Microwave Irradiation Synthesis and Characterization of Reduced-(Graphene Oxide-(Polystyrene-Polymethyl Methacrylate))/Silver Nanoparticle Nanocomposites and Their Anti-Microbial Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of RGO/AgNPs
2.3. Preparation of RGO/AgNPs-(PS-PMMA)
2.4. Preparation of R-(GO-(PS-PMMA)-AgNPs by MWI
2.5. Material Characterization and Testing
3. Results and Discussion
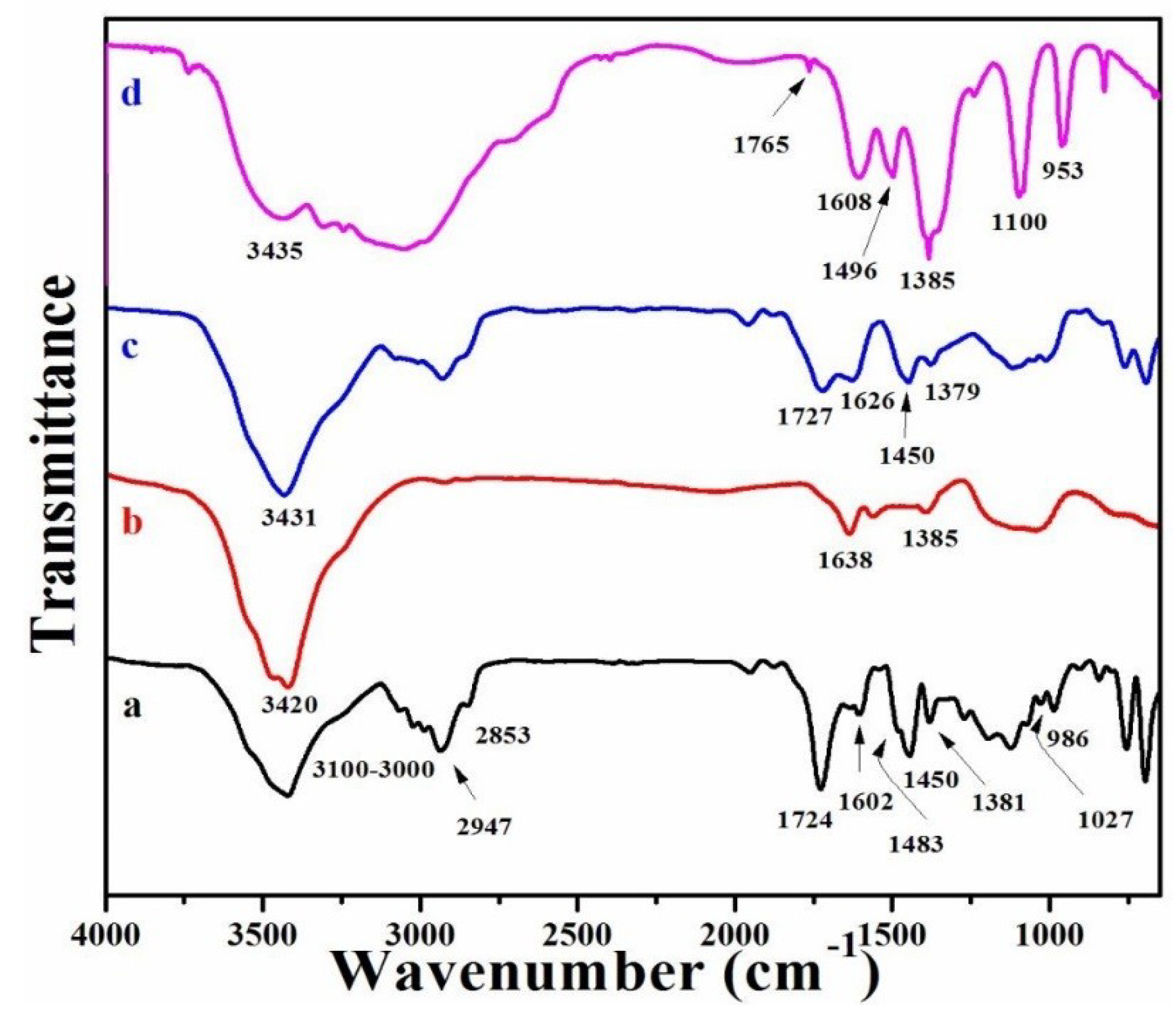
3.1. FT-IR Spectra
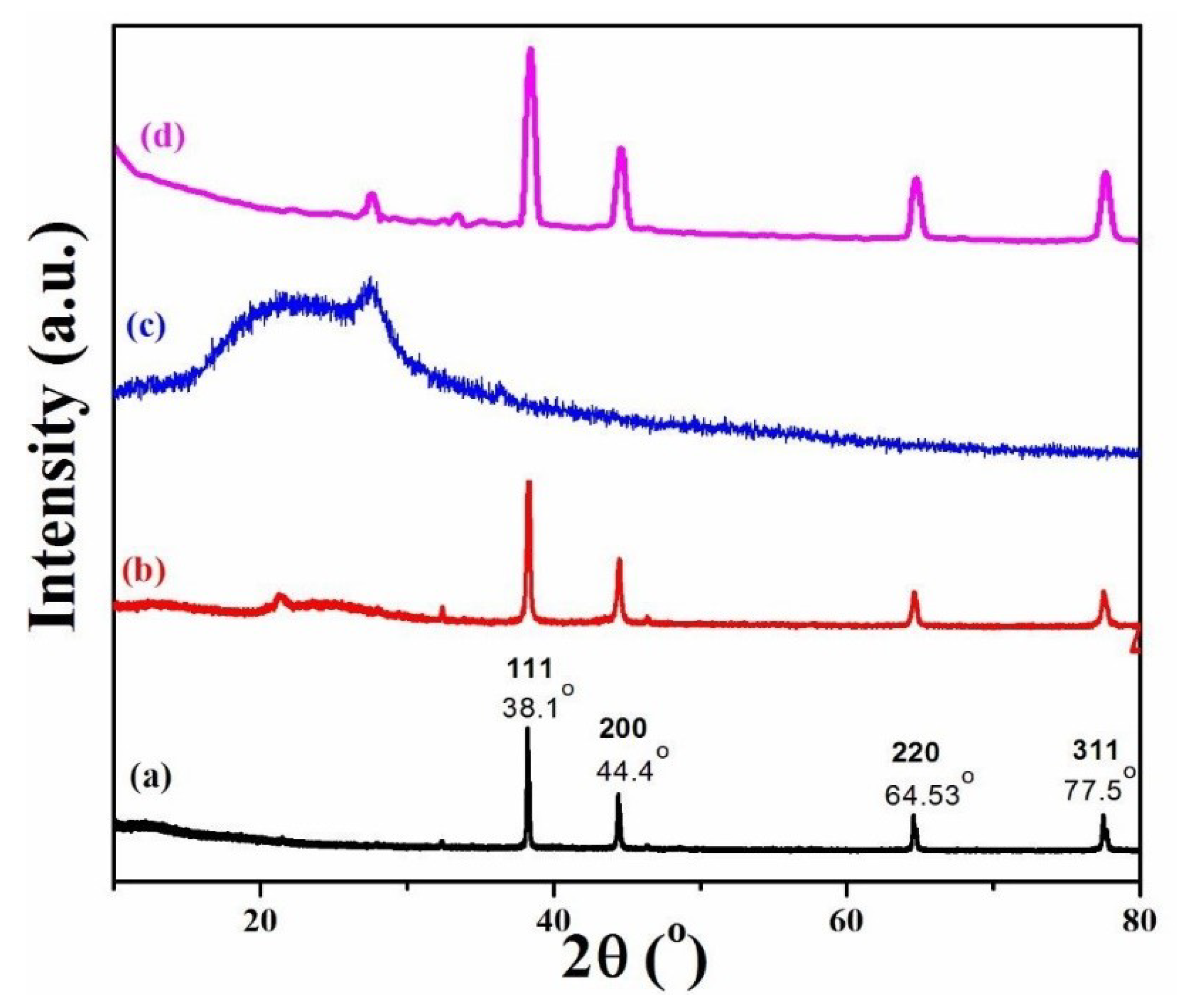
3.2. XRD
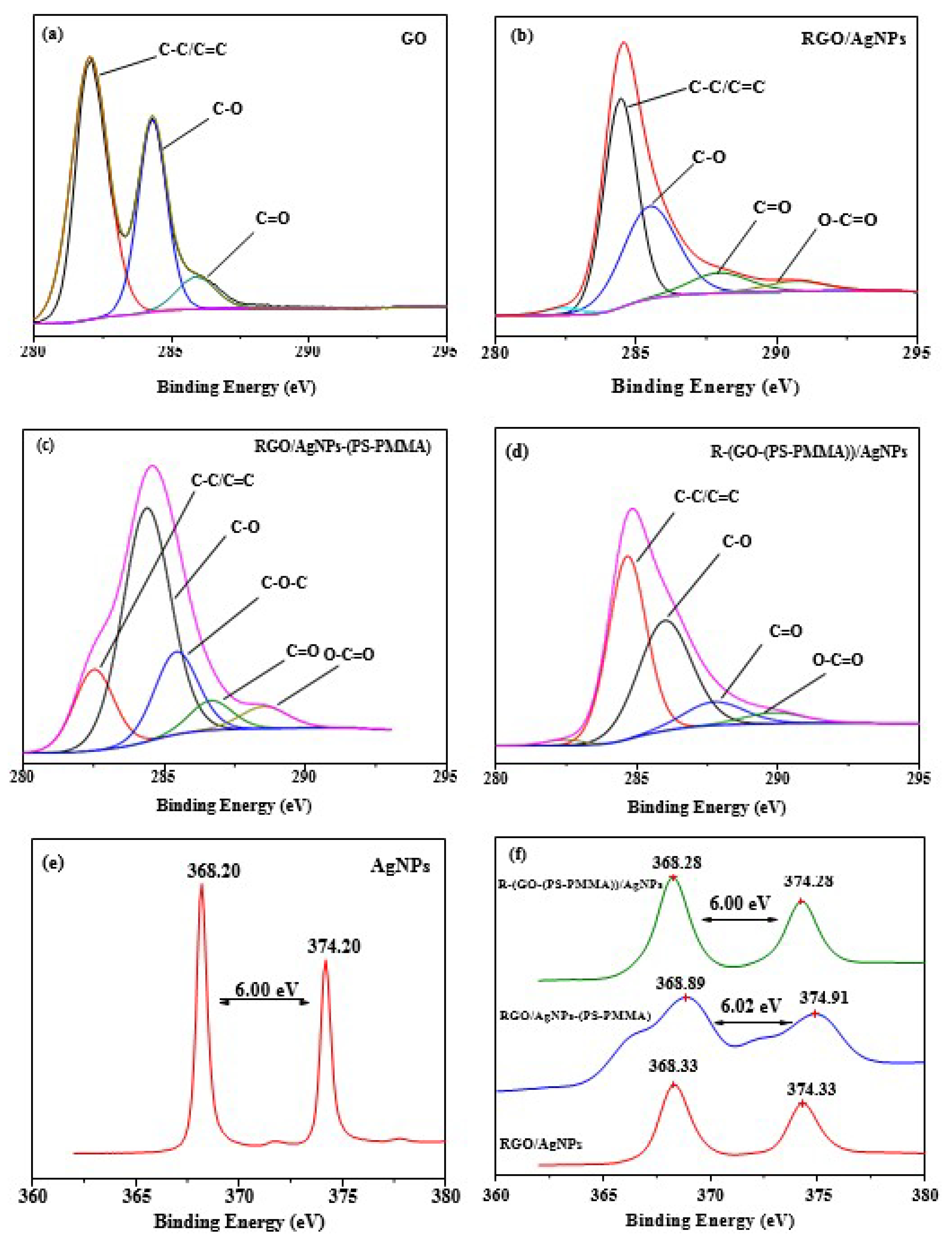
3.3. X-ray Photoelectron Spectra (XPS)
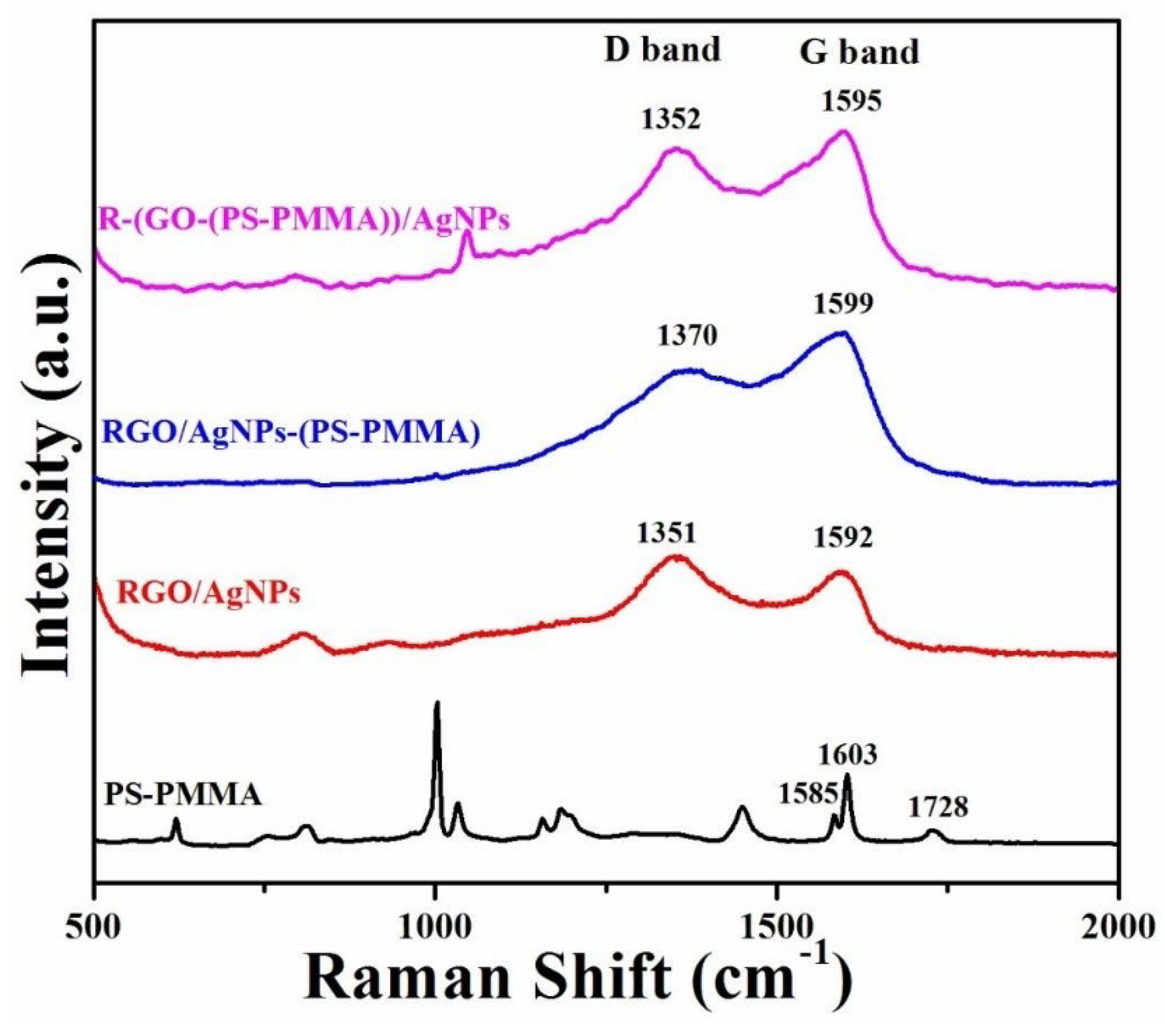
3.4. Raman Spectroscopy
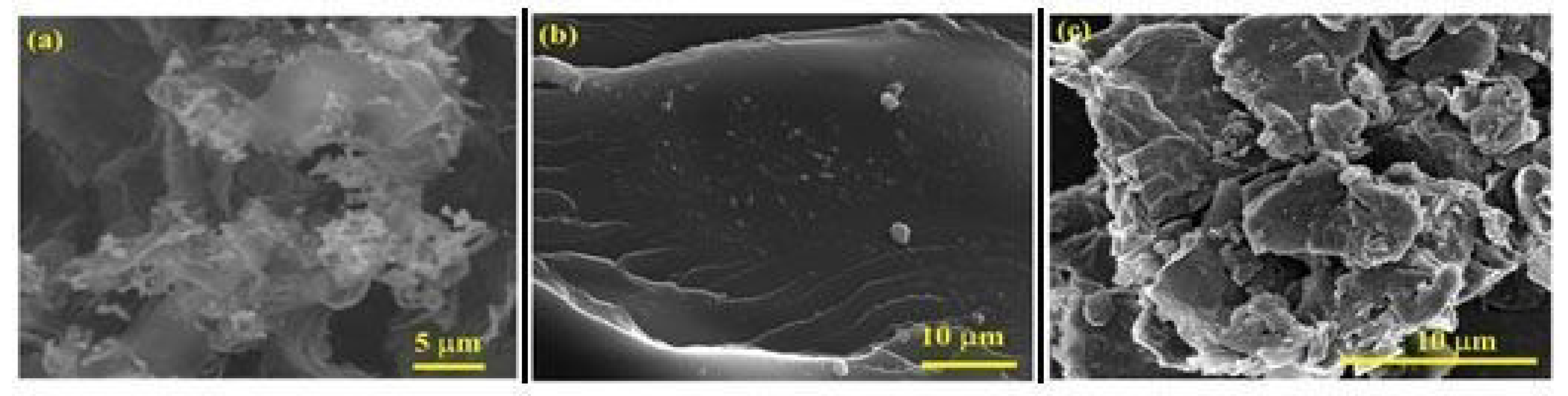
3.5. Scanning Electron Microscopy
3.6. High-Resolution Transmission Electron Microscopy (HR-TEM)
3.7. Thermal Properties of the Nanocomposites
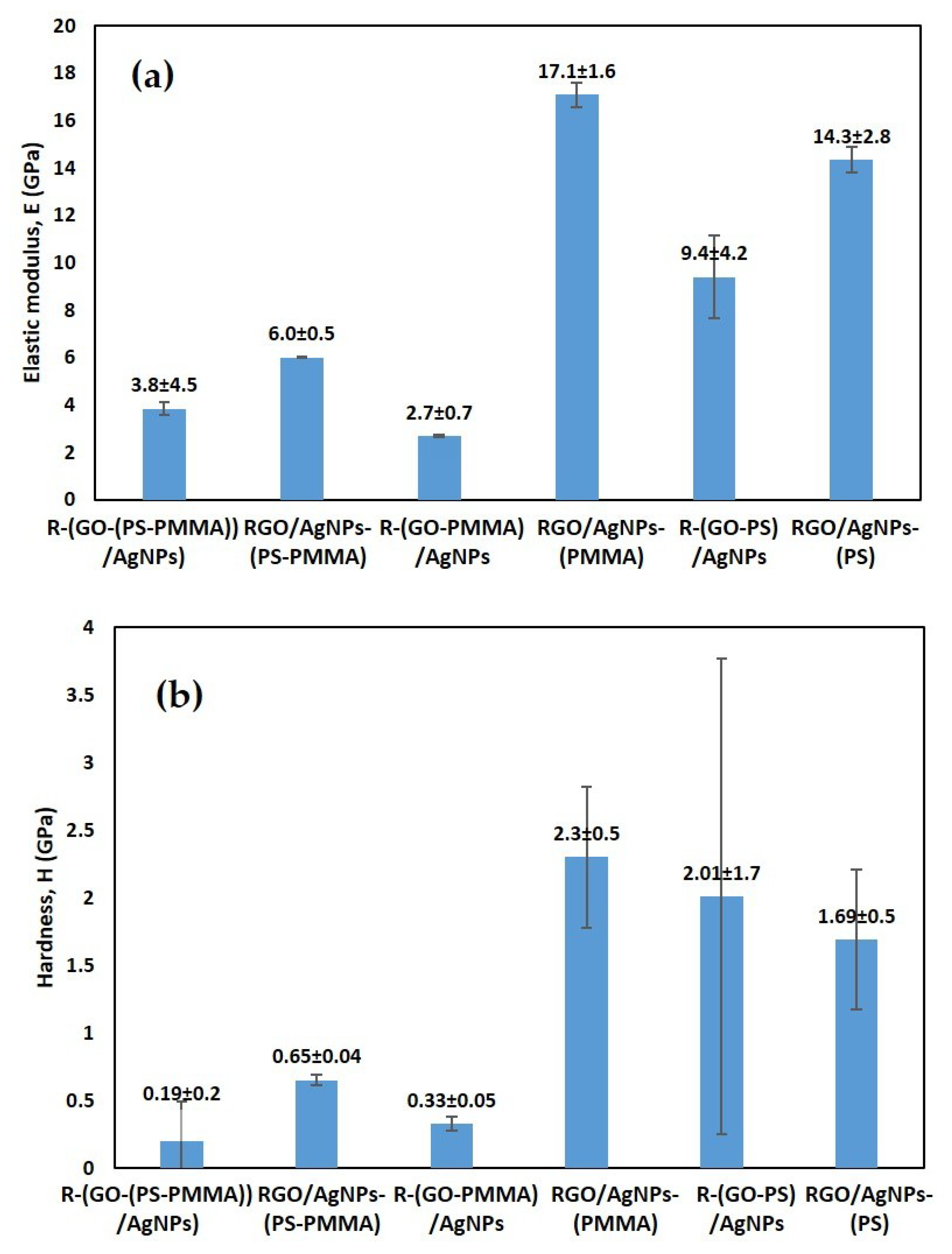
3.8. Mechanical Properties of the Nanocomposites
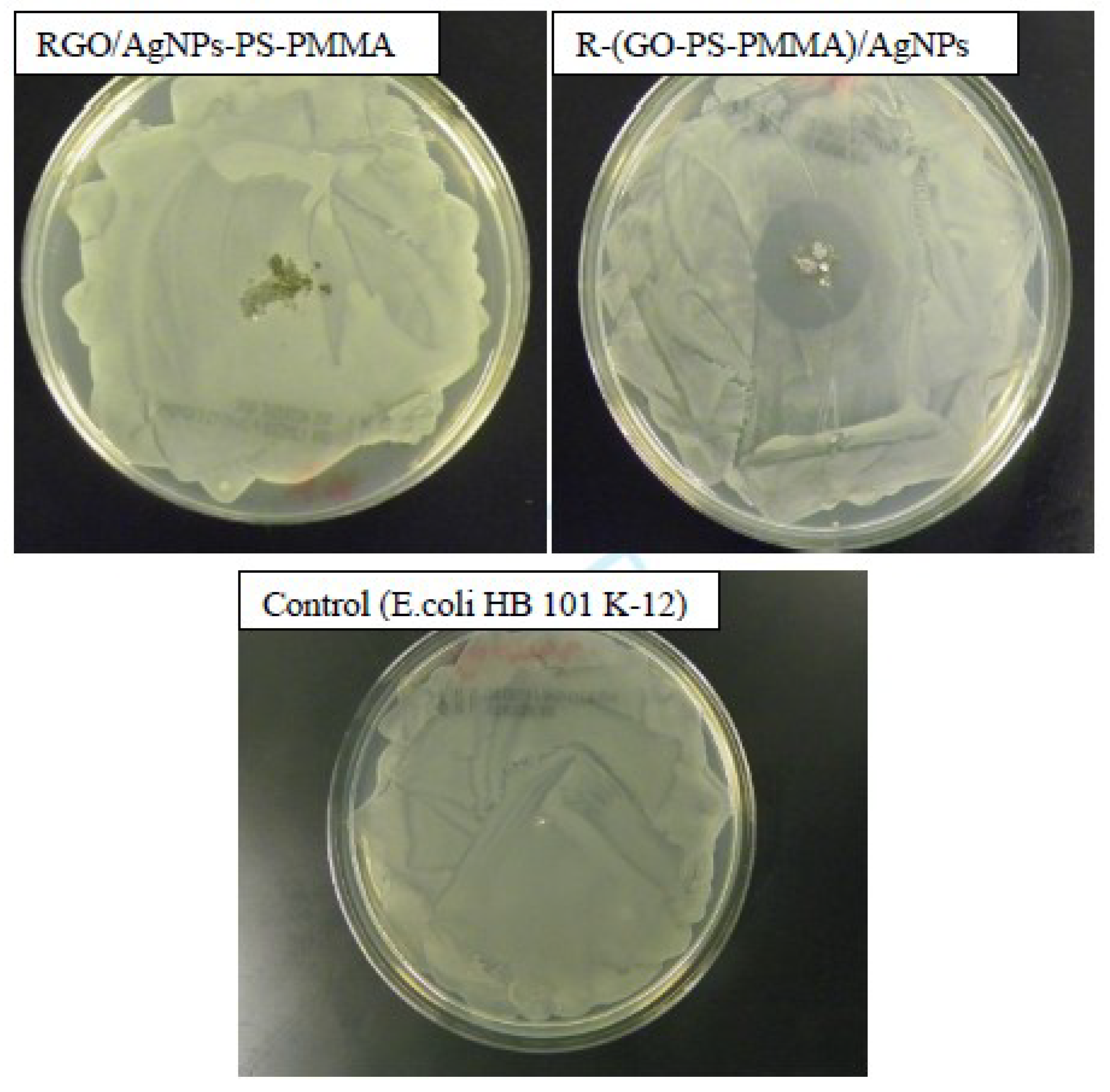
3.9. Antibacterial Activities of the Nanocomposites
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kamp, G.; Dietze, B.; Große-Siestrup, C.; Wendt, C.; Martiny, H. Microbicidal Activity of a New Silver- Containing Polymer, SPI-ARGENT II. Antimicrob. Agents Chemother. 1998, 42, 2440–2442. [Google Scholar] [CrossRef]
- Fang, N.; Lee, H.; Sun, C.; Zhang, X. Sub-Diffraction limited Optical Imaging with Silver Superlens. Science 2005, 308, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Gates, B.; Mayers, B.; Xia, Y. Crystalline Silver Nanowires by Soft Solution Processing. Nano Lett. 2002, 2, 165–168. [Google Scholar] [CrossRef]
- Nicewarner-Pena, S.R.; Griffith Freemean, R.; Reiss, B.D.; He, L.; Pena, D.J.; Walton, I.D.; Cromer, R.; Keating, C.D.; Natan, M.J. Submicrometer Metallic Barcode. Science 2001, 294, 137–141. [Google Scholar] [CrossRef]
- Mock, J.J.; Oldenburg, S.J.; Smith, D.R.; Schultz, D.A.; Schultz, S. Composite Plasmon Resonant Nanowires. Nano Lett. 2002, 5, 465–469. [Google Scholar] [CrossRef]
- Makvandi, P.; Wang, C.-Y.; Ehsan, N.Z.; Borzacchiello, A.; Niu, L.; Tay, F.R. Metal-Based Nanomaterials in Biomedical Applications: Antimicrobial Activity and Cytotoxicity Aspects. Adv. Funct. Mater. 2020. [Google Scholar] [CrossRef]
- Ehsan, N.Z.; Jamaledin, R.; Nazerzadeh, P.; Afjeh-Dana, E.; Ashtari, B.; Hosseinzadeh, M.; Vecchione, R.; Wu, A.; Borzacchiello, A.; Makvandi, P. Metal-Based Nanostructures/PLGA Nanocomposites: Antimicrobial Acitivity, Cytotoxicity, And Their Biomedical Applications. ACS Appl. Mater. Interfaces 2020, 12, 3279–3300. [Google Scholar]
- Makvandi, P.; Ali, G.W.; Sala, F.D.; Abdel-Fattah, W.I.; Borzacchiello, A. Hyaluronic Acid/Corn Silk Extract Based Injectable Nanocomposite: A Biomimetic Antibacterial Scaffold for Bone Tissue Regeneration. Mater. Sci. Eng. C 2020, 107, 110195. [Google Scholar] [CrossRef]
- Makvandi, P.; Ali, G.W.; Sala, F.D.; Abdel-Fattah, W.I.; Borzacchiello, A. Biosynthesis and Characterization of Antibacterial Thermosensitive Hydrogels Based on Corn Silk Extract Hyaluronic Acid and Nanosilver for Potential Wound Healing. Carbohydr. Polym. 2019, 223, 115023. [Google Scholar] [CrossRef]
- Ehsan, N.Z.; Makvandi, P.; Borzacchiello, A.; Tay, F.R.; Ashtari, B.; Padil, V. Antimicrobial Gum Bio-Based Nanocomposites and Their Industrial and Biomedical Applications. Chem. Comm. 2019, 55, 14871–14885. [Google Scholar]
- Ehsan, N.Z.; Makvandi, P.; Ashtari, B.; Rossi, F.; Motahari, A.; Perale, A. Progress in Conductive Polyaniline-Based Nanocomposites for Biomedical Applications: A Review. J. Med. Chem. 2020, 63, 1–22. [Google Scholar]
- Mailard, M.; Huang, O.; Brus, L. Silver Nanodisk Growth by Surface Plasmon Enhanced Protoreduction of Adsorbed [Ag+]. Nano Lett. 2003, 3, 1611–1615. [Google Scholar] [CrossRef]
- Haes, A.J.; Stuart, D.A.; Nie, S.; Van Dyyene, R.P. Using Solution-Phase Nanoparticles, Surface – Confined Nanoparticle Arrays and Single Nanoparticles as Biological Sensing Platforms. J. Fluoresc. 2004, 14, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Mock, J.J.; Barbic, M.; Smith, D.R.; Schultz, D.A.; Schultz, S. Shape Effects in Plasmon Resonance of Individual Colloidal Silver Nanoparticles. J. Chem. Phys. 2002, 116, 6755–6759. [Google Scholar] [CrossRef]
- Gubin, S.P. Metal Containing Nano-particles within Polymeric Matrices: Preparation, Structure, and Properties. Colloids Surf. A 2002, 202, 155–163. [Google Scholar] [CrossRef]
- Alsharaeh, E.H.; Faisal, N.H.; Othman, A.A.; Ahmed, R. Evaluation of Nanomechanical Properties of (Styrene-Methyl methacrylate) Copolymer Composites Containing Graphene Sheets. Ind. Eng. Chem. Res. 2013, 52, 17871–17881. [Google Scholar] [CrossRef]
- Aldosari, M.; Othman, A.; Alsharaeh, E.H. Synthesis and Characterization of the in situ Bulk Polymerization of PMMA Containing Graphene Sheets Using Microwave Irradiation. Molecules 2013, 18, 3152–3167. [Google Scholar] [CrossRef]
- Alsharaeh, E.H.; AlDosari, M.; Othman, A. US Patent, 85579162013. 2013. Available online: https://patents.justia.com/patent/8557916 (accessed on 9 May 2020).
- Alsharaeh, E.H.; AlDosari, M.; Othman, A. Microwave Irradiation Synthesis and Characterization of RGO-Ag NPs/polystyrene nanocomposites. Polym. Compos. 2014, 35, 2318–2323. [Google Scholar] [CrossRef]
- Polychroniadis, E.K.; Oral, A.Y.; Ozer, M. (Eds.) International Multidisciplinary Microscopy Congress; Springer Proceedings in Physics Book Series; 2013; Volume 154. [Google Scholar] [CrossRef]
- Liu, Z.; Robinson, J.T.; Sun, X.; Dai, H. PEGylated Nanographene Oxide for Delivery of Water-insoluble Cancer Drugs. J. Am. Chem. Soc. 2008, 130, 10876–10877. [Google Scholar] [CrossRef]
- Lee, C.; Wei, X.D.; Kysar, J.W.; Hone, J. Measurement of the Elastic Properties and Intrinsic Strength of Monolayer Graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef]
- Balandin, A.A. Thermal Properties of Graphene and Nanostructured Carbon Materials. Nat. Mater. 2011, 10, 569–581. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.A.; Ghosh, S.; Bao, W.Z.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior Thermal Conductivity of Single-layer Graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Ruoff, R.S. Chemical Methods for the Production of Graphenes. Nat. Nanotechol. 2009, 4, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Giovanni, M.; Poh, H.L.; Ambrosi, A.; Zhao, G.; Sofer, Z.; Sanek, F.; Khezri, B.; Webster, R.D.; Pumera, M. Noble Metal (Pd, Ru, Rh, Pt, Au, Ag) Doped Graphene Hybrids for Electrocatalysis. Nanoscale 2012, 4, 5002–5008. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Yang, J.; Bi, K.; Chen, Y. Mode Dependent Lattice Thermal Conductivity of Single Layer Graphene. J. Appl. Phys. 2014, 116, 153503. [Google Scholar] [CrossRef]
- Soldano, C.; Mahmood, A.; Dujardin, E. Production, Properties and Potential of Graphene. Carbon 2010, 48, 2127–2150. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.B.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.; Nguyen, S.T.; Ruoff, R.S. Graphene-based Composite Materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef]
- Dikin, D.A.; Stankovich, S.; Zimney, E.J.; Piner, R.; Dommett, G.H.B.; Evmenenko, G.; Nguyen, S.T.; Ruoff, R.S. Preparation and Characterization of Graphene Oxide Paper. Nature 2007, 448, 457–460. [Google Scholar] [CrossRef]
- Hong, W.; Woo, H.-J.; Choi, H.-W.; Kim, Y.-S.; Kim, G.-D. Optical Property Modification of PMMA by Ion-beam Implantation. Appl. Surd. Sci. 2001, 169–170, 428–432. [Google Scholar] [CrossRef]
- Ali, U.; Karim, K.; Buang, N.Z. A Review of the Properties and Applications of Poly (Methyl Methacrylate) (PMMA). Polym. Rev. 2015, 55, 678–705. [Google Scholar] [CrossRef]
- Victor, A.; Alsharaeh, E.; Samy El-Shall, M. Catalyzed Radical Polymerization of Styrene Vapor on Nanoparticle Surfaces and the Incorporation of Metal and Metal Oxide Nanoparticles within Polystyrene Polymers. J. Phys. Chem. B 2006, 110, 19100–19103. [Google Scholar]
- Matusinovic, Z.; Rogosic, M.; Sipusic, J. Synthesis and Characterization of Poly(styrene-co-methacrylate)/layered double hydroxide Nanocomposites via in situ Polymerization. J. Polym. Degrad. Stab. 2009, 94, 95–101. [Google Scholar] [CrossRef]
- Coskun, M.; Seven, P. Synthesis, Characterization and Investigation of Dielectric Properties of Two-armed Graft Copolymers Prepared with Methyl methacrylate and Styrene onto PVC using Atom Transfer Radical Polymerization. React. Funct. Polym. 2011, 71, 395–401. [Google Scholar] [CrossRef]
- Huang, X.; Yin, Z.Y.; Wu, S.X.; Qi, X.Y.; He, Q.Y.; Zhang, Q.C.; Yan, Q.Y.; Boey, F.; Zhang, H. Graphene-based Materials: Synthesis, Characterization, Properties, and Applications. Small 2011, 7, 1876–1902. [Google Scholar] [CrossRef] [PubMed]
- El-Shall, M.S.; Abdelsayed, V.; Khder, A.E.R.S.; Hassan, H.M.A.; El-Kaderi, H.M.; Reich, T.E. Metallic and Bimetallic Nanocatalysts Incorporated into Highly Porous Coordination Polymer MIL-101. J. Mater. Chem. 2009, 19, 7625–7631. [Google Scholar] [CrossRef]
- Hassan, H.M.A.; Abdelsayed, V.; Khder, A.; AbouZeid, K.M.; Terner, J.; El-Shall, M.S.; Al-Resayes, S.I.; El-Azhary, A.A. Microwave Synthesis of Graphene Sheets Supporting Metal Nanocrystals in Aqueous and Organic Media. J. Mater. Chem. 2009, 19, 3832–3837. [Google Scholar] [CrossRef]
- Zedan, A.F.; Sappal, S.; Moussa, S.; El-Shall, M.S. Hexagonal CdSe Nanocrystals Supported on Graphene. Photoluminescence Quenching by Graphene. J. Phys. Chem. C 2010, 14, 19920–19927. [Google Scholar] [CrossRef]
- Rao, Y.N.; Banerjee, D.; Datta, A.; Das, S.K.; Guin, R.; Saha, A. Gmma Irradiation Route Synthesis of Highly Re-dispersable Natural Polymer Capped Silver Nanoparticles. Radiat. Phys. Chem. 2010, 79, 1240–1246. [Google Scholar] [CrossRef]
- NIST-XPS Database. Available online: http://srdata.nist.gov/xps/2000 (accessed on 5 May 2019).
- Li, J.; Liu, C.-Y.; Liu, Y. Au/Graphene Hydrogel: Synthesis, Characterization and its use for Catalytic Reduction of 4-nitrophenol. J. Mater. Chem. 2012, 22, 8426–8430. [Google Scholar] [CrossRef]
- Zhang, P.; Shao, C.; Zhang, Z.; Zhang, M.; Mu, J.; Guo, Z.; Liu, Y. In situ Assembly of Well Dispersed Ag Nanoparticles (Ag NPs) on Electrospun Carbon Nanofibers (CNFs) for Catalytic Reduction of 4-nitrophenol. Nanoscale 2011, 3, 3357–3363. [Google Scholar] [CrossRef]
- Gutes, A.; Hsia, B.; Sussman, A.; Mickelson, W.; Zettl, A.; Carraro, C.; Maboudian, R. Graphene Decoration with Metal Nanoparticles: Towards Easy Integration for Sensing Applications. Nanoscale 2012, 4, 438–440. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.; Wang, K.; Lu, H.; Yang, Y.; Nutt, S. Covalent Polymer Functionalization of Graphene Nanosheets and Mechanical Properties of Composites. J. Mater. Chem. 2009, 19, 7098–7105. [Google Scholar] [CrossRef]
- Nobel, M.L.; Mendes, E.; Picken, S.J. Acrylic-based Nanocomposite Resins for Coating Applications. J. Appl. Polym. Sci. 2007, 104, 2146–2156. [Google Scholar] [CrossRef]
- Chen, B.; Evans, J.R.G.; Greenwell, C.H.; Boulet, P.; Coveney, P.V.; Bowden, A.A.; Whiting, A. A Critical Appraisal of Polymer-Clay Nanocomposites. Chem. Soc. Rev. 2008, 37, 568–594. [Google Scholar] [CrossRef]
- Chow, W.S.; Mohd Ishak, Z.A. Mechanical, Morphological and Rheological Properties of Polyamide 6/organo-montorillonite Nanocomposites. Polym. Lett. 2007, 1, 77–83. [Google Scholar] [CrossRef]
- Ye, S.; Feng, J. A New Insight into in situ Thermal Reduction of Graphene Oxide Dispersed in a Polymer Matrix. Polym. Chem. 2013, 4, 1765–1768. [Google Scholar] [CrossRef]

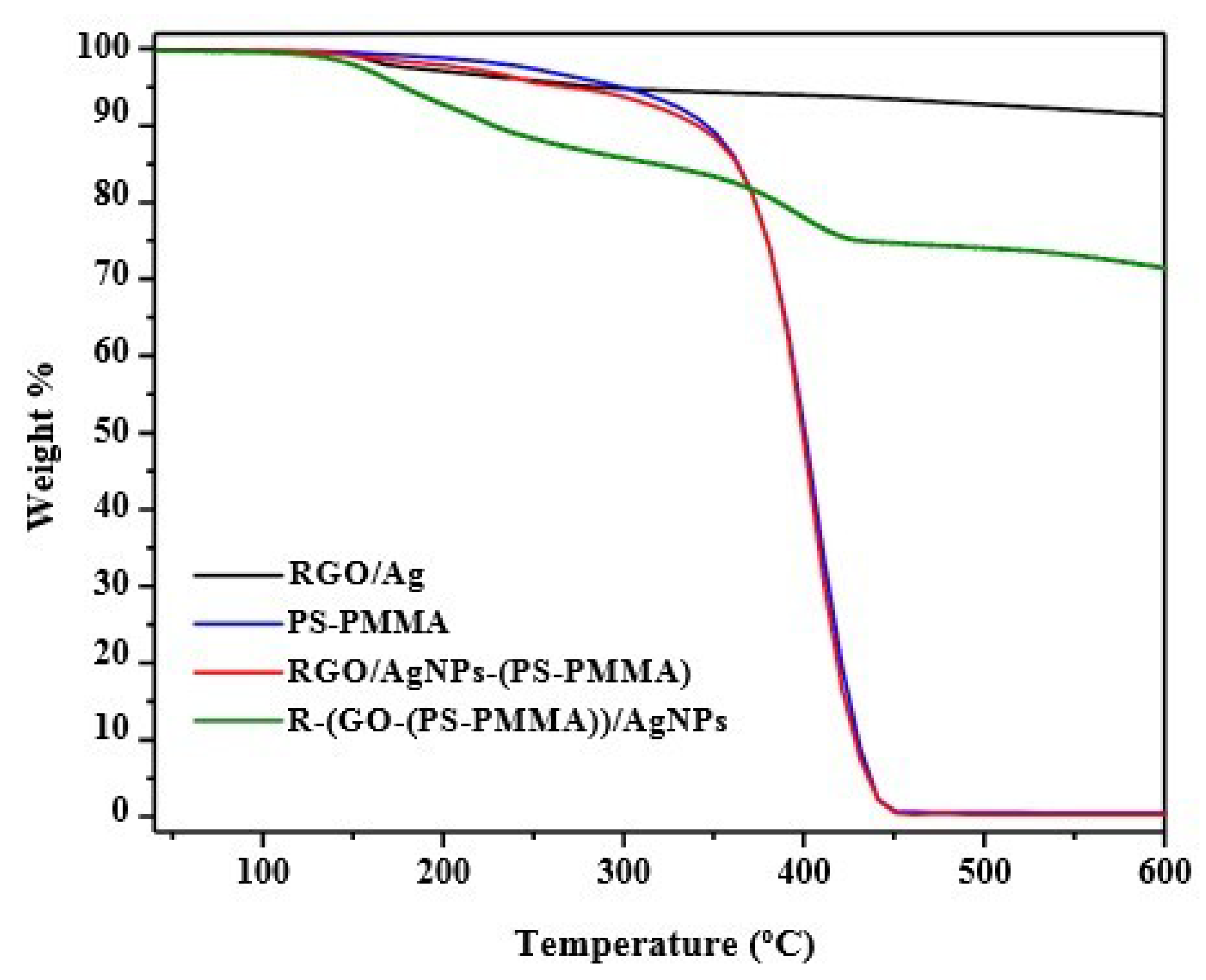
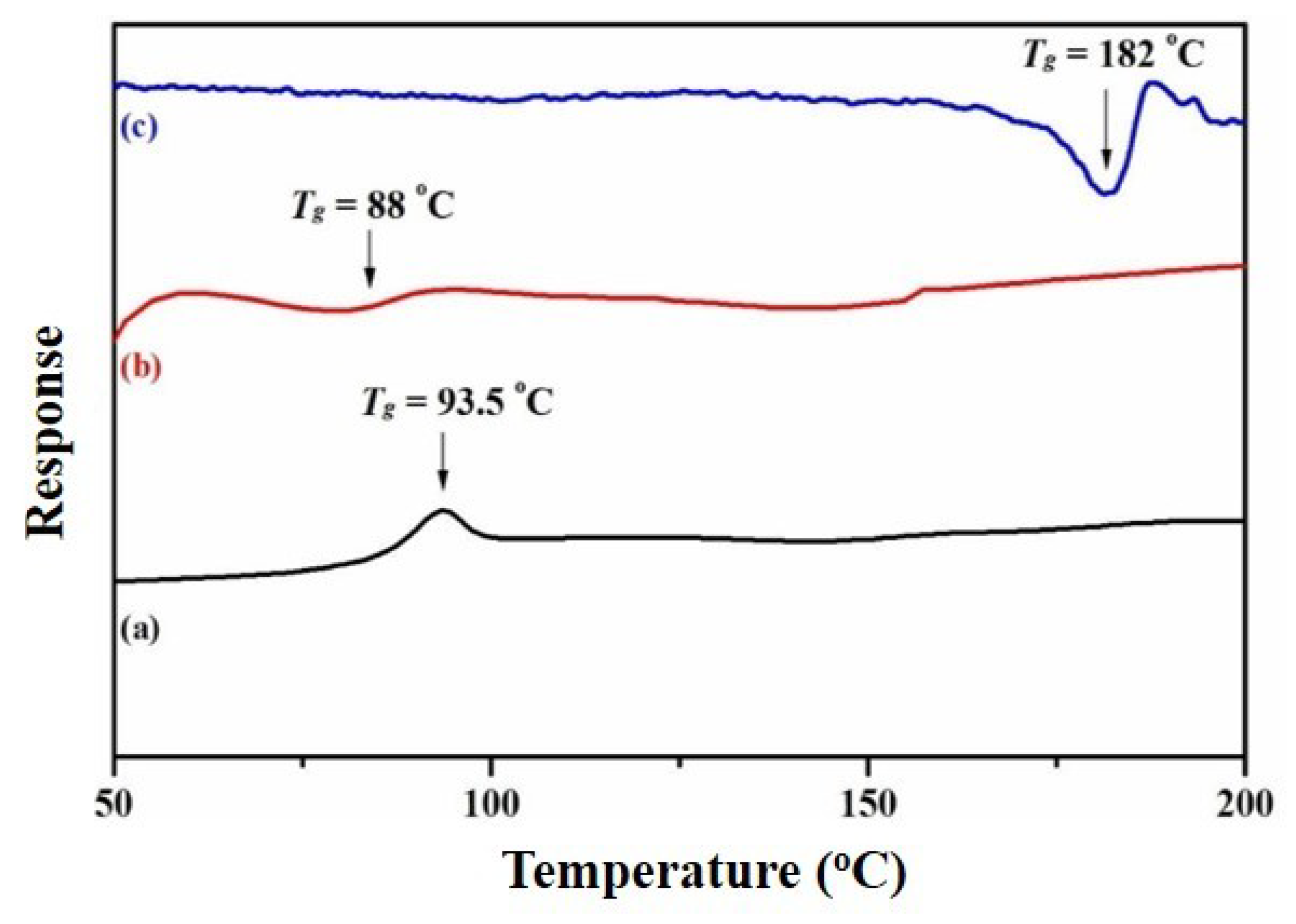
| Sample | Tdeg (°C) | Td (10% Weight Loss) (°C) | Tmax (50% Weight Loss) (°C) | Tg (°C) |
|---|---|---|---|---|
| RGO/Ag NPS | 158 | - | - | - |
| PS-PMMA | 288, 367 | 344 | 397 | 93.5 |
| RGO/Ag NPs-(PS-PMMA) | 400 | 340 | 397 | 88 |
| R-(GO-PS-PMMA)-Ag NPs | 196, 400 | 228 | <800 | 181 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aldosari, M.A.; Alsaud, K.B.B.; Othman, A.; Al-Hindawi, M.; Faisal, N.H.; Ahmed, R.; Michael, F.M.; Krishnan, M.R.; Asharaeh, E. Microwave Irradiation Synthesis and Characterization of Reduced-(Graphene Oxide-(Polystyrene-Polymethyl Methacrylate))/Silver Nanoparticle Nanocomposites and Their Anti-Microbial Activity. Polymers 2020, 12, 1155. https://doi.org/10.3390/polym12051155
Aldosari MA, Alsaud KBB, Othman A, Al-Hindawi M, Faisal NH, Ahmed R, Michael FM, Krishnan MR, Asharaeh E. Microwave Irradiation Synthesis and Characterization of Reduced-(Graphene Oxide-(Polystyrene-Polymethyl Methacrylate))/Silver Nanoparticle Nanocomposites and Their Anti-Microbial Activity. Polymers. 2020; 12(5):1155. https://doi.org/10.3390/polym12051155
Chicago/Turabian StyleAldosari, Mohammad A., Khaled Bin Bandar Alsaud, Ali Othman, Mohammed Al-Hindawi, Nadimul Haque Faisal, Rehan Ahmed, Feven Mattwes Michael, Mohan Raj Krishnan, and Edreese Asharaeh. 2020. "Microwave Irradiation Synthesis and Characterization of Reduced-(Graphene Oxide-(Polystyrene-Polymethyl Methacrylate))/Silver Nanoparticle Nanocomposites and Their Anti-Microbial Activity" Polymers 12, no. 5: 1155. https://doi.org/10.3390/polym12051155
APA StyleAldosari, M. A., Alsaud, K. B. B., Othman, A., Al-Hindawi, M., Faisal, N. H., Ahmed, R., Michael, F. M., Krishnan, M. R., & Asharaeh, E. (2020). Microwave Irradiation Synthesis and Characterization of Reduced-(Graphene Oxide-(Polystyrene-Polymethyl Methacrylate))/Silver Nanoparticle Nanocomposites and Their Anti-Microbial Activity. Polymers, 12(5), 1155. https://doi.org/10.3390/polym12051155






