1. Introduction
Non-invasive and fast measurement techniques are of interest for investigations in the life sciences, i.e., for medical diagnostics and tissue implant characterisation. Optical technologies have already indicated their application potential in these fields. In general, the assessment of tissue properties or disorders requires the reliable detection of subtle changes in tissue structure to be able to initiate measures for treatment in case of diseases. For example, inflammatory skin lesions occur in a broad variety of skin disorders with different pathogenesis. Typical hallmarks of clinically inflamed skin are redness, edema or induration (i.e., hardening and thickening) and scaling. In clinically uncertain cases, taking a skin biopsy for histopathological analysis is usually indicated. Conventional hand-held dermatoscopes serve as essential non-invasive diagnostic tools for evaluating pigmented lesions, but are currently not suitable for further examination of inflamed skin. Indeed, flattening of the skin lesion while using the conventional dermatoscope leads to changes or even reduction of the blood circulation and finally affects the disease-related color of the corresponding skin site. To avoid direct skin contact during examination we developed a prototype for a non-contact, remote digital dermatoscope in previous work [
1,
2]. In an initial study we attempted to visualise skin inflammation of common inflammatory skin diseases such as psoriasis and lichen planus by non-contact remote digital dermoscopy [
3]. We revealed promising first results regarding a visualisation of inflammatory changes within epidermal, i.e., upper, layers of the skin by non-contact remote dermoscopy. It was possible to visualise disease-specific changes within the epidermis like an expansion of the granular layer in lichen planus. Simultaneously, the system generated true-color images of the skin lesion [
3].
However, it remains a challenge to explore changes located deeper than the epidermis as relevant for inflammatory diseases. In patients with sclerodermia circumscripta and granuloma anulare, using the prototype it was challenging to generate optical signals reflecting a distinct disease-related pattern [
3]. In patients with sclerodermia, affected skin lesions are clinically characterized by fibrosis and sclerosis, resulting in a hardening of the skin and hair loss because of a destruction of the hair follicles. Histopathology in sclerodermia circumscripta is characterized by a deposition of collagen in the dermis [
4] and an edematous formation of collagen. Thus, for sufficient non-invasive examination of inflammatory skin disorders further development of non-contact remote digital dermatoscopy is required as they are located deeper in the skin with involvement of collagen formation, such as in patients with sclerodermia.
To generate additional diagnostically relevant information, different techniques are in principle possible for non-contact measurement. Common dermatoscopes are designed for 2D imaging based on digital imaging of the skin by using a charge-coupled device (CCD) or complementary metal–oxide–semiconductor (CMOS) chip. This technique provides spatially resolved spectral information. State-of-the-art camera devices have three different color channels. To generate further information, in theory the use of hyperspectral cameras with more color channels would be possible [
5]. Additionally, optical coherence tomography (OCT) is a common technique for skin investigation with an imaging depth of 2–3 mm in most biological tissues and axial resolution between 1 and 15 µm [
6]. However, the OCT signal is based only on the backscattering intensity from different depths and provides no spectral information [
6]. Ultrasound also enables depth information based on the intensity of measured waves. In this case acoustic waves are used, and the depth is defined by the time the wave propagates in the target. The imaging depth is inversely proportional to the lateral resolution and the required frequency. In muscle tissue, for example, the lateral resolution can be as low as 40 µm [
7]. Another approach to gain more information of possible diagnostic relevance is to use polarized light. Typically, a cross polarization setup is realized, where the sample is illuminated with linearly polarised light. Only light with polarisation perpendicular to the illumination is detected. Cross polarisation is used to suppress surface-reflected signals and, therefore, reveal information on structures below the surface. Even more information could be obtained by measuring the Mueller matrix (MM) of the target [
8]. Fricke et al. devised a non-destructive, cost-efficient and contactless quantification method for the degree of fibre alignment in electrospun polycaprolactone (PCL) fibre scaffolds, based on Mueller matrix (MM) polarimetry [
9].
Motivated by the described limitations of current approaches and to meet demands from dermatology, in this work, we investigate the potential of MM-measurement to obtain information about orientations within collagen and gelatin containing structures. These were chosen as simple tissue phantoms for our initial proof-of-principle investigations, the latter being realised by an electrospinning process, which is a versatile and scalable manufacturing technique ensuring high throughput and cost-efficiency. The method is widely used in tissue engineering (TE), to fabricate porous scaffolds based on fibres with diameters ranging from several hundred nanometers to a few microns. The results achieved indicate that the MM measurement provides valuable insights on tissue parameters, which are independent from fabrication- and application-based factors like scaffold thickness, fibre diameter or polymer type. Furthermore, in order to investigate the influence of the electrospinning process on the proteins in the blended solution and to confirm the collagen content in the transparent films, Raman spectroscopy was used. In the future, based on the results obtained here, we will investigate the suitability of our approach for skin diagnostics. In particular, we aim at differential diagnostics of inflammatory skin diseases in vivo.
2. Materials and Methods
2.1. Electrospun PCL: Gelatin Fibre Scaffolds
In general, an electrospinning setup consists of an emitter, a high voltage supply and a grounded collector (see
Figure 1). An initial droplet of polymeric solution is vertically emitted into an electrical field. As soon as the forces, induced by the electrical field and gravity, overcome the surface tension of the fluid, a fibre jet is emitted. On its way to the grounded collector, the fibre is constantly accelerated and the solvent evaporates. Upon deposition on the collector, the fibre diameter is constantly decreasing. The manufactured fibre scaffolds are strongly influenced by the ambient, process and solution parameters.
In tissue engineering an important task is to combine manufactured scaffolds, mimicking the extra cellular matrix (ECM), with cells and signaling factors. In the case of artificial skin substitutes, a variety of polymers have successfully been used as scaffold material [
10]. The recreation of the ECM calls for specifically oriented fibres. The fibre orientation within the manufactured scaffold is mostly influenced by the relative velocity of the collector; the higher the relative collector velocity, the higher the degree of fibre alignment [
9]. While this is not expected to be a linear relation, we anticipate an increase of the degree of alignment until a certain threshold and a stagnating or decreasing behavior after passing of this mark.
2.1.1. Processing System
We used an electrospinning device according to Fricke et al. that consists of a drum collector, an electric motor, a blunt cannula, polyethylene tubing, a syringe and a syringe pump (see
Supplementary Materials Figure S1) [
9,
11,
12,
13].
2.1.2. Parameter Settings and Experimental Procedure
The fibre scaffolds were fabricated from a blended polymeric solution. The blends consisted of polycaprolactone (PCL, 80 kDa, Sigma-Aldrich Chemistry Corporate, St. Louis, MI, USA) and gelatin from bovine skin (Type B, ~225 g Bloom, Sigma-Aldrich Chemistry Corporate, St. Louis, MI, USA) in 2,2,2-trifluoroethanol (TFE, 99.8%, abcr GmbH, Karlsruhe, Germany) [
14]. In order to enhance the solubility of gelatin in TFE, 2 v% of acetic acid were added. The mass ratio of PCL to gelatin was 5:2, resulting in 125 mg/mL PCL/TFE and 50 mg/mL gelatin/TFE. The polymeric solution preparation was conducted as previously published by Suresh et al. [
14]. Three different fibre scaffolds were manufactured for each of the 13 relative collector velocities of 1.2 to 10.7 m/s at a total process duration of 25 min each (see
Figure 1) [
9,
14]. An additional reference sample for the spectrographic analysis was fabricated from a 175 mg/mL PCL/TFE solution at a relative collector velocity of 2 m/s.
2.2. Collagen Film
The sample used was Viscofan Coffi Folie (Viscofan, Cáseda, Spain). It basically consists (in mass percent) of collagen (59–63.7%), water (15%), glycerin and Sorbitol (14–16%) and oil (4%) [
15]. The product is supplied on a 38 cm wide roll of 50 m length. The film has a thickness of 0.04 mm.
2.3. Raman Spectroscopy
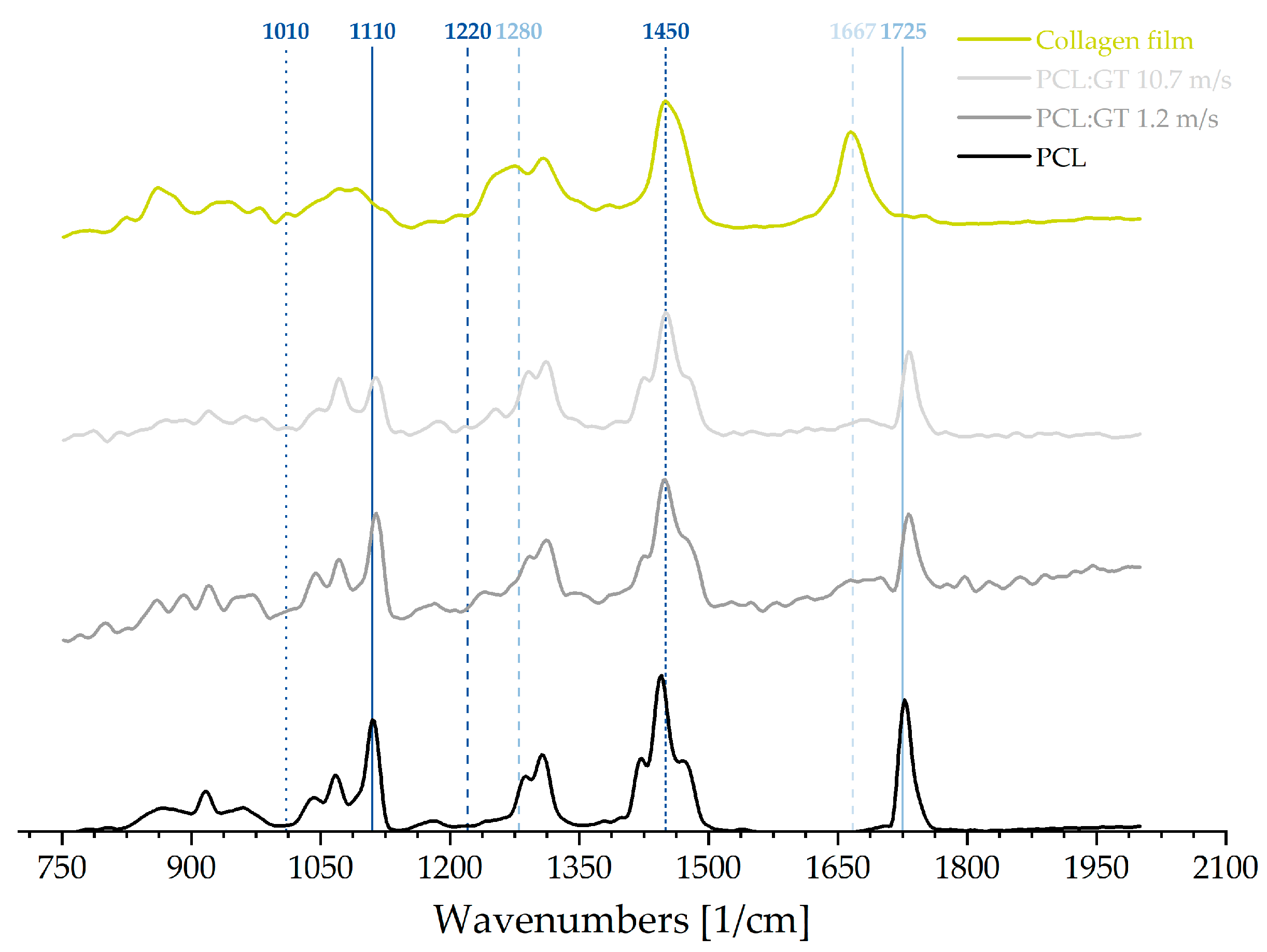
In order to assess the molecular structure and content of specific proteins as well as possible changes (manufacturing and handling), Raman spectroscopy (Alpha 300 RA, Witec GmbH, Ulm, Germany) was performed. The Raman spectrograph contained a laser with 532 nm wavelength and laser power adjusted to 30 mW and a CCD detector for spectra detection. It had an NA of 0.9 and spectral resolution down to 0.1 relative wavenumbers (at 633 nm). The integration time was set to 0.7 s as well as the accumulation to 50. The samples employed for the measurements were prepared from a collagen film, a pure PCL fibre scaffold and PCL:GT scaffolds fabricated with 1.2 m/s and 10.7 m/s relative collector velocity. Afterwards, the resulting spectra were normalized to the CH band intensity at 2923 cm−1. and compared.
2.4. SEM-Based Analysis
According to Fricke et al., SEM (S-3400N, Hitachi High-Tech Analytical Science Ltd., Tubney Woods, Abington, UK) images of sputter coated samples were taken.
2.4.1. Fibre Scaffolds
The degree of fibre alignment was determined, based on nine images, three for each of the three samples per relative collector velocity. Subsequently, fibre orientation was measured by software analysis (AxioVision
®, Carl Zeiss AG, Jena, Germany) [
9].
2.4.2. Collagen Film
Six pictures were taken, three of the upper and three of the lower side of the film. These images were investigated for potential fibre alignment and overall structure.
2.5. Mueller Matrix Measurement System
The Mueller matrix (MM) contains information about the polarisation changing properties of a sample. It can be calculated using intensity measurements where the sample investigated is illuminated with light of different polarisation states. The intensity for different polarisation states of the light after interaction with the sample is measured. The MM measurement system generates these states (with a polarisation state generator (PSG)) and filters the states of interest before sensing (with a polarisation state analyser (PSA)). As the sensor is a camera, 2D-measurements of the MM over a sample surface can be obtained. The MM measurement system used in this work was a self-built lab prototype described in detail in [
9]. To calculate the full MM, 36 measurements of different combinations of illuminating and measured light had to be performed. The device generates and measures horizontal (H), vertical (V) +45° (P), −45° (M), right circular (R) and left circular (L) polarisation states as required for MM calculation. The PSG and PSA consist of a fixed polarizer and two liquid crystal retarders (LCR). By changing the voltage applied to the LCR, all required polarisation states can be generated in a comparably short time of 15 s. Potentially, the measurement time can be decreased to below 1 s in the future, since the system is switched entirely electronically without moving any parts. The system operates in reflection and transmission mode. The illumination is monochromatic and can be switched between 445 nm, 532 nm and 633 nm. To interpret the results, for example, polar decomposition of the MM can be performed [
16]. The matrix is divided in three matrices of known physical properties.
2.6. Statistical Analysis
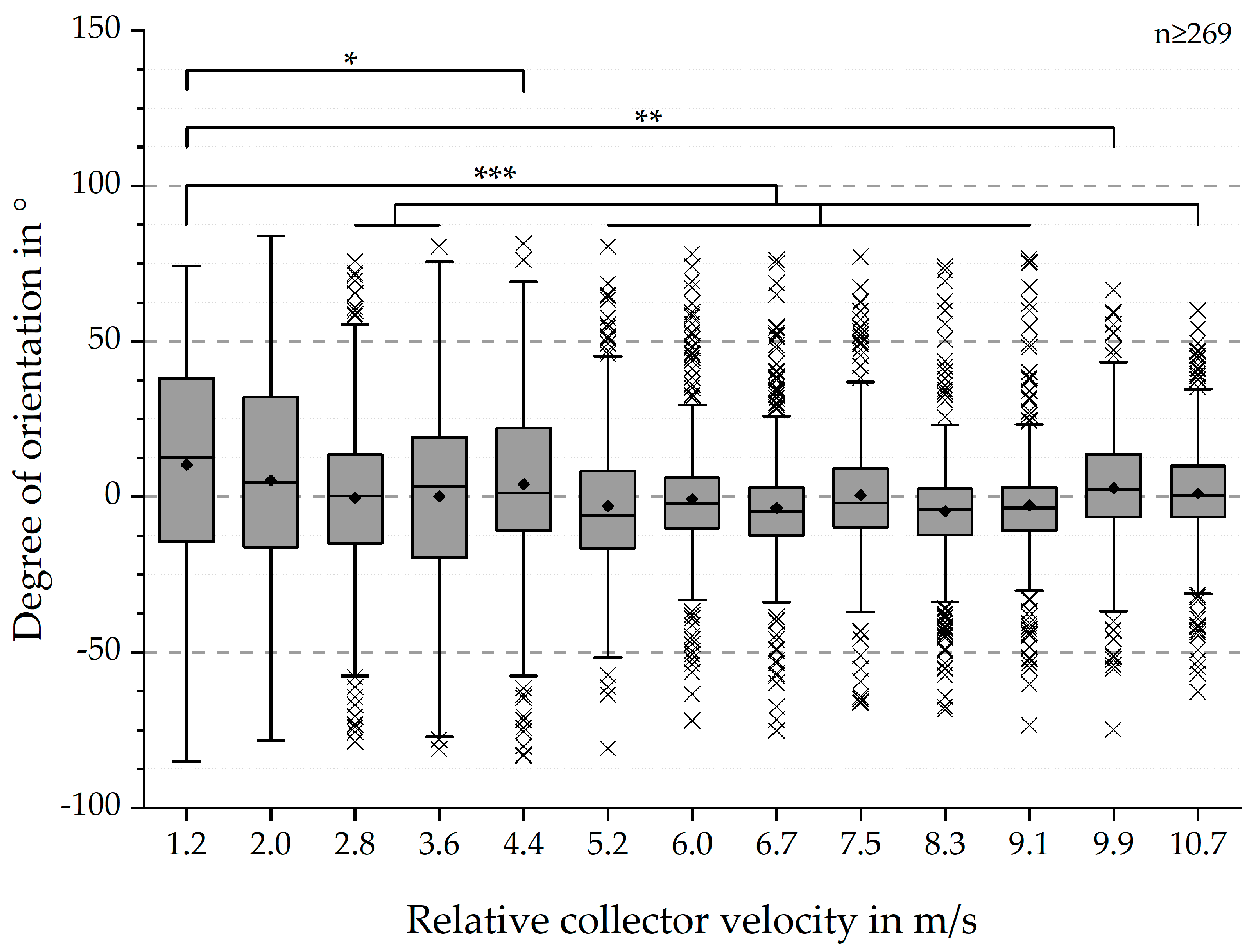
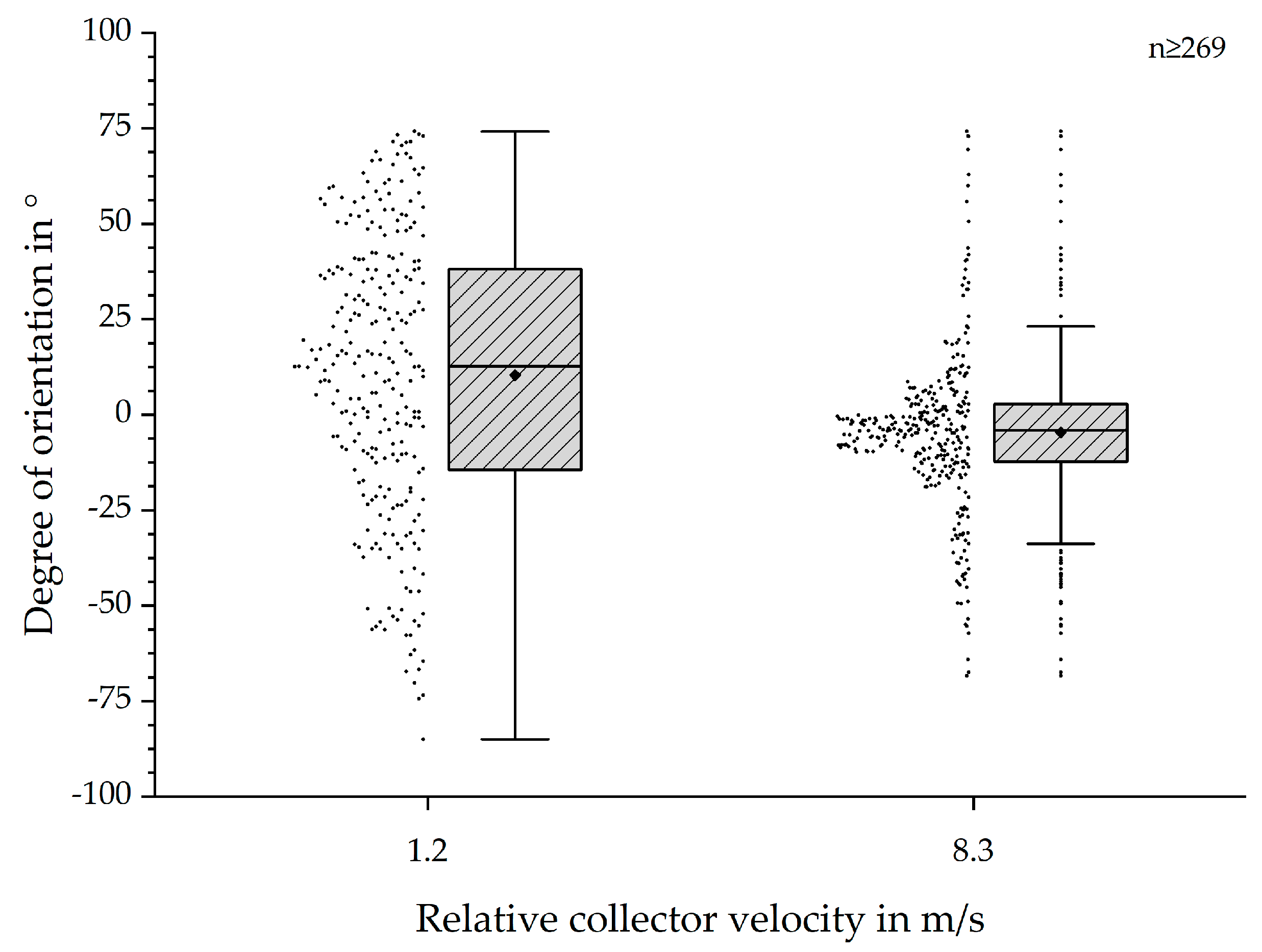
SEM-based results were expressed as boxplots with outliers or means ± standard deviation (SD), if not stated otherwise [
9]. In all boxplots, the interquartile range (IQR), represented by a closed box, was defined as 50% of the measured data. The line within the IQR was defined as the median value, while the displayed whiskers represented 1.5 times the IQR. Distance between whiskers was defined as the dispersion. The mean value was displayed as a rhombus. All values located outside of 1.5 × IQR were displayed as individual crosses. These values were “outliers” with regards to the IQR, but were not necessarily extreme values.
QQ-plots were generated to investigate the application of parametric or non-parametric tests. In order to determine differences between all groups, one-way repeated measures analysis of variance (ANOVA) was conducted. Subsequently, Tukey and Dunnett (reference: 1.2 m/s) post-hoc tests were performed to investigate differences between specific groups. Differences were considered significant at p < 0.05 (*), p < 0.01 (**) and p < 0.001 (***). All data were analysed using statistical analysis software (Origin 2018b, OriginLab Corporation, Northampton, Massachusetts, USA).
4. Discussion
We were able to point out, that a non-contact and thus non-destructive measurement of the orientation of collagen- and gelatin-containing samples is generally possible. The obtained spectral data showed distinct differences in molecular structure for the samples used in this study: PCL, PCL:GT (1.2 m/s and 10.7 m/s) and collagen film. The recorded spectrum for the collagen film indicated a strong presence of proteins, unchanged in their conformation. The appearance of the most prominent peak for collagen, located at 1667 cm
−1, indicates that the film indeed consisted of collagen and not of denaturized proteins. The comparison with the PCL:GT fibre scaffolds shows some similarities, probably caused by the fact that gelatin is derived from native collagen [
23]. Correspondingly, these similarities reveal that the gelatin is not altered by the electrospinning process. The still observable bands for amide II and III (1220 cm
−1 and 1280 cm
−1) distinguish the PCL:GT blends from the pure PCL. Furthermore, the characteristic band for PCL at 1110 cm
−1 changes with increasing the relative collector velocity from 1.2. m/s to 10.7 m/s and thus, indicating changes in the PCL:GT fibre scaffolds properties in comparison to pure PCL. Further analysis, both qualitative and especially quantitative, are necessary to determine the actual influence of the relative collector velocity on these bands and therefore fibre scaffold properties. However, a slight shift of the PCL band between 1725 cm
−1 and 1735 cm
−1 was observed in both PCL:GT spectra. Consequently, an effect on the ratio of amorphous to crystalline phases of the PCL in PCL:GT can be assumed [
19]. The qualitative Raman analysis illustrates an impact of gelatin on the PCL structure and the other way around, within the blended fibre scaffold.
The SEM-based analysis still bears several factors increasing errors with regards to the mean values of the fibre alignment (see
Figure 10) [
9]. However, the statistical analysis and presented boxplots (see
Figure 3 and
Figure 4) clearly indicate a significant increase of the degree of fibre orientation with increasing relative collector velocity. Correspondingly, the initially hypothesised threshold was reached around 8.3 m/s, represented by the smallest IQR for 8.3 m/s and 9.1 m/s. In spite of the smallest IQR for these relative collector velocities, the number of outliers is higher than for all other groups. However, the reason for this observation does probably not originate from the measurement method but from the statistical analysis. The definition of the whiskers is 1.5 times the IQR. Hence, closer distributions will lead to smaller dispersion and therefore apparently more outliers. So, there is no simple answer to the question whether these results represent statistical anomalies or the influence of the measurement protocol [
9]. Overall, these results are consistent to those obtained by Fricke et al. indicating an influence of the relative collector velocity, not being limited only to PCL fibre scaffolds. Further investigations have to be conducted to generate a calibration curve for generic materials.
Furthermore, the analysed fibres exhibited diameters up to 450% smaller than for pure PCL (see
Supplementary Materials). Nevertheless, the observed trend (see
Figure 10) with regard to degree of orientation in dependence of relative collector velocity, is similar to the one described by Fricke et al. [
9].
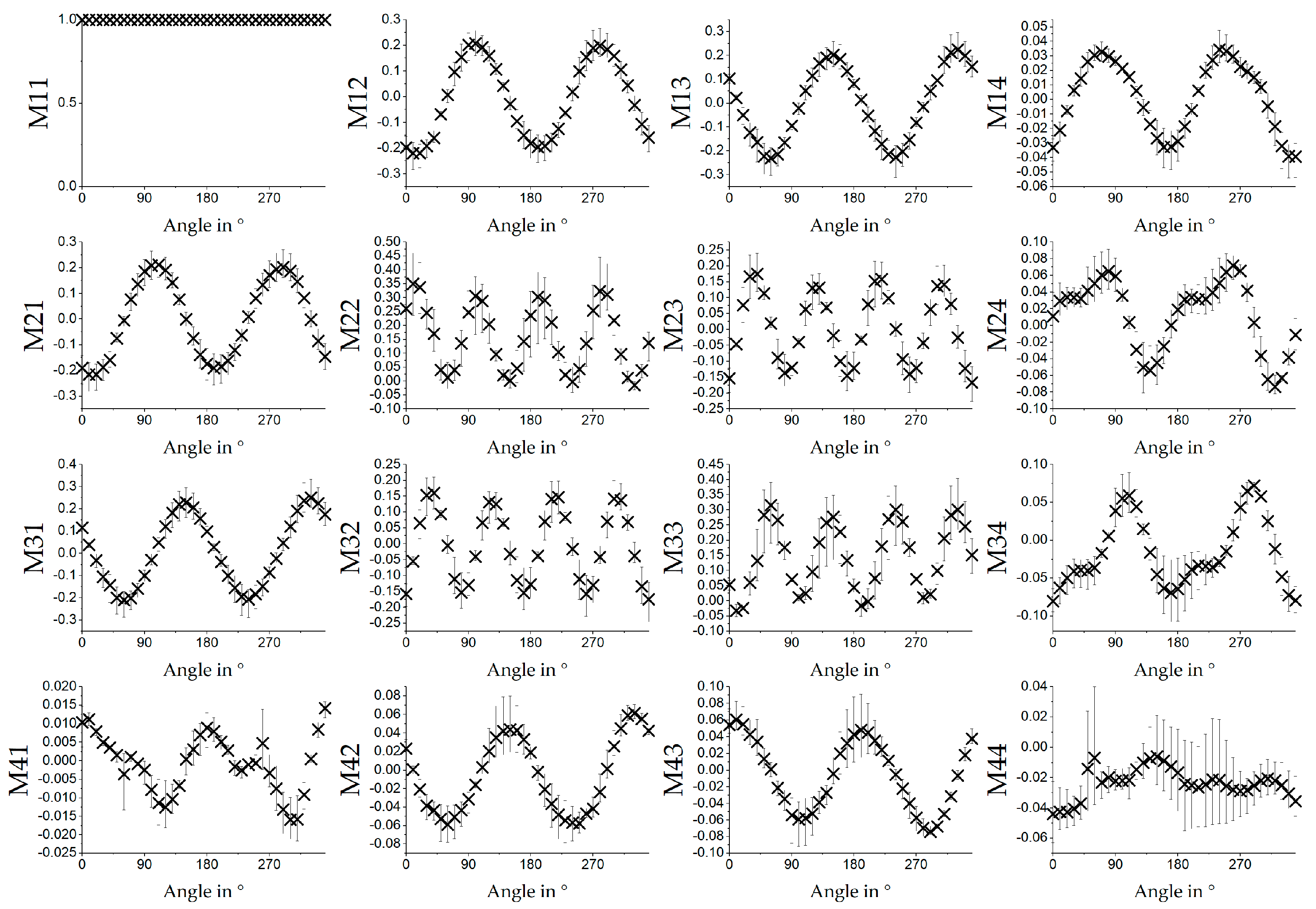
To demonstrate the potential of MM measurements for samples that are not electrospun, the results were compared with the measurements of a transparent collagen film. The correlation between the absolute orientation and polarizance P is clearly depicted in
Figure 10. The MM measurement of PCL:GT showed the dependence of MM entries on the orientation of the sample. Compared with previous results for PCL [
9], a significant difference can be seen. For PCL:GT, in addition, MM entries are sensitive to rotation of the sample, which are probably related to circular diattenuation. This may be explained by the chirality of the gelatin (applies also for collagen) [
24,
25,
26,
27] component while pure PCL has no chiral structure. While results for PCL:GT also show an angle dependence of the entries connected with linear retardation and diattenuation [
22], results for these entries are not as clear for the transparent collagen film. Here mainly MM entries that are connected to circular retardance [
22], were sensitive to rotation of the sample. For the PCL:GT entries like M24, M34 and M41 (see
Figure 6), the data seem to show a superposition of two signals. A reason for this could be the different optical properties of gelatin and PCL. Another explanation could be a bimodal distribution of the orientation of the fibres, which is less likely because no supporting observations were made during the measurements (see
Figure 4). Further calculation of the fast axis of the retardance show that the angle of the sample is strongly connected to the angle of the fast axis for the PCL:GT samples. Here, one MM measurement is sufficient to measure sample orientation. Measurement is more precise compared to previously shown results for PCL fibres [
9]. The angle of the fast axis is not directly connected to the orientation of the fibres as assumed. In particular, it was assumed that fibres are oriented in the direction of rotation of the collector (see
Figure 1). Because light can interact with electrons, which are capable of moving in direction of orientation of the molecules, the fast axis of retardation should be perpendicular to the fibre orientation. As fibres were oriented horizontally at 0°, the Stokes vector of the fast axis of retardation should be vertical. However, there is a difference of about 13°. This difference indicates that the fibres are not oriented in the direction of the rotation of the collector but are oriented with a certain angle due to a side movement during the spinning process. The side movement could be a result of an inhomogeneous electric field of the apparatus. However, the calculation is not suitable to calculate sample orientation of the transparent gelatin film. Here, the orientation of the sample can only be extracted if the amplitude of the signal for the angle dependent MM entries is known.
5. Conclusions
The performed MM measurements are a promising method to measure orientation of bio-polymer structures in skin phantoms or the skin itself. Results showed that angle dependence of the MM entries depend strongly on the samples investigated. That demonstrates the strength of the MM approach, which is suitable to measure any polarisation altering property of the sample. The orientation of PCL:GT samples can be measured with only one MM measurement by calculating the Stokes vector of the fast axis of retardation without prior knowledge of the amplitude of the signal. For the transparent collagen film, orientation can be obtained by using prior knowledge of the amplitude of the angle dependent signal in the MM entries M34 and M43. In order to apply this measurement method to different materials, the algorithms developed might have to be adjusted. Ongoing studies are investigating the power of other decomposing methods and further characterisation of collagen samples within one measurement.
The presented results of Raman spectroscopy, SEM-based analysis of the degree of fibre orientation and MM measurements strongly indicate the suitability of PCL:GT fibre scaffolds and collagen containing samples for calibration of skin disease detection and evaluation by measuring the orientation of given structures and the presence of chiral molecules, which manifest themselves by an angle dependent signal in certain MM entries.