Sulfonimide-Based Dendrimers: Progress in Synthesis, Characterization, and Potential Applications
Abstract
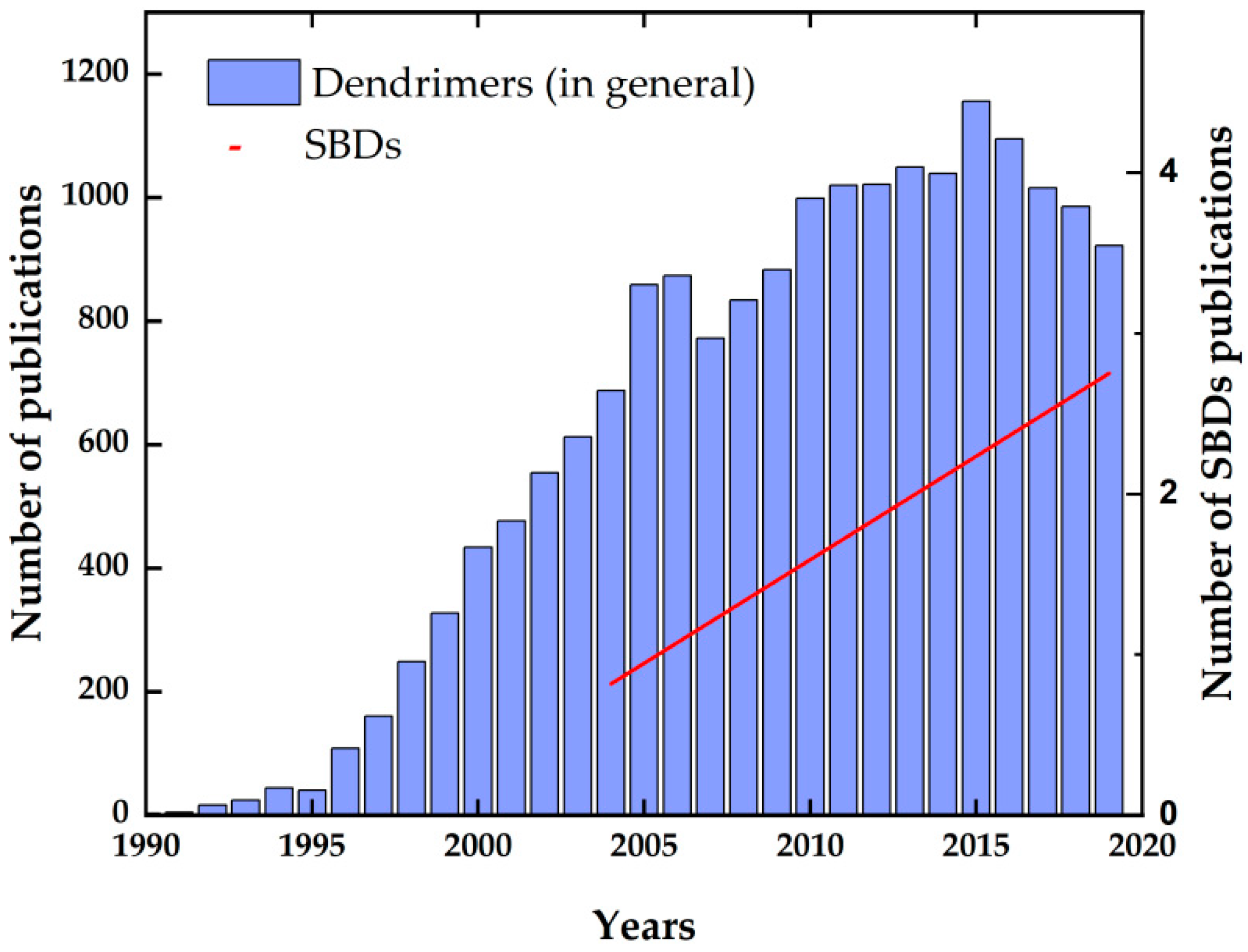
:1. Introduction
2. Properties
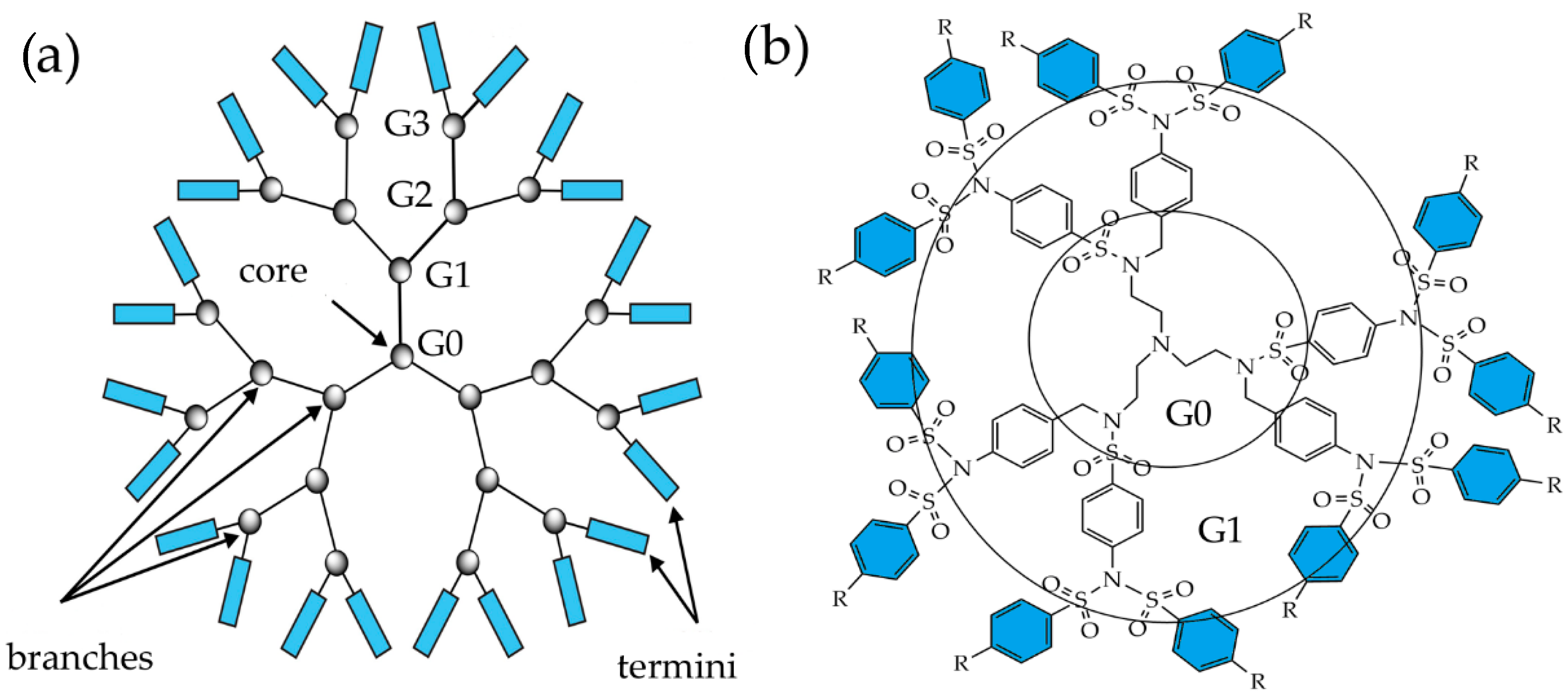
2.1. Structure
2.2. Isomeric Structures
2.3. Functional Groups
2.4. Molar Masses
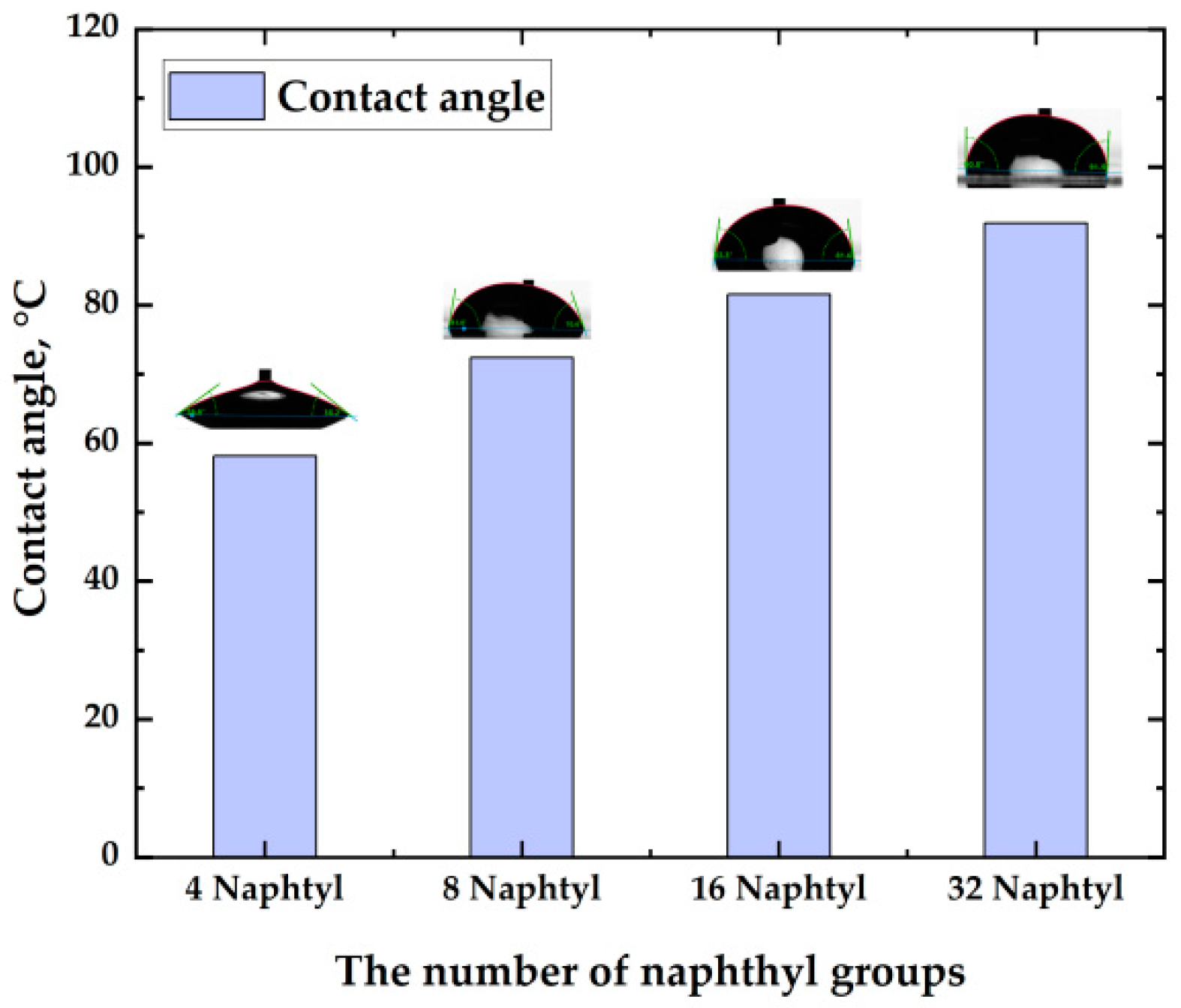
2.5. Solubility
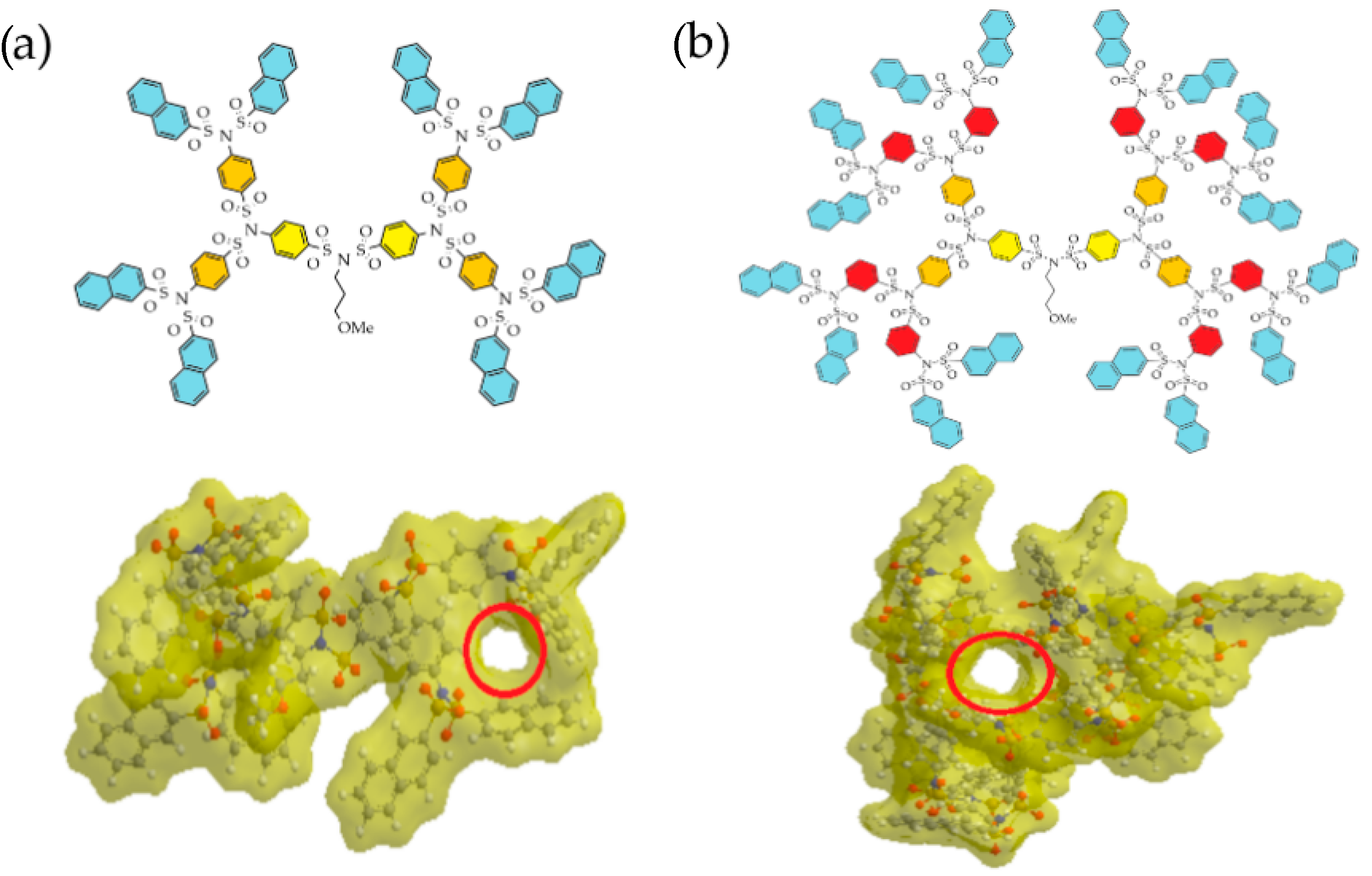
2.6. Voids and Cavities
3. Synthesis
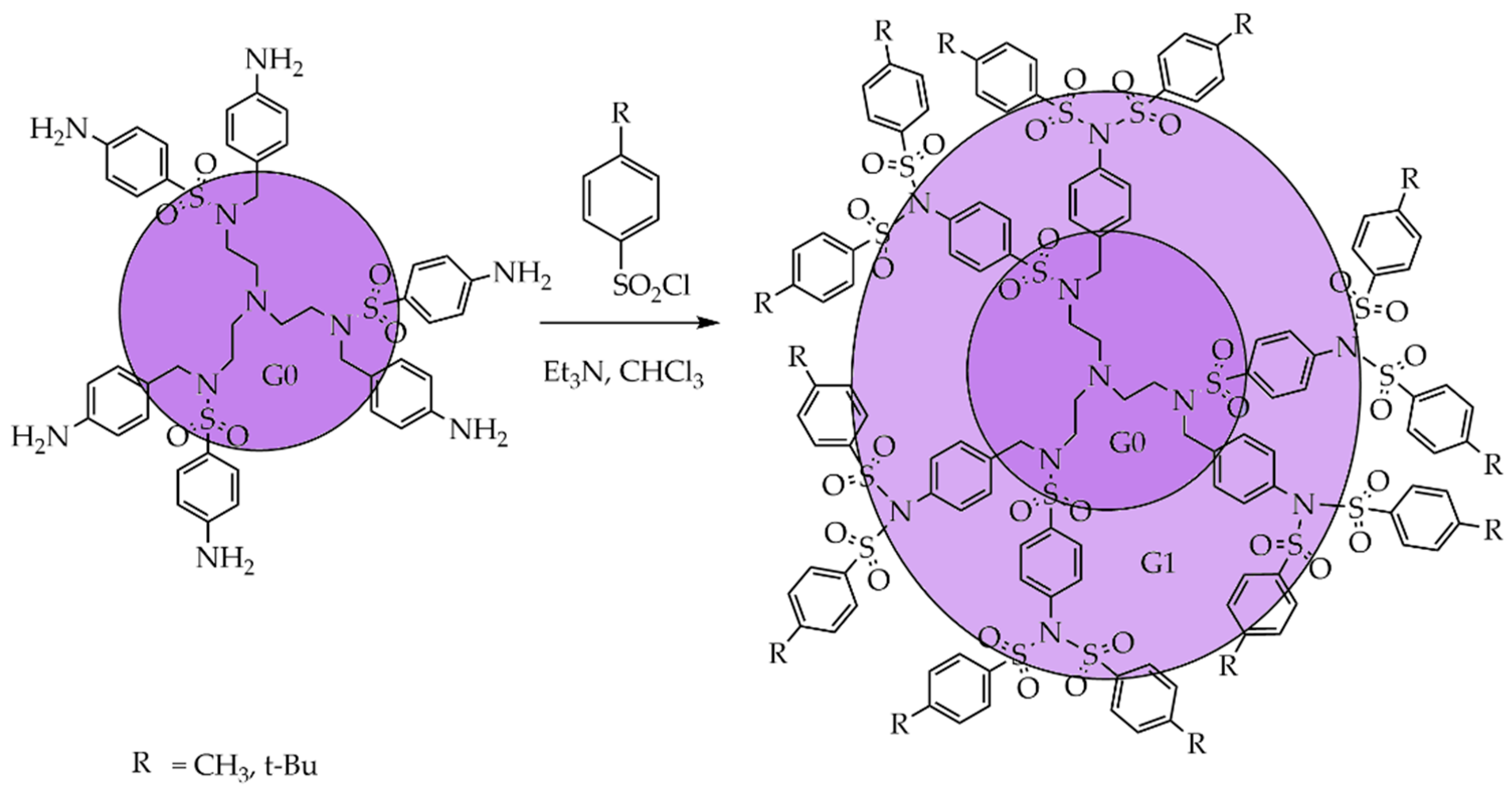
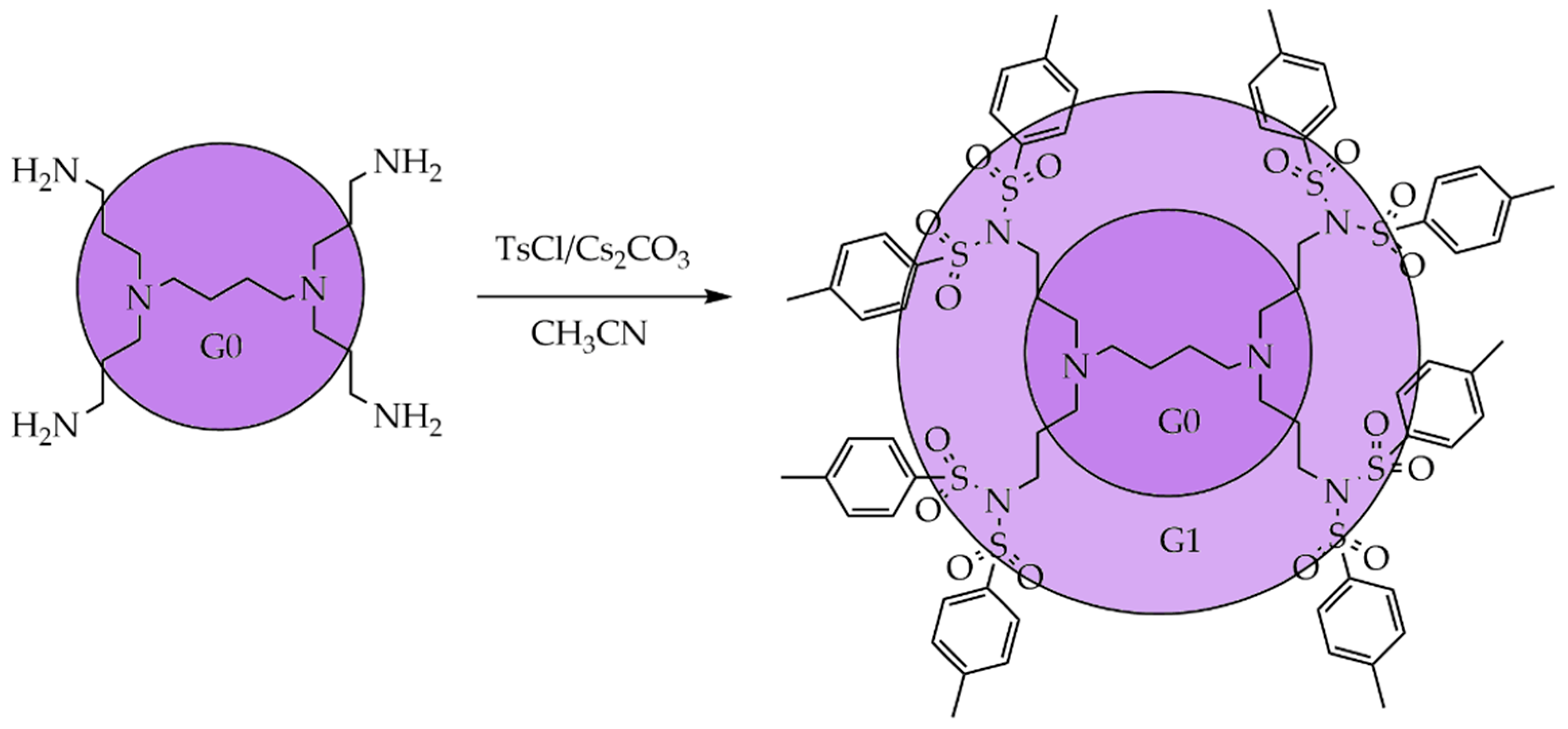
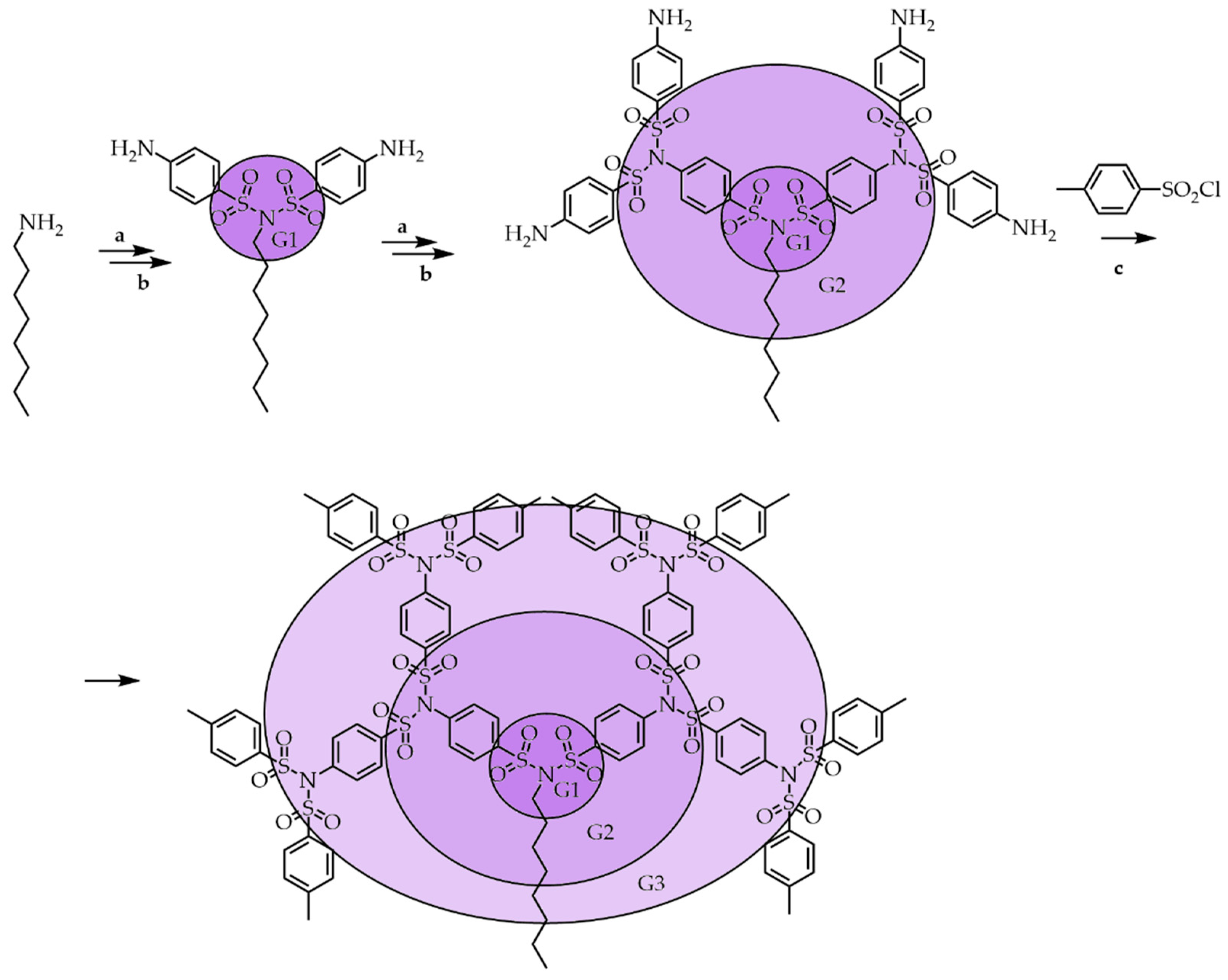
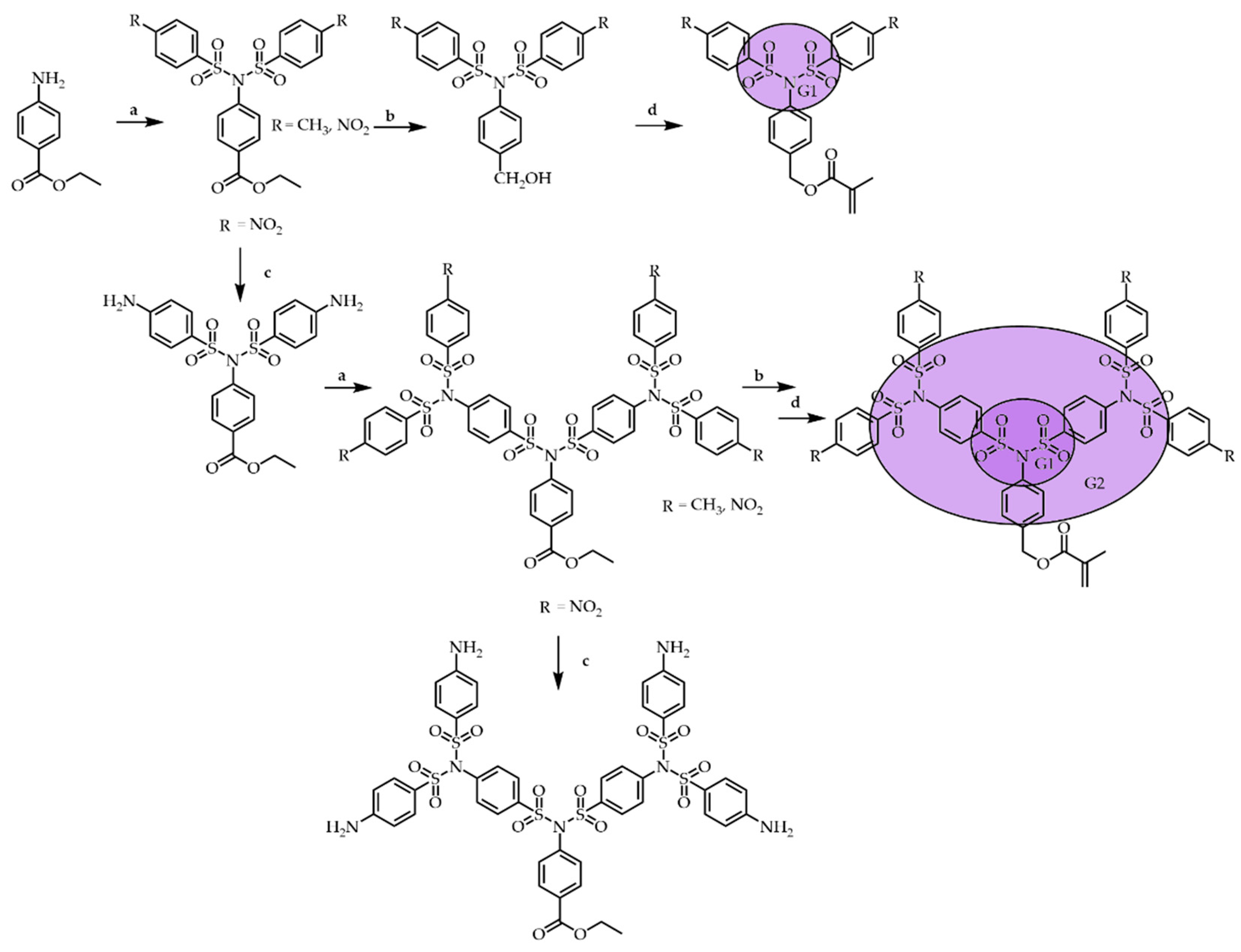
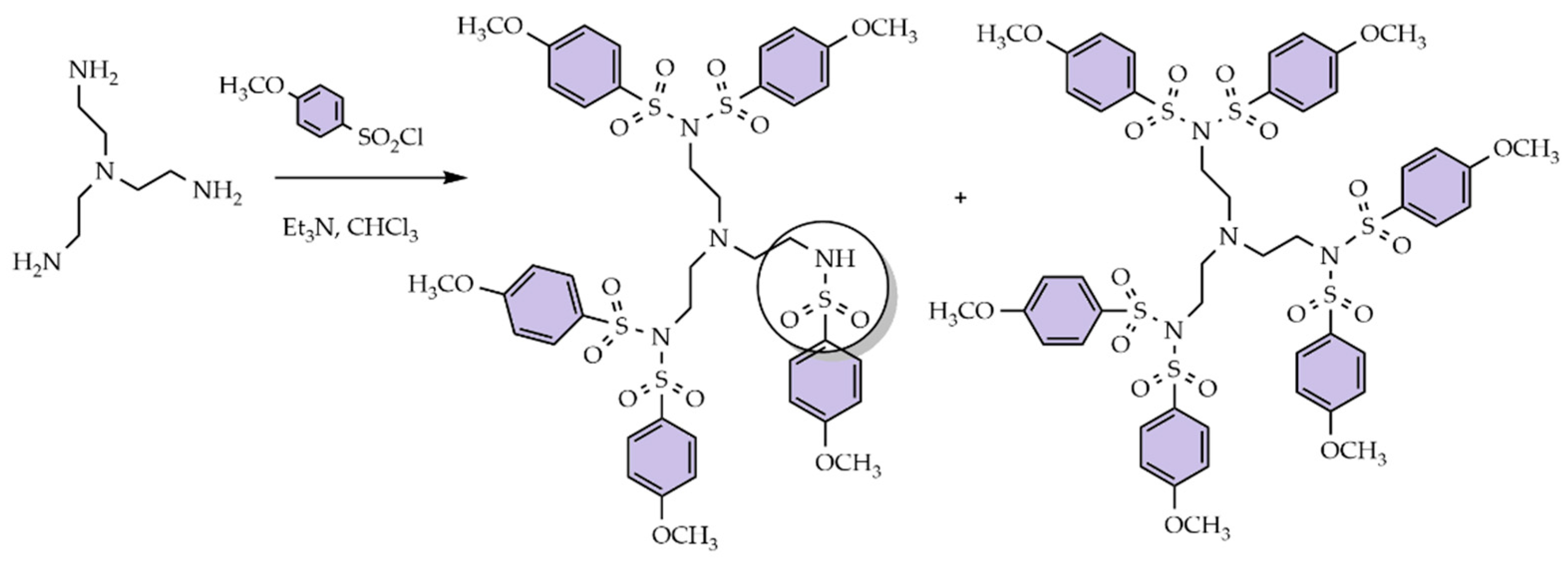
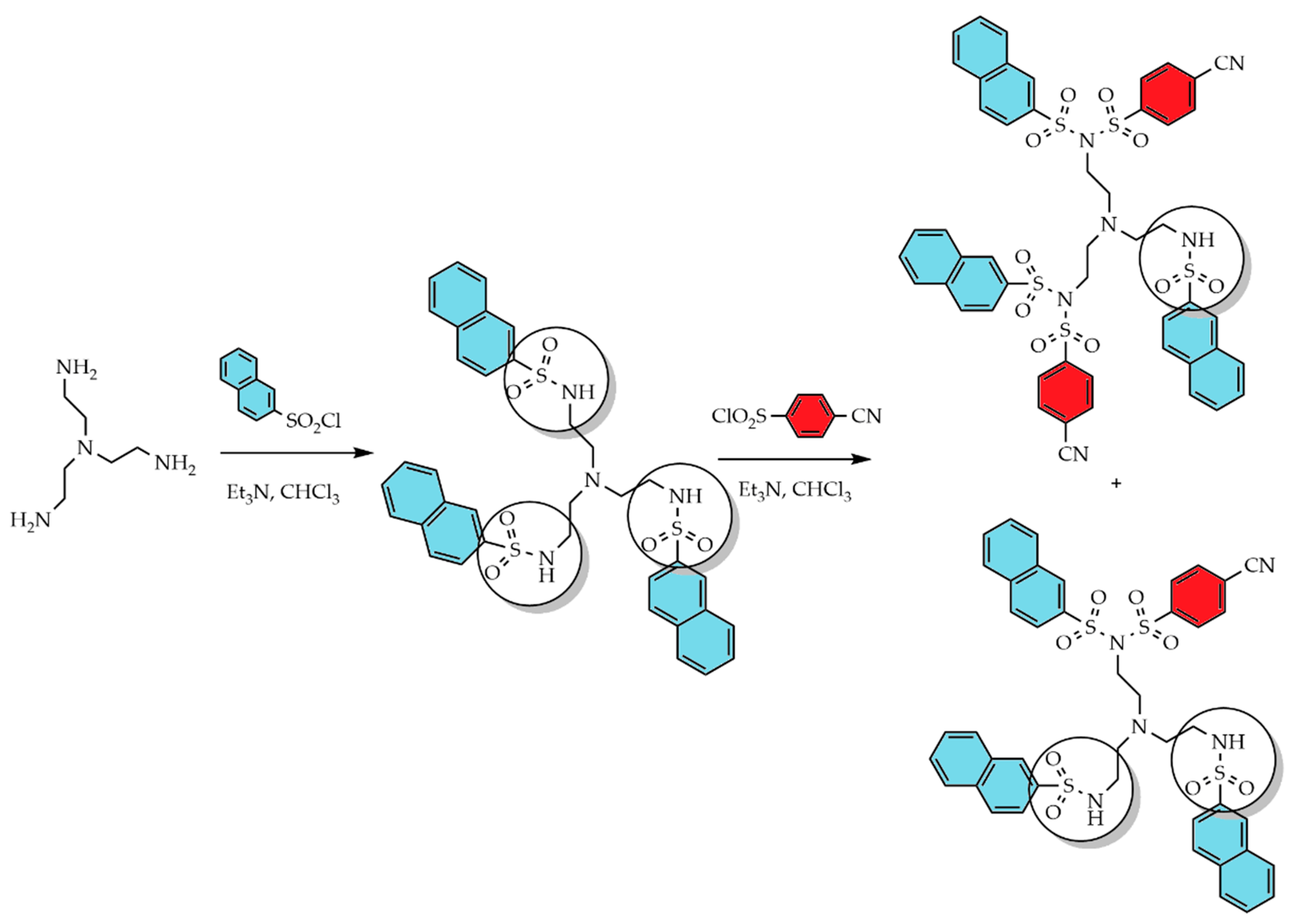
3.1. Divergent Technique
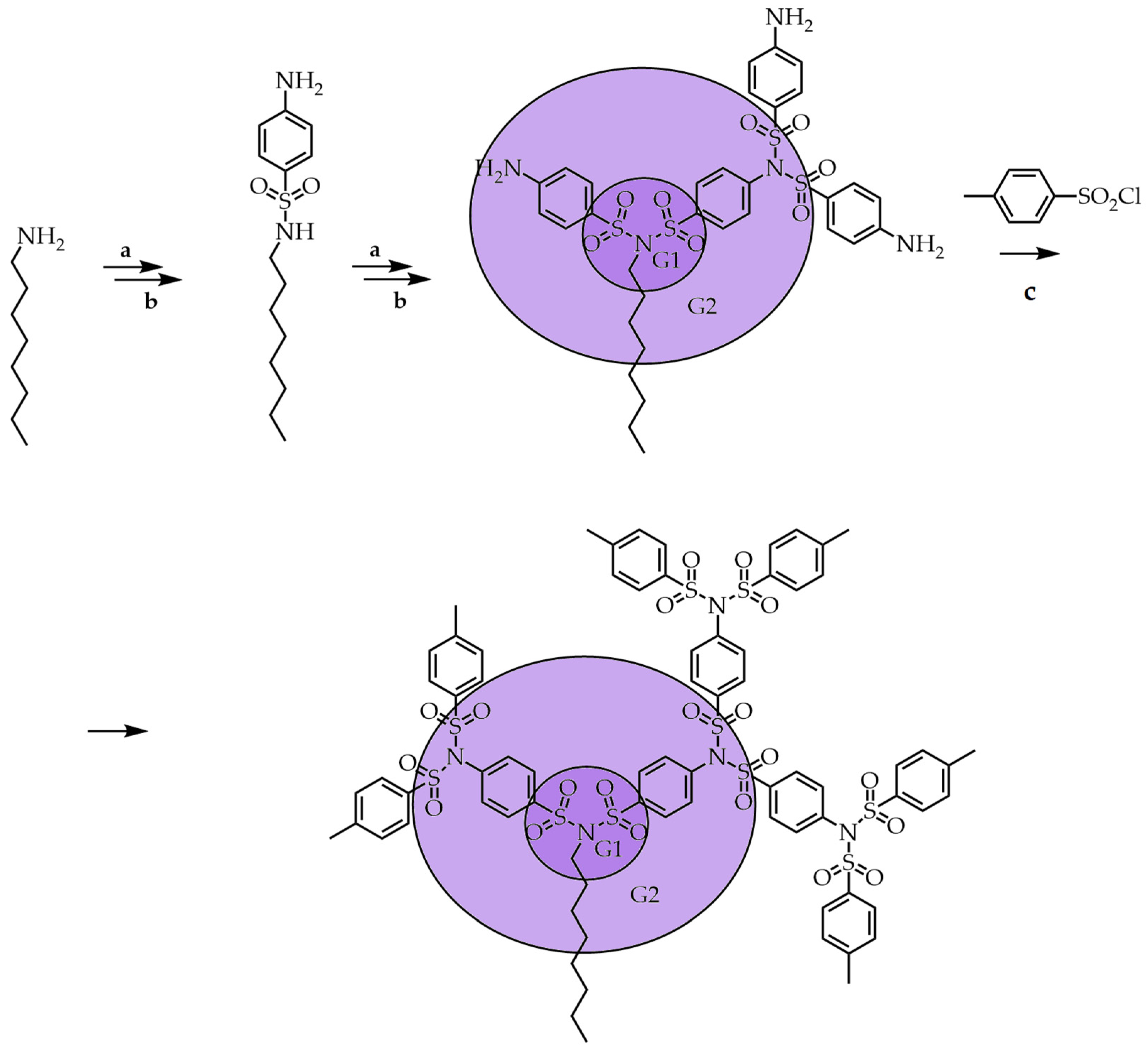
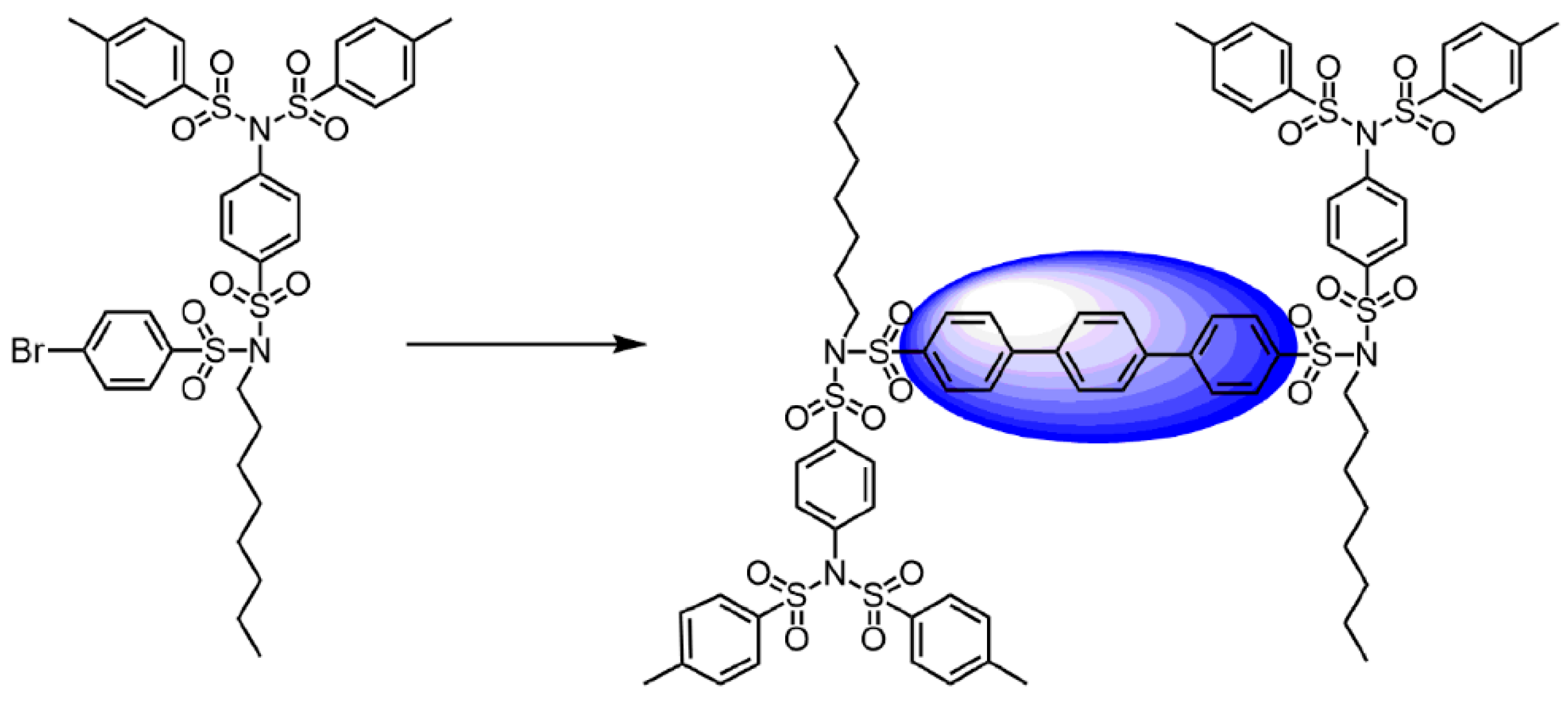
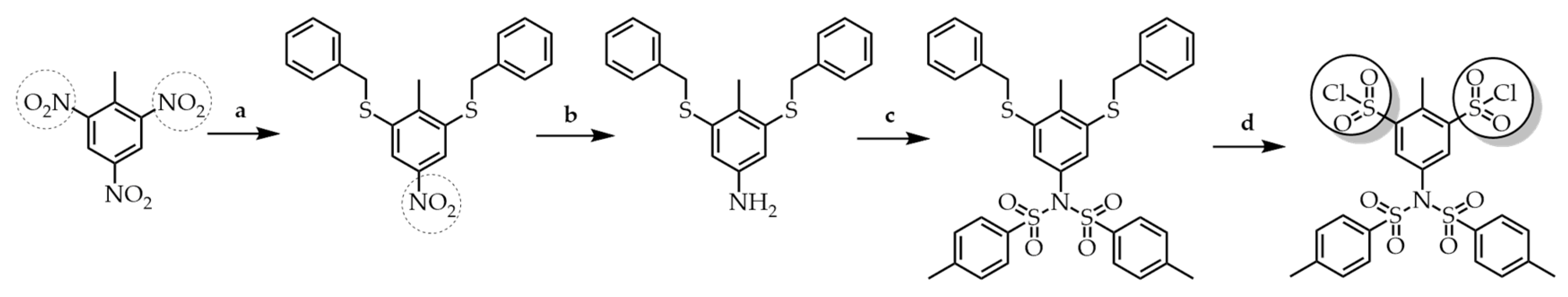
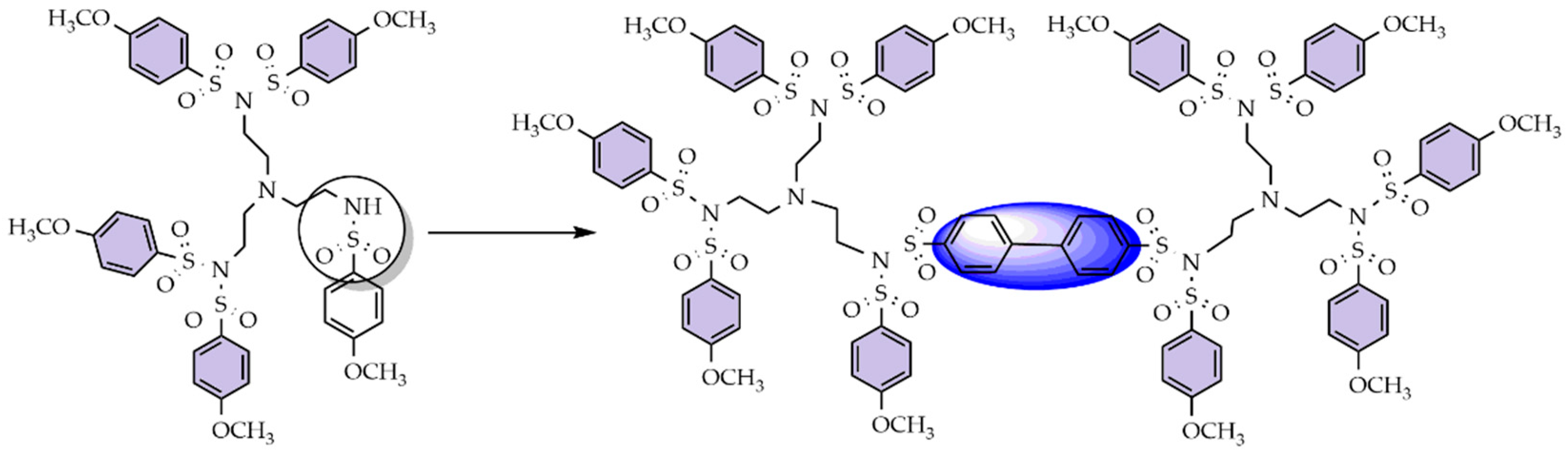
3.2. Convergent Technique
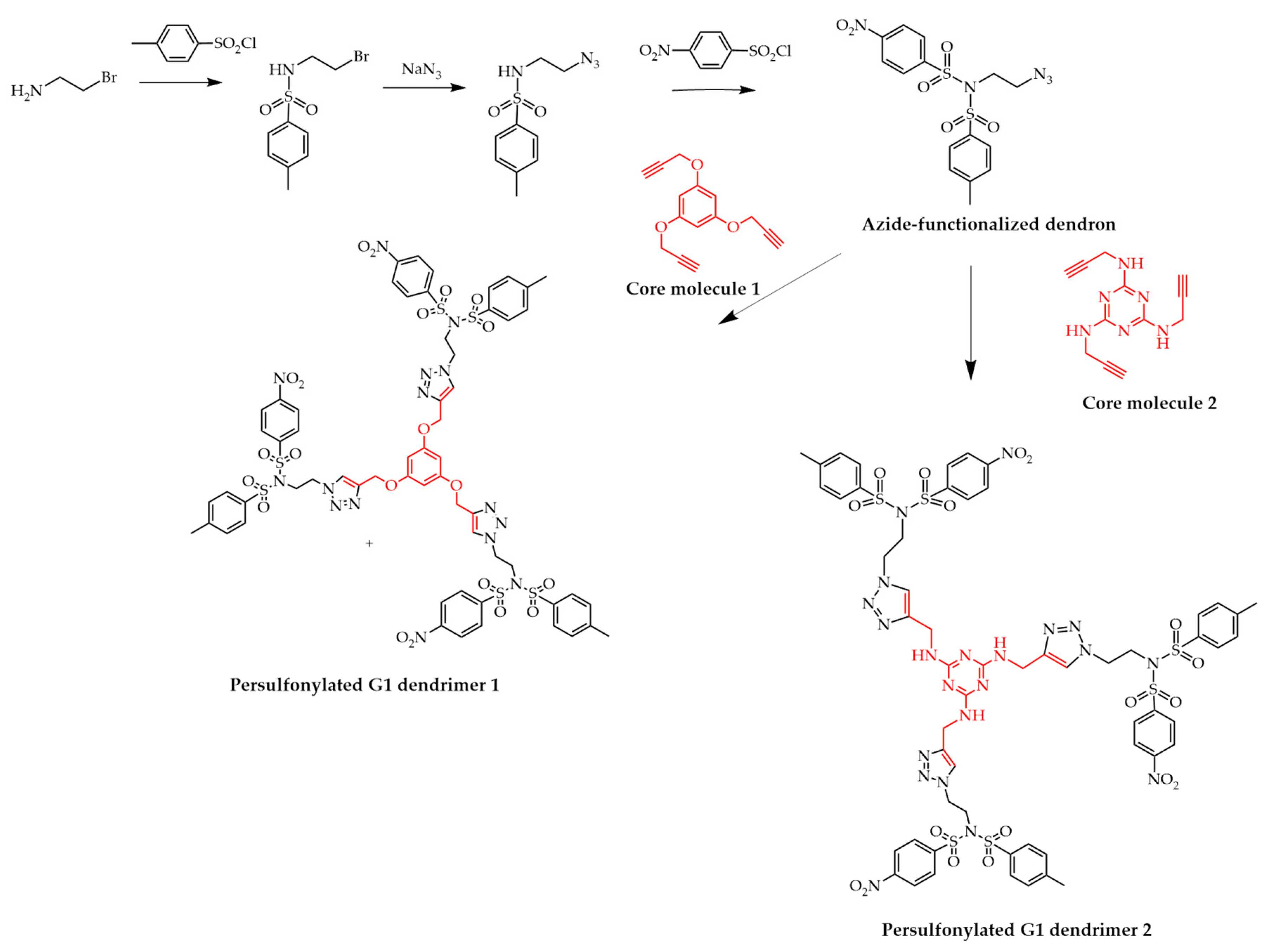
3.3. Click Chemistry
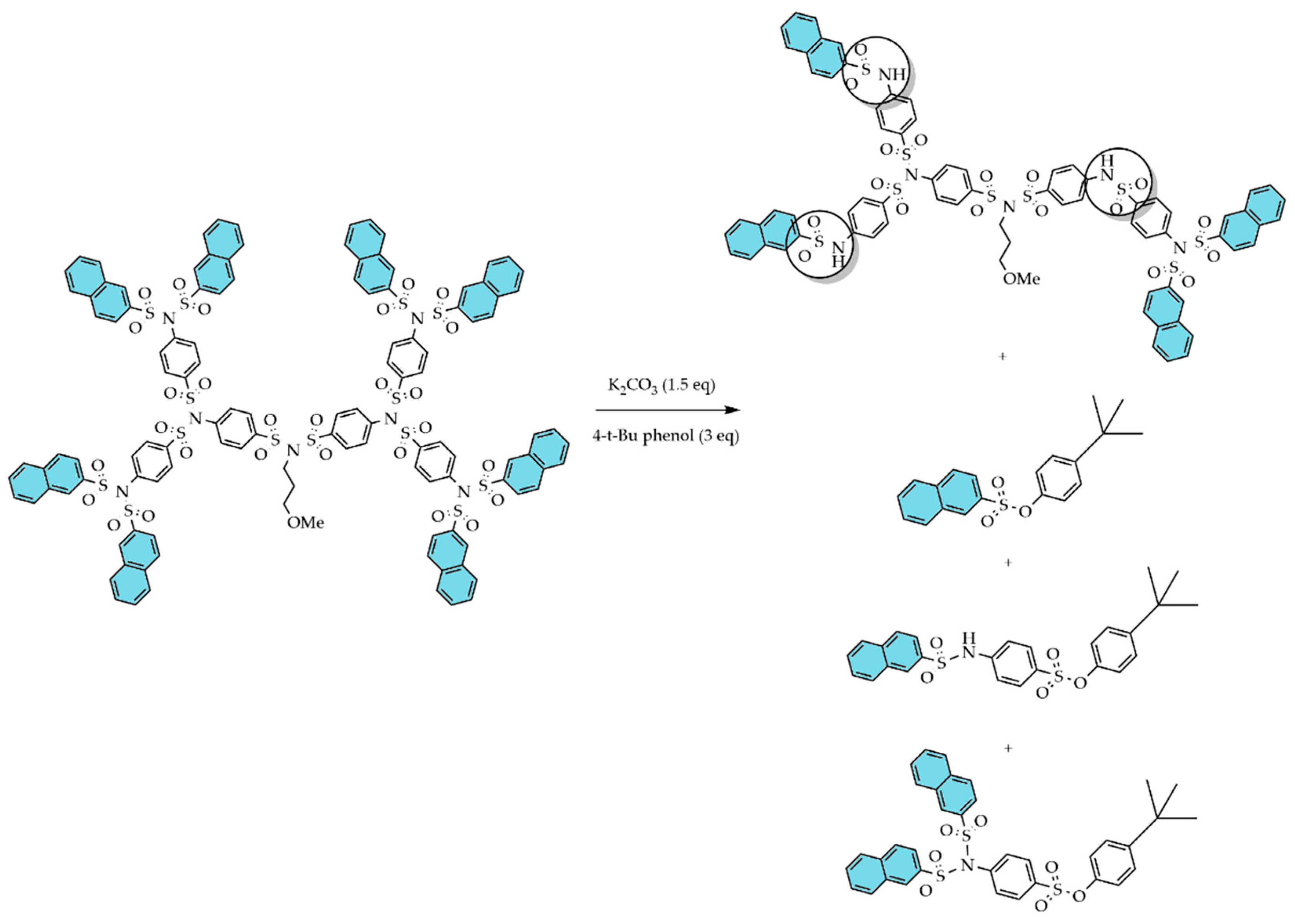
4. Purification and Separation
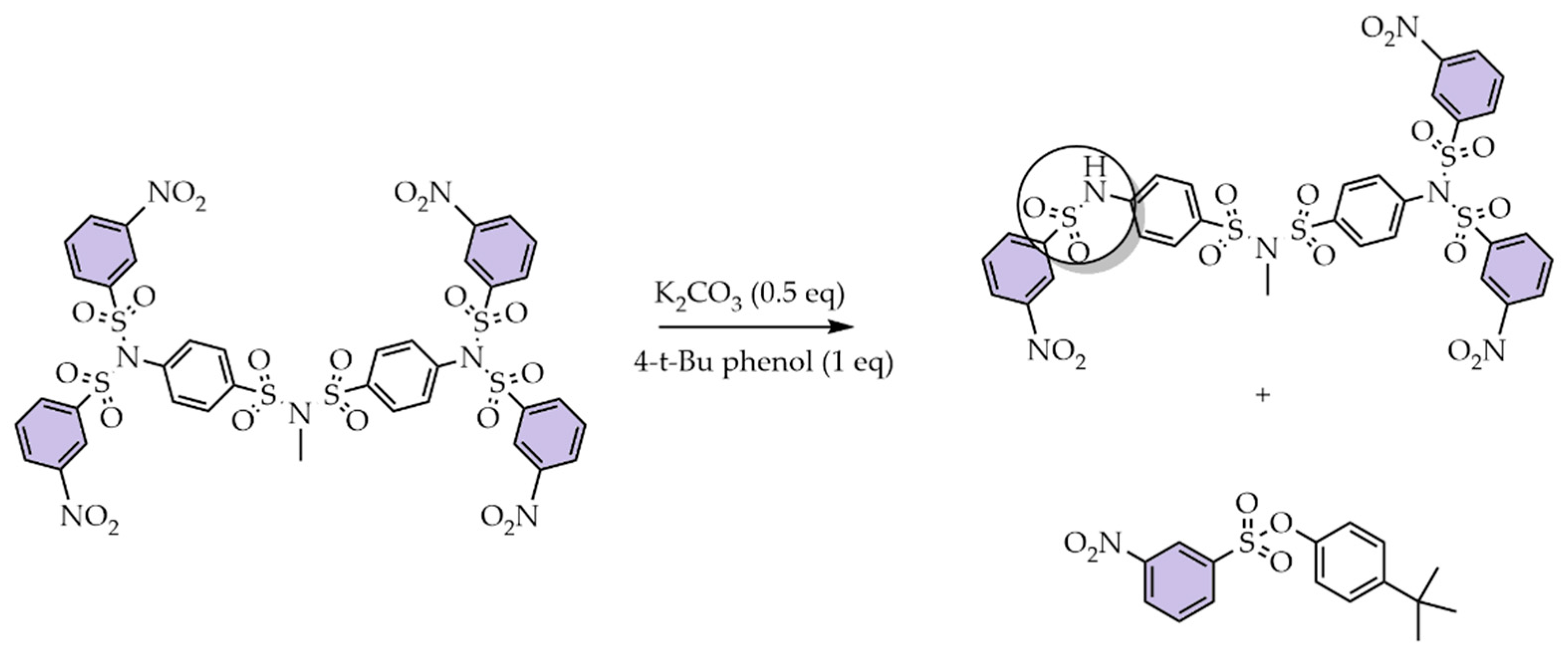
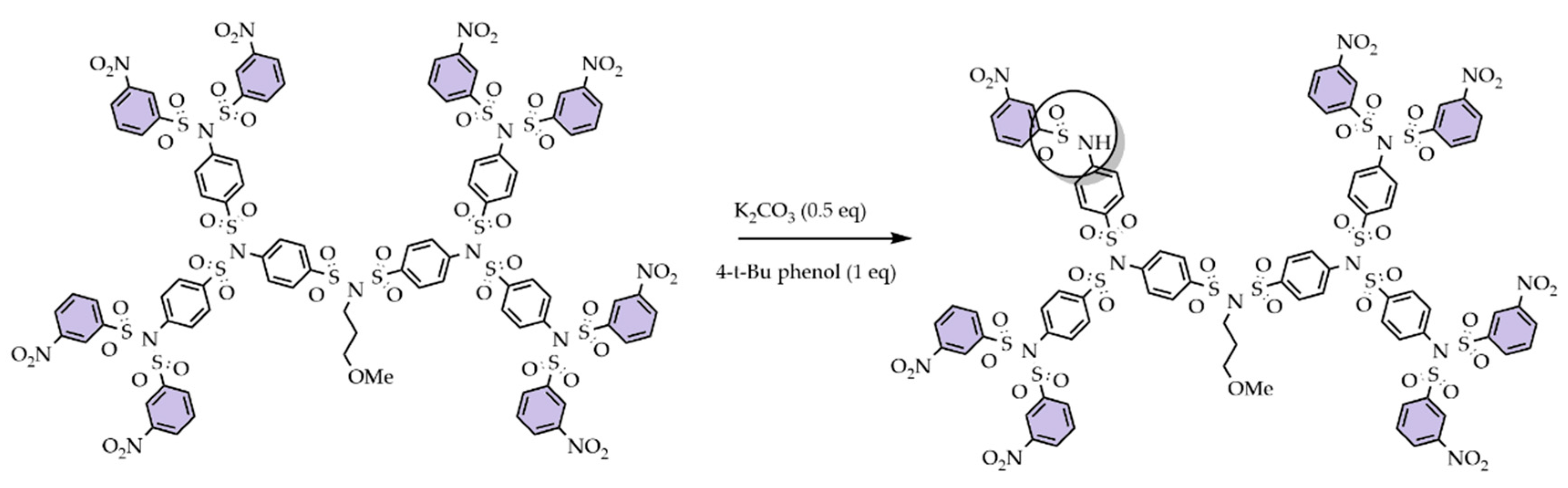
5. Activation of SBDs
6. Characterization
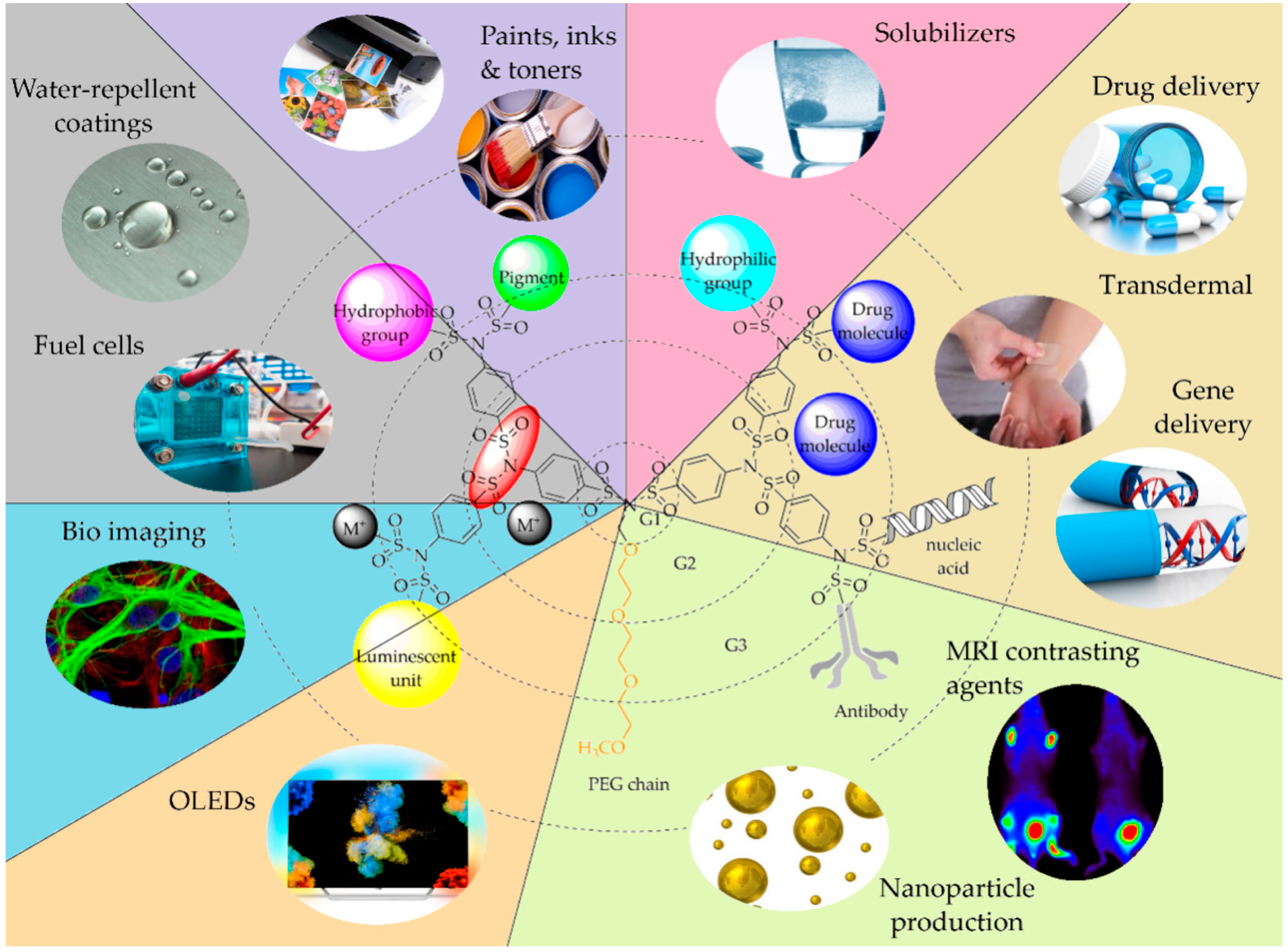
7. Application
7.1. SBDs in Electrochemistry
7.2. SBDs in Optics and Sensors
7.3. SBDs in Medicine
7.4. SBDs as Additives, Printing Inks, and Paints
7.5. SBDs as Coatings
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ratner, B.D.; Hoffman, A.S.; Schoen, F.J.; Lemons, J.E. Biomaterials Science; Elsevier: Amsterdam, The Netherlands, 2013; ISBN 9780123746269. [Google Scholar]
- Liu, X.; Gregurec, D.; Irigoyen, J.; Martinez, A.; Moya, S.; Ciganda, R.; Hermange, P.; Ruiz, J.; Astruc, D. Precise localization of metal nanoparticles in dendrimer nanosnakes or inner periphery and consequences in catalysis. Nat. Commun. 2016, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.V.; Malkoch, M. Simplifying the synthesis of dendrimers: Accelerated approaches. Chem. Soc. Rev. 2012, 41, 4593–4609. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, T.D.; Karellas, P.; Henderson, S.A.; Giannis, M.; O’Keefe, D.F.; Heery, G.; Paull, J.R.A.; Matthews, B.R.; Holan, G. Dendrimers as drugs: Discovery and preclinical and clinical development of dendrimer-based microbicides for HIV and STI prevention. Mol. Pharm. 2005, 2, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Madaan, K.; Kumar, S.; Poonia, N.; Lather, V.; Pandita, D. Dendrimers in drug delivery and targeting: Drug-dendrimer interactions and toxicity issues. J. Pharm. Bioallied Sci. 2014, 6, 139–150. [Google Scholar] [CrossRef]
- Lukin, O.; Gramlich, V.; Kandre, R.; Zhun, I.; Felder, T.; Schalley, C.A.; Dolgonos, G. Designer Dendrimers: Branched Oligosulfonimides with Controllable Molecular Architectures. J. Am. Chem. Soc. 2006, 128, 8964–8974. [Google Scholar] [CrossRef]
- Schubert, D.; Corda, M.; Lukin, O.; Brusilowskij, B.; Fiškin, E.; Schalley, C.A. A topological view of isomeric dendrimers. Eur. J. Org. Chem. 2008, 4148–4156. [Google Scholar] [CrossRef]
- Najafi, F.; Salami-Kalajahi, M.; Roghani-Mamaqani, H.; Kahaie-Khosrowshahi, A. A comparative study on solubility improvement of tetracycline and dexamethasone by poly(propylene imine) and polyamidoamine dendrimers: An insight into cytotoxicity and cell proliferation. J. Biomed. Mater. Res. Part A 2020, 108, 485–495. [Google Scholar] [CrossRef]
- Dvornic, P.R. PAMAMOS: The first commercial silicon-containing dendrimers and their applications. J. Polym. Sci. Part A Polym. Chem. 2006, 44, 2755–2773. [Google Scholar] [CrossRef]
- Najafi, F.; Salami-Kalajahi, M.; Roghani-Mamaqani, H.; Kahaie-Khosrowshahi, A. Effect of grafting ratio of poly(propylene imine) dendrimer onto gold nanoparticles on the properties of colloidal hybrids, their DOX loading and release behavior and cytotoxicity. Colloids Surf. B Biointerfaces 2019, 178, 500–507. [Google Scholar] [CrossRef]
- Choi, J.S.; Joo, D.K.; Kim, C.H.; Kim, K.; Park, J.S. Synthesis of a barbell-like triblock copolymer, poly(L-lysine) dendrimer-block-poly(ethylene glycol)-block-poly(L-lysine) dendrimer, and its self-assembly with plasmid DNA. J. Am. Chem. Soc. 2000, 122, 474–480. [Google Scholar] [CrossRef]
- Weng, Z.; Zaera, F. Synthesis of Chiral Dendrimer-Encapsulated Nanoparticle (DEN) Catalysts. Top. Catal. 2018, 61, 902–914. [Google Scholar] [CrossRef]
- Cao, Y.; Nguyen, G.K.T.; Chuah, S.; Tam, J.P.; Liu, C.F. Butelase-Mediated Ligation as an Efficient Bioconjugation Method for the Synthesis of Peptide Dendrimers. Bioconjugate Chem. 2016, 27, 2592–2596. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Kong, L.; Wang, L.; Fan, Y.; Shen, M.; Shi, X. Construction of core-shell tecto dendrimers based on supramolecular host-guest assembly for enhanced gene delivery. J. Mater. Chem. B 2017, 5, 8459–8466. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhang, H.; Liu, Q.; Sun, S.; Lei, P.; Zhao, Z.; Wu, L.; Wang, Y. The synthesis and biological evaluation of chondroitin sulfate e glycodendrimers. Future Med. Chem. 2019, 11, 1403–1415. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Takeda, K.; Yuba, E.; Harada, A.; Kono, K. Preparation of PEG-modified PAMAM dendrimers having a gold nanorod core and their application to photothermal therapy. J. Mater. Chem. B 2014, 2, 4167–4176. [Google Scholar] [CrossRef]
- Gupta, A.; Dubey, S.; Mishra, M.; Gupta, M.A. Unique Structures, Properties and Applications of Dendrimers. J. Drug Deliv. Ther. 2018, 8, 328–339. [Google Scholar] [CrossRef]
- Bondareva, J.; Luchkin, S.; Dagesyan, S.; Egorov, A.; Evlashin, S.; Lukin, O. Covalent and noncovalent films made up of sulfonimide-based dendrimers. Appl. Surf. Sci. 2020, 146345. [Google Scholar] [CrossRef]
- Rajabi, M.A.; Mousa, S. Lipid Nanoparticles and their Application in Nanomedicine. Curr. Pharm. Biotechnol. 2016, 17, 662–672. [Google Scholar] [CrossRef]
- Bondareva, J.; Rozhkov, V.; Kachala, V.V.; Fetyukhin, V.; Lukin, O. An optimized divergent synthesis of sulfonimide-based dendrimers achieving the fifth generation. Synth. Commun. 2019, 49, 3536–3545. [Google Scholar] [CrossRef]
- Chourasiya, S.S.; Patel, D.R.; Nagaraja, C.M.; Chakraborti, A.K.; Bharatam, P.V. Sulfonamide: Vs. sulfonimide: Tautomerism and electronic structure analysis of N-heterocyclic arenesulfonamides. New J. Chem. 2017, 41, 8118–8129. [Google Scholar] [CrossRef]
- Lill, S.O.N.; Broo, A. Molecule VI: Sulfonimide or sulfonamide? Cryst. Growth Des. 2014, 14, 3704–3710. [Google Scholar] [CrossRef]
- Rothermel, K.; Žabka, M.; Hioe, J.; Gschwind, R.M. Disulfonimides versus Phosphoric Acids in Brønsted Acid Catalysis: The Effect of Weak Hydrogen Bonds and Multiple Acceptors on Complex Structures and Reactivity. J. Org. Chem. 2019, 84, 13221–13231. [Google Scholar] [CrossRef] [PubMed]
- Hornback, J.M. Organic Chemistry; Thomson: Boston, MA, USA, 2005; ISBN 9780534389512. [Google Scholar]
- Craig, C.R.; Stitzel, R.E. Modern Pharmacology with Clinical Applications; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2004; ISBN 9780781737623. [Google Scholar]
- Boucher, B.A.; Hevener, K.E. Chapter 6: Medicinal Chemistry. In The APhA Complete Review for the Foreign Pharmacy Graduate Equivalency Examination, 2nd ed.; The American Pharmacists Association: Washington, DC, USA, 2018. [Google Scholar] [CrossRef]
- Harrold, M.W.; Zavod, R.M. Basic Concepts in Medicinal Chemistry. Drug Dev. Ind. Pharm. 2014, 40, 988. [Google Scholar] [CrossRef]
- Alfei, S.; Castellaro, S.; Taptue, G.B. Synthesis and NMR characterization of dendrimers based on 2, 2-bis-(hydroxymethyl)-propanoic acid (bis-HMPA) containing peripheral amino acid residues for gene transfection. Org. Commun. 2017, 10, 144–177. [Google Scholar] [CrossRef]
- Alfei, S.; Castellaro, S. Synthesis and characterization of polyester-based dendrimers containing peripheral arginine or mixed amino acids as potential vectors for gene and drug delivery. Macromol. Res. 2017, 25, 1172–1186. [Google Scholar] [CrossRef]
- Alfei, S.; Catena, S. Synthesis and characterization of fourth generation polyester-based dendrimers with cationic amino acids-modified crown as promising water soluble biomedical devices. Polym. Adv. Technol. 2018, 29, 2735–2749. [Google Scholar] [CrossRef]
- Schito, A.M.; Alfei, S. Antibacterial Activity of Non-Cytotoxic, Amino Acid-Modified Polycationic Dendrimers against Pseudomonas aeruginosa and Other Non-Fermenting Gram-Negative Bacteria. Polymers 2020, 12, 1818. [Google Scholar] [CrossRef]
- Pina, F.; Passaniti, P.; Maestri, M.; Balzani, V.; Vögtle, F.; Gorka, M.; Lee, S.K.; Van Heyst, J.; Fakhrnabavi, H. Ground and excited-state electronic interactions in poly(propylene amine) dendrimers functionalized with naphthyl units: Effect of protonation and metal complexation. ChemPhysChem 2004, 5, 473–480. [Google Scholar] [CrossRef]
- Mallakpour, S.; Zandi, H. Synthesis and Characterization of Novel Photoactive Polyamides Containing Naphthalene Moieties. Iran. Polym. J. 2006, 15, 619–927. [Google Scholar]
- Saudan, C.; Balzani, V.; Ceroni, P.; Gorka, M.; Maestri, M.; Vicinelli, V.; Vögtle, F. Dendrimers with a cyclam core. Absorption spectra, multiple luminescence, and effect of protonation. Tetrahedron 2003, 59, 3845–3852. [Google Scholar] [CrossRef]
- Wegner, V.G. Topochemische reaktionen von monomeren mit konjugierten dreifachbindungen. II. Mitt.: Eine topochemische farb- und vernetzungsreaktion bei polymeren mit konjugierten acetylengruppen im grundbaustein. Die Makromol. Chem. 1970, 134, 219–229. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Schweizer, B.W.; Lukin, O.; Zhang, A.; Schlüter, A.D. Dendronized Polymers with Aromatic Sulfonimide Dendrons. Macromol. Chem. Phys. 2010, 211, 1538–1549. [Google Scholar] [CrossRef]
- Jansook, P.; Kurkov, S.V.; Loftsson, T. Cyclodextrins as solubilizers: Formation of complex aggregates. J. Pharm. Sci. 2010, 99, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Kesharwani, P.; Jain, K.; Jain, N.K. Dendrimer as nanocarrier for drug delivery. Prog. Polym. Sci. 2014, 39, 268–307. [Google Scholar] [CrossRef]
- Buhleier, E.; Wehner, W.; Vogtle, F. Cascade-chain-like and nonskid-chain-like syntheses of molecular cavity topologies. Synthesis 1978, 2, 155–158. [Google Scholar] [CrossRef]
- Vögtle, F.; Richardt, G.; Werner, N. Dendrimer Chemistry: Concepts, Syntheses, Properties, Applications; John Wiley and Sons: Hoboken, NJ, USA, 2009; ISBN 9783527320660. [Google Scholar]
- Vögtle, F.; Fakhrnabavi, H.; Lukin, O. Controllable, selective per-functionalization of dendritic oligoamines. Org. Lett. 2004, 6, 1075–1078. [Google Scholar] [CrossRef]
- Newkome, G.R.; George, R.; Moorefield, C.N.; Charles, N.; Vögtle, F. Dendrimers and Dendrons: Concepts, Syntheses, Applications; Wiley-VCH: Weinheim, Germany, 2001; ISBN 9783527299973. [Google Scholar]
- de Brabander-van den Berg, E.M.M.; Meijer, E.W. Poly(propylene imine) Dendrimers: Large-Scale Synthesis by Hetereogeneously Catalyzed Hydrogenations. Angew. Chem. Int. Ed. Engl. 1993, 32, 1308–1311. [Google Scholar] [CrossRef] [Green Version]
- Tomalia, D.A.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. Dendritic macromolecules: Synthesis of starburst dendrimers. Macromolecules 1986, 19, 2466–2468. [Google Scholar] [CrossRef]
- Vögtle, F.; Fakhrnabavi, H.; Lukin, O.; Müller, S.; Friedhofen, J.; Schalley, C.A. Towards a selective functionalization of ammo-terminated dendrimers. Eur. J. Org. Chem. 2004, 4717–4724. [Google Scholar] [CrossRef]
- De Christopher, P.J.; Adamek, J.P.; Lyon, G.D.; Klein, S.A.; Baumgarten, R.J. Simple Deaminations. V. Preparation and Some Properties of N-Alkyl-N,N-disulfonimides. J. Org. Chem. 1974, 39, 3525–3532. [Google Scholar] [CrossRef]
- Caminade, A.M.; Laurent, R.; Delavaux-Nicot, B.; Majoral, J.P. “Janus” dendrimers: Syntheses and properties. New J. Chem. 2012, 36, 217–226. [Google Scholar] [CrossRef]
- Lukin, O.; Schubert, D.; Müller, C.; Corda, M.; Kandre, R. Persulfonylation of amines applied to the synthesis of higher generation dendrimers. J. Org. Chem. 2008, 73, 3562–3565. [Google Scholar] [CrossRef] [PubMed]
- Antoni, P.; Nyström, D.; Hawker, C.J.; Hult, A.; Malkoch, M. A chemoselective approach for the accelerated synthesis of well-defined dendritic architectures. Chem. Commun. 2007, 2249–2251. [Google Scholar] [CrossRef] [PubMed]
- Okaniwa, M.; Takeuchi, K.; Asai, M.; Ueda, M. One-pot synthesis of dendritic poly(amide-urea)s via Curtius rearrangement. 2. Synthesis and characterization of dendritic poly(amide-urea)s. Macromolecules 2002, 35, 6232–6238. [Google Scholar] [CrossRef]
- Ruiz, J.; Lafuente, G.; Marcen, S.; Ornelas, C.; Lazare, S.; Cloutet, E.; Blais, J.C.; Astruc, D. Construction of giant dendrimers using a tripodal building block. J. Am. Chem. Soc. 2003, 125, 7250–7257. [Google Scholar] [CrossRef] [PubMed]
- Bergamini, G.; Ceroni, P.; Balzani, V.; Kandre, R.; Lukin, O. Dendrimers with a pentaphenylene core: A photophysical study. ChemPhysChem 2009, 10, 265–269. [Google Scholar] [CrossRef]
- Rozhkov, V.V.; Kolotylo, M.V.; Onys’Ko, P.P.; Lukin, O. Synthesis of sulfonimide-based branched arylsulfonyl chlorides. Tetrahedron Lett. 2016, 57, 308–309. [Google Scholar] [CrossRef]
- Khanam, S.; Pandey, S.K.; Rai, S.K.; Verma, D.; Jasinski, J.P.; Pratap, S.; Tewari, A.K. Synthesis of N,N-Bis-Sulfonylated and N-Alkyl-N-Sulfonylated G1 Dendrimers via Click Reaction: Application of Thiocarbamide based Cu I Catalysts. ChemistrySelect 2017, 2, 6370–6374. [Google Scholar] [CrossRef]
- Khanam, S.; Rai, S.K.; Tewari, A.K. Advancement in the sulfone-based dendrimers: From synthesis to application Advancement in the sulfone-based dendrimers: From synthesis to application. Adv. Mater. Lett. 2017, 8, 1005–1019. [Google Scholar] [CrossRef]
- Binetti, R.; Costamagna, F.M.; Marcello, I. Exponential growth of new chemicals and evolution of information relevant to risk control. Ann. Ist. Super. Sanita. 2008, 44, 13–15. [Google Scholar] [PubMed]
- Mullen, D.G.; Desai, A.; Van Dongen, M.A.; Barash, M.; Baker, J.R.; Holl, M.M.B. Best practices for purification and characterization of PAMAM dendrimer. Macromolecules 2012, 45, 5316–5320. [Google Scholar] [CrossRef] [PubMed]
- Grayson, S.M.; Fréchet, J.M.J. Convergent Dendrons and Dendrimers: From Synthesis to Applications. Chem. Rev. 2001, 101, 3819–3868. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.X.; Redemann, C.T.; Szoka, F.C. In vitro gene delivery by degraded polyamidoamine dendrimers. Bioconjugate Chem. 1996, 7, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Caminade, A.M.; Turrin, C.O.; Laurent, R.; Ouali, A.; Beátrice, D.-N. Dendrimers: Towards Catalytic, Material and Biomedical Uses; Wiley: Hoboken, NJ, USA, 2012; Volume 51. [Google Scholar] [CrossRef]
- Zhou, L.; Russell, D.H.; Zhao, M.; Crooks, R.M. Characterization of Poly(amidoamine) Dendrimers and Their Complexes with Cu 2+ by Matrix-Assisted Laser Desorption Ionization Mass Spectrometry. ACS Publ. 2001, 34, 3567–3573. [Google Scholar] [CrossRef]
- Iavarone, A.T.; Williams, E.R. Mechanism of charging and supercharging molecules in electrospray ionization. J. Am. Chem. Soc. 2003, 125, 2319–2327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schalley, C.A.; Baytekin, B.; Baytekin, H.T.; Engeser, M.; Felder, T.; Rang, A. Mass spectrometry as a tool in dendrimer chemistry: From self-assembling dendrimers to dendrimer gas-phase host–guest chemistry. J. Phys. Org. Chem. 2006, 19, 479–490. [Google Scholar] [CrossRef]
- Weener, J.-W.; van Dongen, J.L.J.; Meijer, E.W. Electrospray Mass Spectrometry Studies of Poly(Propylene Imine) Dendrimers: Probing Reactivity in the Gas Phase. J. Am. Chem. Soc. 1999. [Google Scholar] [CrossRef]
- Schalley, C.A.; Verhaelen, C.; Klärner, F.G.; Hahn, U.; Vögtle, F. Gas-phase host-guest chemistry of dendritic viologens and molecular tweezers: A remarkably strong effect on dication stability. Angew. Chem. Int. Ed. 2005, 44, 477–480. [Google Scholar] [CrossRef]
- Williams, A.A.; Day, B.S.; Kite, B.L.; McPherson, M.K.; Slebodnick, C.; Morris, J.R.; Gandour, R.D. Homologous, long-chain alkyl dendrons form homologous thin films on silver oxide surfaces. Chem. Commun. 2005, 5053–5055. [Google Scholar] [CrossRef]
- Karakaya, B.; Claussen, W.; Gessler, K.; Saenger, W.; Schlüter, A.D. Toward dendrimers with cylindrical shape in solution. J. Am. Chem. Soc. 1997, 119, 3296–3301. [Google Scholar] [CrossRef]
- Polyakov, O.G.; Ivanova, S.M.; Gaudinski, C.M.; Miller, S.M.; Anderson, O.P.; Strauss, S.H. Cu(CO)2 (N(SO2 CF3)2). The First Structurally Characterized Copper(I) Polycarbonyl. Organometallics 1999, 18, 3769–3771. [Google Scholar] [CrossRef]
- De Ridder, D.J.A.; Goubitz, K.; Fontijn, M.; Čapková, P.; Dova, E.; Schenk, H. Influence of aromatic sulfonation on the geometry of [2.2]paracyclophane: Crystal structures of one sulfonate, one disulfonic anhydride and five disulfonimides. Acta Crystallogr. Sect. B Struct. Sci. 2001, 57, 780–790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reese, C.; Bao, Z. Organic single-crystal field-effect transistors. Mater. Today 2007, 10, 20–27. [Google Scholar] [CrossRef]
- Lukin, O.; Schubert, D.; Müller, C.M.; Schweizer, W.B.; Gramlich, V.; Schneider, J.; Dolgonos, G.; Shivanyuk, A. Engineering crystals of dendritic molecules. Proc. Natl. Acad. Sci. USA 2009, 106, 10922–10927. [Google Scholar] [CrossRef] [Green Version]
- Müller-Dethlefs, K.; Hobza, P. Noncovalent Interactions: A Challenge for Experiment and Theory. Chem. Rev. 2000, 100, 143–167. [Google Scholar] [CrossRef]
- Williams, J.H.; Cockcroft, J.K.; Fitch, A.N. Structure of the Lowest Temperature Phase of the Solid Benzene-Hexafluorobenzene Adduct. Angew. Chem. Int. Ed. Engl. 1992, 31, 1655–1657. [Google Scholar] [CrossRef]
- Amabilino, D.B.; Stoddart, J.F.; Williams, D.J. From Solid-State Structures and Superstructures to Self-Assembly Processes. Chem. Mater. 1994, 6, 1159–1167. [Google Scholar] [CrossRef]
- Aslanova, E.T.; Mamedalieva, F.M.; Aslanov, T.A.; Mamedov, B.A. Synthesis of Symmetrical Polyamide–Sulfonimides Based on Bis-saccharin Dicarboxylic Acid Dichlorides. Russ. J. Gen. Chem. 2019, 89, 96–99. [Google Scholar] [CrossRef]
- Ludwig, A. Discovery of new materials using combinatorial synthesis and high-throughput characterization of thin-film materials libraries combined with computational methods. Npj Comput. Mater. 2019, 5. [Google Scholar] [CrossRef]
- Martin, C.; Leurent, H. Technology and Innovation for the Future of Production: Accelerating Value Creation; World Economic Forum in collaboration with A.T. Kearney: Cologny, Switzerland, 2017. [Google Scholar]
- Astruc, D.; Boisselier, E.; Ornelas, C. Dendrimers designed for functions: From physical, photophysical, and supramolecular properties to applications in sensing, catalysis, molecular electronics, photonics, and nanomedicine. Chem. Rev. 2010, 110, 1857–1959. [Google Scholar] [CrossRef]
- Sowinska, M.; Urbanczyk-Lipkowska, Z. Advances in the chemistry of dendrimers. New J. Chem. 2014, 38, 2168–2203. [Google Scholar] [CrossRef]
- Jeon, I.Y.; Noh, H.J.; Baek, J.B. Hyperbranched macromolecules: From synthesis to applications. Molecules 2018, 23, 657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, Y.; Li, S.; Weng, Z.; Gao, C. Hyperbranched polymers: Advances from synthesis to applications. Chem. Soc. Rev. 2015, 44, 4091–4130. [Google Scholar] [CrossRef] [PubMed]
- Schlüter, A.D.; Halperin, A.; Kröger, M.; Vlassopoulos, D.; Wegner, G.; Zhang, B. Dendronized polymers: Molecular objects between conventional linear polymers and colloidal particles. ACS Macro Lett. 2014, 3, 991–998. [Google Scholar] [CrossRef]
- Vögtle, F.; Gorka, M.; Hesse, R.; Ceroni, P.; Maestri, M.; Balzani, V. Photochemical and photophysical properties of poly(propylene amine) dendrimers with peripheral naphthalene and azobenzene groups. Photochem. Photobiol. Sci. 2002, 1, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Katzur, V.; Eichler, M.; Deigele, E.; Stage, C.; Karageorgiev, P.; Geis-Gerstorfer, J.; Schmalz, G.; Ruhl, S.; Rupp, F.; Müller, R. Surface-immobilized PAMAM-dendrimers modified with cationic or anionic terminal functions: Physicochemical surface properties and conformational changes after application of liquid interface stress. J. Colloid Interface Sci. 2012, 366, 179–190. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, C.Y. Transient analysis of polymer electrolyte fuel cells. Electrochim. Acta 2005, 50, 1307–1315. [Google Scholar] [CrossRef]
- Sahu, A.K.; Pitchumani, S.; Sridhar, P.; Shukla, A.K. Nafion and modified-Nafion membranes for polymer electrolyte fuel cells: An overview. Bull. Mater. Sci. 2009, 32, 285–294. [Google Scholar] [CrossRef]
- Allcock, H.R.; Wood, R.M. Design and synthesis of ion-conductive polyphosphazenes for fuel cell applications: Review. J. Polym. Sci. Part B Polym. Phys. 2006, 44, 2358–2368. [Google Scholar] [CrossRef]
- Sutradhar, S.C.; Ahmed, F.; Ryu, T.; Yoon, S.; Lee, S.; Rahman, M.M.; Kim, J.; Lee, Y.; Kim, W.; Jin, Y. A novel synthesis approach to partially fluorinated sulfonimide based poly (arylene ether sulfone) s for proton exchange membrane. Int. J. Hydrog. Energy 2019, 44, 11321–11331. [Google Scholar] [CrossRef]
- Cheng, X.B.; Zhang, R.; Zhao, C.Z.; Wei, F.; Zhang, J.G.; Zhang, Q. A review of solid electrolyte interphases on lithium metal anode. Adv. Sci. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Xiao, S.; Shi, Y.; Yang, Y.; Hou, Y.; Wu, Y. A Composite Gel Polymer Electrolyte with High Performance Based on Poly(Vinylidene Fluoride) and Polyborate for Lithium Ion Batteries. Adv. Energy Mater. 2014, 4, 1300647. [Google Scholar] [CrossRef]
- Hallinan, D.T.; Balsara, N.P. Polymer Electrolytes. Annu. Rev. Mater. Res. 2013, 43, 503–525. [Google Scholar] [CrossRef]
- Rohan, R.; Sun, Y.; Cai, W.; Pareek, K.; Zhang, Y.; Xu, G.; Cheng, H. Functionalized meso/macro-porous single ion polymeric electrolyte for applications in lithium ion batteries. J. Mater. Chem. A 2014, 2, 2960–2967. [Google Scholar] [CrossRef]
- Chen, Y.; Ke, H.; Zeng, D.; Zhang, Y.; Sun, Y.; Cheng, H. Superior polymer backbone with poly(arylene ether) over polyamide for single ion conducting polymer electrolytes. J. Membr. Sci. 2017, 525, 349–358. [Google Scholar] [CrossRef]
- Cao, C.; Li, Y.; Feng, Y.; Long, P.; An, H.; Qin, C.; Han, J.; Li, S.; Feng, W. A sulfonimide-based alternating copolymer as a single-ion polymer electrolyte for high-performance lithium-ion batteries. J. Mater. Chem. A 2017, 5, 22519–22526. [Google Scholar] [CrossRef]
- Puntoriero, F.; Serroni, S.; Galletta, M.; Juris, A.; Licciardello, A.; Chiorboli, C.; Campagna, S.; Scandola, F. A New Heptanuclear Dendritic Ruthenium(II) Complex Featuring Photoinduced Energy Transfer Across High-Energy Subunits. ChemPhysChem 2005, 6, 129–138. [Google Scholar] [CrossRef]
- Xu, M.H.; Lin, J.; Hu, Q.S.; Pu, L. Fluorescent sensors for the enantioselective recognition of mandelic acid: Signal amplification by dendritic branching. J. Am. Chem. Soc. 2002, 124, 14239–14246. [Google Scholar] [CrossRef]
- Lor, M.; Thielemans, J.; Viaene, L.; Cotlet, M.; Hofkens, J.; Weil, T.; Hampel, C.; Müllen, K.; Verhoeven, J.W.; Van der Auweraer, M.; et al. Photoinduced electron transfer in a rigid first generation triphenylamine core dendrimer substituted with a peryleneimide acceptor. J. Am. Chem. Soc. 2002, 124, 9918–9925. [Google Scholar] [CrossRef]
- Montalti, M.; Credi, A.; Prodi, L.; Gandolfi, M.T. Handbook of Photochemistry Third Edition; CRC Press: Boca Raton, FL, USA, 2006; ISBN 9780824723774. [Google Scholar]
- Bergamini, G.; Ceroni, P.; Balzani, V.; Villavieja, M.D.M.; Kandre, R.; Zhun, I.; Lukin, O. A photophysical study of terphenyl core oligosulfonimide dendrimers exhibiting high steady-state anisotropy. Chem. Phys. Chem. 2006, 7, 1980–1984. [Google Scholar] [CrossRef]
- US5098475A—Inks with Dendrimer Colorants—Google Patents. Available online: https://patents.google.com/patent/US5098475A/en (accessed on 24 March 1992).
- Mackay, M.E.; Hong, Y.; Jeong, M.; Hong, S.; Russell, T.P.; Hawker, C.J.; Vestberg, R.; Douglas, J.F. Influence of dendrimer additives on the dewetting of thin polystyrene films. Langmuir 2002, 18, 1877–1882. [Google Scholar] [CrossRef]
- Patel, H.N.; Patel, D.P.M. Dendrimer Applications—A Review. Int. J. Pharm. Bio. Sci. 2013, 4, 454–463. [Google Scholar]
- US8236385B2—Treatment of Substrates for Improving Ink Adhesion to the Substrates—Google Patents. Available online: https://patents.google.com/patent/US8236385B2/en (accessed on 7 August 2012).
| Generations | Molecular Weight, Da | Dimensions, nm | 2D Graphical Representation | 3D Chemical Structure View 1 |
|---|---|---|---|---|
| G2 | 1334 | 2.6 |  | 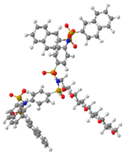 |
| G3 | 2541 | 3.2 | 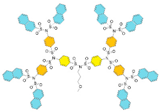 | 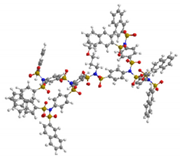 |
| G4 | 5304 | 3.6 | 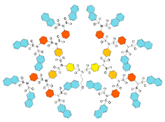 | 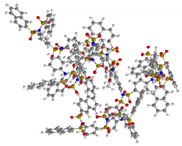 |
| G5 | 10831 | 4.6 | 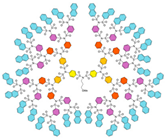 | 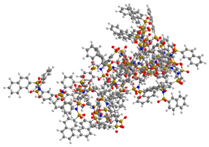 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bondareva, J.V.; Evlashin, S.A.; Lukin, O.V. Sulfonimide-Based Dendrimers: Progress in Synthesis, Characterization, and Potential Applications. Polymers 2020, 12, 2987. https://doi.org/10.3390/polym12122987
Bondareva JV, Evlashin SA, Lukin OV. Sulfonimide-Based Dendrimers: Progress in Synthesis, Characterization, and Potential Applications. Polymers. 2020; 12(12):2987. https://doi.org/10.3390/polym12122987
Chicago/Turabian StyleBondareva, Julia V., Stanislav A. Evlashin, and Oleg V. Lukin. 2020. "Sulfonimide-Based Dendrimers: Progress in Synthesis, Characterization, and Potential Applications" Polymers 12, no. 12: 2987. https://doi.org/10.3390/polym12122987






