Thermomechanical and Morphological Properties of Poly(ethylene terephthalate)/Anhydrous Calcium Terephthalate Nanocomposites
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Terephthalate Salts
2.2. Production of Composite Materials
2.3. Characterization of Terephthalate Salts and rPET/CATAS Nanocomposites
3. Results and Discussion
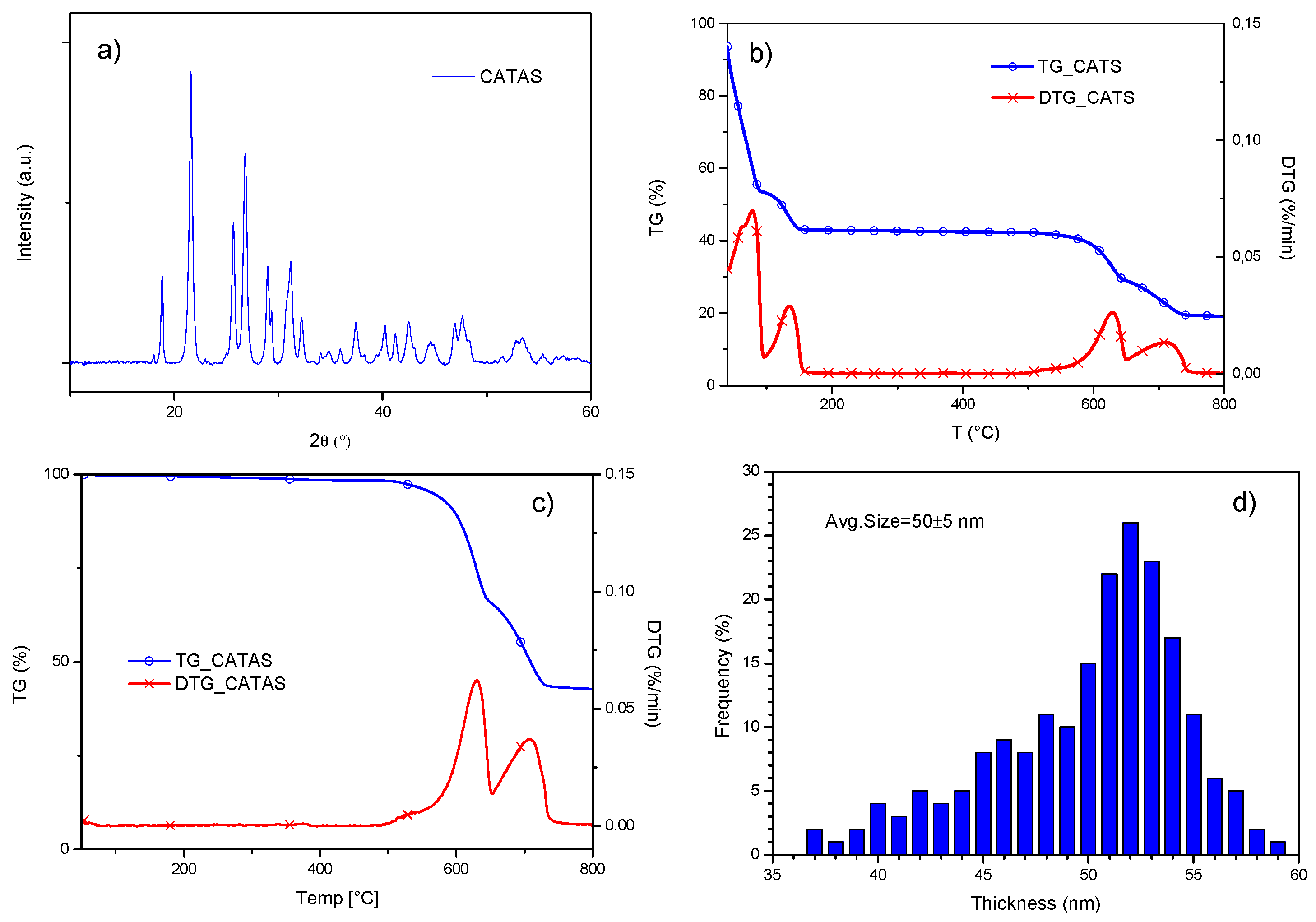
3.1. Characterization of Calcium Terephthalate Salts
3.2. Characteristics of rPET/CATAS Nanocomposites
3.2.1. DMTA (Dynamic Mechanical Thermal Analysis)
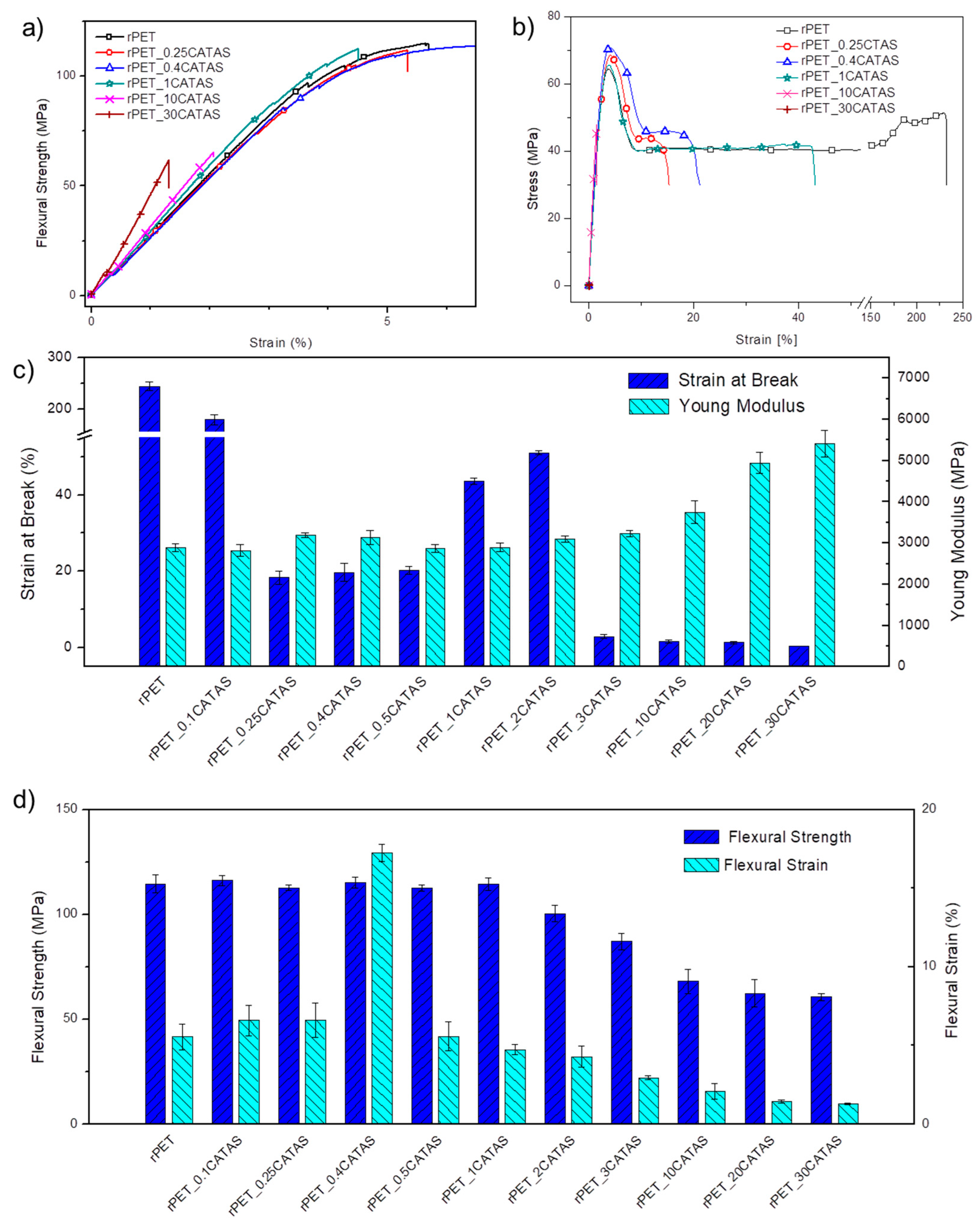
3.2.2. Mechanical Characterization (Flexural and Tensile Tests)
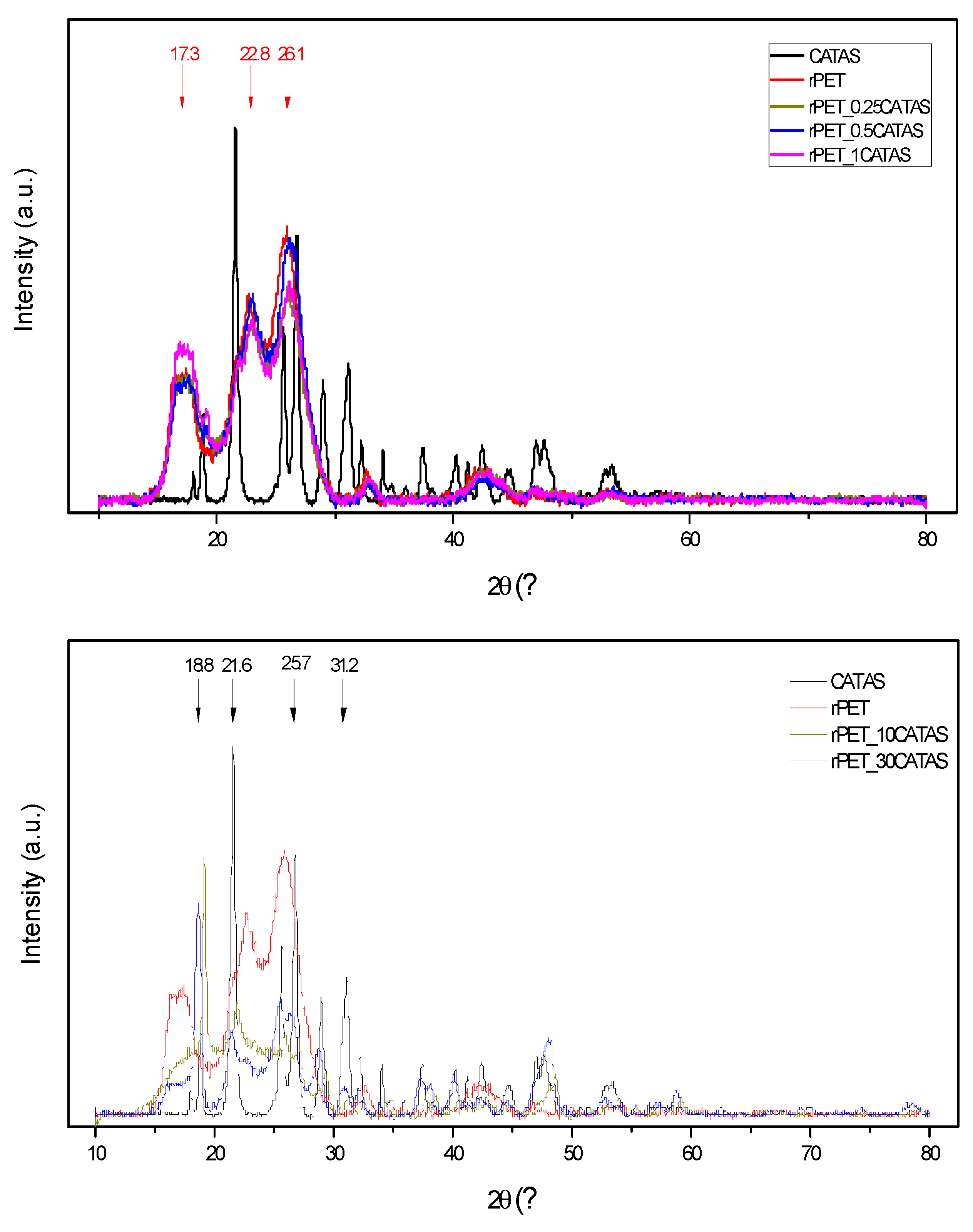
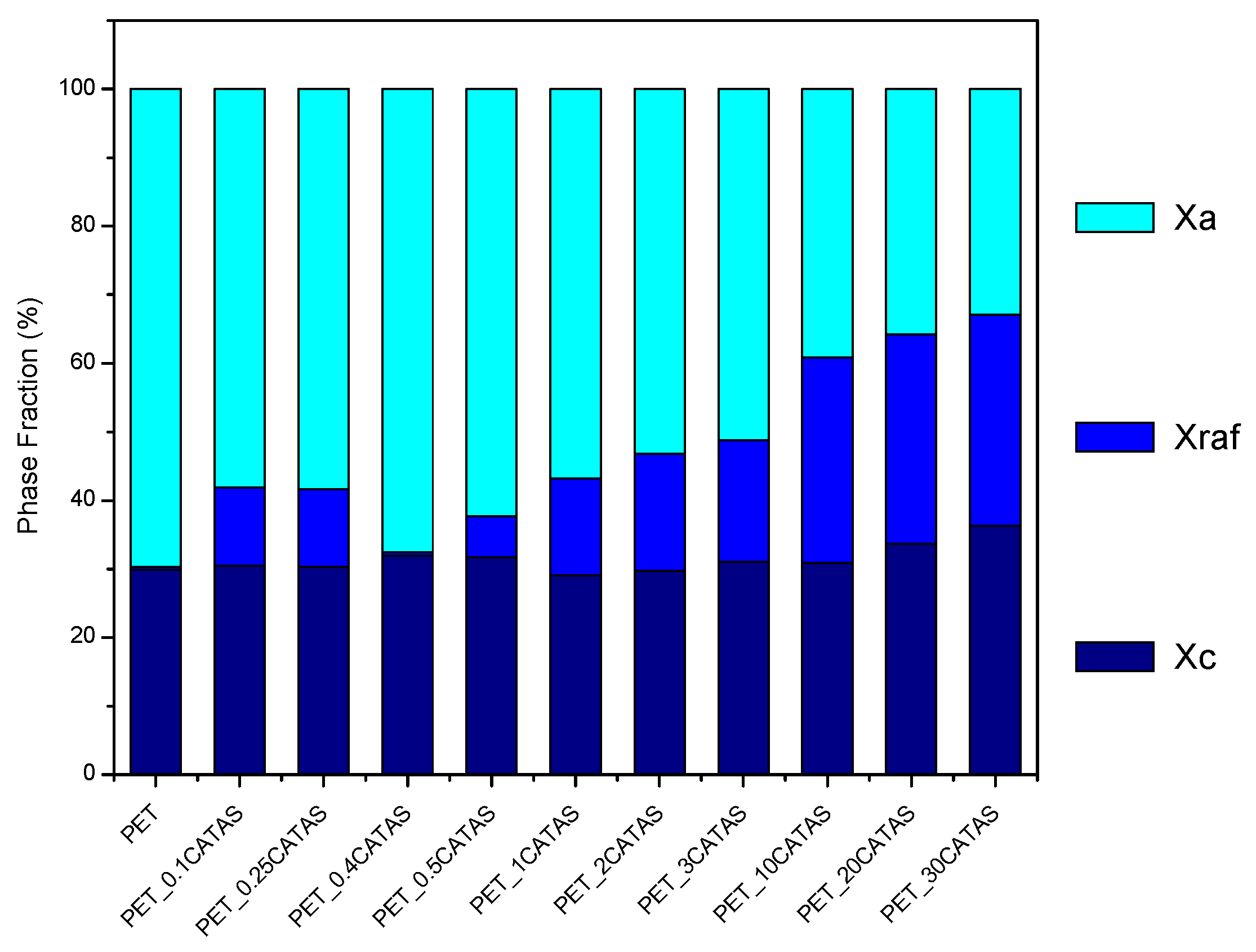
3.2.3. XRD Analysis
3.2.4. Morphological Analysis
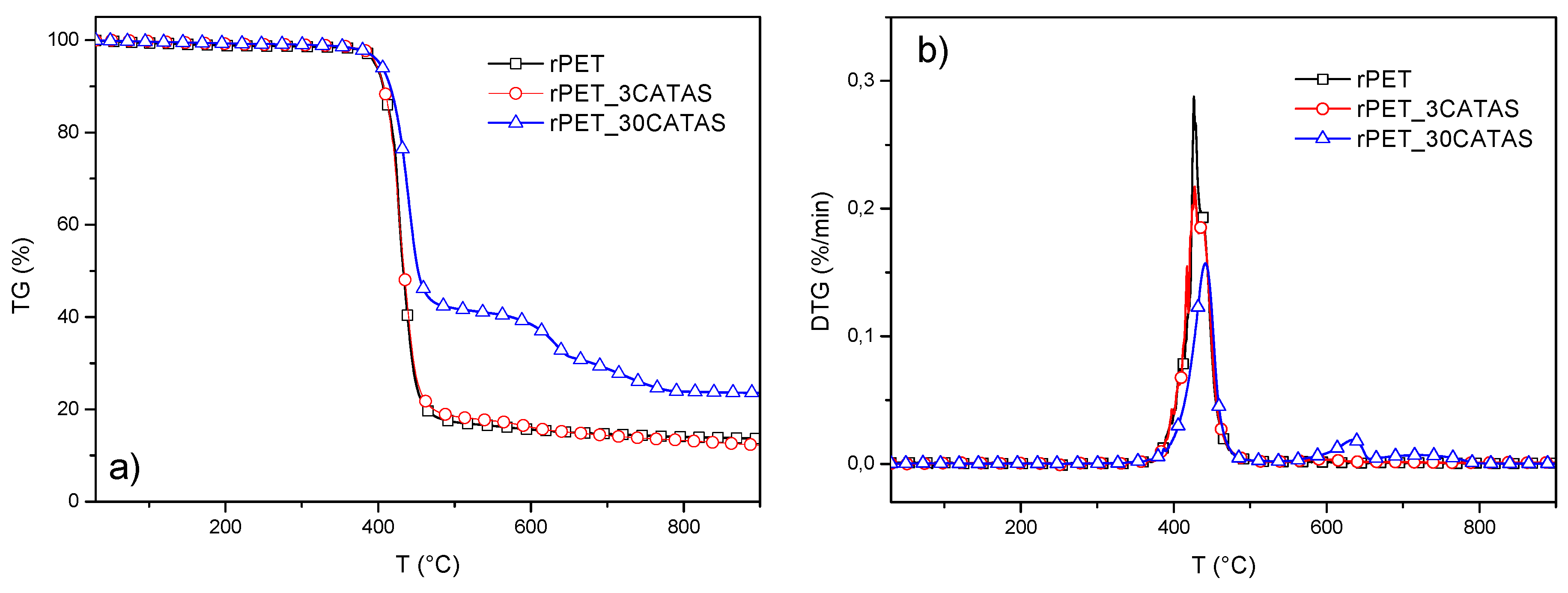
3.2.5. Thermal Characterization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Allen, R.D. Waste PET: A Renewable Resource. Joule 2019, 3, 910. [Google Scholar] [CrossRef]
- Thomas, S.; Rane, A.V.; Kanny, K.; Abitha, V.K.; Thomas, M.G. Recycling of Polyethylene Terephthalate Bottles; William Andrew: Norwich, NY, USA, 2018. [Google Scholar]
- Palkopoulou, S.; Joly, C.; Feigenbaum, A.; Papaspyrides, C.D.; Dole, P. Critical review on challenge tests to demonstrate decontamination of polyolefins intended for food contact applications. Trends Food Sci. Technol. 2016, 49, 110–120. [Google Scholar] [CrossRef]
- Chowreddy, R.R.; Nord-Varhaug, K.; Rapp, F. Recycled Poly(ethylene Terephthalate)/Clay Nanocomposites: Rheology, Thermal and Mechanical Properties. J. Polym. Environ. 2019, 27, 37–49. [Google Scholar] [CrossRef]
- Pegoretti, A.; Kolarik, J.; Peroni, C.; Migliaresi, C. Recycled poly(ethylene terephthalate)/layered silicate nanocomposites: Morphology and tensile mechanical properties. Polymer 2004, 45, 2751–2759. [Google Scholar] [CrossRef]
- Tsai, T.-Y.; Bunekar, N.; Wu, T.-C. The Role of Organomodified Different LDHs on Recycled Poly(ethylene terephthalate) Nanocomposites. J. Chin. Chem. Soc. 2017, 64, 851–859. [Google Scholar] [CrossRef]
- Kráčalík, M.; Mikešová, J.; Puffr, R.; Baldrian, J.; Thomann, R.; Friedrich, C. Effect of 3D structures on recycled PET/organoclay nanocomposites. Polym. Bull. 2007, 58, 313–319. [Google Scholar] [CrossRef]
- Zare, Y. Recent progress on preparation and properties of nanocomposites from recycled polymers: A review. Waste Manag. 2013, 33, 598–604. [Google Scholar] [CrossRef]
- Aoyama, S.; Ismail, I.; Park, Y.T.; Macosko, C.W.; Ougizawa, T. Higher-Order Structure in Amorphous Poly(ethylene terephthalate)/Graphene Nanocomposites and Its Correlation with Bulk Mechanical Properties. ACS Omega 2019, 4, 1228–1237. [Google Scholar] [CrossRef]
- Dyosiba, X.; Ren, J.; Musyoka, N.M.; Langmi, H.W.; Mathe, M.; Onyango, M.S. Preparation of value-added metal-organic frameworks (MOFs) using waste PET bottles as source of acid linker. Sustain. Mater. Technol. 2016, 10, 10–13. [Google Scholar] [CrossRef]
- Doan, V.D.; Do, T.L.; Ho, T.M.T.; Le, V.T.; Nguyen, H.T. Utilization of waste plastic pet bottles to prepare copper-1, 4-benzenedicarboxylate metal-organic framework for methylene blue removal. Sep. Sci. Technol. 2020, 55, 444–455. [Google Scholar] [CrossRef]
- Jones, P.G.; Ossowski, J.; Kus, P.; Dix, I. Three Crystal Structures of Terephthalic Acid Salts of Simple Amines. Z. Für Nat. B 2009, 64, 865–870. [Google Scholar] [CrossRef]
- Kaduk, J.A. Terephthalate salts: Salts of monopositive cations. Acta Crystallogr. Sect. B Struct. Sci. 2000, 56, 474–485. [Google Scholar] [CrossRef] [PubMed]
- Panasyuk, G.P.; Azarova, L.A.; Budova, G.P.; Izotov, A.D. Synthesis and characterization of ammonium terephthalates. Inorg. Mater. 2002, 38, 385–389. [Google Scholar] [CrossRef]
- Panasyuk, G.P.; Khaddaj, M.; Privalov, V.I.; Miroshnichenko, I.V. Poly(ethylene terephthalate) Transformations during Autoclaving in Water Vapor. Plast. Massy 2002, 2, 27–31. [Google Scholar]
- Hou, J.; Ashling, C.W.; Collins, S.M.; Krajnc, A.; Zhou, C.; Longley, L.; Johnstone, D.; Chater, P.A.; Li, S.; Coudert, F. Metal-organic framework crystal-glass composites. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef]
- Mou, C.; Wang, L.; Deng, Q.; Huang, Z.; Li, J. Calcium terephthalate/graphite composites as anode materials for lithium-ion batteries. Ionics 2015, 21, 1893–1899. [Google Scholar] [CrossRef]
- Sachdeva, S.; Koper, S.J.H.; Sabetghadam, A.; Soccol, D.; Gravesteijn, D.J.; Kapteijn, F.; Sudhölter, E.J.R.; Gascon, J.; De Smet, L.C.P.M. Gas phase sensing of alcohols by Metal Organic Framework–polymer composite materials. ACS Appl. Mater. Interfaces 2017, 9, 24926–24935. [Google Scholar] [CrossRef]
- Stock, N.; Biswas, S. Synthesis of metal-organic frameworks (MOFs): Routes to various MOF topologies, morphologies, and composites. Chem. Rev. 2011, 112, 933–969. [Google Scholar] [CrossRef]
- Schmidt, B.V. Metal-Organic Frameworks in Polymer Science: Polymerization Catalysis, Polymerization Environment, and Hybrid Materials. Macromol. Rapid Commun. 2019, 41. [Google Scholar] [CrossRef]
- Wunderlich, B. Thermal Analysis of Polymeric Materials; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Matsuzaki, T.; Iitaka, Y. The crystal structure of calcium terephthalate trihydrate. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1972, 28, 1977–1981. [Google Scholar] [CrossRef]
- Alavi, M.A.; Morsali, A. Alkaline-earth metal carbonate, hydroxide and oxide nano-crystals synthesis methods, size and morphologies consideration. Nanocrystal 2011, 237–262. [Google Scholar] [CrossRef]
- Mazaj, M.; Mali, G.; Rangus, M.; Žunkovič, E.; Kaučič, V.; Zabukovec Logar, N. Spectroscopic studies of structural dynamics induced by heating and hydration: A case of calcium-terephthalate metal–organic framework. J. Phys. Chem. C 2013, 117, 7552–7564. [Google Scholar] [CrossRef]
- Dominici, F.; Puglia, D.; Luzi, F.; Sarasini, F.; Rallini, M.; Torre, L. A Novel Class of Cost Effective and High Performance Nanocomposites Based on Terephthalate Salts Reinforced Polyether Ether Ketone. Polymers 2019, 11, 2097. [Google Scholar] [CrossRef]
- Liang, P.-C.; Liu, H.-K.; Yeh, C.-T.; Lin, C.-H.; Zima, V.T.Z. Supramolecular Assembly of Calcium Metal− Organic Frameworks with Structural Transformations. Cryst. Growth Des. 2011, 11, 699–708. [Google Scholar] [CrossRef]
- Wang, L.; Mou, C.; Sun, Y.; Liu, W.; Deng, Q.; Li, J. Structure-property of metal organic frameworks calcium terephthalates anodes for lithium-ion batteries. Electrochim. Acta 2015, 173, 235–241. [Google Scholar] [CrossRef]
- Mazaj, M.; Zabukovec Logar, N.A. Phase Formation Study of Ca-Terephthalate MOF-Type Materials. Cryst. Growth Des. 2015, 15, 617–624. [Google Scholar] [CrossRef]
- Howarth, A.J.; Liu, Y.; Li, P.; Li, Z.; Wang, T.C.; Hupp, J.T.; Farha, O.K. Chemical, thermal and mechanical stabilities of metal–organic frameworks. Nat. Rev. Mater. 2016, 1, 15018. [Google Scholar] [CrossRef]
- Ahani, M.; Khatibzadeh, M.; Mohseni, M. Preparation and characterization of Poly(ethylene terephthalate)/hyperbranched polymer nanocomposites by melt blending. Nanocomposites 2016, 2, 29–36. [Google Scholar] [CrossRef]
- Golpour, M.; Pakizeh, M. Preparation and characterization of new PA-MOF/PPSU-GO membrane for the separation of KHI from water. Chem. Eng. J. 2018, 345, 221–232. [Google Scholar] [CrossRef]
- Strain, I.N.; Wu, Q.; Pourrahimi, A.M.; Hedenqvist, M.S.; Olsson, R.T.; Andersson, R.L. Electrospinning of recycled PET to generate tough mesomorphic fibre membranes for smoke filtration. J. Mater. Chem. A 2015, 3, 1632–1640. [Google Scholar] [CrossRef]
- Kim, J.; Choi, J.; Soo Kang, Y.; Won, J. Matrix effect of mixed-matrix membrane containing CO2-selective MOFs. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Shabafrooz, V.; Bandla, S.; Allahkarami, M.; Hanan, J.C. Graphene/polyethylene terephthalate nanocomposites with enhanced mechanical and thermal properties. J. Polym. Res. 2018, 25, 256. [Google Scholar] [CrossRef]
- Wang, X.-S.; Li, X.-G.; Yan, D. Thermal decomposition kinetics of poly (trimethylene terephthalate). Polym. Degrad. Stab. 2000, 69, 361–372. [Google Scholar] [CrossRef]
- Cacho-Bailo, F.; Téllez, C.; Coronas, J. Interactive thermal effects on metal–organic framework Polymer composite membranes. Chem. A Eur. J. 2016, 22, 9533–9536. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.-C.; Hahm, W.-G.; Im, S.-S.; Oh, S.-G. Poly(ethylene terephthalate)(PET) nanocomposites filled with fumed silicas by melt compounding. Macromol. Res. 2002, 10, 221–229. [Google Scholar] [CrossRef]
- Qiu, G.; Tang, Z.L.; Huang, N.X.; Gerking, L. Dual melting endotherms in the thermal analysis of Poly(ethylene terephthalate). J. Appl. Polym. Sci. 1998, 69, 729–742. [Google Scholar] [CrossRef]




| Sample Name | rPET [%] wt. | CATAS [%] wt. |
|---|---|---|
| rPET | 100 | --- |
| rPET_0.1CATAS | 99.9 | 0.1 |
| rPET_0.25CATAS | 99.75 | 0.25 |
| rPET_0.4CATAS | 99.6 | 0.4 |
| rPET_0.5CATAS | 99.5 | 0.5 |
| rPET_1CATAS | 99 | 1 |
| rPET_2CATAS | 98 | 2 |
| rPET_3CATAS | 97 | 3 |
| rPET_10CATAS | 90 | 10 |
| rPET_20CATAS | 80 | 20 |
| rPET_30CATAS | 70 | 30 |
| MATERIAL | G’ @ T = 50 °C (×109 Pa) | G’ @ T = 150 °C (×108 Pa) | Tg (at tan δ peak) (°C) |
|---|---|---|---|
| rPET | 0.89 | 0.61 | 90.41 |
| rPET_1CATAS | 1.00 | 0.68 | 90.48 |
| rPET_10CATAS | 1.19 | 0.96 | 90.89 |
| rPET_20CATAS | 1.47 | 1.54 | 91.29 |
| rPET_30CATAS | 1.92 | 2.76 | 93.43 |
| MATERIAL | Young’s Modulus (MPa) | Stress (MPa) | Strain at Maximum Stress (%) | Stress at Break (MPa) | Strain at Break (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| rPET | 2887 | ±178 | 65.5 | ±3.6 | 3.99 | ±0.21 | 41.0 | ±5.2 | 244.2 | ±16.9 |
| rPET_0.1CATAS | 2819 | ±283 | 61.7 | ±4.2 | 3.68 | ±0.23 | 41.7 | ±3.1 | 178.9 | ±20.7 |
| rPET_0.25CATAS | 3186 | ±115 | 68.9 | ±2.3 | 3.98 | ±0.28 | 41.9 | ±1.9 | 18.3 | ±3.6 |
| rPET_0.4CATAS | 3131 | ±342 | 69.4 | ±1.7 | 3.89 | ±0.27 | 42.4 | ±3.2 | 19.7 | ±4.5 |
| rPET_0.5CATAS | 2867 | ±201 | 66.8 | ±0.1 | 3.90 | ±0.28 | 39.6 | ±0.5 | 20.2 | ±2.3 |
| rPET_1CATAS | 2898 | ±206 | 67.4 | ±2.5 | 3.78 | ±0.26 | 43.2 | ±1.7 | 43.6 | ±1.6 |
| rPET_2CATAS | 3099 | ±145 | 64.8 | ±1.4 | 3.12 | ±0.38 | 39.4 | ±0.5 | 51.0 | ±0.9 |
| rPET_3CATAS | 3225 | ±148 | 61.9 | ±8.8 | 2.91 | ±0.36 | 61.8 | ±8.8 | 2.9 | ±1.1 |
| rPET_10CATAS | 3751 | ±549 | 45.5 | ±14.1 | 1.60 | ±0.42 | 45.5 | ±14.1 | 1.6 | ±0.7 |
| rPET_20CATAS | 4943 | ±517 | 36.5 | ±10.2 | 1.34 | ±0.51 | 36.5 | ±10.3 | 1.3 | ±0.5 |
| rPET_30CATAS | 5408 | ±645 | 0.3 | 0.0 | 0.04 | 0.01 | 0.3 | 0.0 | 0.3 | ±0.1 |
| Material | Ton (°C) | T20% (°C) | Tmax (°C) | Residual Mass (%) (at 900 °C) |
|---|---|---|---|---|
| rPET | 350 | 417 | 426 | 13.7 |
| rPET_3CATAS | 350 | 418 | 427 | 12.3 |
| rPET_30CATAS | 335 | 439 | 441 | 23.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dominici, F.; Sarasini, F.; Luzi, F.; Torre, L.; Puglia, D. Thermomechanical and Morphological Properties of Poly(ethylene terephthalate)/Anhydrous Calcium Terephthalate Nanocomposites. Polymers 2020, 12, 276. https://doi.org/10.3390/polym12020276
Dominici F, Sarasini F, Luzi F, Torre L, Puglia D. Thermomechanical and Morphological Properties of Poly(ethylene terephthalate)/Anhydrous Calcium Terephthalate Nanocomposites. Polymers. 2020; 12(2):276. https://doi.org/10.3390/polym12020276
Chicago/Turabian StyleDominici, Franco, Fabrizio Sarasini, Francesca Luzi, Luigi Torre, and Debora Puglia. 2020. "Thermomechanical and Morphological Properties of Poly(ethylene terephthalate)/Anhydrous Calcium Terephthalate Nanocomposites" Polymers 12, no. 2: 276. https://doi.org/10.3390/polym12020276
APA StyleDominici, F., Sarasini, F., Luzi, F., Torre, L., & Puglia, D. (2020). Thermomechanical and Morphological Properties of Poly(ethylene terephthalate)/Anhydrous Calcium Terephthalate Nanocomposites. Polymers, 12(2), 276. https://doi.org/10.3390/polym12020276









