Porous Film Coating Enabled by Polyvinyl Pyrrolidone (PVP) for Enhanced Air Permeability of Fabrics: The Effect of PVP Molecule Weight and Dosage
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Preparation of Latex Film
2.3. Coating of Cotton Fabric
2.4. Characterization
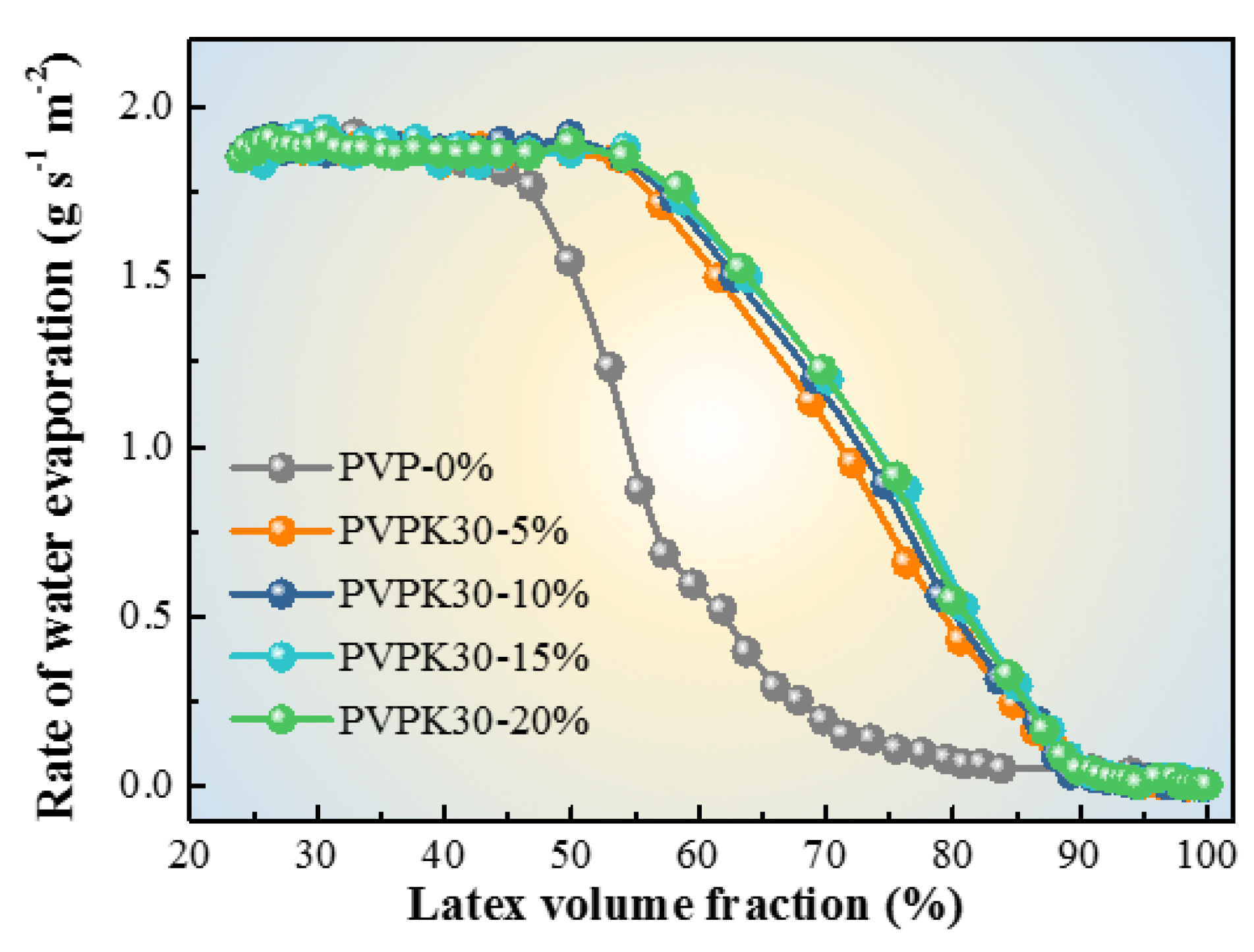
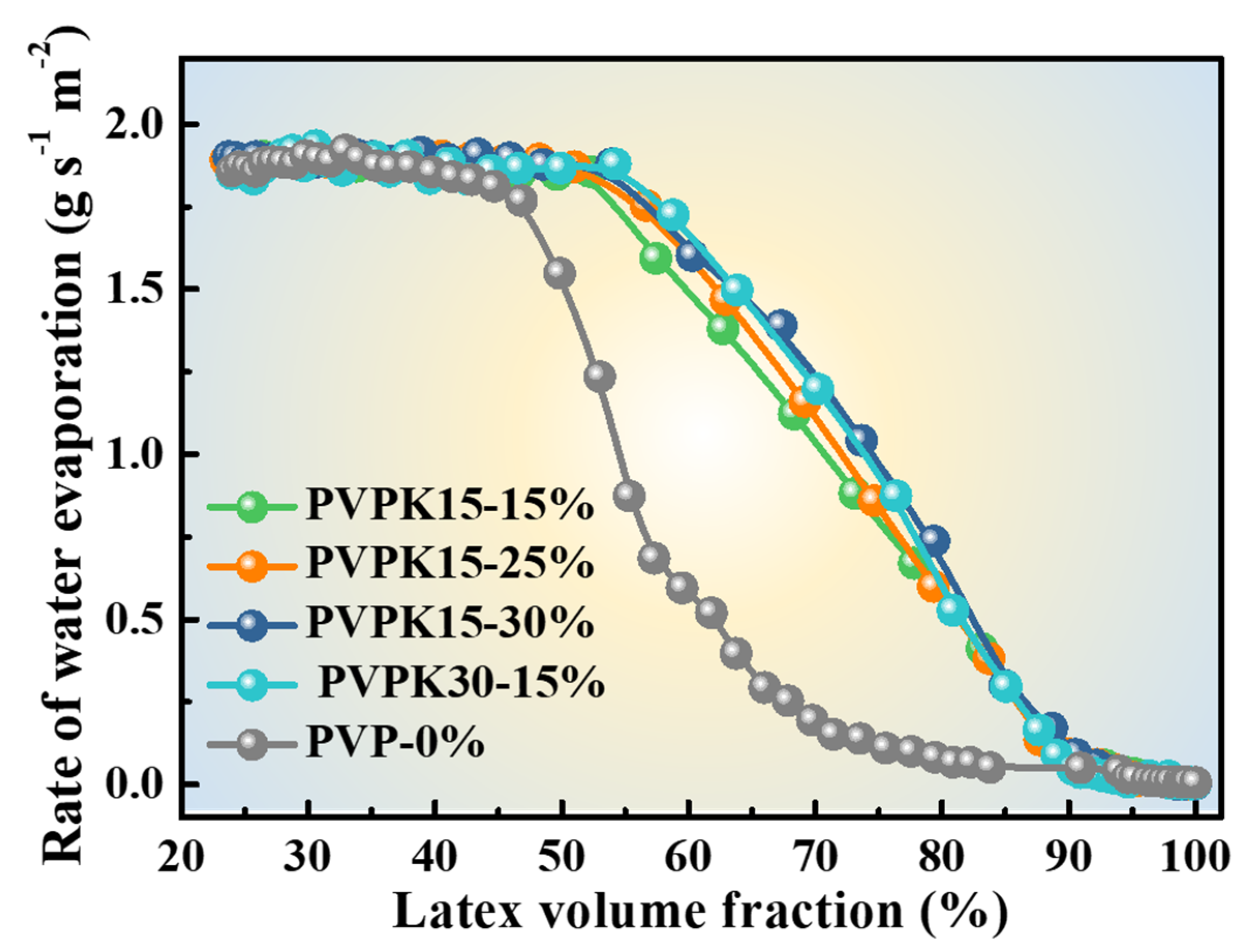
2.4.1. Gravimetric Analysis of Water Evaporation from Latex
2.4.2. Latex Film Morphologies
2.4.3. The Performance of Coated Fabric
3. Results and Discussion
3.1. Morphologies of Latex Films with PVP
3.2. Pore Architecture of Latex Films after Removal of PVP
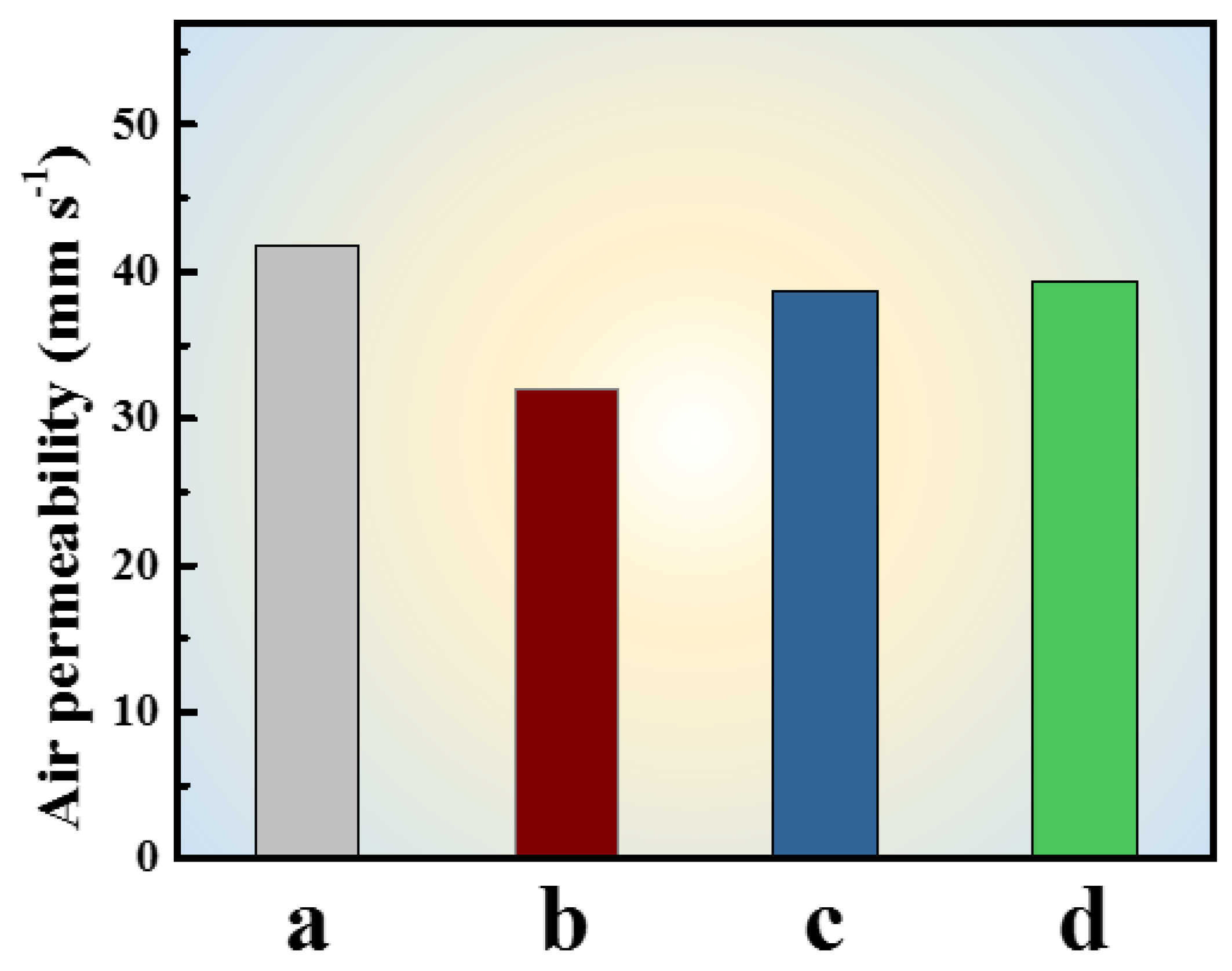
3.3. The Performance of Coated Fabrics
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, Z.J.; Hu, X.D.; Wang, X.H.; Xiang, Z. The morphology of poly(terminal vinyl dimethicone-co-methyl methacrylate-co-n-butyl acrylate)/pigment composite film and its application in pigment printing of polyester fabric. RSC Adv. 2020, 10, 4949–4955. [Google Scholar] [CrossRef] [Green Version]
- Gao, D.; Liang, Z.; Lyu, B.; Feng, J.; Ma, J.; Wei, Q. “Soft” polymer latexes stabilized by a mixture of zinc oxide nanoparticles and polymerizable surfactants: Binders for pigment printing. Prog. Org. Coat. 2016, 101, 262–269. [Google Scholar] [CrossRef]
- Gonzalez-Alvarez, M.J.; Paternoga, J.; Breul, K.; Cho, H.J.; Roshandel, M.Z.; Soleimani, M.; Winnik, M.A. Understanding particle formation in surfactant-free waterborne coatings prepared by emulsification of pre-formed polymers. Polym. Chem. 2017, 8, 2931–2941. [Google Scholar] [CrossRef]
- Gonzalez-Martinez, J.F.; Falk, Y.Z.; Bjorklund, S.; Erkselius, S.; Rehnberg, N.; Sotres, J. Humidity-Induced Phase Transitions of Surfactants Embedded in Latex Coatings Can Drastically Alter Their Water Barrier and Mechanical Properties. Polymers 2018, 10, 284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gurnani, P.; Perrier, S. Controlled radical polymerization in dispersed systems for biological applications. Prog. Polym. Sci. 2020, 102, 101209. [Google Scholar] [CrossRef]
- Kwon, S.; Oh, K.; Shin, S.J.; Lee, H.L. Effects of hydroxyethyl methacrylate comonomer in styrene/acrylate latex on coating structure and printability. Prog. Org. Coat. 2020, 147, 105862. [Google Scholar] [CrossRef]
- Li, L.; Wang, R.; Lu, Q. Influence of polymer latex on the setting time, mechanical properties and durability of calcium sulfoaluminate cement mortar. Constr. Build. Mater. 2018, 169, 911–922. [Google Scholar] [CrossRef]
- Nakanishi, E.Y.; Cabral, M.R.; Gonçalves, P.d.S.; Santos, V.d.; Savastano Junior, H. Formaldehyde-free particleboards using natural latex as the polymeric binder. J. Clean. Prod. 2018, 195, 1259–1269. [Google Scholar] [CrossRef]
- Rodrigues, L.D.A.; Hurtado, C.R.; Macedo, E.F.; Tada, D.B.; Guerrini, L.M.; Oliveira, M.P. Colloidal properties and cytotoxicity of enzymatically hydrolyzed cationic starch-graft-poly(butyl acrylate-co-methyl methacrylate) latex by surfactant-free emulsion polymerization for paper coating application. Prog. Org. Coat. 2020, 145, 105693. [Google Scholar] [CrossRef]
- Abdelrahman, M.; Khattab, T. Development of One-Step Water-Repellent and Flame-Retardant Finishes for Cotton. ChemistrySelect 2019, 4, 3811–3816. [Google Scholar] [CrossRef]
- Abdelrahman, M.S.; Fouda, M.M.G.; Ajarem, J.S.; Maodaa, S.N.; Allam, A.A.; Khattab, T.A. Development of colorimetric cotton swab using molecular switching hydrazone probe in calcium alginate. J. Mol. Struct. 2020, 1216, 128301. [Google Scholar] [CrossRef]
- Camlibel, N.O.; Avinc, O.; Arik, B.; Yavas, A.; Yakin, I. The effects of huntite-hydromagnesite inclusion in acrylate-based polymer paste coating process on some textile functional performance properties of cotton fabric. Cellulose 2019, 26, 1367–1381. [Google Scholar] [CrossRef]
- Ibrahim, N.A.; Eid, B.M.; Khalil, H.M.; Almetwally, A.A. A new approach for durable multifunctional coating of PET fabric. Appl. Surf. Sci. 2018, 448, 95–103. [Google Scholar] [CrossRef]
- Markus, M.; Speck, T.; Speck, O.; Thomas, S.; Heinrich, P. Biomimetics and Technical Textiles: Solving Engineering Problems with the Help of Nature’s Wisdom. Am. J. Bot. 2006, 93, 1455–1465. [Google Scholar]
- Matsuo, T. Fibre materials for advanced technical textiles. Text. Prog. 2008, 40, 87–121. [Google Scholar] [CrossRef]
- Park, S.; Jayaraman, S. Smart Textiles: Wearable Electronic Systems. Mrs Bull. 2003, 28, 585–591. [Google Scholar] [CrossRef]
- Revaiah, R.G.; Kotresh, T.M.; Kandasubramanian, B. Technical textiles for military applications. J. Text. Inst. 2020. [Google Scholar] [CrossRef]
- Schwarz, A.; Langenhove, L.V.; Guermonprez, P.; Deguillemont, D. A roadmap on smart textiles. Text. Prog. 2010, 42, 99–180. [Google Scholar] [CrossRef]
- Willert, A.; Meuser, C.; Baumann, R.R. Printed batteries and conductive patterns in technical textiles. Jpn. J. Appl. Phys. 2018. [Google Scholar] [CrossRef] [Green Version]
- Wu, L.; Ge, Y.; Zhang, L.; Yu, D.; Wu, M.; Ni, H. Enhanced electrical conductivity and competent mechanical properties of polyaniline/polyacrylate (PANI/PA) composites for antistatic finishing prepared at the aid of polymeric stabilizer. Prog. Org. Coat. 2018, 125, 99–108. [Google Scholar] [CrossRef]
- Machotova, J.; Kalendova, A.; Zlamana, B.; Snuparek, J.; Palarcik, J.; Svoboda, R. Waterborne Coating Binders Based on Self-Crosslinking Acrylic Latex with Embedded Inorganic Nanoparticles: A Comparison of Nanostructured ZnO and MgO as Crosslink Density Enhancing Agents. Coatings 2020, 10, 339. [Google Scholar] [CrossRef] [Green Version]
- Kaur, J.; Krishnan, R.; Ramalingam, B.; Jana, S. Hydroxyethyl sulfone based reactive coalescing agents for low-VOC waterborne coatings. RSC Adv. 2020, 10, 17171–17179. [Google Scholar] [CrossRef]
- Liu, Q.; Liao, B.; Pang, H.; Lu, M.G.; Meng, Y.Y. Preparation and characterization of a self -matting coating based on waterborne polyurethane-polyacrylate hybrid dispersions. Prog. Org. Coat. 2020, 143, 105551. [Google Scholar] [CrossRef]
- Wang, Y.X.; Li, C.; Zhang, X.P.; Lin, Q.Q.; Jiang, Y.; Yuan, J.F.; Pan, M.W. Poly(vinylidene chloride)/Poly(chlorotrifluoroethylene-co-acrylates) Composite Latex Coating Cured at Room Temperature Showing an Excellent Corrosion Resistance. Chemistryselect 2020, 5, 6278–6284. [Google Scholar] [CrossRef]
- Lan, T.; An, R.; Liu, Z.; Li, K.; Xiang, J.; Liu, G. Facile fabrication of a biomass-based film with interwoven fibrous network structure as heterogeneous catalysis platform. J. Colloid Interface Sci. 2018, 532, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Costentin, C.; Savéant, J.-M. Molecular approach to catalysis of electrochemical reaction in porous films. Curr. Opin. Electrochem. 2019, 15, 58–65. [Google Scholar] [CrossRef]
- Wei, Y.; Li, J.; Li, Y.; Zhao, B.; Zhang, L.; Yang, X.; Chang, J. Research on permeability coefficient of a polyethylene controlled-release film coating for urea and relevant nutrient release pathways. Polym. Test. 2017, 59, 90–98. [Google Scholar] [CrossRef]
- Ghani, M. Nanocrystalline cellulose as a biotemplate for preparation of porous titania thin film as a sorbent for thin film microextraction of ketorolac, meloxicam, diclofenac and mefenamic acid. J. Chromatogr. B 2020, 1142, 122039. [Google Scholar] [CrossRef]
- Wei, D.; Zhou, R.; Cheng, S.; Feng, W.; Li, B.; Wang, Y.; Jia, D.; Zhou, Y.; Guo, H. Microarc oxidized TiO2 based ceramic coatings combined with cefazolin sodium/chitosan composited drug film on porous titanium for biomedical applications. Mater. Sci. Eng. C 2013, 33, 4118–4125. [Google Scholar] [CrossRef]
- Xiong, X.; Xie, F.; Meng, J. Preparation of superhydrophobic porous coating film with the matrix covered with polydimethylsiloxane for oil/water separation. Prog. Org. Coat. 2018, 125, 365–371. [Google Scholar] [CrossRef]
- Chen, Y.; Krings, S.; Booth, J.R.; Bon, S.A.F.; Hingley-Wilson, S.; Keddie, J.L. Introducing Porosity in Colloidal Biocoatings to Increase Bacterial Viability. Biomacromolecules 2020, 21, 4545–4558. [Google Scholar] [CrossRef]
- Ma, Y.; Davis, H.T.; Scriven, L.E. Microstructure development in drying latex coatings. Prog. Org. Coat. 2005, 52, 46–62. [Google Scholar] [CrossRef]
- Ludwig, I.; Schabel, W.; Kind, M.; Castaing, J.-C.; Ferlin, P. Drying and film formation of industrial waterborne latices. Aiche J. 2007, 53, 549–560. [Google Scholar] [CrossRef]
- Appel, L.E.; Zentner, G.M. Use of Modified Ethylcellulose Lattices for Microporous Coating of Osmotic Tablets. Pharm. Res. 1991, 8, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Hodges, I.C.; Hearn, J. Reactive Latex Films. Langmuir 2001, 17, 3419–3422. [Google Scholar] [CrossRef]
- Yang, S.; Aoki, Y.; Skeldon, P.; Thompson, G.E.; Habazaki, H. Growth of porous anodic alumina films in hot phosphate–glycerol electrolyte. J. Solid State Electrochem. 2011, 15, 689–696. [Google Scholar] [CrossRef] [Green Version]
- Ariyanti, S.; Man, Z.; Bustam, M.A. Improvement of Hydrophobicity of Urea Modified Tapioca Starch Film with Lignin for Slow Release Fertilizer. Adv. Mater. Res. 2013, 626, 350–354. [Google Scholar] [CrossRef]
- Steward, P.A.; Hearn, J.; Wilkinson, M.C. Studies on permeation through polymer latex films, I. Films containing no or only low levels of additives. Polym. Int. 1995, 38, 1–12. [Google Scholar] [CrossRef]
- Fogden, A. Porous polymer films cast from latex–glucose dispersions. Colloids Surf. A Physicochem. Eng. Asp. 2009, 349, 102–109. [Google Scholar] [CrossRef]
- Malik, T.; Razzaq, H.; Razzaque, S.; Nawaz, H.; Siddiqa, A.; Siddiq, M.; Qaisar, S. Design and synthesis of polymeric membranes using water-soluble pore formers: An overview. Polym. Bull. 2019, 76, 4879–4901. [Google Scholar] [CrossRef]
- Sabir, A.; Shafiq, M.; Islam, A.; Khan, S.; Jamil, T.; Zahid, M.; Shafeeq, A.; Shehzad, M.A.; Bhatti, A.; Habib, Y.; et al. Influence of polyethylene glycol 600 on cellulose acetate membranes for reverse osmosis desalination. Polym. Res. J. 2015, 9, 1935–2530. [Google Scholar]
- Calderon-Moreno, J.M.; Preda, S.; Predoana, L.; Zaharescu, M.; Anastasescu, M.; Nicolescu, M.; Stoica, M.; Stroescu, H.; Gartner, M.; Buiu, O.; et al. Effect of polyethylene glycol on porous transparent TiO2 films prepared by sol–gel method. Ceram. Int. 2014, 40, 2209–2220. [Google Scholar] [CrossRef]
- Joshi, M.; Adak, B.; Butola, B.S. Polyurethane nanocomposite based gas barrier films, membranes and coatings: A review on synthesis, characterization and potential applications. Prog. Mater. Sci. 2018, 97, 230–282. [Google Scholar] [CrossRef]
- Zhang, C.; Lyu, P.; Xia, L.; Wang, Y.; Li, C.; Xiang, X.; Dai, F.; Xu, W.; Liu, X.; Deng, B. Regulation of pore morphologies of PU films and thereof water vapor permeability by varying tetrahydrofuran concentration in binary solvent. Polym. Test. 2018, 69, 32–38. [Google Scholar] [CrossRef]
- Wang, P.; Ma, J.; Wang, Z.; Shi, F.; Liu, Q. Enhanced Separation Performance of PVDF/PVP-g-MMT Nanocomposite Ultrafiltration Membrane Based on the NVP-Grafted Polymerization Modification of Montmorillonite (MMT). Langmuir 2012, 28, 4776–4786. [Google Scholar] [CrossRef]
- Shen, Y.; Ye, J.; Yang, L.; Wu, M.; Ni, T. Effect of Polyvinylpyrrolidone on the Morphologies of Latex Films and Air Permeability of the Latex Finished Fabrics. J. Donghua Univ. (Engl. Ed.) 2010, 27, 339–344. [Google Scholar]
- Shen, Y.; Yang, L.; Ye, J.; Chen, L. Preparation of Latex Film with Macropores and Their Applications in Pigment Dyeing. J. Donghua Univ. (Engl. Ed.) 2010, 27, 846–851. [Google Scholar]
- Winnik, M.A.; Feng, J. Latex blends: An approach to zero VOC coatings. J. Coat. Technol. 1996, 68, 39–50. [Google Scholar]
- Lovell, P.A. Emulsion Polymerization and Emulsion Polymers; El-Aasser, M.S., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 1997. [Google Scholar]
- Winnik, M.A. Latex film formation. Curr. Opin. Colloid Interface Sci. 1997, 2, 192–199. [Google Scholar] [CrossRef]
- Vandezande, G.A.; Rudin, A. Film formation of vinyl acrylic latexes; effects of surfactant type, water and latex particle size. J. Coat. Technol. 1996, 68, 63–73. [Google Scholar]
- Vanderhoff, J.W.; Tarkowski, H.L.; Jenkins, M.C.; Bradford, E.B. Theoretical consideration of the interfacial forces involved in the coalescence of latex particles. J. Macromol. Chem. 1967, 40, 1246–1269. [Google Scholar] [CrossRef]
- Vanderhoff, J.W.; Bradford, E.B.; Carrington, W.K. The transport of water through latex films. J. Polym. Sci. 1973, 41, 155–174. [Google Scholar] [CrossRef]
- Sperry, P.R.; Snyder, B.S.; O’Dowd, M.L.; Lesko, P.M. Role of water in particle deformation and compaction in latex film formation. Langmuir 1994, 10, 2619–2628. [Google Scholar] [CrossRef]
- Fernando, I.; Qian, T.; Zhou, Y. Long term impact of surfactants & polymers on the colloidal stability, aggregation and dissolution of silver nanoparticles. Environ. Res. 2019, 179, 108781. [Google Scholar] [PubMed]
- Abu-Noqta, O.A.; Aziz, A.A.; Usman, A.I. Colloidal Stability of Iron Oxide Nanoparticles Coated with Different Capping Agents. Mater. Today Proc. 2019, 17, 1072–1077. [Google Scholar] [CrossRef]
- Kellaway, I.W.; Najib, N.M. The adsorption of hydrophilic polymers at the polystyrene-water interface. Int. J. Pharm. 1980, 6, 285–294. [Google Scholar] [CrossRef]



| Samples | Styrene-butyl Acrylate Copolymer Latex (g) | PVP (g) | Water (g) |
|---|---|---|---|
| PVP 0% | 4 | 0.00 | 0.8 |
| PVP 5% | 4 | 0.06 | 0.8 |
| PVP 10% | 4 | 0.12 | 0.8 |
| PVP 15% | 4 | 0.18 | 0.8 |
| PVP 20% | 4 | 0.24 | 0.8 |
| PVP 25% | 4 | 0.30 | 0.8 |
| PVP 30% | 4 | 0.36 | 0.8 |
| Samples | Thickener KF-386 (g) | Styrene-butyl Acrylate Copolymer Latex (g) | PVPK30 (g) | Water (g) |
|---|---|---|---|---|
| coated fabric with PVP 0% | 0.5 | 4 | 0.00 | 95.50 |
| coated fabric with PVPK30 10% | 0.5 | 4 | 0.12 | 95.38 |
| coated fabric with PVPK30 20% | 0.5 | 4 | 0.24 | 95.26 |
| Fabric | CIE L*, a*, b* | DE* | ||
|---|---|---|---|---|
| L* | a* | b* | ||
| original fabric | 35.11 | 57.47 | 8.21 | |
| coated fabric with PVP 0% | 35.31 | 57.21 | 8.07 | 0.35 |
| coated fabric with PVPK30 10% | 35.71 | 57.79 | 7.93 | 0.74 |
| coated fabric with PVPK30 20% | 35.76 | 57.67 | 7.77 | 0.82 |
| Fabric | Crocking Fastness/Grade | Laundering Fastness/Grade Color Change | |
|---|---|---|---|
| Dry | Wet | ||
| original fabric | 4.5 | 4 | 4 |
| coated fabric with PVP 0% | 4.5 | 4 | 4 |
| coated fabric with PVPK30 10% | 4.5 | 4 | 4 |
| coated fabric with PVPK30 20% | 4.5 | 4 | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, J.; Shen, Y.; Yu, D.; Yang, T.; Wu, M.; Yang, L.; Petru, M. Porous Film Coating Enabled by Polyvinyl Pyrrolidone (PVP) for Enhanced Air Permeability of Fabrics: The Effect of PVP Molecule Weight and Dosage. Polymers 2020, 12, 2961. https://doi.org/10.3390/polym12122961
Jiang J, Shen Y, Yu D, Yang T, Wu M, Yang L, Petru M. Porous Film Coating Enabled by Polyvinyl Pyrrolidone (PVP) for Enhanced Air Permeability of Fabrics: The Effect of PVP Molecule Weight and Dosage. Polymers. 2020; 12(12):2961. https://doi.org/10.3390/polym12122961
Chicago/Turabian StyleJiang, Jiantang, Yifeng Shen, Deyou Yu, Tao Yang, Minghua Wu, Lei Yang, and Michal Petru. 2020. "Porous Film Coating Enabled by Polyvinyl Pyrrolidone (PVP) for Enhanced Air Permeability of Fabrics: The Effect of PVP Molecule Weight and Dosage" Polymers 12, no. 12: 2961. https://doi.org/10.3390/polym12122961
APA StyleJiang, J., Shen, Y., Yu, D., Yang, T., Wu, M., Yang, L., & Petru, M. (2020). Porous Film Coating Enabled by Polyvinyl Pyrrolidone (PVP) for Enhanced Air Permeability of Fabrics: The Effect of PVP Molecule Weight and Dosage. Polymers, 12(12), 2961. https://doi.org/10.3390/polym12122961









