Delayed Addition of Template Molecules Enhances the Binding Properties of Diclofenac-Imprinted Polymers
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Polymers Preparation
2.3. Light Scattering of Polymerization Mixtures
2.4. HPLC Analysis
2.5. Equilibrium Batch Rebinding
3. Results and Discussion
3.1. Synthesis of Imprinted Polymers
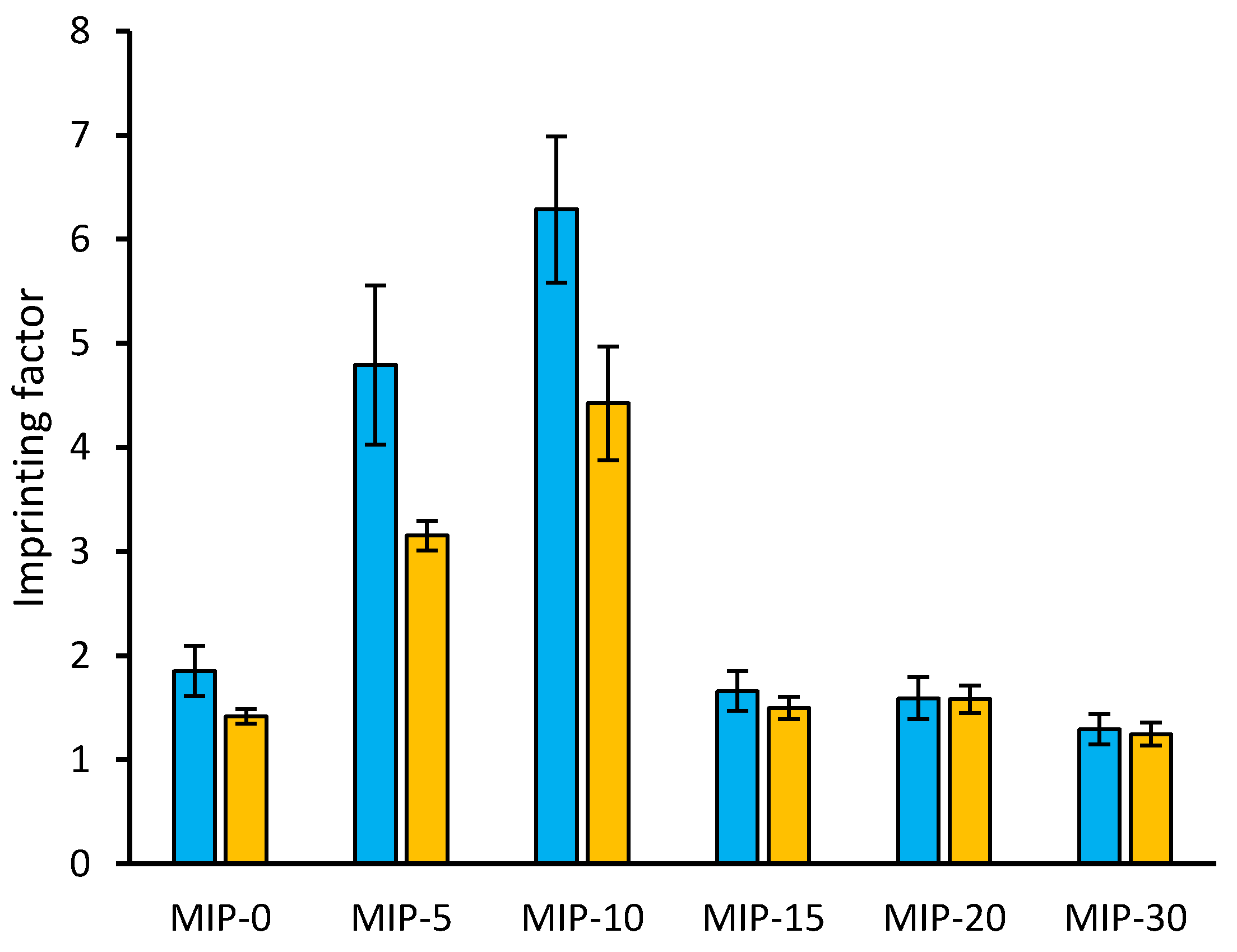
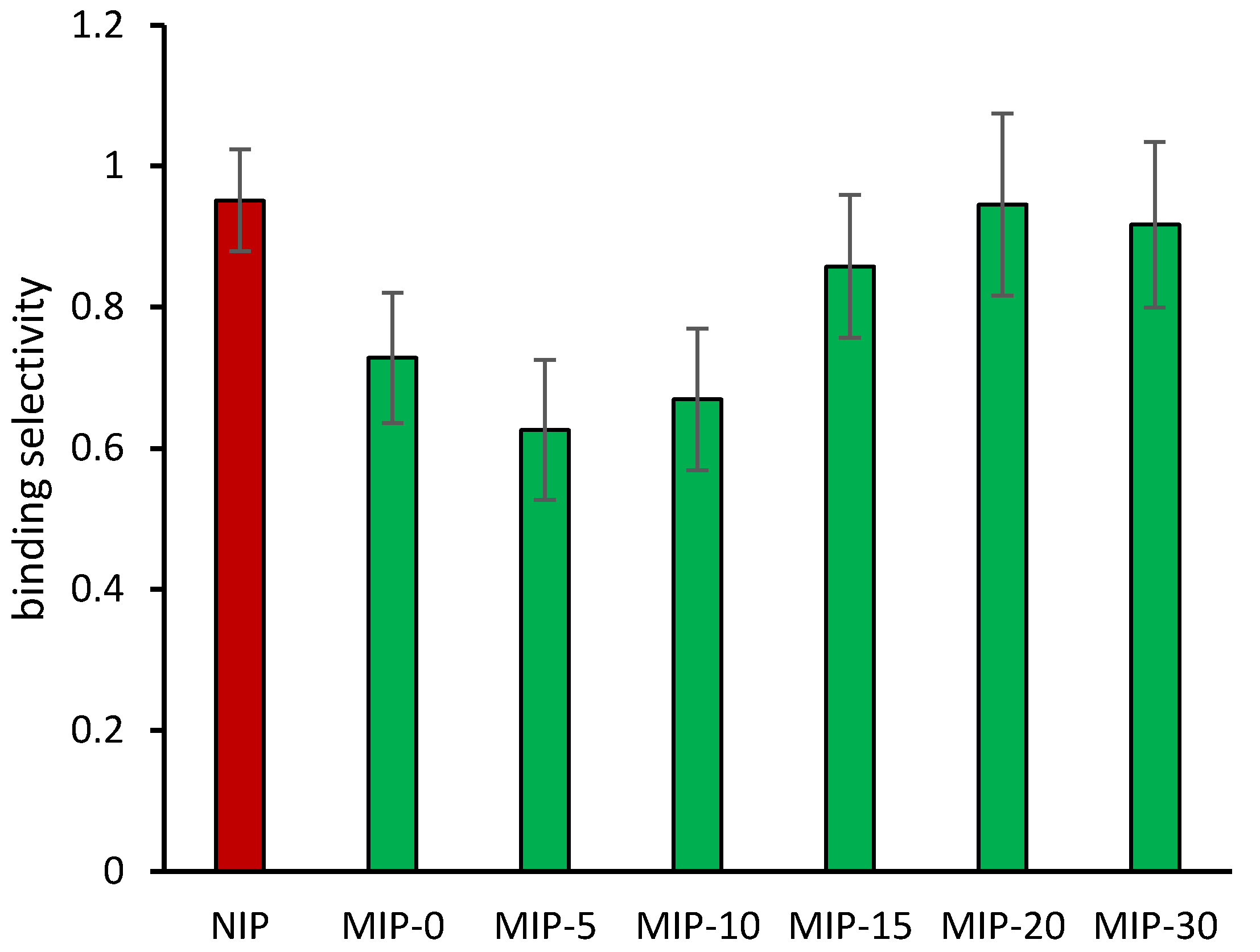
3.2. Binding Properties of Imprinted Polymers
4. Discussion
- (a)
- When template molecules are present in the pre-polymerization mixture from the start, they will find a reaction medium that is very rich in functional monomers, but with no oligomers. Because the polymerization process is fast and dominated by cross-linking steps, weak complexes between template molecules and functional monomers will prevail by producing an MIP with a relatively low imprinting effect.
- (b)
- When template molecules are added after the start of the polymerization process, they will find a reaction medium enriched with functional macromonomers and only a few remaining monomers. Because the functional macromonomers are highly flexible, they will freely rearrange around the template. Chelate effects and chain desolvation greatly stabilize these complexes, producing an MIP with an enhanced imprinting effect.
- (c)
- When template molecules are added very late in the polymerization process, they will find a reaction medium mainly composed of preformed nanogel particles that will form a continuous cross-linked macrogel. In this condition, the interactions between the template molecules and the growing polymer particles become increasingly disadvantaged, since cross-linked nanogel particles are stiffer than functional macromonomers. As a consequence, an MIP with a low or negligible imprinting effect is the more predictable outcome.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Haupt, K.; Linares, A.V.; Bompart, M.; Tse Sum Bui, B. Molecularly imprinted polymers. Top. Curr. Chem. 2012, 325, 1–28. [Google Scholar]
- Chen, L.; Xu, W.; Li, J. Recent advances in molecular imprinting technology: Current status, challenges and highlighted applications. Chem. Soc. Rev. 2011, 40, 2922–2942. [Google Scholar] [CrossRef]
- Chen, L.; Wang, X.; Lu, W.; Wu, X.; Li, J. Molecular imprinting: Perspectives and applications. Chem. Soc. Rev. 2016, 45, 2137–2211. [Google Scholar] [CrossRef]
- Biffis, A.; Graham, N.B.; Siedlaczek, G.; Stalberg, S.; Wulff, G. The synthesis, characterization and molecular recognition properties of imprinted microgels. Macromol. Chem. Phys. 2001, 202, 163–171. [Google Scholar] [CrossRef]
- Salian, V.D.; Byrne, M.E. Living radical polymerization and molecular imprinting: Improving polymer morphology in imprinted polymers. Macromol. Mater. Eng. 2013, 298, 379–390. [Google Scholar] [CrossRef]
- O’Mahony, J.; Molinelli, A.; Nolan, K.; Smyth, M.R.; Mizaikoff, B. Towards the rational development of molecularly imprinted polymers: 1H NMR studies on hydrophobicity and ion-pair interactions as driving forces for selectivity. Biosens. Bioelectron. 2005, 20, 1884–1893. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, J.; Molinelli, A.; Nolan, K.; Smyth, M.R.; Mizaikoff, B. Anatomy of a successful imprint: Analysing the recognition mechanisms of a molecularly imprinted polymer for quercetin. Biosens. Bioelectron. 2006, 21, 1383–1392. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Lu, J.; Chen, Z.; Guo, J. Preparation and recognition performance of cholic acid-imprinted material prepared with novel surface-imprinting technique. Polymer 2009, 50, 3275–3284. [Google Scholar] [CrossRef]
- Gao, B.; Liu, S.; Li, Y. Preparation and recognition performance of uric acid-imprinted material prepared with novel surface imprinting technique. J. Chromatogr. A 2010, 1217, 2226–2236. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Qinyuan, N.; Ruikui, D. Preparation and recognition performance of cytisine alkaloid-imprinted material prepared using novel surface molecular imprinting technique. J. Sep. Sci. 2010, 33, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Matsui, J.; Tamaki, K.; Sugimoto, N. Molecular imprinting in alcohols: Utility of a pre-polymer based strategy for synthesizing stereoselective artificial receptor polymers in hydrophilic media. Anal. Chim. Acta 2002, 466, 11–15. [Google Scholar] [CrossRef]
- Li, Z.; Day, M.; Ding, J.; Faid, K. Synthesis and characterization of functional methacrylate copolymers and their application in molecular imprinting. Macromolecules 2005, 38, 2620–2625. [Google Scholar] [CrossRef]
- Kitayama, Y.; Yoshikawa, K.; Takeuchi, T. Post-cross-linked molecular imprinting with functional polymers as a universal building block for artificial polymeric receptors. Macromolecules 2017, 50, 7526–7534. [Google Scholar] [CrossRef]
- Long, Y.; Xing, X.; Han, R.; Sun, Y.; Wang, Y.; Zhao, Z.; Mi, H. Two-step purification of low-content cellular protein using protein-imprinted polymers. Anal. Biochem. 2008, 380, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Zhao, Z.; Fan, Y.; Wang, C.; Shi, L.; Xia, J.; Long, Y.; Mi, H. Protein-imprinted polymer with immobilized assistant recognition polymer chains. Biomaterials 2006, 27, 4381–4387. [Google Scholar] [CrossRef] [PubMed]
- Han, R.; Xing, X.; Wang, Y.; Long, Y.; Sun, Y.; Zhao, Z.; Mi, H. Separation/enrichment of active natural low content protein using protein imprinted polymer. J. Chromatogr. B 2008, 873, 113–118. [Google Scholar] [CrossRef]
- Gao, J.; Tian, H.; Wang, Y.; Yang, Q.; Liu, D.; Wang, Y.; Mi, H. The design of protein-imprinted polymers as antibody substitutes for investigating proteineprotein interactions. Biomaterials 2012, 33, 3344–3352. [Google Scholar] [CrossRef]
- Liu, D.; Yang, Q.; Jin, S.; Song, Y.; Gao, J.; Wang, Y.; Mi, H. Core-shell molecularly imprinted polymer nanoparticles with assistant recognition polymer chains for effective recognition and enrichment of natural low-abundance protein. Acta Biomater. 2014, 10, 769–775. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, S.; Pan, J.; Jia, H.; Chen, Z.; Guo, T. Multifunctional oligomer immobilized on quartz crystal microbalance: A facile and stabilized molecular imprinting strategy for glycoprotein detection. Anal. Bioanal. Chem. 2019, 411, 3941–3949. [Google Scholar] [CrossRef]
- Zhao, W.; Li, B.; Xu, S.; Huang, X.; Luo, J.; Zhu, Y.; Liu, X. Electrochemical protein recognition based on macromolecular self-assembly of molecularly imprinted polymer: A new strategy to mimic antibody for label-free biosensing. J. Mater. Chem. B 2019, 7, 2311–2319. [Google Scholar] [CrossRef]
- Garcia-Calzon, J.A.; Diaz-Garcia, M.E. Characterization of binding sites in molecularly imprinted polymers. Sens. Actuat. B 2007, 123, 1180–1194. [Google Scholar] [CrossRef]
- Kumar, K.V.; Gadipelli, S.; Wood, B.; Ramisetty, K.A.; Stewart, A.A.; Howard, C.A.; Brett, D.J.L.; Rodriguez-Reinoso, F. Characterization of the adsorption site energies and heterogeneous surfaces of porous materials. J. Mater. Chem. A 2019, 7, 10104–10137. [Google Scholar] [CrossRef]
- Baggiani, C.; Giovannoli, C.; Anfossi, L.; Passini, C.; Baravalle, P.; Giraudi, G. A connection between the binding properties of imprinted and non-imprinted polymers: A change of perspective in molecular imprinting. J. Am. Chem. Soc. 2012, 134, 1513–1518. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, T.; Cui, L.; Wang, J.D.; Piletska, E.V.; Guerreiro, A.R.; Piletsky, S.A. Rational design and synthesis of water-compatible molecularly imprinted polymers for selective solid phase extraction of amiodarone. Anal. Chim. Acta 2012, 709, 98–104. [Google Scholar] [CrossRef]
- Khvostikov, V.A.; Karandashev, V.K.; Burmii, Z.P. Analysis of α-alumina samples by mass spectrometry with inductively coupled plasma and laser ablation. Inorg. Mater. 2015, 51, 1410–1415. [Google Scholar] [CrossRef]
- Li, X.Y.; Ma, L.; Huang, Y.P.; Liu, Z.S.; Aisa, H.A. Preparation of metallic pivot-based imprinted monoliths with a hydrophilic macromonomer. RSC Adv. 2015, 5, 36753–36761. [Google Scholar] [CrossRef]
- Feng, J.; Li, F.; Ran, R.X.; Huang, Y.P.; Liu, Z.S. Synergistic effect of metal ions pivot and macromolecular crowding reagents on affinity of molecularly imprinted polymer. Eur. Polym. J. 2019, 120, 109242. [Google Scholar] [CrossRef]
- Cowen, T.; Stefanucci, E.; Piletska, E.; Marrazza, G.; Canfarotta, F.; Piletsky, S.A. Synthetic mechanism of molecular imprinting at the solid phase. Macromolecules 2020, 53, 1435–1442. [Google Scholar] [CrossRef]

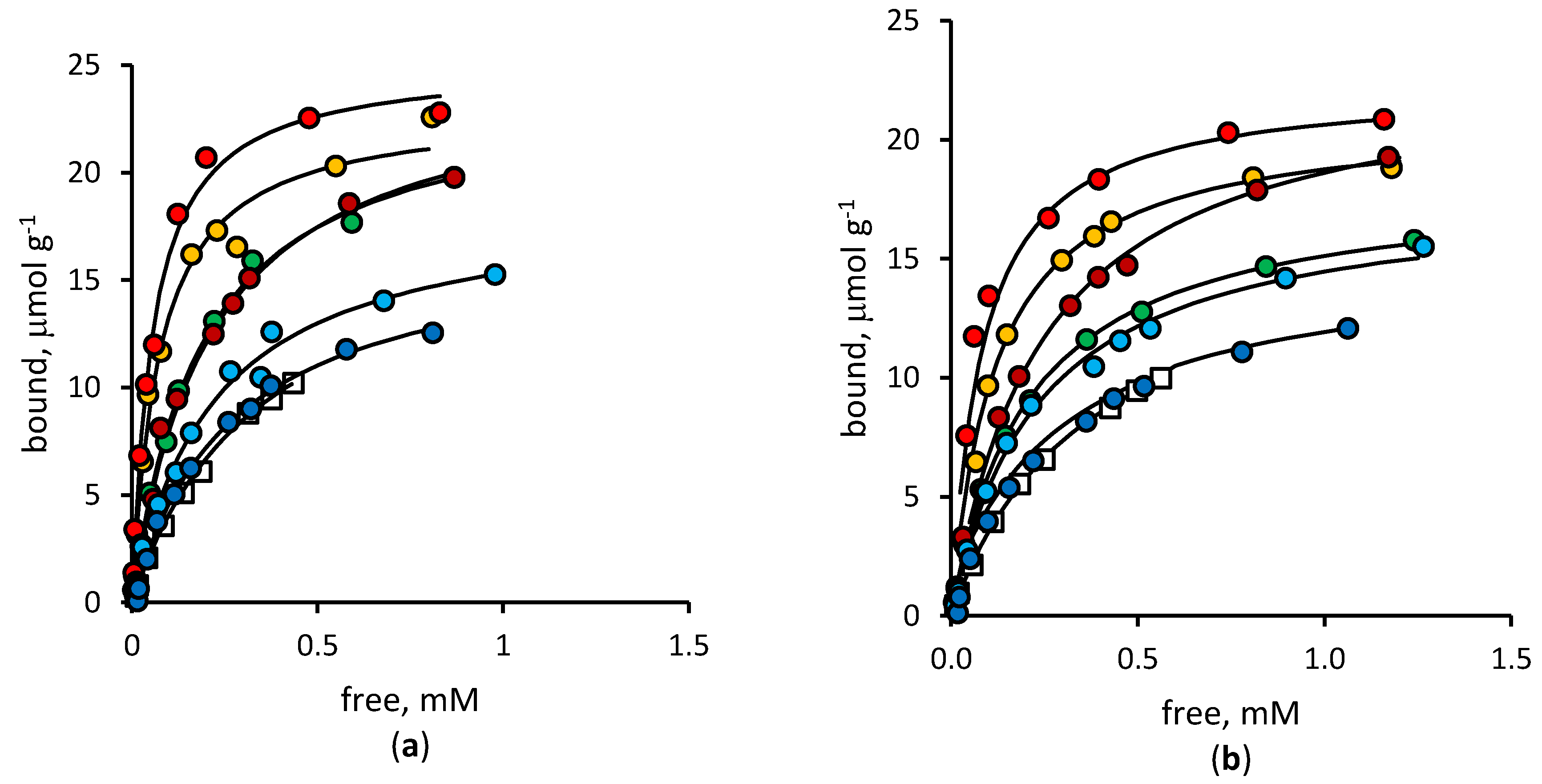
| Diclofenac | Mefenamic Acid | |||
|---|---|---|---|---|
| Polymer | Keq, 103 L mol−1 | Bmax, µmol g−1 | Keq, 103 L mol−1 | Bmax, µmol g−1 |
| NIP | 2.86 ± 0.18 | 18.4 ± 0.7 | 2.72 ± 0.11 | 16.4 ± 0.3 |
| MIP-0 | 5.30 ± 0.62 | 24.1 ± 1.1 | 3.86 ± 0.12 | 18.5 ± 0.2 |
| MIP-5 | 13.70 ± 2.04 | 23.0 ± 1.0 | 8.58 ± 0.20 | 20.9 ± 0.1 |
| MIP-10 | 17.98 ± 1.68 | 25.1 ± 0.7 | 12.03 ± 1.41 | 22.4 ± 1.3 |
| MIP-15 | 4.75 ± 0.47 | 24.8 ± 1.0 | 4.07 ± 0.26 | 23.2 ± 0.6 |
| MIP-20 | 4.55 ± 0.52 | 18.7 ± 0.3 | 4.30 ± 0.33 | 17.8 ± 0.5 |
| MIP-30 | 3.70 ± 0.36 | 17.0 ± 0.3 | 3.39 ± 0.28 | 15.3 ± 0.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anfossi, L.; Cavalera, S.; Di Nardo, F.; Spano, G.; Giovannoli, C.; Baggiani, C. Delayed Addition of Template Molecules Enhances the Binding Properties of Diclofenac-Imprinted Polymers. Polymers 2020, 12, 1178. https://doi.org/10.3390/polym12051178
Anfossi L, Cavalera S, Di Nardo F, Spano G, Giovannoli C, Baggiani C. Delayed Addition of Template Molecules Enhances the Binding Properties of Diclofenac-Imprinted Polymers. Polymers. 2020; 12(5):1178. https://doi.org/10.3390/polym12051178
Chicago/Turabian StyleAnfossi, Laura, Simone Cavalera, Fabio Di Nardo, Giulia Spano, Cristina Giovannoli, and Claudio Baggiani. 2020. "Delayed Addition of Template Molecules Enhances the Binding Properties of Diclofenac-Imprinted Polymers" Polymers 12, no. 5: 1178. https://doi.org/10.3390/polym12051178
APA StyleAnfossi, L., Cavalera, S., Di Nardo, F., Spano, G., Giovannoli, C., & Baggiani, C. (2020). Delayed Addition of Template Molecules Enhances the Binding Properties of Diclofenac-Imprinted Polymers. Polymers, 12(5), 1178. https://doi.org/10.3390/polym12051178









