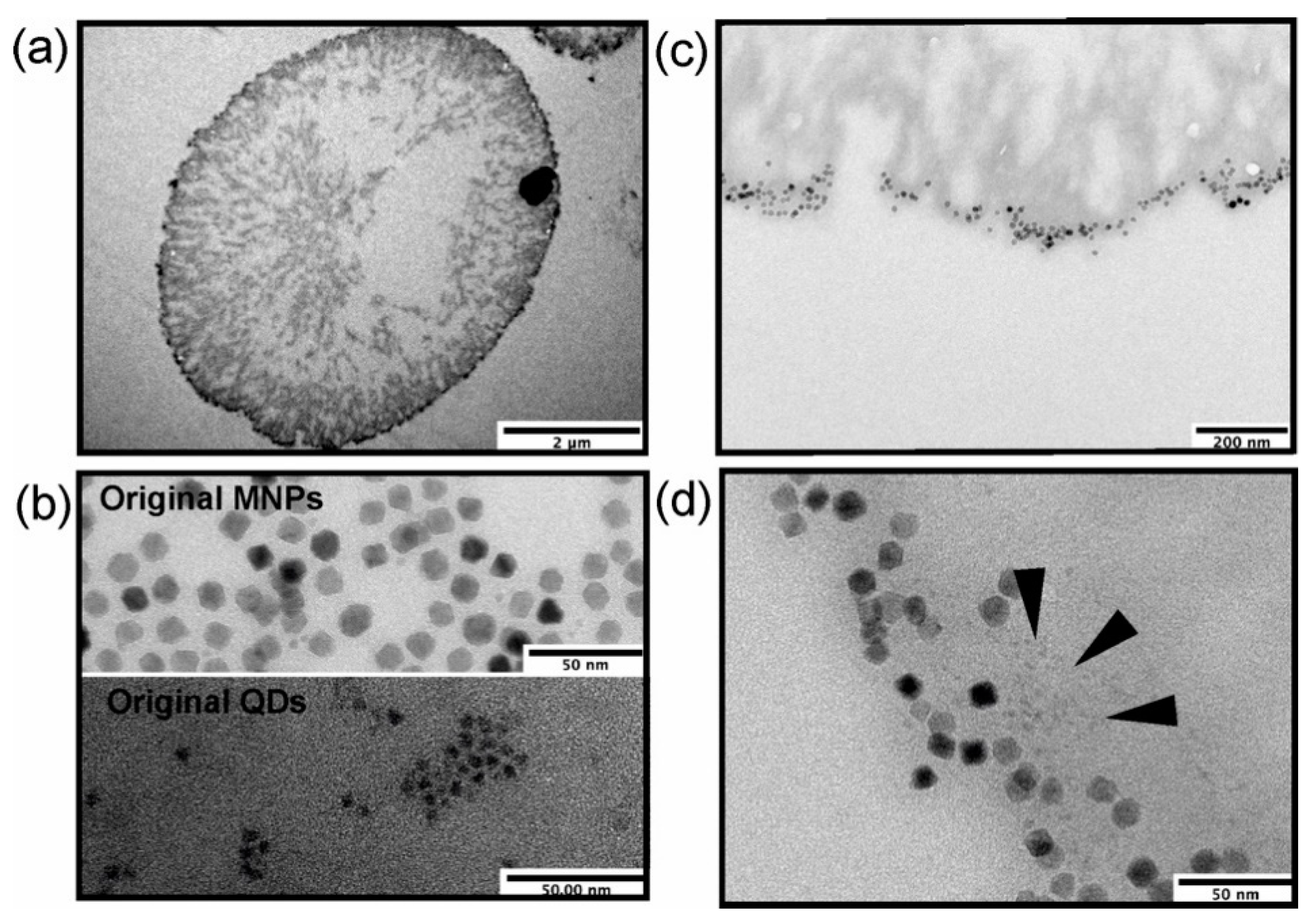
2.4. Transmission Electron Microscopy
2.5. Preparation of Microcapsule Sections and Study of Magnetization Distribution
3. Results and Discussion
3.1. Engineering of Nanoparticle-Doped Hybrid Polyelectrolyte Microcapsules
3.2. Analysis of Microcapsules’ Photoluminescence Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- De Luca, M.; Ferraro, M.M.; Hartmann, R.; Rivera-Gil, P.; Klingl, A.; Nazarenus, M.; Ramirez, A.; Parak, W.J.; Bucci, C.; Rinaldi, R.; et al. Advances in use of capsule-based fluorescent sensors for measuring acidification of endocytic compartments in cells with altered expression of V-ATPase subunit V1G1. ACS Appl. Mater. Interfaces 2015, 7, 15052–15060. [Google Scholar] [CrossRef]
- Novoselova, M.V.; Voronin, D.V.; Abakumova, T.O.; Demina, P.A.; Petrov, A.V.; Petrov, V.V.; Zatsepin, T.S.; Sukhorukov, G.B.; Gorin, D.A. Focused ultrasound-mediated fluorescence of composite microcapsules loaded with magnetite nanoparticles: In vitro and in vivo study. Colloids Surf. B 2019, 181, 680–687. [Google Scholar] [CrossRef]
- Voronin, D.V.; Sindeeva, O.A.; Kurochkin, M.A.; Mayorova, O.; Fedosov, I.V.; Semyachkina-Glushkovskaya, O.; Gorin, D.A.; Tuchin, V.V.; Sukhorukov, G.B. In vitro and in vivo visualization and trapping of fluorescent magnetic microcapsules in a bloodstream. ACS Appl. Mater. Interfaces 2017, 9, 6885–6893. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Poston, R.N.; Gould, D.J.; Sukhorukov, G.B. Magnetically targetable microcapsules display subtle changes in permeability and drug release in response to a biologically compatible low frequency alternating magnetic field. Mater. Sci. Eng. C 2019, 94, 647–655. [Google Scholar] [CrossRef]
- Sciortino, F.; Casterou, G.; Eliat, P.-A.; Troadec, M.-B.; Gaillard, C.; Chevance, S.; Kahn, M.L.; Gauffre, F. Simple engineering of polymer–nanoparticle hybrid nanocapsules. ChemNanoMat 2016, 2, 796–799. [Google Scholar] [CrossRef]
- Mayorova, O.A.; Sindeeva, O.A.; Lomova, M.V.; Gusliakova, O.I.; Tarakanchikova, Y.V.; Tyutyaev, E.V.; Pinyaev, S.I.; Kulikov, O.A.; German, S.V.; Pyataev, N.A.; et al. Endovascular addressing improves the effectiveness of magnetic targeting of drug carrier. Comparison with the conventional administration method. Nanomed. Nanotechnol. Biol. Med. 2020, 28, 102184. [Google Scholar] [CrossRef] [PubMed]
- Gaponik, N.; Radtchenko, I.L.; Sukhorukov, G.B.; Rogach, A.L. Luminescent polymer microcapsules addressable by a magnetic field. Langmuir 2004, 20, 1449–1452. [Google Scholar] [CrossRef]
- Nifontova, G.; Efimov, A.; Agapova, O.; Agapov, I.; Nabiev, I.; Sukhanova, A. Bioimaging tools based on polyelectrolyte microcapsules encoded with fluorescent semiconductor nanoparticles: Design and characterization of the fluorescent properties. Nanoscale Res. Lett. 2019, 14, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.; Di, Y.; Chen, D.; Madrid, K.; Zhang, M.; Tian, C.; Tang, L.; Gu, Y. Combined chemo-and photo-thermal therapy delivered by multifunctional theranostic gold nanorod-loaded microcapsules. Nanoscale 2015, 7, 8884–8897. [Google Scholar] [CrossRef]
- Rui, Y.; Pang, B.; Zhang, J.; Liu, Y.; Hu, H.; Liu, Z.; Ama Baidoo, S.; Liu, C.; Zhao, Y.; Li, S. Near-infrared light-activatable siRNA delivery by microcapsules for combined tumour therapy. Artif. Cells Nanomed. Biotechnol. 2018, 46, 15–24. [Google Scholar] [CrossRef] [Green Version]
- Roy, S.; Zhu, D.; Parak, W.J.; Feliu, N. Lysosomal proton buffering of poly(ethylenimine) measured in situ by fluorescent pH-sensor microcapsules. ACS Nano 2020, 14, 8012–8023. [Google Scholar] [CrossRef]
- Das, P.; Ganguly, S.; Banerjee, S.; Das, N.C. Graphene based emergent nanolights: A short review on the synthesis, properties and application. Rev. Chem. Intermed. 2019, 45, 3823–3853. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, P.; Itzhaki, E.; Hadad, E.; Gedanken, A.; Margel, S. Microwave-synthesized polysaccharide-derived carbon dots as therapeutic cargoes and toughening agents for elastomeric gels. ACS Appl. Mater. Interfaces 2020, 12, 51940–51951. [Google Scholar] [CrossRef]
- Kage, D.; Hoffmann, K.; Nifontova, G.; Krivenkov, V.; Sukhanova, A.; Nabiev, I.; Resch-Genger, U. Tempo-spectral multiplexing in flow cytometry with lifetime detection using QD-encoded polymer beads. Sci. Rep. 2020, 10, 653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brazhnik, K.; Sokolova, Z.; Baryshnikova, M.; Bilan, R.; Efimov, A.; Nabiev, I.; Sukhanova, A. Quantum dot-based lab-on-a-bead system for multiplexed detection of free and total prostate-specific antigens in clinical human serum samples. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1065–1075. [Google Scholar] [CrossRef] [PubMed]
- Linkov, P.; Krivenkov, V.; Nabiev, I.; Samokhvalov, P. High quantum yield CdSe/ZnS/CdS/ZnS multishell quantum dots for biosensing and optoelectronic applications. Mater. Today Proc. 2016, 3, 104–108. [Google Scholar] [CrossRef]
- Resch-Genger, U.; Grabolle, M.; Cavaliere-Jaricot, S.; Nitschke, R.; Nann, T. Quantum dots versus organic dyes as fluorescent labels. Nat. Methods 2008, 5, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Freitas, D.V.; Passos, S.G.B.; Dias, J.M.M.; Mansur, A.; Carvalho, S.M.; Mansur, H.; Navarro, M. Toward greener electrochemical synthesis of composition-tunable luminescent CdX-based (X = Te, Se, S) quantum dots for bioimaging cancer cells. Sens. Actuators B 2017, 250, 233–243. [Google Scholar] [CrossRef]
- Ji, B.; Koley, S.; Slobodkin, I.; Remennik, S.; Banin, U. ZnSe/ZnS core/shell quantum dots with superior optical properties through thermodynamic shell growth. Nano Lett. 2020, 20, 2387–2395. [Google Scholar] [CrossRef]
- Bilan, R.; Nabiev, I.; Sukhanova, A. Quantum dot-based nanotools for bioimaging, diagnostics, and drug delivery. Chembiochem 2016, 22, 2103–2114. [Google Scholar] [CrossRef] [PubMed]
- Rakovich, T.Y.; Mahfoud, O.K.; Mohamed, B.M.; Prina-Mello, A.; Crosbie-Staunton, K.; Van Den Broeck, T.; De Kimpe, L.; Sukhanova, A.; Baty, D.; Rakovich, A.; et al. Highly sensitive single domain antibody–quantum dot conjugates for detection of HER2 biomarker in lung and breast cancer cells. ACS Nano 2014, 6, 5682–5695. [Google Scholar] [CrossRef]
- Xu, J.; Tehrani, K.F.; Kner, P. Multicolor 3D super-resolution imaging by quantum dot stochastic optical reconstruction microscopy. ACS Nano 2015, 3, 2917–2925. [Google Scholar] [CrossRef] [PubMed]
- Hanne, J.; Falk, H.J.; Görlitz, F.; Hoyer, P.; Engelhardt, J.; Sahl, S.J.; Hell, S.W. STED nanoscopy with fluorescent quantum dots. Nat. Commun. 2015, 6, 7127. [Google Scholar] [CrossRef] [Green Version]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of nanoparticle toxicity on their physical and chemical properties. Nanoscale Res. Lett. 2018, 13, 44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cooper, D.R.; Dimitrijevic, N.M.; Nadeau, J.L. Photosensitization of CdSe/ZnS QDs and reliability of assays for reactive oxygen species production. Nanoscale 2010, 2, 114–121. [Google Scholar] [CrossRef]
- Tabish, T.A.; Scotton, C.J.; Ferguson, D.C.J.; Lin, L.; van der Veen, A.; Lowry, S.; Ali, M.; Jabeen, F.; Ali, M.; Winyard, P.G.; et al. Biocompatibility and toxicity of graphene quantum dots for potential application in photodynamic therapy. Nanomedicine 2018, 13, 1923–1937. [Google Scholar] [CrossRef] [Green Version]
- Romoser, A.; Ritter, D.; Majitha, R.; Meissner, K.E.; McShane, M.; Sayes, C.M. Mitigation of quantum dot cytotoxicity by microencapsulation. PLoS ONE 2011, 7, e22079. [Google Scholar] [CrossRef]
- Nifontova, G.; Krivenkov, V.; Zvaigzne, M.; Samokhvalov, P.; Efimov, A.E.; Agapova, O.I.; Agapov, I.I.; Korostylev, E.; Zarubin, S.; Karaulov, A.; et al. Controlling charge transfer from quantum dots to polyelectrolyte layers extends prospective applications of magneto-optical microcapsules. ACS Appl. Mater. Interfaces 2020, 12, 35882–35894. [Google Scholar] [CrossRef]
- Rodríguez-Ramos, A.; Marín-Caba, L.; Iturrioz-Rodríguez, N.; Padín-González, E.; García-Hevia, L.; Oliveira, T.M.; Corea-Duarte, M.A.; Fanarraga, M.L. Design of polymeric and biocompatible delivery systems by dissolving mesoporous silica templates. Int. J. Mol. Sci. 2020, 21, 9573. [Google Scholar] [CrossRef] [PubMed]
- Nifontova, G.; Zvaigzne, M.; Baryshnikova, M.; Korostylev, E.; Ramos-Gomes, F.; Alves, F.; Nabiev, I.; Sukhanova, A. Next-generation theranostic agents based on polyelectrolyte microcapsules encoded with semiconductor nanocrystals: Development and functional characterization. Nanoscale Res. Lett. 2018, 13, 30. [Google Scholar] [CrossRef] [Green Version]
- Nifontova, G.; Ramos-Gomes, F.; Baryshnikova, M.; Alves, F.; Nabiev, I.; Sukhanova, A. Cancer cell targeting with functionalized quantum dot-encoded polyelectrolyte microcapsules. Front. Chem. (Lausanne Switz.) 2019, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Caruso, F.; Dähne, L.; Decher, G.; De Geest, B.G.; Fan, J.; Feliu, N.; Gogotsi, Y.; Hammond, P.T.; Hersam, M.C.; et al. The future of layer-by-layer assembly: A tribute to ACS Nano associate editor Helmuth Möhwald. ACS Nano 2019, 6, 6151–6169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeannot, L.; Bell, M.; Ashwell, R.; Volodkin, D.; Vikulina, A.S. Internal structure of matrix-type multilayer capsules templated on porous vaterite CaCO3 crystals as probed by staining with a fluorescence dye. Micromachines 2018, 9, 547. [Google Scholar] [CrossRef] [Green Version]
- Nifontova, G.; Ramos-Gomes, F.; Alves, F.; Nabiev, I.; Sukhanova, A. Stimulus-sensitive theranostic delivery systems based on microcapsules encoded with quantum dots and magnetic nanoparticles. In Methods in Molecular Biology, 3rd ed.; Fontes, A., Santos, B., Eds.; Humana: New York, NY, USA, 2020; Volume 2135, pp. 199–212. [Google Scholar] [CrossRef]
- Bilan, R.S.; Krivenkov, V.A.; Berestovoy, M.A.; Efimov, A.E.; Agapov, I.I.; Samokhvalov, P.S.; Nabiev, I.; Sukhanova, A. Engineering of optically encoded microbeads with FRET-free spatially separated quantum-dot layers for multiplexed assays. ChemPhysChem 2017, 18, 970–979. [Google Scholar] [CrossRef] [PubMed]
- Krivenkov, V.; Samokhvalov, P.; Zvaigzne, M.; Martynov, I.; Chistyakov, A.; Nabiev, I. Ligand-mediated photobrightening and photodarkening of CdSe/ZnS quantum dot ensembles. J. Phys. Chem. C 2018, 122, 15761–15771. [Google Scholar] [CrossRef]
- Efimov, A.E.; Agapov, I.I.; Agapova, O.I.; Oleinikov, V.A.; Mezin, A.V.; Molinari, M.; Nabiev, I.; Mochalov, K.E. A novel design of a scanning probe microscope integrated with an ultramicrotome for serial block-face nanotomography. Rev. Sci. Instrum. 2017, 88, 023701. [Google Scholar] [CrossRef]
- Hasegawa, U.; Nomura, S.-I.M.; Kaul, S.C.; Hirano, T.; Akiyoshi, K. Nanogel-quantum dot hybrid nanoparticles for live cell imaging. Biochem. Biophys. Res. Commun. 2005, 331, 917–921. [Google Scholar] [CrossRef]
- Bogdanov, A.M.; Bogdanova, E.A.; Chudakov, D.M.; Gorodnicheva, T.V.; Lukyanov, S.; Lukyanov, K.A. Cell culture medium affects GFP photostability: A solution. Nat. Methods 2009, 6, 859–860. [Google Scholar] [CrossRef]
- Nifontova, G.; Kalenichenko, D.; Baryshnikova, M.; Ramos Gomes, F.; Alves, F.; Karaulov, A.; Nabiev, I.; Sukhanova, A. Biofunctionalized polyelectrolyte microcapsules encoded with fluorescent semiconductor nanocrystals for highly specific targeting and imaging of cancer cells. Photonics 2019, 6, 117. [Google Scholar] [CrossRef] [Green Version]
- Kastl, L.; Sasse, D.; Wulf, V.; Hartmann, R.; Mircheski, J.; Ranke, C.; Carregal-Romero, S.; Martínez-López, J.A.; Fernández-Chacón, R.; Parak, W.J.; et al. Multiple internalization pathways of polyelectrolyte multilayer capsules into mammalian cells. ACS Nano 2013, 7, 6605–6618. [Google Scholar] [CrossRef]
- De Koker, S.; De Geest, B.G.; Singh, S.K.; De Rycke, R.; Naessens, T.; Van Kooyk, Y.; Demeester, J.; De Smedt, S.C.; Grooten, J. Polyelectrolyte microcapsules as antigen delivery vehicles to dendritic cells: Uptake, processing, and cross-presentation of encapsulated antigens. Angew. Chem. Int. Ed. 2009, 48, 8485–8489. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, Y.; Mao, Z.; Gao, C. Cellular uptake of covalent poly(allylamine hydrochloride) microcapsules and its influences on cell functions. Macromol. Biosci. 2012, 11, 1534–1545. [Google Scholar] [CrossRef] [PubMed]
- Zharkov, M.N.; Brodovskaya, E.P.; Kulikov, O.A.; Gromova, E.V.; Ageev, V.P.; Atanova, A.V.; Kozyreva, Z.V.; Tishin, A.M.; Pyatakov, A.P.; Pyataev, N.A.; et al. Enhanced cytotoxicity caused by AC magnetic field for polymer microcapsules containing packed magnetic nanoparticles. Colloids Surf. B 2021, 199, 111548. [Google Scholar] [CrossRef] [PubMed]
- Read, J.E.; Luo, D.; Chowdhury, T.T.; Flower, R.J.; Poston, R.N.; Sukhorukov, G.B.; Gould, D.J. Magnetically responsive layer-by-layer microcapsules can be retained in cells and under flow conditions to promote local drug release without triggering ROS production. Nanoscale 2020, 14, 7735–7748. [Google Scholar] [CrossRef]
- Tabish, T.A.; Zhang, S.; Winyard, P.G. Developing the next generation of graphene-based platforms for cancer therapeutics: The potential role of reactive oxygen species. Redox Biol. 2018, 15, 34–40. [Google Scholar] [CrossRef] [PubMed]
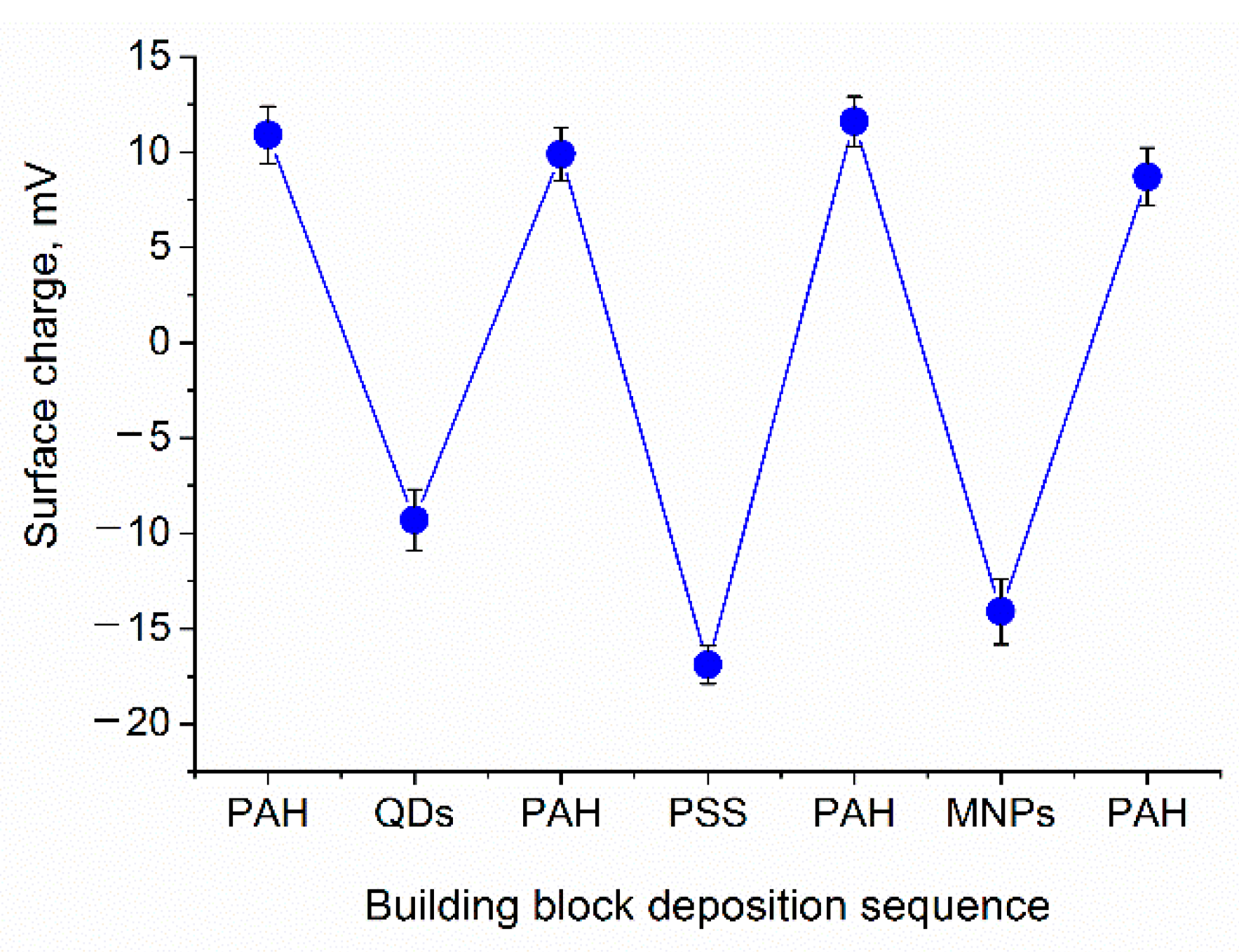

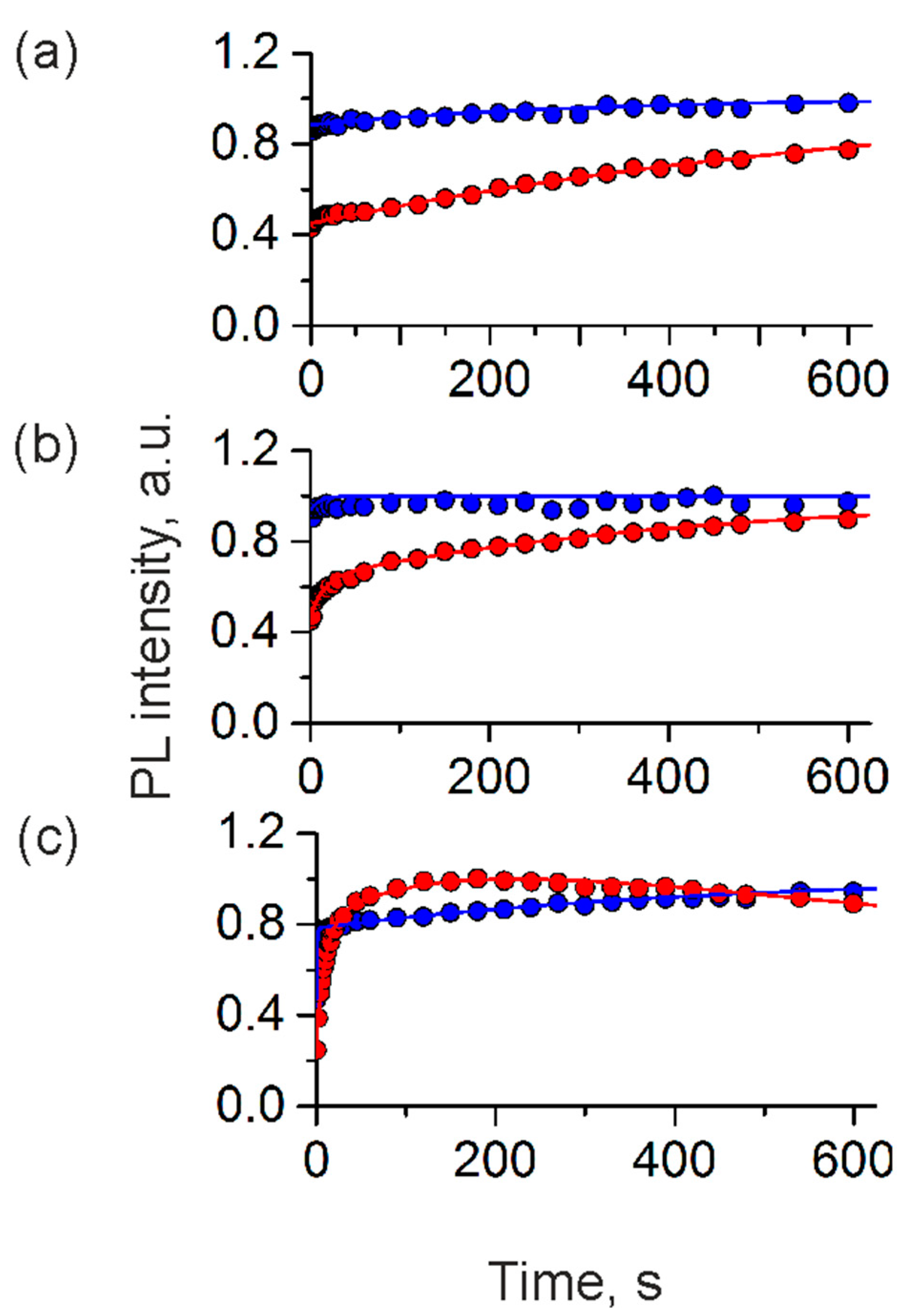
| Building Block | ς-Potential, mV | Hydrodynamic Diameter, nm * |
|---|---|---|
| Calcium carbonate microbeads | −8.86 ± 1.14 | 4137.00 ± 883.70 |
| PSS 70 kDa | −18.10 ± 7.86 | 309.00 ± 88.03 |
| PAH 65 kDa | +25.0 ± 1.85 | 320.60 ± 36.13 |
| PAA 100 kDa | −9.30 ± 2.96 | 298.60 ± 36.90 |
| QDs carboxylated | −36.90 ± 0.31 | 58.70 ± 9.54 |
| MNPs carboxylated | −41.80 ± 4.24 | 31.22 ± 0.46 |
| Parameter | Water | Regular Cell Culture Medium | Phenol-Red-Free Cell Culture Medium | |||
|---|---|---|---|---|---|---|
| Positively charged | Negatively charged | Positively charged | Negatively charged | Positively charged | Negatively charged | |
| A1 | −1.900 | −0.100 | −0.363 | 0.065 | −1.420 | −0.135 |
| k1 | 0.0534 | 0.063 | −0.067 | 0.096 | −0.001 | −0.003 |
| A2 | +2.500 | −0.520 | −0.807 | - | - | - |
| k2 | 0.001 | 0.003 | −0.002 | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nifontova, G.; Krivenkov, V.; Zvaigzne, M.; Efimov, A.; Korostylev, E.; Zarubin, S.; Karaulov, A.; Nabiev, I.; Sukhanova, A. Nanoparticle-Doped Hybrid Polyelectrolyte Microcapsules with Controlled Photoluminescence for Potential Bioimaging Applications. Polymers 2021, 13, 4076. https://doi.org/10.3390/polym13234076
Nifontova G, Krivenkov V, Zvaigzne M, Efimov A, Korostylev E, Zarubin S, Karaulov A, Nabiev I, Sukhanova A. Nanoparticle-Doped Hybrid Polyelectrolyte Microcapsules with Controlled Photoluminescence for Potential Bioimaging Applications. Polymers. 2021; 13(23):4076. https://doi.org/10.3390/polym13234076
Chicago/Turabian StyleNifontova, Galina, Victor Krivenkov, Mariya Zvaigzne, Anton Efimov, Evgeny Korostylev, Sergei Zarubin, Alexander Karaulov, Igor Nabiev, and Alyona Sukhanova. 2021. "Nanoparticle-Doped Hybrid Polyelectrolyte Microcapsules with Controlled Photoluminescence for Potential Bioimaging Applications" Polymers 13, no. 23: 4076. https://doi.org/10.3390/polym13234076
APA StyleNifontova, G., Krivenkov, V., Zvaigzne, M., Efimov, A., Korostylev, E., Zarubin, S., Karaulov, A., Nabiev, I., & Sukhanova, A. (2021). Nanoparticle-Doped Hybrid Polyelectrolyte Microcapsules with Controlled Photoluminescence for Potential Bioimaging Applications. Polymers, 13(23), 4076. https://doi.org/10.3390/polym13234076