Bioactivity Comparison of Electrospun PCL Mats and Liver Extracellular Matrix as Scaffolds for HepG2 Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Production and Preparation of Scaffolds
2.2. Physical Characterization of Scaffolds
2.3. Cell Seeding and Cell Cultivation on Scaffolds
2.4. Cell Transfection
2.5. Statistical Analysis
3. Results
3.1. Physical Characterization of the Scaffolds
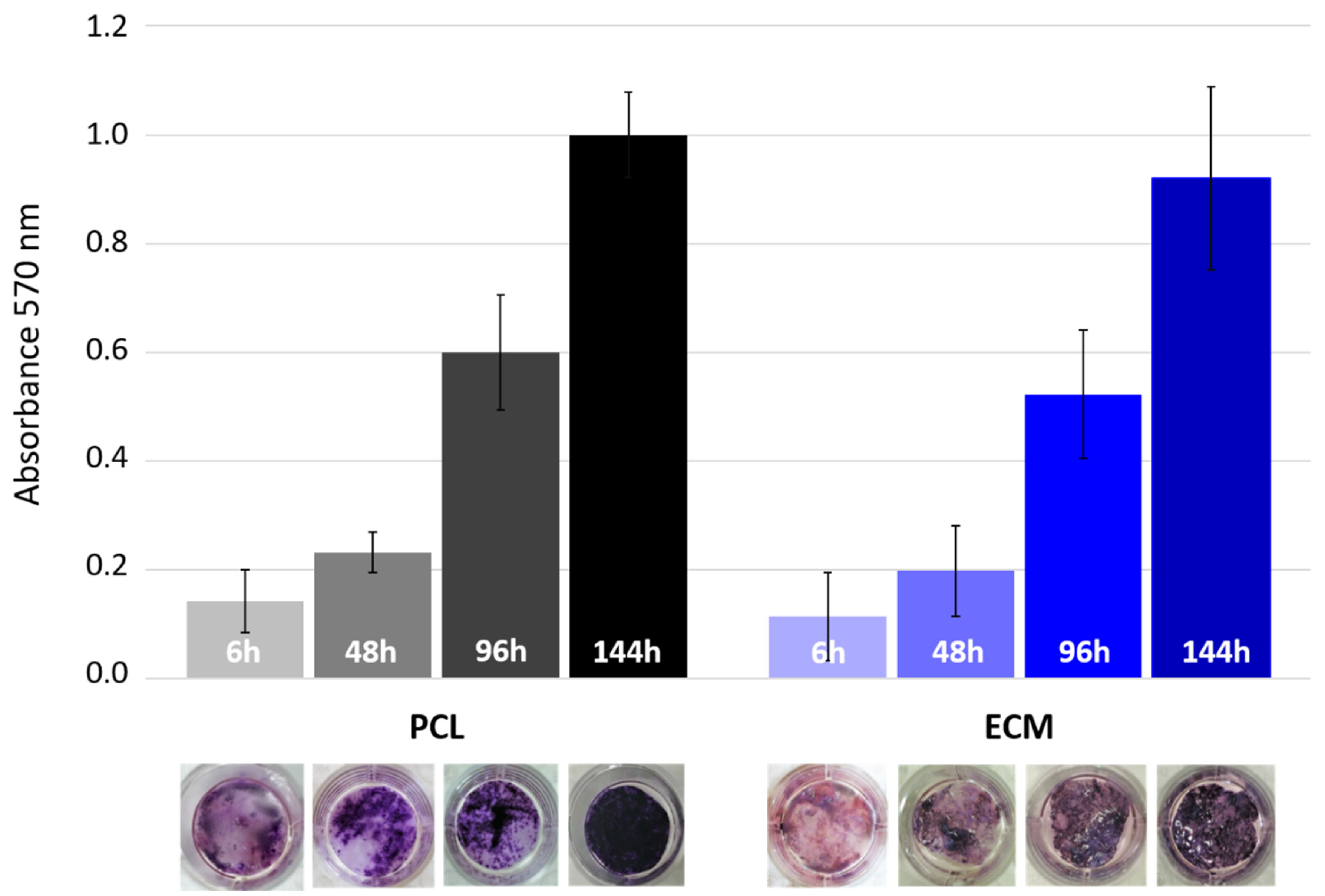
3.2. HepG2 Cell Seeding and Cultivation on the Scaffolds
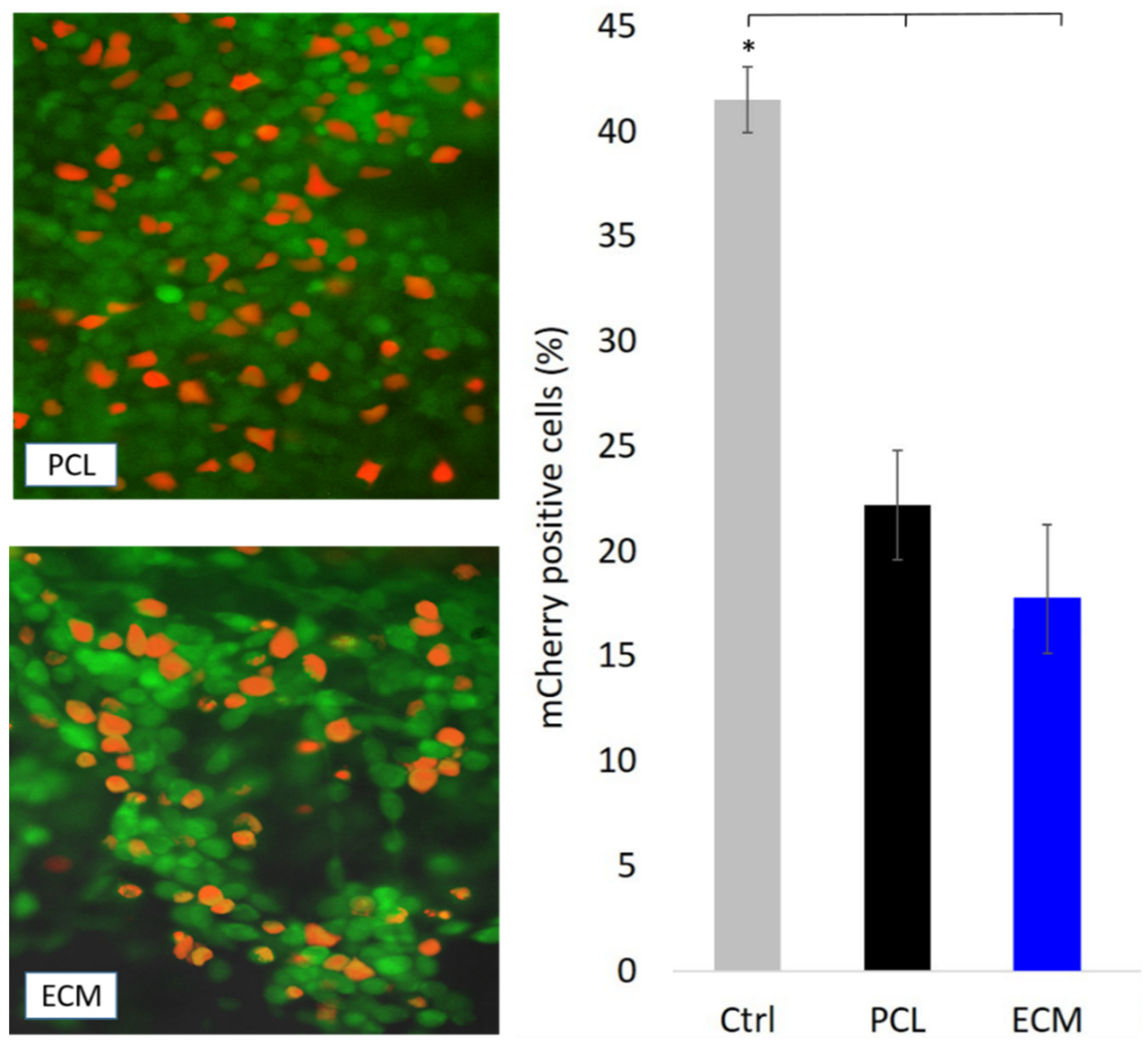
3.3. Transfection of HepG2 Cells Growing on the Scaffolds
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bramhill, J.; Ross, S.; Ross, G. Bioactive Nanocomposites for Tissue Repair and Regeneration: A review. Int. J. Environ. Res. Public Health 2017, 14, 66. [Google Scholar] [CrossRef]
- Patterson, J.; Martino, M.M.; Hubbell, J.A. Biomimetic Materials in Tissue Engineering. Mater. Today 2010, 13, 14–22. [Google Scholar] [CrossRef]
- Xie, Y.; Yang, S.T.; Kniss, D.A. Three-dimensional cell-scaffold constructs promote efficient gene transfection: Implications for cell-based gene therapy. Tissue Eng. 2001, 7, 585–598. [Google Scholar] [CrossRef]
- Siddiqui, N.; Asawa, S.; Birru, B.; Baadhe, R.; Rao, S. PCL-based Composite Scaffold Matrices for Tissue Engineering Applications. Molec. Biotechnol. 2018, 60, 506–532. [Google Scholar] [CrossRef] [PubMed]
- Malikmammadov, E.; Tanir, T.E.; Kiziltay, A.; Hasirci, V.; Hasirci, N. PCL and PCL-based Materials in Biomedical Applications. J. Biomater. Sci. Polym. Ed. 2018, 29, 863–893. [Google Scholar] [CrossRef]
- Tominac Trcin, M.; Zdraveva, E.; Dolenec, T.; Vrgoč Zimić, I.; Bujić Mihica, M.; Batarilo, I.; Dekaris, I.; Blažević, V.; Slivac, I.; Holjevac Grgurić, T.; et al. Poly(ε-caprolactone) Titanium Dioxide and Defuroxime Antimicrobial Scaffolds for Cultivation of Human Limbal Stem Cells. Polymers 2020, 12, 1758. [Google Scholar] [CrossRef]
- Wang, M.; Wang, K.; Yang, Y.; Liu, Y.; Yu, D.-G. Electrospun Environment Remediation Nanofibers Using Unspinnable Liquids as the Sheath Fluids: A Review. Polymers 2020, 12, 103. [Google Scholar] [CrossRef]
- Kang, S.; Hou, S.; Chen, X.; Yu, D.-G.; Wang, L.; Li, X.; Williams, R.G. Energy-Saving Electrospinning with a Concentric Teflon-Core Rod Spinneret to Create Medicated Nanofibers. Polymers 2020, 12, 2421. [Google Scholar] [CrossRef]
- Abdullah, M.F.; Nuge, T.; Andriyana, A.; Ang, B.C.; Muhamad, F. Core–Shell Fibers: Design, Roles, and Controllable Release Strategies in Tissue Engineering and Drug Delivery. Polymers 2019, 11, 2008. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, D.; Li, J.; Li, S.; Chen, Z.; Yu, D.-G.; Liu, Z.; Guo, J.Z. Electrospun Janus zein–PVP Nanofibers Provide a Two-stage Controlled Release of Poorly Water-soluble Drugs. Mater. Des. 2020, 196, 109075. [Google Scholar] [CrossRef]
- Ding, Y.; Dou, C.; Chang, S.; Xie, Z.; Yu, D.-G.; Liu, Y.; Shao, J. Core–Shell Eudragit S100 Nanofibers Prepared via Triaxial Electrospinning to Provide a Colon-Targeted Extended Drug Release. Polymers 2020, 12, 2034. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Wang, M.; Zhang, F.; Liu, Y.; Liu, X.; Yu, D.-G.; Shen, H. Sheath-separate-core Nanocomposites Fabricated Using a Trifluid Electrospinning. Mater. Des. 2020, 192, 108782. [Google Scholar] [CrossRef]
- Katsogiannis, K.A.G.; Vladisavljević, G.T.; Georgiadou, S. Porous Electrospun Polycaprolactone (PCL) Fibres by Phase Separation. Eur. Polym. J. 2015, 69, 284–295. [Google Scholar] [CrossRef]
- Coronado, R.E.; Somaraki-Cormier, M.; Natesan, S.; Christy, R.J.; Ong, J.L.; Halff, G.A. Decellularization and Solubilization of Porcine Liver for Use as a Substrate for Porcine Hepatocyte Culture: Method Optimization and Comparison. Cell Transplant. 2017, 26, 1840–1854. [Google Scholar] [CrossRef] [PubMed]
- Faulk, D.M.; Wildemann, J.D.; Badylak, S.F. Decellularization and Cell Seeding of Whole Liver Biologic Scaffolds Composed of Extracellular Matrix. J. Clin. Exp. Hepatol. 2015, 5, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Park, K.M.; Hussein, K.H.; Hong, S.H.; Ahn, C.; Yang, S.R.; Park, S.M.; Kweon, O.-K.; Kim, B.-M.; Woo, H.-M. Decellularized Liver Extracellular Matrix as Promising Tools for Transplantable Bioengineered Liver Promotes Hepatic Lineage Commitments of Induced Pluripotent Stem Cells. Tissue Eng. Part A 2016, 22, 449–460. [Google Scholar] [CrossRef]
- DeQuach, J.A.; Yuan, S.H.; Goldstein, L.S.B.; Christman, K.L. Decellularized Porcine Brain Matrix for Cell Culture and Tissue Engineering Scaffolds. Tissue Eng. A 2011, 17, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, V.; Maroufi, N.F.; Saghati, S.; Asadi, N.; Darabi, M.; Nazari, S.; Hosseinkhani, H.; Rahbarghazi, R. Current Progress in Hepatic Tissue Regeneration by Tissue Engineering. J. Transl. Med. 2019, 17, 383. [Google Scholar] [CrossRef]
- Li, Y.S.; Harn, H.; Hsieh, D.K.; Wen, T.C.; Subeq, Y.M.; Sun, L.Y.; Li, S.Z.; Chiou, T.W. Cells and Materials for Liver Tissue Engineering. Cell Transplant. 2013, 22, 685–700. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.C.; Choi, J.S.; Kim, B.S.; Kim, J.D.; Yoon, H.I.; Cho, Y.W. Decellularized Extracellular Matrix Derived from Porcine Adipose Tissue as a Xenogeneic Biomaterial for Tissue Engineering. Tissue Eng. Part C Methods 2012, 18, 866–876. [Google Scholar] [CrossRef] [PubMed]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory; Cold Spring Harbor: New York, NY, USA, 2001. [Google Scholar]
- Hanahan, D.; Jessee, J.; Bloom, F.R. Plasmid Transformation of Escherichia coli and Other Bacteria. In Bacterial Genetic Systems; Miller, J.H., Ed.; Academic Press: Cambridge, MA, USA, 1991; Volume 204, Chapter 4; p. 63. [Google Scholar]
- Jensen, G.; Morrill, C.; Huang, Y. Tissue Engineering, an Emerging Technique for Pharmaceutical Research. Acta Pharm. Sin. 2018, 8, 756–766. [Google Scholar] [CrossRef]
- Jiang, T.; Carbone, E.J.; Lo, K.W.H.; Laurencin, C.T. Electrospinning of polymer nanofibers for tissue regeneration. Prog. Polym. Sci. 2015, 46, 1–24. [Google Scholar] [CrossRef]
- Huang, Z.M.; Zhang, Y.Z.; Kotaki, M.; Ramakrishna, S. A Review on Polymer Nanofibers by Electrospinning and Their Applications in Nanocomposites. Compos. Sci. Technol. 2003, 6, 2223–2253. [Google Scholar] [CrossRef]
- Lang, R.; Stern, M.M.; Smith, L.; Liu, Y.; Bharadwaj, S.; Liu, G.; Baptista, P.M.; Bergman, C.R.; Soker, S.; Yoo, J.J.; et al. Three-Dimensional Culture of Hepatocytes on Porcine Liver Tissue-derived Extracellular Matrix. Biomaterials 2011, 32, 7042–7052. [Google Scholar] [CrossRef]
- Akhmanova, M.; Osidak, E.; Domogatsk, S.; Rodin, S.; Domogatskaya, A. Physical, Spatial, and Molecular Aspects of Extracellular Matrix of in vivo Niches and Artificial Scaffolds Relevant to Stem Cells Research. Stem Cells Int. 2015, 2015, 167025. [Google Scholar] [CrossRef]
- Innocenti, B.; Larrieu, J.C.; Lambert, P.; Pianigiani, S. Automatic Characterization of Soft Tissues Material Properties During Mechanical Tests. Muscles Ligaments Tendons J. 2018, 7, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Yang, J.; Huang, K.; Lee, Z.; Lee, X. A Comparison of Biomechanical Properties Between Human and Porcine Cornea. J. Biomech. 2001, 34, 533–537. [Google Scholar] [CrossRef]
- Chen, L.; Yan, C.; Zheng, Z. Functional Polymer Surfaces for Controlling Cell Behaviors. Mater. Today 2018, 21, 38–59. [Google Scholar] [CrossRef]
- Kurzyk, A.; Ostrowska, B.; Święszkowski, W.; Pojda, Z. Characterization and Optimization of the Seeding Process of Adipose Stem Cells on the Polycaprolactone Scaffold. Stem Cells Int. 2019, 2019, 1201927. [Google Scholar] [CrossRef] [PubMed]
- Rijal, G.; Li, W.A. Versatile 3D Tissue Matrix Scaffold System for Tumor Modeling and Drug Screening. Sci. Adv. 2017, 3, e1700764. [Google Scholar] [CrossRef] [PubMed]
- Stylianopoulos, T.; Diop-Frimpong, B.; Munn, L.L.; Jain, R.K. Diffusion anisotropy in collagen gels and tumors: The effect of fiber network orientation. Biophys. J. 2010, 99, 3119–3128. [Google Scholar] [CrossRef] [PubMed]
- Slivac, I.; Guay, D.; Mangion, M.; Champeil, J.; Gaillet, B. Non-viral Nucleic Acid Delivery Methods. Expert. Opin. Biol. Ther. 2017, 17, 105–111. [Google Scholar] [CrossRef]
- Bengali, Z.; Pannier, A.K.; Segura, T.; Anderson, B.C.; Jang, J.H.; Mustoe, T.A.; Shea, L.D. Gene Delivery through Cell Culture Substrate Adsorbed DNA Complexes. Biotechnol. Bioeng. 2005, 90, 290–302. [Google Scholar] [CrossRef] [PubMed]

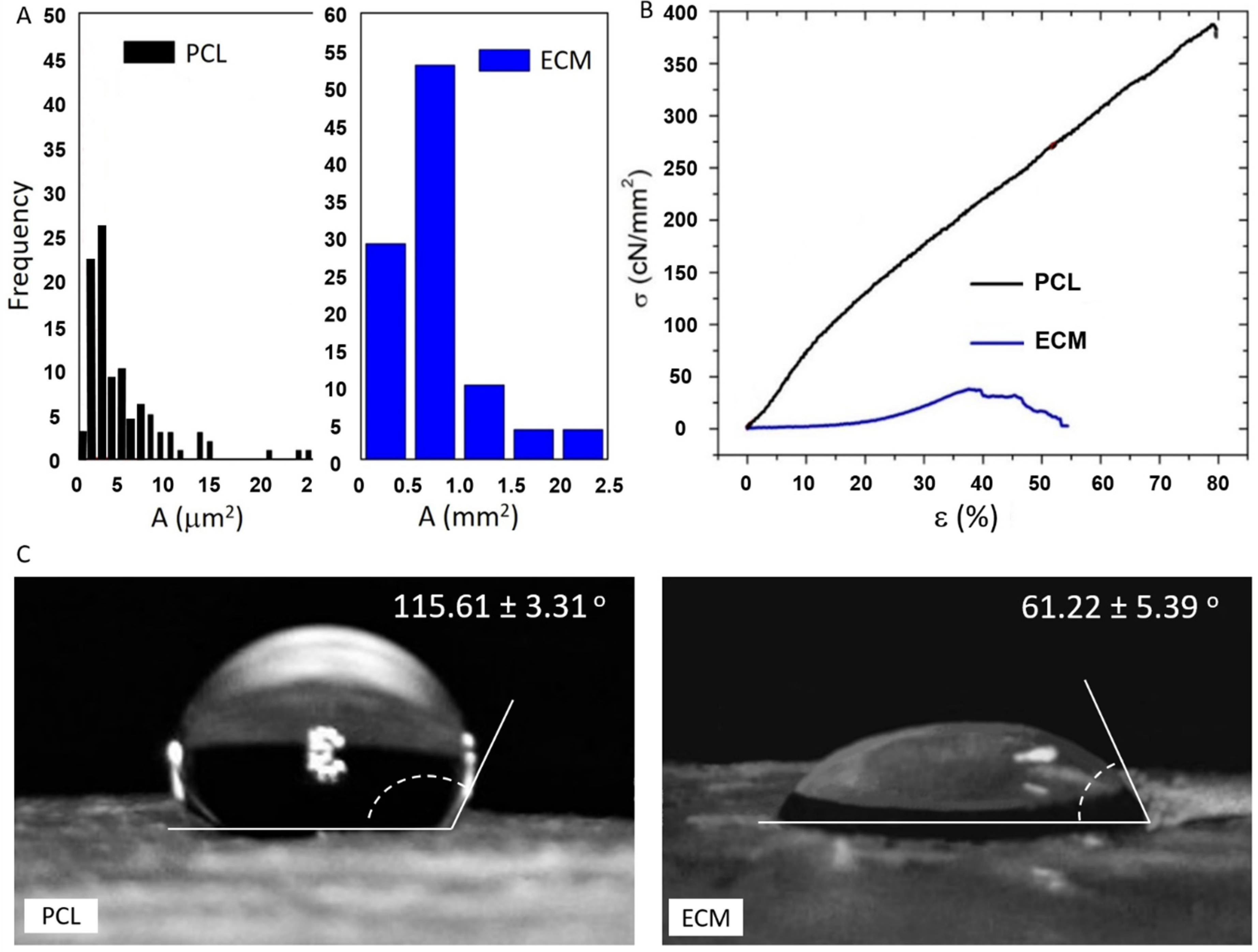
| Scaffold | m (g) | h (cm) | ρ (g/cm3) | dfibre (µm) | Apore (µm2) | p (%) | F (cN) | ε (%) | σ (cN/mm2) |
|---|---|---|---|---|---|---|---|---|---|
| PCL | 0.079 ± 0.034 | 0.064 ± 0.017 | 0.663 | 0.874 ± 0.402 | 6.126 ± 2.899 | 81.4 | 592.67 | 86.63 | 383.18 |
| ECM | 0.051 ± 0.006 | 0.093 ± 0.036 | 0.295 | - | 785.5 ± 27.6 | - | 640 | 66.28 | 32 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slivac, I.; Zdraveva, E.; Ivančić, F.; Žunar, B.; Holjevac Grgurić, T.; Gaurina Srček, V.; Svetec, I.-K.; Dolenec, T.; Bajsić, E.G.; Tominac Trcin, M.; et al. Bioactivity Comparison of Electrospun PCL Mats and Liver Extracellular Matrix as Scaffolds for HepG2 Cells. Polymers 2021, 13, 279. https://doi.org/10.3390/polym13020279
Slivac I, Zdraveva E, Ivančić F, Žunar B, Holjevac Grgurić T, Gaurina Srček V, Svetec I-K, Dolenec T, Bajsić EG, Tominac Trcin M, et al. Bioactivity Comparison of Electrospun PCL Mats and Liver Extracellular Matrix as Scaffolds for HepG2 Cells. Polymers. 2021; 13(2):279. https://doi.org/10.3390/polym13020279
Chicago/Turabian StyleSlivac, Igor, Emilija Zdraveva, Fran Ivančić, Bojan Žunar, Tamara Holjevac Grgurić, Višnja Gaurina Srček, Ivan-Krešimir Svetec, Tamara Dolenec, Emi Govorčin Bajsić, Mirna Tominac Trcin, and et al. 2021. "Bioactivity Comparison of Electrospun PCL Mats and Liver Extracellular Matrix as Scaffolds for HepG2 Cells" Polymers 13, no. 2: 279. https://doi.org/10.3390/polym13020279
APA StyleSlivac, I., Zdraveva, E., Ivančić, F., Žunar, B., Holjevac Grgurić, T., Gaurina Srček, V., Svetec, I.-K., Dolenec, T., Bajsić, E. G., Tominac Trcin, M., & Mijović, B. (2021). Bioactivity Comparison of Electrospun PCL Mats and Liver Extracellular Matrix as Scaffolds for HepG2 Cells. Polymers, 13(2), 279. https://doi.org/10.3390/polym13020279








