Surface and Structural Properties of Medical Acrylonitrile Butadiene Styrene Modified with Silver Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Manufacturing
2.2. Material Evaluation
2.2.1. Scanning Electron Microscopy
2.2.2. Roughness
2.2.3. Surface Wettability
2.2.4. Tensile Test
2.2.5. Differential Scanning Calorimetry and Thermogravimetry
2.2.6. Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy
2.2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Feng, J.; Carpanese, C.; Fina, A. Thermal decomposition investigation of ABS containing Lewis-acid type metal salts. Polym. Degrad. Stab. 2016, 129, 319–327. [Google Scholar] [CrossRef]
- McKeen, L.W. Fatigue and Tribological Properties of Plastics and Elastomers, 3rd ed.; William Andrew: Norwich, NY, USA, 2016. [Google Scholar]
- Martinho, G.; Pires, A.; Saraiva, L.; Ribeiro, R. Composition of plastics from wasteelectrical and electronic equipment (WEEE) by direct sampling. Waste Manag. 2012, 32, 1213–1217. [Google Scholar] [CrossRef]
- Huang, G.; Huo, S.; Xu, X.; Chen, W.; Jin, Y.; Li, R.; Song, P.; Wang, H. Realizing simultaneous improvements in mechanical strength, flame retardancy and smoke suppression of ABS nanocomposites from multifunctional graphene. Compos. Part B Eng. 2019, 177, 107377. [Google Scholar] [CrossRef]
- Elix Polymers Brochure High Performance ABS Products for Medical Applications. Available online: www.elix-polymers.com (accessed on 4 August 2016).
- Ziąbka, M. A review of materials used in middle ear prosthetics. Ceram. Mater. 2018, 70, 65–85. [Google Scholar]
- Ziąbka, M.; Dziadek, M.; Menaszek, E.; Banasiuk, R.; Królicka, A. Middle ear prosthesis with bactericidal efficacy—In Vitro investigation. Molecules 2017, 22, 1681. [Google Scholar] [CrossRef] [PubMed]
- Ziąbka, M.; Menaszek, E.; Tarasiuk, J.; Wroński, S. Biocompatible Nanocomposite Implant with Silver Nanoparticles for Otology—In Vivo Evaluation. Nanomaterials 2018, 8, 764. [Google Scholar] [CrossRef] [PubMed]
- Teo, A.J.T.; Mishra, A.; Park, I.; Kim, Y.-J.; Park, W.-T.; Yoon, Y.-J. Polymeric Biomaterials for Medical Implants and Devices. ACS Biomater. Sci. Eng. 2016, 2, 454–472. [Google Scholar] [CrossRef]
- Joseph Francis, P.J.; Arun, K.J.; Navas, A.A.; Irene, J. Biomedical Applications of Polymers—An Overview. Curr. Trends Biomed. Eng. Biosci. Mini Rev. 2018, 15, 1–2. [Google Scholar]
- Kammula, R.G.; Morris, J.M. Considerationsfor the biocompatibility evaluation of medical devices. Med. Device Diagn. Ind. 2001, 23, 82–92. [Google Scholar]
- Williams, D.F. On the mechanisms of biocompatibility. Biomaterials 2008, 29, 2941–2953. [Google Scholar] [CrossRef]
- Chudobova, D.; Cihalova, K.; Kopel, P.; Melichar, L.; Ruttkay-Nedecky, B.; Vaculovicova, M.; Adam, V.; Kizek, R. Complexes of Metal-Based Nanoparticles with Chitosan Suppressing the Risk of Staphylococcus aureus and Escherichia coli Infections. Nanotechnol. Diagn. Treat. Prophyl. Infect. Dis. 2015, 217–232. [Google Scholar] [CrossRef]
- Radeshkumar, C.; Munstedt, H. Antimicrobial Polymers from Polypropylene/SilverComposites-Ag+ Release Measured by Anode Stripping Voltametry. React. Funct. Polym. 2006, 66, 780–788. [Google Scholar] [CrossRef]
- Capek, I. Polymer decorated gold nanoparticles in nanomedicine conjugates. Adv. Colloid Interface Sci. 2017, 249, 386–399. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, K.S.; Rahman, A.; Husen, T.; Husen, A. Properties of Zinc Oxide Nanoparticles and Their Activity Against Microbes. Nanoscale Res. Lett. 2018, 13, 141. [Google Scholar] [CrossRef] [PubMed]
- Essa, A.M.M.; Khallaf, M.K. Antimicrobial potential of consolidation polymers loaded with biological copper nanoparticles. BMC Microbiol. 2016, 16, 144. [Google Scholar] [CrossRef]
- Schmidt, G.; Malwitz, M.M. Properties of polymer–nanoparticle composites. Curr. Opin. Colloid Interface Sci. 2003, 8, 103–108. [Google Scholar] [CrossRef]
- Bitar, D.; Parvizi, J. Biological response to prosthetic debris. World J. Orthop. 2015, 6, 172–189. [Google Scholar] [CrossRef]
- Günther, D.; Scharnweber, D.; Hess, R.; Wolf-Brandstetter, C.; Grosse Holthaus, M.; Lasagni, A.F. High precision patterning of biomaterials using the direct laser interference patterning technology. Laser Surf. Modif. Biomater. 2016, 3–33. [Google Scholar] [CrossRef]
- Torrado, A.R.; Shemelya, C.M.; English, J.D.; Lin, Y.; Wicker, R.B.; Roberson, D.A. Characterizing the Effect of Additives to ABS on the Mechanical Property Anisotropy of Specimens Fabricated by Material Extrusion 3D Printing. Addit. Manuf. 2015, 6, 1–47. [Google Scholar] [CrossRef]
- Al-Hydarya, I.A.D.; Al-Rubiae, M.S.J. The Role of Anatase Nanoparticles on the Mechanical Properties and the Bacterial Adhesion to Acrylonitrile-Butadiene-Styrene Terpolymer. Mater. Res. 2019, 22. [Google Scholar] [CrossRef]
- ISO. 10993-13:2010 Biological Evaluation of Medical Devices—Part 13: Identification and Quantification of Degradation Products from Polymeric Medical Devices; International Organization for Standardization: Geneva, Switzerland, 2010. [Google Scholar]
- PN-EN. ISO 527-1:2012 Plastics—Determination of Tensile Properties—Part 1: General Principles; International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- Aid, S.; Eddhahak, A.; Ortega, Z.; Froelich, D.; Tcharkhtchi, A. Experimental study of the miscibility of ABS/PC polymer blends and investigation of the processing effect. J. Appl. Polym. Sci. 2017, 134, 44975. [Google Scholar] [CrossRef]
- Yang, B.; Huang, W.M.; Li, C.; Chor, J.H. Effects of moisture on the glass transition temperature of polyurethane shape memory polymer filled with nano-carbon powder. Eur. Polym. J. 2005, 41, 1123–1128. [Google Scholar] [CrossRef]
- Jian, R.-K.; Chen, L.; Zhao, B.; Yan, Y.-W.; Li, X.-F.; Wang, Y.-Z. Acrylonitrile−Butadiene−Styrene Terpolymer with Metal Hypophosphites: Flame Retardance and Mechanism Research. Ind. Eng. Chem. Res. 2014, 53, 2299–2307. [Google Scholar] [CrossRef]
- Thanh Truc, N.T.; Lee, B.-K. Selective separation of ABS/PC containing BFRs from ABSs mixture of WEEE by developing hydrophilicity with ZnO coating under microwave treatment. J. Hazard Mater. 2017, 329, 84–91. [Google Scholar] [CrossRef]
- Li, J.; Chen, F.; Yang, L.; Jiang, L.; Dan, Y. FTIR analysis on aging characteristics of ABS/PC blend under UV-irradiation in air. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2017, 184, 361–367. [Google Scholar] [CrossRef]
- Sánchez-Valdes, S.; Ortega-Ortiz, H.; Ramos-de Valle, L.F.; Medellín-Rodríguez, F.J.; Guedea-Miranda, R. Mechanical and antimicrobial properties of multilayer films with a polyethylene/silver nanocomposite layer. J. Appl. Polym. Sci. 2008, 111, 953–962. [Google Scholar] [CrossRef]
- Zendehnam, A.; Robatmili, N.; Hosseini, S.M.; Arabzadegan, M.; Madaeni, S.S. Fabrication of novel (acrylonitrile butadiene styrene/activated carbon/silver nanoparticles) heterogeneous anion exchange membrane: Physico-chemical and antibacterial characteristics. J. Taiwan Inst. Chem. Eng. 2013, 44, 670–677. [Google Scholar] [CrossRef]
- Avila-Alfaro, J.A.; Sánchez-Valdes, S.; Ramos-deValle, L.F.; Ortega-Ortiz, H.; Méndez-Nonell, J.; Patiño-Soto, A.P.; Narro-Cespedes, R.I.; Perera-Mercado, Y.A.; Avalos-Belmontes, F. Ultrasound irradiation coating of silver nanoparticle on ABS sheet surface. J. Inorg. Organomet. Polym. Mater. 2013, 23, 673–683. [Google Scholar] [CrossRef]
- Ziąbka, M.; Dziadek, M.; Menaszek, E. Biocompatibility of Poly(acrylonitrile-butadiene-styrene) Nanocomposites Modified with Silver Nanoparticles. Polymers 2018, 10, 1257. [Google Scholar] [CrossRef]
- Chatterjee, A. Effect of nanoTiO2 addition on poly methyl methacrylate. J. Appl. Polym. Sci. 2010, 16, 3396–3407. [Google Scholar]
- Ziąbka, M.; Dziadek, M. Long-lasting examinations of Surface and Structural Properties of Medical Polypropylene Modified with Silver Nanoparticles. Polymers 2019, 11, 2018. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.K.; Braihi, A.J.; Jabbar, R.S. Studying the Effect of Nano Silver on Some Properties of Acrylonitrile Butadiene Styrene Copolymer for Medical Devices. Int. J. Eng. Technol. 2019, 8, 208–213. [Google Scholar]
- Köroğlu, A.; Şahin, O.; Kürkçüoğlu, I.; Dede, D.O.; Özdemir, T.; Hazer, B. Silver nanoparticle incorporation effect on mechanical and thermal properties of denture base acrylic resins. J. Appl. Oral Sci. 2016, 24, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Sodagar, A.; Kassaee, M.Z.; Akhavan, A.; Javadi, N.; Arab, S.; Kharazifard, M.J. Effect of silver nano particles on flexural strength of acrylic resins. J. Prosthodont. Res. 2012, 56, 120–124. [Google Scholar] [CrossRef]



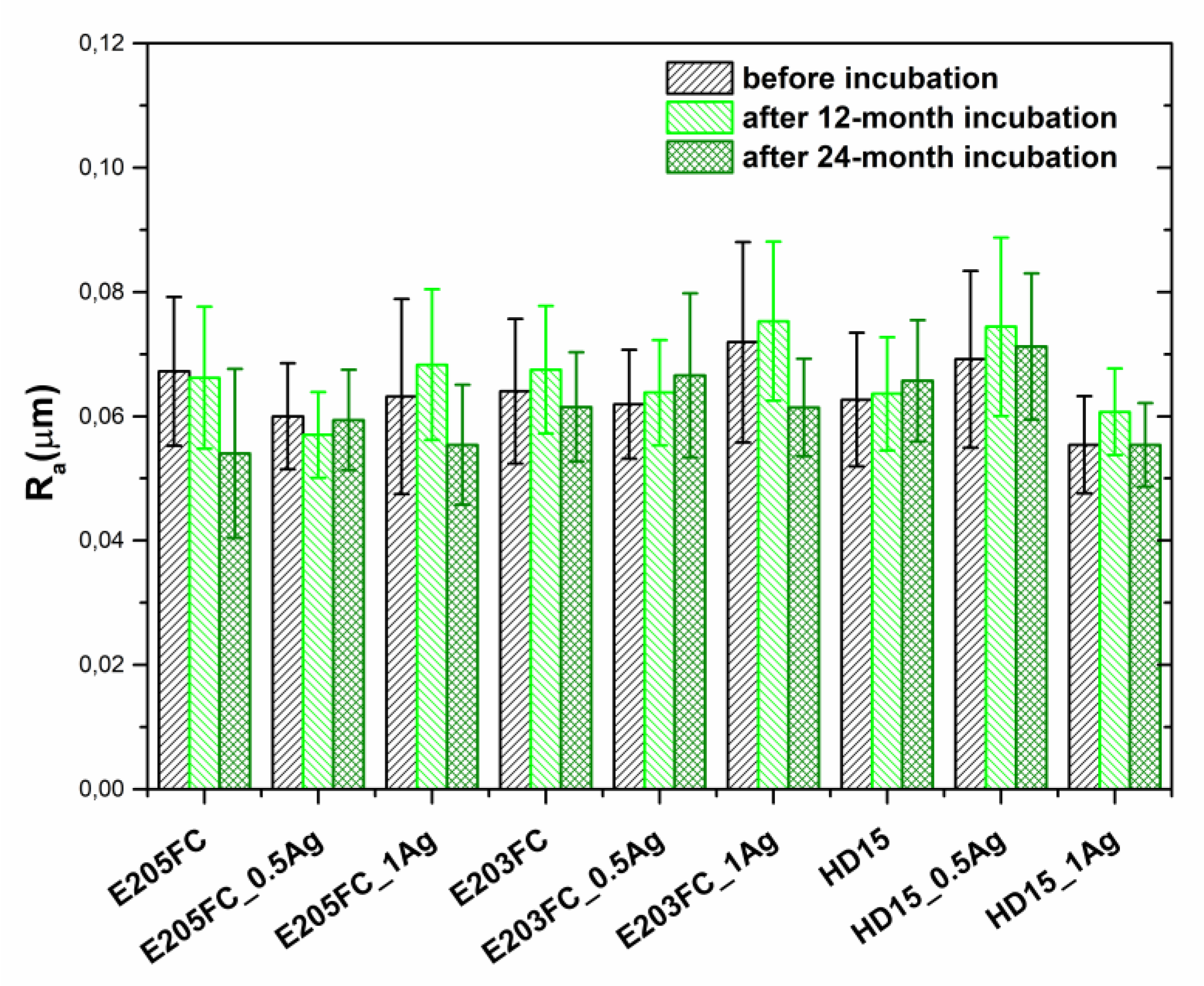
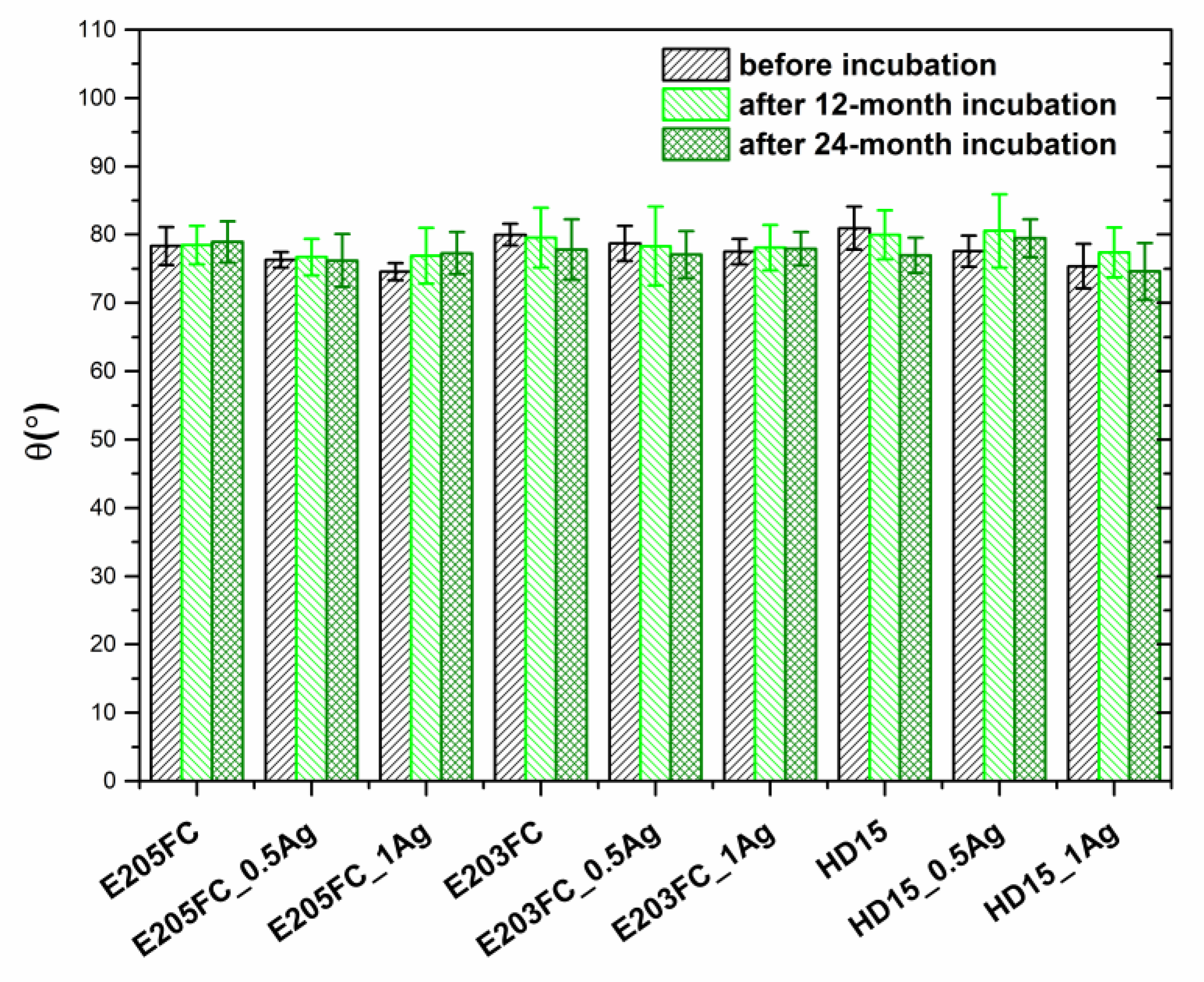
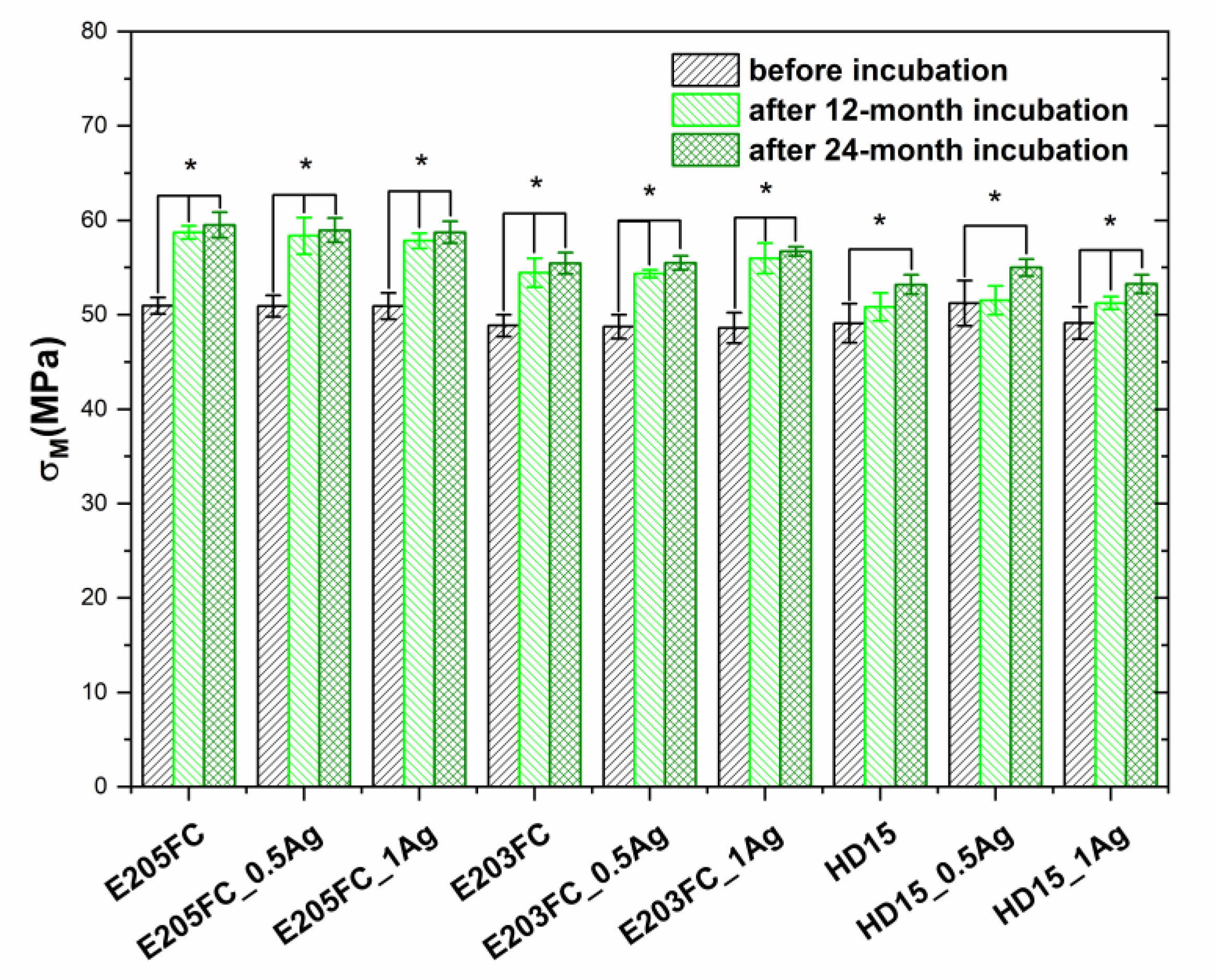
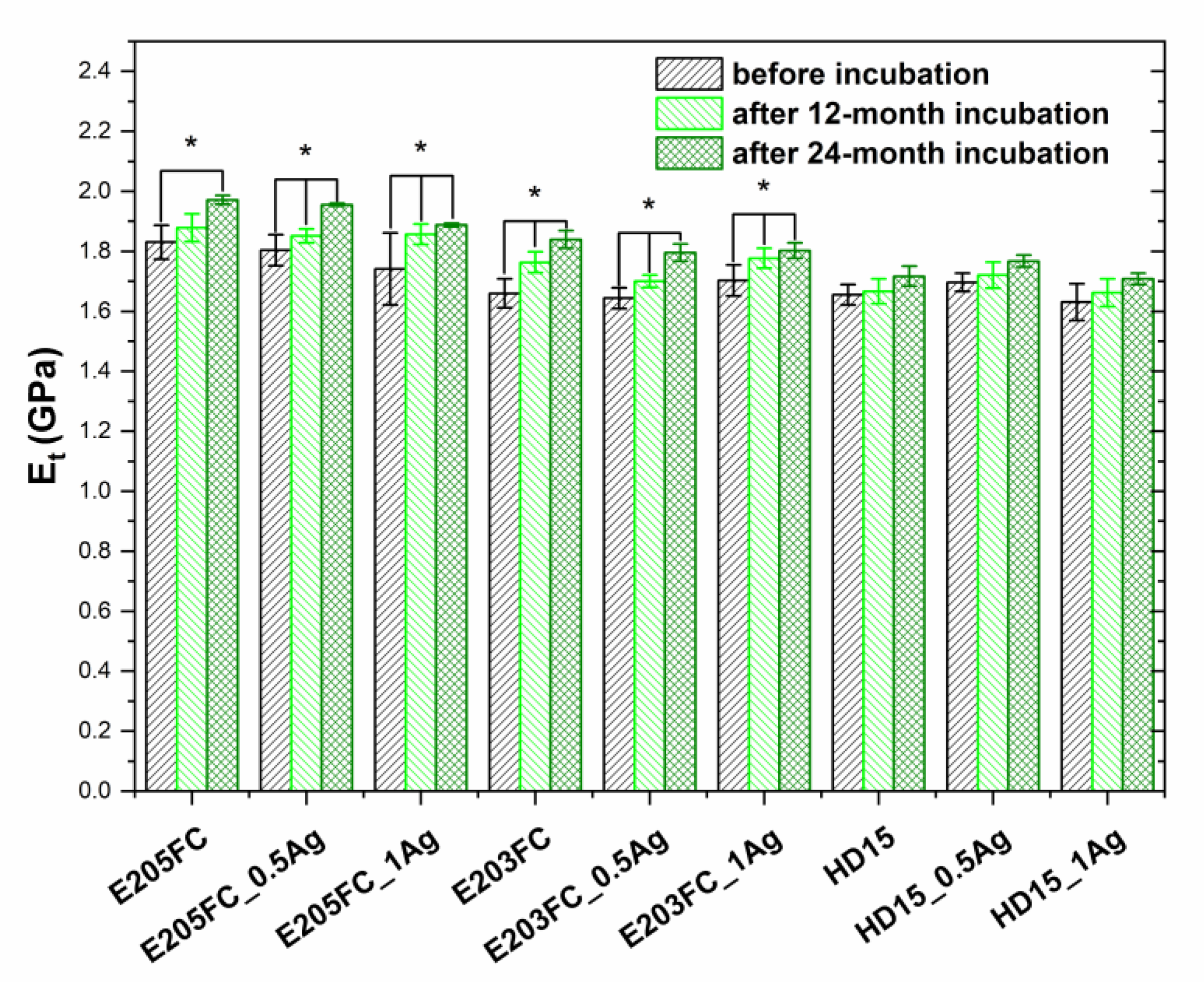
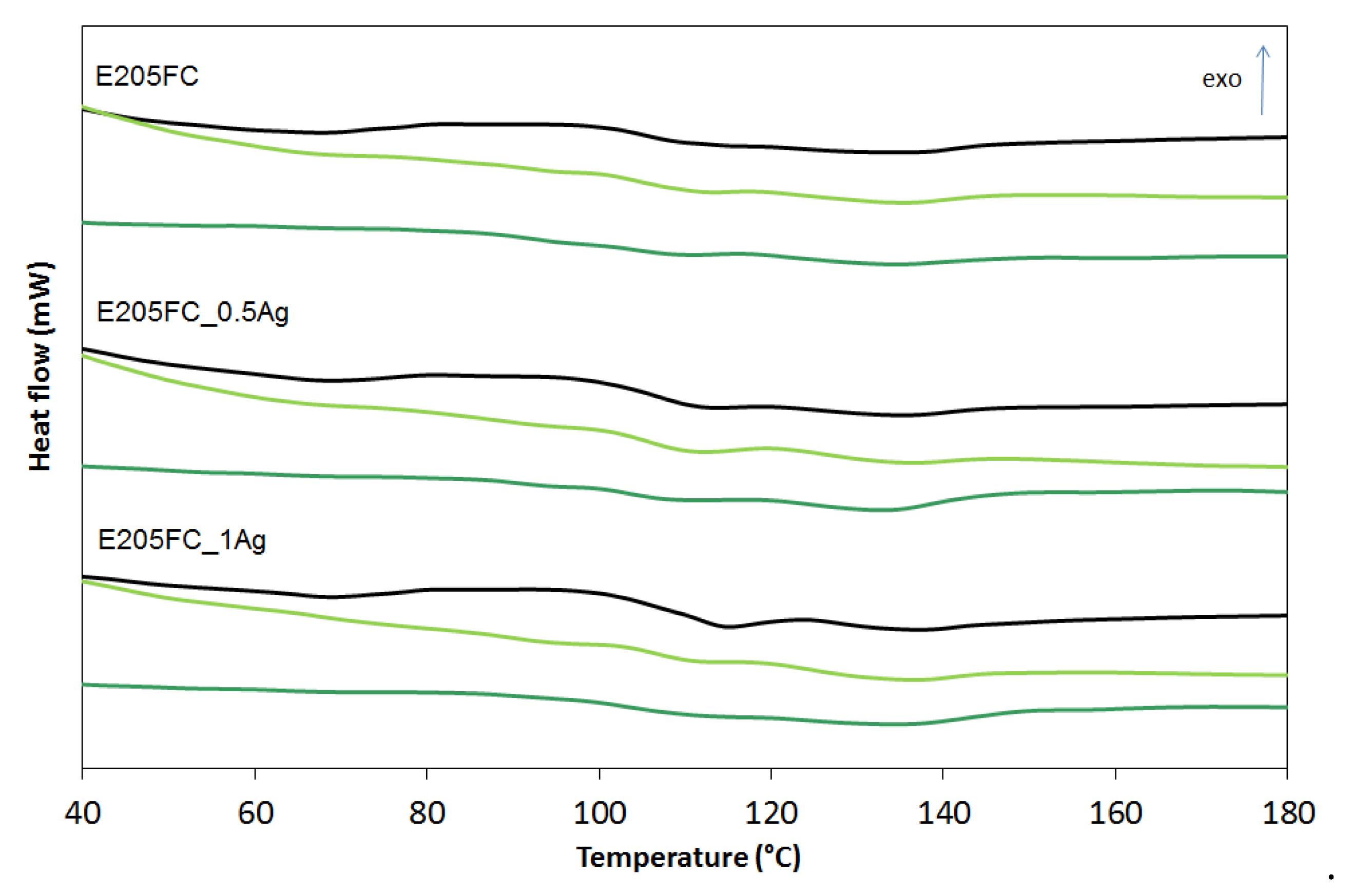

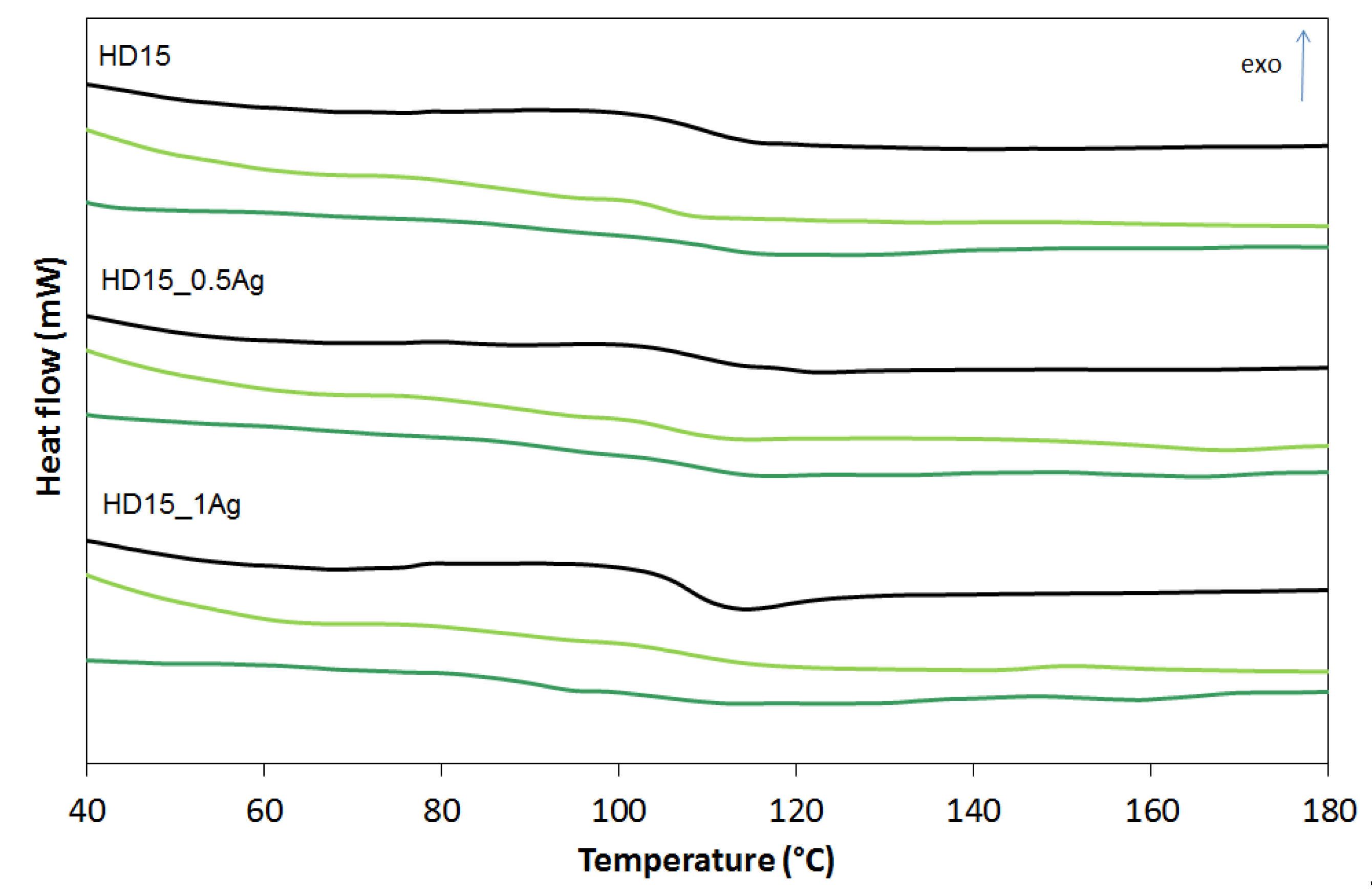
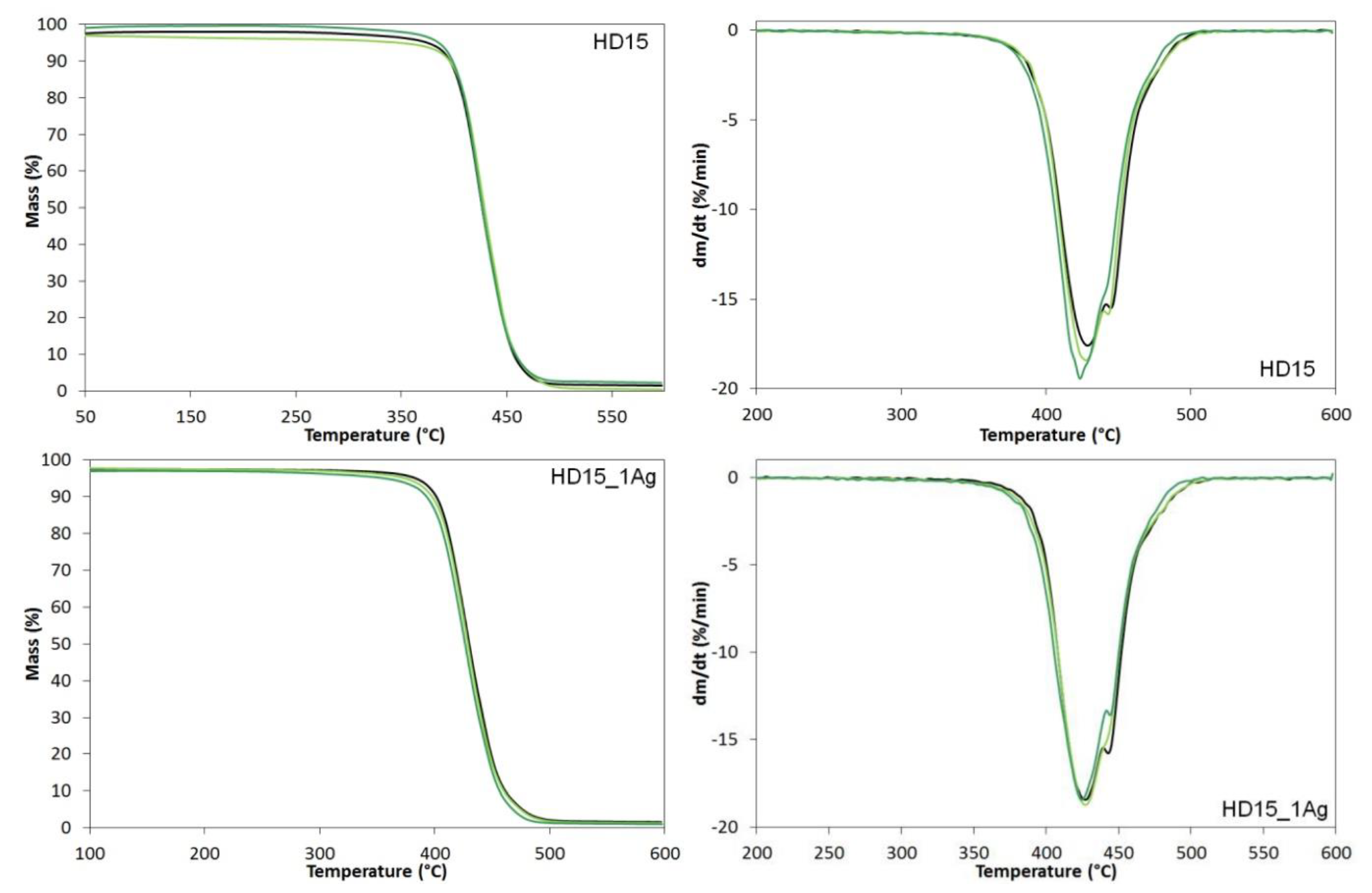
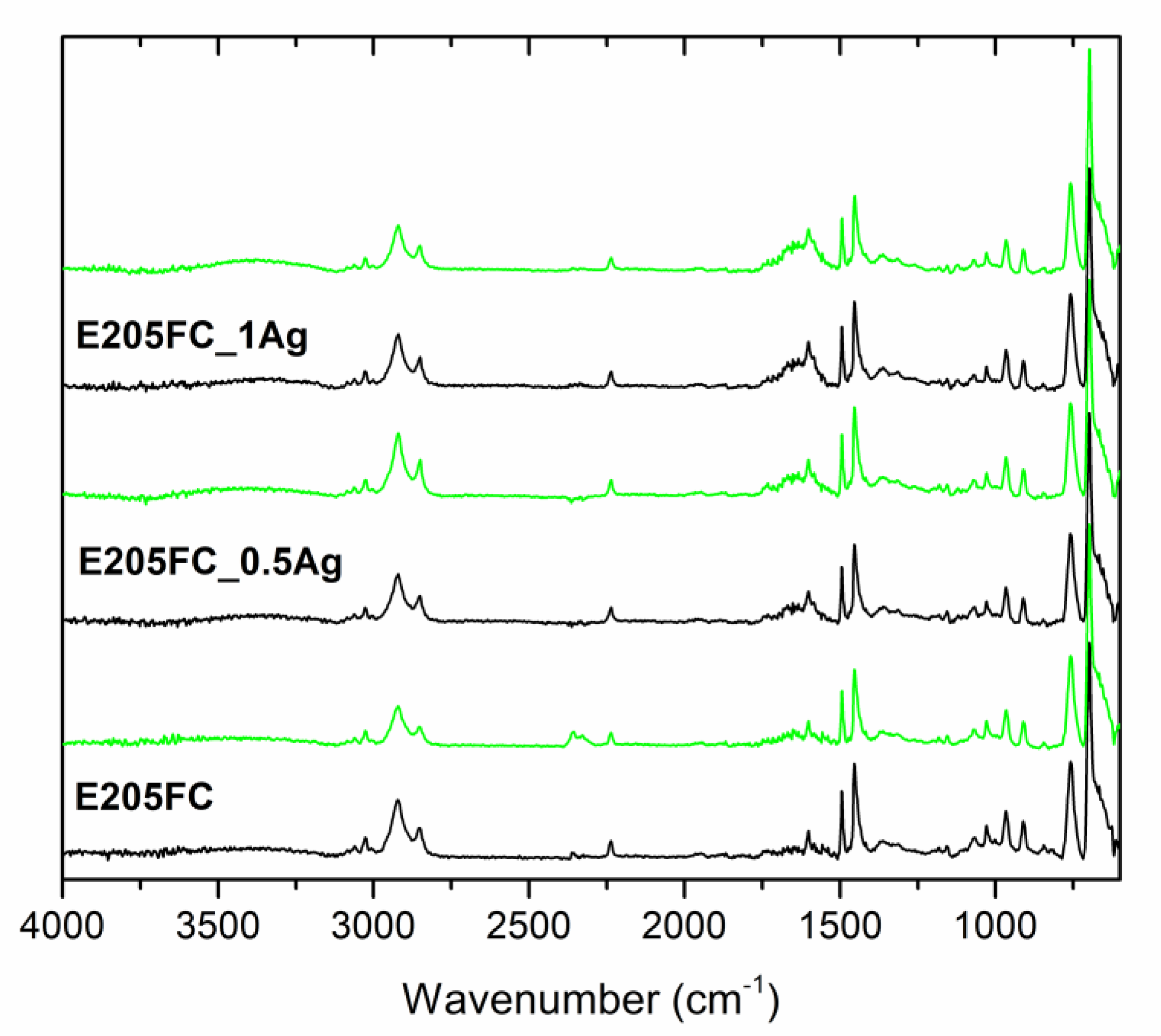
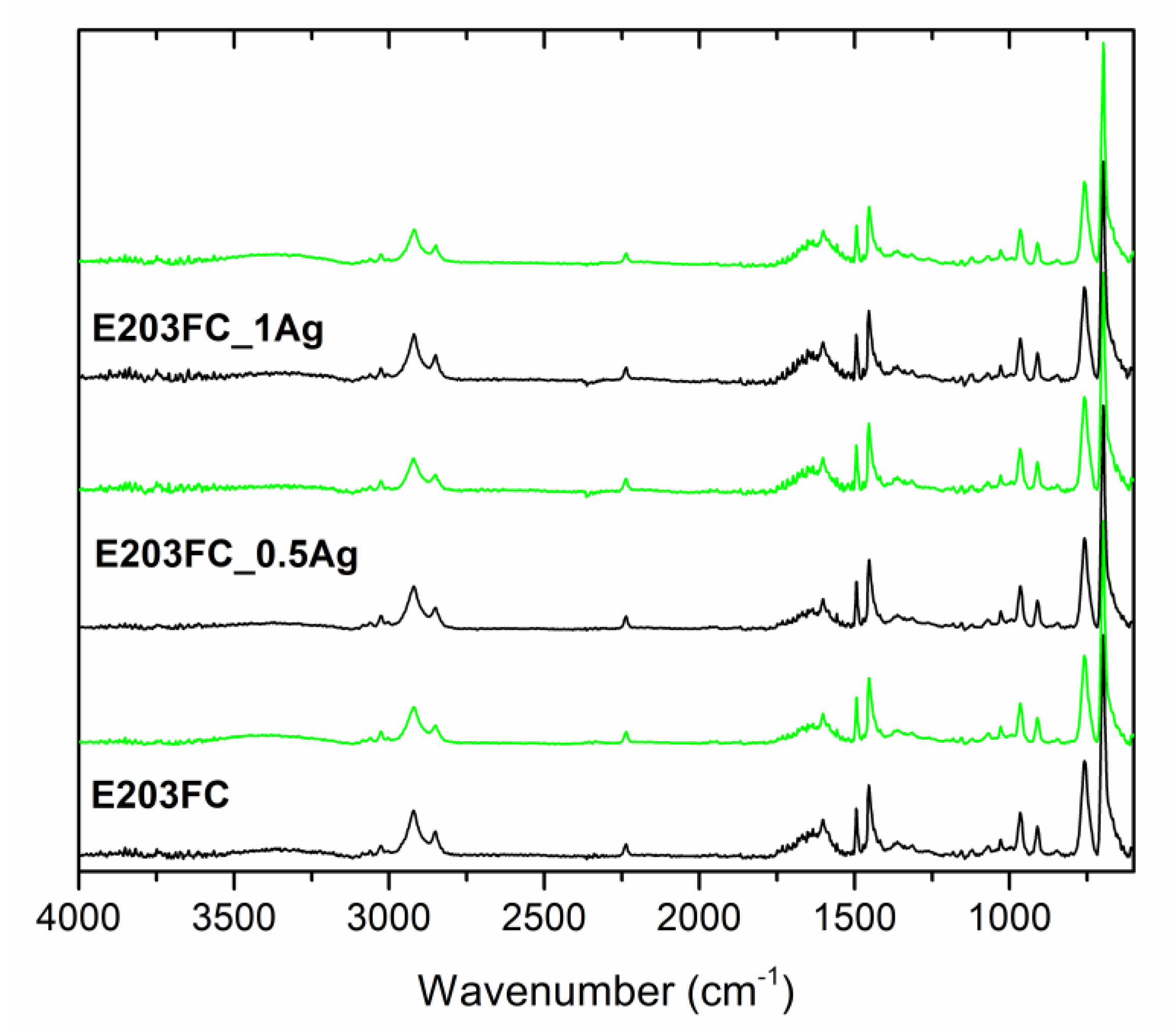
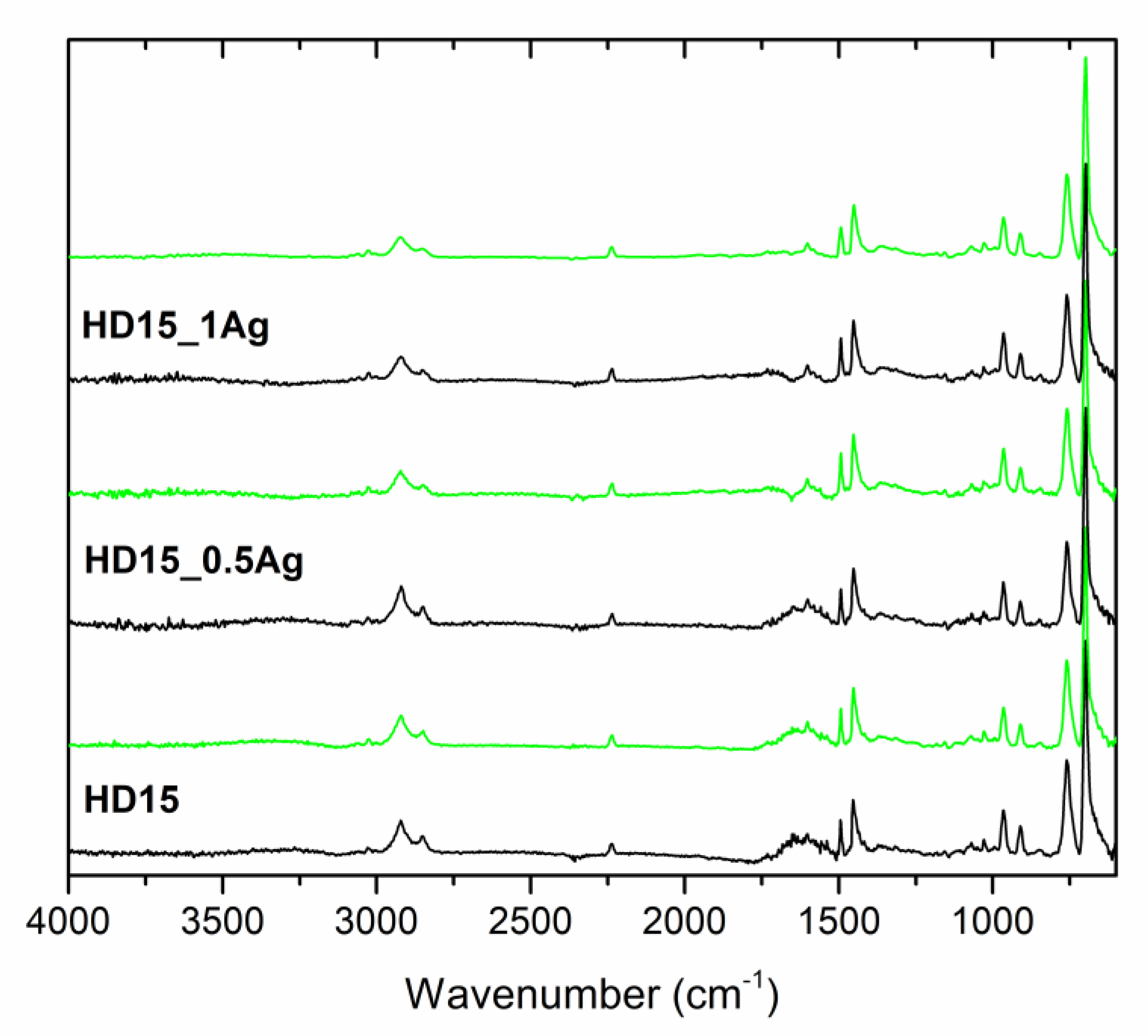
| Sample | Before Incubation | After 12-months Incubation | After 24-Months Incubation | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mechanic. Prop. | DSC | Mechanic. Prop. | DSC | Mechanic. Prop. | DSC | |||||||
| Et (GPa) | σM (MPa) | Tg (°C) | ΔCp [J/gK] | Et (GPa) | σM (MPa) | Tg (°C) | ΔCp [J/gK] | Et (GPa) | σM (MPa) | Tg (°C) | ΔCp [J/gK] | |
| E205FC | 1.83 ± 0.06 | 51.0 ± 0.9 | 105 | 0.260 | 1.88 ± 0.05 | 58.7 ± 0.7 | 104 | 0.216 | 1.97 ± 0.02 | 59.5 ± 1.4 | 104 | 0.256 |
| E205FC_0.5Ag | 1.80 ± 0.05 | 50.9 ± 1.1 | 107 | 0.384 | 1.85 ± 0.02 | 58.4 ± 1.9 | 105 | 0.237 | 1.96 ± 0.01 | 59.0 ± 1.3 | 104 | 0.158 |
| E205FC_1Ag | 1.74 ± 0.12 | 50.9 ± 1.4 | 112 | 0.520 | 1.86 ± 0.03 | 57.8 ± 0.8 | 107 | 0.186 | 1.89 ± 0.01 | 58.7 ± 1.2 | 103 | 0.144 |
| E203FC | 1.66 ± 0.05 | 48.9 ± 1.2 | 106 | 0.324 | 1.76 ± 0.03 | 54.4 ± 1.5 | 107 | 0.291 | 1.84 ± 0.03 | 55.5 ± 1.1 | 103 | 0.330 |
| E203FC_0.5Ag | 1.64 ± 0.04 | 48.7 ± 1.3 | 115 | 0.365 | 1.70 ± 0.02 | 54.3 ± 0.4 | 105 | 0.380 | 1.80 ± 0.03 | 55.5 ± 0.7 | 104 | 0.126 |
| E203FC_1Ag | 1.70 ± 0.05 | 48.6 ± 1.6 | 109 | 0.421 | 1.78 ± 0.03 | 56.0 ± 1.6 | 104 | 0.134 | 1.80 ± 0.03 | 56.7 ± 0.5 | 104 | 0.053 |
| HD15 | 1.65 ± 0.03 | 49.1 ± 2.1 | 110 | 0.385 | 1.67 ± 0.04 | 50.8 ± 1.5 | 105 | 0.196 | 1.72 ± 0.03 | 53.2 ± 1.0 | 111 | 0.138 |
| HD15_0.5Ag | 1.70 ± 0.03 | 51.2 ± 2.4 | 109 | 0.373 | 1.72 ± 0.04 | 51.5 ± 1.5 | 105 | 0.217 | 1.78 ± 0.02 | 55.0 ± 0.9 | 109 | 0.146 |
| HD15_1Ag | 1.63 ± 0.06 | 49.1 ± 1.7 | 108 | 0.433 | 1.66 ± 0.05 | 51.2 ± 0.7 | 107 | 0.202 | 1.71 ± 0.02 | 53.3 ± 1.0 | 110 | 0.154 |
| Sample | T1% [°C] | T3% [°C] | T5% [°C] | T10% [°C] | T50% [°C] | TDTGmax [°C] | Char Residue [%] |
|---|---|---|---|---|---|---|---|
| After Preparation | |||||||
| E205FC | 346 | 380 | 390 | 402 | 430 | 426.0 | 9.61 |
| E205FC_0.5Ag | 348 | 381 | 391 | 402 | 428 | 424.5 | 2.05 |
| E205FC_1Ag | 346 | 384 | 392 | 401 | 427 | 423.2 | 6.29 |
| E203FC | 340 | 377 | 388 | 400 | 429 | 426.5 | 2.23 |
| E203FC_0.5Ag | 341 | 380 | 390 | 401 | 430 | 426.3 445.5 | 2.25 |
| E203FC_1Ag | 348 | 380 | 390 | 402 | 430 | 426.6 | 2.65 |
| HD15 | 328 | 376 | 390 | 403 | 432 | 428.7 444.6 | 2.14 |
| HD15_0.5Ag | 339 | 382 | 392 | 404 | 431 | 427.3 443.5 | 1.92 |
| HD15_1Ag | 357 | 387 | 396 | 405 | 431 | 427.1 442.8 | 2.36 |
| After 12 Months of Incubation | |||||||
| E205FC | 343 | 380 | 390 | 401 | 428 | 421.4 | 0.51 |
| E205FC_0.5Ag | 340 | 379 | 390 | 402 | 428 | 424.5 | 1.59 |
| E205FC_1Ag | 333 | 378 | 389 | 402 | 427 | 422.1 | 7.06 |
| E203FC | 319 | 377 | 389 | 401 | 429 | 426.6 | 0.11 |
| E203FC_0.5Ag | 340 | 378 | 389 | 401 | 430 | 428.2 | 4.06 |
| E203FC_1Ag | 336 | 379 | 389 | 401 | 430 | 426.6 | 2.20 |
| HD15 | 316 | 376 | 390 | 403 | 431 | 427.5 442.7 | 3.59 |
| HD15_0.5Ag | 319 | 378 | 391 | 403 | 431 | 425.8 443.5 | 1.87 |
| HD15_1Ag | 323 | 377 | 390 | 403 | 430 | 427.1 | 2.66 |
| After 24 Months of Incubation | |||||||
| E205FC | 344 | 368 | 378 | 394 | 422 | 419.5 | 5.56 |
| E205FC_0.5Ag | 344 | 366 | 378 | 393 | 422 | 420.8 | 9.04 |
| E205FC_1Ag | 344 | 370 | 382 | 396 | 423 | 420.3 | 6.37 |
| E203FC | 336 | 371 | 383 | 396 | 426 | 425.8 | 2.02 |
| E203FC_0.5Ag | 343 | 373 | 384 | 398 | 427 | 424.0 | 0.12 |
| E203FC_1Ag | 317 | 366 | 381 | 362 | 427 | 424.5 | 3.26 |
| HD15 | 324 | 376 | 388 | 400 | 427 | 423.3 | 0.77 |
| HD15_0.5Ag | 326 | 376 | 388 | 401 | 428 | 424.7 | 1.47 |
| HD15_1Ag | 322 | 374 | 387 | 400 | 428 | 424.4 440.0 | 1.04 |
| Band Position | Band Assignment |
|---|---|
| 3200–3000 cm−1 | stretching vibrations of aromatic C–H bonds |
| 3000–2800 cm−1 | stretching vibrations of aliphatic C–H bonds |
| 2237 cm−1 | stretching vibrations of C≡N bonds in acrylonitrile units |
| 1635 cm−1 | stretching vibrations of C=C bonds in butadiene units |
| 1600, 1582, and 1492 cm−1 | stretching vibrations of aromatic ring in styrene units |
| 1452 cm−1 | scissoring vibrations of CH2 groups |
| 967 and 911 cm−1 | deformation of C–H bonds for hydrogen atoms attached to alkenic carbons in butadiene units |
| 756 and 698 cm−1 | bending vibrations of C–H bonds in monosubstituted benzene rings |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ziąbka, M.; Dziadek, M.; Pielichowska, K. Surface and Structural Properties of Medical Acrylonitrile Butadiene Styrene Modified with Silver Nanoparticles. Polymers 2020, 12, 197. https://doi.org/10.3390/polym12010197
Ziąbka M, Dziadek M, Pielichowska K. Surface and Structural Properties of Medical Acrylonitrile Butadiene Styrene Modified with Silver Nanoparticles. Polymers. 2020; 12(1):197. https://doi.org/10.3390/polym12010197
Chicago/Turabian StyleZiąbka, Magdalena, Michał Dziadek, and Kinga Pielichowska. 2020. "Surface and Structural Properties of Medical Acrylonitrile Butadiene Styrene Modified with Silver Nanoparticles" Polymers 12, no. 1: 197. https://doi.org/10.3390/polym12010197
APA StyleZiąbka, M., Dziadek, M., & Pielichowska, K. (2020). Surface and Structural Properties of Medical Acrylonitrile Butadiene Styrene Modified with Silver Nanoparticles. Polymers, 12(1), 197. https://doi.org/10.3390/polym12010197







