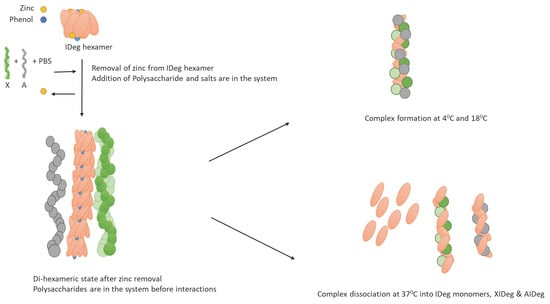
Clinically Relevant Insulin Degludec and Its Interaction with Polysaccharides: A Biophysical Examination
Abstract
1. Introduction
2. Methodology
2.1. Materials
2.2. Preparation of Polymeric-Insulin Complex (PIC) for Stability Test Using Hydrodynamical Analysis
2.3. Intrinsic Viscosity
2.4. Particle Size Distribution (PSD)
2.5. Zeta Potential (ζ) Analysis
2.6. Sedimentation Velocity
2.7. Statistical Analysis
3. Results and Discussion
3.1. Intrinsic Viscosity
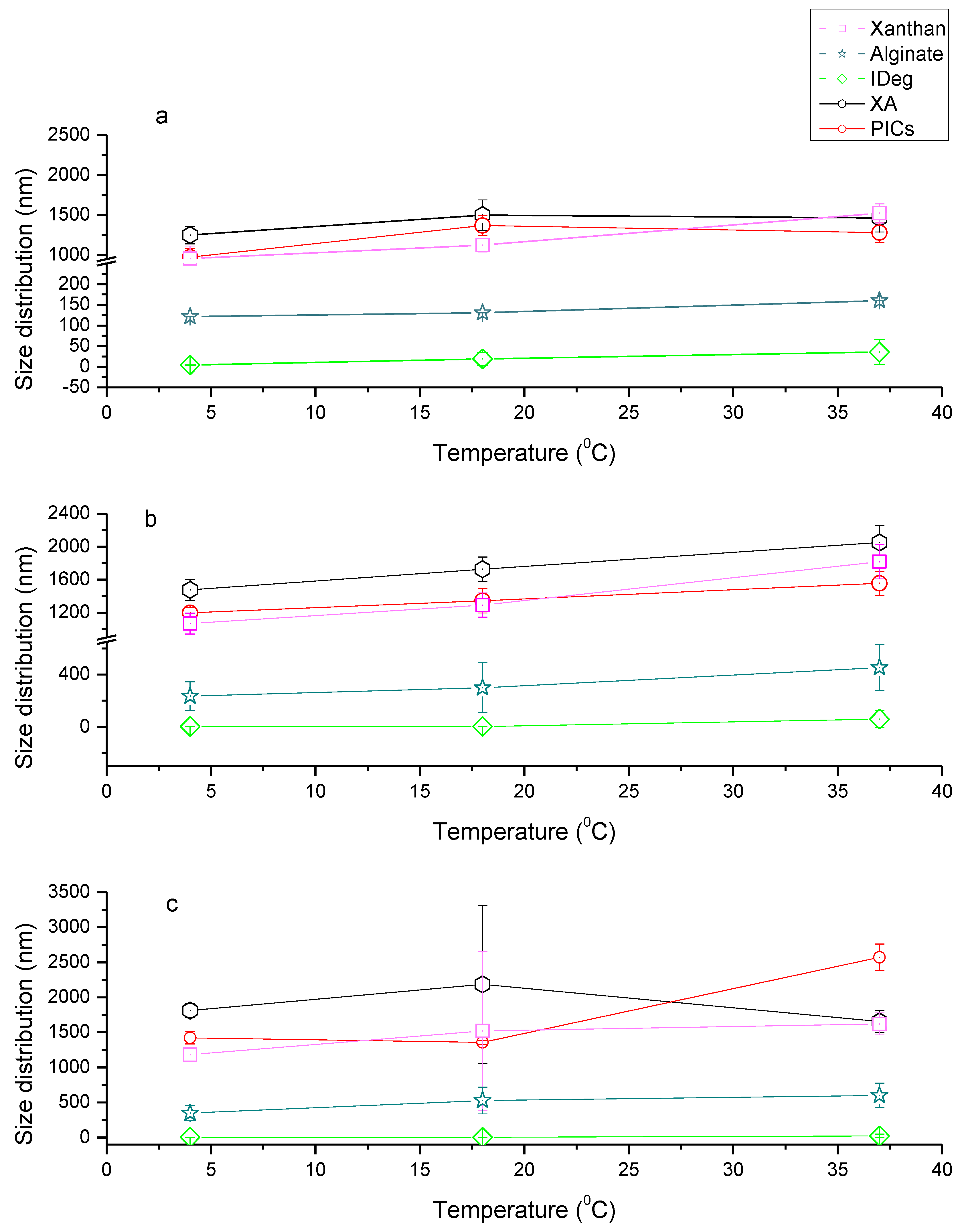
3.2. Particle Size Distribution
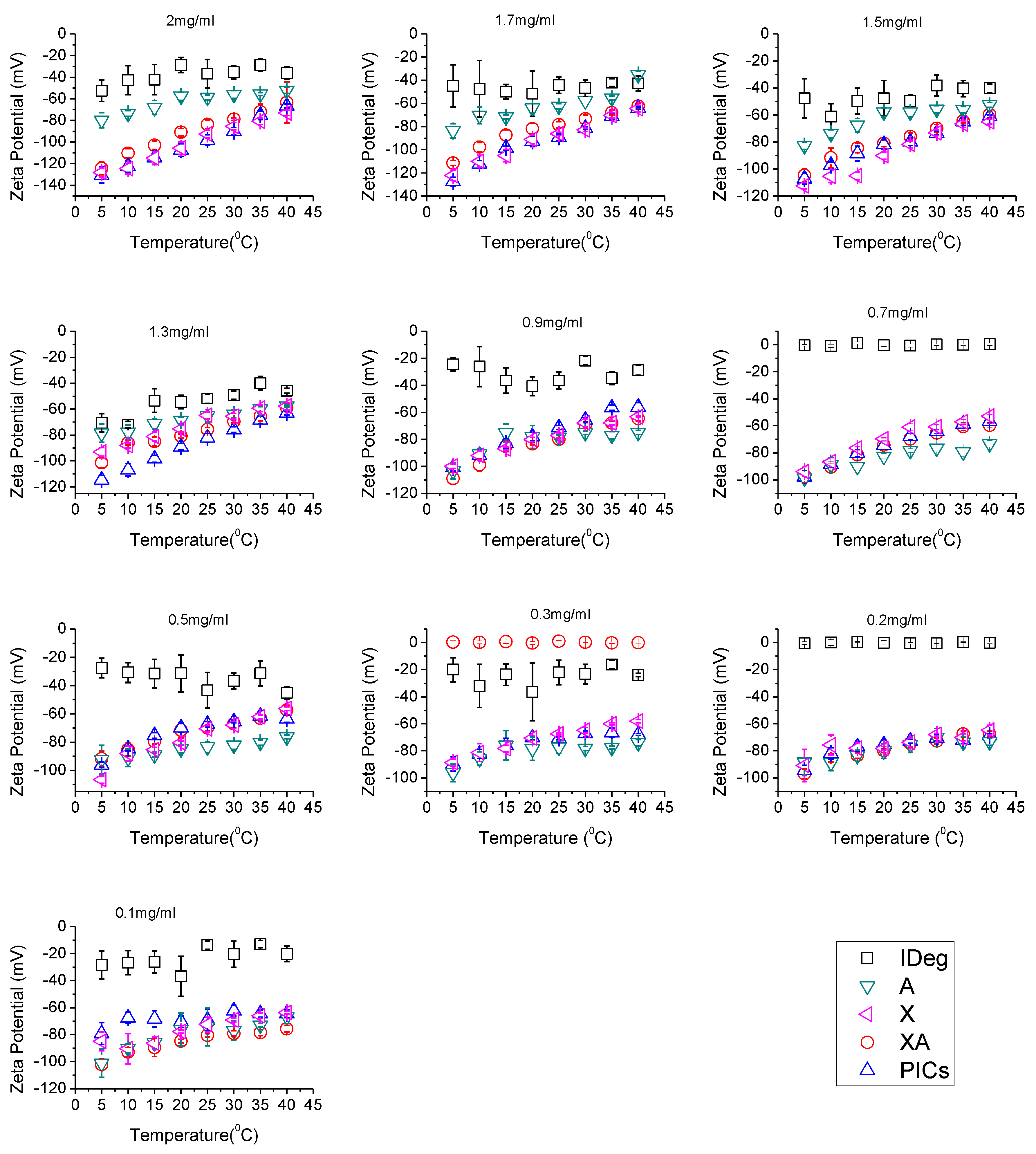
3.3. Zeta (ζ) Potential Analysis
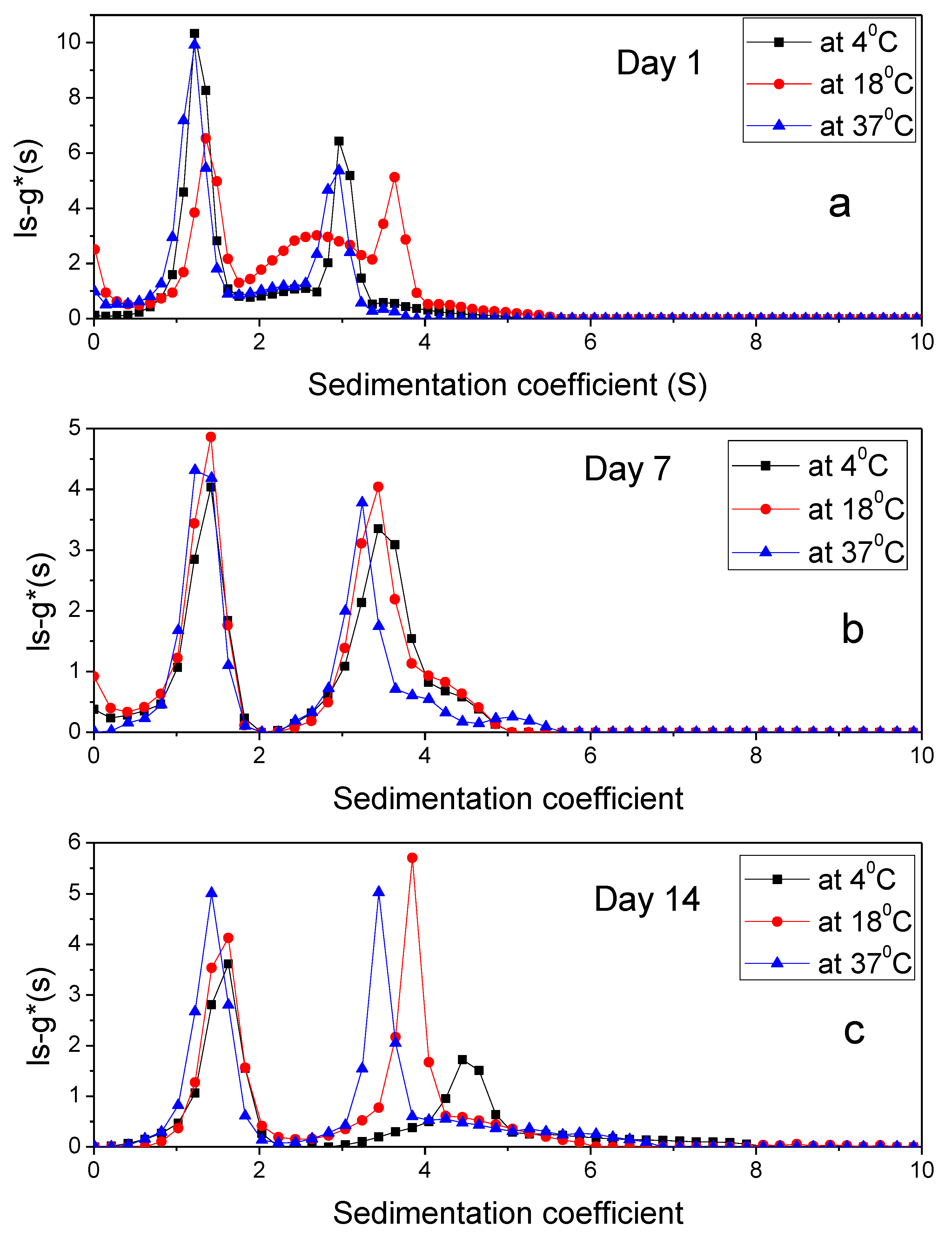
3.4. Sedimentation Velocity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| A | Alginate |
| X | Xanthan |
| IDeg | Insulin degludec |
| XA | Mixture of xanthan and alginate |
| PBS | Phosphate Buffer Saline |
| PIC | Polymeric insulin complex |
| PPC | Protein–polysaccharide complexes |
| s020w | Sedimentation coefficient, corrected for non-ideality, temperature and buffer conditions (1S = 1Svedberg = 1 × 10−13 sec). |
References
- Shelke, N.B.; James, R.; Laurencin, C.T.; Kumbar, S.G. Polysaccharide biomaterials for drug delivery and regenerative engineering. Polym. Adv. Technol. 2014, 25, 448–460. [Google Scholar] [CrossRef]
- Poli, A.; Anzelmo, G.; Fiorentino, G.; Nicolaus, B.; Tommonaro, G.; Di, P. Polysaccharides from Wastes of Vegetable Industrial Processing: New Opportunities for Their Eco-Friendly Re-Use. In Biotechnology of Biopolymers; IntechOpen: Rijeka, Croatia, 2011; pp. 33–56. [Google Scholar]
- Malafaya, P.B.; Silva, G.A.; Reis, R.L. Natural–origin polymers as carriers and scaffolds for biomolecules and cell delivery in tissue engineering applications. Adv. Drug Deliv. Rev. 2007, 59, 207–233. [Google Scholar] [CrossRef] [PubMed]
- Xu, A.Y.; Melton, L.D.; Williams, M.A.; McGillivray, D.J. Protein and polysaccharide conjugates as emerging scaffolds for drug delivery systems. Int. J. Nanotechnol. 2017, 14, 470–480. [Google Scholar] [CrossRef]
- Woo, V.C. New Insulins and New Aspects in Insulin Delivery. Can. J. Diabetes 2015, 39, 335–343. [Google Scholar] [CrossRef]
- Vora, J.; Cariou, B.; Evans, M.; Gross, J.L.; Harris, S.; Landstedt-Hallin, L.; Mithal, A.; Rodriguez, M.R.; Meneghini, L. Clinical use of insulin degludec. Diabetes Res. Clin. Pr. 2015, 109, 19–31. [Google Scholar] [CrossRef]
- Rees, D.A.; Welsh, E.J. Secondary and Tertiary Structure of Polysaccharides in Solutions and Gels. Angew. Chem. Int. Ed. 1977, 16, 214–224. [Google Scholar] [CrossRef]
- García-Ochoa, F.; Santos, V.E.; Casas, J.A.; Gomez, E. Xanthan gum: production, recovery, and properties. Biotechnol. Adv. 2000, 18, 549–579. [Google Scholar]
- Katzbauer, B. Properties and applications of xanthan gum. Polym. Degrad. Stab. 1998, 59, 81–84. [Google Scholar] [CrossRef]
- Matthew, B. The Phase Behaviour of Xanthan Based Biopolymer Mixtures. Ph.D. Thesis, University of Nottingham, Nottingham, UK, September 2005. [Google Scholar]
- Fareez, I.M.; Lim, S.M.; Mishra, R.K.; Ramasamy, K. Chitosan coated alginate–xanthan gum bead enhanced pH and thermotolerance of Lactobacillus plantarum LAB12. Int. J. Boil. Macromol. 2015, 72, 1419–1428. [Google Scholar] [CrossRef]
- Pongjanyakul, T.; Puttipipatkhachorn, S. Xanthan–alginate composite gel beads: Molecular interaction and in vitro characterization. Int. J. Pharm. 2007, 331, 61–71. [Google Scholar] [CrossRef]
- Kumar, A.; Rao, K.M.; Han, S.S. Development of sodium alginate-xanthan gum based nanocomposite scaffolds reinforced with cellulose nanocrystals and halloysite nanotubes. Polym. Test. 2017, 63, 214–225. [Google Scholar] [CrossRef]
- Mounica, R.M.; Shanmugam, V.; Rajesh, K. Design and Characterization of Insulin Nanoparticles for Oral Delivery. Int. J. Innov. Pharm. Res. 2012, 3, 238–243. [Google Scholar]
- Kulkarni, R.V.; Sa, B. Electroresponsive Polyacrylamide-grafted-xanthan Hydrogels for Drug Delivery. J. Bioact. Compat. Polym. 2009, 24, 368–384. [Google Scholar] [CrossRef]
- Shalviri, A.; Liu, Q.; Abdekhodaie, M.J.; Wu, X.Y. Novel modified starch–xanthan gum hydrogels for controlled drug delivery: Synthesis and characterization. Carbohydr. Polym. 2010, 79, 898–907. [Google Scholar] [CrossRef]
- Caddeo, C.; Nácher, A.; Díez-Sales, O.; Merino-Sanjuán, M.; Fadda, A.M.; Manconi, M. Chitosan–xanthan gum microparticle-based oral tablet forcolon-targeted and sustained delivery of quercetin. J. Microencapsul. 2014, 31, 1464–5246. [Google Scholar] [CrossRef]
- Tahtat, D.; Mahlous, M.; Benamer, S.; Khodja, A.N.; Oussedik-Oumehdi, H.; Laraba-Djebari, F. Oral delivery of insulin from alginate/chitosan crosslinked by glutaraldehyde. Int. J. Boil. Macromol. 2013, 58, 160–168. [Google Scholar] [CrossRef]
- Reis, C.P.; Ribeiro, A.J.; Neufeld, R.J.; Veiga, F. Alginate microparticles as novel carrier for oral insulin delivery. Biotechnol. Bioeng. 2007, 96, 977–989. [Google Scholar] [CrossRef]
- Sarmento, B.; Ribeiro, A.; Veiga, F.; Sampaio, P.; Neufeld, R.; Ferreira, D. Alginate/Chitosan Nanoparticles are Effective for Oral Insulin Delivery. Pharm. Res. 2007, 24, 2198–2206. [Google Scholar] [CrossRef]
- Schmitt, C.; Turgeon, S.L. Protein/polysaccharide complexes and coacervates in food systems. Adv. Colloid Interface Sci. 2011, 167, 63–70. [Google Scholar] [CrossRef]
- Ganzevles, R.A.; Zinoviadou, K.; Van Vliet, T.; Stuart, M.A.C.; De Jongh, H.H.J. Modulating Surface Rheology by Electrostatic Protein/Polysaccharide Interactions. Langmuir 2006, 22, 10089–10096. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Bandyopadhyay, P. Polysaccharide-protein interactions and their relevance in food colloids. Complex World Polysacch. 2012, 14, 395–406. [Google Scholar]
- Devi, N.; Sarmah, M.; Khatun, B.; Maji, T.K. Encapsulation of active ingredients in polysaccharide–protein complex coacervates. Adv. Colloid Interface Sci. 2017, 239, 136–145. [Google Scholar] [CrossRef]
- Van Witteloostuijn, S.B.; Pedersen, S.L.; Jensen, K.J. Half-Life Extension of Biopharmaceuticals using Chemical Methods: Alternatives to PEGylation. ChemMedChem 2016, 11, 2474–2495. [Google Scholar] [CrossRef]
- Fukushima, M.; Hirayama, T.; Ichikawa, M.; Kitazawa, I.; Kojima, K.; Sakai, T.; Takatsu, Y.; Ohtaki, T. Glycosaminoglycan-Conjugated Insulin Derivatives Suitable for Once-Daily Formulations. ACS Omega 2019, 4, 5517–5525. [Google Scholar] [CrossRef]
- Miltenburg, A.M.M.; Prohn, M.; van Kuijk, J.H.M.; Tiessen, R.G.; de Kort, M.; Berg, R.J.W. Half-life prolongation of therapeutic proteins by conjugation to atiii-binding pentasaccharides: A first-in-human study of carbocarrier® insulin. Br. J. Clin. Pharmacol. 2013, 75, 1221–1230. [Google Scholar] [CrossRef]
- Theisen, A. Refractive Increment Data- Book for Polymer and Biomolecular Scientists; Nottingham University Press: Nottingham, UK, 2000. [Google Scholar]
- Harding, S.E. The intrinsic viscosity of Biological Macromolecules. Progress in Measurement, Interaction and Application to Structure in Dilute Solution. Prog. Biophys. Mol. Biol. 1997, 68, 207–262. [Google Scholar] [CrossRef]
- Dam, J.; Schuck, P. Calculating Sedimentation Coefficient Distributions by Direct Modeling of Sedimentation Velocity Concentration Profiles. Methods Enzymol. 2004, 384, 185–212. [Google Scholar]
- Brunchi, C.-E.; Bercea, M.; Morariu, S.; Dascalu, M. Some properties of xanthan gum in aqueous solutions: effect of temperature and pH. J. Polym. Res. 2016, 23, 123. [Google Scholar] [CrossRef]
- Higiro, J.; Herald, T.J.; Alavi, S. Rheological study of xanthan and locust beam gum interaction in dilute solution. Food Res. Int. 2005, 39, 165–175. [Google Scholar] [CrossRef]
- Milas, M.; Rinaudo, M.; Tinland, B. The viscosity dependence on concentration, molecular weight and shear rate of xanthan solutions. Polym. Bull. 1985, 14, 157–164. [Google Scholar] [CrossRef]
- Belalia, F.; Djelali, N.E. Rheological properties of sodium alginate solutions. Rev. Roum. Chim. 2014, 59, 135–145. [Google Scholar]
- Galván, Z.R.N.; Soares, L.D.S.; Medeiros, E.A.A.; Soares, N.D.F.F.; Ramos, A.M.; Coimbra, J.S.D.R.; De Oliveira, E.B. Rheological Properties of Aqueous Dispersions of Xanthan Gum Containing Different Chloride Salts Are Impacted by both Sizes and Net Electric Charges of the Cations. Food Biophys. 2018, 13, 186–197. [Google Scholar] [CrossRef]
- Seright, R.; Henrici, B. Xanthan Stability at Elevated Temperatures. SPE Reserv. Eng. 1990, 5, 52–60. [Google Scholar] [CrossRef]
- Morris, E.R. Ordered conformation of xanthan in solutions and “weak gels”: Single helix, double helix – or both? Food Hydrocoll. 2019, 86, 18–25. [Google Scholar] [CrossRef]
- Adams, G.G.; Alzahrani, Q.; Jiwani, S.I.; Meal, A.; Morgan, P.S.; Coffey, F.; Kok, S.; Rowe, A.J.; Harding, S.E.; Chayen, N.; et al. Glargine and degludec: Solution behaviour of higher dose synthetic insulins. Sci. Rep. 2017, 7, 7287. [Google Scholar] [CrossRef]
- Havelund, S.; Ribel, U.; Hubálek, F.; Hoeg-Jensen, T.; Wahlund, P.-O.; Jonassen, I. Investigation of the Physico-Chemical Properties that Enable Co-Formulation of Basal Insulin Degludec with Fast-Acting Insulin Aspart. Pharm. Res. 2015, 32, 2250–2258. [Google Scholar] [CrossRef]
- Jonassen, I.; Havelund, S.; Hoeg-Jensen, T.; Steensgaard, D.B.; Wahlund, P.O.; Ribel, U. Design of the novel protraction mechanism of insulin degludec, an ultra-long-acting basal insulin. Pharm. Res.-Dordr. 2012, 29, 2104–2114. [Google Scholar] [CrossRef]
- Salgın, S.; Takaç, S.; Özdamar, T.H. Effect of ionic environments on the adsorption and diffusion characteristics of serine alkaline protease enzyme in polyethersulfone ultrafiltration membranes. J. Colloid Interface Sci. 2006, 299, 806–814. [Google Scholar] [CrossRef]
- Malhotra, A.; Coupland, J.N. The effect of surfactants on the solubility, zeta potential, and viscosity of soy protein isolates. Food Hydrocoll. 2004, 18, 101–108. [Google Scholar] [CrossRef]
- Hunter, R.J. Zeta Potential in Colloid Science Principles and Applications; Academic Press Inc. Ltd.: London, UK, 1981. [Google Scholar]
- Drab, S.R.; Philis-Tsimikas, A. A new option for glycemic control: Insulin degludec, a new-generation basal insulin with an ultralong duration of action. Pharmacotherapy 2014, 34, 291–302. [Google Scholar] [CrossRef]
- Dhami, R.; Harding, S.E.; Jones, T.; Hughes, T.; Mitchell, J.R.; To, K.-M. Physico-chemical studies on a commercial food-grade xanthan—I. Characterisation by sedimentation velocity, sedimentation equilibrium and viscometry. Carbohydr. Polym. 1995, 27, 93–99. [Google Scholar] [CrossRef]
- Abdelhameed, A.S.; Ang, S.; Morris, G.A.; Smith, I.; Lawson, C.; Gähler, R.; Wood, S.; Harding, S.E. An analytical ultracentrifuge study on ternary mixtures of konjac glucomannan supplemented with sodium alginate and xanthan gum. Carbohydr. Polym. 2010, 81, 145–148. [Google Scholar] [CrossRef]
- Tinland, B.; Rinaudo, M. Dependence of the stiffness of the xanthan chain on the external salt concentration. Macromolecules 1989, 22, 1863–1865. [Google Scholar] [CrossRef]
- Chazeau, L.; Milas, M.; Rinaudo, M. Conformations of Xanthan in Solution: Analysis by Steric Exclusion Chromatography. Int. J. Polym. Anal. Charact. 1995, 2, 21–29. [Google Scholar] [CrossRef]
- Steensgaard, D.B.; Schluckebier, G.; Strauss, H.M.; Norrman, M.; Thomsen, J.K.; Friderichsen, A.V.; Havelund, S.; Jonassen, I. Ligand-Controlled Assembly of Hexamers, Dihexamers, and Linear Multihexamer Structures by the Engineered Acylated Insulin Degludec. Biochemistry 2013, 52, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.Q.; Hu, X.; Wang, C.; Ai, L. Polysaccharides: Structure and Solubility. In Solubility of Polysaccharides; Xu, Z., Ed.; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar]
- Harding, S.E.; Tombs, M.P. An Introduction to Polysaccharide Biotechnology; Taylor and Francis: London, UK, 1998. [Google Scholar]
- Correia, J.J.; Johnson, M.L.; Welss, G.H.; Yphantis, D.A. Numerical study of the Johnston-Ogston effect in two-component systems. Biophys. Chem. 1976, 5, 255–264. [Google Scholar] [CrossRef]

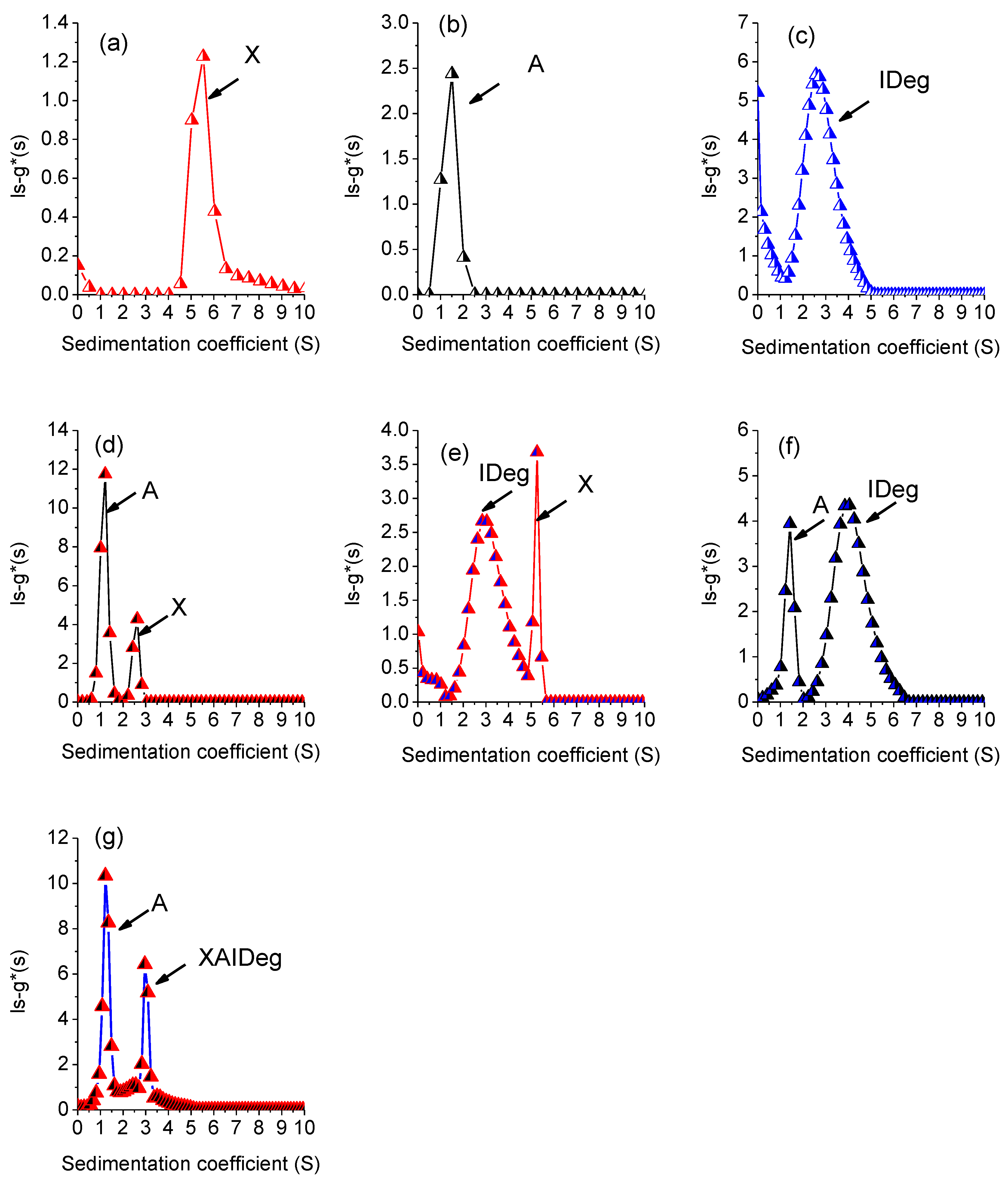
| Polymer | Sedimentation Coefficient (S) | Area Under Curve (AUC) |
|---|---|---|
| Alginate | 1.5 ± 0.016 | 2.09 |
| Xanthan | 5.5 ± 0.310 | 1.35 |
| IDeg | 2.6 ± 0.002 | 9.85 |
| Mixture | Experimental AUC Values | Interaction Status | |||
|---|---|---|---|---|---|
| Expected AUC | Identity | Sedimentation Coefficient (S) | AUC (Fringe Units) | ||
| A + X | 3.44 | A | 1.0 ± 0.003 | 4.18 | S value decreased for both, 1.5 to 1S for A and 5.5 to 2.5S for X. No interactions due to co-sedimentation |
| X | 2.5 ± 0.002 | 1.11 | |||
| X + IDeg | 11.20 | X | 5.25 ± 0.001 | 0.91 | S value for IDeg change from 2.6S to 2.83S and for xanthan from 5.5S to 5.25S. No complex formation as sum of the two components AUC is very low. Earlier sedimentation |
| IDeg | 2.83 ± 0.003 | 4.79 | |||
| A + IDeg | 11.94 | A | 1.42 ± 0.003 | 1.93 | S value for Alginate decreased (from 1.5 to 1.42S) and Insulin increased (from 2.6 to 4.04S). Reduction in AUC for both polymers. Some interactions |
| IDeg | 4.04 ± 0.001 | 7.84 | |||
| 4 °C | 18 °C | 37 °C | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Days | Sedimentation Coefficient (S) | AUC (Fringe Units) | Interaction Status | Sedimentation Coefficient (S) | AUC (Fringe Units) | Interaction Status | Sedimentation Coefficient (S) | AUC (Fringe Units) | Interaction Status |
| 1 | 1.21 ± 0.002 2.96 ± 0.001 | 3.69 2.05 | Interaction occurred. A part of alginate sedimented separately and PIC formation occurred | 1.39 ± 0.002 2.66 ± 0.007 | 3.77 1.54 | Interaction occurred. A part of alginate sedimented separately and PIC formation occurred | 1.32 ± 0.002 3.25 ± 0.007 | 2.029 2.605 | Interaction occurred. A part of alginate sedimented separately and PIC formation occurred |
| 7 | 1.41 ± 0.004 3.43 ± 0.004 | 1.89 6.212 | 1.46 ± 0.003 4.03 ± 0.002 | 2.56 8.64 | 1.26 ± 0.003 3.34 ± 0.002 5.00 ± 0.015 | 2.35 1.17 1.17 | Some interactions may have occurred but most of the components remained separated | ||
| 14 | 1.6 ± 0.004 4.3 ± 0.003 | 3.76 2.198 | 1.48 ± 0.002 3.85 ± 0.002 | 2.43 8.53 | 1.37 ± 0.003 3.65 ± 0.004 | 2.32 1.75 | Some interactions may have occurred but most of the components remained separated and sedimented quickly, hence only two peaks were visible | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiwani, S.I.; Huang, S.; Beji, O.; Gyasi-Antwi, P.; Gillis, R.B.; Adams, G.G. Clinically Relevant Insulin Degludec and Its Interaction with Polysaccharides: A Biophysical Examination. Polymers 2020, 12, 390. https://doi.org/10.3390/polym12020390
Jiwani SI, Huang S, Beji O, Gyasi-Antwi P, Gillis RB, Adams GG. Clinically Relevant Insulin Degludec and Its Interaction with Polysaccharides: A Biophysical Examination. Polymers. 2020; 12(2):390. https://doi.org/10.3390/polym12020390
Chicago/Turabian StyleJiwani, Shahwar Imran, Sha Huang, Oritsegidenene Beji, Philemon Gyasi-Antwi, Richard B. Gillis, and Gary G. Adams. 2020. "Clinically Relevant Insulin Degludec and Its Interaction with Polysaccharides: A Biophysical Examination" Polymers 12, no. 2: 390. https://doi.org/10.3390/polym12020390
APA StyleJiwani, S. I., Huang, S., Beji, O., Gyasi-Antwi, P., Gillis, R. B., & Adams, G. G. (2020). Clinically Relevant Insulin Degludec and Its Interaction with Polysaccharides: A Biophysical Examination. Polymers, 12(2), 390. https://doi.org/10.3390/polym12020390