Biobased Waterborne Polyurethane-Urea/SWCNT Nanocomposites for Hydrophobic and Electrically Conductive Textile Coatings
Abstract
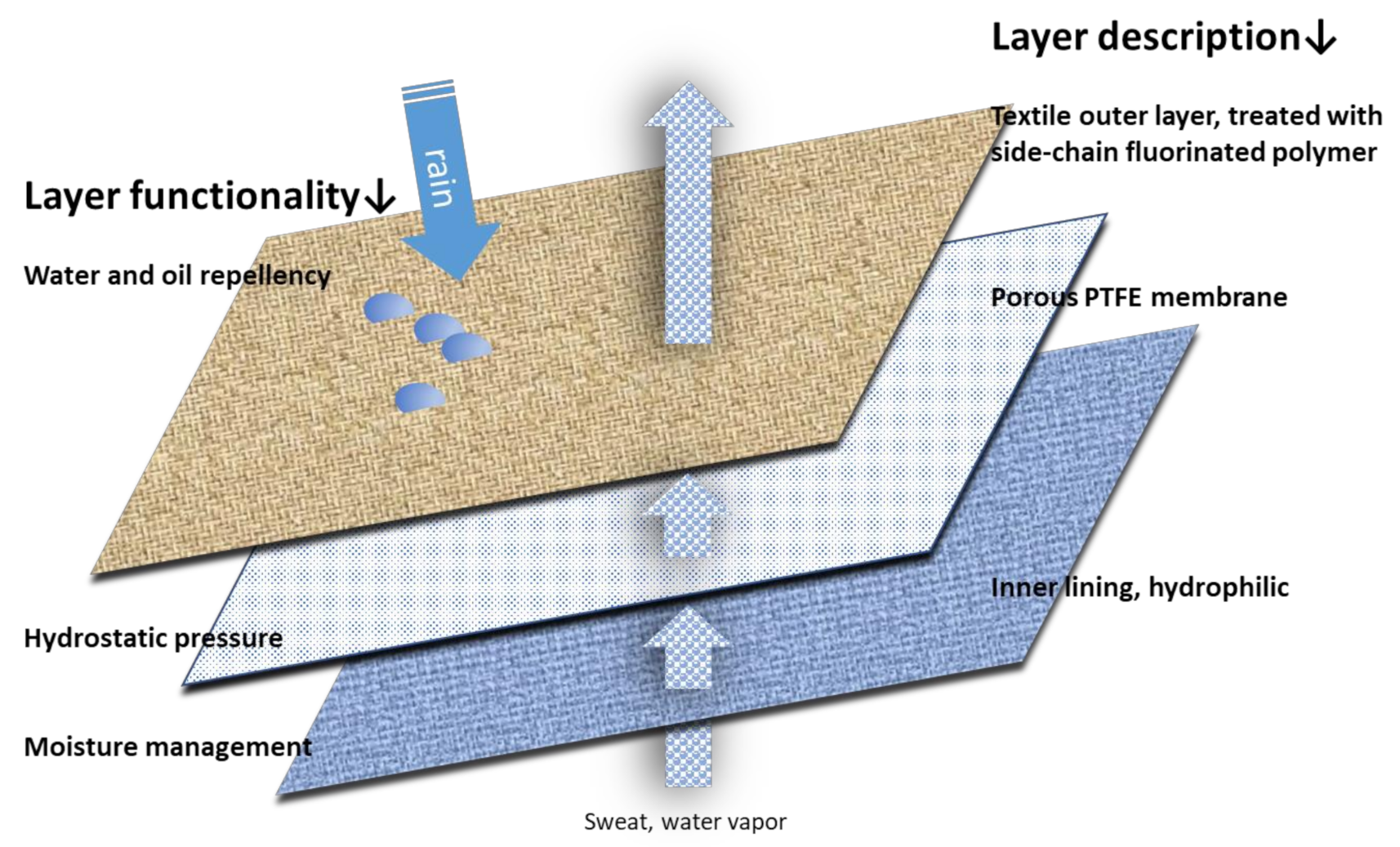
1. Introduction
2. Materials and Methods
2.1. Materials
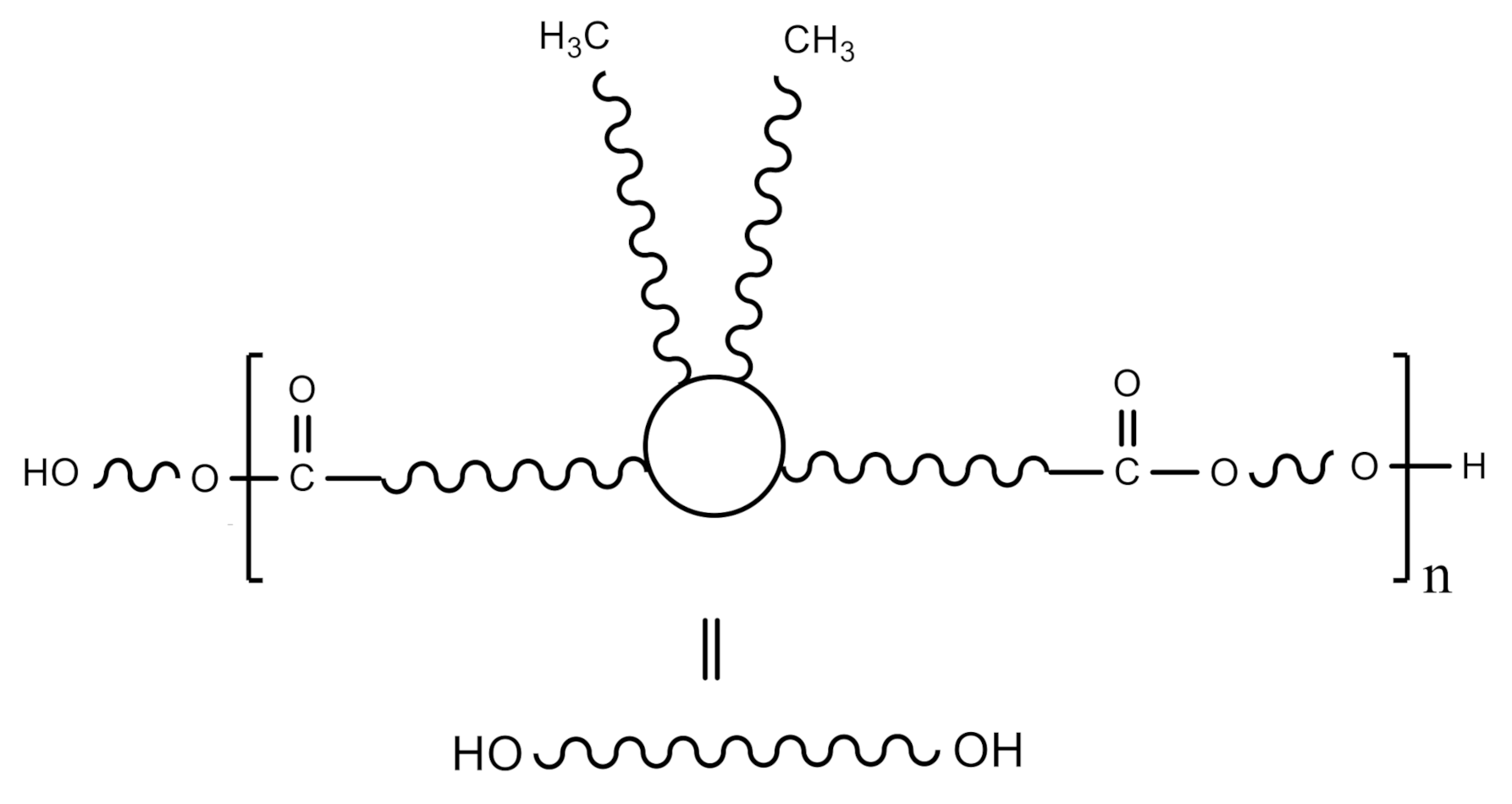
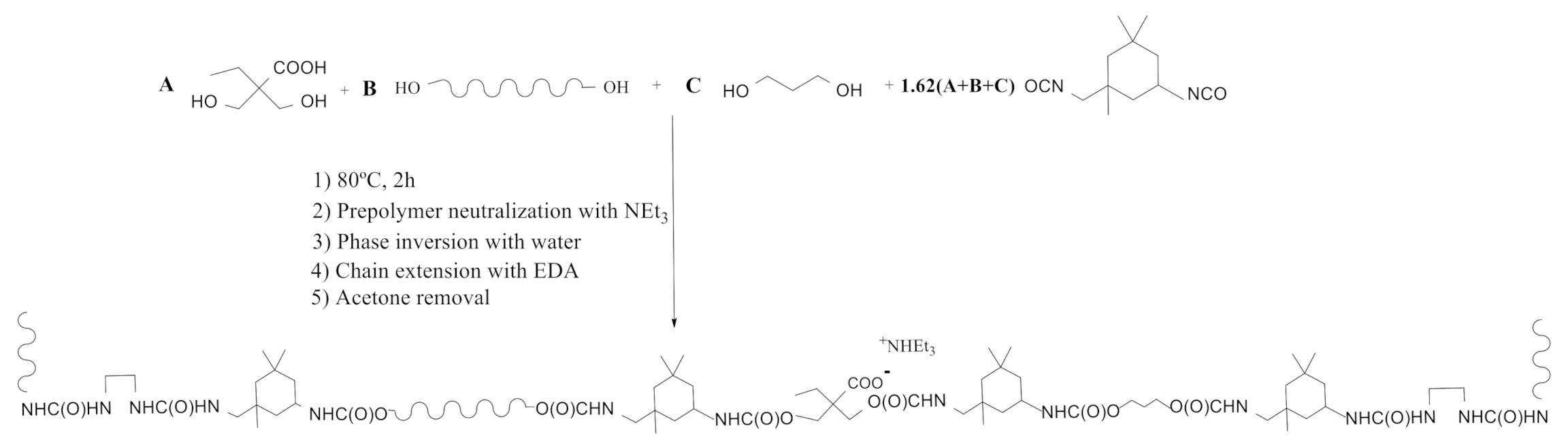
2.2. Synthesis of Waterborne Polyurethane-Urea Dispersions
2.3. Additivation of WPUD with SWCNT
2.4. Application of WPUD on Fabrics by Coating
2.5. Characterization Techniques
2.5.1. Characterization of Synthesized Polymers and Dispersions
2.5.2. Characterization of Coated Textiles
3. Results and Discussion
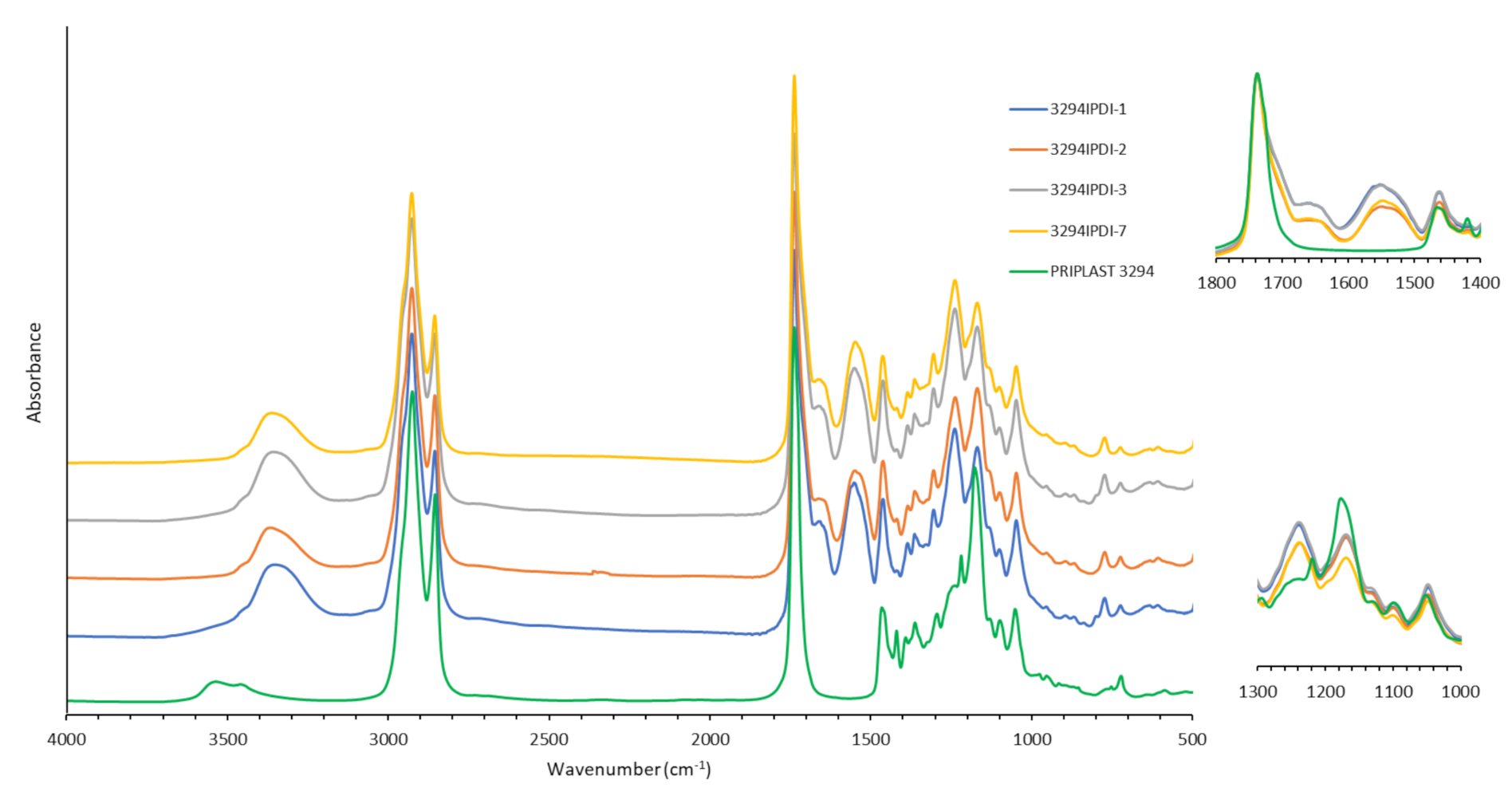
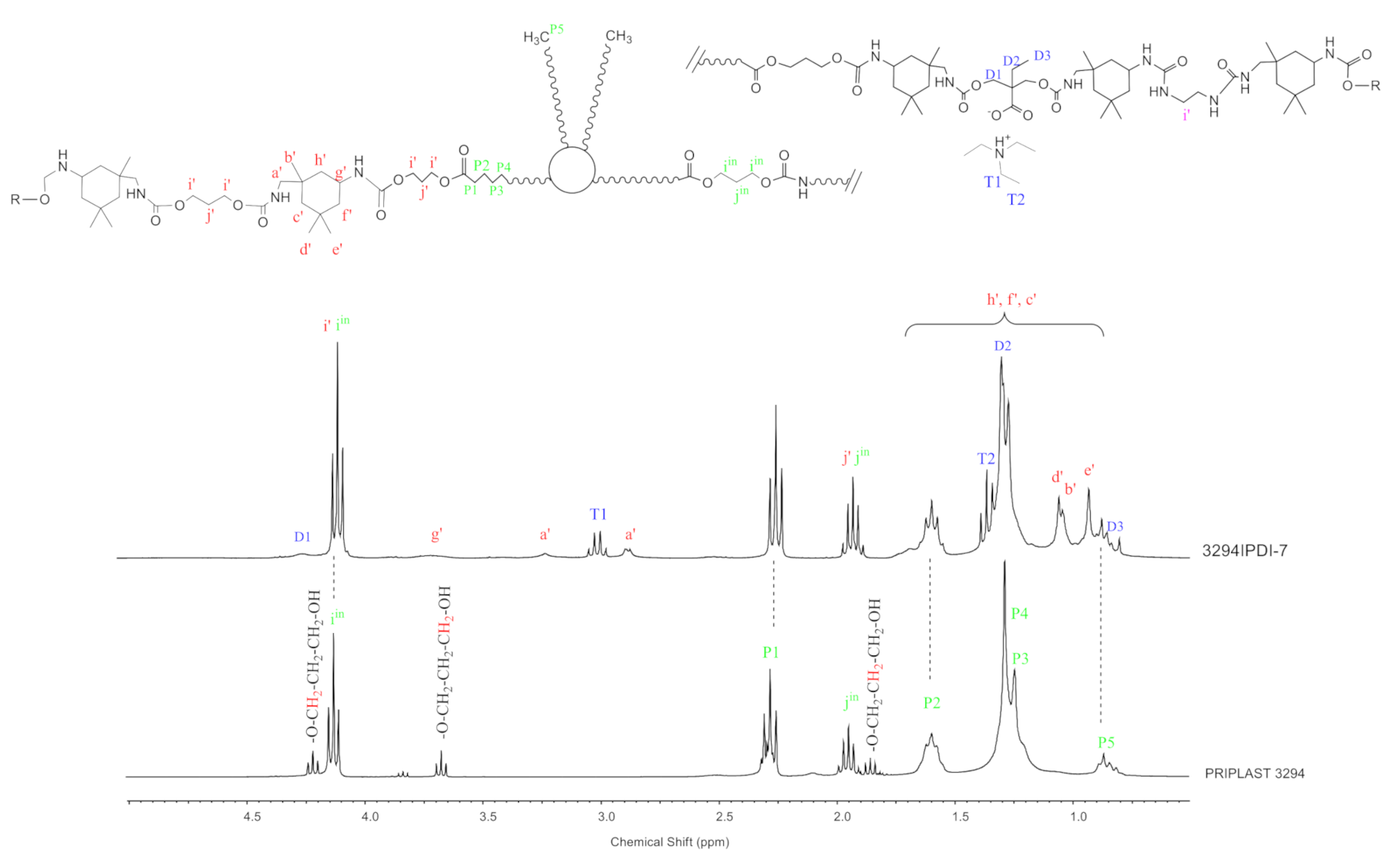
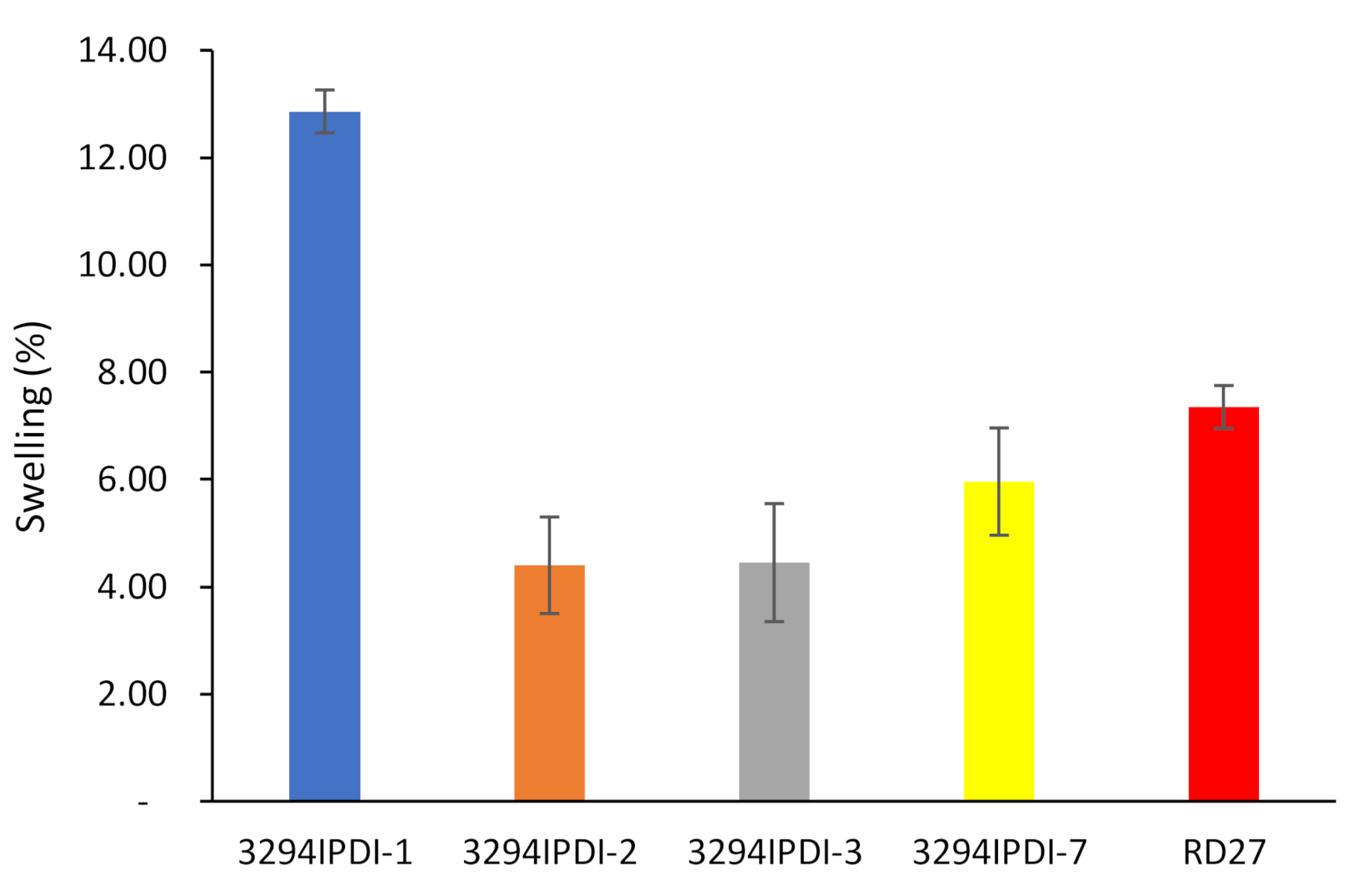
3.1. Synthesis and Characterization of WPUD
- IPDI CH2 in α-position to -NHC(O) group (a’), 3.24 ppm and 2.88 ppm corresponding to trans isomer (25% abundance) and cis isomer (75% abundance), respectively.
- IPDI CH in α-position to -NHC(O) group (g’), 3.73 ppm.
3.2. Additivation of WPUD with SWCNT
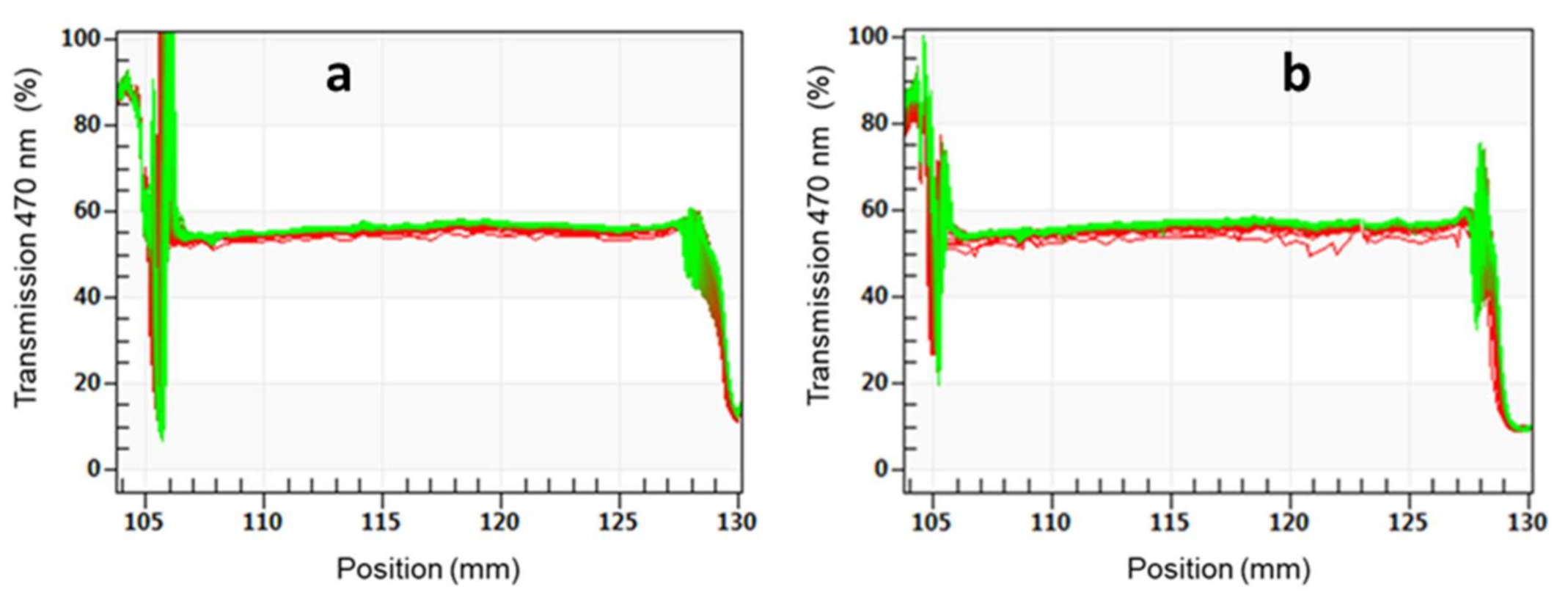
3.3. Characterization of Textiles Coated with WPUD
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Buck, R.C.; Franklin, J.; Berger, U.; Conder, J.M.; Cousins, I.T.; De Voogt, P.; Jensen, A.A.; Kannan, K.; Mabury, S.A.; van Leeuwen, S.P.J. Perfluoroalkyl and polyfluoroalkyl substances in the environment: Terminology, classification, and origins. Integr. Environ. Assess. Manag. 2011, 7, 513–541. [Google Scholar] [CrossRef] [PubMed]
- Giesy, J.P.; Kannan, K. Global distribution of perfluorooctane sulfonate in wildlife. Environ. Sci. Technol. 2001, 35, 1339–1342. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.W.; Mabury, S.A.; Solomon, K.R.; Muir, D.C.G. Progress toward understanding the bioaccumulation of perfluorinated alkyl acids. Environ. Toxicol. Chem. 2013, 32, 2421–2423. [Google Scholar] [CrossRef] [PubMed]
- Apelberg, B.J.; Witter, F.R.; Herbstman, J.B.; Calafat, A.M.; Halden, R.U.; Needham, L.L.; Goldman, L.R. Cord serum concentrations of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) in relation to weight and size at birth. Environ. Health Perspect. 2007, 115, 1670–1676. [Google Scholar] [CrossRef] [PubMed]
- Fei, C.; McLaughlin, J.K.; Tarone, R.E.; Olsen, J. Perfluorinated chemicals and fetal growth: A study within the Danish national birth cohort. Environ. Health Perspect. 2007, 115, 1677–1682. [Google Scholar] [CrossRef]
- Vierke, L.; Möller, A.; Klitzke, S. Transport of perfluoroalkyl acids in a water-saturated sediment column investigated under near-natural conditions. Environ. Pollut. 2014, 186, 7–13. [Google Scholar] [CrossRef]
- Luz, A.L.; Anderson, J.K.; Goodrum, P.; Durda, J. Perfluorohexanoic acid toxicity, part I: Development of a chronic human health toxicity value for use in risk assessment. Regul. Toxicol. Pharmacol. 2019, 103, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Klaunig, J.E.; Shinohara, M.; Iwai, H.; Chengelis, C.P.; Kirkpatrick, J.B.; Wang, Z.; Bruner, R.H. Evaluation of the chronic toxicity and carcinogenicity of perfluorohexanoic acid (PFHxA) in Sprague-Dawley Rats. Toxicol. Pathol. 2015, 43, 209–220. [Google Scholar] [CrossRef]
- Brusseau, M.L.; Anderson, R.H.; Guo, B. PFAS concentrations in soils: Background levels versus contaminated sites. Sci. Total Environ. 2020, 740, 140017. [Google Scholar] [CrossRef]
- Stoiber, T.; Evans, S.; Naidenko, O.V. Disposal of products and materials containing per- and polyfluoroalkyl substances (PFAS): A cyclical problem. Chemosphere 2020, 260, 127659. [Google Scholar] [CrossRef]
- Lohmann, R.; Cousins, I.T.; DeWitt, J.C.; Glüge, J.; Goldenman, G.; Herzke, D.; Lindstrom, A.B.; Miller, M.F.; Ng, C.A.; Patton, S.; et al. Are Fluoropolymers Really of Low Concern for Human and Environmental Health and Separate from Other PFAS? Environ. Sci. Technol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Geiser, M.; Schürch, S.; Gehr, P. Influence of surface chemistry and topography of particles on their immersion into the lung’s surface-lining layer. J. Appl. Physiol. 2003, 94, 1793–1801. [Google Scholar] [CrossRef]
- Groh, K.J.; Geueke, B.; Muncke, J. Food contact materials and gut health: Implications for toxicity assessment and relevance of high molecular weight migrants. Food Chem. Toxicol. 2017, 109, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Washington, J.W.; Jenkins, T.M.; Rankin, K.; Naile, J.E. Decades-scale degradation of commercial, side-chain, fluorotelomer-based polymers in soils and water. Environ. Sci. Technol. 2015, 49, 915–923. [Google Scholar] [CrossRef] [PubMed]
- Cobbing, M.; Campione, C.; Kopp, M. PFC Revolution in the Outdoor Sector; Greenpeace: Zürich, Switzerland, 2017. [Google Scholar]
- Zahid, M.; Mazzon, G.; Athanassiou, A.; Bayer, I.S. Environmentally benign non-wettable textile treatments: A review of recent state-of-the-art. Adv. Colloid Interface Sci. 2019, 270, 216–250. [Google Scholar] [CrossRef] [PubMed]
- Lassen, C.; Jensen, A.A.; Warming, M. Alternatives to Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) in Textiles; The Danish Environmental Protection Agency: Copenhagen, Danish, 2015. [Google Scholar]
- Lacruz, A.; Salvador, M.; Blanco, M.; Vidal, K.; Martínez de Ilarduya, A. Development of fluorine-free waterborne textile finishing agents for anti-stain and solvent-water separation based on low surface energy (co)polymers. Prog. Org. Coat. 2021, 150. [Google Scholar] [CrossRef]
- Parsons, G.N.; Atanasov, S.E.; Dandley, E.C.; Devine, C.K.; Gong, B.; Jur, J.S.; Lee, K.; Oldham, C.J.; Peng, Q.; Spagnola, J.C.; et al. Mechanisms and reactions during atomic layer deposition on polymers. Coord. Chem. Rev. 2013, 257, 3323–3331. [Google Scholar] [CrossRef]
- Iwai, Y.; Sameshima, S.; Yonezawa, S.; Katayama, S. Fabrication of conductive cotton by electroless plating method with supercritical carbon dioxide. J. Supercrit. Fluids 2015, 100, 46–51. [Google Scholar] [CrossRef]
- Jiang, S.Q.; Newton, E.; Yuen, C.W.M.; Kan, C.W. Chemical Silver Plating on Cotton and Polyester Fabrics and its Application on Fabric Design. Text. Res. J. 2006, 76, 57–65. [Google Scholar] [CrossRef]
- Trovato, V.; Teblum, E.; Kostikov, Y.; Pedrana, A.; Re, V.; Nessim, G.D.; Rosace, G. Sol-gel approach to incorporate millimeter-long carbon nanotubes into fabrics for the development of electrical-conductive textiles. Mater. Chem. Phys. 2020, 240, 122218. [Google Scholar] [CrossRef]
- Etxaniz, I.; Llorente, O.; Aizpurua, J.; Martín, L.; González, A.; Irusta, L. Dispersion Characteristics and Curing Behaviour of Waterborne UV Crosslinkable Polyurethanes Based on Renewable Dimer Fatty Acid Polyesters. J. Polym. Environ. 2019, 27, 189–197. [Google Scholar] [CrossRef]
- Poussard, L.; Lazko, J.; Mariage, J.; Raquez, J.M.; Dubois, P. Biobased waterborne polyurethanes for coating applications: How fully biobased polyols may improve the coating properties. Prog. Org. Coat. 2016, 97, 175–183. [Google Scholar] [CrossRef]
- Cavallo, D.; Gardella, L.; Soda, O.; Sparnacci, K.; Monticelli, O. Fully bio-renewable multiblocks copolymers of poly(lactide) and commercial fatty acid-based polyesters polyols: Synthesis and characterization. Eur. Polym. J. 2016, 81, 247–256. [Google Scholar] [CrossRef]
- Wu, S.; Ladani, R.B.; Zhang, J.; Bafekrpour, E.; Ghorbani, K.; Mouritz, A.P.; Kinloch, A.J.; Wang, C.H. Aligning multilayer graphene flakes with an external electric field to improve multifunctional properties of epoxy nanocomposites. Carbon 2015, 94, 607–618. [Google Scholar] [CrossRef]
- Wang, Q.; Dai, J.; Li, W.; Wei, Z.; Jiang, J. The effects of CNT alignment on electrical conductivity and mechanical properties of SWNT/epoxy nanocomposites. Compos. Sci. Technol. 2008, 68, 1644–1648. [Google Scholar] [CrossRef]
- Monti, M.; Natali, M.; Torre, L.; Kenny, J.M. The alignment of single walled carbon nanotubes in an epoxy resin by applying a DC electric field. Carbon 2012, 50, 2453–2464. [Google Scholar] [CrossRef]
- Yan, H.; Tang, Y.; Long, W.; Li, Y. Enhanced thermal conductivity in polymer composites with aligned graphene nanosheets. J. Mater. Sci. 2014, 49, 5256–5264. [Google Scholar] [CrossRef]
- Lerche, D.; Sobisch, T. Direct and accelerated characterization of formulation stability. J. Dispers. Sci. Technol. 2011, 32, 1799–1811. [Google Scholar] [CrossRef]
- Poussard, L.; Mecheri, A.; Mariage, J.; Barakat, I.; Bonnaud, L.; Raquez, J.M.; Dubois, P. Synthesis of oligo(butylene succinate)-based polyurethanes: Influence of the chemical structure on thermal and mechanical properties. In Proceedings of the Journal of Renewable Materials; Scrivener Publishing LLC: Beverly, MA, USA, 2014; Volume 2, pp. 13–22. [Google Scholar]
- Bueno-Ferrer, C.; Hablot, E.; Perrin-Sarazin, F.; Garrigós, M.C.; Jiménez, A.; Averous, L. Structure and Morphology of New Bio-Based Thermoplastic Polyurethanes Obtained From Dimeric Fatty Acids. Macromol. Mater. Eng. 2012, 297, 777–784. [Google Scholar] [CrossRef]
- Lomölder, R.; Plogmann, F.; Speier, P. Selectivity of isophorone diisocyanate in the urethane reaction influence of temperature, catalysis, and reaction partners. J. Coat. Technol. 1997, 69, 51–57. [Google Scholar] [CrossRef]
- Mukherjea, R.N.; Saha, K.K.; Sanyal, S.K. Plasticizing effect of acetylated castor oil on castor oil-based, moisture-cured polyurethane film. J. Am. Oil Chem. Soc. 1978, 55, 653–656. [Google Scholar] [CrossRef]
- Xu, J.; Li, T.; Zhao, W.; Li, P.; Wu, Y. Synthesis and characterization of waterborne polyurethane emulsions based on poly(butylene itaconate) ester. Des. MonoMers Polym. 2016, 19, 309–318. [Google Scholar] [CrossRef]
- Sun, Y.; Zhao, X.; Liu, R.; Chen, G.; Zhou, X. Synthesis and characterization of fluorinated polyacrylate as water and oil repellent and soil release finishing agent for polyester fabric. Prog. Org. Coat. 2018, 123, 306–313. [Google Scholar] [CrossRef]
- Li, D.; Müller, M.B.; Gilje, S.; Kaner, R.B.; Wallace, G.G. Processable aqueous dispersions of graphene nanosheets. Nat. Nanotechnol. 2008, 3, 101–105. [Google Scholar] [CrossRef]
- Sheng, L.; Zhang, X.; Ge, Z.; Liang, Z.; Liu, X.; Chai, C.; Luo, Y. Preparation and properties of waterborne polyurethane modified by stearyl acrylate for water repellents. J. Coat. Technol. Res. 2018, 15, 1283–1292. [Google Scholar] [CrossRef]
- Yuan, F.; Xu, D.; Qi, X.; Zhao, J.; Gao, Y. Impact of High Hydrostatic Pressure on the Emulsifying Properties of Whey Protein Isolate-Chitosan Mixtures. Food Bioprocess Technol. 2013, 6, 1024–1031. [Google Scholar] [CrossRef]
- Gao, L.; McCarthy, T.J. Contact angle hysteresis explained. Langmuir 2006, 22, 6234–6237. [Google Scholar] [CrossRef]
- Baxter, S. Wetting and contact-angle hysteresis. Nature 1950, 165, 198. [Google Scholar] [CrossRef]
- Zhang, R.; Deng, H.; Valenca, R.; Jin, J.; Fu, Q.; Bilotti, E.; Peijs, T. Carbon nanotube polymer coatings for textile yarns with good strain sensing capability. Sens. Actuators A Phys. 2012, 179, 83–91. [Google Scholar] [CrossRef]
- Narkis, M.; Lidor, G.; Vaxman, A.; Zuri, L. New injection moldable electrostatic dissipative(ESD) composites based on very low carbon black loadings. J. Electrostat. 1999, 47, 201–214. [Google Scholar] [CrossRef]
- Dai, M.; Zhai, Y.; Zhang, Y. A green approach to preparing hydrophobic, electrically conductive textiles based on waterborne polyurethane for electromagnetic interference shielding with low reflectivity. Chem. Eng. J. 2020. [Google Scholar] [CrossRef]
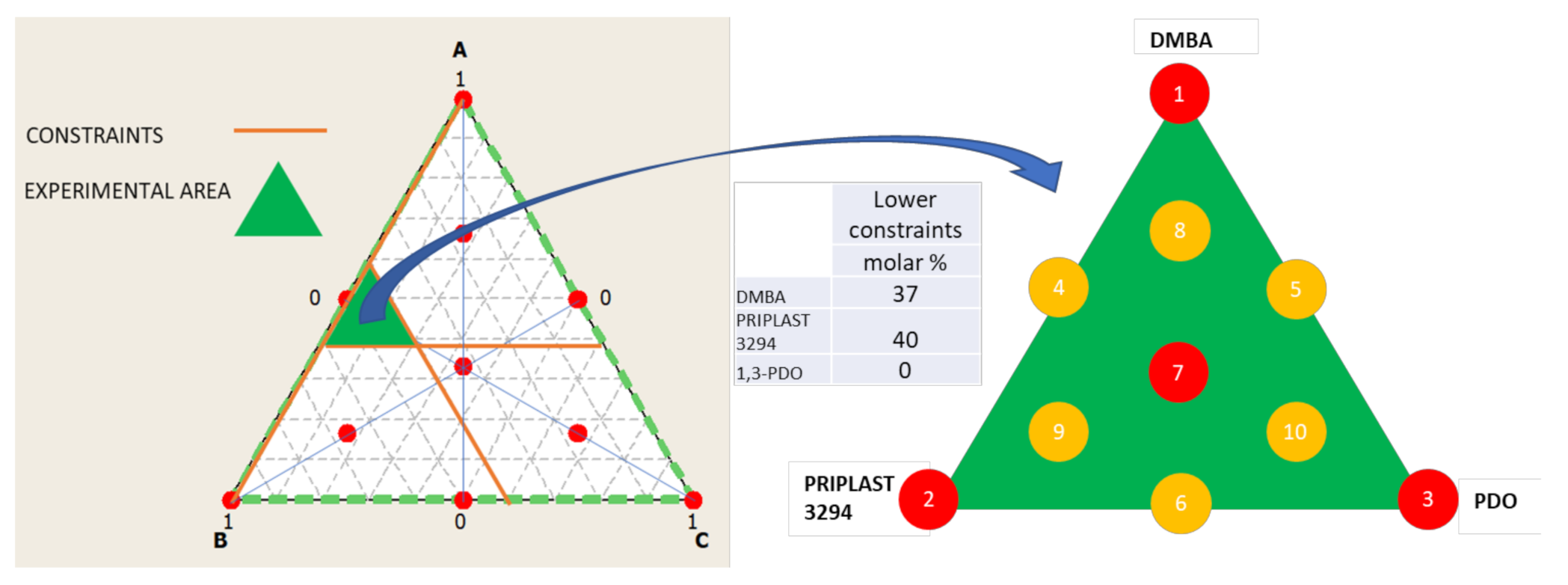


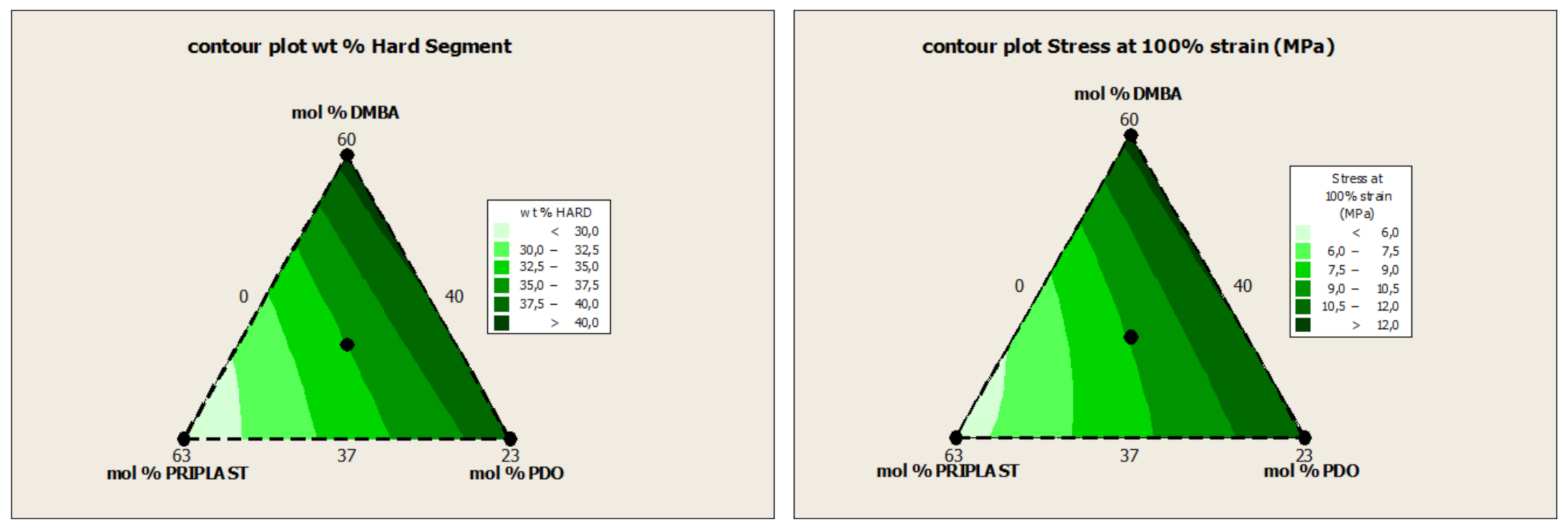
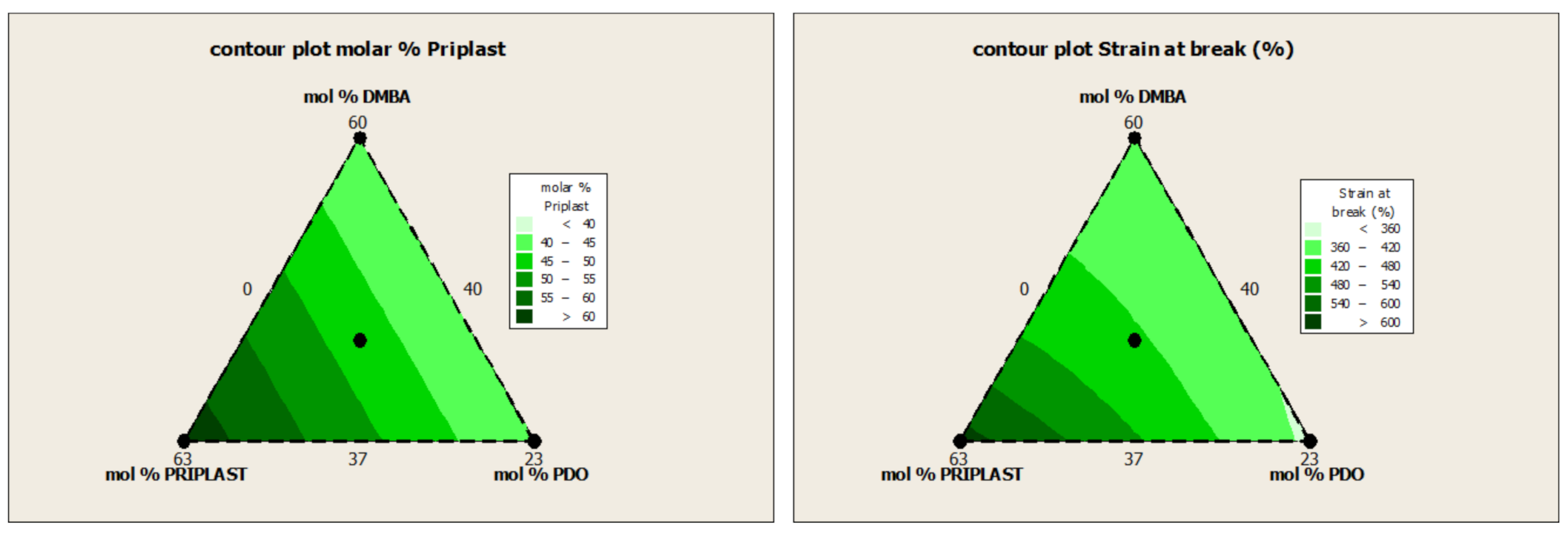
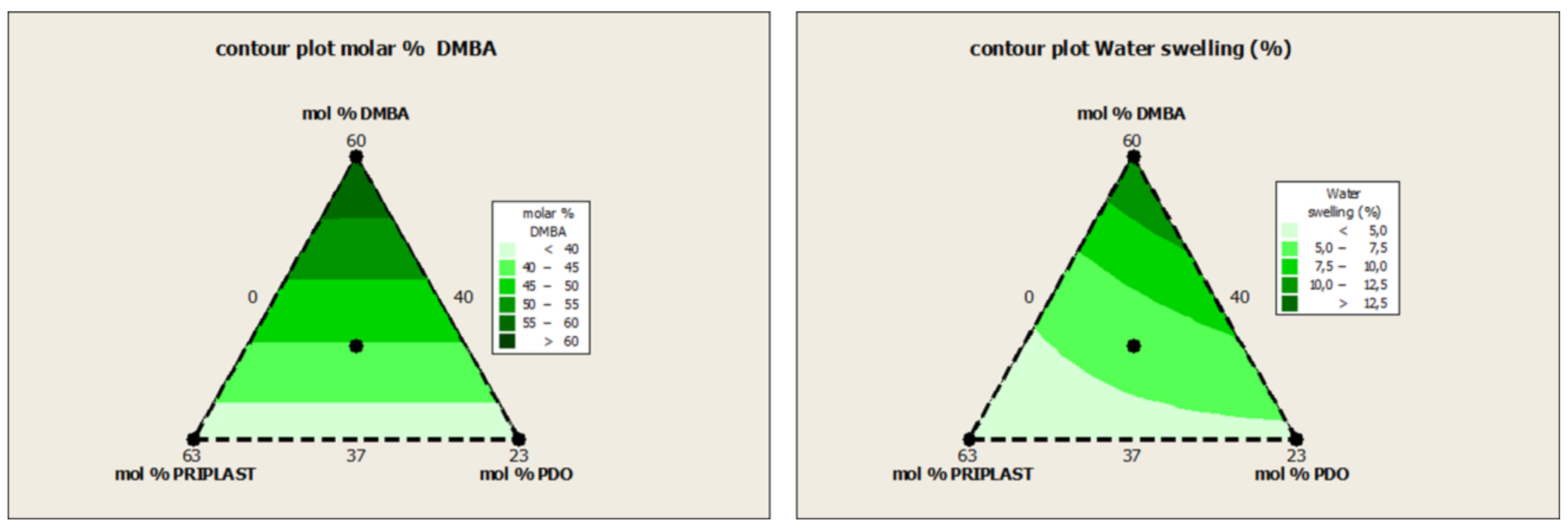




| Experiment | Matrix | Molar % | Hard Segment (wt. %) | Bio-Based Content (wt. %) | ||||
|---|---|---|---|---|---|---|---|---|
| DMBA | Priplast 3294 | 1,3-PDO | DMBA | Priplast 3294 | 1,3-PDO | |||
| 3294IPDI-1 | 1 | 0 | 0 | 60.00 | 40.00 | 0.00 | 40.6 | 59.4 |
| 3294IPDI-2 | 0 | 1 | 0 | 37.00 | 63.00 | 0.00 | 28.0 | 72.0 |
| 3294IPDI-3 | 0 | 0 | 1 | 37.00 | 40.00 | 23.00 | 38.8 | 62.6 |
| 4 | 1/2 | 1/2 | 0 | 48.50 | 51.50 | 0.00 | 33.4 | 66.6 |
| 5 | 1/2 | 0 | 1/2 | 48.50 | 40.00 | 11.50 | 39.7 | 61.0 |
| 6 | 0 | 1/2 | 1/2 | 37.00 | 51.50 | 11.50 | 32.6 | 68.0 |
| 3294IPDI-7 | 1/3 | 1/3 | 1/3 | 44.67 | 47.67 | 7.67 | 35.0 | 65.4 |
| 8 | 2/3 | 1/6 | 1/6 | 52.33 | 43.83 | 3.83 | 37.7 | 62.5 |
| 9 | 1/6 | 2/3 | 1/6 | 40.83 | 55.33 | 3.83 | 31.2 | 69.0 |
| 10 | 1/6 | 1/6 | 2/3 | 40.83 | 43.83 | 15.33 | 36.8 | 64.1 |
| Composition | Printing Paste Formulation (g) |
|---|---|
| WPUD | 85 |
| water | 15 |
| Defoamer PR | 1 |
| Complex DG | 2 |
| DMEA | 1 |
| Thickener L-120 | Added drop by drop under high shear until viscosity of 18,000 ± 50 cPs is reached (Brookfield RV 6/30) |
| Dry polyurethane-urea content | 30 ± 1% |
| Property | Standard |
|---|---|
| Determination of resistance to water penetration-Hydrostatic pressure test (water column) | EN 20811:1992 |
| Fabric stiffness | ČSN 80 0858 |
| Determination of the permeability of fabrics to air | ISO 9237:1995 |
| Electrostatic Properties | Standard | Minimum Compliance Values |
|---|---|---|
| Surface resistivity & Specific surface resistivity | EN 1149-1 | Surface resistance ≤ 2.5 × 109 |
| Vertical resistance | EN 1149-2 | Electrical resistance > 105 |
| Charge decay (inductive charge) | EN 1149-3, method 2 | t50 < 4 s or S > 0.2 values |
| Electrostatic properties. Performance requirements and material design. | EN 1149-5 | t50 < 4 s or S > 0.2 values; or surface resistance is ≤ 2.5·109 |
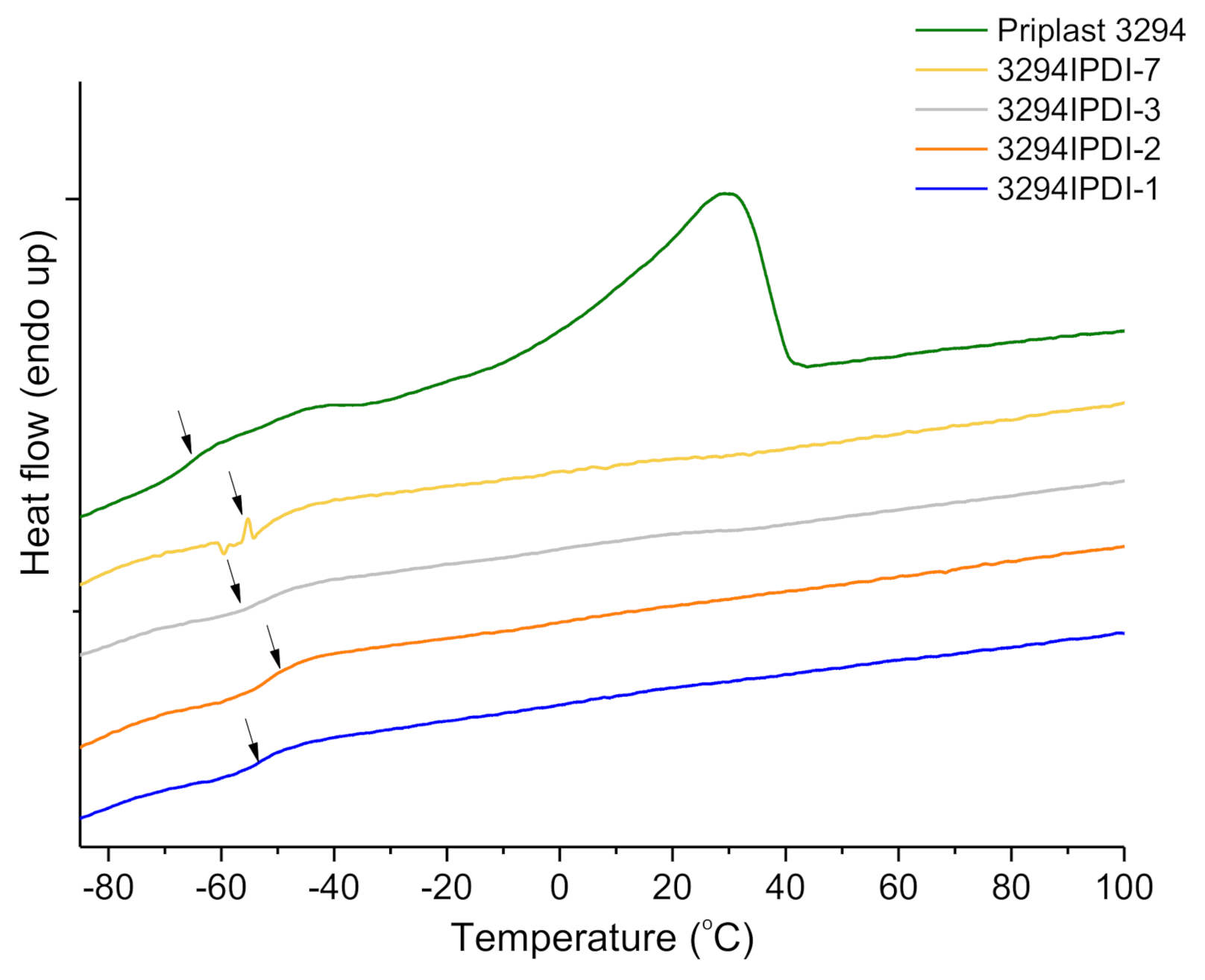
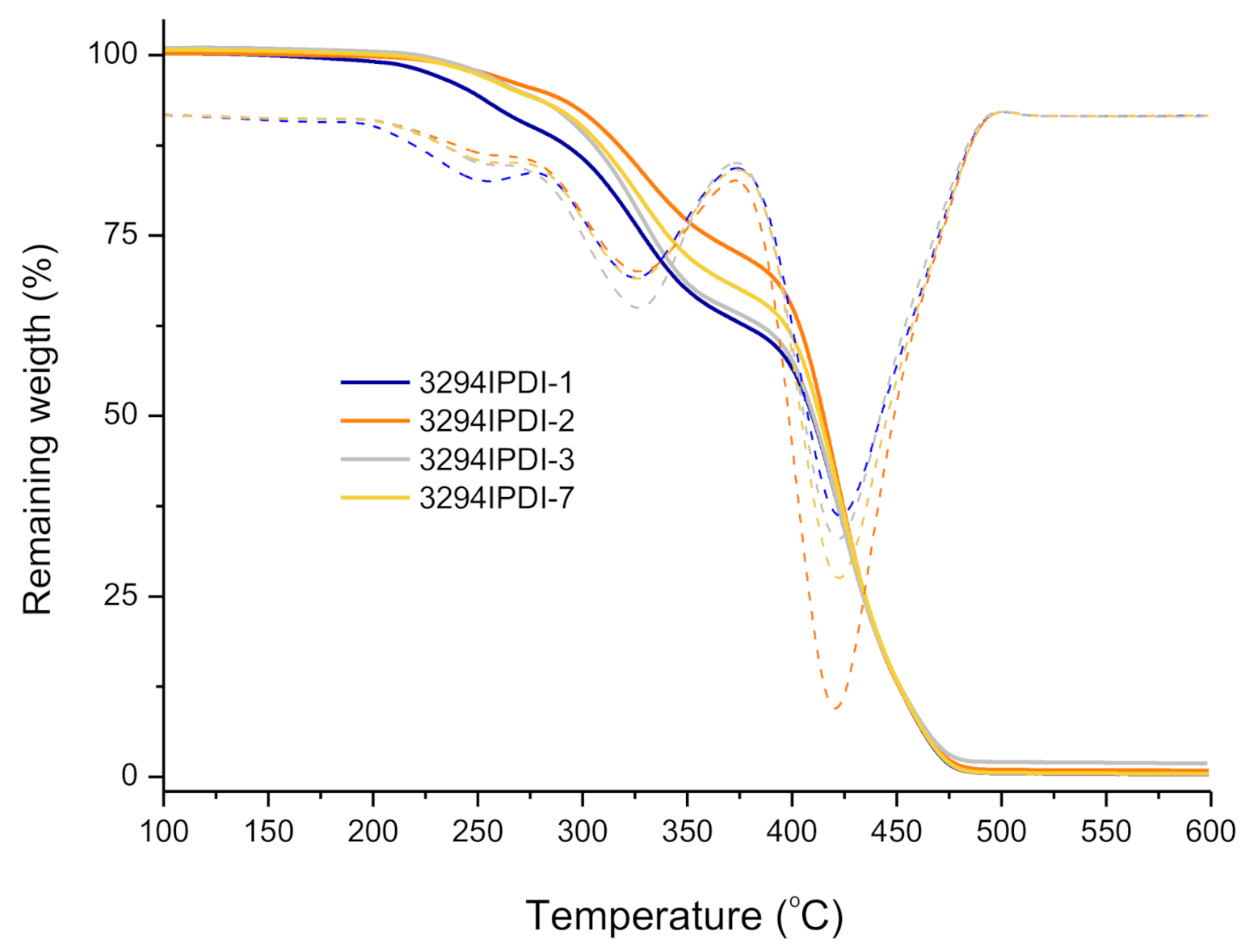
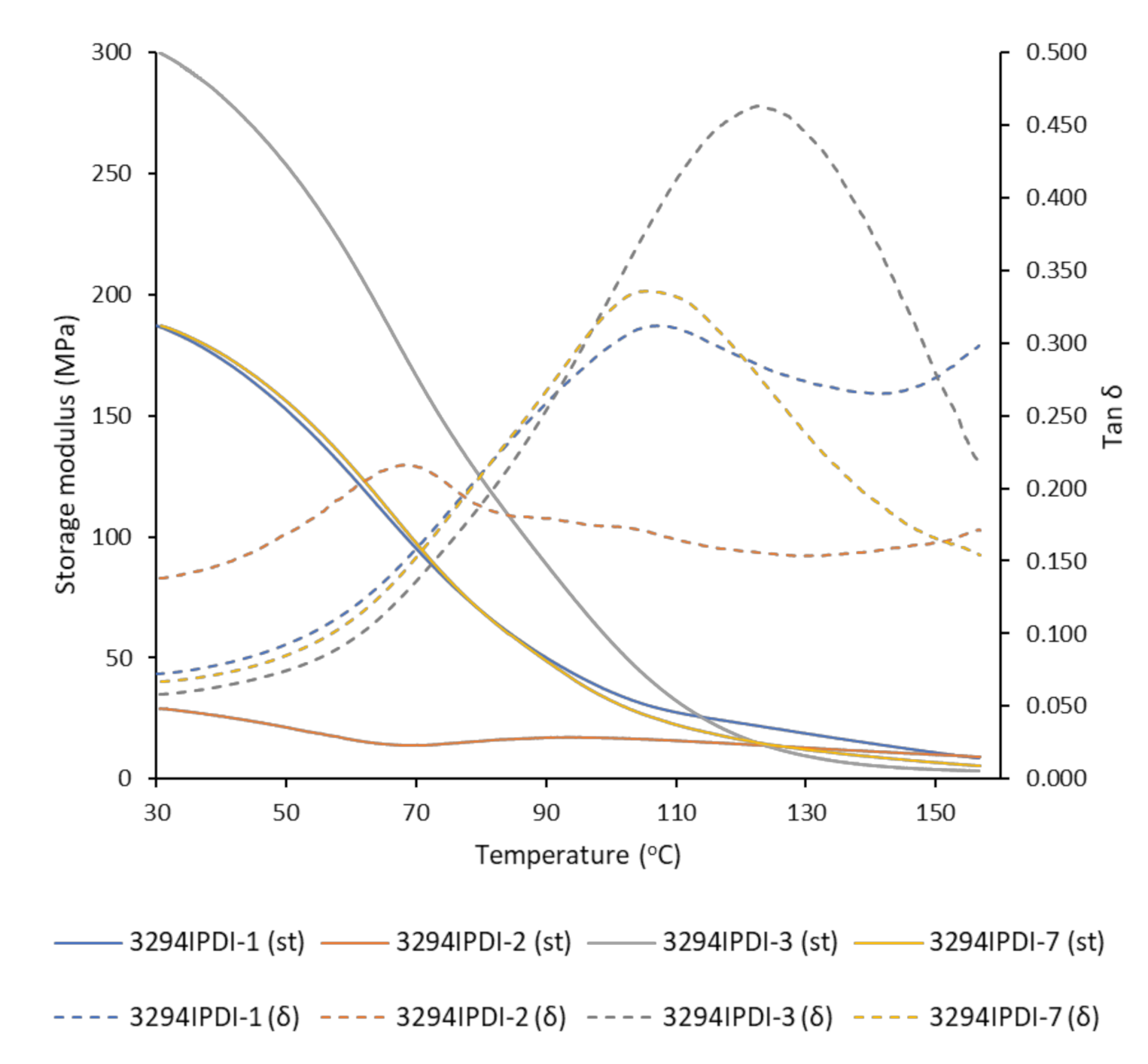
| Reference | DSC a | TGA b | DMTA c | |||
|---|---|---|---|---|---|---|
| Tg (°C) | T10% (°C) | TdS1 (°C) | TdS2 (°C) | TdS3 (°C) | Tα (°C) | |
| 3294IPDI-1 | −53.8 | 276.5 | 254.9 | 326.1 | 423.2 | 109.2 |
| 3294IPDI-2 | −50.8 | 308.6 | 263.3 | 327.2 | 420.6 | 68.9 |
| 3294IPDI-3 | −53.7 | 297.6 | 261.1 | 326.8 | 422.5 | 123.5 |
| 3294IPDI-7 | −51.2 | 300.1 | 261.8 | 326.2 | 422.7 | 106.2 |
| Priplast 3294 | −66.0 | 402.7 | - | - | 414.4 | N.D. d |
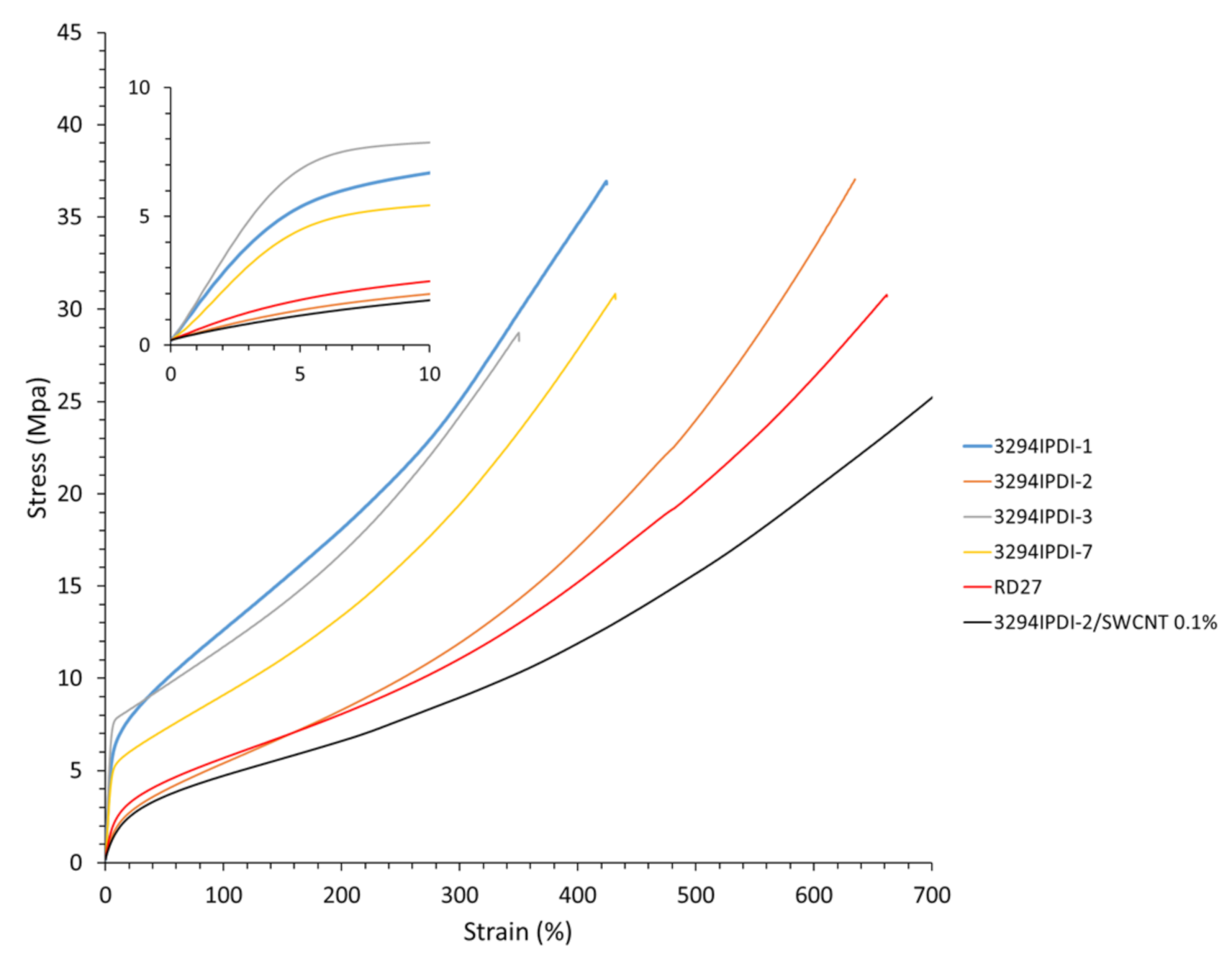
| Reference | E a (Mpa) | σ100% a (Mpa) | σb a (Mpa) | εb a (%) |
|---|---|---|---|---|
| 3294IPDI-1 | 120.6 ± 4.0 | 12.5 ± 0.1 | 35.9 ± 1.2 | 415.4 ± 11.4 |
| 3294IPDI-2 | 25.1 ± 0.3 | 5.4 ± 0.0 | 35.4 ± 1.4 | 625.4 ± 15.8 |
| 3294IPDI-3 | 161.0 ± 9.3 | 11.7 ± 0.2 | 28.4 ± 2.0 | 347.8 ± 11.9 |
| 3294IPDI-7 | 103.9 ± 6.1 | 9.1 ± 0.1 | 28.7 ± 1.3 | 427.3 ± 8.3 |
| 3294IPDI-2/ SWCNT 0.1 wt. %b | 19.3 ± 1.2 | 4.8 ± 0.2 | 25.8 ± 6.1 | 735.0 ± 6.1 |
| RD27 b | 35.4 ± 0.4 | 5.6 ± 0.0 | 33.4 ± 4.7 | 700.7 ± 80.5 |
| Reference | pH at 35% of Solid Content | Average Particle Size (nm) | PdI | Z Potential (mV) |
|---|---|---|---|---|
| 3294IPDI-1 | 8.1 | 50 ± 7 | 0.12 ± 0.1 | −40.5 ± 6.0 |
| 3294IPDI-2 | 8.2 | 217 ± 40 | 0.11 ± 0.1 | −38.7 ± 7.9 |
| 3294IPDI-3 | 8.1 | 49 ± 10 | 0.20 ± 0.1 | −39.8 ± 4.3 |
| 3294IPDI-7 | 8.0 | 48 ± 15 | 0.19 ± 0.1 | −43.5 ± 4.7 |
| Coating Reference | Substrate | |||||||
|---|---|---|---|---|---|---|---|---|
| Glass Slides | UPRON a | |||||||
| WCA (°) | θAv (°) | θRe (°) | CAHb (°) | WCA (°) | θAv (°) | θRe (°) | CAH b (°) | |
| Uncoated | spreads | - | - | - | wets | - | - | - |
| 3294IPDI-1 | 98.4 ± 1.3 | 97.0 ± 3.3 | 94.8 ± 0.8 | 2.3 ± 3.4 | 121.4 ± 1.8 | 122.7 ± 3.8 | 119.4 ± 5.1 | 3.2 ± 1.5 |
| 3294IPDI-2 | 86.0 ± 1.9 | 91.5 ± 4.5 | 84.4 ± 5.6 | 7.1 ± 6.5 | 119.7 ± 4.0 | 125.1 ± 4.4 | 121.6 ± 5.7 | 2.4 ± 2.2 |
| 3294IPDI-3 | 92.4 ± 2.7 | 92.9 ± 1.0 | 87.1 ± 4.5 | 5.9 ± 4.6 | 116.3 ± 2.8 | 117.1 ± 1.1 | 113.8 ± 1.1 | 3.3 ± 1.0 |
| 3294IPDI-7 | 86.9 ± 0.6 | 90.4 ± 2.2 | 88.2 ± 2.2 | 2.2 ± 3.1 | 113.2 ± 7.0 | 115.3 ± 6.4 | 111.8 ± 5.7 | 3.5 ± 1.0 |
| RD27 | 78.2 ± 1.0 | 79.4 ± 1.0 | 77.0 ± 1.3 | 2.4 ± 1.6 | 106.8 ± 2.4 | 111.2 ± 0.4 | 110.6 ± 0.7 | 0.6 ± 1.0 |
| 3294IPDI-2/ SWCNT 0.05 wt. % | 78.3 ± 1.2 | 90.8 ± 3.6 | 86.2 ± 1.2 | 4.6 ± 3.8 | 111.7 ± 1.4 | 112.7 ± 3.8 | 109.4 ± 2.8 | 3.3 ± 3.2 |
| 3294IPDI-2/ SWCNT 0.1 wt. % | 89.2 ± 1.6 | 96.0 ± 3.0 | 94.3 ± 2.7 | 1.6 ± 4.1 | 114.9 ± 5.0 | 120.5 ± 1.4 | 115.7 ± 1.6 | 4.8 ± 0.9 |
| Coating Reference | Dry Add-on (g/m2) | Stiffness [mN] | Water Column (cm) | Air Permeability (mm/s) | |
|---|---|---|---|---|---|
| Warp | Weft | ||||
| Uncoated | - | 17.4 | 9.7 | <15 soaked | 85.3 |
| 3294IPDI-1 | 23.5 | 153.0 | 50.3 | 26.9 | 3.7 |
| 3294IPDI-2 | 27.0 | 127.0 | 42.2 | 38.0 | 0.7 |
| 3294IPDI-3 | 24.9 | 154.0 | 50.3 | 28.9 | 2.6 |
| 3294IPDI-7 | 26.0 | 161.0 | 47.3 | 29.3 | 1.8 |
| RD27 | 22.0 | 155.0 | 43.9 | 34.0 | 0.4 |
| 3294IPDI-2/SWCNT 0.05 wt. % | 24.1 | 124.0 | 38.9 | 32.7 | 4.4 |
| 3294IPDI-2/SWCNT 0.1 wt. % | 24.0 | 87.0 | 31.9 | 33.8 | 3.6 |
| Coating Reference | Electrostatic Property | Unit | Limit | Value | Requirements EN 1149-5 |
|---|---|---|---|---|---|
| 3294IPDI-2 | Surface resistivity | Ω | ≤2.5 × 109 | 9.2 × 1012 | FAIL |
| Charge decay (inductive charging) | s | t50 < 4 | >30 | ||
| - | S > 0.2 | 0.03 | |||
| Vertical resistance | Ω | >105 | 1.1.1012 | ||
| 3294IPDI-2/SWCNT 0.05 wt. % | Surface resistivity | Ω | ≤2.5 × 109 | 3.8 × 106 | PASS |
| Charge decay (inductive charging) | s | t50 < 4 | <0.01 | ||
| - | S > 0.2 | 0.99 | |||
| Vertical resistance | Ω | >105 | 2.8 × 1011 | ||
| 3294IPDI-2/SWCNT 0.1 wt. % | Surface resistivity | Ω | ≤2.5 × 109 | 3.6 × 106 | PASS |
| Charge decay (inductive charging) | s | t50 < 4 | <0.01 | ||
| - | S > 0.2 | 0.99 | |||
| Vertical resistance | Ω | >105 | 3.2 × 1011 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lacruz, A.; Salvador, M.; Blanco, M.; Vidal, K.; Goitandia, A.M.; Martinková, L.; Kyselka, M.; de Ilarduya, A.M. Biobased Waterborne Polyurethane-Urea/SWCNT Nanocomposites for Hydrophobic and Electrically Conductive Textile Coatings. Polymers 2021, 13, 1624. https://doi.org/10.3390/polym13101624
Lacruz A, Salvador M, Blanco M, Vidal K, Goitandia AM, Martinková L, Kyselka M, de Ilarduya AM. Biobased Waterborne Polyurethane-Urea/SWCNT Nanocomposites for Hydrophobic and Electrically Conductive Textile Coatings. Polymers. 2021; 13(10):1624. https://doi.org/10.3390/polym13101624
Chicago/Turabian StyleLacruz, Amado, Mireia Salvador, Miren Blanco, Karmele Vidal, Amaia M. Goitandia, Lenka Martinková, Martin Kyselka, and Antxon Martínez de Ilarduya. 2021. "Biobased Waterborne Polyurethane-Urea/SWCNT Nanocomposites for Hydrophobic and Electrically Conductive Textile Coatings" Polymers 13, no. 10: 1624. https://doi.org/10.3390/polym13101624
APA StyleLacruz, A., Salvador, M., Blanco, M., Vidal, K., Goitandia, A. M., Martinková, L., Kyselka, M., & de Ilarduya, A. M. (2021). Biobased Waterborne Polyurethane-Urea/SWCNT Nanocomposites for Hydrophobic and Electrically Conductive Textile Coatings. Polymers, 13(10), 1624. https://doi.org/10.3390/polym13101624







