In Silico Characterization of the Interaction between the PBP2a “Decoy” Protein of Resistant Staphylococcus aureus and the Monomeric Units of Eudragit E-100 and Poly(Maleic Acid-alt-Octadecene) Polymers
Abstract
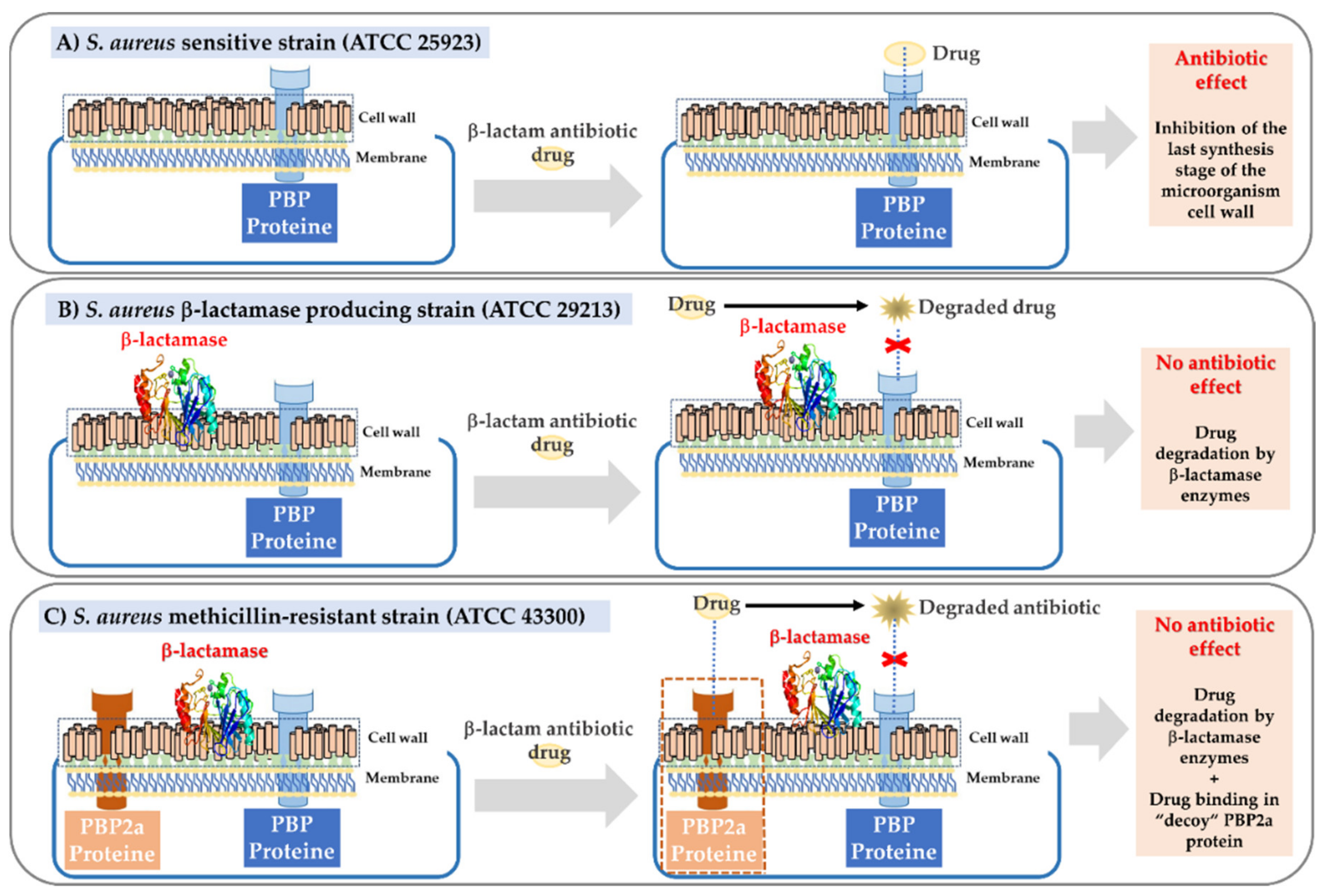
1. Introduction
2. Materials and Methods
2.1. Obtaining 3D Ligands
2.2. PBP2a Proteine
2.3. Docking Molecular
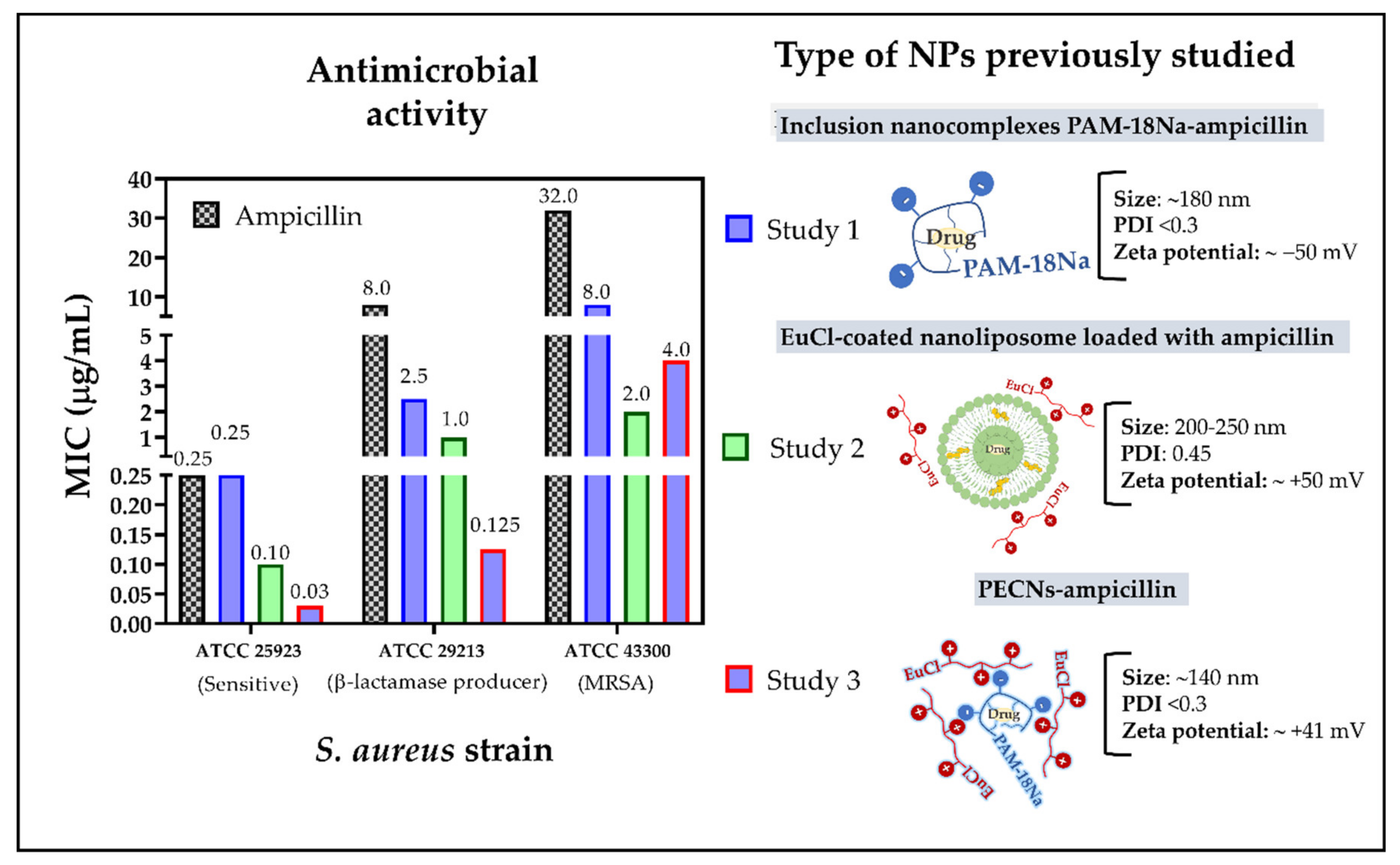
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carlier, M.; Stove, V.; Wallis, S.C.; De Waele, J.J.; Verstraete, A.G.; Lipman, J.; Roberts, J.A. Assays for therapeutic drug monitoring of β-lactam antibiotics: A structured review. Int. J. Antimicrob. Agents 2015, 46, 367–375. [Google Scholar] [CrossRef]
- Sergelidis, D.; Angelidis, A.S. Methicillin-resistant Staphylococcus aureus: A controversial food-borne pathogen. Lett. Appl. Microbiol. 2017, 64, 409–418. [Google Scholar] [CrossRef]
- Archer, G.L. Staphylococcus aureus: A well-armed pathogen. Clin. Infect. Dis. 1998, 26, 1179–1181. [Google Scholar] [CrossRef] [PubMed]
- Raynor, B.D. Penicillin and ampicillin. Prim. Care Update OB/GYNS 1997, 4, 147–152. [Google Scholar] [CrossRef]
- Nhung, N.T.; Chansiripornchai, N.; Carrique-Mas, J.J. Antimicrobial resistance in bacterial poultry pathogens: A review. Front. Vet. Sci. 2017, 4, 126. [Google Scholar] [CrossRef] [PubMed]
- Santajit, S.; Indrawattana, N. Mechanisms of Antimicrobial Resistance in ESKAPE Pathogens. Biomed Res. Int. 2016, 2016, 2475067. [Google Scholar] [CrossRef]
- Abushaheen, M.A.; Muzaheed; Fatani, A.J.; Alosaimi, M.; Mansy, W.; George, M.; Acharya, S.; Rathod, S.; Divakar, D.D.; Jhugroo, C.; et al. Antimicrobial resistance, mechanisms and its clinical significance. Disease-a-Month 2020, 66, 100971. [Google Scholar] [CrossRef]
- Aslam, A.; Gajdács, M.; Zin, C.S.; Rahman, N.S.A.; Ahmed, S.I.; Zafar, M.Z.; Jamshed, S. Evidence of the practice of self-medication with antibiotics among the lay public in low-and middle-income countries: A scoping review. Antibiotics 2020, 9, 597. [Google Scholar] [CrossRef]
- Fuda, C.; Suvorov, M.; Vakulenko, S.B.; Mobashery, S. The basis for resistance to β-lactam antibiotics by penicillin-binding protein 2a of methicillin-resistant Staphylococcus aureus. J. Biol. Chem. 2004, 279, 40802–40806. [Google Scholar] [CrossRef] [PubMed]
- Fishovitz, J.; Hermoso, J.A.; Chang, M.; Mobashery, S. Penicillin-binding protein 2a of methicillin-resistant Staphylococcus aureus. IUBMB Life 2014, 66, 572–577. [Google Scholar] [CrossRef]
- Lv, N.; Kong, Q.; Zhang, H.; Li, J. Discovery of novel Staphylococcus aureus penicillin binding protein 2a inhibitors by multistep virtual screening and biological evaluation. Bioorgan. Med. Chem. Lett. 2021, 41, 128001. [Google Scholar] [CrossRef] [PubMed]
- Helfand, M.S.; Totir, M.A.; Carey, M.P.; Hujer, A.M.; Bonomo, R.A.; Carey, P.R. Following the Reactions of Mechanism-Based Inhibitors with β-Lactamase by Raman Crystallography. Biochemistry 2003, 42, 13386–13392. [Google Scholar] [CrossRef]
- Abioye, A.O.; Tangyie-Chi, G.; Kola-Mustapha, A.T.; Ruparelia, K.; Beresford, K.; Arroo, R. Polymer-Drug Nanoconjugate—An Innovative Nanomedicine: Challenges and Recent Advancements in Rational Formulation Design for Effective Delivery of Poorly Soluble Drugs. Pharm. Nanotechnol. 2016, 4, 38–79. [Google Scholar] [CrossRef][Green Version]
- Erdoğar, N.; Akkın, S.; Bilensoy, E. Nanocapsules for Drug Delivery: An Updated Review of the Last Decade. Recent Pat. Drug Deliv. Formul. 2019, 12, 252–266. [Google Scholar] [CrossRef]
- El-Hammadi, M.M.; Arias, J.L. An update on liposomes in drug delivery: A patent review (2014–2018). Expert Opin. Ther. Pat. 2019, 29, 891–907. [Google Scholar] [CrossRef]
- Salamanca, C.H.; Yarce, C.J.; Roman, Y.; Davalos, A.F.; Rivera, G.R. Application of nanoparticle technology to reduce the anti-microbial resistance through B-lactam antibiotic polymer inclusion nano-complex. Pharmaceuticals 2018, 11, 19. [Google Scholar] [CrossRef]
- Arévalo, M.L.; Yarce, J.C.; Oñate-Garzón, J.; Salamanca, H.C. Decrease of Antimicrobial Resistance through Polyelectrolyte-Coated Nanoliposomes Loaded with β-Lactam Drug. Pharmaceuticals 2018, 12, 1. [Google Scholar] [CrossRef]
- Montero, N.; Alhajj, M.J.; Sierra, M.; Oñate-Garzon, J.; Yarce, C.J.; Salamanca, C.H. Development of polyelectrolyte complex nanoparticles-PECNs loaded with ampicillin by means of polyelectrolyte complexation and ultra-high pressure homogenization (UHPH). Polymers 2020, 12, 1168. [Google Scholar] [CrossRef]
- Tosco, P.; Stiefl, N.; Landrum, G. Bringing the MMFF force field to the RDKit: Implementation and validation. J. Cheminform. 2014, 6, 37–41. [Google Scholar] [CrossRef]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and testing of a general Amber force field. J. Comput. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef]
- DeLano, W. Pymol: An Open-Source Molecular Graphics Tool; Ballard, C., Howard-Eales, M., Eds.; Daresbury Laboratory: Warrington, UK, 2002. [Google Scholar]
- Kuhn, B.; Kollman, P.A. Binding of a diverse set of ligands to avidin and streptavidin: An accurate quantitative prediction of their relative affinities by a combination of molecular mechanics and continuum solvent models. J. Med. Chem. 2000, 43, 3786–3791. [Google Scholar] [CrossRef]
- Peacock, S.J.; Paterson, G.K. Mechanisms of methicillin resistance in Staphylococcus aureus. Annu. Rev. Biochem. 2015, 84, 577–601. [Google Scholar] [CrossRef]
- Salamanca, C.H.; Castillo, D.F.; Villada, J.D.; Rivera, G.R. Physicochemical characterization of in situ drug-polymer nanocomplex formed between zwitterionic drug and ionomeric material in aqueous solution. Mater. Sci. Eng. C 2017, 72, 405–414. [Google Scholar] [CrossRef]
- Reyes, I.; Palacio, M.M.; Yarce, C.J.; Oñate-Garzón, J.; Salamanca, C.H. Relationship between the ionization degree and the inter-polymeric aggregation of the poly(maleic acid-alt-octadecene) salts regarding time. Polymers 2020, 12, 1036. [Google Scholar] [CrossRef]
- Foxley, M.A.; Friedline, A.W.; Jensen, J.M.; Nimmo, S.L.; Scull, E.M.; King, J.B.; Strange, S.; Xiao, M.T.; Smith, B.E.; Thomas Iii, K.J.; et al. Efficacy of ampicillin against methicillin-resistant Staphylococcus aureus restored through synergy with branched poly(ethylenimine). J. Antibiot. 2016, 69, 871–878. [Google Scholar] [CrossRef]

| Molecule | Binding Energy (kcal/mol) | HI | HB | EI |
|---|---|---|---|---|
| Ampicillin | −6.4 | 5 | 1 | 1 |
| Eudragit (ionic form) | −4.6 | 3 | 2 | 1 |
| Eudragit (neutral form) | −4.5 | 2 | 4 | 2 |
| PAM18 (ionic form) | −5.2 | 12 | 0 | 0 |
| PAM18 (neutral form) | −4.7 | 7 | 3 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liscano, Y.; Amú, A.; González, A.; Oñate-Garzón, J.; Salamanca, C.H. In Silico Characterization of the Interaction between the PBP2a “Decoy” Protein of Resistant Staphylococcus aureus and the Monomeric Units of Eudragit E-100 and Poly(Maleic Acid-alt-Octadecene) Polymers. Polymers 2021, 13, 2320. https://doi.org/10.3390/polym13142320
Liscano Y, Amú A, González A, Oñate-Garzón J, Salamanca CH. In Silico Characterization of the Interaction between the PBP2a “Decoy” Protein of Resistant Staphylococcus aureus and the Monomeric Units of Eudragit E-100 and Poly(Maleic Acid-alt-Octadecene) Polymers. Polymers. 2021; 13(14):2320. https://doi.org/10.3390/polym13142320
Chicago/Turabian StyleLiscano, Yamil, Ana Amú, Astrid González, Jose Oñate-Garzón, and Constain H. Salamanca. 2021. "In Silico Characterization of the Interaction between the PBP2a “Decoy” Protein of Resistant Staphylococcus aureus and the Monomeric Units of Eudragit E-100 and Poly(Maleic Acid-alt-Octadecene) Polymers" Polymers 13, no. 14: 2320. https://doi.org/10.3390/polym13142320
APA StyleLiscano, Y., Amú, A., González, A., Oñate-Garzón, J., & Salamanca, C. H. (2021). In Silico Characterization of the Interaction between the PBP2a “Decoy” Protein of Resistant Staphylococcus aureus and the Monomeric Units of Eudragit E-100 and Poly(Maleic Acid-alt-Octadecene) Polymers. Polymers, 13(14), 2320. https://doi.org/10.3390/polym13142320








