Improving the Electrochemical Performance and Stability of Polypyrrole by Polymerizing Ionic Liquids
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
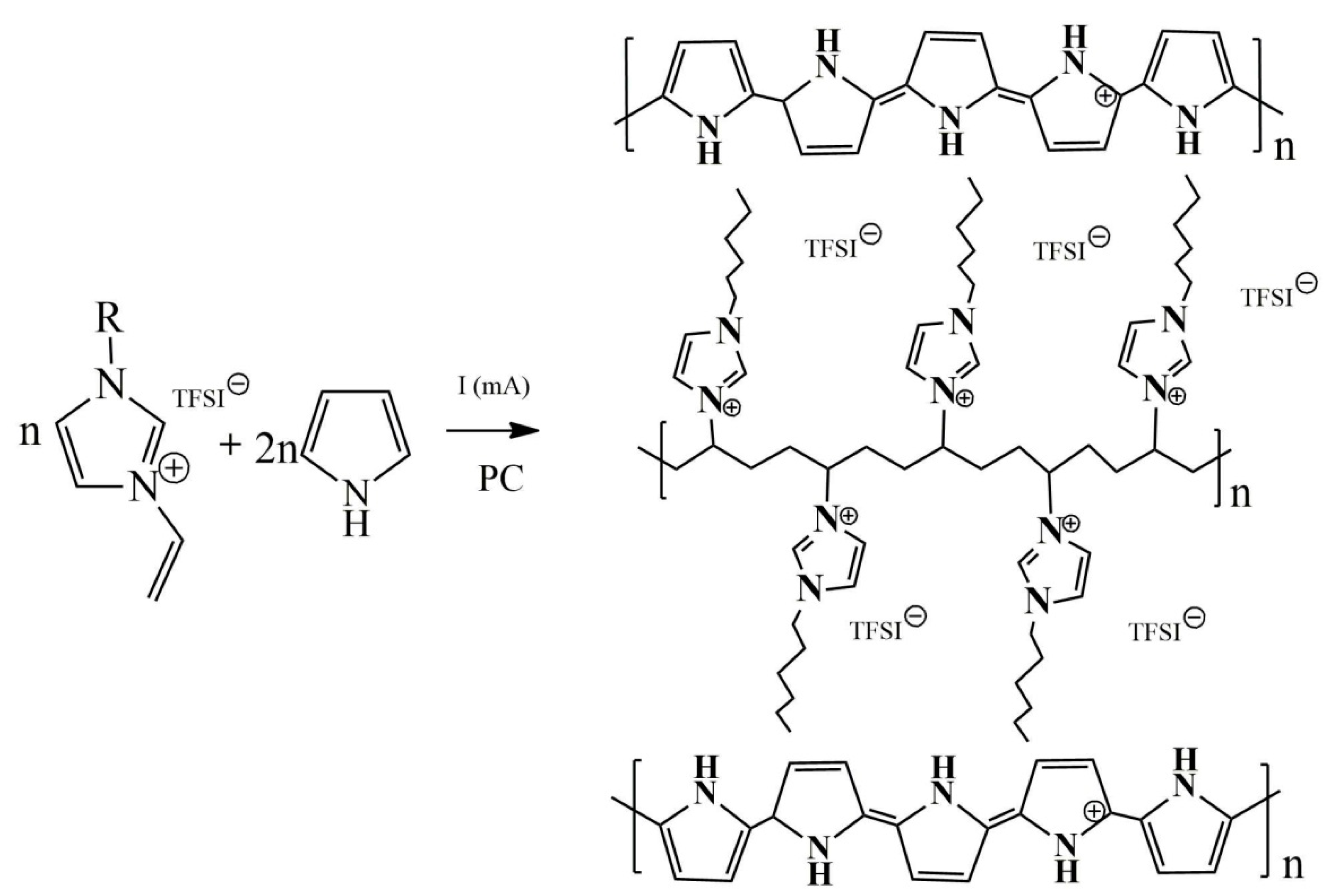
2.2. Electrochemical Synthesis of PPyPIL
2.3. Characterization
3. Results and Discussion
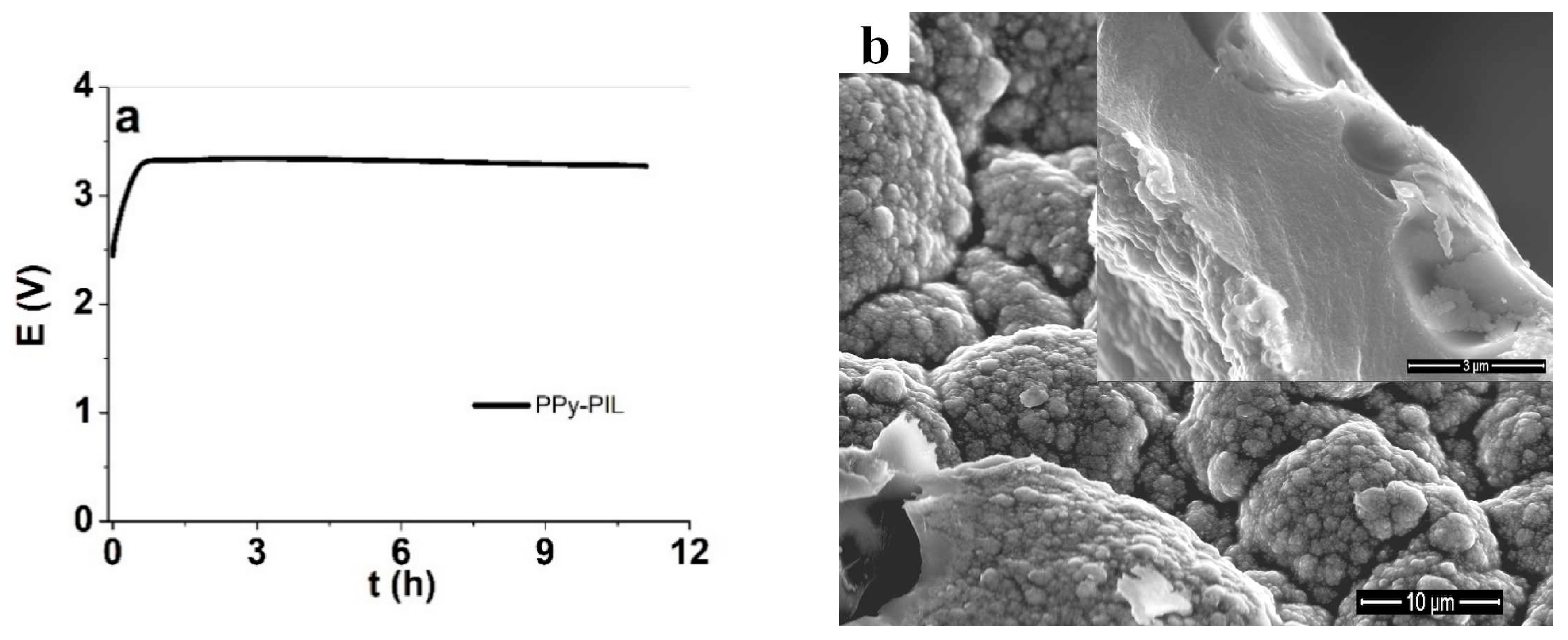
3.1. PPyPIL Films
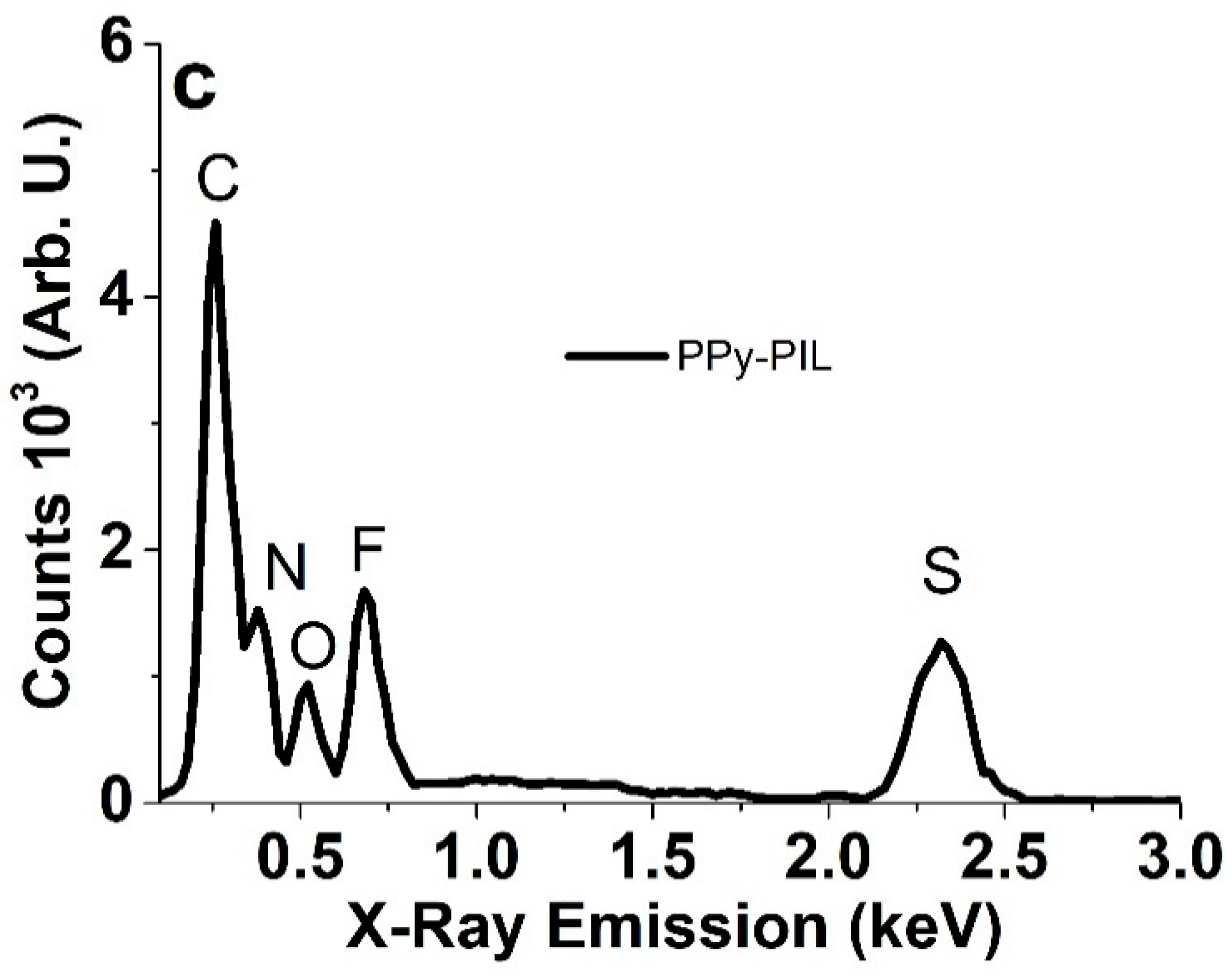
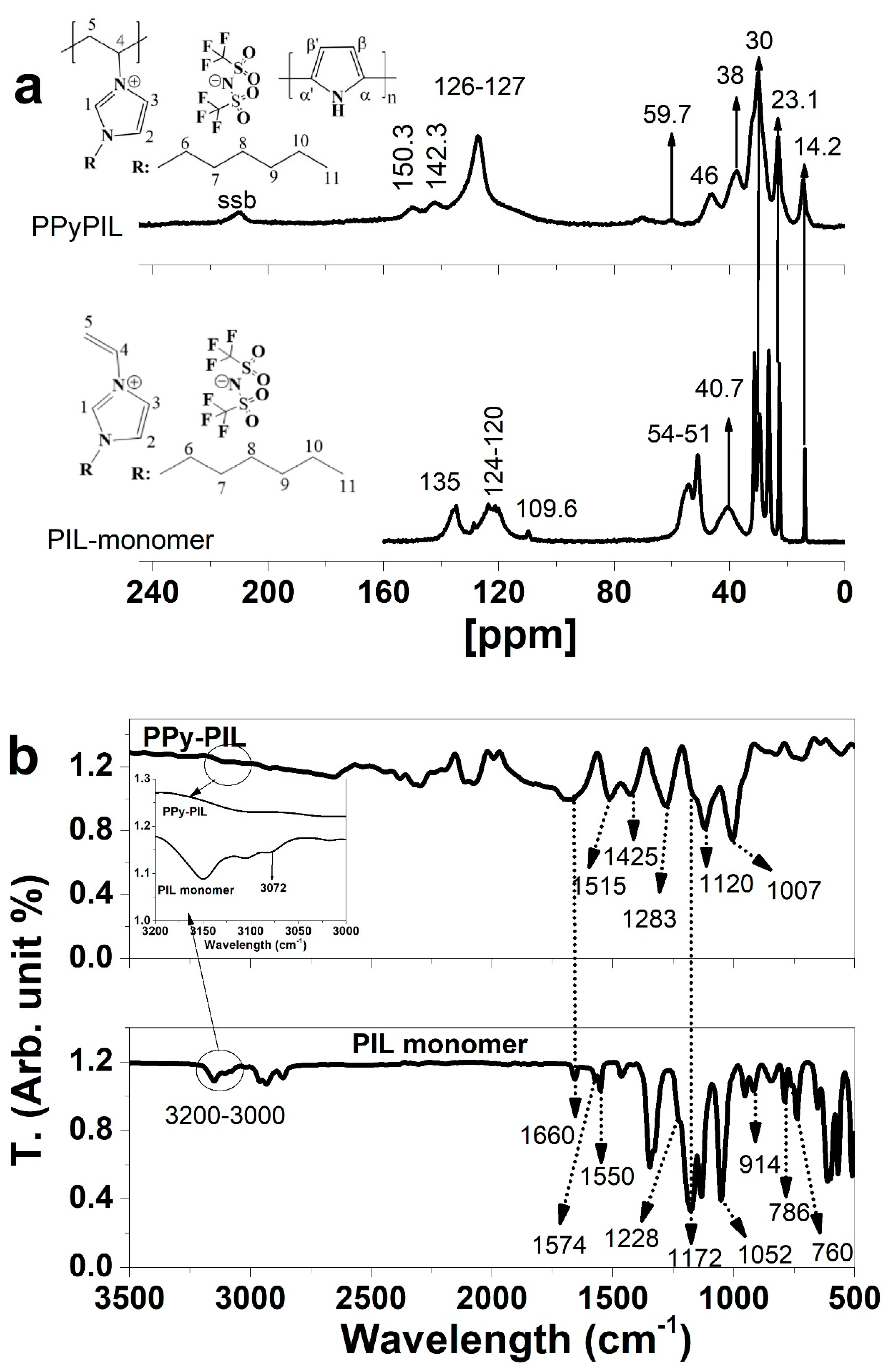
3.2. Structural Analysis of PPyPIL with ssNMR and FTIR
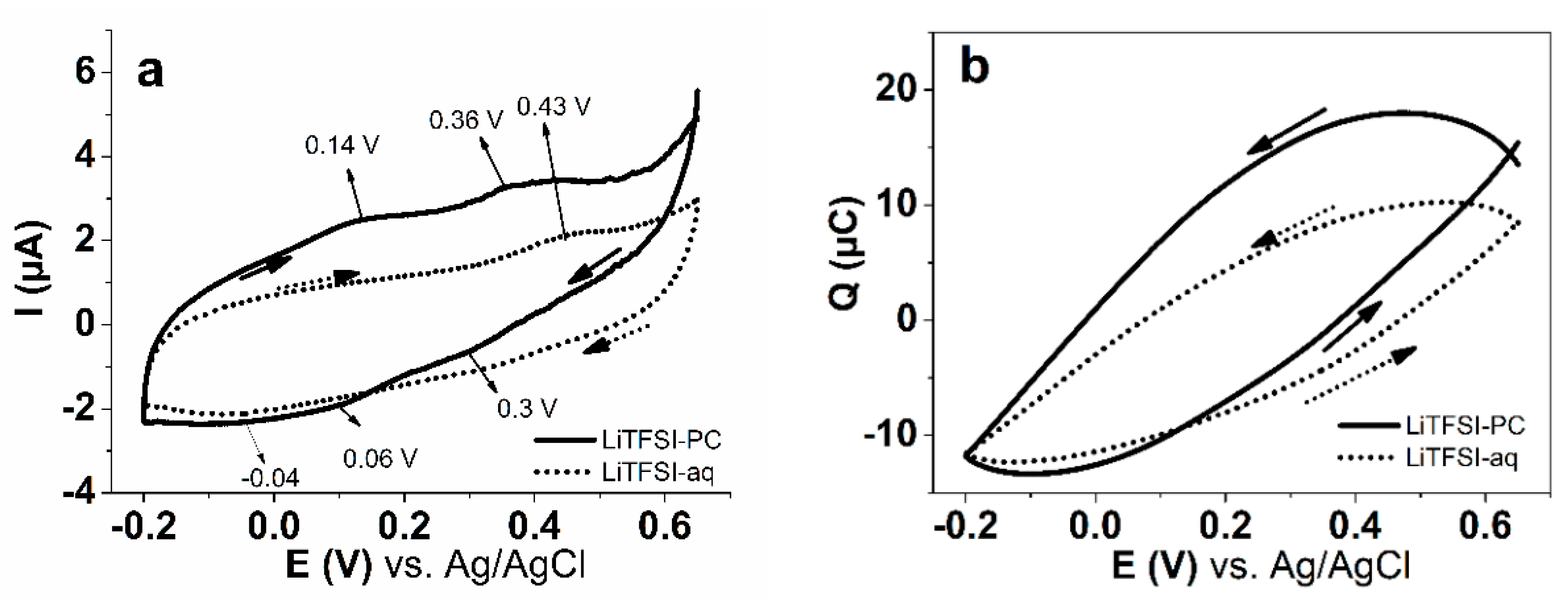
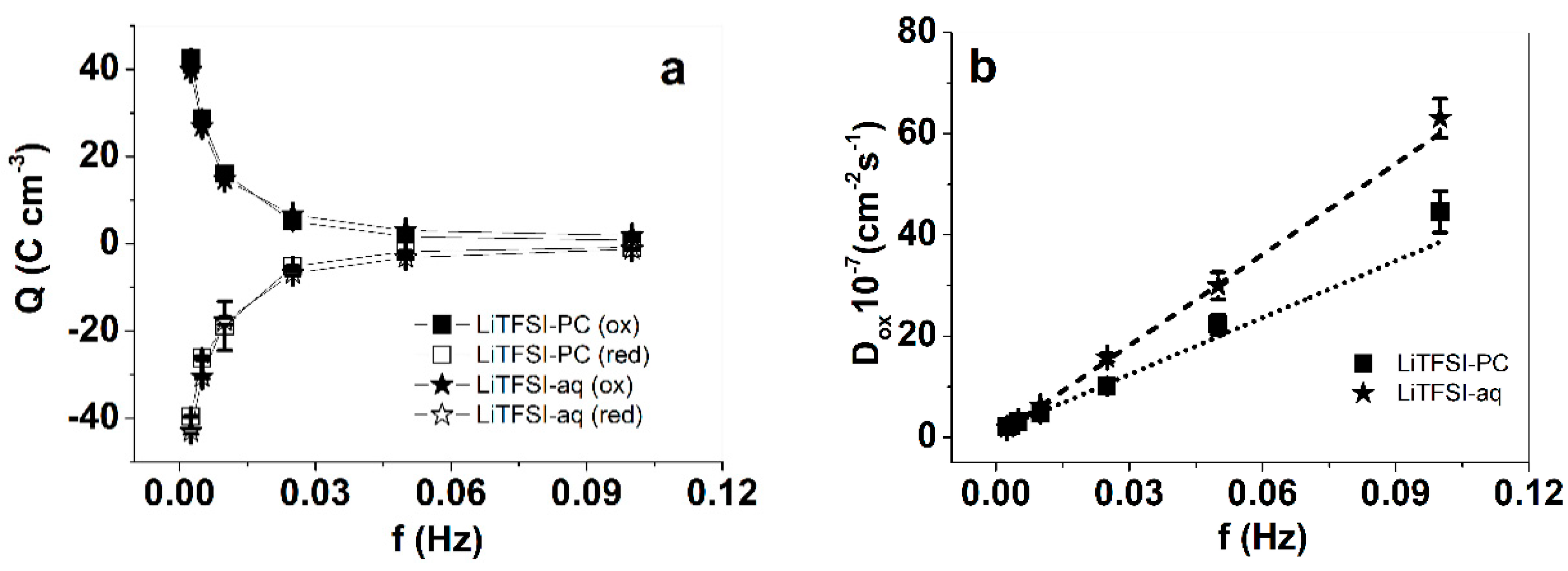
3.3. Modified SICM Measurements of PPyPIL Films
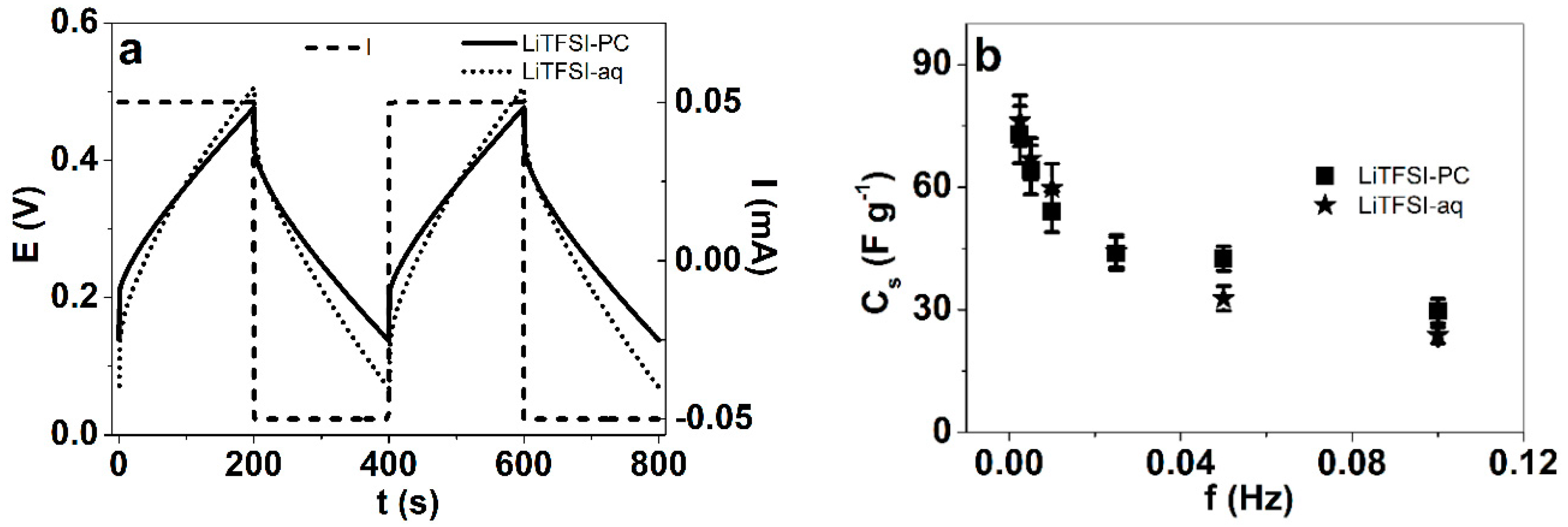
3.4. Potential (Current) Square Wave Step Measurements of PPyPIL Films
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gao, F.; Zhang, N.; Fang, X.; Ma, M. Bioinspired Design of Strong, Tough, and Highly Conductive Polyol-Polypyrrole Composites for Flexible Electronics. ACS Appl. Mater. Interfaces 2017, 9, 5692–5698. [Google Scholar] [CrossRef] [PubMed]
- Entezami, A.A.; Massoumi, B. Artificial muscles, biosensors and drug delivery systems based on conducting polymers: A review. Iran. Polym. J. 2006, 15, 13–30. [Google Scholar]
- Singh, M.; Kathuroju, P.K.; Jampana, N. Polypyrrole based amperometric glucose biosensors. Sens. Actuators B Chem. 2009, 143, 430–443. [Google Scholar] [CrossRef]
- García-Córdova, F.; Valero, L.; Ismail, Y.A.; Otero, T.F. Biomimetic polypyrrole based all three-in-one triple layer sensing actuators exchanging cations. J. Mater. Chem. 2011, 21, 17265–17272. [Google Scholar] [CrossRef]
- Aphale, A.; Maisuria, K.; Mahapatra, M.K.; Santiago, A.; Singh, P.; Patra, P. Hybrid electrodes by in-situ integration of graphene and carbon-nanotubes in polypyrrole for supercapacitors. Sci. Rep. 2015, 5, 14445. [Google Scholar] [CrossRef]
- Song, H.K.; Palmore, G.T.R. Redox-active polypyrrole: Toward polymer-based batteries. Adv. Mater. 2006, 18, 1764–1768. [Google Scholar] [CrossRef]
- Zheng, W.; Alici, G.; Clingan, P.R.; Munro, B.J.; Spinks, G.M.; Steele, J.R.; Wallace, G.G. Polypyrrole stretchable actuators. J. Polym. Sci. Part B Polym. Phys. 2013, 51, 57–63. [Google Scholar] [CrossRef]
- Maziz, A.; Concas, A.; Khaldi, A.; Stålhand, J.; Persson, N.-K.; Jager, E.W.H. Knitting and weaving artificial muscles. Sci. Adv. 2017, 3, 1–12. [Google Scholar] [CrossRef]
- Hara, S.; Zama, T.; Takashima, W.; Kaneto, K. TFSI-doped polypyrrole actuator with 26% strain. J. Mater. Chem. 2004, 14, 1516. [Google Scholar] [CrossRef]
- Bober, P.; Capáková, Z.; Acharya, U.; Zasońska, B.A.; Humpolíček, P.; Hodan, J.; Hromádková, J.; Stejskal, J. Highly conducting and biocompatible polypyrrole/poly(vinyl alcohol) cryogels. Synth. Met. 2019, 252, 122–126. [Google Scholar] [CrossRef]
- Ghanbari, R.; Ghorbani, S.R.; Arabi, H.; Foroughi, J. The charge transport mechanisms in conducting polymer polypyrrole films and fibers. Mater. Res. Express 2018, 5, 1–6. [Google Scholar] [CrossRef]
- Khanh, T.T.; Kesküla, A.; Zondaka, Z.; Harjo, M.; Kivilo, A.; Khorram, M.S.; Tamm, T.; Kiefer, R. Role of polymerization temperature on the performance of polypyrrole/dodecylbenzenesulphonate linear actuators. Synth. Met. 2019, 247, 53–58. [Google Scholar] [CrossRef]
- Hagiwara, T.; Hirasaka, M.; Sato, K.; Yamaura, M. Enhancement of the electrical conductivity of polypyrrole film by stretching: Influence of polymerization conditions. Synth. Met. 1990, 36, 241–252. [Google Scholar] [CrossRef]
- Maddison, D.S.; Unsworth, J. Optimization of synthesis conditions of polypyrrole from aqueous solutions. Synth. Met. 1989, 30, 47–55. [Google Scholar] [CrossRef]
- Kupila, E.L.; Kankare, J. Influence of electrode pretreatment, counter anions and additives on the electropolymerization of pyrrole in aqueous solutions. Synth. Met. 1995, 74, 241–249. [Google Scholar] [CrossRef]
- Aydemir, N.; Kilmartin, P.A.; Travas-Sejdic, J.; Kesküla, A.; Peikolainen, A.-L.; Parcell, J.; Harjo, M.; Aabloo, A.; Kiefer, R. Electrolyte and solvent effects in PPy/DBS linear actuators. Sens. Actuators B Chem. 2015, 216, 24–32. [Google Scholar] [CrossRef]
- Stankoviç, R.; Pavloviç, O.; Vojnoviç, M.; Jovanoviç, S. The effects of preparation conditions on the properties of electrochemically synthesized thick films of polypyrrole. Eur. Polym. J. 1994, 30, 385–393. [Google Scholar] [CrossRef]
- Maw, S.; Smela, E.; Yoshida, K.; Stein, R.B. Effects of monomer and electrolyte concentrations on actuation of PPy(DBS) bilayers. Synth. Met. 2005, 155, 18–26. [Google Scholar] [CrossRef]
- Demoustier-Champagne, S.; Stavaux, P.-Y. Effect of Electrolyte Concentration and Nature on the Morphology and the Electrical Properties of Electropolymerized Polypyrrole Nanotubules. Chem. Mater. 1999, 11, 829–834. [Google Scholar] [CrossRef]
- Kiefer, R.; Kesküla, A.; Martinez, J.G.; Anbarjafari, G.; Torop, J.; Otero, T.F. Interpenetrated triple polymeric layer as electrochemomechanical actuator: Solvent influence and diffusion coefficient of counterions. Electrochim. Acta 2017, 230, 461–469. [Google Scholar] [CrossRef]
- Põldsalu, I.; Rohtlaid, K.; Nguyen, T.M.G.; Plesse, C.; Vidal, F.; Khorram, M.S.; Peikolainen, A.L.; Tamm, T.; Kiefer, R. Thin ink-jet printed trilayer actuators composed of PEDOT:PSS on interpenetrating polymer networks. Sens. Actuators B Chem. 2018, 258, 1072–1079. [Google Scholar] [CrossRef]
- Ebrahimi, S.; Nasiri, M.; Agbolaghi, S.; Abbasi, F.; Sarvari, R. A focus on polystyrene tacticity in synthesized conductive PEDOT:PSS thin films. J. Polym. Res. 2018, 25, 18–20. [Google Scholar] [CrossRef]
- Ohno, H. Molten salt type polymer electrolytes. Electrochim. Acta 2001, 46, 1407–1411. [Google Scholar] [CrossRef]
- Ohno, H.; Yoshizawa, M.; Ogihara, W. Development of new class of ion conductive polymers based on ionic liquids. Electrochim. Acta 2004, 50, 255–261. [Google Scholar] [CrossRef]
- Mecerreyes, D. Polymeric ionic liquids: Broadening the properties and applications of polyelectrolytes. Prog. Polym. Sci. 2011, 36, 1629–1648. [Google Scholar] [CrossRef]
- Winterton, N. Solubilization of polymers by ionic liquids. J. Mater. Chem. 2006, 16, 4281–4293. [Google Scholar] [CrossRef]
- Green, O.; Grubjesic, S.; Lee, S.; Firestone, M.A. The design of polymeric ionic liquids for the preparation of functional materials. Polym. Rev. 2009, 49, 339–360. [Google Scholar] [CrossRef]
- Devasurendra, A.M.; Zhang, C.; Young, J.A.; Tillekeratne, L.M.V.; Anderson, J.L.; Kirchho, J.R. Electropolymerized Pyrrole-Based Conductive Polymeric Ionic Liquids and Their Application for Solid-Phase Microextraction. ACS Appl. Mater. Interfaces 2017, 9, 24955–24963. [Google Scholar] [CrossRef]
- Marcilla, R.; Ochoteco, E.; Pozo-Gonzalo, C.; Grande, H.; Pomposo, J.A.; Mecerreyes, D. New organic dispersions of conducting polymers using polymeric ionic liquids as stabilizers. Macromol. Rapid Commun. 2005, 26, 1122–1126. [Google Scholar] [CrossRef]
- Ponkratov, D.O.; Lozinskaya, E.I.; Vlasov, P.S.; Aubert, P.H.; Plesse, C.; Vidal, F.; Vygodskii, Y.S.; Shaplov, A.S. Synthesis of novel families of conductive cationic poly(ionic liquid)s and their application in all-polymer flexible pseudo-supercapacitors. Electrochim. Acta 2018, 281, 777–788. [Google Scholar] [CrossRef]
- Mao, H.; Liang, J.; Ji, C.; Zhang, H.; Pei, Q.; Zhang, Y.; Zhang, Y.; Hisaeda, Y.; Song, X.M. Poly(zwitterionic liquids) functionalized polypyrrole/graphene oxide nanosheets for electrochemically detecting dopamine at low concentration. Mater. Sci. Eng. C 2016, 65, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Mao, H.; Zhang, H.; Jiang, W.; Liang, J.; Sun, Y.; Zhang, Y.; Wu, Q.; Zhang, G.; Song, X.M. Poly(ionic liquid) functionalized polypyrrole nanotubes supported gold nanoparticles: An efficient electrochemical sensor to detect epinephrine. Mater. Sci. Eng. C 2017, 75, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Laslau, C.; Williams, D.E.; Travas-Sejdic, J. Progress in Polymer Science The application of nanopipettes to conducting polymer fabrication, imaging and electrochemical characterization. Prog. Polym. Sci. 2012, 37, 1177–1191. [Google Scholar] [CrossRef]
- Feng, X.; Gao, C.; Guo, Z.; Zhou, Y.; Wang, J. Pore structure controllable synthesis of mesoporous poly(ionic liquid)s by copolymerization of alkylvinylimidazolium salts and divinylbenzene. RSC Adv. 2014, 4, 23389–23395. [Google Scholar] [CrossRef]
- Wasserscheid, P.; Welton, T. (Eds.) Ionic Liquids in Synthesis; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2002; ISBN 3527305157. [Google Scholar]
- Otero, T.F.; Martinez, J.G. Activation energy for polypyrrole oxidation: Film thickness influence. J. Solid State Electrochem. 2011, 15, 1169–1178. [Google Scholar] [CrossRef]
- Wu, T.Y.; Hao, L.; Chen, P.R.; Liao, J.W. Ionic conductivity and transporting properties in LiTFSI-Doped Bis(trifluoromethanesulfonyl)imide-based ionic liquid electrolyte. Int. J. Electrochem. Sci. 2013, 8, 2606–2624. [Google Scholar]
- Sadki, S.; Schottland, P.; Brodiec, N.; Sabouraud, G. The mechanisms of pyrrole electropolymerization. Chem. Soc. Rev. 2000, 29, 283–293. [Google Scholar]
- Grzeszczuk, M.; Ozsakarya, R. Surface morphology and corresponding electrochemistry of polypyrrole films electrodeposited using a water miscible ionic liquid. RSC Adv. 2014, 4, 22214–22223. [Google Scholar] [CrossRef]
- Ogihara, W.; Washiro, S.; Nakajima, H.; Ohno, H. Effect of cation structure on the electrochemical and thermal properties of ion conductive polymers obtained from polymerizable ionic liquids. Electrochim. Acta 2006, 51, 2614–2619. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, B.; Elageed, E.H.M.; Qin, L.; Ni, B.; Xiuli, L.; Gao, G. Swelling Poly(ionic liquid)s: Synthesis and Application as Quasi-Homogeneous Catalysts in the Reaction of Ethylene Carbonate with Aniline. ACS Macro Lett. 2016, 5, 435–438. [Google Scholar] [CrossRef]
- Gao, C.; Chen, G.; Wang, X.; Li, J.; Zhou, Y.; Wang, J. A hierarchical meso-macroporous poly(ionic liquid) monolith derived from a single soft template. Chem. Commun. 2015, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, P.M.; Grande, H.J.; Cortazar, M.; Alberdi, J.M.; Areizaga, J.; Pomposo, J.A. Structure-conductivity relationships in chemical polypyrroles of low, medium and high conductivity. Synth. Met. 2006, 156, 420–425. [Google Scholar] [CrossRef]
- Forsyth, M.; Truong, V.; Smith, M.E. Structural characterization of conducting polypyrrole using 13C cross-polarization/magic-angle spinning solid-state nuclear magnetic resonance spectroscopy. Polymer 1994, 35, 1593–1601. [Google Scholar] [CrossRef]
- Hagiwara, T.; Yamaura, M.; Iwata, K. Structural analysis of deprotonated polyaniline by solid-state 13C N.M.R. Synth. Met. 1988, 26, 195–201. [Google Scholar] [CrossRef]
- Martina, S.; Enkelmann, V.; Wegner, G.; Zotti, G.; Zerbi, G. Synthesis and electrochemical and spectroscopical STUDIES of 2.5-pyrrole oligomers and well-defined short-chain poly(2.5-pyrrole). Synth. Met. 1993, 57, 1096–1101. [Google Scholar] [CrossRef]
- Rahman, T.M.; Barikbin, Z.; Badruddoza, A.Z.M.; Doyle, P.S.; Khan, S.A. Monodisperse Polymeric Ionic Liquid Microgel Beads with Multiple Chemically Switchable Functionalities. Langmuir 2013, 29, 9535–9543. [Google Scholar] [CrossRef]
- Wang, X.; Xing, L.; Shu, Y.; Chen, X.; Wang, J. Novel Polymeric Ionic Liquid Microspheres with High Exchange Capacity for Fast Extraction of Plasmid DNA. Anal. Chim. Acta 2014, 837, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wang, L.; Yang, B.; Du, T.; Zhang, Y. Facile preparation of polymer electrolytes based on the polymerized ionic liquid poly ((4-vinylbenzyl) trimethylammonium bis (trifluoromethanesulfonylimide)) for lithium secondary batteries. Electrochim. Acta 2014, 123, 296–302. [Google Scholar] [CrossRef]
- Lamprakopoulos, S.; Yfantis, D.; Yfantis, A.; Schmeisser, D.; Anastassopoulou, J.; Theophanides, T. An FTIR study of the role of H2O and D2O in the aging mechanism of conductive polypyrroles. Synth. Met. 2004, 144, 229–234. [Google Scholar] [CrossRef]
- Hosono, K.; Matsubara, I.; Murayama, N.; Shin, W.; Izu, N.; Kanzaki, S. Structure and properties of plasma polymerized and 4-ethylbenzenesulfonic acid-doped polypyrrole films. Thin Solid Films 2003, 441, 72–75. [Google Scholar] [CrossRef]
- Chaban, V. Solvation of the fluorine containing anions and their lithium salts in propylene carbonate and dimethoxyethane. J. Mol. Model. 2015, 21, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Valero, L.; Otero, T.F.; Martinez, J.G.; Martínez, J.G. Exchanged Cations and Water during Reactions in Polypyrrole Macroions from Artificial Muscles. ChemPhysChem 2014, 15, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Khadka, R.; Zondaka, Z.; Kesküla, A.; Safaei Khorram, M.; Thien Khanh, T.; Tamm, T.; Travas-Sejdic, J.; Kiefer, R.; Minh City, C. Influence of solvent on linear polypyrrole-polyethylene oxide actuators. J. Appl. Polym. Sci. 2018, 46831, 1–7. [Google Scholar] [CrossRef]
- Kiefer, R.; Kilmartin, P.A.; Bowmaker, G.A.; Travas-Sejdic, J. Electrochemomechanical behaviour of free standing PEDOT films in organic and aqueous electrolytes. SPIE Smart Struct. Mater. Nondestruct. Eval. Health Monit. 2008, 6927, 692720. [Google Scholar]
- Põldsalu, I.; Harjo, M.; Tamm, T.; Uibu, M.; Peikolainen, A.L.; Kiefer, R. Inkjet-printed hybrid conducting polymer-activated carbon aerogel linear actuators driven in an organic electrolyte. Sens. Actuators B Chem. 2017, 250, 44–51. [Google Scholar] [CrossRef]
- Zhang, Y.; Feng, H.; Wu, X.; Wang, L.; Zhang, A.; Xia, T.; Dong, H.; Li, X.; Zhang, L. Progress of electrochemical capacitor electrode materials: A review. Int. J. Hydrogen Energy 2009, 34, 4889–4899. [Google Scholar] [CrossRef]
- Zondaka, Z.; Kesküla, A.; Tamm, T.; Kiefer, R. Polypyrrole linear actuation tuned by phosphotungstic acid. Sens. Actuators B Chem. 2017, 247, 742–748. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kesküla, A.; Heinmaa, I.; Tamm, T.; Aydemir, N.; Travas-Sejdic, J.; Peikolainen, A.-L.; Kiefer, R. Improving the Electrochemical Performance and Stability of Polypyrrole by Polymerizing Ionic Liquids. Polymers 2020, 12, 136. https://doi.org/10.3390/polym12010136
Kesküla A, Heinmaa I, Tamm T, Aydemir N, Travas-Sejdic J, Peikolainen A-L, Kiefer R. Improving the Electrochemical Performance and Stability of Polypyrrole by Polymerizing Ionic Liquids. Polymers. 2020; 12(1):136. https://doi.org/10.3390/polym12010136
Chicago/Turabian StyleKesküla, Arko, Ivo Heinmaa, Tarmo Tamm, Nihan Aydemir, Jadranka Travas-Sejdic, Anna-Liisa Peikolainen, and Rudolf Kiefer. 2020. "Improving the Electrochemical Performance and Stability of Polypyrrole by Polymerizing Ionic Liquids" Polymers 12, no. 1: 136. https://doi.org/10.3390/polym12010136
APA StyleKesküla, A., Heinmaa, I., Tamm, T., Aydemir, N., Travas-Sejdic, J., Peikolainen, A.-L., & Kiefer, R. (2020). Improving the Electrochemical Performance and Stability of Polypyrrole by Polymerizing Ionic Liquids. Polymers, 12(1), 136. https://doi.org/10.3390/polym12010136







