Characterization and Preparation of Furanic-Glyoxal Foams
Abstract
1. Introduction
2. Materials and Methods
2.1. Foams Preparation
2.2. SEM Observation
2.3. ATR FT-MIR Analyses
2.4. MALDI-TOF Analysis
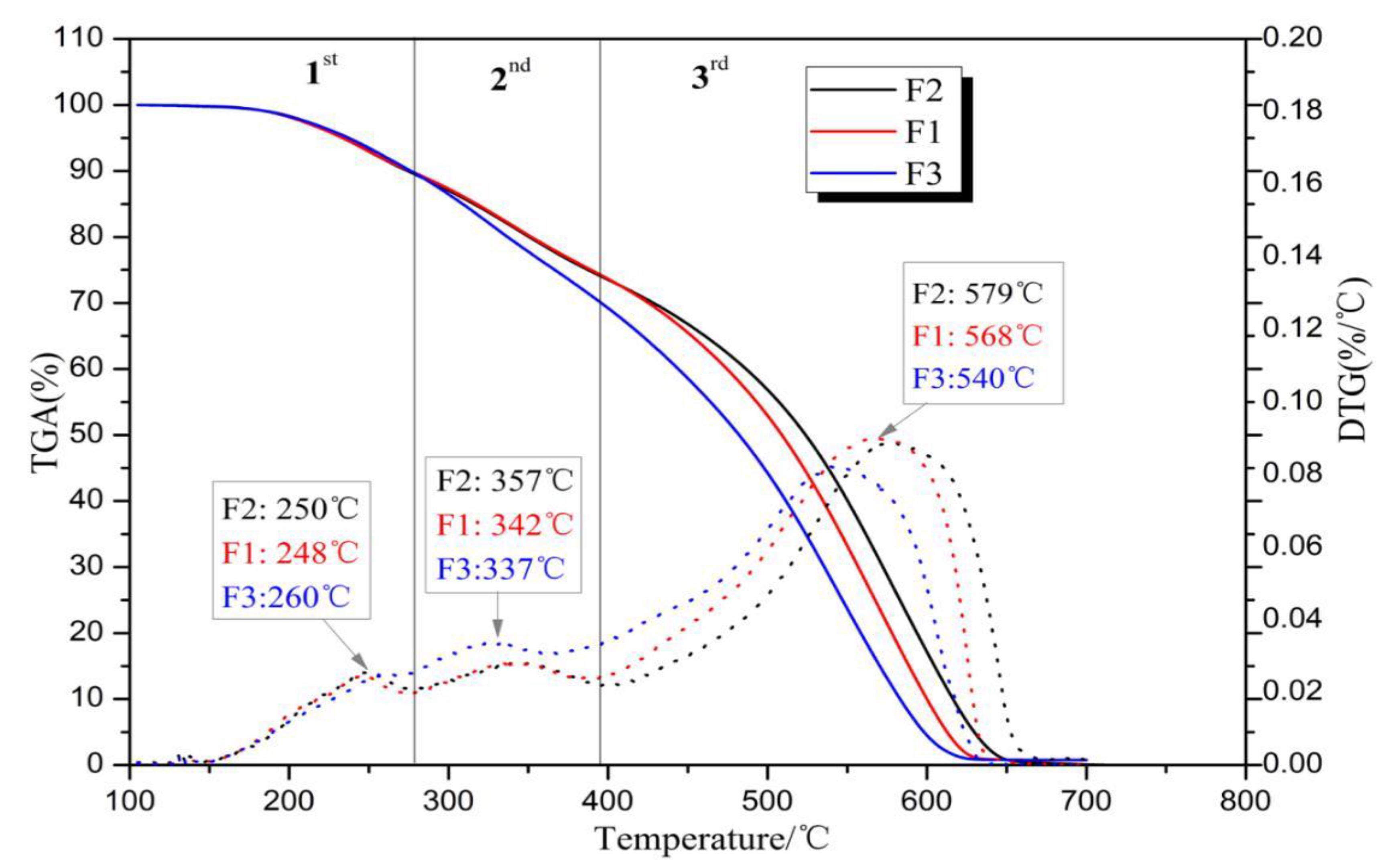
2.5. Thermogravimetric Analysis (TGA)
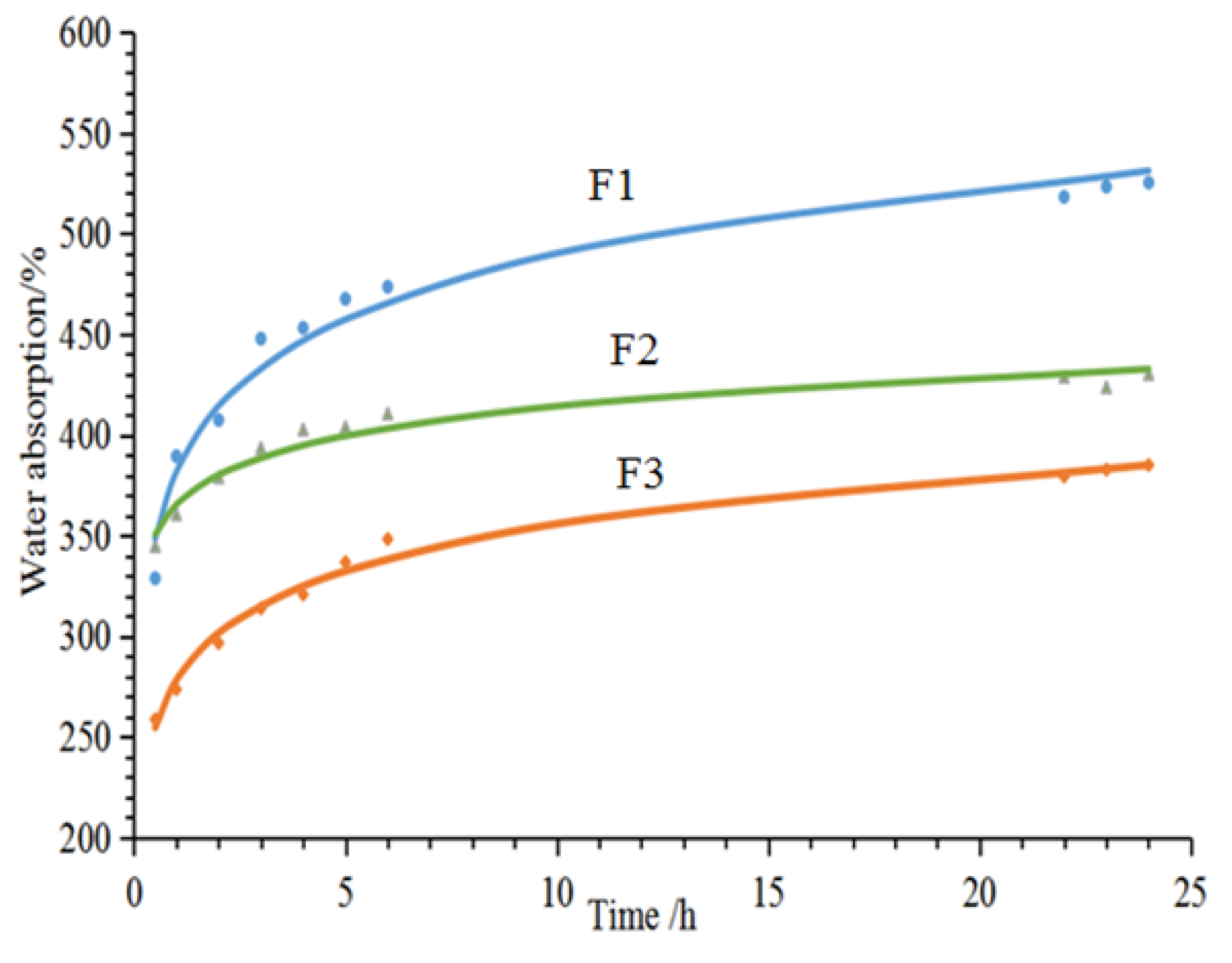
2.6. 24-h Water Absorption
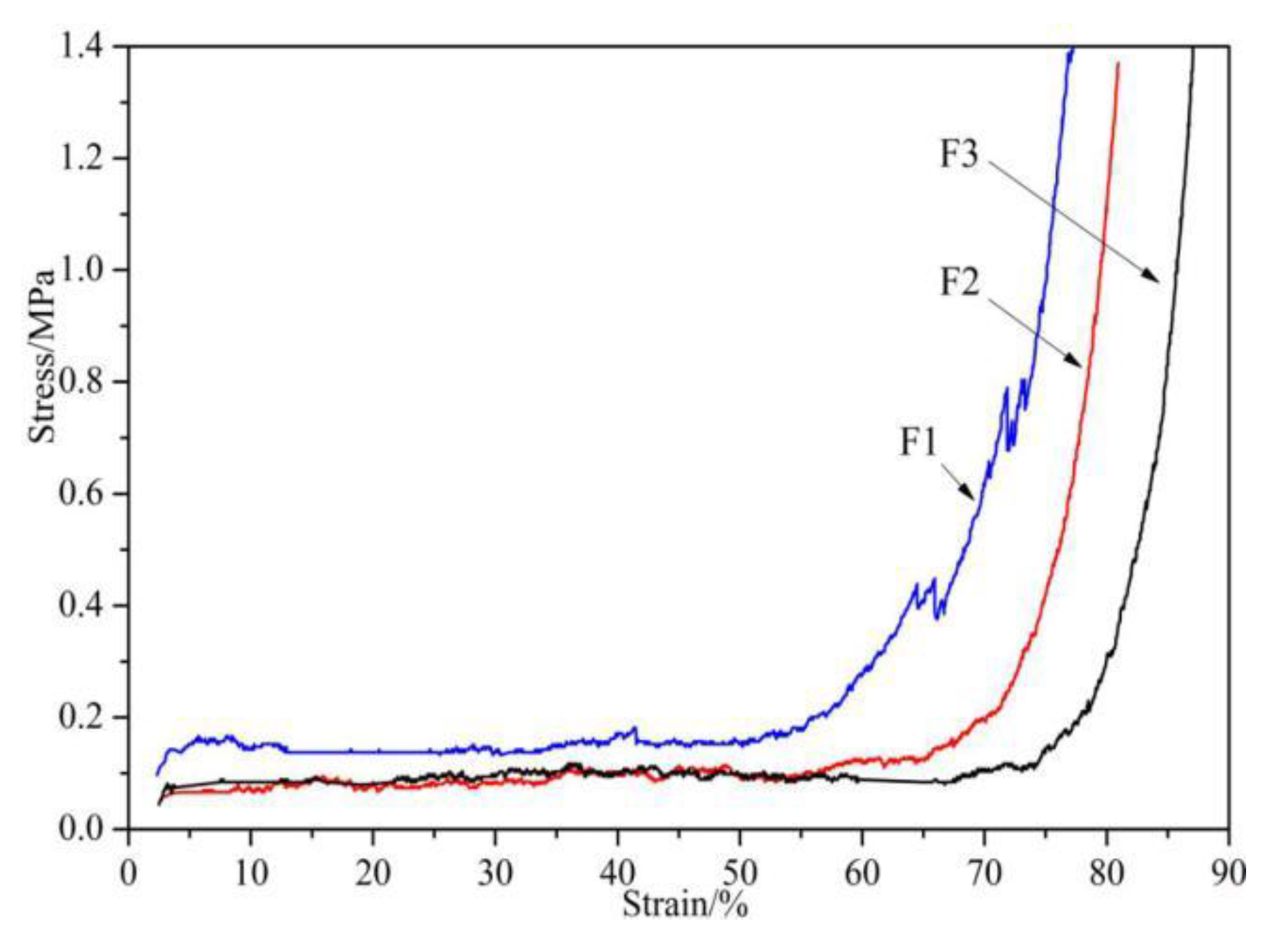
2.7. Compression
3. Results and Discussions
3.1. Physical Properties of Furanic-Glyoxal Foams
3.2. SEM Analysis
3.3. Thermogravimetric Analysis (TGA)
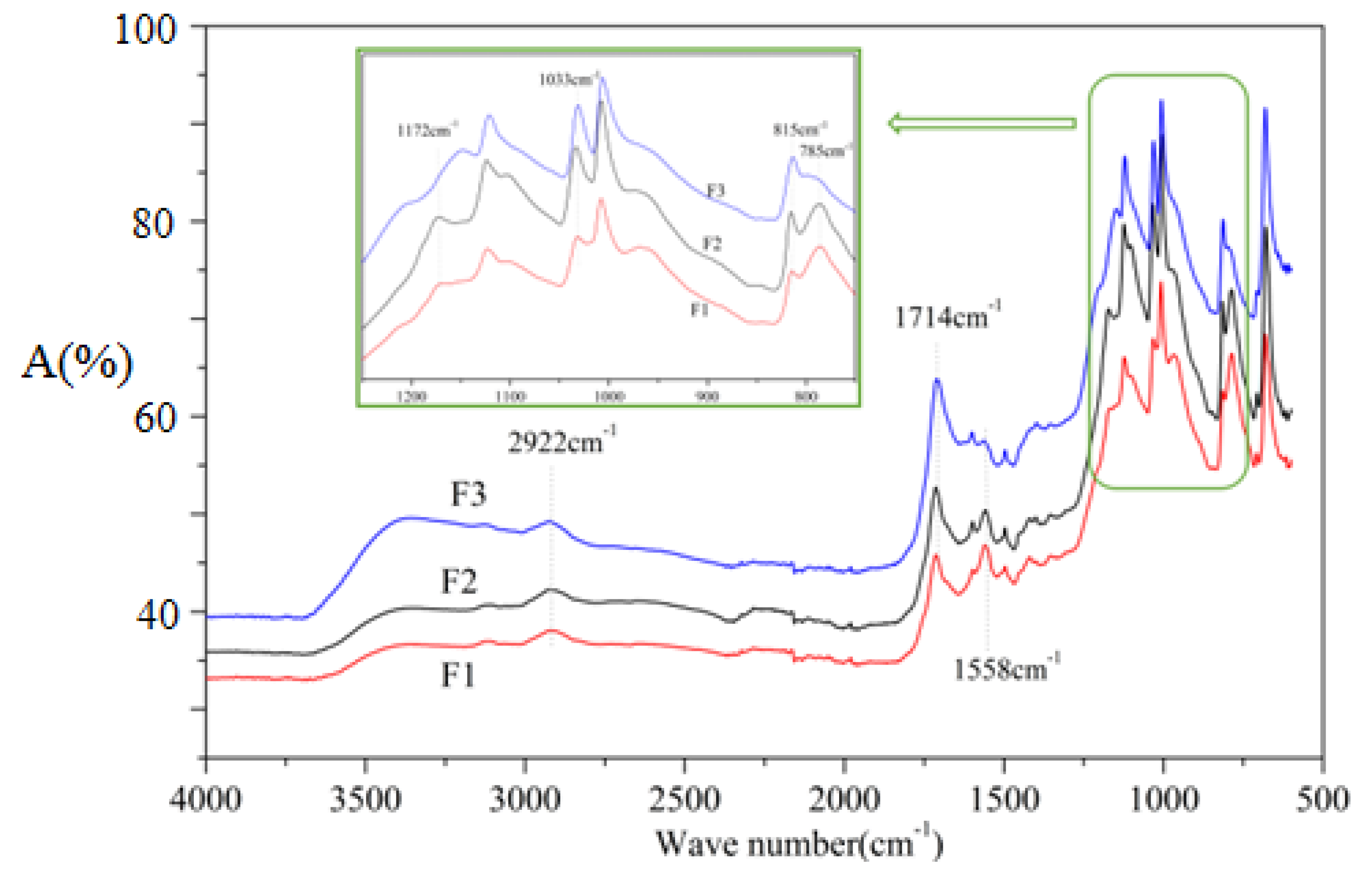
3.4. FTIR Analysis
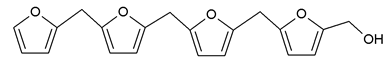
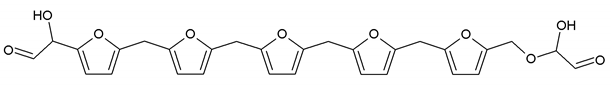
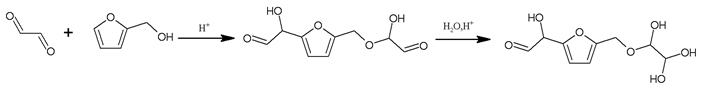
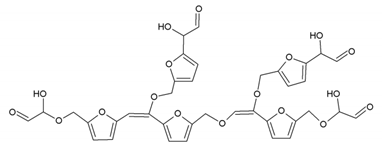
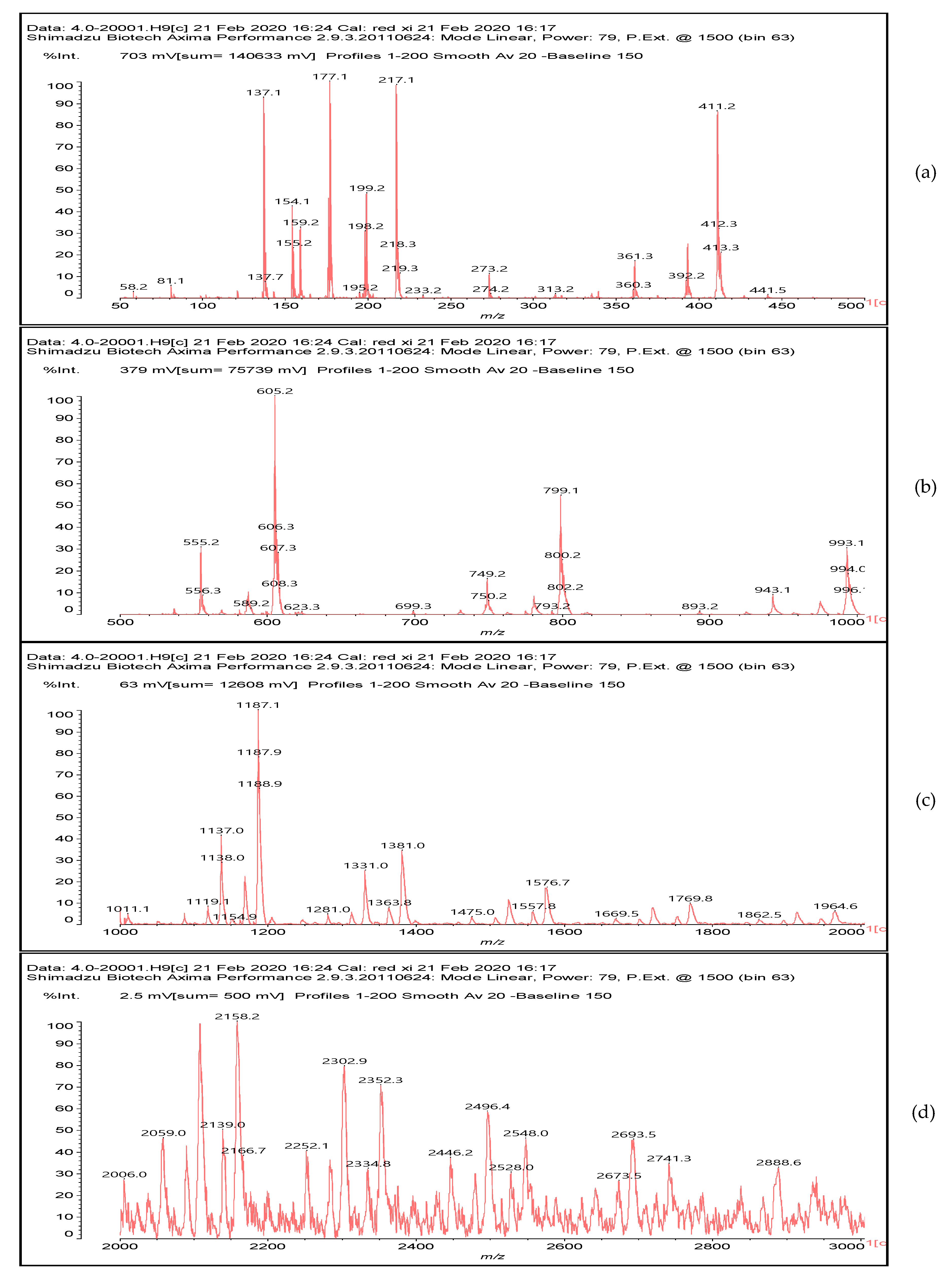
3.5. MALDI ToF Analysis
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Gautam, R.; Bassi, A.S.; Yanful, E.K. A review of biodegradation of synthetic plastic and foams. Appl. Biochem. Biotechnol. 2007, 141, 85–108. [Google Scholar] [CrossRef]
- Landrock, A.H. Handbook of Plastic Foams: Types, Properties, Manufacture and Applications; Elsevier: Amsterdam, The Netherlands, 1995. [Google Scholar]
- Thomson, T. Polyurethanes as Specialty Chemicals: Principles and Applications; CRC press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Singh, H.; Jain, A.K. Ignition, combustion, toxicity, and fire retardancy of polyurethane foams: A comprehensive review. J. Appl. Polym. Sci. 2009, 111, 1115–1143. [Google Scholar] [CrossRef]
- Zia, K.M.; Bhatti, H.N.; Bhatti, I.A. Methods for polyurethane and polyurethane composites, recycling and recovery: A review. React. Funct. Polym. 2007, 67, 675–692. [Google Scholar] [CrossRef]
- Kausar, A. Polyurethane composite foams in high-performance applications: A review. Polym. Plast. Technol. Eng. 2018, 57, 346–369. [Google Scholar] [CrossRef]
- Han, X.; Zeng, C.; Lee, L.J. Extrusion of polystyrene nanocomposite foams with supercritical CO2. Polym. Eng. Sci. 2003, 43, 1261–1275. [Google Scholar] [CrossRef]
- Arora, K.A.; Lesser, A.J.; McCarthy, T.J. Preparation and characterization of microcellular polystyrene foams processed in supercritical carbon dioxide. Macromolecules 1998, 31, 4614–4620. [Google Scholar] [CrossRef]
- Luong, D.D.; Pinisetty, D.; Gupta, N. Compressive properties of closed-cell polyvinyl chloride foams at low and high strain rates: Experimental investigation and critical review of state of the art. Compos. Part B Eng. 2013, 44, 403–416. [Google Scholar] [CrossRef]
- Anderson, J.R.; Okamoto, K.T. Polyethylene Foams and Methods of their Production. U.S. Patent 6,376,059, 23 April 2002. [Google Scholar]
- Zhang, Y.; Rodriguez, D.; Ait-Kadi, A. High-density polyethylene foams. I. Polymer and foam characterization. J. Appl. Polym. Sci. 2003, 90, 2111–2119. [Google Scholar] [CrossRef]
- Mougel, C.; Garnier, T.; Cassagnau, P. Phenolic foams: A review of mechanical properties, fire resistance and new trends in phenol substitution. Polymer 2019, 164, 86–117. [Google Scholar] [CrossRef]
- Pizzi, A. Tannin-based biofoams—A review. J. Renew. Mater. 2019, 7, 474–489. [Google Scholar] [CrossRef]
- Meikleham, N.E.; Pizzi, A. Acid-and alkali-catalyzed tannin-based rigid foams. J. Appl. Polym. Sci. 1994, 53, 1547–1556. [Google Scholar] [CrossRef]
- Tondi, G.; Pizzi, A. Tannin-based rigid foams: Characterization and modification. Ind. Crops Prod. 2009, 29, 356–363. [Google Scholar] [CrossRef]
- Tondi, G.; Zhao, W.; Pizzi, A. Tannin-based rigid foams: A survey of chemical and physical properties. Bioresour. Technol. 2009, 100, 5162–5169. [Google Scholar] [CrossRef]
- Tondi, G.; Fierro, V.; Pizzi, A. Tannin-based carbon foams. Carbon 2009, 47, 1480–1492. [Google Scholar] [CrossRef]
- Pizzi, A.; Tondi, G.; Pasch, H. Matrix-assisted laser desorption/ionization time-of-flight structure determination of complex thermoset networks: Polyflavonoid tannin–furanic rigid foams. J. Appl. Polym. Sci. 2008, 110, 1451–1456. [Google Scholar] [CrossRef]
- Szczurek, A.; Fierro, V.; Pizzi, A. A new method for preparing tannin-based foams. Ind. Crops Prod. 2014, 54, 40–53. [Google Scholar] [CrossRef]
- Pizzi, A. Tannins: Prospectives and actual industrial applications. Biomolecules 2019, 9, 344. [Google Scholar] [CrossRef]
- Zhou, X.; Li, B.; Xu, Y. Tannin-furanic resin foam reinforced with cellulose nanofibers (CNF). Ind. Crops Prod. 2019, 134, 107–112. [Google Scholar] [CrossRef]
- Pizzi, A. Wood Adhesives: Chemistry and Technology; Dekker: New York, NY, USA, 1983. [Google Scholar]
- Tondi, G.; Pizzi, A.; Pasch, H. MALDI-ToF investigation of furanic polymer foams before and after carbonization: Aromatic rearrangement and surviving furanic structures. Eur. Polym. J. 2008, 44, 2938–2943. [Google Scholar] [CrossRef]
- Srivastava, V.K.; Pizzi, A. Characterization and preparation of wood-furanic foams. J. Renew. Mater. 2014, 2, 201–206. [Google Scholar] [CrossRef]
- Xi, X.; Pizzi, A.; Amirou, S. Melamine–Glyoxal–Glutaraldehyde Wood Panel Adhesives without Formaldehyde. Polymers 2018, 10, 22. [Google Scholar]
- Xi, X.; Wu, Z.; Pizzi, A. Furfuryl alcohol-aldehyde plywood adhesive resins. J. Adhes. 2019, 95, 1–25. [Google Scholar] [CrossRef]
- Li, J.; Zhang, A.; Zhang, S.; Gao, Q.; Zhang, W.; Li, J. Larch tannin-based rigid phenolic foam with high compressive strength, low friability, and low thermal conductivity reinforced by cork powder. Compos. Part B Eng. 2019, 156, 368–377. [Google Scholar] [CrossRef]
- Wang, L.; Xiu, S.; Shahbazi, A. Combustion characteristics of bio-oil from swine manure/crude glycerol co-liquefaction by thermogravimetric analysis technology. Energy Sources Part A 2016, 38, 2250–2257. [Google Scholar] [CrossRef]
- Merle, J.; Birot, M.; Deleuze, H.; Mitterer, C.; Carré, H.; Charrier-El Bouhtoury, F. New biobased foams from wood byproducts. Mater. Des. 2016, 91, 186–192. [Google Scholar] [CrossRef]
- Li, X.; Lacoste, C.; Pizzi, A.; Fierro, V.; Celzard, A. Physical properties of tannin/furanic resin foamed by different blowing agents. Bioresources 2013, 8, 743–752. [Google Scholar] [CrossRef]
- Lacoste, C.; Pizzi, A.; Basso, M.C.; Laborie, M.-P.; Celzard, A. Pinus pinaster tannin/furanic foams: Part 2: Physical properties. Ind. Crops Prod. 2014, 61, 531–536. [Google Scholar] [CrossRef]
- Basso, M.C.; Lagel, M.C.; Pizzi, A.; Celzard, A.; Abdalla, S. First tools for tannin-furanic foams design. Bioresources 2015, 10, 5233–5241. [Google Scholar] [CrossRef]
- Tondi, G.; Link, M.; Kolbitsch, C. Lignin-based foams: Production process and characterization. BioResources 2016, 11, 2972–2986. [Google Scholar] [CrossRef]
- Hu, X.M.; Wang, D.M.; Cheng, W.M. Effect of polyethylene glycol on the mechanical property, microstructure, thermal stability, and flame resistance of phenol–urea–formaldehyde foams. J. Mater. Sci. 2014, 49, 1556–1565. [Google Scholar] [CrossRef]
- Jayaraman, K.; Kok, M.V.; Gokalp, I. Combustion properties and kinetics of different biomass samples using TG–MS technique. J. Therm. Anal. Calorim. 2017, 127, 1361–1370. [Google Scholar] [CrossRef]
- Li, P.S.; Wang, Q.; Xu, Q. Combustion reaction mechanism of four typical Chinese biomass by TG and DTG. Asia Pac. J. Chem. Eng. 2012, 7, S209–S215. [Google Scholar] [CrossRef]
- Yu, L.J.; Wang, S.; Jiang, X.M. Thermal analysis studies on combustion characteristics of seaweed. J. Therm. Anal. Calorim. 2008, 93, 611–617. [Google Scholar] [CrossRef]
- Chen, W.H.; Lin, Y.Y.; Liu, H.C. A comprehensive analysis of food waste derived liquefaction bio-oil properties for industrial application. Appl. Energy 2019, 237, 283–291. [Google Scholar] [CrossRef]
- Xi, X.; Pizzi, A.; Gerardin, C. Glucose-biobased Non-Isocyanate Polyurethane Rigid Foams. J. Renew. Mater. 2019, 7, 301–312. [Google Scholar] [CrossRef]
- Shindo, A.; Izumino, K. Structural variation during pyrolysis of furfuryl alcohol and furfural-furfuryl alcohol resins. Carbon 1994, 32, 1233–1243. [Google Scholar] [CrossRef]
- Guigo, N.; Mija, A.; Vincent, L. Chemorheological analysis and model-free kinetics of acid catalysed furfuryl alcohol polymerization. Phys. Chem. Chem. Phys. 2007, 9, 5359–5366. [Google Scholar] [CrossRef]
- Maciel, G.E.; Chuang, I.S.; Myers, G.E. Carbon 13NMR study of cured furfuryl alcohol resins using cross polarization and magic-angle spinning. Macromolecules 1982, 15, 1218–1220. [Google Scholar] [CrossRef]
- Deng, S.; Du, G.; Li, X. Performance, reaction mechanism, and characterization of glyoxal–monomethylol urea (G–MMU) resin. Ind. Eng. Chem. Res. 2014, 53, 5421–5431. [Google Scholar] [CrossRef]



| No. | Furfuryl Alcohol (g) | Glyoxal (g) | Olive Powder (g) | p-TSA (g) | Blowing Agent (g) |
|---|---|---|---|---|---|
| F1 (4.1) | 14.7 | 11.6 | 4 | 3.6 | 1.5 |
| F2 (4.0) | 14.7 | 14.5 | 4 | 3.6 | 1.5 |
| F3 (4.2) | 11.76 | 14.5 | 4 | 3.6 | 1.5 |
| No. | Tf/℃ | tp/min | Density (kg/m3) | 2 h Water Absorption (%) | Cell Diameter (μm) |
|---|---|---|---|---|---|
| F1 | 75 ± 2 | 7 | 61.1 ± 3 | 352.6 | 758 ± 15 |
| F2 | 65 ± 2 | 10 | 69.1 ± 2 | 279.3 | 850 ± 20 |
| F3 | 57 ± 2 | 11 | 61.7 ± 3 | 332.4 | 795.3 ± 15 |
| Combustion Properties | F1 | F2 | F3 |
|---|---|---|---|
| Ti (°C) | 197.22 | 197.08 | 201.52 |
| Tmax (°C) | 567.67 | 578.33 | 539.33 |
| Tf (°C) | 625 | 647.33 | 615.00 |
| DTGmax (% °C−1) | 0.090 | 0.089 | 0.082 |
| DTGmean (% °C−1) | 4.55 | 4.33 | 4.70 |
| S (%2 K−3 min−2) × 10−7 | 0.1687 | 0.153 | 0.154 |
156 Da= Furfuryl alcohol-Glyoxal |
232 Da= Glyoxal-Furfuryl alcohol-Glyoxal hydrates. One of the aldehyde groups combines with water to form hydrates.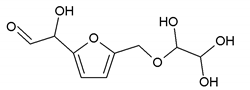 |
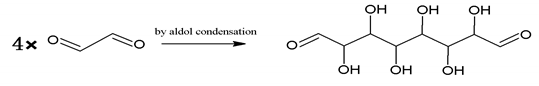
| 232 = 4 × glyoxal, by aldol condensation |
273 Da = Glyoxal hydrates-Furfuryl alcohol-Glyoxal hydrates with Na+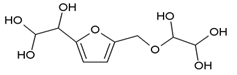 |
360.5 Da = 361 Da (with Na+)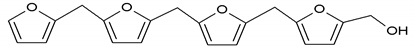 |
410 Da = Furfuryl alcohol-Glyoxal-Furfuryl alcohol-Glyoxal-Furfuryl alcohol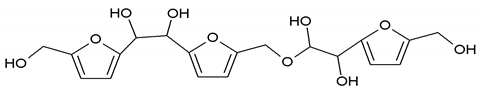 |
| 605 Da (cal 604 Da) = FA-G-FA-G-FA-G-FA-G, minus one molecule of water |
| 623 Da = Furfuryl alcohol-Glyoxal-Furfuryl alcohol-Glyoxal-Furfuryl alcohol-Glyoxal-Furfuryl alcohol-Glyoxal |
| 799 Da = G-FA-G-FA-G-FA-G-FA-G-FA-G, minus two molecule of water |
| 993 Da = G-FA-G-FA-G-FA-G-FA-G-FA-G-G-FA-G, minus three molecule of water |
| 1187 Da = G-FA-G-FA-G-FA-G-FA-G-FA-G-G-FA-G-G-FA-G, minus four molecule of water |
| 1381 Da = G-FA-G-FA-G-FA-G-FA-G-FA-G-G-FA-G-G-FA-G-G-FA-G, minus five molecule of water |
| 1576 Da (cal 1575Da) =G-FA-G-FA-G-FA-G-FA-G-FA-G-G-FA-G-G-FA-G-G-FA-G-G-FA-G, minus six molecule of water |
| 1769 Da = G-FA-G-FA-G-FA-G-FA-G-FA-(-G-G-FA-G-)5, minus 7 molecule of water |
| 1964 Da (cal 1963) = G-FA-G-FA-G-FA-G-FA-G-FA-(-G-G-FA-G-)6, minus 8 molecule of water |
| 2158 Da (cal 2157) = G-FA-G-FA-G-FA-G-FA-G-FA-(-G-G-FA-G-)7, minus 9 molecule of water |
| 2352 Da (cal 2351) = G-FA-G-FA-G-FA-G-FA-G-FA-(-G-G-FA-G-)8, minus 10 molecule of water |
| 2546 Da (cal 2545) = G-FA-G-FA-G-FA-G-FA-G-FA-(-G-G-FA-G-)9, minus 11 molecule of water |
| 2741 Da (cal 2739) = G-FA-G-FA-G-FA-G-FA-G-FA-(-G-G-FA-G-)10, minus 12 molecule of water |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xi, X.; Pizzi, A.; Lei, H.; Du, G.; Zhou, X.; Lin, Y. Characterization and Preparation of Furanic-Glyoxal Foams. Polymers 2020, 12, 692. https://doi.org/10.3390/polym12030692
Xi X, Pizzi A, Lei H, Du G, Zhou X, Lin Y. Characterization and Preparation of Furanic-Glyoxal Foams. Polymers. 2020; 12(3):692. https://doi.org/10.3390/polym12030692
Chicago/Turabian StyleXi, Xuedong, Antonio Pizzi, Hong Lei, Guanben Du, Xiaojian Zhou, and Yuying Lin. 2020. "Characterization and Preparation of Furanic-Glyoxal Foams" Polymers 12, no. 3: 692. https://doi.org/10.3390/polym12030692
APA StyleXi, X., Pizzi, A., Lei, H., Du, G., Zhou, X., & Lin, Y. (2020). Characterization and Preparation of Furanic-Glyoxal Foams. Polymers, 12(3), 692. https://doi.org/10.3390/polym12030692









