History of Cyclodextrin Nanosponges
Abstract
1. Introduction
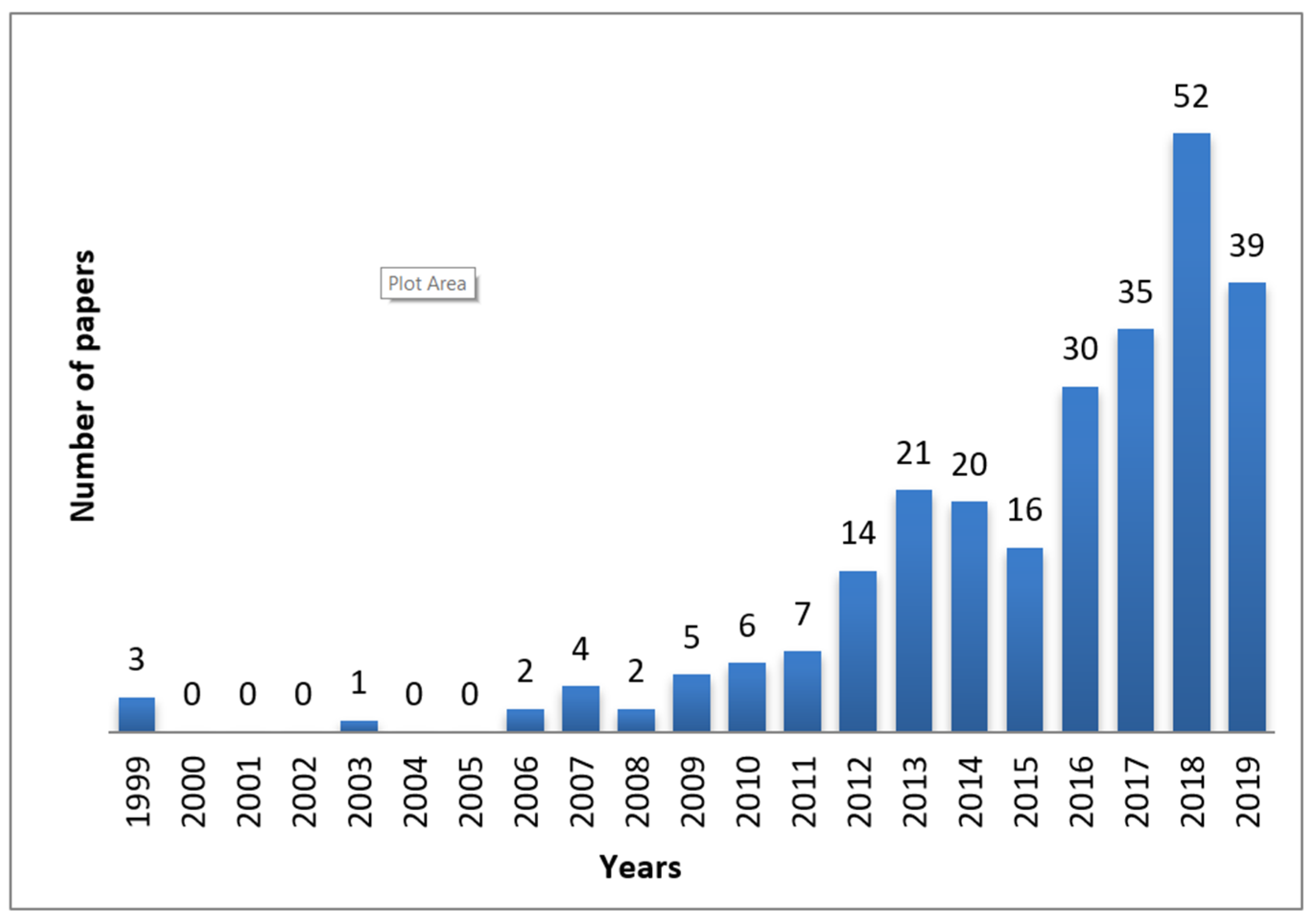
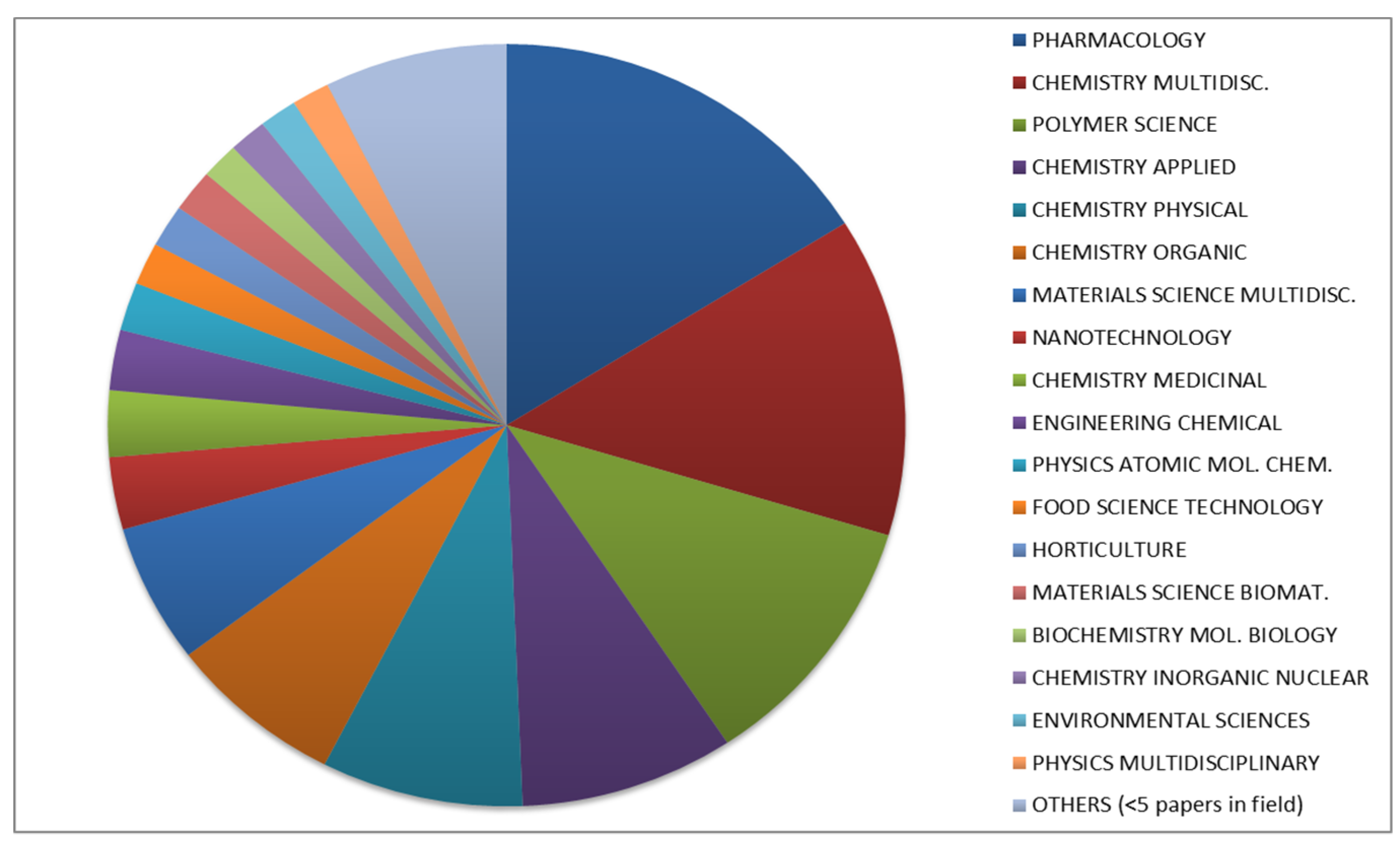
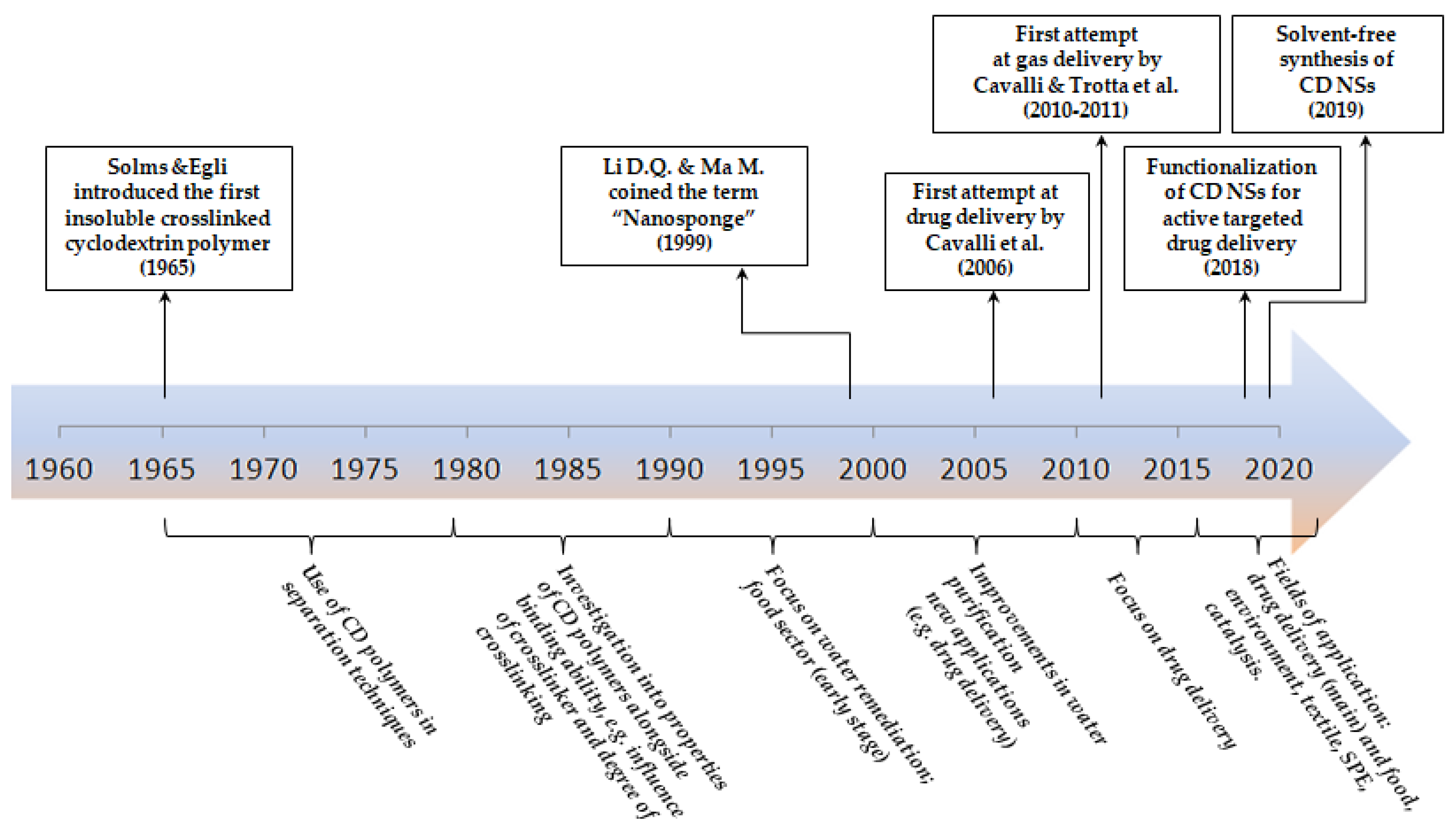
2. Origin and Historical Development of Nanosponges Over the Last 50 Years
2.1. From the 1960s to the 1980s: Origin of Insoluble Crosslinked Cyclodextrin Polymers
2.2. From the 1980s to the 1990s: Investigation on Polymer Properties and Applications
2.3. 1999:“In the Beginning Was the Word”
2.4. From 2000 to 2009: New Millennium Came with New Applications
2.5. From 2010 to 2015: Focus on Nanosponges as Delivery Systems
2.6. From 2016 to Present: State-of-the-Art and Future Prospects of Nanosponges
3. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Torne, S.J.; Ansari, K.A.; Vavia, P.R.; Trotta, F.; Cavalli, R. Enhanced oral paclitaxel bioavailability after administration of paclitaxel-loaded nanosponges. Drug Deliv. 2010, 17, 419–425. [Google Scholar] [CrossRef]
- Cavalli, R.; Trotta, F.; Tumiatti, W. Cyclodextrin-based nanosponges for drug delivery. J. Incl. Phenom. Macrocycl. Chem. 2006, 56, 209–213. [Google Scholar] [CrossRef]
- Vyas, A.; Saraf, S.; Saraf, S. Cyclodextrin based novel drug delivery systems. J. Incl. Phenom. Macrocycl. Chem. 2008, 62, 23–42. [Google Scholar] [CrossRef]
- Trotta, F. Cyclodextrin Nanosponges and Their Applications; Bilensoy, E., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011. [Google Scholar]
- Subramanian, S.; Singireddy, A.; Krishnamoorthy, K.; Rajappan, M. Nanosponges: A novel class of drug delivery system—Review. J. Pharm. Pharm. Sci. 2012, 15, 103–111. [Google Scholar] [CrossRef]
- Shringirishi, M.; Prajapati, S.K.; Mahor, A.; Alok, S.; Yadav, P.; Verma, A. Nanosponges: A potential nanocarrier for novel drug delivery-a review. Asian Pac. J. Trop. Dis. 2014, 4, S519–S526. [Google Scholar] [CrossRef]
- Chilajwar, S.V.; Pednekar, P.P.; Jadhav, K.R.; Gupta, G.J.; Kadam, V.J. Cyclodextrin-based nanosponges: A propitious platform for enhancing drug delivery. Expert Opin. Drug Deliv. 2014, 11, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Trotta, F.; Dianzani, C.; Caldera, F.; Mognetti, B.; Cavalli, R. The application of nanosponges to cancer drug delivery. Expert Opin. Drug Deliv. 2014, 11, 931–941. [Google Scholar] [CrossRef]
- Trotta, F.; Zanetti, M.; Cavalli, R. Cyclodextrin-based nanosponges as drug carriers. Beilstein J. Org. Chem. 2012, 8, 2091–2099. [Google Scholar] [CrossRef]
- Liu, Z.; Jiao, Y.; Wang, Y.; Zhou, C.; Zhang, Z. Polysaccharides-based nanoparticles as drug delivery systems. Adv. Drug Deliv. Rev. 2008, 60, 1650–1662. [Google Scholar] [CrossRef]
- Caldera, F.; Tannous, M.; Cavalli, R.; Zanetti, M.; Trotta, F. Evolution of Cyclodextrin Nanosponges. Int. J. Pharm. 2017, 531, 470–479. [Google Scholar] [CrossRef]
- Kutova, O.M.; Guryev, E.L.; Sokolova, E.A.; Alzeibak, R.; Balalaeva, I.V. Targeted delivery to tumors: Multidirectional strategies to improve treatment efficiency. Cancers 2019, 11, 68. [Google Scholar] [CrossRef] [PubMed]
- Trotta, F.; Rubin Pedrazzo, A. Processo Per La Preparazione Di Una Nanospugna. IT Patent P021499IT-01, 15 November 2019. [Google Scholar]
- Pushpalatha, R.; Selvamuthukumar, S.; Kilimozhi, D. Hierarchy analysis of different cross-linkers used for the preparation of cross-linked cyclodextrin as drug nanocarriers. Chem. Eng. Commun. 2018, 205, 759–771. [Google Scholar] [CrossRef]
- Pushpalatha, R.; Selvamuthukumar, S.; Kilimozhi, D. Cyclodextrin nanosponge based hydrogel for the transdermal co-delivery of curcumin and resveratrol: Development, optimization, in vitro and ex vivo evaluation. J. Drug Deliv. Sci. Technol. 2019, 52, 55–64. [Google Scholar] [CrossRef]
- Rao, M.R.P.; Raut, S.P.; Shirsath, C.T.; Jadhav, M.B.; Chandanshive, P.A. Self-nanoemulsifying Drug Delivery System of Mebendazole for Treatment of Lymphatic Filariasis. Indian J. Pharm. Sci. 2018, 80, 1057–1068. [Google Scholar] [CrossRef]
- Kamble, M.; Zaheer, Z.; Mokale, S.; Zainuddin, R. Formulation Optimization and Biopharmaceutical Evaluation of Imatinib Mesylate Loaded β-Cyclodextrin Nanosponges. Pharm. Nanotechnol. 2019, 7, 343–361. [Google Scholar] [CrossRef]
- Solms, J.; Egli, R.H. Harze mit Einschlusshohlräumen von Cyclodextrin-Struktur. Helv. Chim. Acta 1965, 48, 1225–1228. [Google Scholar] [CrossRef]
- Hoffman, J.L. Chromatography of nucleic acids on cross-linked cyciodextrin gels having inclusion-forming capacity. J. Macromol. Sci. Part A—Chem. 1973, 7, 1147–1157. [Google Scholar] [CrossRef]
- Harada, A.; Furue, M.; Nozakura, S.-I. Optical resolution of mandelic acid derivatives by column chromatography on crosslinked cyclodextrin gels. J. Polym. Sci. Polym. Chem. Ed. 1978, 16, 189–196. [Google Scholar] [CrossRef]
- Mizobuchi, Y.; Tanaka, M.; Shono, T. Preparation and sorption behaviour of cyclodextrin polyurethane resins. J. Chromatogr. A 1980, 194, 153–161. [Google Scholar] [CrossRef]
- Sugiura, I.; Komiyama, M.; Toshima, N.; Hirai, H. Immobilized β-Cyclodextrins. Preparation with Various Crosslinking Reagents and the Guest Binding Properties. Bull. Chem. Soc. Jpn. 1989, 62, 1643–1651. [Google Scholar] [CrossRef]
- Shaw, P.E.; Buslig, B.S. Selective removal of bitter compounds from grapefruit juice and from aqueous solution with cyclodextrin polymers and with Amberlite XAD-4. J. Agric. Food Chem. 1986, 34, 837–840. [Google Scholar] [CrossRef]
- Su, C.-S.; Yang, C.-P. Partial removal of various food components from aqueous solution using crosslinked polymers of cyclodextrins with epichlorohydrin. J. Sci. Food Agric. 1991, 54, 635–643. [Google Scholar] [CrossRef]
- Shao, Y.; Martel, B.; Morcellet, M.; Weltrowski, M.; Crini, G. Sorption of textile dyes on β-Cyclodextrin-epichlorhydrin gels. J. Incl. Phenom. Mol. Recognit. Chem. 1996, 25, 209–212. [Google Scholar] [CrossRef]
- Crini, G.; Bertini, S.; Torri, G.; Naggi, A.; Sforzini, D.; Vecchi, C.; Janus, L.; Lekchiri, Y.; Morcellet, M. Sorption of aromatic compounds in water using insoluble cyclodextrin polymers. J. Appl. Polym. Sci. 1998, 68, 1973–1978. [Google Scholar] [CrossRef]
- Li, D.Q.; Ma, M. Nanosponges: From inclusion chemistry to water purifying technology. Chemtech 1999, 29, 31–37. [Google Scholar]
- Li, D.Q.; Ma, M. Nanoporous polymers: New nanosponge absorbent media. Filtr. Sep. 1999, 36, 26–28. [Google Scholar] [CrossRef]
- Orprecio, R.; Evans, C.H. Polymer-immobilized cyclodextrin trapping of model organic pollutants in flowing water streams. J. Appl. Polym. Sci. 2003, 90, 2103–2110. [Google Scholar] [CrossRef]
- Mamba, B.B.; Krause, R.W.; Malefetse, T.J.; Nxumalo, E.N. Monofunctionalized cyclodextrin polymers for the removal of organic pollutants from water. Environ. Chem. Lett. 2007, 5, 79–84. [Google Scholar] [CrossRef]
- Li, D.; Ma, M. Nanosponges for water purification. Clean Prod. Process. 2000, 2, 112–116. [Google Scholar] [CrossRef]
- Trotta, F.; Tumiatti, W. Cross-Linked Polymers Based on Cyclodextrins for Removing Polluting Agents. U.S. Patent US20050154198A1, 14 July 2005. [Google Scholar]
- Mhlanga, S.D.; Mamba, B.B.; Krause, R.W.; Malefetse, T.J. Removal of organic contaminants from water using nanosponge cyclodextrin polyurethanes. J. Chem. Technol. Biotechnol. 2007, 82, 382–388. [Google Scholar] [CrossRef]
- Mhlongo, S.H.; Mamba, B.B.; Krause, R.W. Monitoring the prevalence of nitrosamines in South African waters and their removal using cyclodextrin polyurethanes. Phys. Chem. Earth 2009, 34, 819–824. [Google Scholar] [CrossRef]
- Berto, S.; Bruzzoniti, M.C.; Cavalli, R.; Perrachon, D.; Prenesti, E.; Sarzanini, C.; Trotta, F.; Tumiatti, W. Synthesis of new ionic β-cyclodextrin polymers and characterization of their heavy metals retention. J. Incl. Phenom. Macrocycl. Chem. 2007, 57, 631–636. [Google Scholar] [CrossRef]
- Berto, S.; Bruzzoniti, M.C.; Cavalli, R.; Perrachon, D.; Prenesti, E.; Sarzanini, C.; Trotta, F.; Tumiatti, W. Highly crosslinked ionic β-cyclodextrin polymers and their interaction with heavy metals. J. Incl. Phenom. Macrocycl. Chem. 2007, 57, 637–643. [Google Scholar] [CrossRef]
- Trotta, F.; Cavalli, R. Characterization and applications of new hyper-cross-linked cyclodextrins. Compos. Interfaces 2009, 16, 39–48. [Google Scholar] [CrossRef]
- Swaminathan, S.; Vavia, P.R.; Trotta, F.; Torne, S. Formulation of betacyclodextrin based nanosponges of itraconazole. J. Incl. Phenom. Macrocycl. Chem. 2007, 57, 89–94. [Google Scholar] [CrossRef]
- Di Nardo, G.; Roggero, C.; Campolongo, S.; Valetti, F.; Trotta, F.; Gilardi, G. Catalytic properties of catechol 1,2-dioxygenase from Acinetobacter radioresistens S13 immobilized on nanosponges. Dalt. Trans. 2009, 6507–6512. [Google Scholar] [CrossRef] [PubMed]
- Trotta, F.; Cavalli, R.; Martina, K.; Biasizzo, M.; Vitillo, J.; Bordiga, S.; Vavia, P.; Ansari, K. Cyclodextrin nanosponges as effective gas carriers. J. Incl. Phenom. Macrocycl. Chem. 2011, 71, 189–194. [Google Scholar] [CrossRef]
- Cavalli, R.; Akhter, A.K.; Bisazza, A.; Giustetto, P.; Trotta, F.; Vavia, P. Nanosponge formulations as oxygen delivery systems. Int. J. Pharm. 2010, 402, 254–257. [Google Scholar] [CrossRef]
- Seglie, L.; Martina, K.; Devecchi, M.; Roggero, C.; Trotta, F.; Scariot, V. The effects of 1-MCP in cyclodextrin-based nanosponges to improve the vase life of Dianthus caryophyllus cut flowers. Postharvest Biol. Technol. 2011, 59, 200–205. [Google Scholar] [CrossRef]
- Seglie, L.; Spadaro, D.; Trotta, F.; Devecchi, M.; Gullino, M.L.; Scariot, V. Use of 1-methylcylopropene in cyclodextrin-based nanosponges to control grey mould caused by Botrytis cinerea on Dianthus caryophyllus cut flowers. Postharvest Biol. Technol. 2012, 64, 55–57. [Google Scholar] [CrossRef]
- Ansari, K.A.; Torne, S.J.; Vavia, P.R.; Trotta, F.; Cavalli, R. Paclitaxel Loaded Nanosponges: In-Vitro Characterization and Cytotoxicity Study on MCF-7 Cell Line Culture. Curr. Drug Deliv. 2011, 8, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Mognetti, B.; Barberis, A.; Marino, S.; Berta, G.; De Francia, S.; Trotta, F.; Cavalli, R. In vitro enhancement of anticancer activity of paclitaxel by a Cremophor free cyclodextrin-based nanosponge formulation. J. Incl. Phenom. Macrocycl. Chem. 2012, 74, 201–210. [Google Scholar] [CrossRef]
- Minelli, R.; Cavalli, R.; Ellis, L.; Pettazzoni, P.; Trotta, F.; Ciamporcero, E.; Barrera, G.; Fantozzi, R.; Dianzani, C.; Pili, R. Nanosponge-encapsulated camptothecin exerts anti-tumor activity in human prostate cancer cells. Eur. J. Pharm. Sci. 2012, 47, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Torne, S.; Darandale, S.; Vavia, P.; Trotta, F.; Cavalli, R. Cyclodextrin-based nanosponges: Effective nanocarrier for Tamoxifen delivery. Pharm. Dev. Technol. 2013, 18, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Shende, P.; Deshmukh, K.; Trotta, F.; Caldera, F. Novel cyclodextrin nanosponges for delivery of calcium in hyperphosphatemia. Int. J. Pharm. 2013, 456, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Darandale, S.S.; Vavia, P.R. Cyclodextrin-based nanosponges of curcumin: Formulation and physicochemical characterization. J. Incl. Phenom. Macrocycl. Chem. 2013, 75, 315–322. [Google Scholar] [CrossRef]
- Cavallaro, V.; Trotta, F.; Gennari, M.; Di Silvestro, I.; Pellegrino, A.; Barbera, A.C. Effects of the complex nanosponges-naphthaleneacetic acid and β cyclodextrins on in vitro rhizogenesis of globe artichoke. Acta Hortic. 2013, 983, 369–372. [Google Scholar] [CrossRef]
- Lembo, D.; Swaminathan, S.; Donalisio, M.; Civra, A.; Pastero, L.; Aquilano, D.; Vavia, P.; Trotta, F.; Cavalli, R. Encapsulation of Acyclovir in new carboxylated cyclodextrin-based nanosponges improves the agent’s antiviral efficacy. Int. J. Pharm. 2013, 443, 262–272. [Google Scholar] [CrossRef]
- Swaminathan, S.; Vavia, P.R.; Trotta, F.; Cavalli, R. Nanosponges encapsulating dexamethasone for ocular delivery: Formulation design, physicochemical characterization, safety and corneal permeability assessment. J. Biomed. Nanotechnol. 2013, 9, 998–1007. [Google Scholar] [CrossRef]
- Bastiancich, C.; Scutera, S.; Alotto, D.; Cambieri, I.; Fumagalli, M.; Casarin, S.; Rossi, S.; Trotta, F.; Stella, M.; Cavalli, R.; et al. Cyclodextrin-Based Nanosponges as a Nanotechnology Strategy for Imiquimod Delivery in Pathological Scarring Prevention and Treatment. J. Nanopharm. Drug Deliv. 2015, 2, 311–324. [Google Scholar] [CrossRef]
- Conte, C.; Caldera, F.; Catanzano, O.; D’Angelo, I.; Ungaro, F.; Miro, A.; Pellosi, D.S.; Trotta, F.; Quaglia, F. β-cyclodextrin nanosponges as multifunctional ingredient in water-containing semisolid formulations for skin delivery. J. Pharm. Sci. 2014, 103, 3941–3949. [Google Scholar] [CrossRef]
- Shende, P.K.; Gaud, R.S.; Bakal, R.; Patil, D. Effect of inclusion complexation of meloxicam with β-cyclodextrin- and β-cyclodextrin-based nanosponges on solubility, in vitro release and stability studies. Colloids Surf. B Biointerfaces 2015, 136, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Castiglione, F.; Punta, C.; Melone, L.; Panzeri, W.; Rossi, B.; Trotta, F.; Mele, A. Anomalous diffusion of ibuprofen in cyclodextrin nanosponge hydrogels: An HRMAS NMR study. Beilstein J. Org. Chem. 2014, 10, 2715–2723. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Ambrosi, M.; Caldera, F.; Trotta, F.; Berrueta, L.; Gallo, B. Encapsulation of apple polyphenols in β-CD nanosponges. J. Incl. Phenom. Macrocycl. Chem. 2014, 80, 85–92. [Google Scholar] [CrossRef]
- Ansari, K.A.; Vavia, P.R.; Trotta, F.; Cavalli, R. Cyclodextrin-based nanosponges for delivery of resveratrol: In vitro characterisation, stability, cytotoxicity and permeation study. AAPS PharmSciTech 2011, 12, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Anandam, S.; Selvamuthukumar, S. Fabrication of cyclodextrin nanosponges for quercetin delivery: Physicochemical characterization, photostability, and antioxidant effects. J. Mater. Sci. 2014, 49, 8140–8153. [Google Scholar] [CrossRef]
- Sapino, S.; Carlotti, M.E.; Cavalli, R.; Ugazio, E.; Berlier, G.; Gastaldi, L.; Morel, S. Photochemical and antioxidant properties of gamma-oryzanol in beta-cyclodextrin-based nanosponges. J. Incl. Phenom. Macrocycl. Chem. 2013, 75, 69–76. [Google Scholar] [CrossRef]
- Roggero, C.M.; Di Carlo, S.; Tumiatti, V.; Tumiatti, M.; Vecchi, M.; Scariot, V.; Kapila, S. Use of Functionalised Nanosponges for the Growth, Conservation, Protection and Disinfection of Vegetable Organisms. WO Patent 2013046165, A1, 4 April 2013. [Google Scholar]
- Vercelli, M.; Gaino, W.; Contartese, V.; Gallo, L.; Di Carlo, S.; Tumiatti, V.; Larcher, F.; Scariot, V. Preliminary studies on the effect of Fe-nanosponge complex in horticulture. Acta Sci. Pol. Hortorum Cultus 2015, 14, 51–58. [Google Scholar]
- Appell, M.; Jackson, M.A. Sorption of ochratoxin A from aqueous solutions using β-cyclodextrin-polyurethane polymer. Toxins 2012, 4, 98–109. [Google Scholar] [CrossRef]
- Alongi, J.; Pošsković, M.; Frache, A.; Trotta, F. Novel flame retardants containing cyclodextrin nanosponges and phosphorus compounds to enhance EVA combustion properties. Polym. Degrad. Stab. 2010, 95, 2093–2100. [Google Scholar] [CrossRef]
- Alongi, J.; Poskovic, M.; Visakh, P.M.; Frache, A.; Malucelli, G. Cyclodextrin nanosponges as novel green flame retardants for PP, LLDPE and PA6. Carbohydr. Polym. 2012, 88, 1387–1394. [Google Scholar] [CrossRef]
- Lai, X.; Zeng, X.; Li, H.; Yin, C.; Zhang, H.; Liao, F. Synergistic effect of phosphorus-containing nanosponges on intumescent flame-retardant polypropylene. J. Appl. Polym. Sci. 2012, 125, 1758–1765. [Google Scholar] [CrossRef]
- Fulekar, M.H.; Pathak, B. Environmental Nanotechnology; CRC Press: Boca Raton, FL, USA, 2017; Volume 1, ISBN 9781498726245. [Google Scholar]
- Swaminathan, S.; Cavalli, R.; Trotta, F. Cyclodextrin-based nanosponges: A versatile platform for cancer nanotherapeutics development. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2016, 8, 579–601. [Google Scholar] [CrossRef]
- Shende, P.; Kulkarni, Y.A.; Gaud, R.S.; Deshmukh, K.; Cavalli, R.; Trotta, F.; Caldera, F. Acute and Repeated Dose Toxicity Studies of Different β-Cyclodextrin-Based Nanosponge Formulations. J. Pharm. Sci. 2015, 104, 1856–1863. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, S.; Pastero, L.; Serpe, L.; Trotta, F.; Vavia, P.; Aquilano, D.; Trotta, M.; Zara, G.P.; Cavalli, R. Cyclodextrin-based nanosponges encapsulating camptothecin: Physicochemical characterization, stability and cytotoxicity. Eur. J. Pharm. Biopharm. 2010, 74, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.R.P.; Bhingole, R.C. Nanosponge-based pediatric-controlled release dry suspension of Gabapentin for reconstitution. Drug Dev. Ind. Pharm. 2015, 41, 2029–2036. [Google Scholar] [CrossRef] [PubMed]
- Shende, P.; Chaphalkar, R.; Deshmukh, K.; Gaud, R.S. Physicochemical Investigation of Engineered Nanosuspensions Containing Model Drug, Lansoprazole. J. Dispers. Sci. Technol. 2016, 37, 504–511. [Google Scholar] [CrossRef]
- Singireddy, A.; Subramanian, S. Cyclodextrin nanosponges to enhance the dissolution profile of quercetin by inclusion complex formation. Part. Sci. Technol. 2016, 34, 341–346. [Google Scholar] [CrossRef]
- Dora, C.P.; Trotta, F.; Kushwah, V.; Devasari, N.; Singh, C.; Suresh, S.; Jain, S. Potential of erlotinib cyclodextrin nanosponge complex to enhance solubility, dissolution rate, in vitro cytotoxicity and oral bioavailability. Carbohydr. Polym. 2016, 137, 339–349. [Google Scholar] [CrossRef]
- Daga, M.; Ulllio, C.; Argenziano, M.; Dianzani, C.; Cavalli, R.; Trotta, F.; Ferretti, C.; Zara, G.P.; Gigliotti, C.L.; Ciamporcero, E.S.; et al. GSH-targeted nanosponges increase doxorubicin-induced toxicity “in vitro” and “in vivo” in cancer cells with high antioxidant defenses. Free Radic. Biol. Med. 2016, 97, 24–37. [Google Scholar] [CrossRef]
- Ferro, M.; Castiglione, F.; Pastori, N.; Punta, C.; Melone, L.; Panzeri, W.; Rossi, B.; Trotta, F.; Mele, A. Dynamics and interactions of ibuprofen in cyclodextrin nanosponges by solid-state NMR spectroscopy. Beilstein J. Org. Chem. 2017, 13, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Coviello, V.; Sartini, S.; Quattrini, L.; Baraldi, C.; Gamberini, M.C.; La Motta, C. Cyclodextrin-based nanosponges for the targeted delivery of the anti-restenotic agent DB103: A novel opportunity for the local therapy of vessels wall subjected to percutaneous intervention. Eur. J. Pharm. Biopharm. 2017, 117, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Sundararajan, M.; Thomas, P.A.; Venkadeswaran, K.; Jeganathan, K.; Geraldine, P. Synthesis and characterization of chrysin-loaded β-cyclodextrin-based nanosponges to enhance in-vitro solubility, photostability, drug release, antioxidant effects and antitumorous efficacy. J. Nanosci. Nanotechnol. 2017, 17, 8742–8751. [Google Scholar] [CrossRef]
- Shringirishi, M.; Mahor, A.; Gupta, R.; Prajapati, S.K.; Bansal, K.; Kesharwani, P. Fabrication and characterization of nifedipine loaded β-cyclodextrin nanosponges: An in vitro and in vivo evaluation. J. Drug Deliv. Sci. Technol. 2017, 41, 344–350. [Google Scholar] [CrossRef]
- Gigliotti, C.L.; Minelli, R.; Cavalli, R.; Occhipinti, S.; Barrera, G.; Pizzimenti, S.; Cappellano, G.; Boggio, E.; Conti, L.; Fantozzi, R.; et al. In vitro and in vivo therapeutic evaluation of camptothecin-encapsulated β-cyclodextrin nanosponges in prostate cancer. J. Biomed. Nanotechnol. 2016, 12, 114–127. [Google Scholar] [CrossRef]
- Gigliotti, C.L.; Ferrara, B.; Occhipinti, S.; Boggio, E.; Barrera, G.; Pizzimenti, S.; Giovarelli, M.; Fantozzi, R.; Chiocchetti, A.; Argenziano, M.; et al. Enhanced cytotoxic effect of camptothecin nanosponges in anaplastic thyroid cancer cells in vitro and in vivo on orthotopic xenograft tumors. Drug Deliv. 2017, 24, 670–680. [Google Scholar] [CrossRef]
- Dubey, P.; Sharma, H.K.; Shah, S.; Tyagi, C.K.; Chandekar, A.R.; Jadon, R.S. Formulations and evaluation of Cyclodextrin complexed Ceadroxil loaded nanosponges. Int. J. Drug Deliv. 2017, 9, 84. [Google Scholar] [CrossRef]
- Rao, M.R.P.; Shirsath, C. Enhancement of Bioavailability of Non-nucleoside Reverse Transciptase Inhibitor Using Nanosponges. AAPS PharmSciTech 2017, 18, 1728–1738. [Google Scholar] [CrossRef]
- Rana, Z.; Zahid, Z.; Jaiprakash, N. Sangshetti Mufassir, M. Enhancement of oral bioavailability of anti-HIV drug rilpivirine HCl through nanosponge formulation. Drug Dev. Ind. Pharm. 2017, 43, 2076–2084. [Google Scholar]
- Momin, M.M.; Zaheer, Z.; Zainuddin, R.; Jaiprakash, N. Extended release delivery of erlotinib glutathione nanosponge for targeting lung cancer. Artif. Cells Nanomed. Biotechnol. 2017, 46, 1064–1075. [Google Scholar] [CrossRef]
- Mady, F.M.; Ragab, S.; Ibrahim, M. Cyclodextrin-based nanosponge for improvement of solubility and oral bioavailability of Ellagic acid. Pak. J. Pharm. Sci. 2018, 31, 2069–2076. [Google Scholar]
- Deshmukh, K.; Shende, P. Toluene diisocyanate cross-linked β-cyclodextrin nanosponges as a pH-sensitive carrier for naproxen. Mater. Res. Express 2018, 5. [Google Scholar] [CrossRef]
- Cecone, C.; Caldera, F.; Trotta, F.; Bracco, P.; Zanetti, M. Controlled Release of DEET Loaded on Fibrous Mats from Electrospun PMDA/Cyclodextrin Polymer. Molecules 2018, 23, 1694. [Google Scholar] [CrossRef]
- Rao, M.R.P.; Chaudhari, J.; Trotta, F.; Caldera, F. Investigation of Cyclodextrin-Based Nanosponges for Solubility and Bioavailability Enhancement of Rilpivirine. AAPS PharmSciTech 2018, 19, 2358–2369. [Google Scholar] [CrossRef] [PubMed]
- Zidan, M.F.; Ibrahim, H.M.; Afouna, M.I.; Ibrahim, E.A. In vitro and in vivo evaluation of cyclodextrin-based nanosponges for enhancing oral bioavailability of atorvastatin calcium. Drug Dev. Ind. Pharm. 2018, 44, 1243–1253. [Google Scholar] [CrossRef]
- Mendes, C.; Meirelles, G.C.; Germano, C.; Assreuy, J.; Silva, M.A.S.; Ponchel, G. Cyclodextrin based nanosponge of norfloxacin: Intestinal permeation enhancement and improved antibacterial activity. Carbohydr. Polym. 2018, 195, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Ren, X.; Guo, T.; Wu, L.; Shakya, S.; He, Y. Biofunctionalization of β -cyclodextrin nanosponges using cholesterol. Carbohydr. Polym. 2018, 190, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Nait Bachir, Y.; Nait Bachir, R.; Hadj-Ziane-Zafour, A. Nanodispersions stabilized by β-cyclodextrin nanosponges: Application for simultaneous enhancement of bioactivity and stability of sage essential oil. Drug Dev. Ind. Pharm. 2019, 45, 333–347. [Google Scholar] [CrossRef]
- Pushpalatha, R.; Selvamuthukumar, S.; Kilimozhi, D. Cross-linked, cyclodextrin-based nanosponges for curcumin delivery—Physicochemical characterization, drug release, stability and cytotoxicity. J. Drug Deliv. Sci. Technol. 2018, 45, 45–53. [Google Scholar] [CrossRef]
- Pushpalatha, R.; Selvamuthukumar, S.; Kilimozhi, D. Carbonyl and carboxylate crosslinked cyclodextrin as a nanocarrier for resveratrol: In silico, in vitro and in vivo evaluation. J. Incl. Phenom. Macrocycl. Chem. 2018, 92, 261–272. [Google Scholar] [CrossRef]
- Rezaei, A.; Varshosaz, J.; Fesharaki, M.; Farhang, A.; Jafari, S.M. Improving the solubility and in vitro cytotoxicity (anticancer activity) of ferulic acid by loading it into cyclodextrin nanosponges. Int. J. Nanomed. 2019, 14, 4589–4599. [Google Scholar] [CrossRef] [PubMed]
- Gholibegloo, E.; Mortezazadeh, T.; Salehian, F.; Forootanfar, H.; Firoozpour, L.; Foroumadi, A.; Ramazani, A.; Khoobi, M. Folic acid decorated magnetic nanosponge: An efficient nanosystem for targeted curcumin delivery and magnetic resonance imaging. J. Colloid Interface Sci. 2019, 556, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Gholibegloo, E.; Mortezazadeh, T.; Salehian, F.; Ramazani, A.; Amanlou, M.; Khoobi, M. Improved curcumin loading, release, solubility and toxicity by tuning the molar ratio of cross-linker to β-cyclodextrin. Carbohydr. Polym. 2019, 213, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Dhakar, N.K.; Caldera, F.; Bessone, F.; Cecone, C.; Rubin, A.; Cavalli, R.; Dianzani, C.; Trotta, F. Evaluation of solubility enhancement, antioxidant activity, and cytotoxicity studies of kynurenic acid loaded cyclodextrin nanosponge. Carbohydr. Polym. 2019, 224. [Google Scholar] [CrossRef]
- Sherje, A.P.; Surve, A.; Shende, P. CDI cross-linked β-cyclodextrin nanosponges of paliperidone: Synthesis and physicochemical characterization. J. Mater. Sci. Med. 2019, 30. [Google Scholar] [CrossRef]
- Argenziano, M.; Haimhoffer, A.; Bastiancich, C.; Caldera, F.; Trotta, F.; Scutera, S.; Alotto, D.; Fumagalli, M.; Musso, T.; Castagnoli, C.; et al. In Vitro Enhanced Skin Permeation and Retention of Imiquimod Loaded in β -Cyclodextrin Nanosponge Hydrogel. Pharmaceutics 2019, 11, 138. [Google Scholar] [CrossRef]
- Garcia-Fernandez, M.J.; Tabary, N.; Chai, F.; Cazaux, F.; Blanchemain, N.; Flament, M.P.; Martel, B. New multifunctional pharmaceutical excipient in tablet formulation based on citric acid-cyclodextrin polymer. Int. J. Pharm. 2016, 511, 913–920. [Google Scholar] [CrossRef]
- Gangadharappa, H.V.; Chandra Prasad, S.M.; Singh, R.P. Formulation, in vitro and in vivo evaluation of celecoxib nanosponge hydrogels for topical application. J. Drug Deliv. Sci. Technol. 2017, 41, 488–501. [Google Scholar] [CrossRef]
- Dhakar, N.K.; Matencio, A.; Caldera, F.; Argenziano, M.; Cavalli, R.; Dianzani, C.; Zanetti, M.; López-Nicolás, J.M.; Trotta, F. Comparative evaluation of solubility, cytotoxicity and photostability studies of resveratrol and oxyresveratrol loaded nanosponges. Pharmaceutics 2019, 11, 545. [Google Scholar] [CrossRef]
- Ncube, P.; Krause, R.W.M.; Mamba, B.B. Detection of chloroform in water using an azo dye-modified β-cyclodextrin—Epichlorohydrin copolymer as a fluorescent probe. Phys. Chem. Earth 2014, 67–69, 79–85. [Google Scholar] [CrossRef]
- Trotta, F.; Caldera, F.; Dianzani, C.; Argenziano, M.; Barrera, G.; Cavalli, R. Glutathione Bioresponsive Cyclodextrin Nanosponges. Chempluschem 2016, 81, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Argenziano, M.; Lombardi, C.; Ferrara, B.; Trotta, F.; Caldera, F.; Blangetti, M.; Koltai, H.; Kapulnik, Y.; Yarden, R.; Gigliotti, L.; et al. Glutathione/pH-responsive nanosponges enhance strigolactone delivery to prostate cancer cells. Oncotarget 2018, 9, 35813–35829. [Google Scholar] [CrossRef] [PubMed]
- Fontana, R.M.; Milano, N.; Barbara, L.; Di Vincenzo, A.; Gallo, G.; Meo, P. Lo Cyclodextrin-Calixarene Nanosponges as Potential Platforms for pH-Dependent Delivery of Tetracycline. ChemistrySelect 2019, 4, 9743–9747. [Google Scholar] [CrossRef]
- Trotta, F.; Caldera, F.; Cavalli, R.; Soster, M.; Riedo, C.; Biasizzo, M.; Uccello Barretta, G.; Balzano, F.; Brunella, V. Molecularly imprinted cyclodextrin nanosponges for the controlled delivery of L-DOPA: Perspectives for the treatment of Parkinson’s disease. Expert Opin. Drug Deliv. 2016, 13, 1671–1680. [Google Scholar] [CrossRef]
- Byrne, M.E.; Park, K.; Peppas, N.A. Molecular imprinting within hydrogels. Adv. Drug Deliv. Rev. 2002, 54, 149–161. [Google Scholar] [CrossRef]
- Seong, H.; Lee, H.B.; Park, K. Glucose binding to molecularly imprinted polymers. J. Biomater. Sci. Polym. Ed. 2002, 13, 637–649. [Google Scholar] [CrossRef][Green Version]
- Deshmukh, K.; Tanwar, Y.S.; Shende, P.; Cavalli, R. Biomimetic estimation of glucose using non-molecular and molecular imprinted polymer nanosponges. Int. J. Pharm. 2015, 494, 244–248. [Google Scholar] [CrossRef]
- Rousseau, J.; Menuel, S.; Rousseau, C.; Hapiot, F.; Monflier, E. Cyclodextrins as Porous Material for Catalysis. In Organic Nanoreactors: From Molecular to Supramolecular Organic Compounds; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 15–42. ISBN 9780128018101. [Google Scholar]
- Zaidi, S.A. Molecular imprinting: A useful approach for drug delivery. Mater. Sci. Energy Technol. 2020, 3, 72–77. [Google Scholar] [CrossRef]
- Pei, M.; Pai, J.Y.; Du, P.; Liu, P. Facile Synthesis of Fluorescent Hyper-Cross-Linked β-Cyclodextrin-Carbon Quantum Dot Hybrid Nanosponges for Tumor Theranostic Application with Enhanced Antitumor Efficacy. Mol. Pharm. 2018, 15, 4084–4091. [Google Scholar] [CrossRef]
- Peila, R.; Scordino, P.; Shanko, D.B.; Caldera, F.; Trotta, F.; Ferri, A. Synthesis and characterization of β-cyclodextrin nanosponges for N,N-diethyl-meta-toluamide complexation and their application on polyester fabrics. React. Funct. Polym. 2017, 119, 87–94. [Google Scholar] [CrossRef]
- Cecone, C.; Caldera, F.; Anceschi, A.; Scalarone, D.; Trotta, F.; Bracco, P.; Zanetti, M. One-step facile process to obtain insoluble polysaccharides fibrous mats from electrospinning of water-soluble PMDA/cyclodextrin polymer. J. Appl. Polym. Sci. 2018, 135, 46490. [Google Scholar] [CrossRef]
- Cecone, C.; Zanetti, M.; Anceschi, A.; Caldera, F.; Trotta, F.; Bracco, P. Microfibers of microporous carbon obtained from the pyrolysis of electrospun β-cyclodextrin/pyromellitic dianhydride nanosponges. Polym. Degrad. Stab. 2019, 161, 277–282. [Google Scholar] [CrossRef]
- Ma, M.; Li, D.Q. New organic nanoporous polymers and their inclusion complexes. Chem. Mater. 1999, 11, 872–874. [Google Scholar] [CrossRef]
- Femminò, S.; Penna, C.; Bessone, F.; Caldera, F.; Dhakar, N.; Cau, D.; Pagliaro, P.; Cavalli, R.; Trotta, F. α-Cyclodextrin and α-Cyclodextrin Polymers as Oxygen Nanocarriers to Limit Hypoxia/Reoxygenation Injury: Implications from an In Vitro Model. Polymers 2018, 10, 211. [Google Scholar] [CrossRef]
- Sadjadi, S.; Heravi, M.M.; Daraie, M. Cyclodextrin nanosponges: A potential catalyst and catalyst support for synthesis of xanthenes. Res. Chem. Intermed. 2017, 43, 843–857. [Google Scholar] [CrossRef]
- Silva, F.; Caldera, F.; Trotta, F.; Nerín, C.; Domingues, F.C. Encapsulation of coriander essential oil in cyclodextrin nanosponges: A new strategy to promote its use in controlled-release active packaging. Innov. Food Sci. Emerg. Technol. 2019, 56. [Google Scholar] [CrossRef]
- Simionato, I.; Domingues, F.C.; Nerín, C.; Silva, F. Encapsulation of cinnamon oil in cyclodextrin nanosponges and their potential use for antimicrobial food packaging. Food Chem. Toxicol. 2019, 132. [Google Scholar] [CrossRef]
- Silva, F.; Simionato, I.; Domeño, C.; Domingues, F.C. Cyclodextrin nanosponges as a new encapsulating agent for essential oils and their effectiveness against foodborne pathogens. Facta Univ. 2018, 16, 42. [Google Scholar]
- Salgin, S.; Salgin, U.; Vatansever, Ö. Synthesis and Characterization of β-Cyclodextrin Nanosponge and Its Application for the Removal of p-Nitrophenol from Water. Clean Soil Air Water 2017, 45. [Google Scholar] [CrossRef]
- Pedrazzo, A.R.; Smarra, A.; Caldera, F.; Musso, G.; Dhakar, N.K.; Cecone, C.; Hamedi, A.; Corsi, I.; Trotta, F. Eco-Friendly beta-cyclodextrin and Linecaps Polymers for the Removal of Heavy Metals. Polymers 2019, 11, 1658. [Google Scholar] [CrossRef]
- Liao, X.; Zhang, Q. Mesoporous Polymer Nanosponges Immobilized with Functional Polyols for Rapid Removal of Boric Acid and Organic Micropollutants. ACS Appl. Polym. Mater. 2019, 1, 2089–2098. [Google Scholar] [CrossRef]
- Massella, D.; Argenziano, M.; Ferri, A.; Guan, J.; Giraud, S.; Cavalli, R.; Barresi, A.A.; Salaün, F. Bio-Functional Textiles: Combining Pharmaceutical Nanocarriers with Fibrous Materials for Innovative Dermatological Therapies. Pharmaceutics 2019, 11, 403. [Google Scholar] [CrossRef] [PubMed]
- Mihailiasa, M.; Caldera, F.; Li, J.; Peila, R.; Ferri, A.; Trotta, F. Preparation of functionalized cotton fabrics by means of melatonin loaded β-cyclodextrin nanosponges. Carbohydr. Polym. 2016, 142, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Gentili, A. Cyclodextrin-based sorbents for solid phase extraction. J. Chromatogr. A 2019, 460654. [Google Scholar] [CrossRef]
- Sadjadi, S.; Heravi, M.M.; Daraie, M. A novel hybrid catalytic system based on immobilization of phosphomolybdic acid on ionic liquid decorated cyclodextrin-nanosponges: Efficient catalyst for the green synthesis of benzochromeno-pyrazole through cascade reaction: Triply green. J. Mol. Liq. 2017, 231, 98–105. [Google Scholar] [CrossRef]
- Kumar, S.; Rao, R. Analytical tools for cyclodextrin nanosponges in pharmaceutical field: A review. J. Incl. Phenom. Macrocycl. Chem. 2019, 94, 11–30. [Google Scholar] [CrossRef]
- Zhao, F.; Repo, E.; Yin, D.; Meng, Y.; Jafari, S.; Sillanpää, M. EDTA-Cross-Linked β-Cyclodextrin: An Environmentally Friendly Bifunctional Adsorbent for Simultaneous Adsorption of Metals and Cationic Dyes. Environ. Sci. Technol. 2015, 49, 10570–10580. [Google Scholar] [CrossRef]
- Venuti, V.; Rossi, B.; D’Amico, F.; Mele, A.; Castiglione, F.; Punta, C.; Melone, L.; Crupi, V.; Majolino, D.; Trotta, F.; et al. Combining Raman and infrared spectroscopy as a powerful tool for the structural elucidation of cyclodextrin-based polymeric hydrogels. Phys. Chem. Chem. Phys. 2015, 17, 10274–10282. [Google Scholar] [CrossRef]
- Hayiyana, Z.; Choonara, Y.; Makgotloe, A.; Toit, L.; Kumar, P.; Pillay, V. Ester-Based Hydrophilic Cyclodextrin Nanosponges for Topical Ocular Drug Delivery. Curr. Pharm. Des. 2017, 22, 6988–6997. [Google Scholar] [CrossRef]
- Pawar, S.; Shende, P.; Trotta, F. Diversity of β -cyclodextrin-based nanosponges for transformation of actives. Int. J. Pharm. 2019, 565, 333–350. [Google Scholar] [CrossRef]
- Morin-Crini, N.; Winterton, P.; Fourmentin, S.; Wilson, L.D.; Fenyvesi, É.; Crini, G. Water-insoluble β-cyclodextrin–epichlorohydrin polymers for removal of pollutants from aqueous solutions by sorption processes using batch studies: A review of inclusion mechanisms. Prog. Polym. Sci. 2018, 78, 1–23. [Google Scholar] [CrossRef]
- Deshmukh, K.; Tanwar, Y.S.; Sharma, S.; Shende, P.; Cavalli, R. Functionalized nanosponges for controlled antibacterial and antihypocalcemic actions. Biomed. Pharmacother. 2016, 84, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Saladino, M.L.; Chillura Martino, D.; Lo Meo, P.; Noto, R. Polyaminocyclodextrin nanosponges: Synthesis, characterization and pH-responsive sequestration abilities. RSC Adv. 2016, 6, 49941–49953. [Google Scholar] [CrossRef]
- Junthip, J.; Promma, W.; Sonsupap, S.; Boonyanusith, C. Adsorption of paraquat from water by insoluble cyclodextrin polymer crosslinked with 1,2,3,4-butanetetracarboxylic acid. Iran. Polym. J. 2019, 28, 213–223. [Google Scholar] [CrossRef]
- Singireddy, A.; Pedireddi, S.R.; Subramanian, S. Optimization of reaction parameters for synthesis of Cyclodextrin nanosponges in controlled nanoscopic size dimensions. J. Polym. Res. 2019, 26. [Google Scholar] [CrossRef]
| Year | Nanosponge/Cross-Linker | Drug | Indication | References |
|---|---|---|---|---|
| 2010–2015 | β-CD/DPC | Camptothecin | Pharmaceutical application | [46,70] |
| β-CD/CDI | Resveratrol | Pharmaceutical application | [58] | |
| Fluorescent NS | Paclitaxel | Pharmaceutical application | [45] | |
| Carboxylated nanosponges | Acyclovir | Antivirial efficacy | [51] | |
| β-CD/CDI | Rutin, phloridzin and chlorogenic acid | Anti-cancer, anti-diabetic, antiobesity, neuronal protective properties etc. | [57] | |
| β-CD/DPC | Gabapentin | Treatment of partial seizures in pediatric | [71] | |
| 2016 | β-CD/PMDA | Lansoprazole | Gastric ulcers | [72] |
| β-CD/DPC | Quercetin | Cancer treatment | [73] | |
| β-CD /CDI | Erlotinib hydrochloride (ERL) | Cancer treatment | [74] | |
| GSH–NS (β-CD/PMDA) | Doxorubicin | Anticancer activity | [75] | |
| 2017 | β-CD/EDTA | Ibuprofen | diseases | [76] |
| β-CD/DPC | 2-(3,4-dimethoxyphenyl)-3-phenyl-4H-pyrido [1,2-a] pyrimidin-4-one (DB103) | Cardiovascular diseases, drug-eluting stents (DES) | [77] | |
| β-CD/DPC | Chrysin | Optimal anti-oxidant and anti-tumorous properties | [78] | |
| β-CD/DPC | Nifedipine | Treatment of angina pectoris and hypertension | [79] | |
| β-CD/CDI | Camptothecin (CPT) | Antitumor efficacy, inhibition effect on prostate cancer | [80,81] | |
| β-CD/DPC | Cefadroxil (CFD) | Against variety of Gram-positive and Gram-negative bacteria | [82] | |
| β-CD/DPC | Efavirenz and Rilpivirine HCl | HIV | [83,84] | |
| β-CD/PMDA | Erlotinib glutathione | Cancer | [85] | |
| 2018 | β-CD/DMC | Ellagic acid | Cancer | [86] |
| β-CD/DPC | Mebendazole | Lymphatic worm infestations | [16] | |
| β-CD/TDI | Naproxen | Inflammation | [87] | |
| Electrospun | N,N-diethyl-3-toluamide | Infectious diseases | [88] | |
| β-CD/PMDA | ||||
| β-CD/CDI; PMDA | Rilpivrine | HIV | [89] | |
| β-CD/CDI | Atorvastatin calcium | Dyslipidemia | [90] | |
| β-CD/DPC | Norfloxacin | Urinary tract infections | [91] | |
| β-CD/CDI | Doxorubicin | Cancer | [92] | |
| 2019 | β-CD/NDCA | Sage essential oil | Diabetes | [93] |
| β-CD/DPC; PMDA | Curcumin and Resveratrol | Cancer | [15,94,95] | |
| β-CD / DPC | Ferulic acid and Imatinib Mesylate | Cancer | [17,96] | |
| β-CD/EPI | Curcumin | Cancer | [97,98] | |
| β-CD/CDI | Kynurenic acid and Paliperidone | Neurological disorders and Schizophrenia | [99,100] | |
| β-CD/PMDA | Imiquimod | Topical diseases | [101] |
| Years | Nanosponge/Crosslinker | Adsorbate | Field | References |
|---|---|---|---|---|
| 1980–2000 | CDs/HMDI, phenyl isocyanate, EPI | Naringin, limonin, debittering of grapefruit juice, | Food | [23] |
| CDs/EPI | Caffeine, vanillin and theobromine | [24] | ||
| CDs / EPI, HMDI and Phenyl isocyanate | Textile dyes | Textile | [25] | |
| Aromatic pollutants, such as phenol, p-nitrophenol, benzoic acid, p-nitrobenzoic acid, β-naphthol, chlorophenols and 4-tert-butylbenzoic acid | Environment | [26] | ||
| CDs/HMDI or toluene 2,6-diisocyanate | Organic pollutants | Water Treatment (Environment) | [119] | |
| 2000–2009 | β-CD/EPI | Naphthalene, 2-naphthol and naproxen | Water Treatment (Environment) | [29] |
| CDs/CDI, DMC | Chlorinated persistent organic pollutants (POPs) | Water Treatment (Environment) | [32] | |
| CD/HMDI or toluene-2,4-diisocyanate | p-nitrophenol and pentachlorophenol, | [30] | ||
| β-CD/PMDA | Heavy metals (Al (III), Mn (II), Co (II), Ni (II), Cu (II), Cd (II) etc.) | Water Treatment (Environment) | [35] | |
| Carcinogenic N-nitrosodimethylamine (NDMA) | Water Treatment (Environment) | [34] | ||
| Carbonate NSs | Increase the thermostability, pH stability and storage stability of catechol 1,2-dioxygenase | As a substrate for enzyme immobilization | [39] | |
| 2010–2019 | β-CD/DPC | Phosphorus derivatives | Flame retardancy | [64] |
| α, β, γ-CD / CDI; PMDA | Oxygen | Hypoxia/Reoxygenation and Biomedical application | [41,120] | |
| β-CD /CDI | Oxygen, carbon dioxide and 1-methylcyclopropene 1-MCP carriers. | biomedical, environmental and floriculture application | [40] | |
| β-CD/PMDA | Iron fertilizer | Agricultural application | [62] | |
| CD/DPC | Xanthene derivatives | Catalytic Activity | [121] | |
| CD/CDI | Coriander essential oil | food packaging | [122] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krabicová, I.; Appleton, S.L.; Tannous, M.; Hoti, G.; Caldera, F.; Rubin Pedrazzo, A.; Cecone, C.; Cavalli, R.; Trotta, F. History of Cyclodextrin Nanosponges. Polymers 2020, 12, 1122. https://doi.org/10.3390/polym12051122
Krabicová I, Appleton SL, Tannous M, Hoti G, Caldera F, Rubin Pedrazzo A, Cecone C, Cavalli R, Trotta F. History of Cyclodextrin Nanosponges. Polymers. 2020; 12(5):1122. https://doi.org/10.3390/polym12051122
Chicago/Turabian StyleKrabicová, Ilona, Silvia Lucia Appleton, Maria Tannous, Gjylije Hoti, Fabrizio Caldera, Alberto Rubin Pedrazzo, Claudio Cecone, Roberta Cavalli, and Francesco Trotta. 2020. "History of Cyclodextrin Nanosponges" Polymers 12, no. 5: 1122. https://doi.org/10.3390/polym12051122
APA StyleKrabicová, I., Appleton, S. L., Tannous, M., Hoti, G., Caldera, F., Rubin Pedrazzo, A., Cecone, C., Cavalli, R., & Trotta, F. (2020). History of Cyclodextrin Nanosponges. Polymers, 12(5), 1122. https://doi.org/10.3390/polym12051122








