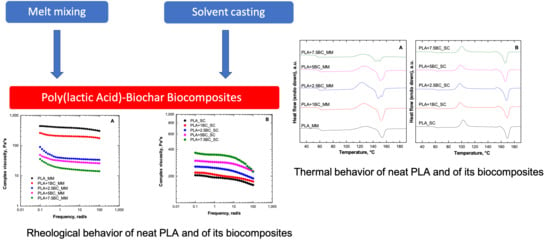
Poly(lactic Acid)–Biochar Biocomposites: Effect of Processing and Filler Content on Rheological, Thermal, and Mechanical Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Processing
2.3. Characterization Techniques
- -
- First heating up from 30 to 200 °C at 10 °C/min;
- -
- Cooling down from 200 to 0 °C at 10 °C/min;
- -
- Second heating up from 0 to 180 °C at 10 °C/min.
3. Results and Discussion
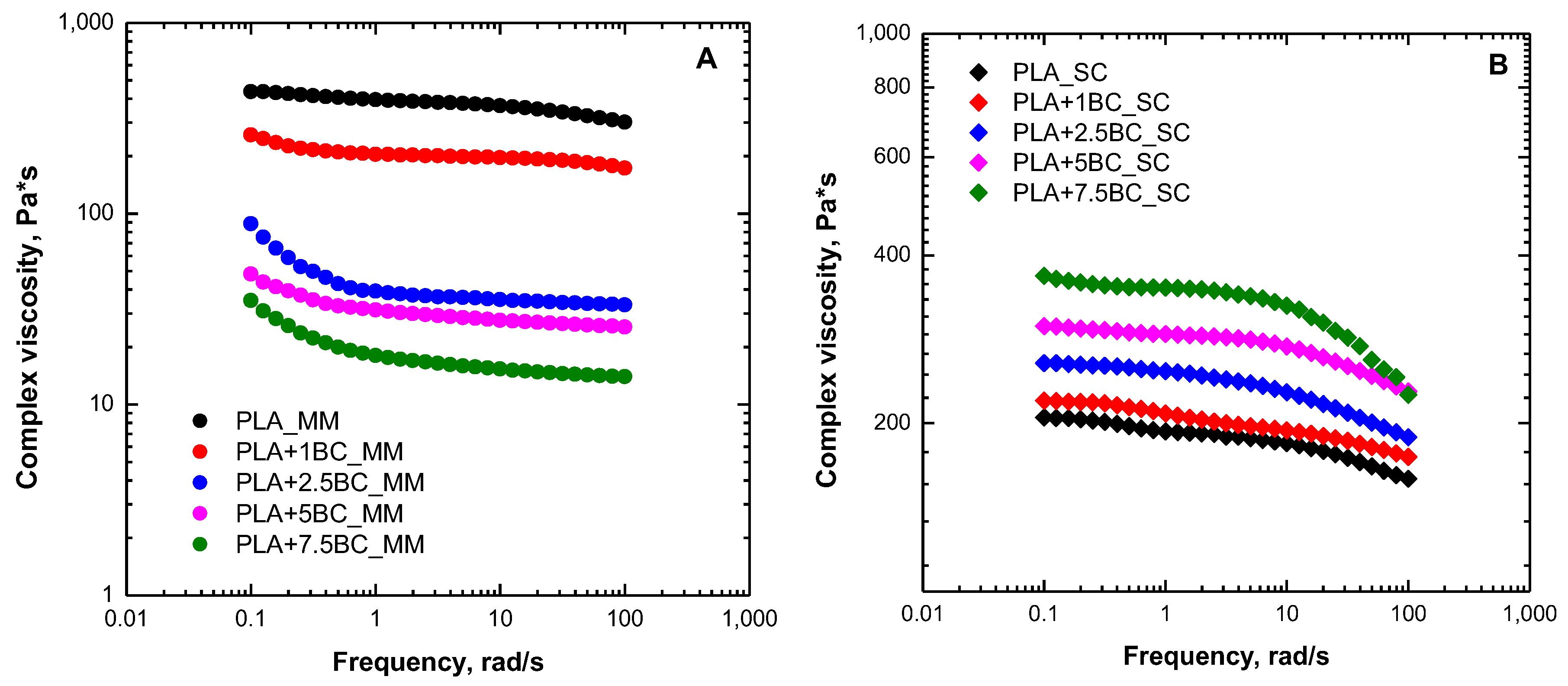
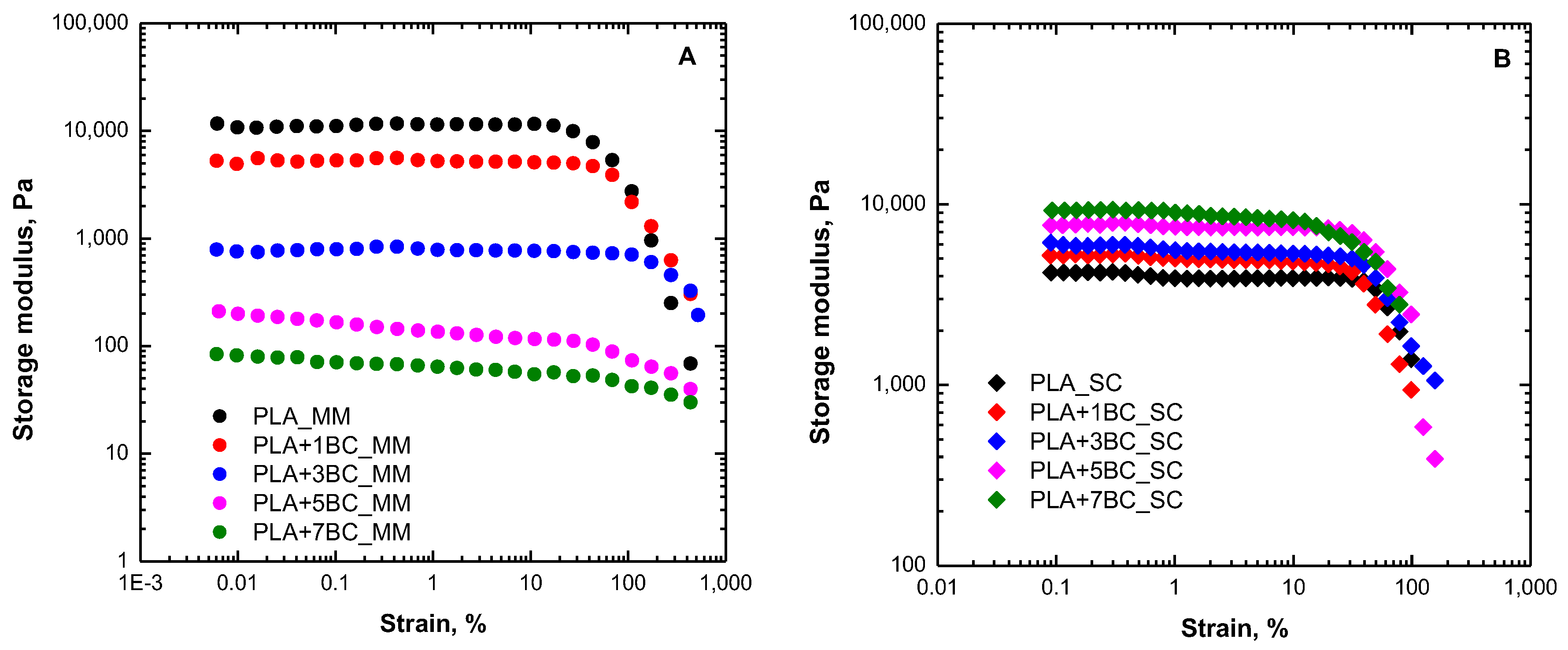
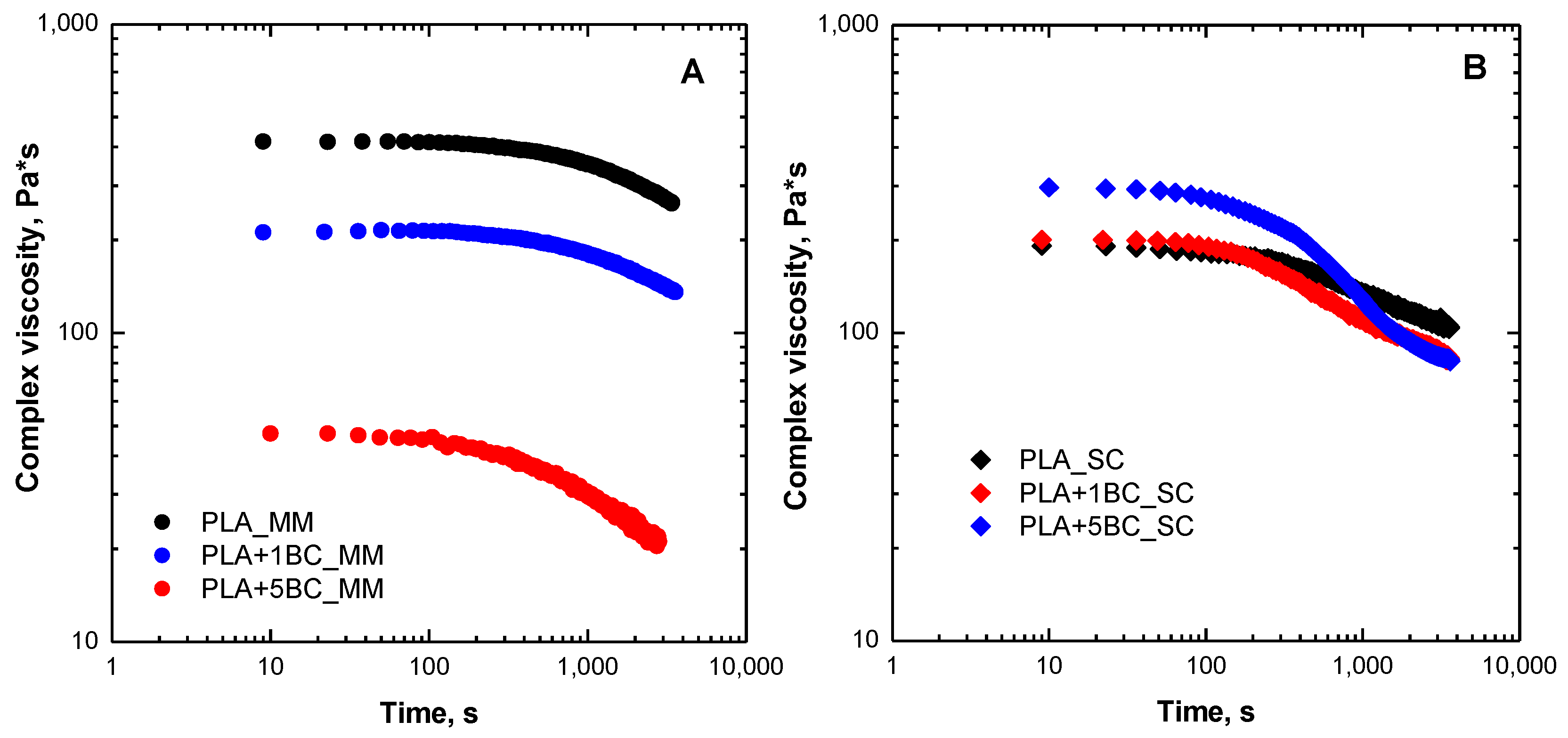
3.1. Rheological Behavior
3.2. Morphology
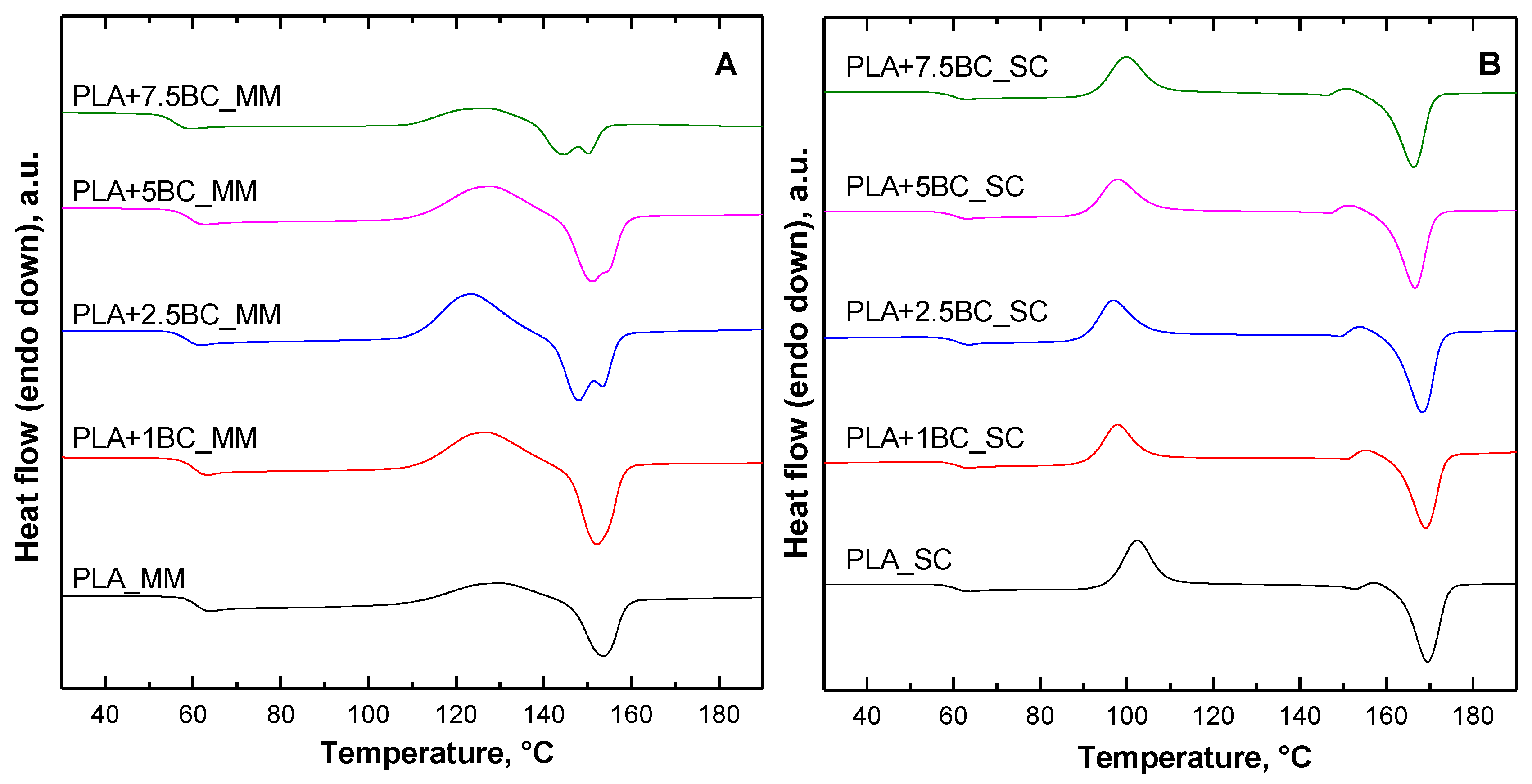
3.3. Thermal Properties
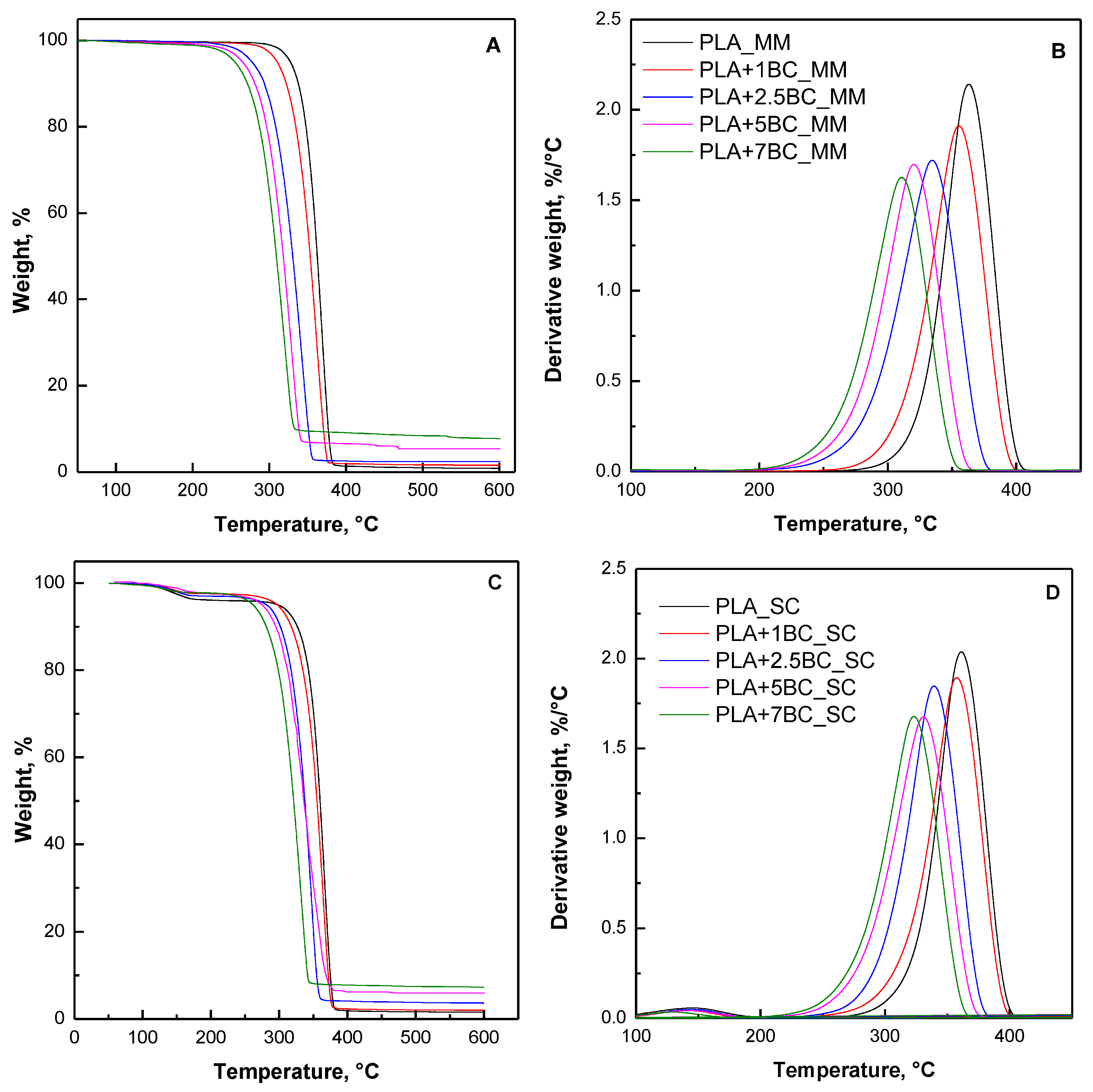
3.4. Thermal Stability
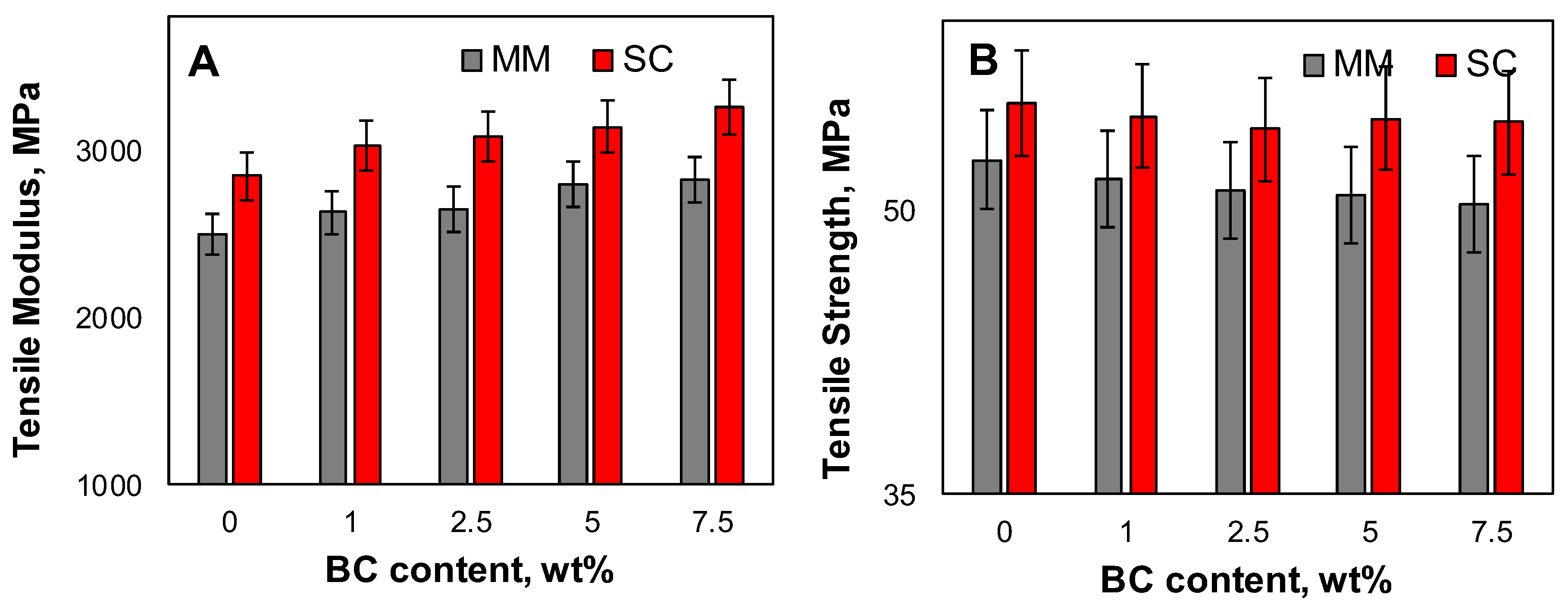
3.5. Mechanical Properties
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yu, L.; Dean, K.; Li, L. Polymer blends and composites from renewable resources. Prog. Polym. Sci. 2006, 31, 576–602. [Google Scholar] [CrossRef]
- Dintcheva, N.T.; La Mantia, F.P.; Arrigo, R. Natural compounds as light stabilizer for a starch-based biodegradable polymer. J. Polym. Eng. 2014, 34, 441–449. [Google Scholar] [CrossRef]
- Jamróz, E.; Kulawik, P.; Kopel, P. The effect of nanofillers on the functional properties of biopolymer-based films: A review. Polymers 2019, 11, 675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armentano, I.; Bitinis, N.; Fortunati, E.; Mattioli, S.; Rescignano, N.; Verdejo, R.; Lopez-Manchado, M.A.; Kenny, J.M. Multifunctional nanostructured PLA materials for packaging and tissue engineering. Prog. Polym. Sci. 2013, 38, 1720–1747. [Google Scholar] [CrossRef] [Green Version]
- Raquez, J.M.; Habibi, Y.; Murariu, M.; Dubois, P. Polylactide (PLA)-based nanocomposites. Prog. Polym. Sci. 2013, 38, 1504–1542. [Google Scholar] [CrossRef]
- Serizawa, S.; Inoue, K.; Iji, M. Kenaf-fiber-reinforced poly(lactic acid) used for electronic products. J. Appl. Polym. Sci. 2006, 100, 618–624. [Google Scholar] [CrossRef]
- Rasal, R.M.; Hirt, D.E. Toughness decrease of PLA-PHBHHx blend films upon surface-confined photopolymerization. J. Biomed. Mater. Res. 2009, 88, 1079–1086. [Google Scholar] [CrossRef]
- Abd Alsaheb, R.A.; Aladdin, A.; Othman, N.Z.; Abd Malek, R.; Leng, O.M.; Aziz, R.; El Enshasy, H.A. Recent applications of polylactic acid in pharmaceutical and medical industries. J. Chem. Pharm. Res. 2015, 7, 51–63. [Google Scholar]
- Lyu, Y.; Pang, J.; Gao, Z.; Zhang, Q.; Shi, X. Characterization of the compatibility of PVC/PLA blends by Aid of Rheological Responses. Polymer 2019, 176, 20–29. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Samper, M.D.; Aldas, M.; López, J. On the use of PLA-PHB blends for sustainable food packaging applications. Materials 2017, 10, 1008. [Google Scholar] [CrossRef]
- Morreale, M.; Liga, A.; Mistretta, M.C.; Ascione, L.; La Mantia, F.P. Mechanical, thermomechanical and reprocessing behavior of green composites from biodegradable polymer and wood flour. Materials 2015, 8, 7536–7548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozyhar, T.; Baradel, F.; Zoppe, J. Effect of functional mineral additive on processability and material properties of wood-fiber reinforced poly(lactic acid) (PLA) composites. Compos. Part A Appl. Sci. Manuf. 2020, 132, 105827. [Google Scholar] [CrossRef]
- Gonçalves, C.; Gonçalves, I.C.; Magalhães, F.D.; Pinto, A.M. Poly(lactic acid) composites containing carbon-based nanomaterials: A review. Polymers 2017, 9, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mokhena, T.C.; Sefadi, J.S.; Sadiku, E.R.; John, M.J.; Mochane, M.J.; Mtibe, A. Thermoplastic processing of PLA/Cellulose nanomaterials composites. Polymers 2018, 10, 1363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiore, V.; Botta, L.; Scaffaro, R.; Valenza, A.; Pirrotta, A. PLA based biocomposites reinforced with Arundo donax fillers. Compos. Sci. Technol. 2014, 105, 110–117. [Google Scholar] [CrossRef]
- Battegazzore, D.; Abt, T.; Maspoch, M.L.; Frache, A. Multilayer cotton fabric bio-composites based on PLA and PHB copolymer for industrial load carrying applications. Compos. Part B Eng. 2019, 163, 761–768. [Google Scholar] [CrossRef]
- Nair, S.S.; Chen, H.; Peng, Y.; Huang, Y.; Yan, N. Polylactic acid biocomposites reinforced with nanocellulose fibrils with high lignin content for improved mechanical, thermal, and barrier properties. ACS Sustain. Chem. Eng. 2018, 6, 10058–10068. [Google Scholar] [CrossRef]
- Tesfaye, M.; Patwa, R.; Gupta, A.; Kashyap, M.J.; Katiyar, V. Recycling of poly(lactic acid)/silk based bionanocomposites films and its influence on thermal stability, crystallization kinetics, solution and melt rheology. Int. J. Biol. Macromol. 2017, 101, 580–594. [Google Scholar] [CrossRef]
- Giorcelli, M.; Khan, A.; Pugno, N.M.; Rosso, C.; Tagliaferro, A. Biochar as a cheap and environmental friendly filler able to improve polymer mechanical properties. Biomass Bioenerg. 2019, 120, 219–223. [Google Scholar] [CrossRef] [Green Version]
- Ogunsona, E.O.; Codou, A.; Misra, M.; Mohanty, A.K. Thermally stable pyrolytic biocarbon as an effective and sustainable reinforcing filler for polyamide bio-composites fabrication. J. Polym. Environ. 2018, 26, 3574. [Google Scholar] [CrossRef]
- Behazina, E.; Misra, M.; Mohanty, A.K. Sustainable biocarbon from pyrolyzed perennial grasses and their effects on impact modified polypropylene biocomposites. Compos. Part B Eng. 2017, 118, 116–124. [Google Scholar] [CrossRef]
- Tomczyk, A.; Sokołowska, Z.; Boguta, P. Biochar physicochemical properties: Pyrolysis temperature and feedstock kind effects. Rev. Environ. Sci. Biotechnol. 2020, 19, 191–215. [Google Scholar] [CrossRef] [Green Version]
- Vardon, D.R.; Moser, B.R.; Zheng, W.; Witkin, K.; Evangelista, R.L.; Strathmann, T.J.; Rajagopalan, K.; Sharma, B.K. Complete Utilization of Spent Coffee Grounds To Produce Biodiesel, Bio-Oil, and Biochar. ACS Sustain. Chem. Eng. 2013, 1, 1286–1294. [Google Scholar] [CrossRef]
- Gezahegn, S.; Lai, R.; Huang, L.; Chen, L.; Huang, F.; Blozowski, N.; Thomas, S.C.; Saina, M.; Tjong, J.; Jaffer, S.; et al. Porous graphitic biocarbon and reclaimed carbon fiber derived environmentally benign lightweight composites. Sci. Total Environ. 2019, 664, 363–373. [Google Scholar] [CrossRef]
- Li, S.Y.; Li, X.Y.; Chen, C.C.; Wang, H.Y.; Deng, Q.Y.; Gong, M. Development of electrically conductive nano bamboo charcoal/ultra-high molecular weight polyethylene composites with a segregated network. Compos. Sci. Technol. 2016, 132, 31–37. [Google Scholar] [CrossRef]
- Das, O.; Bhattacharyya, D.; Hui, D.; Lau, K.T. Mechanical and flammability characteristics of biochar/polypropylene biocomposites. Comp. Part B Eng. 2016, 106, 120–128. [Google Scholar] [CrossRef]
- Idrees, M.; Jeelani, S.; Rangari, V. Three-dimensional-printed sustainable biochar-recycled PET composites. ACS Sustain. Chem. Eng. 2018, 6, 13940–13948. [Google Scholar] [CrossRef]
- Schmidt, A.P. 55 Uses of Biochar. Ithaka J. 2012, 1, 286–289. [Google Scholar]
- Nagarajan, V.; Mohanty, A.K.; Misra, M. Biocomposites with size-fractionated biocarbon: Influence of the microstructure on macroscopic properties. ACS Omega 2016, 1, 636–647. [Google Scholar] [CrossRef]
- She, D.; Dong, J.; Zhang, J.; Liu, L.; Sun, Q.; Geng, Z. Development of black and biodegradable biochar/gutta percha composite films with high stretchability and barrier properties. Compos. Sci. Technol. 2019, 175, 1–5. [Google Scholar] [CrossRef]
- Moustafa, H.; Guizani, C.; Dupont, C.; Martin, V.; Jeguirim, M.; Dufresne, A. Utilization of torrefied coffee grounds as reinforcing agent to produce high-quality biodegradable PBAT composites for food packaging applications. ACS Sustain. Chem. Eng. 2017, 5, 1906–1916. [Google Scholar] [CrossRef]
- Arrigo, R.; Jagdale, P.; Bartoli, M.; Tagliaferro, A.; Malucelli, G. Structure–property relationships in polyethylene-based composites filled with biochar derived from waste coffee grounds. Polymers 2019, 11, 1336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Anna, A.; Arrigo, R.; Frache, A. PLA/PHB blends: Biocompatibilizer effects. Polymers 2019, 11, 1416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iozzino, V.; Askanian, H.; Leroux, F.; Verney, V. Poly(Lactic Acid)-based nanobiocomposites with modulated degradation rate. Materials 2018, 11, 1943. [Google Scholar]
- Wang, Y.; Xu, C.; Wu, D.; Xie, W.; Wang, K.; Xia, Q.; Yang, H. Rheology of the cellulose nanocrystals filled poly(ε-caprolactone) biocomposites. Polymer 2018, 140, 167–178. [Google Scholar] [CrossRef]
- Dintcheva, N.T.; Arrigo, R.; Carroccio, S.; Curcuruto, G.; Guenzi, M.; Gambarotti, C.; Filippone, G. Multi-functional polyhedral oligomeric silsesquioxane-functionalized carbon nanotubes for photo-oxidative stable Ultra-High Molecular Weight Polyethylene-based nanocomposites. Eur. Polym. J. 2016, 75, 525–537. [Google Scholar] [CrossRef]
- Gleissle, W.; Hochstein, B. Validity of the Cox–Merz rule for concentrated suspensions. J. Rheol. 2003, 47, 897. [Google Scholar] [CrossRef]
- Hyun, K.; Wilhelm, M.; Klein, C.O.; Cho, K.S.; Nam, J.G.; Ahn, K.H.; Lee, S.J.; Ewoldt, R.H.; McKinley, G.H. A review of nonlinear oscillatory shear tests: Analysis and application of large amplitude oscillatory shear (LAOS). Prog. Polym. Sci. 2011, 36, 1697–1753. [Google Scholar] [CrossRef]
- Giorcelli, M.; Bartoli, M. Development of coffee biochar filler for the production of electrical conductive reinforced plastic. Polymers 2019, 11, 1916. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.; Fortunati, E.; Dominici, F.; Kenny, J.M.; Puglia, D. Effect of processing conditions and lignin content on thermal, mechanical and degradative behavior of lignin nanoparticles/polylactic (acid) bionanocomposites prepared by melt extrusion and solvent casting. Eur. Polym. J. 2015, 71, 126–139. [Google Scholar] [CrossRef]
- Ho, M.; Lau, K.; Hui, D. Improvement of the properties of polylactic acid (PLA) using bamboo charcoal particles. Compos. Part B Eng. 2015, 81, 14–25. [Google Scholar] [CrossRef]
- Oliveira, M.; Santos, E.; Araújo, A.; Fechine, G.J.M.; Machado, A.V.; Botelho, G. The role of shear and stabilizer on PLA degradation. Polym. Test. 2016, 51, 109–116. [Google Scholar] [CrossRef]
- Lee, J.; Kim, K.-H.; Kwon, E.E. Biochar as a Catalyst. Renew. Sustain. Energy Rev. 2017, 77, 70–79. [Google Scholar] [CrossRef]

| [°C] | [°C] | [°C] | [°C] | [°C] | [°C] | [°C] | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| MM | SC | ||||||||||
| PLA | 60.7 | 129.3 | - | 153.4 | 1.2 | 0 | 61.1 | 102.5 | 169.5 | 13.0 | 13.9 |
| PLA+1BC | 60.4 | 127.2 | - | 152.2 | 3.7 | 0 | 60.9 | 97.8 | 169.0 | 21.4 | 23.2 |
| PLA+2.5BC | 59.9 | 123.9 | 148 | 153.4 | 2.3 | 0 | 61.2 | 96.9 | 168.3 | 24.9 | 29.7 |
| PLA+5BC | 59.4 | 128.3 | 151.1 | 152.8 | 1.6 | 0 | 60.4 | 97.7 | 166.7 | 20.2 | 22.9 |
| PLA+7.5BC | 56.1 | 126.6 | 144.8 | 150.2 | 0.8 | 0 | 59.9 | 99.8 | 166.3 | 14.2 | 16.5 |
| Sample | T5% [°C] | T10% [°C] | Tmax [°C] | Residue at 600 °C [%] | T5% [°C] | T10% [°C] | Tmax [°C] | Residue at 600 °C [%] |
|---|---|---|---|---|---|---|---|---|
| MM | SC | |||||||
| PLA | 324.3 | 335.9 | 366.9 | 0.70 | 300.4 | 326.5 | 366.0 | 0.50 |
| PLA+1BC | 310.0 | 322.2 | 361.9 | 1.35 | 295.0 | 317.5 | 363.8 | 2.00 |
| PLA+2.5BC | 277.2 | 293.6 | 342.1 | 2.36 | 282.1 | 301.4 | 347.6 | 3.16 |
| PLA+5BC | 284.0 | 300.1 | 343.9 | 5.51 | 272.4 | 291.7 | 342.6 | 5.30 |
| PLA+7.5BC | 254.8 | 271.9 | 320.2 | 7.41 | 263.4 | 281.3 | 332.0 | 7.82 |
| Element | Composition (wt.%) |
|---|---|
| C | 74.0 |
| K | 21.6 |
| Ca | 4.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arrigo, R.; Bartoli, M.; Malucelli, G. Poly(lactic Acid)–Biochar Biocomposites: Effect of Processing and Filler Content on Rheological, Thermal, and Mechanical Properties. Polymers 2020, 12, 892. https://doi.org/10.3390/polym12040892
Arrigo R, Bartoli M, Malucelli G. Poly(lactic Acid)–Biochar Biocomposites: Effect of Processing and Filler Content on Rheological, Thermal, and Mechanical Properties. Polymers. 2020; 12(4):892. https://doi.org/10.3390/polym12040892
Chicago/Turabian StyleArrigo, Rossella, Mattia Bartoli, and Giulio Malucelli. 2020. "Poly(lactic Acid)–Biochar Biocomposites: Effect of Processing and Filler Content on Rheological, Thermal, and Mechanical Properties" Polymers 12, no. 4: 892. https://doi.org/10.3390/polym12040892
APA StyleArrigo, R., Bartoli, M., & Malucelli, G. (2020). Poly(lactic Acid)–Biochar Biocomposites: Effect of Processing and Filler Content on Rheological, Thermal, and Mechanical Properties. Polymers, 12(4), 892. https://doi.org/10.3390/polym12040892