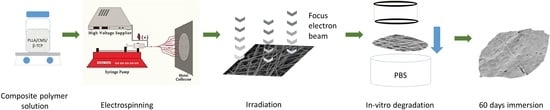
Electron-Beam Irradiation of the PLLA/CMS/β-TCP Composite Nanofibers Obtained by Electrospinning
Abstract
:1. Introduction
2. Materials and Methods
2.1. Characterizations
2.1.1. Surface Morphology
2.1.2. Chemical Interactions
2.1.3. Thermal Behavior
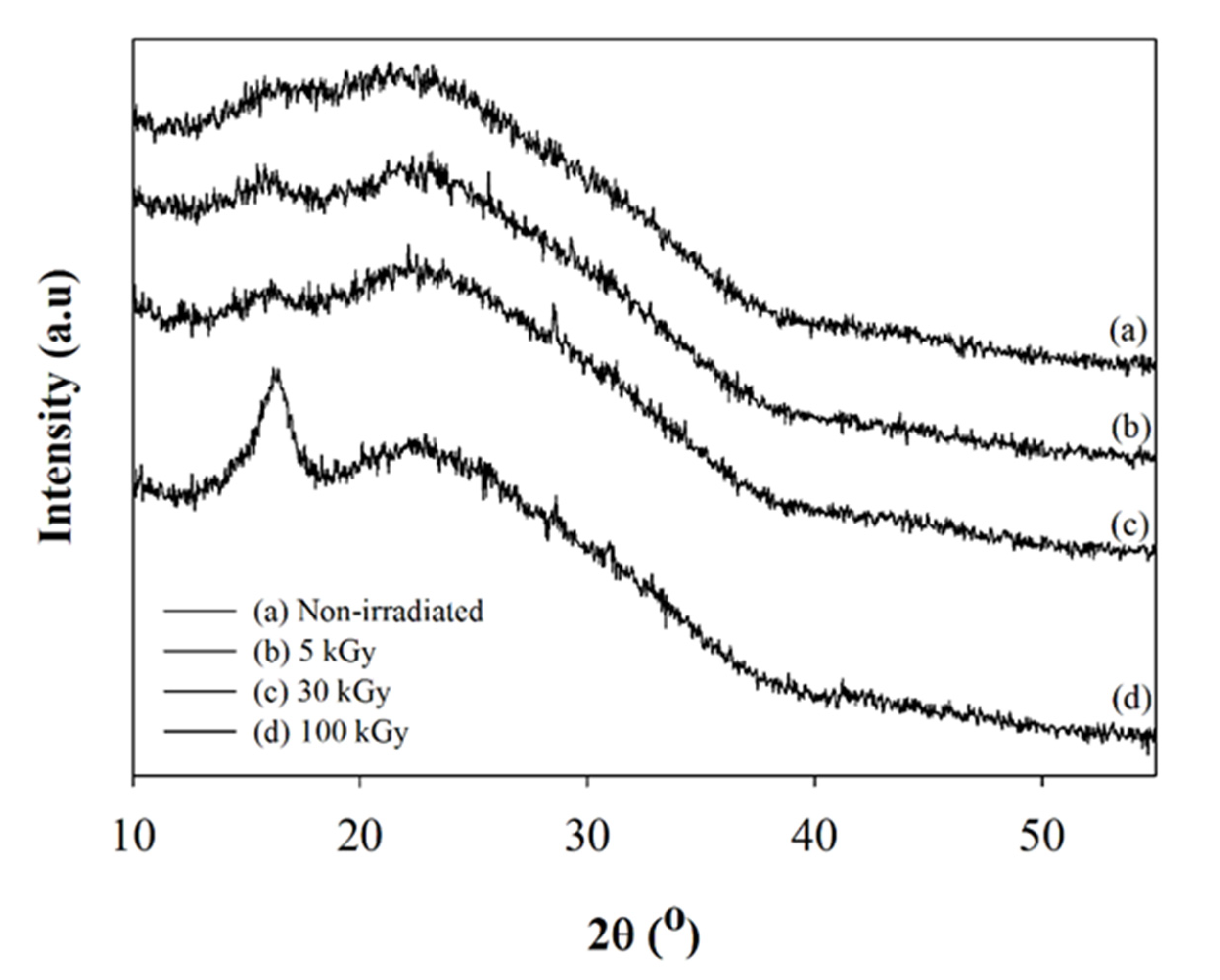
2.1.4. X-ray Diffractions (XRD)
2.1.5. Wettability
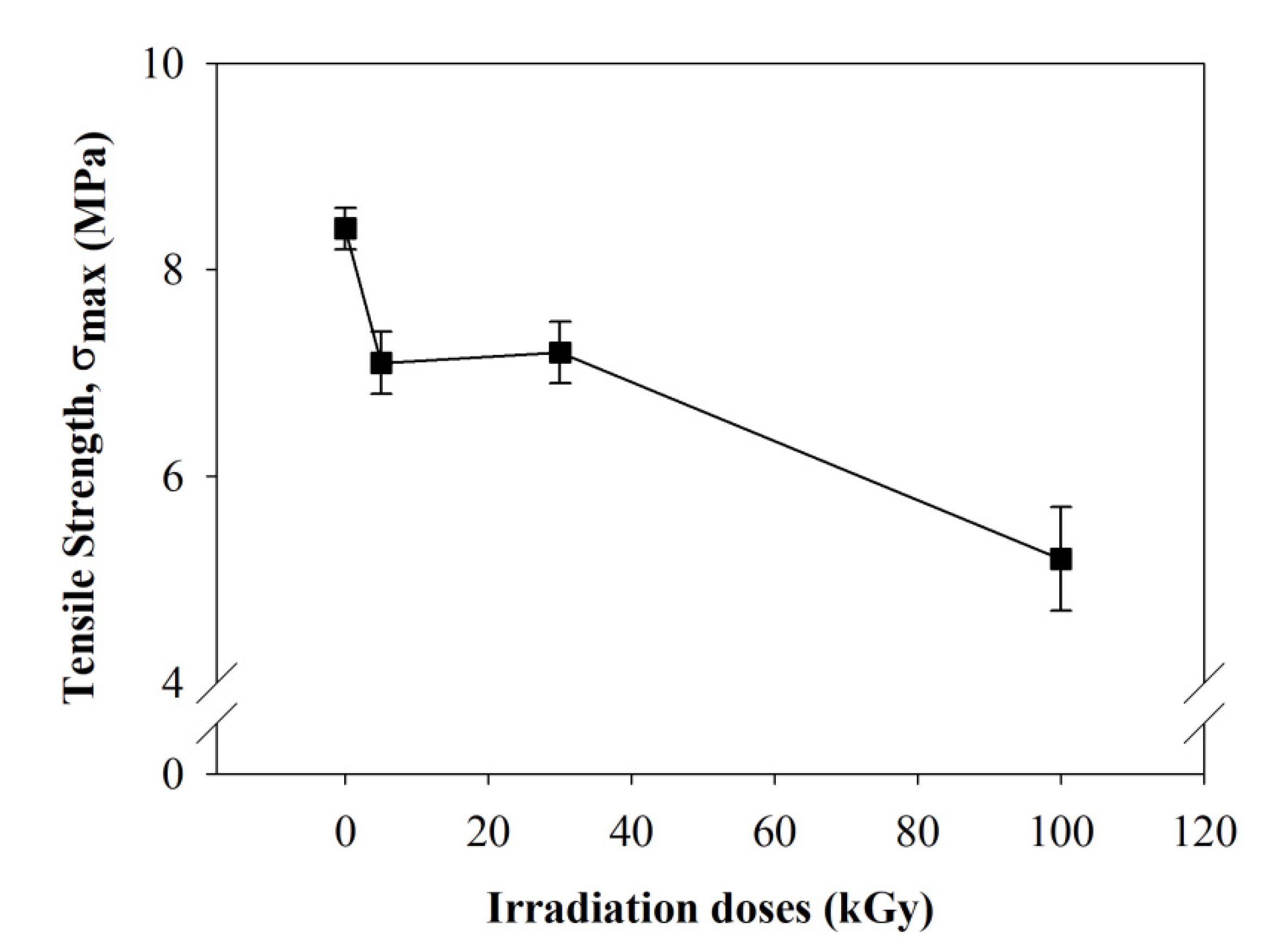
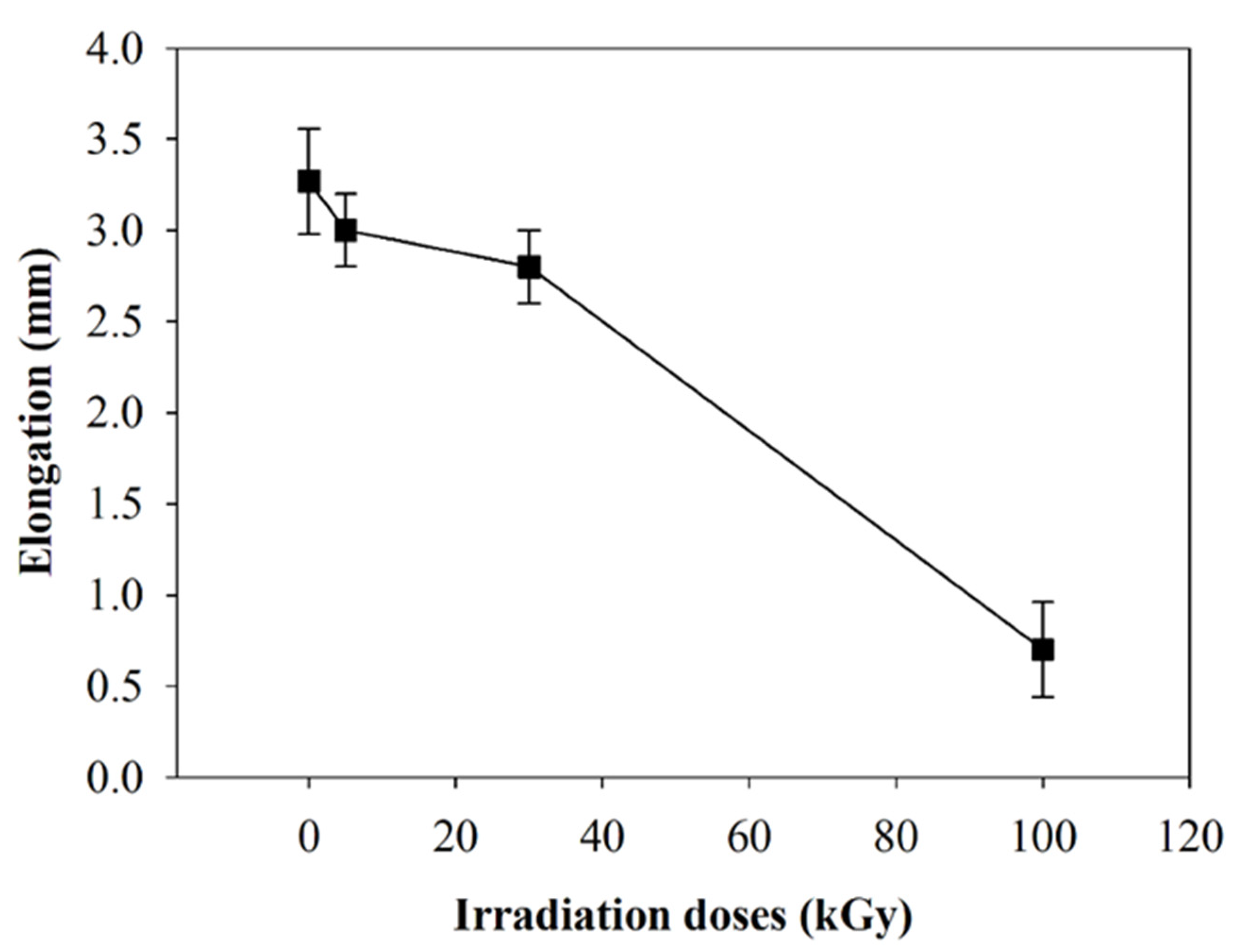
2.1.6. Mechanical Properties
2.1.7. Irradiation
2.1.8. In Vitro Degradation
3. Results
4. Conclusions
- the fiber diameter of composite nanofibers changed slightly after beam irradiation.
- WCA dropped from 125 to 118° due to the formation of hydroxyl groups after the highest dose of beam irradiation.
- FTIR, DSC, and XRD results indicated that there are chemical changes to the structure of composite nanofibers after the highest dose of beam irradiation.
- A tensile test was run to observe the mechanical strength before and after beam irradiation. Apparently, the material decreased its mechanical properties by 2.6 times due to the breaking of the polymer chains after the highest beam-irradiation dose. Moreover, rapid reduction in the elongation showed that polymer degradation took place after beam irradiation.
- SEM images after electron-beam irradiation showed that degradation took place after 30 and 60 days in vitro test.
- Electron-beam irradiation enhanced the in vitro degradation of PLLA matrix since the PLLA component has a longer degradation period. However, higher-irradiation doses retarded the mechanical integrity of the composite.
Author Contributions
Funding
Acknowledgments
- 1-
- Fundamental Research Grant Scheme (FRGS/1/2018/STG07/UKM/02/03—Hybrid Nanofibers under Ionizing Radiation as a Guided Bone Regeneration Membrane, Universiti Kebangsaan Malaysia, Ministry of Higher Education.
- 2-
- The Ministry of Education, Youth and Sports of the Czech Republic and the European Union - European Structural and Investment Funds in the frames of Operational Program Research, Development and Education - project Hybrid Materials for Hierarchical Structures (HyHi, Reg. No. CZ.02.1.01/0.0/0.0/16_019/0000843).
Conflicts of Interest
References
- Fokin, N.; Grothe, T.; Mamun, A.; Trabelsi, M.; Klöcker, M.; Sabantina, L.; Döpke, C.; Blachowicz, T.; HütteAn, A.; Ehrmann, A. Magnetic Properties of Electrospun Magnetic Nanofiber Mats after Stabilization and Carbonization. Materials 2020, 13, 1552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozior, T.; Mamun, A.; Trabelsi, M.; Wortmann, M.; Lilia, S.; Ehrmann, A. Electrospinning on 3D Printed Polymers for Mech. Stab. Filter Compos. Polym. 2019, 11, 2034. [Google Scholar] [CrossRef] [Green Version]
- Yalcinkaya, F. A review on advanced nanofiber technology for membrane distillation. J. Eng. Fibers Fabr. 2019, 14, 1558925018824901. [Google Scholar] [CrossRef]
- Yusof, M.R.; Shamsudin, R.; Abdullah, Y.; Yalcinkaya, F.; Yaacob, N. Electrospinning of carboxymethyl starch/poly(L-lactide acid) composite nanofiber. Polym. Adv. Technol. 2018, 29, 1843–1851. [Google Scholar] [CrossRef]
- Wehlage, D.; Blattner, H.; Mamun, A.; Kutzli, I.; Diestelhorst, E.; Rattenholl, A.; Gudermann, F.; Lütkemeyer, D.; Ehrmann, A. Cell growth on electrospun nanofiber mats from polyacrylonitrile (PAN) blends. Aims Bioeng. 2020, 7, 43. [Google Scholar] [CrossRef]
- Blachowicz, T.; Ehrmann, A. Most recent developments in electrospun magnetic nanofibers: A review. J. Eng. Fibers Fabr. 2020, 15, 1558925019900843. [Google Scholar] [CrossRef]
- Blachowicz, T.; Ehrmann, A. Recent developments in electrospun ZnO nanofibers: A short review. J. Eng. Fibers Fabr. 2020, 15, 1558925019899682. [Google Scholar] [CrossRef] [Green Version]
- Blachowicz, T.; Ehrmann, A. Conductive Electrospun Nanofiber Mats. Materials 2019, 13, 152. [Google Scholar] [CrossRef] [Green Version]
- Trabelsi, M.; Mamun, A.; Klöcker, M.; Sabantina, L.; Großerhode, C.; Blachowicz, T.; Ehrmann, A. Increased Mechanical Properties of Carbon Nanofiber Mats for Possible Medical Applications. Fibers 2019, 7, 98. [Google Scholar] [CrossRef] [Green Version]
- Yalcinkaya, F.; Komarek, M.; Lubasova, D.; Sanetrnik, F.; Maryska, J. Preparation of Antibacterial Nanofibre/Nanoparticle Covered Composite Yarns. Available online: https://www.hindawi.com/journals/jnm/2016/7565972/ (accessed on 25 June 2020).
- Yalcinkaya, F.; Siekierka, A.; Bryjak, M. Preparation of Fouling-Resistant Nanofibrous Composite Membranes for Separation of Oily Wastewater. Polymers (Basel) 2017, 9, 679. [Google Scholar] [CrossRef] [Green Version]
- He, C.; Feng, W.; Cao, L.; Fan, L. Crosslinking of poly(L-lactide) nanofibers with triallyl isocyanutrate by gamma-irradiation for tissue engineering application. J. Biomed. Mater. Res. A 2011, 99, 655–665. [Google Scholar] [CrossRef]
- Hu, X.; Liu, S.; Zhou, G.; Huang, Y.; Xie, Z.; Jing, X. Electrospinning of polymeric nanofibers for drug delivery applications. J. Control. Release 2014, 185, 12–21. [Google Scholar] [CrossRef]
- Alghoraibi, I.; Alomari, S. Different Methods for Nanofiber Design and Fabrication. In Handbook of Nanofibers; Barhoum, A., Bechelany, M., Makhlouf, A., Eds.; Springer International Publishing: Cham, Germany, 2018; pp. 1–46. ISBN 978-3-319-42789-8. [Google Scholar]
- Yalcinkaya, F.; Yalcinkaya, B.; Jirsak, O. Influence of Salts on Electrospinning of Aqueous and Nonaqueous Polymer Solutions. J. Nanomater. 2015, 2015, 12. [Google Scholar] [CrossRef]
- Zeng, J.; Haoqing, H.; Schaper, A.; Wendorff, J.H.; Greiner, A. Poly-L-lactide nanofibers by electrospinning—Influence of solution viscosity and electrical conductivity on fiber diameter and fiber morphology. e-Polymers 2003, 1, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Ren, K.; Wang, Y.; Sun, T.; Yue, W.; Zhang, H. Electrospun PCL/gelatin composite nanofiber structures for effective guided bone regeneration membranes. Mater. Sci. Eng. C 2017, 78, 324–332. [Google Scholar] [CrossRef]
- Morikawa, K.; Green, M.; Naraghi, M. A Novel Approach for Melt Electrospinning of Polymer Fibers. Procedia Manuf. 2018, 28, 205–208. [Google Scholar] [CrossRef]
- Koenig, K.; Beukenberg, K.; Langensiepen, F.; Seide, G. A new prototype melt-electrospinning device for the production of biobased thermoplastic sub-microfibers and nanofibers. Biomater. Res. 2019, 23, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Ramakrishna, S.; Fujihara, K.; Wee, E.T.; Teik, C.L.; Zuwei, M. An Introduction to Electrospinning and Nanofibers 2019; World Scientific: Singapore, 2019. [Google Scholar]
- Fang, J.; Shao, H.; Niu, H.; Lin, T. Applications of Electrospun Nanofibers for Electronic Devices. In Handbook of Smart Textiles; Xiaoming, T., Ed.; Springer Science: Singapore, 2015; pp. 617–652. ISBN 978-981-4451-44-4. [Google Scholar]
- Xue, J.; Xie, J.; Liu, W.; Xia, Y. Electrospun nanofibers: New concepts, materials and applications. Acc. Chem. Res. 2017, 50, 1976–1987. [Google Scholar] [CrossRef]
- Vasita, R.; Katti, D.S. Nanofibers and their applications in tissue engineering. Int. J. Nanomed. 2006, 1, 15–30. [Google Scholar] [CrossRef]
- Zulkifli, F.H.; Shahitha, F.; Yusuff, M.M.; Hamidon, N.N.; Chahal, S. Cross-Linking Effect on Electrospun Hydroxyethyl Cellulose/Poly(Vinyl Alcohol) Nanofibrous Scaffolds. Procedia Eng. 2013, 53, 689–695. [Google Scholar] [CrossRef] [Green Version]
- Shi, Q.; Zhou, C.; Yue, Y.; Guo, W.; Wu, Y.; Wu, Q. Mechanical properties and in vitro degradation of electrospun bio-nanocomposite mats from PLA and cellulose nanocrystals. Carbohydr. Polym. 2012, 90, 301–308. [Google Scholar] [CrossRef]
- Cairns, M.-L.; Dickson, G.R.; Orr, J.F.; Farrar, D.; Hawkins, K.; Buchanan, F.J. Electron-beam treatment of poly(lactic acid) to control degradation profiles. Polym. Degrad. Stab. 2011, 96, 76–83. [Google Scholar] [CrossRef] [Green Version]
- Said, H.M. Effects of gamma irradiation on the crystallization, thermal and mechanical properties of poly(l-lactic acid)/ethylene-co-vinyl acetate blends. J. Radiat. Res. Appl. Sci. 2013, 6, 11–20. [Google Scholar] [CrossRef] [Green Version]
- Walo, M.; Przybytniak, G.; Nowicki, A.; Świeszkowski, W. Radiation-induced effects in gamma-irradiated PLLA and PCL at ambient and dry ice temperatures. J. Appl. Polym. Sci. 2011, 122, 375–383. [Google Scholar] [CrossRef]
- Lee, J.B.; Ko, Y.-G.; Cho, D.; Park, W.H.; Kim, B.N.; Lee, B.C.; Kang, I.-K.; Kwon, O.H. Modification of PLGA Nanofibrous Mats by Electron Beam Irradiation for Soft Tissue Regeneration. Available online: https://www.hindawi.com/journals/jnm/2015/295807/ (accessed on 25 June 2020).
- Loo, S.C.J.; Ooi, C.P.; Boey, Y.C.F. Radiation effects on poly(lactide-co-glycolide) (PLGA) and poly(l-lactide) (PLLA). Polym. Degrad. Stab. 2004, 83, 259–265. [Google Scholar] [CrossRef]
- Jeun, J.P.; Jeon, Y.K.; Nho, Y.C.; Kang, P.H. Effects of gamma irradiation on the thermal and mechanical properties of chitosan/PVA nanofibrous mats. J. Ind. Eng. Chem. 2009, 15, 430–433. [Google Scholar] [CrossRef]
- Zhang, X.; Kotaki, M.; Okubayashi, S.; Sukigara, S. Effect of electron beam irradiation on the structure and properties of electrospun PLLA and PLLA/PDLA blend nanofibers. Acta Biomater. 2010, 6, 123–129. [Google Scholar] [CrossRef]
- Yusof, M.R. Fabrikasi Ddan Pencirian Komposit Gentian Nano Asid Poli L-Laktik/Kanji Karboksimetil/β-Trikalsium Fosfat melalui Teknik Elektroputaran dan kesan Terhadap Iradiasi Alur Elektron. Ph.D. Thesis, Universiti Kebangsaan Malaysia, Bangi, Malaysia, 2020. [Google Scholar]
- Yusof, M.R.; Shamsudin, R.; Zakaria, S.; Abdul Hamid, M.A.; Yalcinkaya, F.; Abdullah, Y.; Yacob, N. Fabrication and Characterization of Carboxymethyl Starch/Poly(l-Lactide) Acid/β-Tricalcium Phosphate Composite Nanofibers via Electrospinning. Polymers (Basel) 2019, 11, 1468. [Google Scholar] [CrossRef] [Green Version]
- Bohari, Y.; Iqbal, M.A.M.C.; Kamaruddin, H.; Bukhori, A.B. Optimization of Reaction Conditions for Carboxymethylated Sago Starch. Iran. Polym. J. 2011, 20, 195–204. [Google Scholar]
- Randolph, S.J.; Fowlkes, J.D.; Rack, P.D. Effects of heat generation during electron-beam-induced deposition of nanostructures. J. Appl. Phys. 2005, 97, 124312. [Google Scholar] [CrossRef] [Green Version]
- Malinowski, R.; Rytlewski, P.; Janczak, K.; Raszkowska-Kaczor, A.; Moraczewski, K.; Stepczyńska, M.; Żuk, T. Studies on functional properties of PCL films modified by electron radiation and TAIC additive. Polym. Test. 2015, 48, 169–174. [Google Scholar] [CrossRef]
- Nagasawa, N.; Kaneda, A.; Kanazawa, S.; Yagi, T.; Mitomo, H.; Yoshii, F.; Tamada, M. Application of poly(lactic acid) modified by radiation crosslinking. Nucl. Instrum. Methods Phys. Res. B 2005, 236, 611–616. [Google Scholar] [CrossRef]
- Gómez-Pachón, E.Y.; Vera-Graziano, R.; Campos, R.M. Structure of poly(lactic-acid) PLA nanofibers scaffolds prepared by electrospinning. IOP Conf. Ser. Mater. Sci. Eng. 2014, 59, 012003. [Google Scholar] [CrossRef] [Green Version]
- Kim, B.K.; Cho, D.; Kwon, O.H.; Park, W.H.; Lee, J.-H. Effects of electron beam irradiation on the gel fraction, thermal and mechanical properties of poly(butylene succinate) crosslinked by multi-functional monomer. Mater. Des. 2015, 87, 428–435. [Google Scholar] [CrossRef]
- Bouscaud, D.; Pesci, R.; Berveiller, S.; Patoor, E. Estimation of the electron beam-induced specimen heating and the emitted X-rays spatial resolution by Kossel microdiffraction in a scanning electron microscope. Ultramicroscopy 2012, 115, 115–119. [Google Scholar] [CrossRef] [Green Version]
- Cho, A.-R.; Shin, D.M.; Jung, H.W.; Hyun, J.C.; Lee, J.S.; Cho, D.; Joo, Y.L. Effect of annealing on the crystallization and properties of electrospun polylatic acid and nylon 6 fibers. J. Appl. Polym. Sci. 2011, 120, 752–758. [Google Scholar] [CrossRef]
- Ryuji, I.; Masaya, K.; Ramakrishna, S. Structure and properties of electrospun PLLA single nanofibres. Nanotechnology 2005, 16, 208–219. [Google Scholar] [CrossRef]
- Zulkifli, F.H.; Jahir Hussain, F.S.; Abdull Rasad, M.S.B.; Mohd Yusoff, M. In vitro degradation study of novel HEC/PVA/collagen nanofibrous scaffold for skin tissue engineering applications. Polym. Degrad. Stab. 2014, 110, 473–481. [Google Scholar] [CrossRef] [Green Version]










© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yusof, M.R.; Shamsudin, R.; Zakaria, S.; Azmi Abdul Hamid, M.; Yalcinkaya, F.; Abdullah, Y.; Yacob, N. Electron-Beam Irradiation of the PLLA/CMS/β-TCP Composite Nanofibers Obtained by Electrospinning. Polymers 2020, 12, 1593. https://doi.org/10.3390/polym12071593
Yusof MR, Shamsudin R, Zakaria S, Azmi Abdul Hamid M, Yalcinkaya F, Abdullah Y, Yacob N. Electron-Beam Irradiation of the PLLA/CMS/β-TCP Composite Nanofibers Obtained by Electrospinning. Polymers. 2020; 12(7):1593. https://doi.org/10.3390/polym12071593
Chicago/Turabian StyleYusof, Mohd Reusmaazran, Roslinda Shamsudin, Sarani Zakaria, Muhammad Azmi Abdul Hamid, Fatma Yalcinkaya, Yusof Abdullah, and Norzita Yacob. 2020. "Electron-Beam Irradiation of the PLLA/CMS/β-TCP Composite Nanofibers Obtained by Electrospinning" Polymers 12, no. 7: 1593. https://doi.org/10.3390/polym12071593
APA StyleYusof, M. R., Shamsudin, R., Zakaria, S., Azmi Abdul Hamid, M., Yalcinkaya, F., Abdullah, Y., & Yacob, N. (2020). Electron-Beam Irradiation of the PLLA/CMS/β-TCP Composite Nanofibers Obtained by Electrospinning. Polymers, 12(7), 1593. https://doi.org/10.3390/polym12071593