3.3.1. Bionanocomposites with Lemon Hybrid Pigments
The optimized lemon hybrid pigments with HT and MMT were incorporated into INZEA biopolymer for the development and characterization of bio-nanocomposites at different hybrid pigments loading (3, 5, and 7 wt%). Blank samples from MMT and HT pure nanoclays were also extruded and used to compare color, thermal, and mechanical properties of the developed bio-nanocomposites.
The selection of the proposed weight amounts was done considering that, regardless of the final color of the samples, enhancement in mechanical performance in polymer-based composites is generally obtained at similar contents, where the agglomeration of the nanoscale additives is usually limited. Visual images of the produced materials are reported in
Figure 4. A darker color was evidenced for MMT containing samples due to the intrinsic color of this nanoclay compared to HT. The results obtained for color parameters are included in
Table 6. A positive correlation between the nanoclay type, its content and the dye absorbed by the nanocomposites was evidenced. As already observed by other authors, the disperse dyeing behavior of composites depends on the dye’s affinity to the polymer, chemical structure of the dispersed dye, and active area [
45].
Previous research articles have focused on the dyeing properties of polypropylene and polyamide six-layered clay incorporated in nanocomposites prepared by melt compounding. Toshniwal et al. [
46] suggested that polypropylene fibers could be made dyeable with disperse dyes by the addition of nanoclay particles in the polymer matrix. Another research work done by Razafimahefa et al. [
47] showed that the introduction of nanoclays improved the dyeing ability of nylon with dispersed dyes.
In the case of INZEA-based samples, the neat matrix was characterized by a high lightness value (
Table 6), induced by the color of the neat polymer. The addition of MMT and HT in INZEA-based systems produced, respectively, a reduction and increase in L* values. This phenomenon was due to the MMT and HT powders’ intrinsic colors, which were able to modify the final aesthetic quality and appearance of the different samples. The L* value was maintained stable with increasing the ratio nanofiller/polymer. Moreover, no variation of a* and b* values was observed with increasing the amount of MMT and HT in the INZEA_MMT and INZEA_HT-based systems (
Table 6).
The presence of lemon hybrid pigments in INZEA-based systems determined a remarkable variation in CIELAB values. In INZEA MMT_LEM-based systems, a reduction in L* and increase in b* values, due to the color of the bio-based nanofillers, were observed. Lemon pigments, in terms of CIELAB parameters, were influenced by the color of the lemon skin from which they were extracted [
48]. A reduction in lightness and an increase in a* and b* coordinates were registered with loading INZEA with HT_LEM nanofiller. The influence of MMT_LEM and HT_LEM hybrid pigments were more visible with increasing the nanofiller ratio. Comparing INZEA MMT_LEM and INZEA HT_LEM, it is possible to highlight that HT_LEM-based samples were characterized by a bright yellow color compared to MMT_LEM samples. Essentially, the values registered for b* parameter and INZEA HT_LEM were higher than those obtained for INZEA MMT_LEM samples. Lastly, gloss values were reduced by using unmodified MMT and HT fillers or using MMT_LEM and HT_LEM pigments, this variation were more evident increasing the nanofiller content. A similar behavior has been reported for different polymeric matrices and nanofillers [
49].
Tensile characterization results of INZEA-based samples are reported in
Table 7. In general, a decrease in terms of strength and strain at break was observed for all formulations, regardless of the type or weight amount of the nanopigment introduced in the polymer matrix. Specifically, tensile strength maintained an acceptable deformation level only when 3 wt% of unmodified MMT was considered, while HT, even at the same lower amount, strongly decreased the ductility of the matrix. It has been reported [
50] that a dye−clay nanopigment, synthesized via a cationic exchange reaction between mineral nanoclays and organic dyes, can be considered to gain the advantages of nanoclay reinforcement and colorant features, simultaneously. Nevertheless, even if the dispersion in polymer matrices can offer superior colorimetric properties and color fastness compared to the inorganic counterparts, mechanical enhancement can be obtained only if the nanopigment has been properly intercalated or exfoliated in the formulation. In our case, the probable formation of some aggregates and a limited extent of intercalation/exfoliation of MMT and HT in the polymer matrix could explain the observed results. This observation is strictly correlated with the experimental results, which showed a strong decrease in ductility, in parallel with a limited reinforcement effect (values of elastic modulus were essentially maintained constant for all formulations). As already observed by Marchante et al. in the case of EVA-based systems [
51], a decrease in elastic modulus (E) could be related to an overall decrease in the rigidity provided by the nanoclay, as the specific surface area was decreased and the stress was transferred through the matrix to the nanoclay. On the other hand, almost all of the samples presented lower deformation values at break (ε
b). In this case, the presence of tactoids could reduce the capacity of the material to absorb energy as a consequence of a reduction in the polymer’s mobility chain. The strain at break also decreased when the amount of the nanoadditive increased and this reduction was more noticeable in samples with nanopigments compared to samples with unmodified nanoclays, confirming that the added nanopigments reduced the compatibility between MMT (and HT) and the polyester-based matrix [
52].
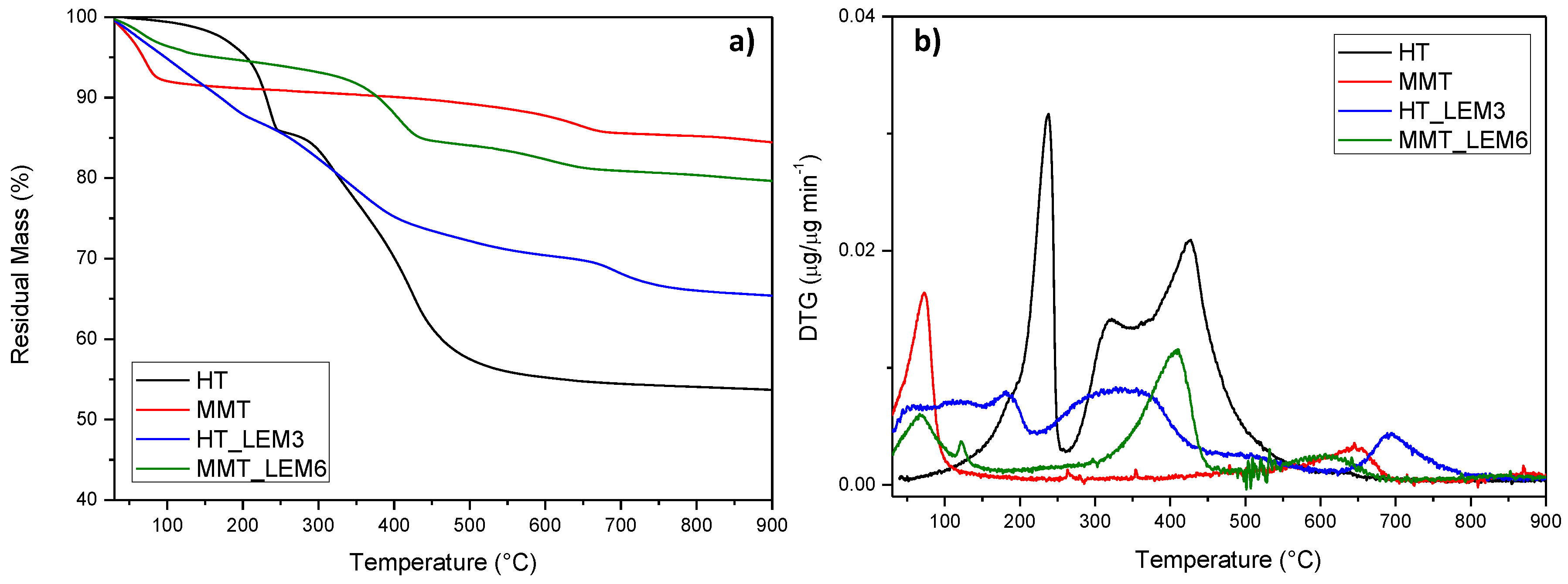
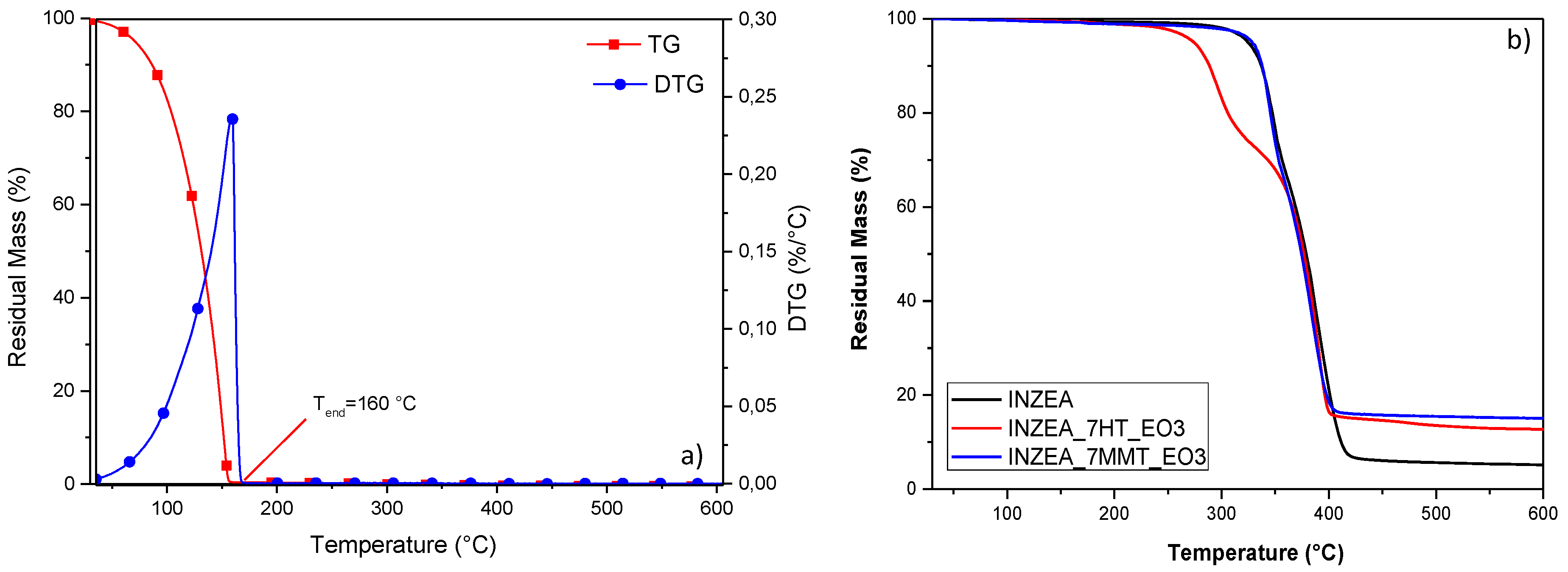
Thermal characterization of INZEA-based formulations containing the lemon hybrid pigments was also performed, by considering the overall stability of the pigmented nanoclays during the heating occurring in the extrusion phase. TGA analysis in nitrogen atmosphere of MMT_LEM and HT_LEM samples was performed and the resulting TG (a) and DTG (b) curves are presented in
Figure 5. In particular, TGA for neat nanoclays and hybrid materials were reported. According to the literature, the first step observed in the TGA curves, up to 200° C, was attributed to the desorption of water molecules from MMT’s pores (non constitutional water) and between the layers. A further weight loss above 400 °C was attributed to the dihydroxylation of the remaining OH groups of MMT. The final residue corresponded to the thermal degradation of the phyllosilicate into an amorphous phase [
53]. In the case of HT, the weight loss curve can be divided into three main thermal steps. The first one was associated with the loss of hydration water occurring below 200 °C, followed by the loss of hydroxyls between 200 and 600 °C [
54]. Since natural dyes are susceptible to temperature, the evaluation of their thermal stability is an important issue to be considered for their correct functional use in thermally processed materials.
Hybrid systems showed a different thermal behavior according to the chemical nature of the nanoclay used [
55,
56]. In the case of MMT_LEM, the hybrid compound exhibited a thermal behavior consisting of the dehydration of the compound (loss of water adsorbed and intercalated into the MMT), followed by a weak shoulder event centered at 122 °C). After that, the decomposition of the organic dye occurred, starting at about 260 °C and completing at nearly 460 °C. Nevertheless, the overall stability of the modified MMT was maintained, at least in the temperature frame of the polyester-based matrix selected for the present study [
57]. On the other hand, the presence of the lemon dye between HT layers significantly affected the thermal behavior of the nanoclay in the lower temperature range, since a distinct weight loss was observed (with a residual weight at 200 °C of 87% and 95%, respectively, for HT_LEM and unmodified HT). So, even if HT was thermally stabilized in the presence of the dye (residual mass at 900 °C was 65% and 54%, respectively, for HT_LEM and unmodified HT), it was easily degradable at the processing temperature range of the selected matrix [
16].
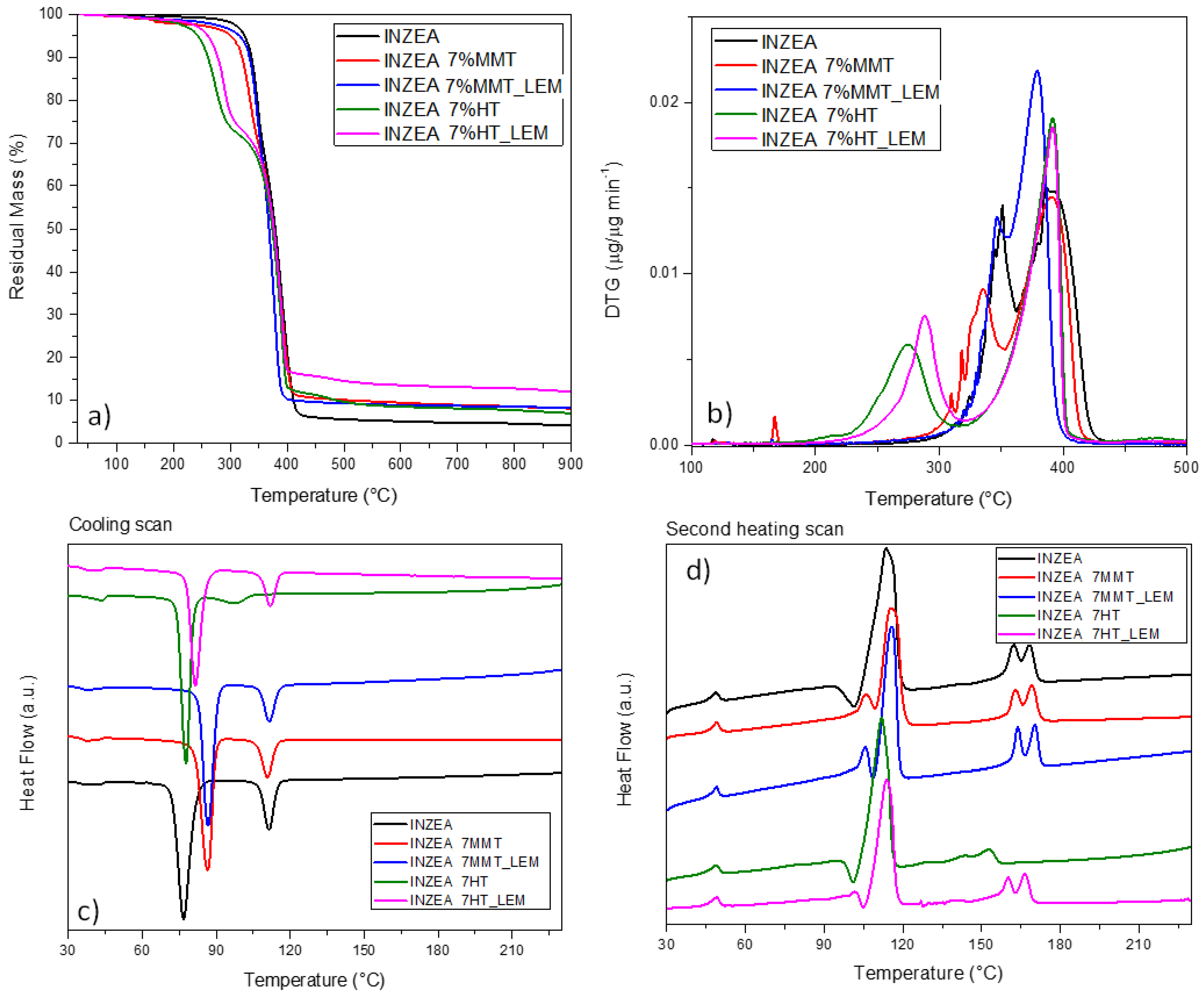
The observed behavior for lemon hybrid pigments greatly influenced also the response of INZEA-based systems. In particular, the curves related to mass loss (a) and derivative weight loss (b) for the system containing 7 wt% of nanopigments (representative of the same trend observed for the formulations containing 3 and 5 wt% of nanopigments) are reported in
Figure 6a,b. As it has been reported [
58], a double degradation peak can be found for the INZEA neat matrix, with two main steps centered at 350 and 400 °C, that could match with the possible degradation temperatures of PLA and poly(butylene succinate) (PBS) polyesters, respectively. It is also important to note that at 900 °C, unmodified INZEA matrix maintained a residual mass of ca. 5 wt%, which was increased when the nanofillers were introduced. This behavior is in accordance with the possible presence of an inorganic filler initially in the formulation of the commercial product.
A deep analysis of the DTG curves for the systems containing pure HT or MMT and the lemon hybrid pigments clearly indicated that, in the case of HT, the thermal stability of the overall blend was strongly affected. Specifically, while the Tpeak2 remained substantially unaffected (only a small variation in the degradation rate was noted), the Tpeak1 for the less stable component was shifted from 350 °C for neat INZEA to 287 and 275 °C, respectively, for INZEA 7%HT_and INZEA 7%HT_LEM. An analogous behavior, even if limited in the shift values, was noted for MMT containing materials, where Tpeak1 for the less stable component shifted to 346 and 335 °C, respectively, for INZEA 7%MMT and INZEA 7%MMT_LEM. In the case of MMT loading, a shift towards lower temperatures was even measured for the more stable component of the blend (from 392 to 379 °C for INZEA 7%MMT_LEM).
In accordance with the literature, this degradation path, differing from that of the neat matrix, can be rationalized by considering the organic nature of the nanopigments, that were indeed responsible of the decrease in thermal stability of the polyester matrix [
36]. The reduction in the thermal stability in the first step of the decomposition process of INZEA can be justified by the fact that unmodified nanoclay could catalyze the hydrolysis reaction, MMT platelets having –OH moieties on the surface promoting degradation [
59] and organo-modified Al layered double hydroxide (LDH) for the presence of moisture in the interlayer spacing [
60].
As previously reported by Wei et al. [
61], the presence of residual metals, such as Al, Fe, Zn and Sn, in LDH could cause an apparent thermal degradation of the polyester component, catalyzing the inter- and intra-molecular transesterification reactions of the polyesters. On the other hand, the decrease in thermal stability can be also explained due to the barrier properties of the nanoclay, which depend on the level of exfoliation. Only when the clay layers are exfoliated, they may have reduced the volatilization of the degradation products, and then, the thermal stability could increase [
51,
62]. In our case, the observed limited mechanical enhancement, due to reduced exfoliation of the nanoclays, can also justify the reduced thermal stability of the same samples.
Figure 6c,d shows the DSC thermograms obtained during the DSC cooling and second heating scans for all formulations. Two different peaks were observed for neat INZEA, indicating the presence of two main polyesters in the polymer matrix, in agreement with the behavior previously observed by Ramos et al [
58]. The analysis of the experimental curves indicated that no substantial variations were introduced in the glass transition temperatures, while the main effect was related to the shift of melting and crystallization temperatures, in parallel with the evidence or disappearance of multiple melting peaks. In detail, MMT and HT differently affected the processes: while INZEA MMT and INZEA MMT_LEM hybrid materials both modified the crystallization event, by shifting the T
c of the low melting polyester to higher temperatures, the effect on the high melting polyester was not visible. It can be considered that MMT could be located and better dispersed in the PBS phase than in the polylactic fraction, due to the higher interaction between PBS and MMT than that between PLA and MMT [
63], being the modification of the melt crystallization event evident only in the PBS fraction. This effect was also reflected in the heat flow curve during the second heating (
Figure 6d), where the cold crystallization was progressively inhibited and a double melting peak appeared, confirming the formation of crystals with different thickness, connected with a minute fraction of thinner and/or less perfect crystals originated during cooling and melting of the originally crystallized material [
64]. No valuable variations, in terms of temperature and curve shape, were noted in the case of INZEA_7%MMT_LEM.
On the other hand, it was found that the presence of nanopigments intercalated in HT was beneficial to the cooling and melting behavior of the PLA fraction. In detail, while INZEA_7%HT_LEM substantially maintained the same crystallization/melting attitude of the neat polyester blend, the unmodified HT was responsible for a general decrease in melting temperature in the polylactic fraction. It is well known that when the melting endotherm of neat PLA is bimodal, it can be ascribed to the formation of crystals with different degrees of perfection: the first endothermic is related to the melting of cold crystallized PLA chains, whereas the second peak can be attributed to those PLA crystals which have reorganized to more perfect crystals once they found sufficient thermal energy in the course of cold crystallization. In our case, this evidence can be correlated to the limited activity, as nucleating agent, of the HT reinforcement in the polyester blend and to the reduction in PLA molecular weight, due to the presence of surface hydroxyl groups on LDH and/or metal-catalyzed degradation [
65,
66].
3.3.2. Bionanocomposites with Lemon Hybrid EOs
Analogously to the thermomechanical characterization of INZEA formulations containing lemon hybrid pigments, the evaluation of the main parameters of lemon-hybrid EO containing samples was also performed. Specifically, the role of LEO, at the best synthesis conditions previously identified (100 wt%, 10 wt% surfactant concentration) when it was introduced in the polymer matrix was further investigated. Results of tensile characterization of the produced INZEA-based samples are reported in
Table 8. A significant increase in the Young’s modulus of the nanocomposite materials was observed compared to the biopolymer matrix. In particular, the incorporation of 7 wt% of MMT added with LEO (INZEA_7MMT.EO3) showed an average value of 2249 MPa significantly higher (+64.8%) than the neat matrix, that showed a value of E = 1365 MPa. A similar result was observed for the formulation with 7 wt% of HT and LEO (INZEA_7HT.EO.3) which increased the modulus to 2193 MPa (+60.7%). The increase of stiffness in the nanocomposites due to the addition of nanoclays has been widely demonstrated [
67]. This increase in the elastic modulus corresponded to a considerable decrease in deformability for both formulations, reducing the deformation at break of the nanocomposites with MMT and HT to 10 and 8%, respectively. This result is indicative of a poor interface bonding between the nanofillers and the matrix, probably due to the scarce compatibility between the biopolymer and the nanoparticles added with LEO. It was demonstrated that the compatibility between a hydrophobic matrix and nanofillers such as MMT and HT is often poor and can be improved through the use of compatibilizers that mitigate the hydrophilic nature of the nanoclays [
68]. It was hypothesized that the presence of essential oils and nanofillers could have two antagonistic effects: on the one hand, EO linked to the filler could mitigate the hydrophilicity of the nanoclays by improving matrix/filler compatibility; on the other hand, the presence of free essential oils in the formulation could reduce the mechanical strength of the composites by acting as a plasticizer and intermolecular lubricant [
69]. The results show that both the composites with essential oils increase their moduli with respect to the corresponding nanocomposites without EO, suggesting that an improvement of compatibility matrix/filler occurs. In addition, a slight increase in elongation without strength reduction suggests that free essential oils are almost completely absent. The overlap of these effects does not produce substantial changes in the mechanical tensile characteristics of composites with and without EO, except for an increase in the elastic modulus of composites with added EO. It can be seen that the presence of 7 wt% of nanofiller limits the deformability of the composites. The reduction in maximum elongation with respect to the matrix, for both nanocomposites, was responsible of a reduction in both the yield stress and strain at break.
The limited, but superior behavior of INZEA_7MMT.EO.3 in terms of deformability, compared to the HT-loaded system, can be justified by considering that the EO containing nanofillers underwent a thermal degradation event when heated during extrusion. In order to justify the narrow effect of the EO in terms of improved ductility, the release of the added EO during a dynamic heating scan, in a TGA inert environment, for INZEA_7MMT.EO.3 and INZEA_7HT.EO.3 was performed. The results of dynamic heating scans are included in
Figure 7a,b. In particular, the results of
Figure 7a confirmed that the heat stability of the EO was quite limited (the evaporation was completed at 160 °C), but its inclusion into the nanoclays efficiently retained the observed loss during heating of the polyester matrix, especially in the case of MMT (
Figure 7b).
In the case of MMT samples functionalized with 300 wt% LEO, the presence of EO.WC.T50 and EO.WC.T100 in INZEA matrix was determined and evaluated, in terms of mechanical performance, by means of tensile tests.
Table 9 summarizes the results of mechanical properties obtained for INZEA-based materials in the presence of different fillers (EO.WC.T50 and EO.WC.T100). A clear difference in terms of mechanical properties (decrease in stress and strain (%) at break and improvement in Young’s modulus) was observed. Specifically, the addition of the filler changed completely the mechanical response of the different samples. Neat INZEA showed a ductile behavior which was turned into a fragile behavior in the case of the presence of the nanofillers. A similar behavior was detected in PLA/MMT-based formulations by Othman and co-authors who observed that a high concentration of MMT in PLA changed the ductility of the polyester [
70]. No particular variations were observed between the different INZEA+7%EO.WC.T50 and INZEA+7%EO.WC.T100-based materials. This phenomenon highlights that the presence of LEO at the different concentrations used resulted ineffective in terms of improving ductility, but only an increase in stiffness was revealed.
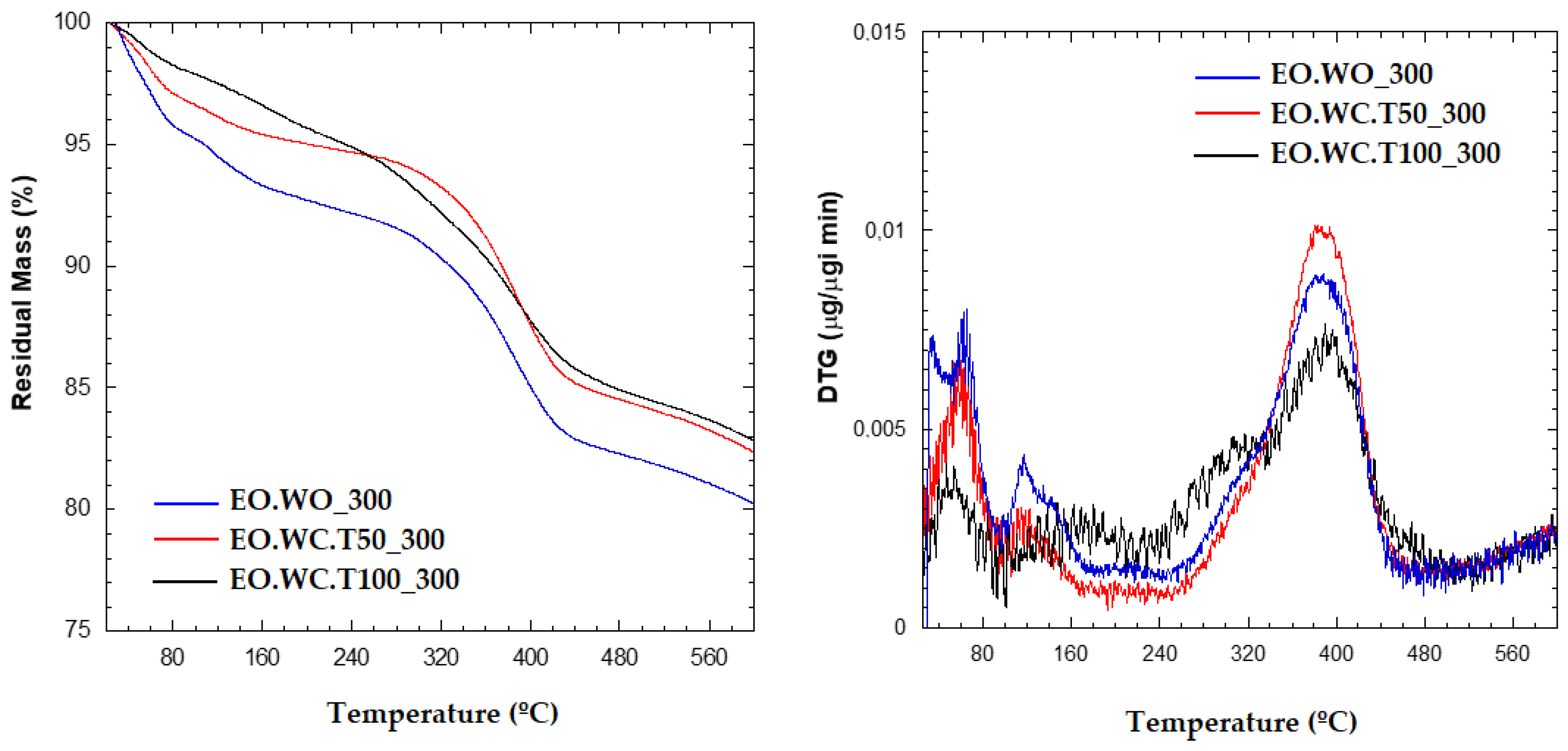
With the aim of evaluating the presence of LEO in the polymer matrix, INZEA+7%EO_W0_300, INZEA+7%EO_WC.T50_300, and INZEA+7%EO_WC.T100_300 samples were tested. First, the thermal stability of the obtained samples was studied by TGA (
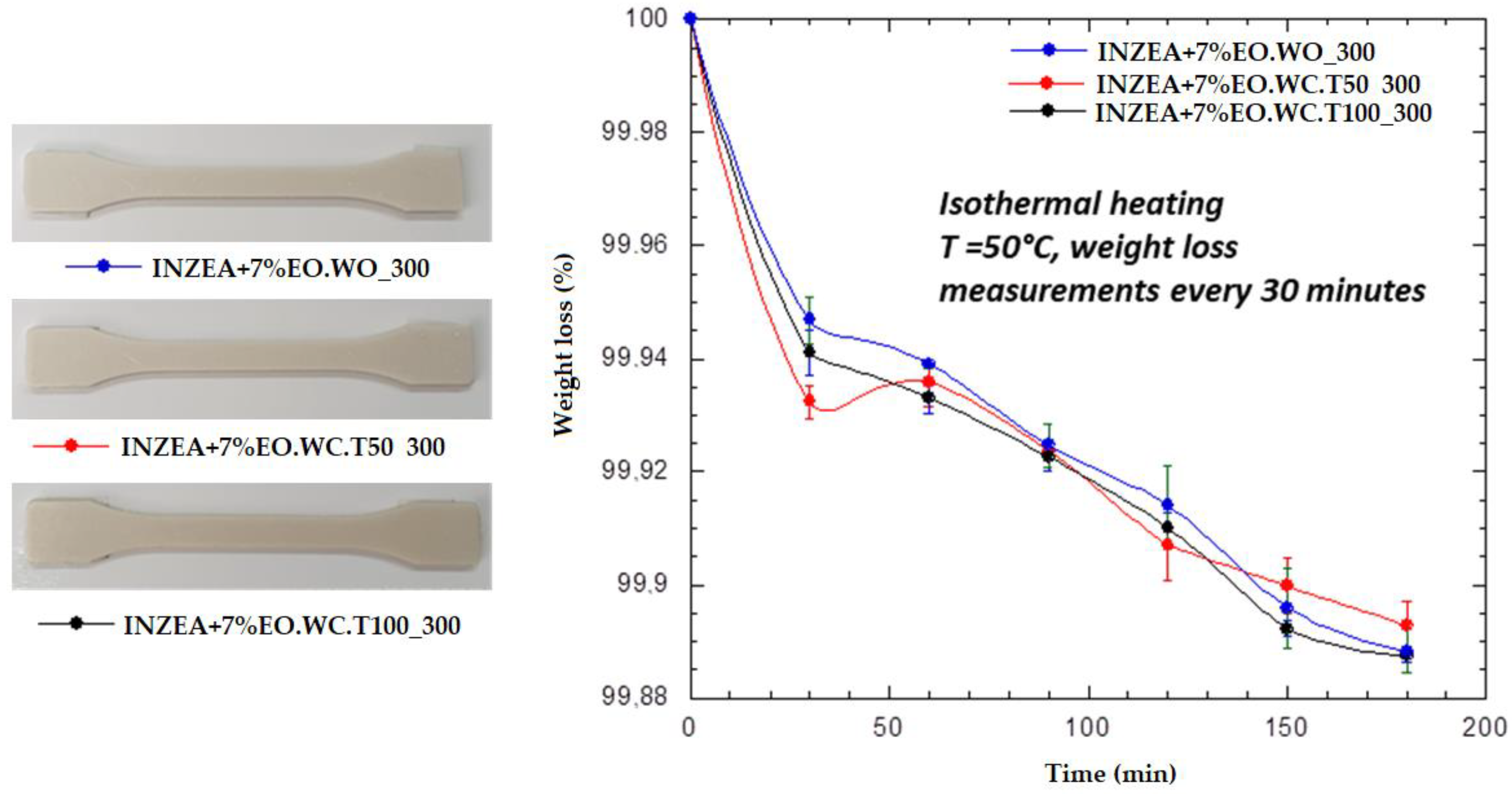
Figure 8); sample EO.WC_T100_300 exhibited the lower weight loss up to 200 °C, being the more thermally stable in the entire temperature range. The release of LEO during thermal processing was also verified by heating the bio-nanocomposites by TGA isothermally at 50 °C. Weight measurements were taken every 30 min and the obtained results (
Figure 9) indicated quite similar kinetics for LEO release in all samples, although these tests should be run for a longer time. Therefore, the obtained results for the developed LEO hybrid systems with MMT confirmed that LEO release during heating was better controlled in the case of sample EO_WC_T100_300, which was also the one showing a higher final lemon smell compared to the other ones.