Manufacturing and Characterization of Coatings from Polyamide Powders Functionalized with Nanosilica
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Manufacturing of PA11 Coatings
2.2. Characterization and Testing
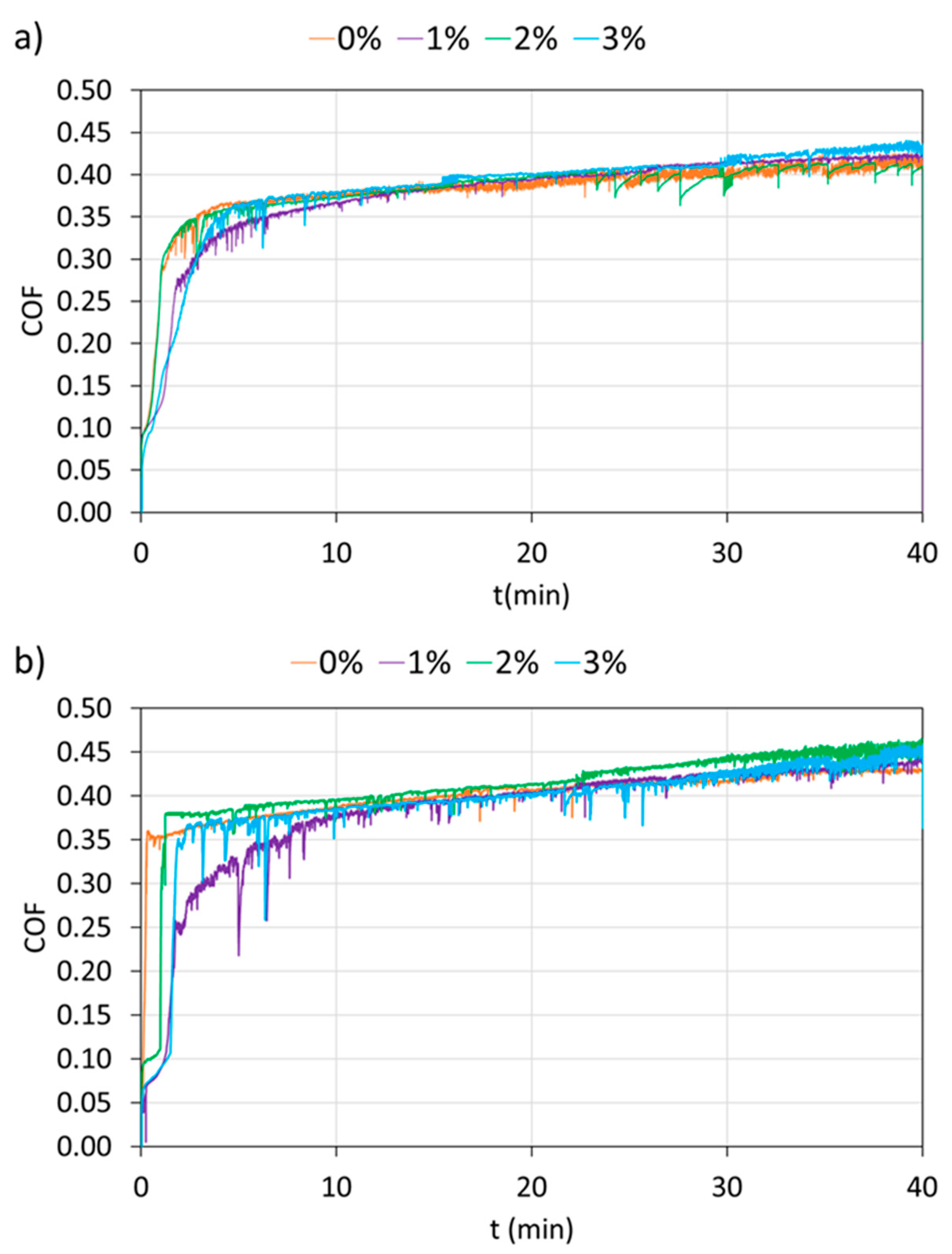
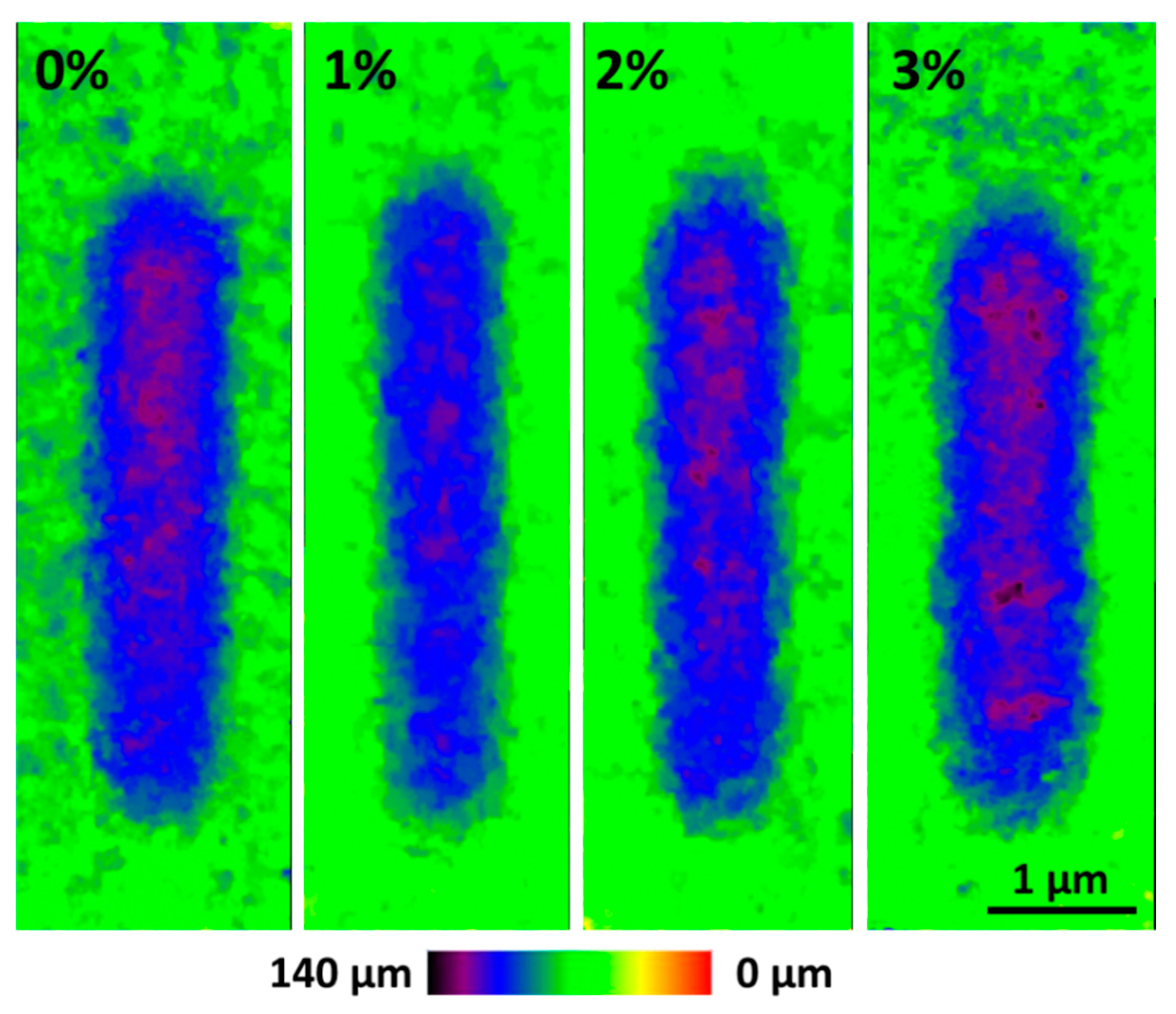
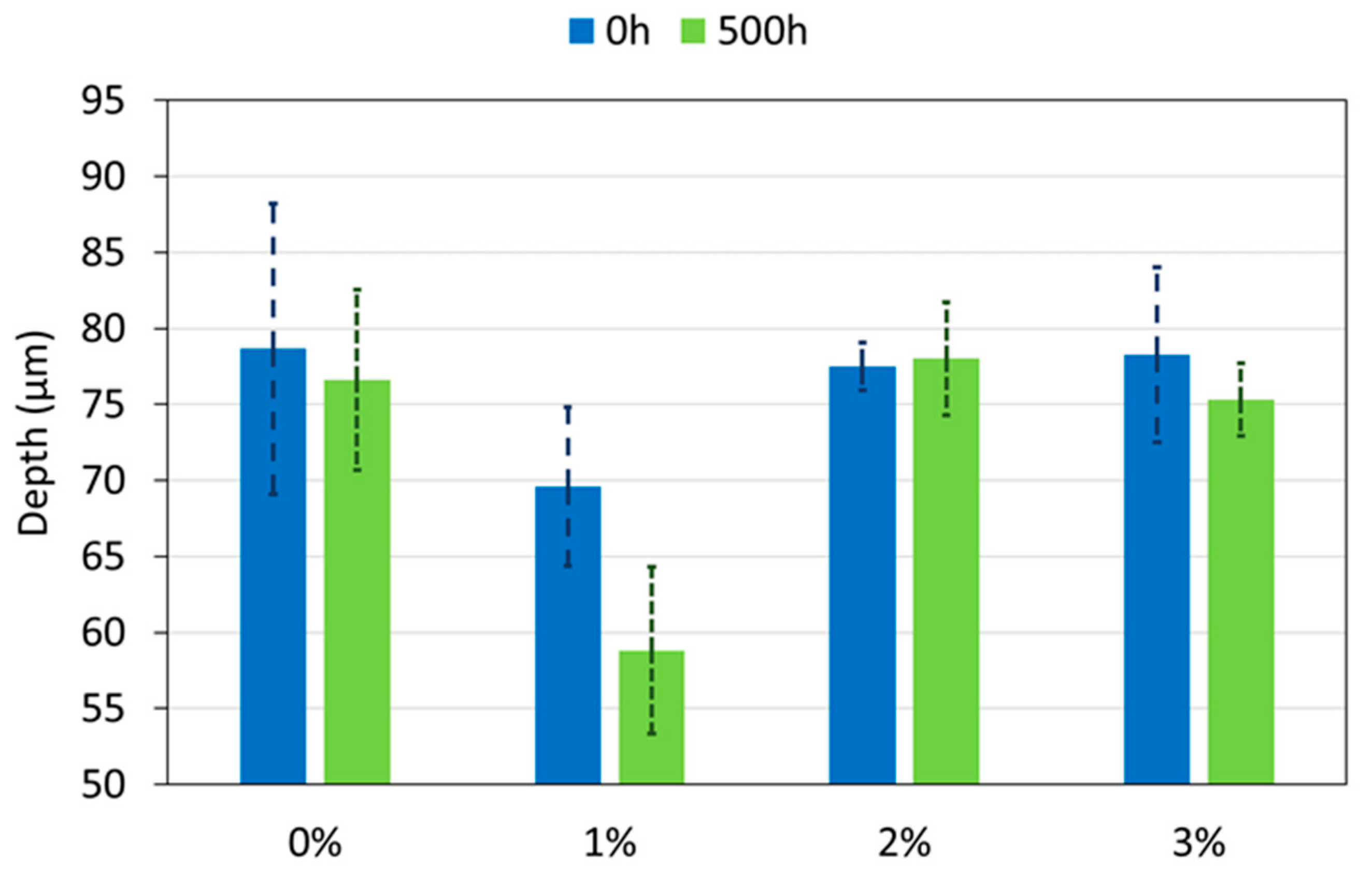
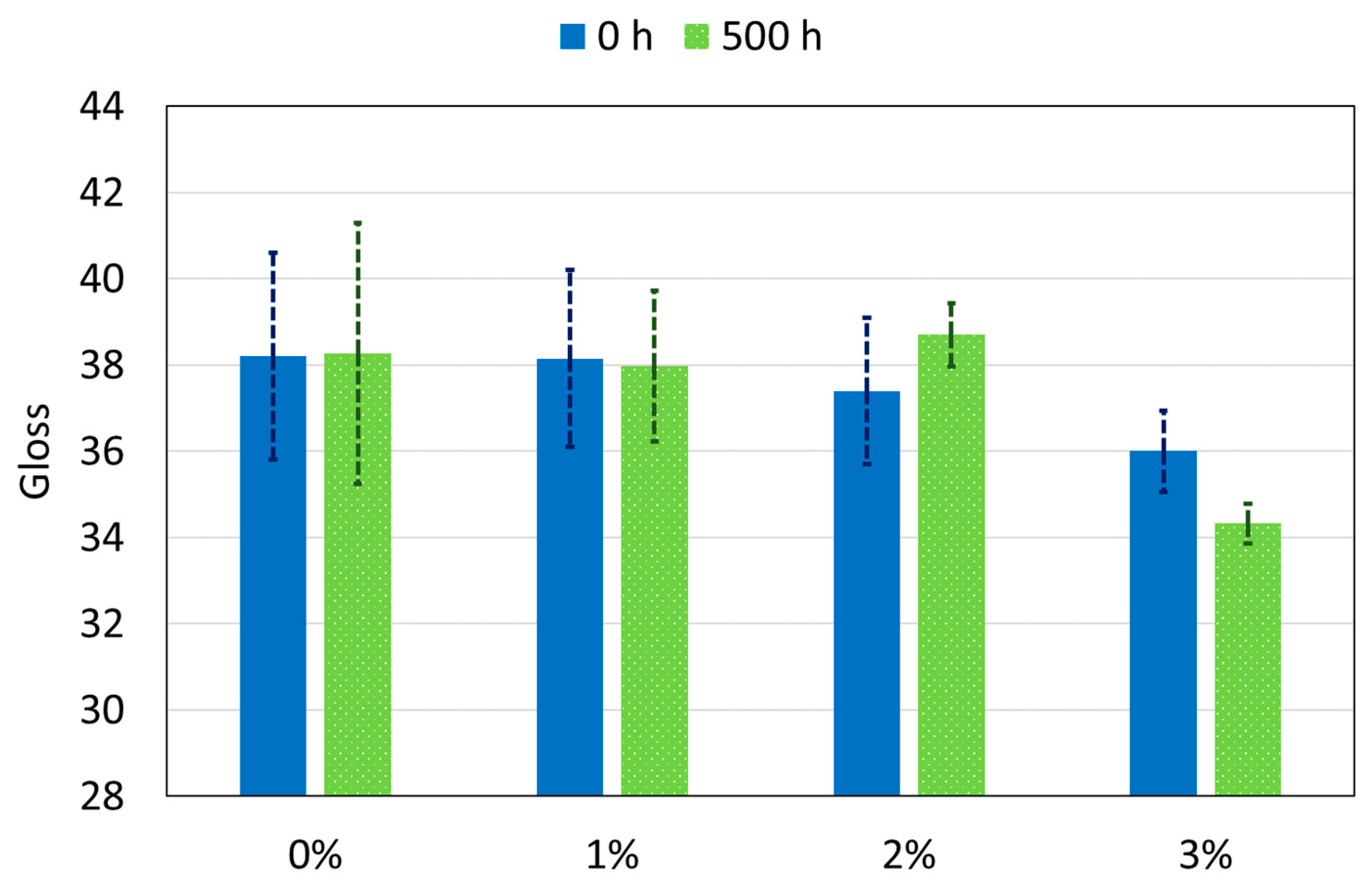
3. Results
4. Conclusions
- The addition of SiO2 nanoparticles to powder polyamide promotes the hardening and stiffening of the coatings manufactured, while slightly reducing their crystallinity;
- The wear behavior of studied coatings implies an abrasive mechanism, with material removed and bonded again to the PA11 coatings, due to their plasticity;
- Xenon exposure does not promote important chemical changes in the coatings. Only the formation of hard imides and carboxylic acid groups has been suggested by the obtained FTIR-ATR results;
- After xenon exposure, 1% coating increases its hardness and stiffness, showing the lowest loss of crystallinity. This material presents the best wear behavior after irradiation, unlike the other organic coatings under study;
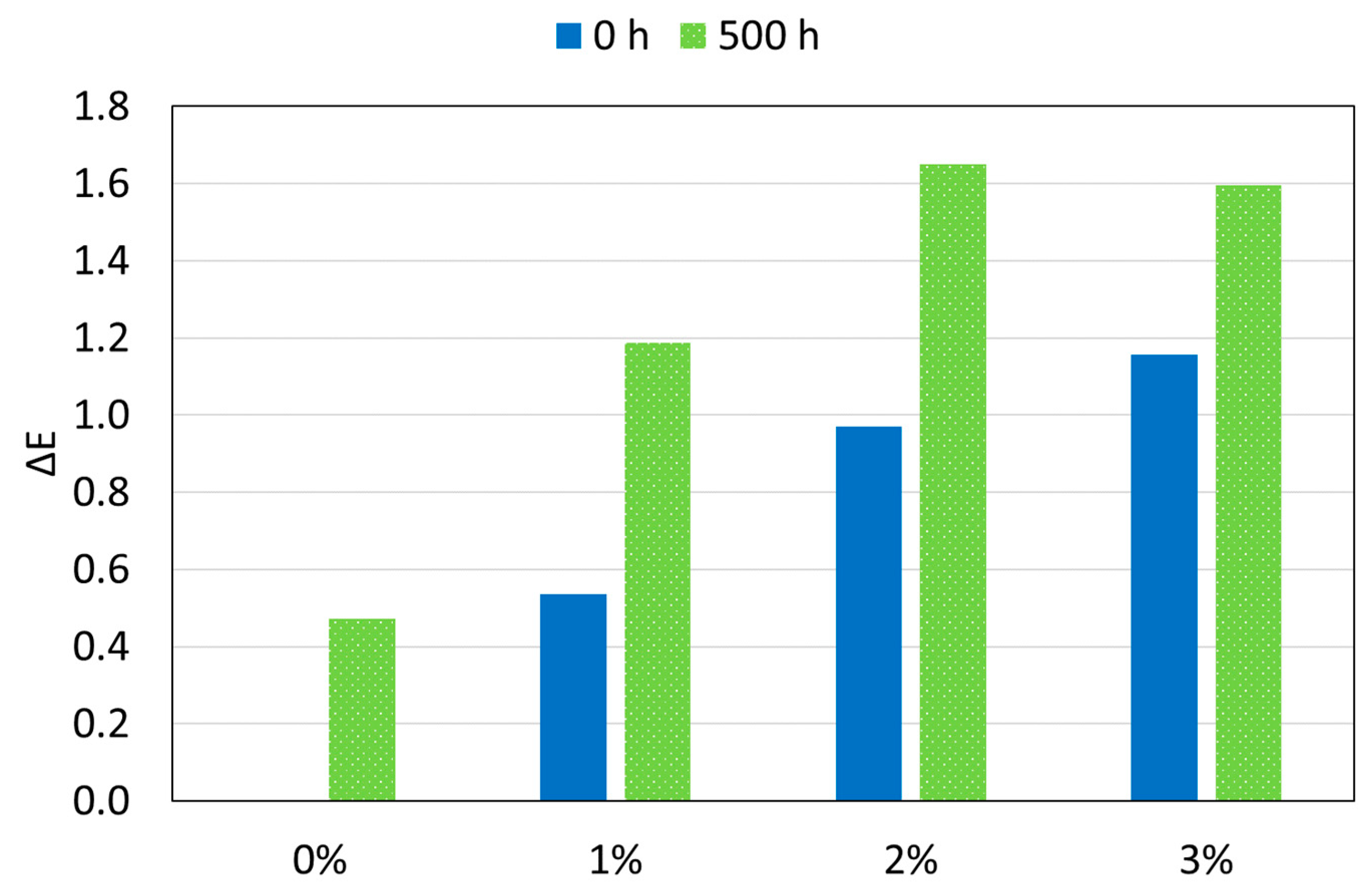
- All the studied polyamide-based coatings present excellent aesthetic properties, with very small changes in gloss (except for 3%) and color after 500 h of exposure to the xenon radiation.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Díez-Pascual, A.M. Nanoparticle reinforced polymers. Polymers 2019, 11, 625. [Google Scholar] [CrossRef]
- Montemor, M. Functional and smart coatings for corrosion protection: A review of recent advances. Surf. Coat. Technol. 2014, 258, 17–37. [Google Scholar] [CrossRef]
- Li, Y.D.; Ma, Y.Z.; Yan, F.F.; Cao, S.K. Nonisothermal crystallization kinetics of flame-sprayed polyamide 1010/nano-ZrO2 composite coatings. J. Appl. Polym. Sci. 2007, 106, 1535–1543. [Google Scholar] [CrossRef]
- Piazza, D.; Silveira, D.S.; Lorandi, N.P.; Birriel, E.J.; Scienza, L.C.; Zattera, A.J. Polyester-based powder coatings with montmorillonite nanoparticles applied on carbon steel. Prog. Org. Coat. 2012, 73, 42–46. [Google Scholar] [CrossRef]
- Allahverdi, A.; Ehsani, M.; Janpour, H.; Ahmadi, S. The effect of nanosilica on mechanical, thermal and morphological properties of epoxy coating. Prog. Org. Coat. 2012, 75, 543–548. [Google Scholar] [CrossRef]
- Bautista, Y.; González, J.; Gilabert, J.; Ibáñez, M.J.; Sanz, V. Correlation between the wear resistance, and the scratch resistance, for nanocomposite coatings. Prog. Org. Coat. 2011, 70, 178–185. [Google Scholar] [CrossRef]
- Isarn, I.; Bonnaud, L.; Massagués, L.; Serra, À.; Ferrando, F. Enhancement of thermal conductivity in epoxy coatings through the combined addition of expanded graphite and boron nitride fillers. Prog. Org. Coat. 2019, 133, 299–308. [Google Scholar] [CrossRef]
- El-Fattah, M.A.; El Saeed, A.M.; El-Ghazawy, R.A. Chemical interaction of different sized fumed silica with epoxy via ultrasonication for improved coating. Prog. Org. Coat. 2019, 129, 1–9. [Google Scholar] [CrossRef]
- Jarka, P.; Tański, T.; Matysiak, W.; Krzemiński, Ł.; Hajduk, B.; Bilewicz, M. Manufacturing and investigation of surface morphology and optical properties of composite thin films reinforced by TiO2, Bi2O3 and SiO2 nanoparticles. Appl. Surf. Sci. 2017, 424, 206–212. [Google Scholar] [CrossRef]
- Ngo, T.H.A.; Nguyen, D.T.; Do, K.D.; Minh Nguyen, T.T.; Mori, S.; Tran, D.T. Surface modification of polyamide thin film composite membrane by coating of titanium dioxide nanoparticles. J. Sci. Adv. Mater. Devices 2016, 1, 468–475. [Google Scholar] [CrossRef]
- Fernàndez-Àlvarez, M.; Velasco, F.; Bautista, A. Effect on wear resistance of nanoparticles addition to a powder polyester coating through ball milling. J. Coat. Technol. Res. 2018, 15, 771–779. [Google Scholar] [CrossRef]
- Fernández-Álvarez, M.; Velasco, F.; Bautista, A.; Galiana, B. Functionalizing organic powder coatings with nanoparticles through ball milling for wear applications. Appl. Surf. Sci. 2020, 513, 145834. [Google Scholar] [CrossRef]
- Barletta, M.; Gisario, A.; Rubino, G. Scratch response of high-performance thermoset and thermoplastic powders deposited by the electrostatic spray and ‘hot dipping’ fluidised bed coating methods: The role of the contact condition. Surf. Coat. Technol. 2011, 205, 5186–5198. [Google Scholar] [CrossRef]
- Mirabedini, S.M.; Kiamanesh, A. The effect of micro and nano-sized particles on mechanical and adhesion properties of a clear polyester powder coating. Prog. Org. Coat. 2013, 76, 1625–1632. [Google Scholar] [CrossRef]
- Doğan, E.; Acar, A.E. The use of anhydride linkages to increase the glass transition temperatures of polymers containing carboxyl end groups: A perspective in powder coatings. Prog. Org. Coat. 2013, 76, 513–518. [Google Scholar] [CrossRef]
- Gioia, C.; Minesso, A.; Cavalieri, R.; Marchese, P.; Celli, A.; Colonna, M. Powder coatings for indoor applications from renewable resources and recycled polymers. J. Coat. Technol. Res. 2015, 12, 555–562. [Google Scholar] [CrossRef]
- Rossi, S.; Fedel, M.; Petrolli, S.; Deflorian, F. Accelerated weathering and chemical resistance of polyurethane powder coatings. J. Coat. Technol. Res. 2016, 13, 427–437. [Google Scholar] [CrossRef]
- Du, Z.; Wen, S.; Wang, J.; Yin, C.; Yu, D.; Luo, J. The Review of Powder Coatings. J. Mater. Sci. Chem. Technol. 2016, 4, 54–59. [Google Scholar] [CrossRef]
- Joshi, G.S.; Adewar, S.; Joshi, S. Modelling and Product customization of PVC Coating Machine. Mater. Today Proc. 2018, 5, 4787–4792. [Google Scholar] [CrossRef]
- McAndrew, T.P.; Audenaert, M.; Petersheim, J.; Garcia, D.; Richards, T. Polyamide-11 powder coatings: Exceptional resistance to cavitation erosion. ACS Symp. Ser. 2007, 962, 190–200. [Google Scholar] [CrossRef]
- Kaci, M.; Focke, W.W.; van der Merwe, E.M. The effects of unmodified vermiculite on photooxidation of polyamide 11 nanocomposites under accelerated UV test. AIP Conf. Proc. 2018, 1981, 9–12. [Google Scholar] [CrossRef]
- Oliver-Ortega, H.; Méndez, J.A.; Mutjé, P.; Tarrés, Q.; Espinach, F.X.; Ardanuy, M. Evaluation of thermal and thermomechanical behaviour of bio-based polyamide 11 based composites reinforced with lignocellulosic fibres. Polymers 2017, 9, 522. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ma, Y.; Xie, B.; Cao, S.; Wu, Z. Dry friction and wear behavior of flame-sprayed polyamide1010/n-SiO2 composite coatings. Wear 2007, 262, 1232–1238. [Google Scholar] [CrossRef]
- Bagheri, H.; Roostaie, A. Electrospun modified silica-polyamide nanocomposite as a novel fiber coating. J. Chromatogr. A 2014, 1324, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Álvarez, M.; Velasco, F.; Bautista, A.; Abenojar, J. Effect of silica nanoparticles on the curing kinetics and erosion wear of an epoxy powder coating. J. Mater. Res. Technol. 2019, 9, 455–464. [Google Scholar] [CrossRef]
- Fernández-Álvarez, M.; Velasco, F.; Bautista, A. Epoxy powder coatings hot mixed with nanoparticles to improve their abrasive wear. Wear 2020, 448–449, 203211. [Google Scholar] [CrossRef]
- Naeimi, F.; Bagheri, R.; Hedayati, M.; Salehi, M.; Panjepour, M. Tribological and mechanical properties of amorphous and semi-crystalline PEEK/SiO2 nanocomposite coatings deposited on the plain carbon steel by electrostatic powder spray technique. Prog. Org. Coat. 2012, 74, 50–58. [Google Scholar] [CrossRef]
- García, M.; García-Turiel, J.; Norder, B.; Chavez, F.; Kooi, B.J.; Van Zyl, W.E.; Verweij, H.; Blank, D.H.A. Polyamide-6/silica nanocomposites. Adv. Eng. Mater. 2004, 6, 724–729. [Google Scholar] [CrossRef]
- Petrovicova, E.; Knight, R.; Schadler, L.S.; Twardowski, T.E. Nylon 11/silica nanocomposite coatings applied by the HVOF process. II. Mechanical and barrier properties. J. Appl. Polym. Sci. 2000, 78, 2272–2289. [Google Scholar] [CrossRef]
- Brusentseva, T.A.; Filippov, A.A.; Fomin, V.M.; Smirnov, S.V.; Veretennikova, I.A. Modification of epoxy resin with silica nanoparticles and process engineering of composites based on them. Mech. Compos. Mater. 2015, 51, 531–538. [Google Scholar] [CrossRef]
- Khodair, Z.T.; Khadom, A.A.; Jasim, H.A. Corrosion protection of mild steel in different aqueous media via epoxy/nanomaterial coating: Preparation, characterization and mathematical views. J. Mater. Res. Technol. 2018, 8, 424–435. [Google Scholar] [CrossRef]
- Karami, P.; Shojaei, A. Improvement of dry sliding tribological properties of polyamide 6 using diamond nanoparticles. Tribol. Int. 2017, 115, 370–377. [Google Scholar] [CrossRef]
- Sabatini, V.; Taroni, T.; Rampazzo, R.; Bompieri, M.; Maggioni, D.; Meroni, D.; Ortenzi, M.A.; Ardizzone, S. PA6 and Halloysite Nanotubes Composites with Improved Hydrothermal Ageing Resistance: Role of Filler Physicochemical Properties, Functionalization and Dispersion Technique. Polymers 2020, 12, 211. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Zhao, M. Photo-oxidative degradation of poly(vinyl chloride) based nanocomposites under ultraviolet irradiation. J. Dispers. Sci. Technol. 2010, 31, 1167–1172. [Google Scholar] [CrossRef]
- Gheno, G.; Ganzerla, R.; Bortoluzzi, M.; Paganica, R. Accelerated weathering degradation behaviour of polyester thermosetting powder coatings. Prog. Org. Coat. 2016, 101, 90–99. [Google Scholar] [CrossRef]
- Inoue, M. Studies on crystallization of high polymers by differential thermal analysis. J. Polym. Sci. Part A Gen. Pap. 1963, 1, 2697–2709. [Google Scholar] [CrossRef]
- Panaitescu, D.M.; Frone, A.N.; Nicolae, C. Micro- and nano-mechanical characterization of polyamide 11 and its composites containing cellulose nanofibers. Eur. Polym. J. 2013, 49, 3857–3866. [Google Scholar] [CrossRef]
- Velasco, F.; Blanco, G.; Bautista, A.; Martínez, M.A. Effect of welding on local mechanical properties of stainless steels for concrete structures using universal hardness tests. Constr. Build. Mater. 2009, 23, 1883–1891. [Google Scholar] [CrossRef]
- Kaci, M.; Dehouche, N.; Focke, W.W.; van der Merwe, E.M. A degradation study of polyamide 11/vermiculite nanocomposites under accelerated UV test. Polym. Eng. Sci. 2019, 59, 2449–2457. [Google Scholar] [CrossRef]
- Wan, J.; Bu, Z.Y.; Li, C.; Fan, H.; Li, B.G. Preparation, melting, glass relaxation and nonisothermal crystallization kinetics of a novel dendritic nylon-11. Thermochim. Acta 2011, 524, 117–127. [Google Scholar] [CrossRef]
- Domingos, E.; Pereira, T.M.C.; de Castro, E.V.R.; Romão, W.; de Sena, G.L.; Guimarães, R.C.L. Monitorando a degradação da poliamida 11 (PA-11) via espectroscopia na região do infravermelho médio com transformada de fourier (FTIR). Polímeros 2012, 23, 37–41. [Google Scholar] [CrossRef]
- Parodi, E.; Govaert, L.E.; Peters, G.W.M. Glass transition temperature versus structure of polyamide 6: A flash-DSC study. Thermochim. Acta 2017, 657, 110–122. [Google Scholar] [CrossRef]
- Yang, F.; Ou, Y.; Yu, Z. Polyamide 6/silica nanocomposites prepared by in situ polymerization. J. Appl. Polym. Sci. 1998, 69, 355–361. [Google Scholar] [CrossRef]
- Han, C.; Sahle-Demessie, E.; Zhao, A.Q.; Richardson, T.; Wang, J. Environmental aging and degradation of multiwalled carbon nanotube reinforced polypropylene. Carbon 2018, 129, 137–151. [Google Scholar] [CrossRef]
- Giraldo, L.F.; Echeverri, M.; López, B.L. Reinforcement of polyamide 6 with nanoparticles. Macromol. Symp. 2007, 258, 119–128. [Google Scholar] [CrossRef]
- Dechet, M.A.; Lanzl, L.; Wilden, A.; Sattes, M.-M.; Drummer, D.; Peukert, W.; Schmidt, J. Manufacturing of Nanoparticle-Filled PA11 Composite Particles for Selective Laser Sintering. In Proceedings of the 30th Annual International Solid Freedom Fabrication Symposium—An Additive Manufacturing Conference—2019, The University of Texas at Austin, Austin, TX, USA, 12–14 August 2019; pp. 1022–1030. [Google Scholar]
- Carrasco, F.; Pagès, P.; Pascual, S.; Colom, X. Artificial aging of high-density polyethylene by ultraviolet irradiation. Eur. Polym. J. 2001, 37, 1457–1464. [Google Scholar] [CrossRef]
- Khadem-Hosseini, A.; Pazokifard, S.; Mirabedini, S.M. Durability and mechanical performance of a photo-catalytic water-based nanocomposite coating. Prog. Org. Coat. 2017, 112, 254–262. [Google Scholar] [CrossRef]
- Yu, F.; Gao, J.; Liu, C.; Chen, Y.; Zhong, G.; Hodges, C.; Chen, M.; Zhang, H. Preparation and UV aging of nano-SiO2/fluorinated polyacrylate polyurethane hydrophobic composite coating. Prog. Org. Coat. 2020, 141, 105556. [Google Scholar] [CrossRef]
- Eslami, R.; Bagheri, R.; Hashemzadeh, Y.; Salehi, M. Optical and mechanical properties of transparent acrylic based polyurethane nano silica composite coatings. Prog. Org. Coat. 2014, 77, 1184–1190. [Google Scholar] [CrossRef]
- Amrollahi, S.; Mohseni, M.; Ramezanzadeh, B. Fabrication of a low surface energy acrylic/melamine clearcoat with enhanced weathering and biological resistances: Investigation of the role of organic UV absorber and nanosilica particles. Prog. Org. Coat. 2017, 105, 132–142. [Google Scholar] [CrossRef]
- Liu, X.-Q.; Wang, Y.; Yang, W.; Liu, Z.Y.; Luo, Y.; Xie, B.H.; Yang, M.B. Control of morphology and properties by the selective distribution of nano-silica particles with different surface characteristics in PA6/ABS blends. J. Mater. Sci. 2012, 47, 4620–4631. [Google Scholar] [CrossRef]
- Varsavas, S.D.; Kaynak, C. Weathering degradation performance of PLA and its glass fiber reinforced composite. Mater. Today Commun. 2018, 15, 344–353. [Google Scholar] [CrossRef]
- Reynaud, E.; Jouen, T.; Gauthier, C.; Vigier, G.; Varlet, J. Nanofillers in polymeric matrix: A study on silica reinforced PA6. Polymer 2001, 42, 8759–8768. [Google Scholar] [CrossRef]
- Kumar, S.; Panneerselvam, K. Two-body abrasive wear behavior of nylon 6 and glass fiber reinforced (GFR) nylon 6 composite. Procedia Technol. 2016, 25, 1129–1136. [Google Scholar] [CrossRef]
- Schön, J. Coefficient of friction and wear of a carbon fiber epoxy matrix composite. Wear 2004, 257, 395–407. [Google Scholar] [CrossRef]
- Shipway, P.H.; Ngao, N.K. Microscale abrasive wear of polymeric materials. Wear 2003, 255, 742–750. [Google Scholar] [CrossRef]
- Sahnoune, M.; Kaci, M.; Taguet, A.; Delbé, K.; Mouffok, S.; Abdi, S.; Lopez-Cuesta, J.M.; Focke, W.W. Tribological and mechanical properties of polyamide-11/halloysite nanotube nanocomposites. J. Polym. Eng. 2018, 39, 25–34. [Google Scholar] [CrossRef]
- Valdesueiro, D.; Hettinga, H.; Drijfhout, J.P.; Lips, P.; Meesters, G.M.H.; Kreutzer, M.T.; Ruud van Ommen, J. Tuning roughness and gloss of powder coating paint by encapsulating the coating particles with thin Al2O3 films. Powder Technol. 2017, 318, 401–410. [Google Scholar] [CrossRef]
- Malshe, V.C.; Waghoo, G. Weathering study of epoxy paints. Prog. Org. Coat. 2004, 51, 267–272. [Google Scholar] [CrossRef]
- Franceschi, E.; Letardi, P.; Luciano, G. Colour measurements on patinas and coating system for outdoor bronze monuments. J. Cult. Herit. 2006, 7, 166–170. [Google Scholar] [CrossRef]
- Lopez de Armentia, S.; Pantoja, M.; Abenojar, J.; Martinez, M.A. Development of silane-based coatings with zirconia nanoparticles combining wetting, tribological, and aesthetical properties. Coatings 2018, 8, 368. [Google Scholar] [CrossRef]
- Ghasemi-Kahrizsangi, A.; Neshati, J.; Shariatpanahi, H.; Akbarinezhad, E. Improving the UV degradation resistance of epoxy coatings using modified carbon black nanoparticles. Prog. Org. Coat. 2015, 85, 199–207. [Google Scholar] [CrossRef]
- Jalili, M.M.; Moradian, S. Deterministic performance parameters for an automotive polyurethane clearcoat loaded with hydrophilic or hydrophobic nano-silica. Prog. Org. Coat. 2009, 66, 359–366. [Google Scholar] [CrossRef]
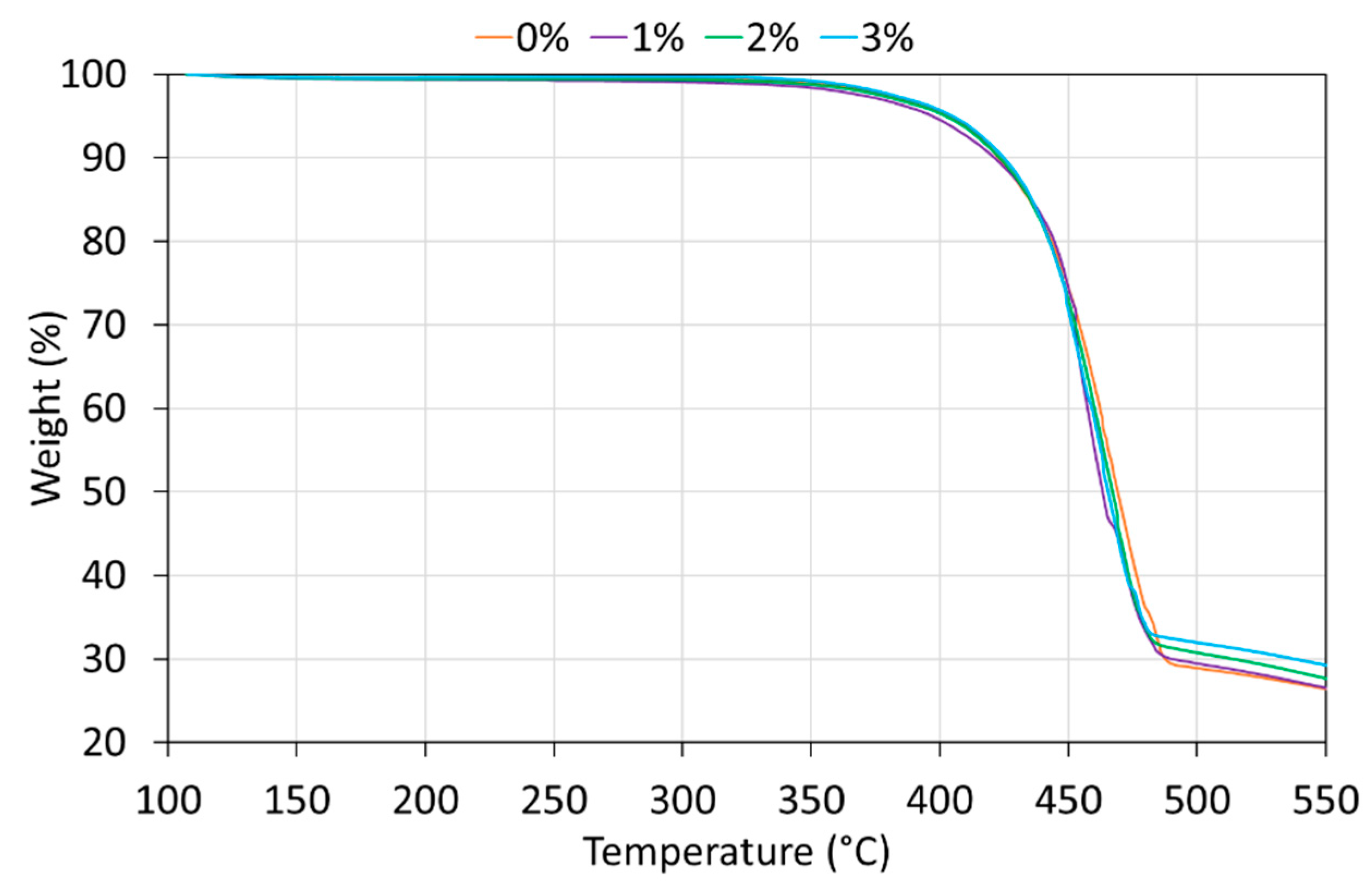
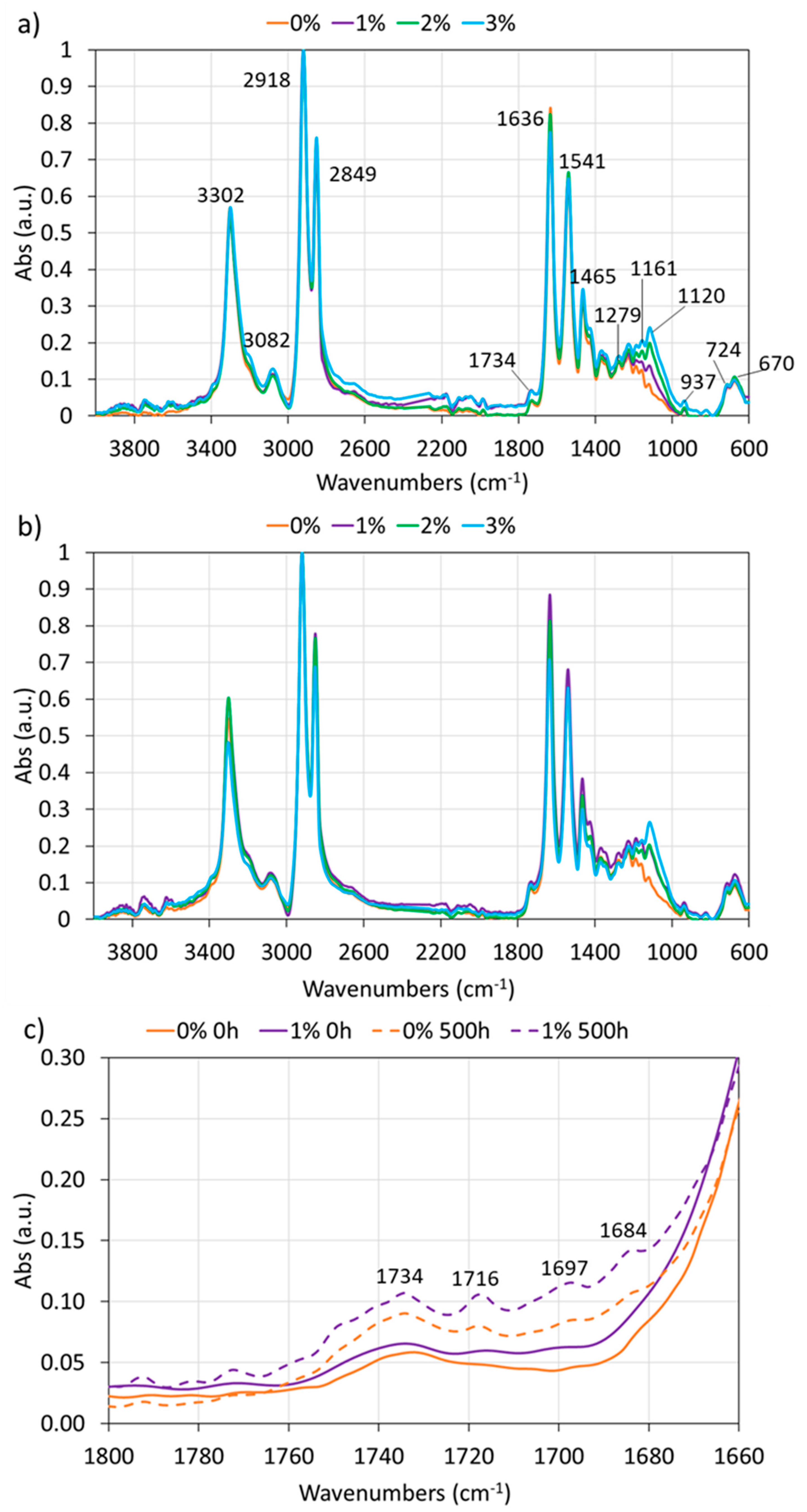
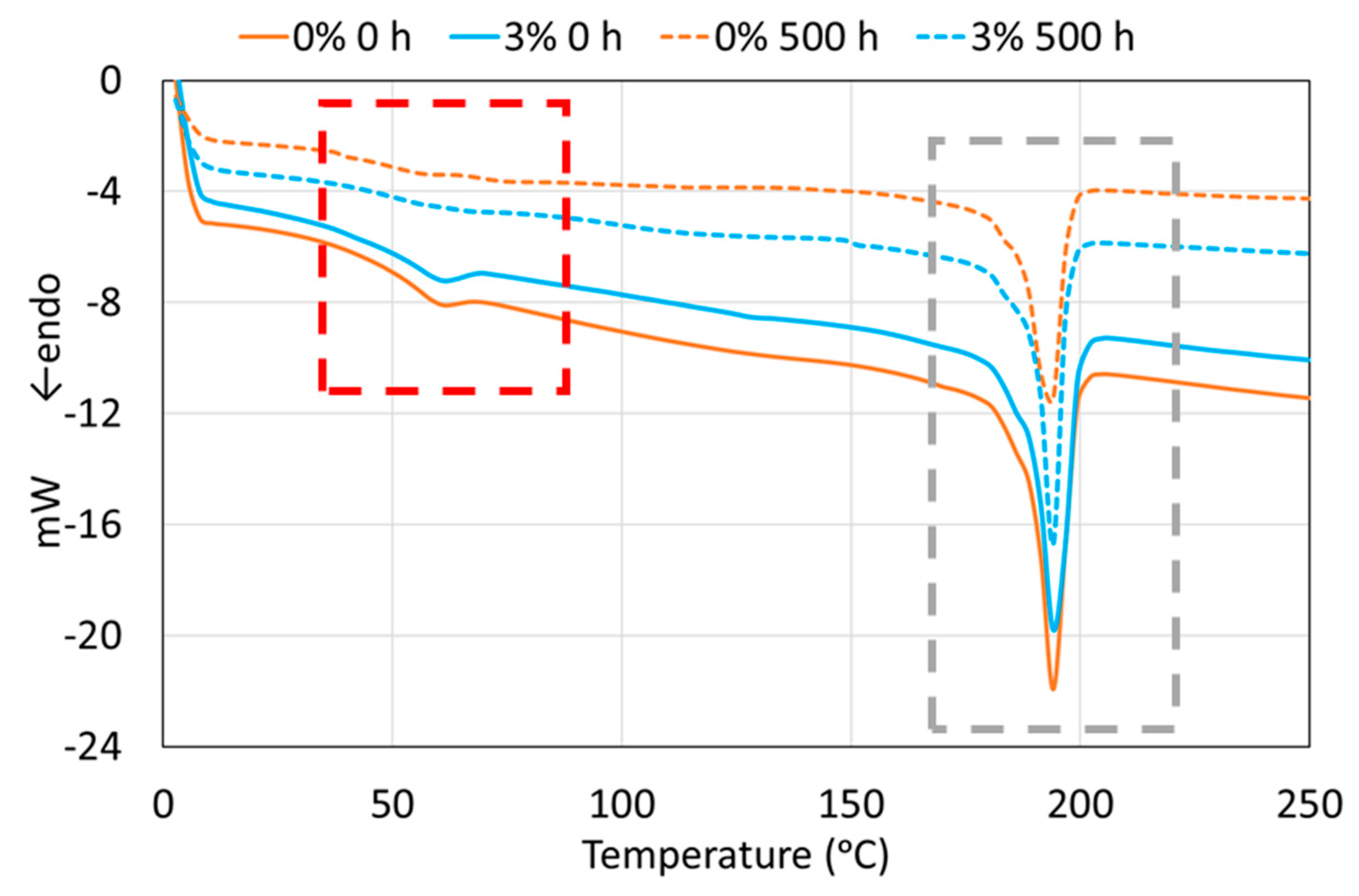


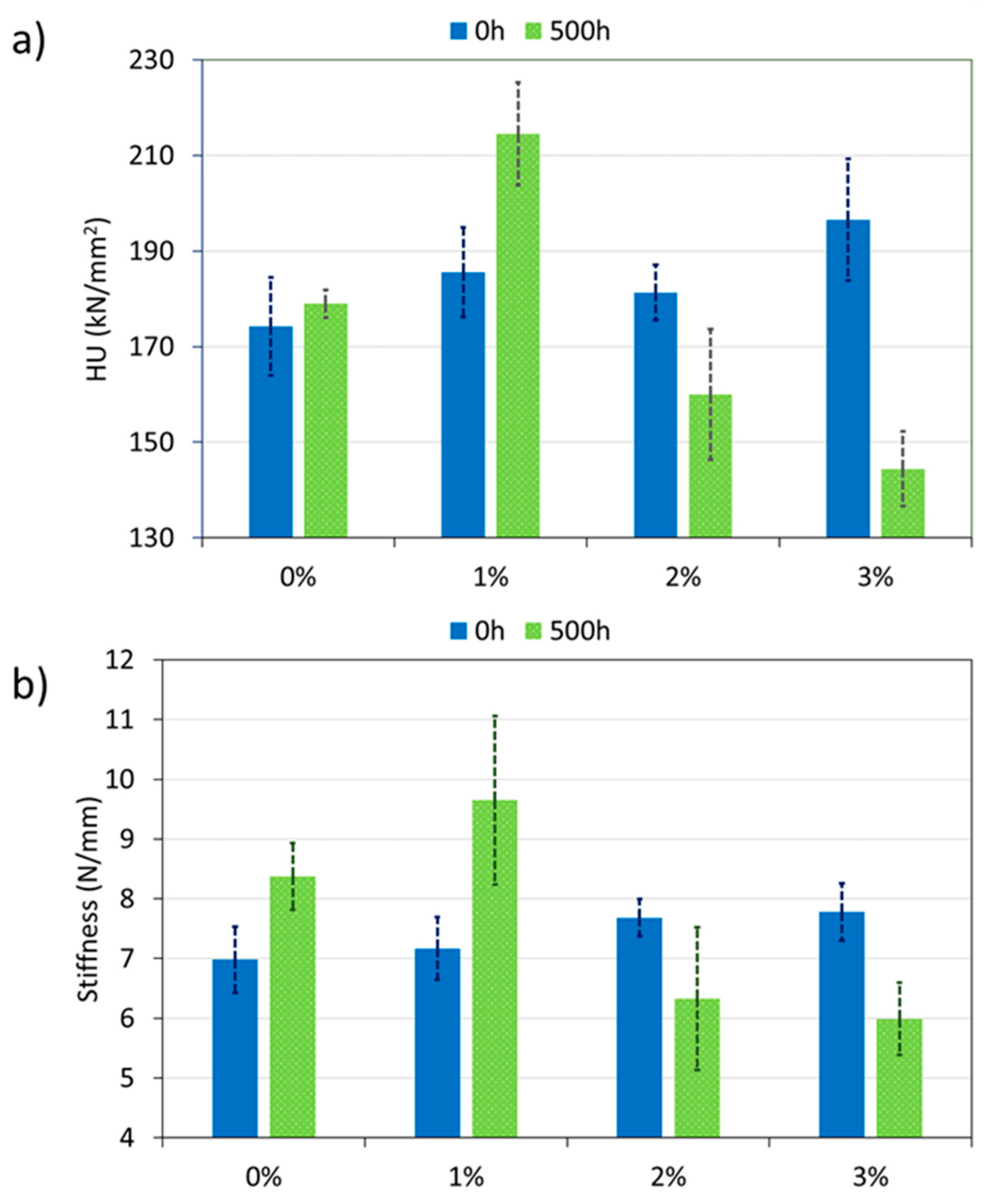


| Wavenumbers (cm−1) | Assignation |
|---|---|
| 3302 | N–H stretch |
| 3082 | N–H (Amide II) |
| 2918 | Asymmetric CH2 stretch |
| 2849 | Symmetric CH2 stretch |
| 1734 | Imides and impurities |
| 1636 | C=O stretch (Amide I) |
| 1541 | N–H and C–O (Amide II) |
| 1465 | CH2 scissoring vibration |
| 1279 | C–N–H (Amide III) |
| 1161 | O=C–N stretch |
| 1114 | Si–O–Si |
| 937 | Amide IV |
| 724 | CH2 rocking |
| 670 | Amide V |
| PA11 Coatings (0 h) | Tg (°C) | Tm (°C) | ΔHm (J·g−1) | XC (%) |
|---|---|---|---|---|
| 0% | 48.7 ± 0.8 | 190.4 ± 1.3 | 27.3 ± 0.4 | 17.2 ± 0.2 |
| 1% | 50.4 ± 0.4 | 190.2 ± 0.6 | 25.1 ± 0.3 | 16.1 ± 0.2 |
| 2% | 48.3 ± 0.6 | 190.5 ± 1.8 | 23.9 ± 0.3 | 15.5 ± 0.2 |
| 3% | 49.1 ± 0.4 | 191.1 ± 0.4 | 24.0 ± 0.4 | 15.8 ± 0.3 |
| PA11 Coatings (500 h) | Tg (°C) | Tm (°C) | ΔHm (J·g−1) | XC (%) |
|---|---|---|---|---|
| 0% | 40.6 ± 1.6 | 191.9 ± 0.4 | 20.6 ± 1.6 | 12.6 ± 0.5 |
| 1% | 46.9 ± 0.4 | 192.7 ± 0.1 | 24.5 ± 0.6 | 15.4 ± 0.1 |
| 2% | 45.2 ± 0.2 | 193.5 ± 0.1 | 22.7 ± 0.9 | 14.7 ± 0.6 |
| 3% | 45.4 ± 0.8 | 193.3 ± 1.1 | 23.2 ± 0.6 | 15.3 ± 0.4 |
| 0 h | 500 h | |||||
|---|---|---|---|---|---|---|
| Sample | L* | a* | b* | L* | a* | b* |
| 0% | 93.5 | −1.2 | 0.4 | 93.6 | −1.5 | 0.1 |
| 1% | 93.5 | −1.1 | 0.9 | 94.3 | −1.5 | 0.1 |
| 2% | 93.7 | −0.9 | 1.3 | 94.6 | −1.5 | 0.2 |
| 3% | 93.4 | −0.6 | 1.4 | 94.2 | −1.5 | 0.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Álvarez, M.; Velasco, F.; Bautista, A.; Lobo, F.C.M.; Fernandes, E.M.; Reis, R.L. Manufacturing and Characterization of Coatings from Polyamide Powders Functionalized with Nanosilica. Polymers 2020, 12, 2298. https://doi.org/10.3390/polym12102298
Fernández-Álvarez M, Velasco F, Bautista A, Lobo FCM, Fernandes EM, Reis RL. Manufacturing and Characterization of Coatings from Polyamide Powders Functionalized with Nanosilica. Polymers. 2020; 12(10):2298. https://doi.org/10.3390/polym12102298
Chicago/Turabian StyleFernández-Álvarez, Maria, Francisco Velasco, Asuncion Bautista, Flavia Cristina M. Lobo, Emanuel M. Fernandes, and Rui L. Reis. 2020. "Manufacturing and Characterization of Coatings from Polyamide Powders Functionalized with Nanosilica" Polymers 12, no. 10: 2298. https://doi.org/10.3390/polym12102298
APA StyleFernández-Álvarez, M., Velasco, F., Bautista, A., Lobo, F. C. M., Fernandes, E. M., & Reis, R. L. (2020). Manufacturing and Characterization of Coatings from Polyamide Powders Functionalized with Nanosilica. Polymers, 12(10), 2298. https://doi.org/10.3390/polym12102298










