Effect of Sodium Hypochlorite Concentration in Continuous Chelation on Dislodgement Resistance of an Epoxy Resin and Hydraulic Calcium Silicate Sealer
Abstract
:1. Introduction
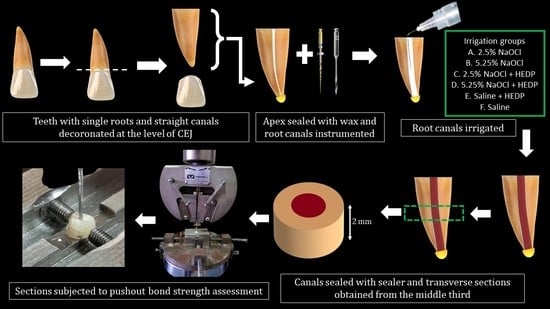
2. Materials and Methods
2.1. Ethics and Teeth
2.2. Treatment Groups and Sample Size Estimation
2.3. Fourier-Transform Infrared Spectroscopy (FTIR)
2.4. Push-Out Bond Strength
2.5. Statistical Analysis
3. Results
3.1. FTIR Analysis
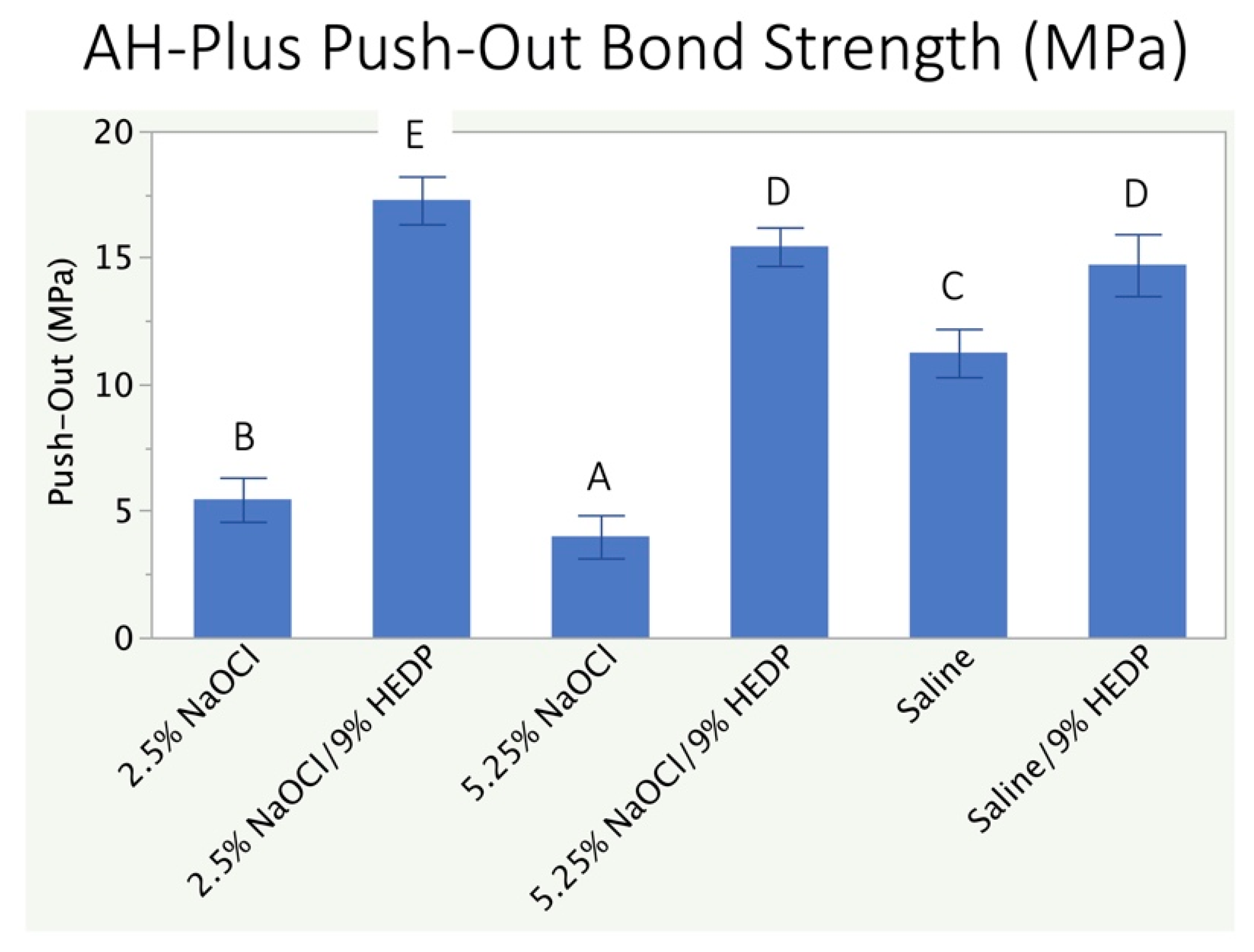
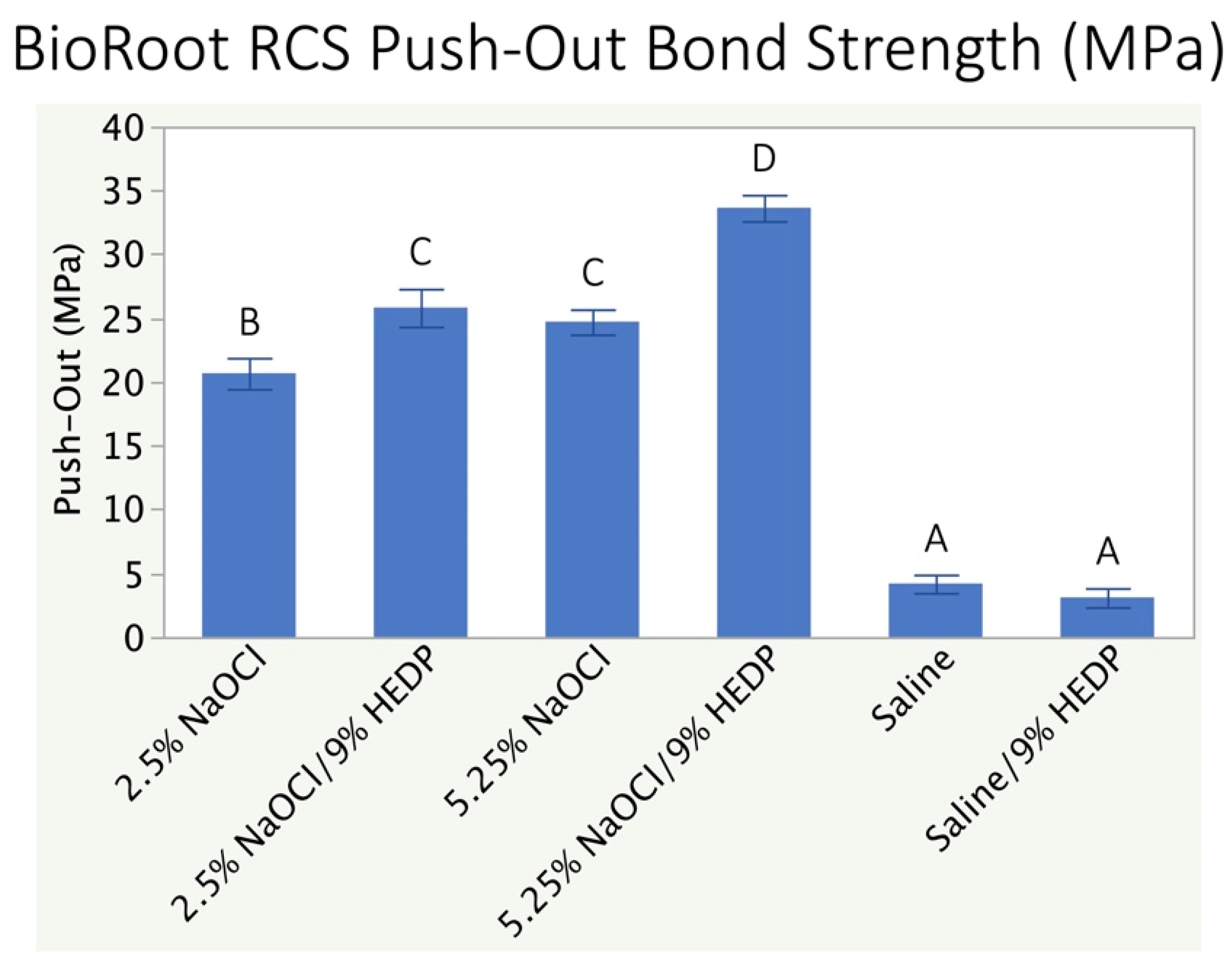
3.2. Push-Out Bond Strength Analysis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McComb, D.; Smith, D.C.; Beagrie, G.S. The Results of in Vivo Endodontic Chemomechanical Instrumentation—A Scanning Electron Microscopic Study. Int. Endod. J. 1976, 9, 11–18. [Google Scholar] [CrossRef]
- Violich, D.R.; Chandler, N.P. The Smear Layer in Endodontics—A Review. Int. Endod. J. 2010, 43, 2–15. [Google Scholar] [CrossRef]
- Lester, K.S.; Boyde, A. Scanning electron microscopy of instrumented, irrigated and filled root canals. Br. Dent. J. 1977, 143, 359–365. [Google Scholar] [CrossRef]
- Lottanti, S.; Gautschi, H.; Sener, B.; Zehnder, M. Effects of Ethylenediaminetetraacetic, Etidronic and Peracetic Acid Irrigation on Human Root Dentin and the Smear Layer. Int. Endod. J. 2009, 42, 335–343. [Google Scholar] [CrossRef]
- Ballal, N.V.; Kandian, S.; Mala, K.; Bhat, K.S.; Acharya, S. Comparison of the Efficacy of Maleic Acid and Ethylenediaminetetraacetic Acid in Smear Layer Removal from Instrumented Human Root Canal: A Scanning Electron Microscopic Study. J. Endod. 2009, 35, 1573–1576. [Google Scholar] [CrossRef]
- Ballal, N.V.; Kundabala, M.; Bhat, S.; Rao, N.; Rao, B.S.S. A Comparative in Vitro Evaluation of Cytotoxic Effects of EDTA and Maleic Acid: Root Canal Irrigants. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2009, 108, 633–638. [Google Scholar] [CrossRef]
- Saleh, I.M.; Ruyter, I.E.; Haapasalo, M.; Ørstavik, D. The Effects of Dentin Pretreatment on the Adhesion of Root-Canal Sealers. Int. Endod. J. 2002, 35, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, U.; Saji, S.; Clarkson, R.; Lalloo, R.; Moule, A.J. Free Active Chlorine in Sodium Hypochlorite Solutions Admixed with Octenidine, Smearoff, Chlorhexidine, and EDTA. J. Endod. 2017, 43, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Rasimick, B.J.; Nekich, M.; Hladek, M.M.; Musikant, B.L.; Deutsch, A.S. Interaction between Chlorhexidine Digluconate and EDTA. J. Endod. 2008, 34, 1521–1523. [Google Scholar] [CrossRef] [PubMed]
- Zehnder, M.; Schmidlin, P.; Sener, B.; Waltimo, T. Chelation in Root Canal Therapy Reconsidered. J. Endod. 2005, 31, 817–820. [Google Scholar] [CrossRef] [PubMed]
- Tartari, T.; Guimarães, B.M.; Amoras, L.S.; Duarte, M.A.H.; Silva e Souza, P.A.R.; Bramante, C.M. Etidronate Causes Minimal Changes in the Ability of Sodium Hypochlorite to Dissolve Organic Matter. Int. Endod. J. 2015, 48, 399–404. [Google Scholar] [CrossRef] [PubMed]
- De-Deus, G.; Namen, F.; Galan Jr, J.; Zehnder, M. Soft Chelating Irrigation Protocol Optimizes Bonding Quality of Resilon/Epiphany Root Fillings. J. Endod. 2008, 34, 703–705. [Google Scholar] [CrossRef] [PubMed]
- Paqué, F.; Rechenberg, D.-K.; Zehnder, M. Reduction of Hard-Tissue Debris Accumulation during Rotary Root Canal Instrumentation by Etidronic Acid in a Sodium Hypochlorite Irrigant. J. Endod. 2012, 38, 692–695. [Google Scholar] [CrossRef]
- Ballal, N.V.; Das, S.; Rao, B.S.S.; Zehnder, M.; Mohn, D. Chemical, Cytotoxic and Genotoxic Analysis of Etidronate in Sodium Hypochlorite Solution. Int. Endod. J. 2019, 52, 1228–1234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zollinger, A.; Mohn, D.; Zeltner, M.; Zehnder, M. Short-term Storage Stability of Na OC l Solutions When Combined with Dual Rinse HEDP. Int. Endod. J. 2018, 51, 691–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marending, M.; Luder, H.U.; Brunner, T.J.; Knecht, S.; Stark, W.J.; Zehnder, M. Effect of Sodium Hypochlorite on Human Root Dentin–Mechanical, Chemical and Structural Evaluation. Int. Endod. J. 2007, 40, 786–793. [Google Scholar] [CrossRef]
- Li, G.; Niu, L.; Zhang, W.; Olsen, M.; De-Deus, G.; Eid, A.A.; Chen, J.; Pashley, D.H.; Tay, F.R. Ability of New Obturation Materials to Improve the Seal of the Root Canal System: A Review. Acta Biomater. 2014, 10, 1050–1063. [Google Scholar] [CrossRef] [Green Version]
- Friedman, S.; Komorowski, R.; Maillet, W.; Klimaite, R.; Nguyen, H.Q.; Torneck, C.D. In Vivo Resistance of Coronally Induced Bacterial Ingress by an Experimental Glass Ionomer Cement Root Canal Sealer. J. Endod. 2000, 26, 1–5. [Google Scholar] [CrossRef]
- Saunders, W.P.; Saunders, E.M. The Root Filling and Restoration Continuum—Prevention of Long-Term Endodontic Failures. Alpha Omegan 1997, 90, 40–46. [Google Scholar]
- Zmener, O.; Spielberg, C.; Lamberghini, F.; Rucci, M. Sealing Properties of a New Epoxy Resin-based Root-canal Sealer. Int. Endod. J. 1997, 30, 332–334. [Google Scholar] [CrossRef]
- Resende, L.M.; Rached-Junior, F.J.A.; Versiani, M.A.; Souza-Gabriel, A.E.; Miranda, C.E.S.; Silva-Sousa, Y.T.C.; Sousa Neto, M.D. A Comparative Study of Physicochemical Properties of AH Plus, Epiphany, and Epiphany SE Root Canal Sealers. Int. Endod. J. 2009, 42, 785–793. [Google Scholar] [CrossRef]
- Donnermeyer, D.; Bürklein, S.; Dammaschke, T.; Schäfer, E. Endodontic Sealers Based on Calcium Silicates: A Systematic Review. Odontology 2019, 107, 421–436. [Google Scholar] [CrossRef]
- Pane, E.S.; Palamara, J.E.A.; Messer, H.H. Critical Evaluation of the Push-out Test for Root Canal Filling Materials. J. Endod. 2013, 39, 669–673. [Google Scholar] [CrossRef]
- Neelakantan, P.; Sharma, S.; Shemesh, H.; Wesselink, P.R. Influence of Irrigation Sequence on the Adhesion of Root Canal Sealers to Dentin: A Fourier Transform Infrared Spectroscopy and Push-out Bond Strength Analysis. J. Endod. 2015, 41, 1108–1111. [Google Scholar] [CrossRef]
- Nunes, V.H.; Silva, R.G.; Alfredo, E.; Sousa-Neto, M.D.; Silva-Sousa, Y.T.C. Adhesion of Epiphany and AH Plus Sealers to Human Root Dentin Treated with Different Solutions. Braz. Dent. J. 2008, 19, 46–50. [Google Scholar] [CrossRef] [Green Version]
- Benezra, M.K.; Wismayer, P.S.; Camilleri, J. Interfacial Characteristics and Cytocompatibility of Hydraulic Sealer Cements. J. Endod. 2018, 44, 1007–1017. [Google Scholar] [CrossRef]
- Sarkar, N.K.; Caicedo, R.; Ritwik, P.; Moiseyeva, R.; Kawashima, I. Physicochemical Basis of the Biologic Properties of Mineral Trioxide Aggregate. J. Endod. 2005, 31, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Prüllage, R.-K.; Urban, K.; Schäfer, E.; Dammaschke, T. Material Properties of a Tricalcium Silicate–Containing, a Mineral Trioxide Aggregate–Containing, and an Epoxy Resin–Based Root Canal Sealer. J. Endod. 2016, 42, 1784–1788. [Google Scholar] [CrossRef] [PubMed]
- Zamparini, F.; Siboni, F.; Prati, C.; Taddei, P.; Gandolfi, M.G. Properties of calcium silicate-monobasic calcium phosphate materials for endodontics containing tantalum pentoxide and zirconium oxide. Clin. Oral Investig. 2019, 23, 445–457. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.M.; Palma, P.J.; Ramos, J.C.; Cabrita, A.S.; Friedman, S. Periapical Inflammation Subsequent to Coronal Inoculation of Dog Teeth Root Filled with Resilon/Epiphany in 1 or 2 Treatment Sessions with Chlorhexidine Medication. J. Endod. 2014, 40, 837–841. [Google Scholar] [CrossRef]
- Neelakantan, P.; Varughese, A.A.; Sharma, S.; Subbarao, C.V.; Zehnder, M.; De-Deus, G. Continuous Chelation Irrigation Improves the Adhesion of Epoxy Resin-based Root Canal Sealer to Root Dentin. Int. Endod. J. 2012, 45, 1097–1102. [Google Scholar] [CrossRef] [PubMed]
- de Lopes, C.C.A.; Limirio, P.H.J.O.; Novais, V.R.; Dechichi, P. Fourier Transform Infrared Spectroscopy (FTIR) Application Chemical Characterization of Enamel, Dentin and Bone. Appl. Spectrosc. Rev. 2018, 53, 747–769. [Google Scholar] [CrossRef]
- Tartari, T.; Bachmann, L.; Zancan, R.F.; Vivan, R.R.; Duarte, M.A.H.; Bramante, C.M. Analysis of the Effects of Several Decalcifying Agents Alone and in Combination with Sodium Hypochlorite on the Chemical Composition of Dentin. Int. Endod. J. 2018, 51, e42–e54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamalasanan, R.R.; Devarasanahalli, S.V.; Aswathanarayana, R.M.; Rashmi, K.; Gowda, Y.; Nadig, R.R. Effect of 5% Chlorine Dioxide Irrigant on Micro Push out Bond Strength of Resin Sealer to Radicular Dentin: An in Vitro Study. J. Clin. Diagnostic Res. 2017, 11, ZC49. [Google Scholar] [CrossRef] [PubMed]
- de Rebolloso, B.E.; Gancedo, C.L.; García, B.E.; Pérez, H.J.J. Effect of exposure to root canal irrigants on the push-out bond strength of calcium silicate-based cements. Clin. Oral Investig. 2021, 35, 267–274. [Google Scholar] [CrossRef]
- Neelakantan, P.; Subbarao, C.; Subbarao, C.V.; De-Deus, G.; Zehnder, M. The Impact of Root Dentin Conditioning on Sealing Ability and Push-out Bond Strength of an Epoxy Resin Root Canal Sealer. Int. Endod. J. 2011, 44, 491–498. [Google Scholar] [CrossRef] [Green Version]
- Paqué, F.; Sirtes, G. Apical Sealing Ability of Resilon/Epiphany versus Gutta-percha/AH Plus: Immediate and 16-months Leakage. Int. Endod. J. 2007, 40, 722–729. [Google Scholar] [CrossRef]
- Barborka, B.J.; Woodmansey, K.F.; Glickman, G.N.; Schneiderman, E.; He, J. Long-Term Clinical Outcome of Teeth Obturated with Resilon. J. Endod. 2017, 43, 556–560. [Google Scholar] [CrossRef]
- Tartari, T.; Bachmann, L.; Maliza, A.G.A.; Andrade, F.B.; Duarte, M.A.H.; Bramante, C.M. Tissue Dissolution and Modifications in Dentin Composition by Different Sodium Hypochlorite Concentrations. J. Appl. Oral Sci. 2016, 24, 291–298. [Google Scholar] [CrossRef]
- Srivastava, A.; Yadav, D.S.; Rao, M.; Rao, H.M.; Arun, A.; Siddique, R. Evaluation of Push-out Bond Strength of BioRoot RCS and AH Plus after Using Different Irrigants: An in Vitro Study. J. Conserv. Dent. 2020, 23, 26. [Google Scholar] [CrossRef]
- Alfawaz, H.; Alqedairi, A.; Alebdi, A.; Allahem, Z.; Alsubait, S.; Alharbi, H. Bond strength of BioRoot RCS relative to different endodontic bioceramic sealers: In vitro push-out test. Pak. Oral Dent. J. 2018, 38, 528–531. [Google Scholar]
- Donnermeyer, D.; Dornseifer, P.; Schäfer, E.; Dammaschke, T. The Push-out Bond Strength of Calcium Silicate-Based Endodontic Sealers. Head Face Med. 2018, 14, 13. [Google Scholar] [CrossRef]
- Doğan, H.; Çalt, S. Effects of Chelating Agents and Sodium Hypochlorite on Mineral Content of Root Dentin. J. Endod. 2001, 27, 578–580. [Google Scholar] [CrossRef] [PubMed]
- Vilanova, W.V.; Carvalho-Junior, J.R.; Alfredo, E.; Sousa-Neto, M.D.; Silva-Sousa, Y.T.C. Effect of Intracanal Irrigants on the Bond Strength of Epoxy Resin-based and Methacrylate Resin-based Sealers to Root Canal Walls. Int. Endod. J. 2012, 45, 42–48. [Google Scholar] [CrossRef]
- Rath, P.P.; Yiu, C.K.Y.; Matinlinna, J.P.; Kishen, A.; Neelakantan, P. The Effects of Sequential and Continuous Chelation on Dentin. Dent. Mater. 2020, 36, 1655–1665. [Google Scholar] [CrossRef] [PubMed]
- Zancan, R.F.; Hadis, M.; Burgess, D.; Zhang, Z.J.; di Maio, A.; Tomson, P.; Duarte, M.A.H.; Camilleri, J. A Matched Irrigation and Obturation Strategy for Root Canal Therapy. Sci. Rep. 2021, 11, 4666. [Google Scholar] [CrossRef] [PubMed]
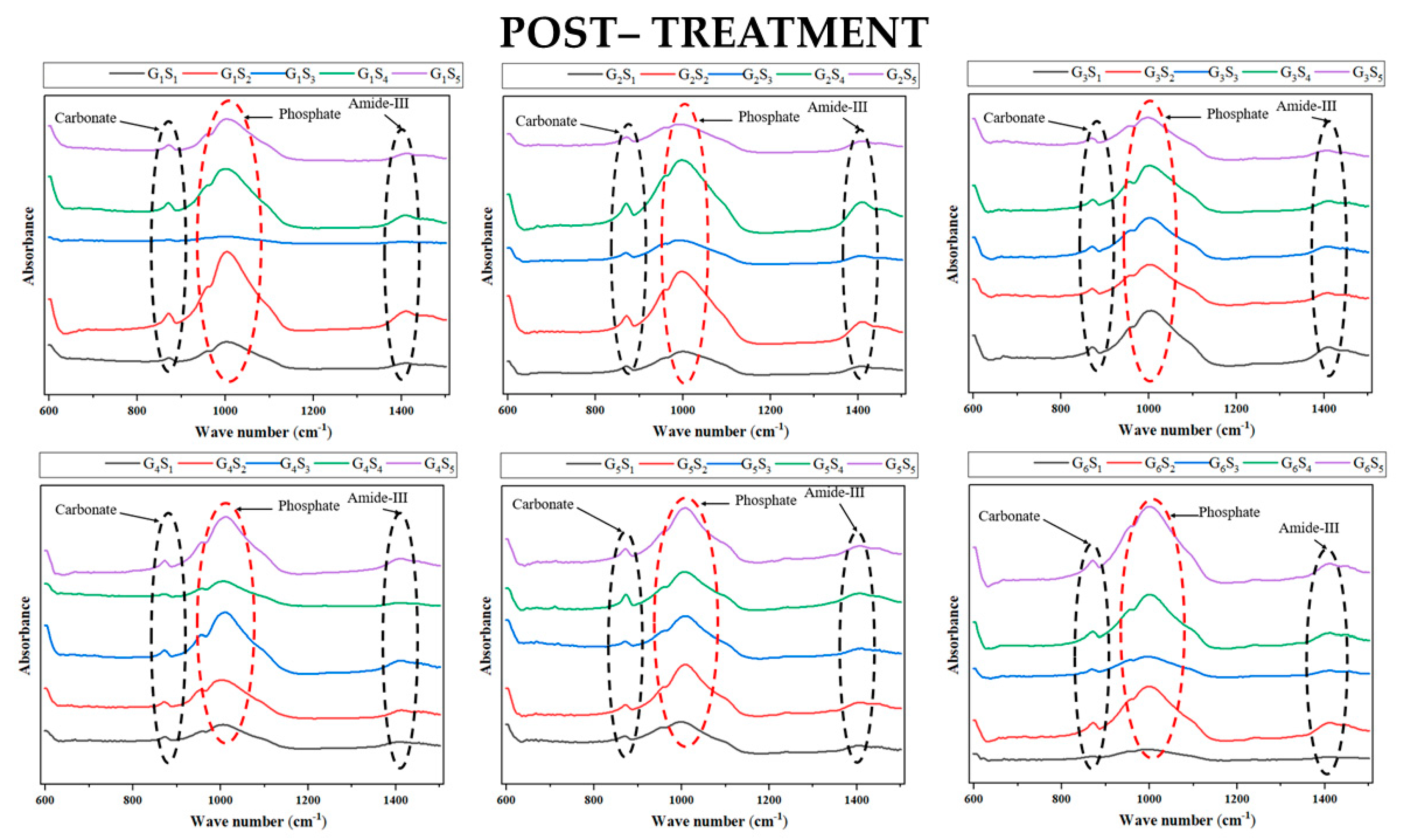
| Irrigant | Amide III/Phosphate | Carbonate/Phosphate | ||
|---|---|---|---|---|
| Pre | Post | Pre | Post | |
| Saline = control | 7.4 ± 0.6 A | 7.0 ± 1.3 A | 22.0 ± 1.9 a | 21.1 ± 2.2 a |
| 9% HEDP in saline | 7.9 ± 0.6 A | 8.0 ± 1.1 A | 23.1± 4.0 a | 21.7± 2.2 a |
| 2.5% NaOCl | 7.3 ± 0.7 A | 0.4 ± 0.2 B | 23.6 ± 1.0 a | 22.2 ± 1.3 a |
| 9% HEDP in 2.5% NaOCl | 7.6 ± 1.1 A | 6.1 ± 2.2 A | 25.2 ± 0.7 a | 21.1 ± 1.6 a |
| 5.25% NaOCl | 7.3 ± 0.8 A | 0.7 ± 0.5 B | 21.9 ± 1.7 a | 29.6 ± 3.0 b |
| 9% HEDP in 5.25% NaOCl | 7.5 ± 0.7 A | 2.2 ± 1.2 B | 22.0 ± 2.1 a | 18.2 ± 3.4 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ballal, N.V.; Roy, A.; Zehnder, M. Effect of Sodium Hypochlorite Concentration in Continuous Chelation on Dislodgement Resistance of an Epoxy Resin and Hydraulic Calcium Silicate Sealer. Polymers 2021, 13, 3482. https://doi.org/10.3390/polym13203482
Ballal NV, Roy A, Zehnder M. Effect of Sodium Hypochlorite Concentration in Continuous Chelation on Dislodgement Resistance of an Epoxy Resin and Hydraulic Calcium Silicate Sealer. Polymers. 2021; 13(20):3482. https://doi.org/10.3390/polym13203482
Chicago/Turabian StyleBallal, Nidambur Vasudev, Amal Roy, and Matthias Zehnder. 2021. "Effect of Sodium Hypochlorite Concentration in Continuous Chelation on Dislodgement Resistance of an Epoxy Resin and Hydraulic Calcium Silicate Sealer" Polymers 13, no. 20: 3482. https://doi.org/10.3390/polym13203482
APA StyleBallal, N. V., Roy, A., & Zehnder, M. (2021). Effect of Sodium Hypochlorite Concentration in Continuous Chelation on Dislodgement Resistance of an Epoxy Resin and Hydraulic Calcium Silicate Sealer. Polymers, 13(20), 3482. https://doi.org/10.3390/polym13203482