Microstructure and Mechanical Properties of Carboxylated Nitrile Butadiene Rubber/Epoxy/XNBR-grafted Halloysite Nanotubes Nanocomposites
Abstract
1. Introduction
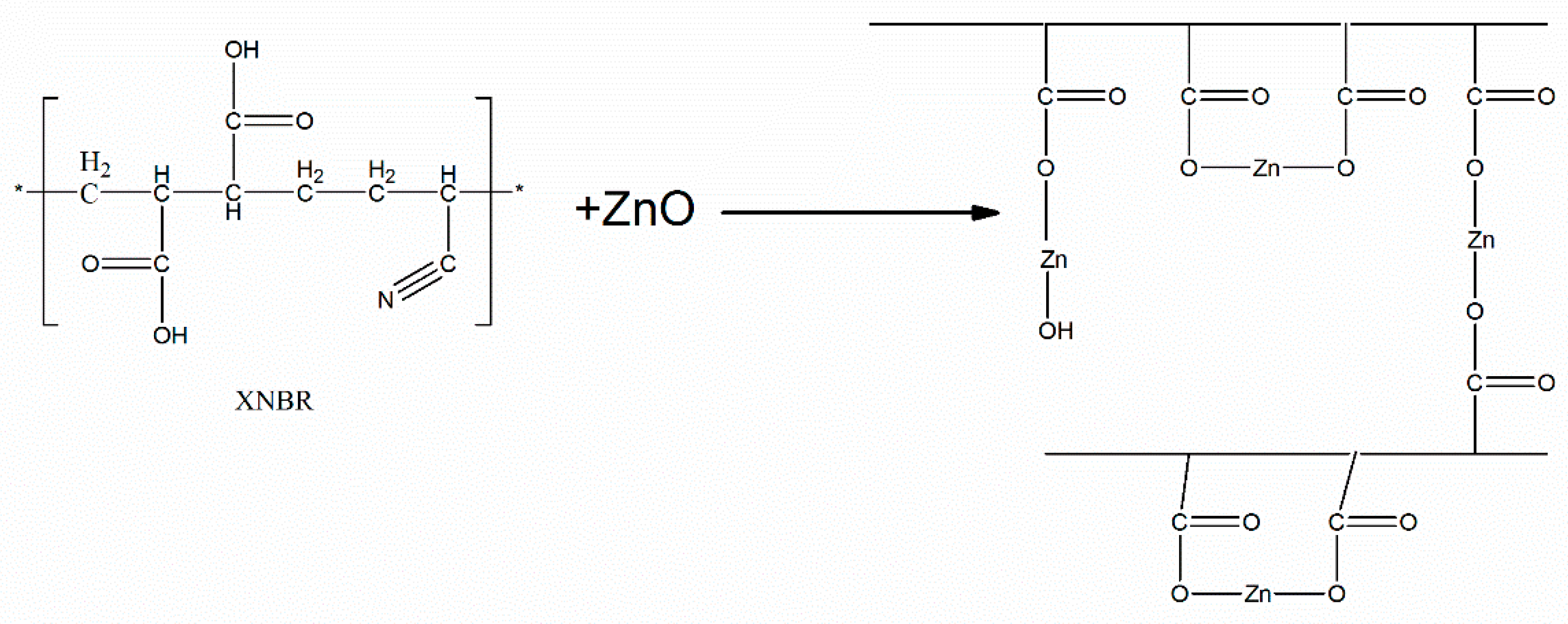
2. Theoretical Background
3. Experimental
3.1. Materials
3.2. Nanocomposite Preparation
3.3. Characterization
4. Results and Discussion
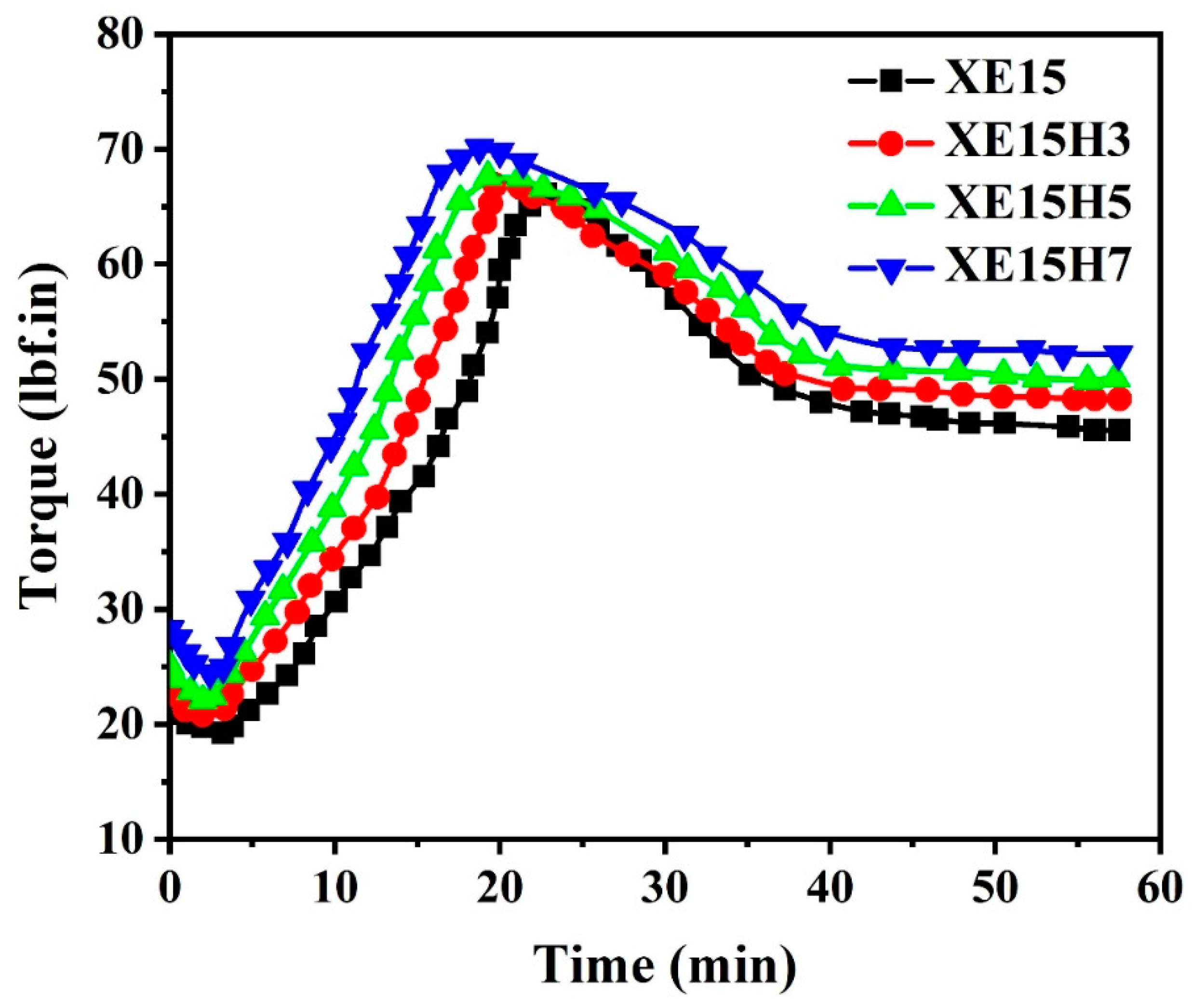
4.1. Cure Characteristics
4.2. Morphologic Observations
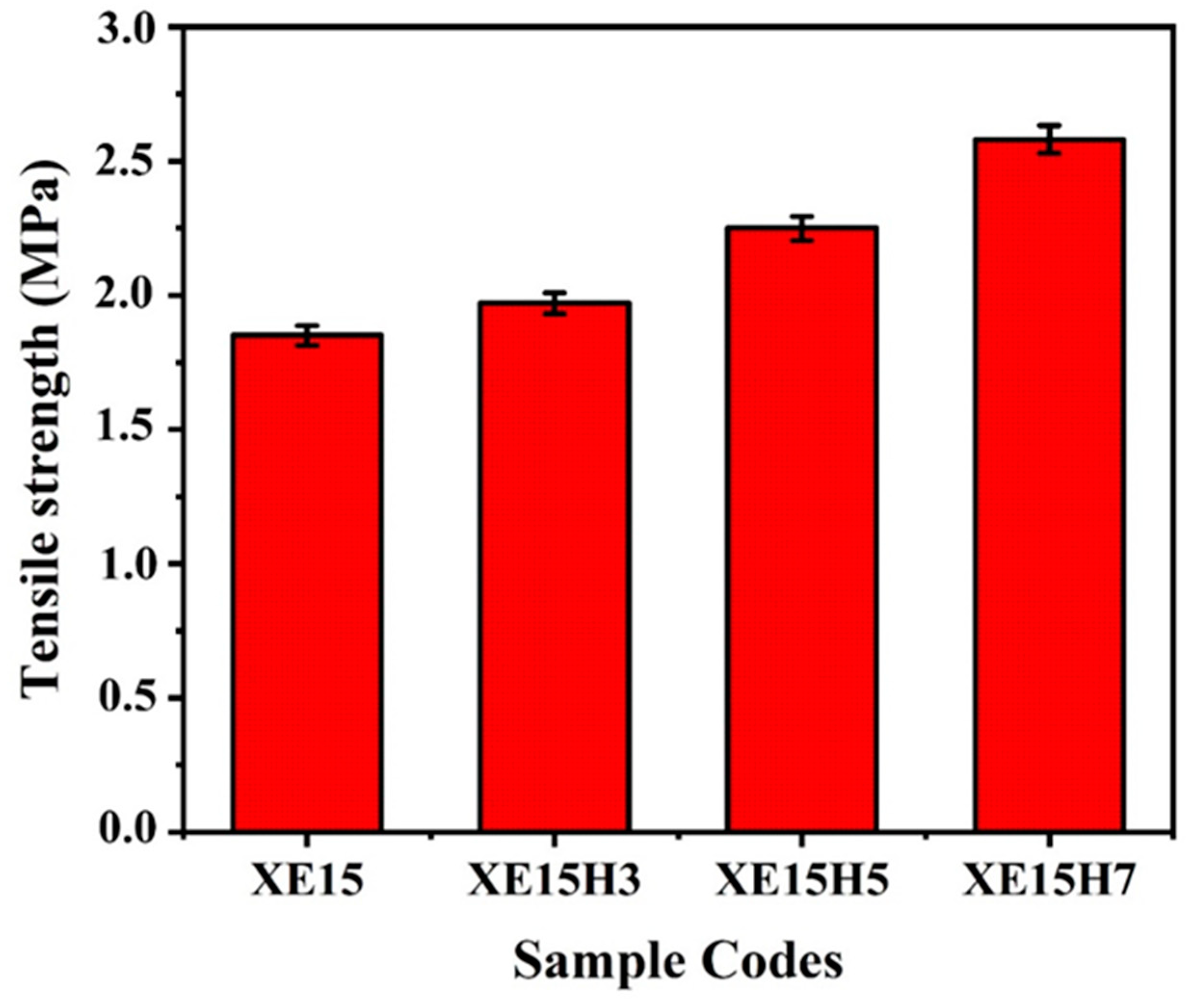
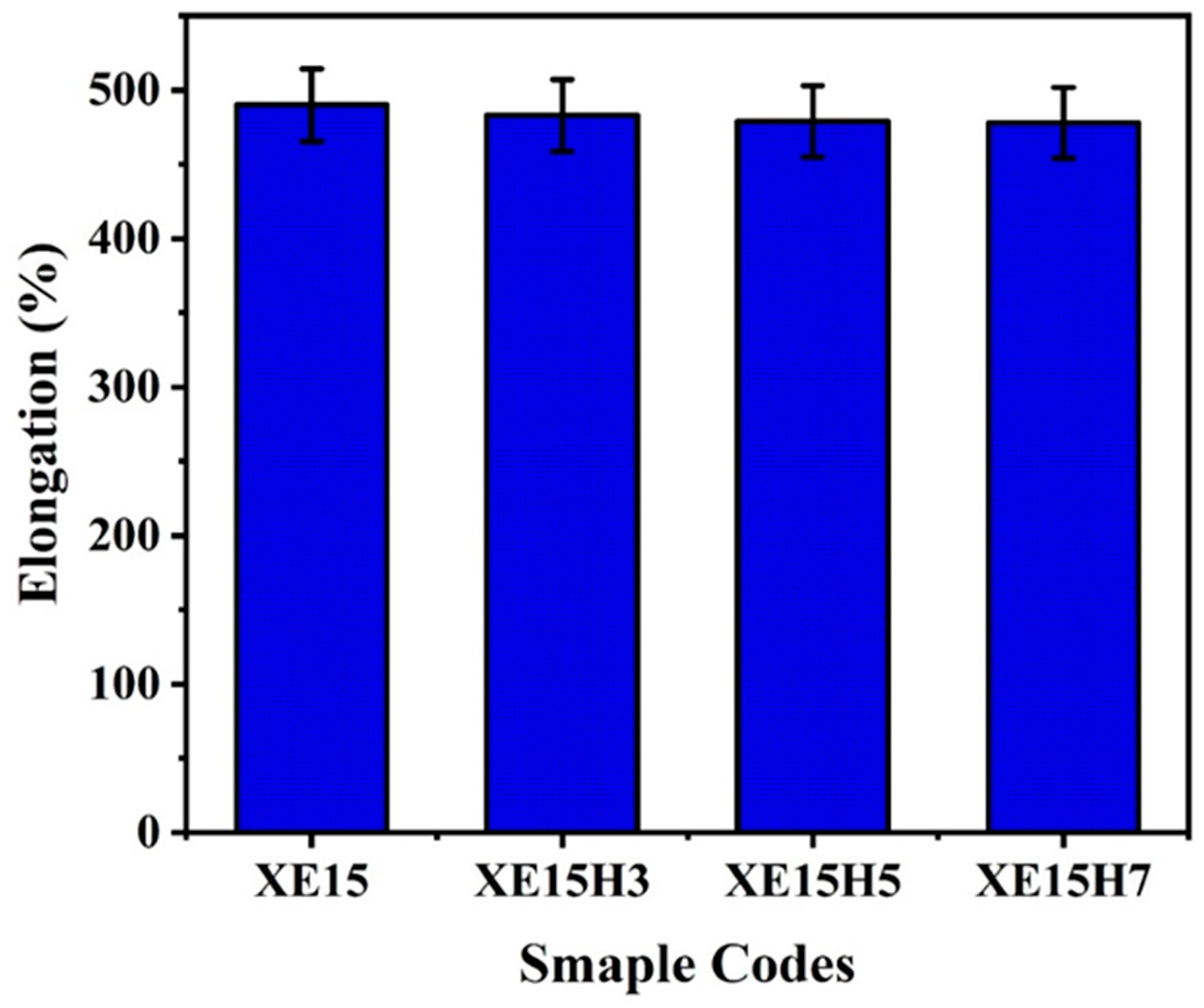
4.3. Mechanical Properties
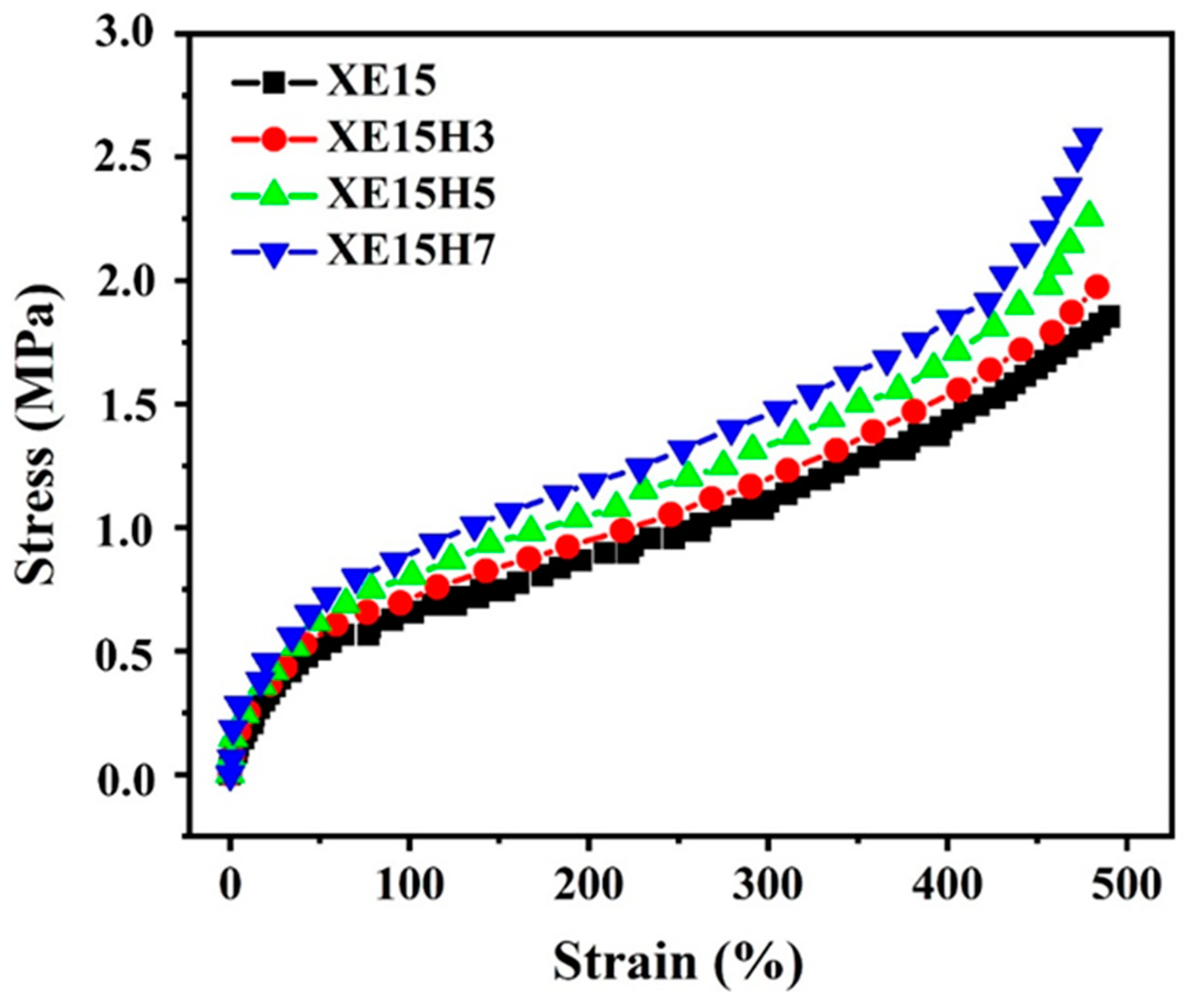
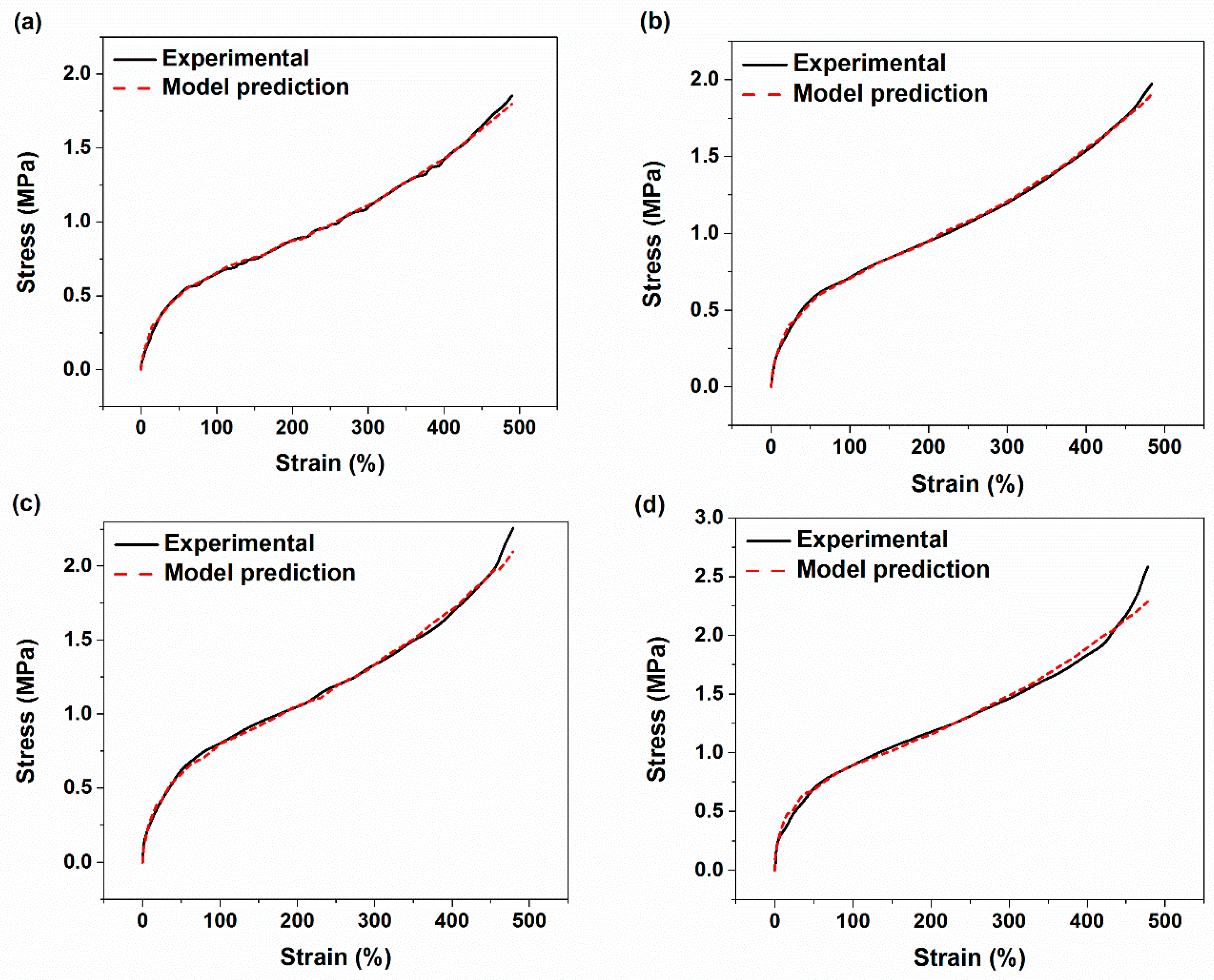
4.4. Stress–Strain Analyses
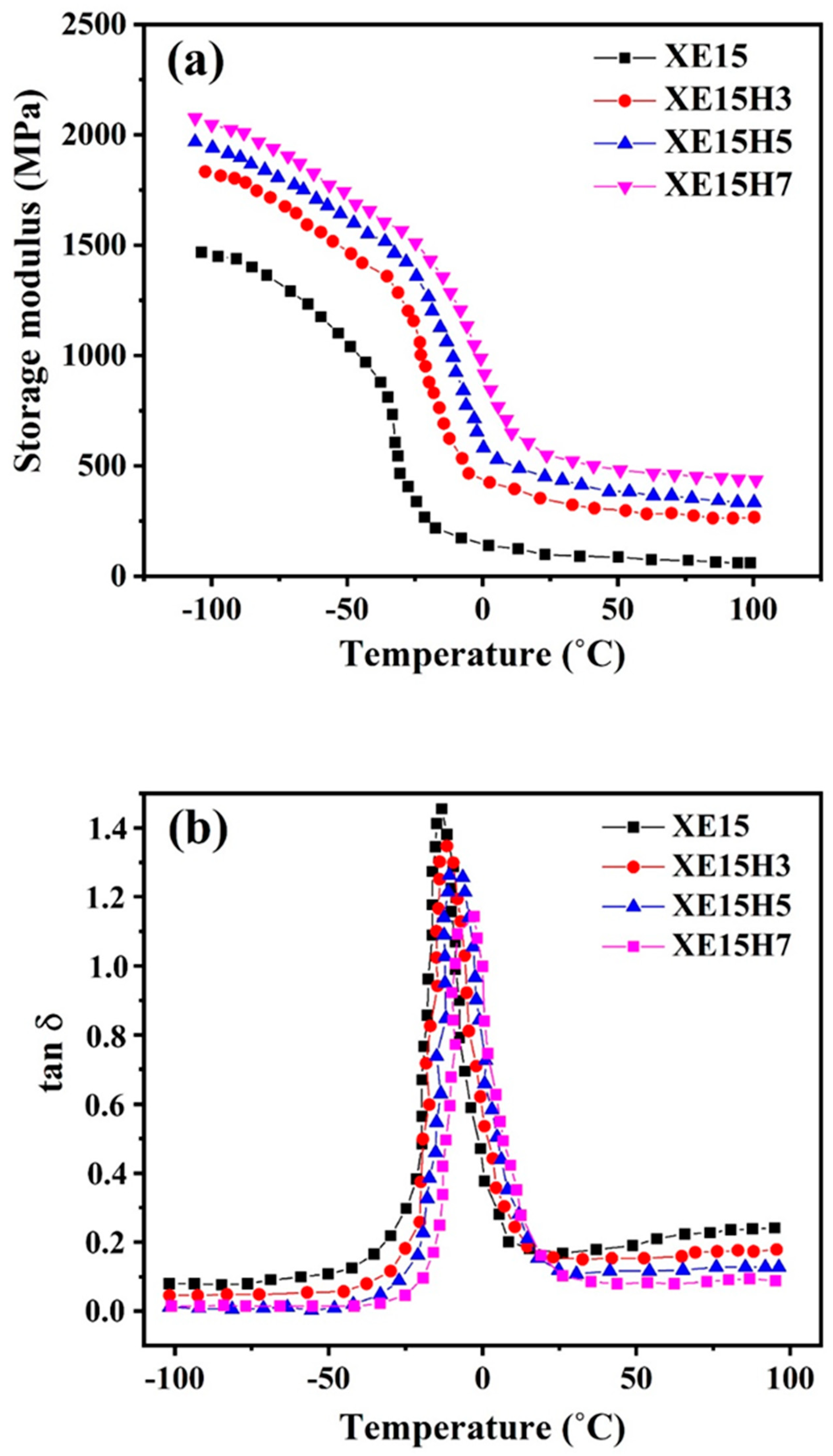
4.5. DMTA Investigations
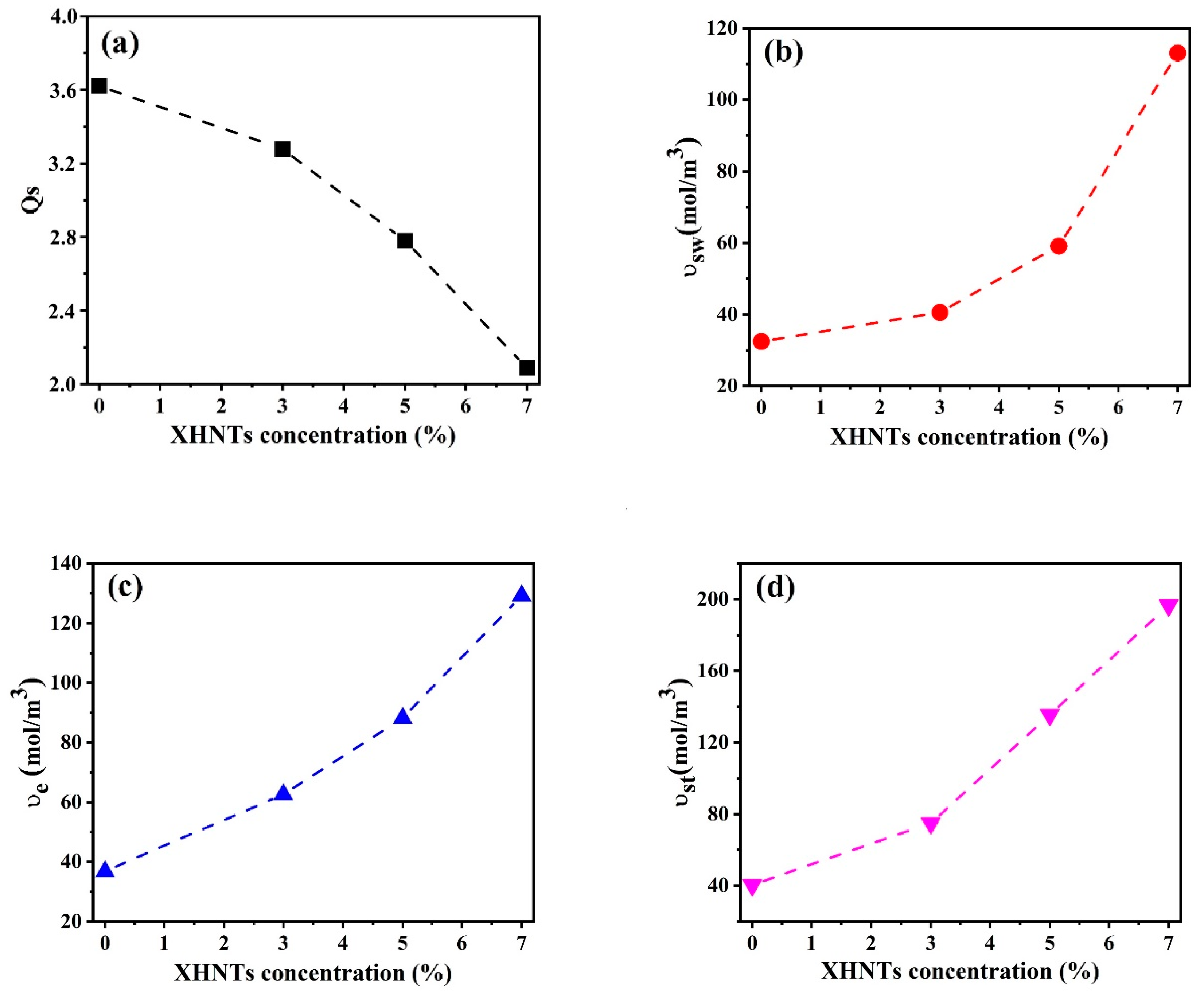
4.6. Swelling Characteristics
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Utrera-Barrios, S.; Hernández Santana, M.; Verdejo, R.; López-Manchado, M.A. Design of rubber composites with autonomous self-healing capability. ACS Omega 2020, 5, 1902–1910. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Karimi, M.; Saeb, M.R. Fabrication methods of carbon-based rubber nanocomposites. In Carbon-Based Nanofillers and Their Rubber Nanocomposites; Elsevier: Kidlington, UK, 2019; pp. 355–380. [Google Scholar]
- Zedler, Ł.; Colom, X.; Cañavate, J.; Saeb, M.R.; Haponiuk, J.T.; Formela, K. Investigating the impact of curing system on structure-property relationship of natural rubber modified with brewery by-product and ground tire rubber. Polymers 2020, 12, 545. [Google Scholar] [CrossRef] [PubMed]
- Hernández, M.; del Mar Bernal, M.; Verdejo, R.; Ezquerra, T.A.; López-Manchado, M.A. Overall performance of natural rubber/graphene nanocomposites. Compos. Sci. Technol. 2012, 73, 40–46. [Google Scholar] [CrossRef]
- Bhagavatheswaran, E.S.; Das, A.; Rastin, H.; Saeidi, H.; Jafari, S.H.; Vahabi, H.; Najafi, F.; Khonakdar, H.A.; Formela, K.; Jouyandeh, M. The taste of waste: The edge of eggshell over calcium carbonate in acrylonitrile butadiene rubber. J. Polym. Environ. 2019, 27, 2478–2489. [Google Scholar] [CrossRef]
- Jahromi, A.E.; Jahromi, H.R.E.; Hemmati, F.; Saeb, M.R.; Goodarzi, V.; Formela, K. Morphology and mechanical properties of polyamide/clay nanocomposites toughened with NBR/NBR-g-GMA: A comparative study. Compos. B Eng. 2016, 90, 478–484. [Google Scholar] [CrossRef]
- Pal, K.; Pal, S.K.; Das, C.K.; Kim, J.K. Effect of fillers on morphological properties and wear characteristics of XNBR/NR blends. J. Appl. Polym. Sci. 2011, 120, 710–718. [Google Scholar] [CrossRef]
- Severe, G.; White, J.L. Dynamically vulcanized blends of oil-resistant elastomers with HNBR. J. Appl. Polym. Sci. 2005, 95, 2–5. [Google Scholar] [CrossRef]
- Chakraborty, S.K.; De, S.K. Epoxy-resin-cured carboxylated nitrile rubber. J. Appl. Polym. Sci. 1982, 27, 4561–4576. [Google Scholar] [CrossRef]
- Brown, H. Crosslinking reactions of carboxylic elastomers. Rubber Chem. Technol. 1963, 36, 931–962. [Google Scholar] [CrossRef]
- Cheng, C.; Chen, Z.; Huang, Z.; Zhang, C.; Tusiime, R.; Zhou, J.; Sun, Z.; Liu, Y.; Yu, M.; Zhang, H. Simultaneously improving mode i and mode ii fracture toughness of the carbon fiber/epoxy composite laminates via interleaved with uniformly aligned pes fiber webs. Compos. A Appl. Sci. Manuf. 2020, 129, 105696. [Google Scholar] [CrossRef]
- Yang, X.; Fan, S.; Li, Y.; Guo, Y.; Li, Y.; Ruan, K.; Zhang, S.; Zhang, J.; Kong, J.; Gu, J. Synchronously improved electromagnetic interference shielding and thermal conductivity for epoxy nanocomposites by constructing 3d copper nanowires/thermally annealed graphene aerogel framework. Compos. A Appl. Sci. Manuf. 2020, 128, 105670. [Google Scholar] [CrossRef]
- Song, P.; Qiu, H.; Wang, L.; Liu, X.; Zhang, Y.; Zhang, J.; Kong, J.; Gu, J. Honeycomb structural rGO-MXene/epoxy nanocomposites for superior electromagnetic interference shielding performance. Sustain. Mater. Technol. 2020, 24, e00153. [Google Scholar] [CrossRef]
- Laskowska, A.; Zaborski, M.; Boiteux, G.; Gain, O.; Marzec, A.; Maniukiewicz, W. Ionic elastomers based on carboxylated nitrile rubber (XNBR) and magnesium aluminum layered double hydroxide (hydrotalcite). Express Polym. Lett. 2014, 8, 374–386. [Google Scholar] [CrossRef]
- Sahoo, S.; Bhowmick, A.K. Influence of ZnO nanoparticles on the cure characteristics and mechanical properties of carboxylated nitrile rubber. J. Appl. Polym. Sci. 2007, 106, 3077–3083. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Naderi, G.; Ghoreishy, M.R. Effect of halloysite nanotube on microstructure, rheological and mechanical properties of dynamically vulcanized PA6/NBR thermoplastic vulcanizates. Soft Mater. 2016, 14, 127–139. [Google Scholar] [CrossRef]
- Bitinis, N.; Hernández, M.; Verdejo, R.; Kenny, J.M.; Lopez-Manchado, M.A. Recent advances in clay/polymer nanocomposites. Advan. Mater. 2011, 23, 5229–5236. [Google Scholar] [CrossRef] [PubMed]
- Vahabi, H.; Sonnier, R.; Taguet, A.; Otazaghine, B.; Saeb, M.R.; Beyer, G. Halloysite nanotubes (HNTs)/polymer nanocomposites: Thermal degradation and flame retardancy. In Clay Nanoparticles; Elsevier: Kidlington, UK, 2020; pp. 67–93. [Google Scholar]
- Paran, S.M.R.; Naderi, G.; Ghoreishy, M. Microstructure and mechanical properties of thermoplastic elastomer nanocomposites based on PA6/NBR/HNT. Polym. Compos. 2017, 38, E451–E461. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Vahabi, H.; Ducos, F.; Formela, K.; Zarrintaj, P.; Laachachi, A.; Cuesta, J.-M.L.; Saeb, M.R. Crystallization kinetics study of dynamically vulcanized PA6/NBR/HNTs nanocomposites by nonisothermal differential scanning calorimetry. J. Appl. Polym. Sci. 2018, 135, 46488. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Vahabi, H.; Jouyandeh, M.; Ducos, F.; Formela, K.; Saeb, M.R. Thermal decomposition kinetics of dynamically vulcanized polyamide 6–acrylonitrile butadiene rubber–halloysite nanotube nanocomposites. J. Appl. Polym. Sci. 2019, 136, 47483. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Naderi, G.; Ghoreishy, M. XNBR-grafted halloysite nanotube core-shell as a potential compatibilizer for immiscible polymer systems. Appl. Surf. Sci. 2016, 382, 63–72. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Abdorahimi, M.; Shekarabi, A.; Khonakdar, H.A.; Jafari, S.H.; Saeb, M.R. Modeling and analysis of nonlinear elastoplastic behavior of compatibilized polyolefin/polyester/clay nanocomposites with emphasis on interfacial interaction exploration. Compos. Sci. Technol. 2018, 154, 92–103. [Google Scholar] [CrossRef]
- Boyce, M.C.; Arruda, E.M. Constitutive models of rubber elasticity: A review. Rubber Chem. Technol. 2000, 73, 504–523. [Google Scholar] [CrossRef]
- Bergstrom, J.S.; Boyce, M.C. Mechanical behavior of particle filled elastomers. Rubber Chem. Technol. 1999, 72, 633–656. [Google Scholar] [CrossRef]
- Valentini, L.; Bon, S.B.; Pugno, N.; Santana, M.H.; López-Manchado, M.A.; Giorgi, G. Synergistic icephobic behaviour of swollen nitrile butadiene rubber graphene and/or carbon nanotube composites. Compos. B Eng. 2019, 166, 352–360. [Google Scholar] [CrossRef]
- Valentini, L.; Bon, S.B.; Hernandez, M.; López-Manchado, M.A.; Pugno, N. Nitrile butadiene rubber composites reinforced with reduced graphene oxide and carbon nanotubes show superior mechanical, electrical and icephobic properties. Compos. Sci. Technol. 2018, 166, 109–114. [Google Scholar] [CrossRef]
- McKenna, G.B.; Flynn, K.M.; Chen, Y. Swelling in crosslinked natural rubber: Experimental evidence of the crosslink density dependence of χ. Polymer 1990, 31, 1937–1945. [Google Scholar] [CrossRef]
- Hill, L.W. Calculation of crosslink density in short chain networks. Prog. Org. Coat. 1997, 31, 235–243. [Google Scholar] [CrossRef]
- Orwoll, R.A.; Arnold, P.A. Polymer–solvent interaction parameter χ. In Physical Properties of Polymers Handbook; Springer: Berlin, Germany, 2007; pp. 233–257. [Google Scholar]
- Sekkar, V.; Narayanaswamy, K.; Scariah, K.; Nair, P.; Sastri, K.; Ang, H.G. Evaluation by various experimental approaches of the crosslink density of urethane networks based on hydroxyl-terminated polybutadiene. J. Appl. Polym. Sci. 2007, 103, 3129–3133. [Google Scholar] [CrossRef]
- Paran, S.; Naderi, G.; Ghoreishy, M. Mechanical properties development of high-ACN nitrile-butadiene rubber/organoclay nanocomposites. Plast. Rubber Compos. 2016, 45, 389–397. [Google Scholar] [CrossRef]
- Mousa, A.; Heinrich, G.; Wagenknecht, U. Cure characteristics and mechanical properties of carboxylated nitrile butadiene rubber (XNBR) vulcanizate reinforced by organic filler. Polym. Plast. Technol. Eng. 2011, 50, 1388–1392. [Google Scholar] [CrossRef]
- Ibarra, L.; Marcos-Fernandez, A.; Alzorriz, M. Mechanistic approach to the curing of carboxylated nitrile rubber (XNBR) by zinc peroxide/zinc oxide. Polymer 2002, 43, 1649–1655. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Naderi, G.; Ghoreishy, M.; Dubois, C. Multiscale modeling of polymer systems comprising nanotube-like inclusions by considering interfacial debonding under plastic deformations. Compos. Struct. 2018, 194, 302–315. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Naderi, G.; Ghoreishy, M.; Heydari, A. Enhancement of mechanical, thermal and morphological properties of compatibilized graphene reinforced dynamically vulcanized thermoplastic elastomer vulcanizates based on polyethylene and reclaimed rubber. Compos. Sci. Technol. 2018, 161, 57–65. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Naderi, G.; Babakhani, A. An experimental study of the effect of CNTs on the mechanical properties of CNTs/NR/EPDM nanocomposite. Polym. Compos. 2018, 39, 4071–4079. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Saeb, M.R.; Formela, K.; Goodarzi, V.; Vijayan, P.; Puglia, D.; Khonakdar, H.A.; Thomas, S. To what extent can hyperelastic models make sense the effect of clay surface treatment on the mechanical properties of elastomeric nanocomposites? Macromol. Mater. Eng. 2017, 302, 1700036. [Google Scholar] [CrossRef]
- Pasbakhsh, P.; Ismail, H.; Fauzi, M.A.; Bakar, A.A. EPDM/modified halloysite nanocomposites. Appl. Clay Sci. 2010, 48, 405–413. [Google Scholar] [CrossRef]
- Hu, C.; Chang, S.; Li, B.; Wang, J.; Zhang, Z. Error analysis and correction of lever-type stylus profilometer based on Nelder-Mead Simplex method. In Proceedings of the AOPC 2017: 3D Measurement Technology for Intelligent Manufacturing, Beijing, China, 4–6 June 2017; p. 1045802. [Google Scholar]
- Bréchet, Y.; Cavaille, J.-Y.; Chabert, E.; Chazeau, L.; Dendievel, R.; Flandin, L.; Gauthier, C. Polymer based nanocomposites: Effect of filler-filler and filler-matrix interactions. Adv. Eng. Mater. 2001, 3, 571–577. [Google Scholar] [CrossRef]
- Sussman, T.; Bathe, K.J. A model of incompressible isotropic hyperelastic material behavior using spline interpolations of tension–compression test data. Int. J. Numer. Meth. Biomed. Eng. 2009, 25, 53–63. [Google Scholar] [CrossRef]
- Du, M.; Guo, B.; Lei, Y.; Liu, M.; Jia, D. Carboxylated butadiene–styrene rubber/halloysite nanotube nanocomposites: Interfacial interaction and performance. Polymer 2008, 49, 4871–4876. [Google Scholar] [CrossRef]
- Quintavalla, S.; Johnson, S. Extension of the Bergstrom-Boyce model to high strain rates. Rubber Chem. Technol. 2004, 77, 972–981. [Google Scholar] [CrossRef]
- Ghoreishy, M.; Firouzbakht, M.; Naderi, G. Parameter determination and experimental verification of Bergström–Boyce hysteresis model for rubber compounds reinforced by carbon black blends. Mater. Des. 2014, 53, 457–465. [Google Scholar] [CrossRef]
- Paran, S.; Naderi, G.; Ghoreishy, M.; Dubois, C. Essential work of fracture and failure mechanisms in dynamically vulcanized thermoplastic elastomer nanocomposites based on PA6/NBR/XNBR-grafted HNTs. Eng. Fract. Mech. 2018, 200, 251–262. [Google Scholar] [CrossRef]
- Berahman, R.; Raiati, M.; Mazidi, M.M.; Paran, S.M.R. Preparation and characterization of vulcanized silicone rubber/halloysite nanotube nanocomposites: Effect of matrix hardness and HNT content. Mater. Des. 2016, 104, 333–345. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Naderi, G.; Javadi, F.; Shemshadi, R.; Saeb, M.R. Experimental and theoretical analyses on mechanical properties and stiffness of hybrid graphene/graphene oxide reinforced EPDM/NBR nanocomposites. Mater. Today Commun. 2020, 22, 100763. [Google Scholar] [CrossRef]
- Paran, S.M.R.; Naderi, G.; Shokoohi, S.; Ebadati, J.; Dubois, C. Mechanical and thermal properties of green thermoplastic elastomer vulcanizate nanocomposites based on poly(vinyl chloride) and nitrile butadiene rubber containing organoclay and rice straw natural fibers. J. Polym. Environ. 2019, 27, 2017–2026. [Google Scholar] [CrossRef]
- Swapna, V.; Stephen, R.; Greeshma, T.; Sharan Dev, C.; Sreekala, M. Mechanical and swelling behavior of green nanocomposites of natural rubber latex and tubular shaped halloysite nano clay. Polym. Compos. 2016, 37, 602–611. [Google Scholar] [CrossRef]
- Kang, H.; Tang, Y.; Yao, L.; Yang, F.; Fang, Q.; Hui, D. Fabrication of graphene/natural rubber nanocomposites with high dynamic properties through convenient mechanical mixing. Compos. B Eng. 2017, 112, 1–7. [Google Scholar] [CrossRef]


| Sample Code | XNBR/Epoxy (70/30) | XHNT | ZnO | Stearic Acid |
|---|---|---|---|---|
| XE15 | 100 | 0 | 6 | 2 |
| XE15H3 | 100 | 3 | 6 | 2 |
| XE15H5 | 100 | 5 | 6 | 2 |
| XE15H7 | 100 | 7 | 6 | 2 |
| Sample Code | Mh (lbf.in) a | Ml (lbf.in) b | (Mh-Ml) (lbf.in) | ts2 (min) c | tc90 (min) d |
|---|---|---|---|---|---|
| XE15 | 66.60 | 18.77 | 47.83 | 3.39 | 20.3 |
| XE15H3 | 66.80 | 19.73 | 47.07 | 2.75 | 19.9 |
| XE15H5 | 67.60 | 21.69 | 45.91 | 2.68 | 19.5 |
| XE15H7 | 69.90 | 24.07 | 45.83 | 2.51 | 18.8 |
| Sample Codes | μ (MPa) | κ (MPa) | S | ζ | C | τbase | m | τcut |
|---|---|---|---|---|---|---|---|---|
| XE15 | 0.23 | 29.67 | 103.14 | 3.62 × 10−9 | −0.89 | 0.39 | 4.06 | 0.001 |
| XE15H3 | 0.24 | 39.31 | 103.15 | 1.58 × 10−7 | −0.78 | 0.44 | 4.14 | 0.033 |
| XE15H5 | 0.26 | 47.79 | 103.17 | 1.96 × 10−7 | −0.76 | 0.48 | 4.15 | 0.060 |
| XE15H7 | 0.29 | 55.40 | 103.18 | 1.97 × 10−7 | −0.65 | 0.49 | 4.21 | 0.070 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paran, S.M.R.; Naderi, G.; Mosallanezhad, H.; Movahedifar, E.; Formela, K.; Saeb, M.R. Microstructure and Mechanical Properties of Carboxylated Nitrile Butadiene Rubber/Epoxy/XNBR-grafted Halloysite Nanotubes Nanocomposites. Polymers 2020, 12, 1192. https://doi.org/10.3390/polym12051192
Paran SMR, Naderi G, Mosallanezhad H, Movahedifar E, Formela K, Saeb MR. Microstructure and Mechanical Properties of Carboxylated Nitrile Butadiene Rubber/Epoxy/XNBR-grafted Halloysite Nanotubes Nanocomposites. Polymers. 2020; 12(5):1192. https://doi.org/10.3390/polym12051192
Chicago/Turabian StyleParan, Seyed Mohamad Reza, Ghasem Naderi, Heydar Mosallanezhad, Elnaz Movahedifar, Krzysztof Formela, and Mohammad Reza Saeb. 2020. "Microstructure and Mechanical Properties of Carboxylated Nitrile Butadiene Rubber/Epoxy/XNBR-grafted Halloysite Nanotubes Nanocomposites" Polymers 12, no. 5: 1192. https://doi.org/10.3390/polym12051192
APA StyleParan, S. M. R., Naderi, G., Mosallanezhad, H., Movahedifar, E., Formela, K., & Saeb, M. R. (2020). Microstructure and Mechanical Properties of Carboxylated Nitrile Butadiene Rubber/Epoxy/XNBR-grafted Halloysite Nanotubes Nanocomposites. Polymers, 12(5), 1192. https://doi.org/10.3390/polym12051192








