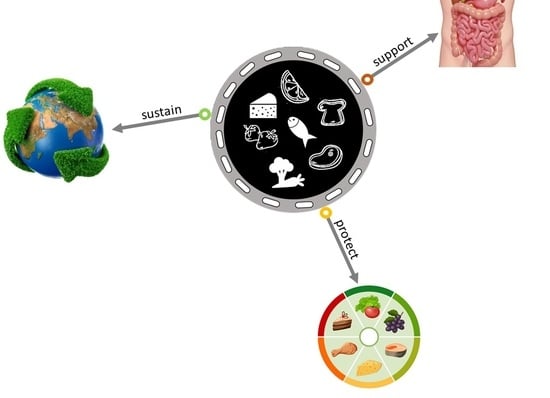
Edible Films and Coatings Functionalization by Probiotic Incorporation: A Review
Abstract
1. Introduction
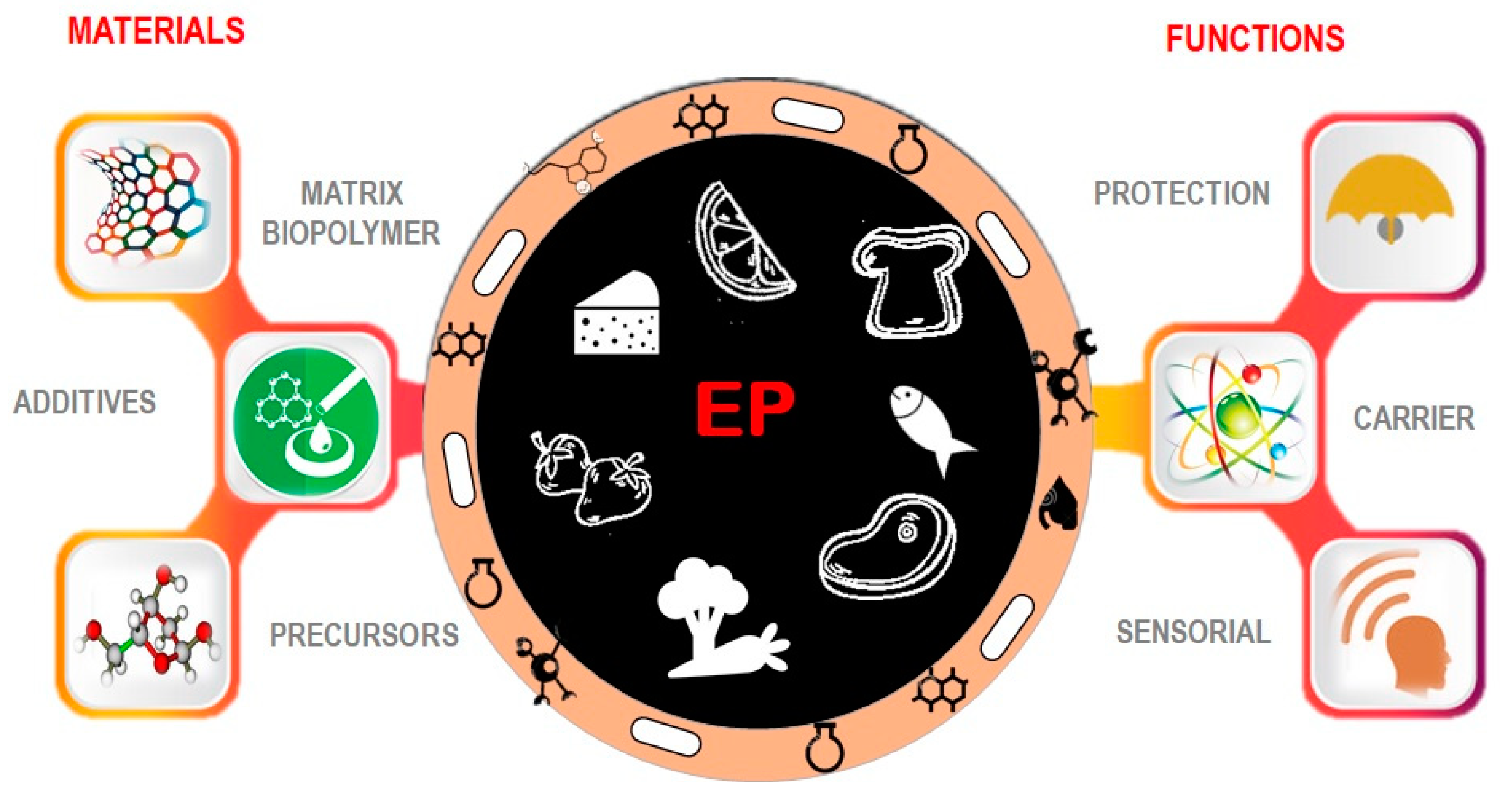
2. Bioactive Molecules in EP and Perspectives in Food Industry
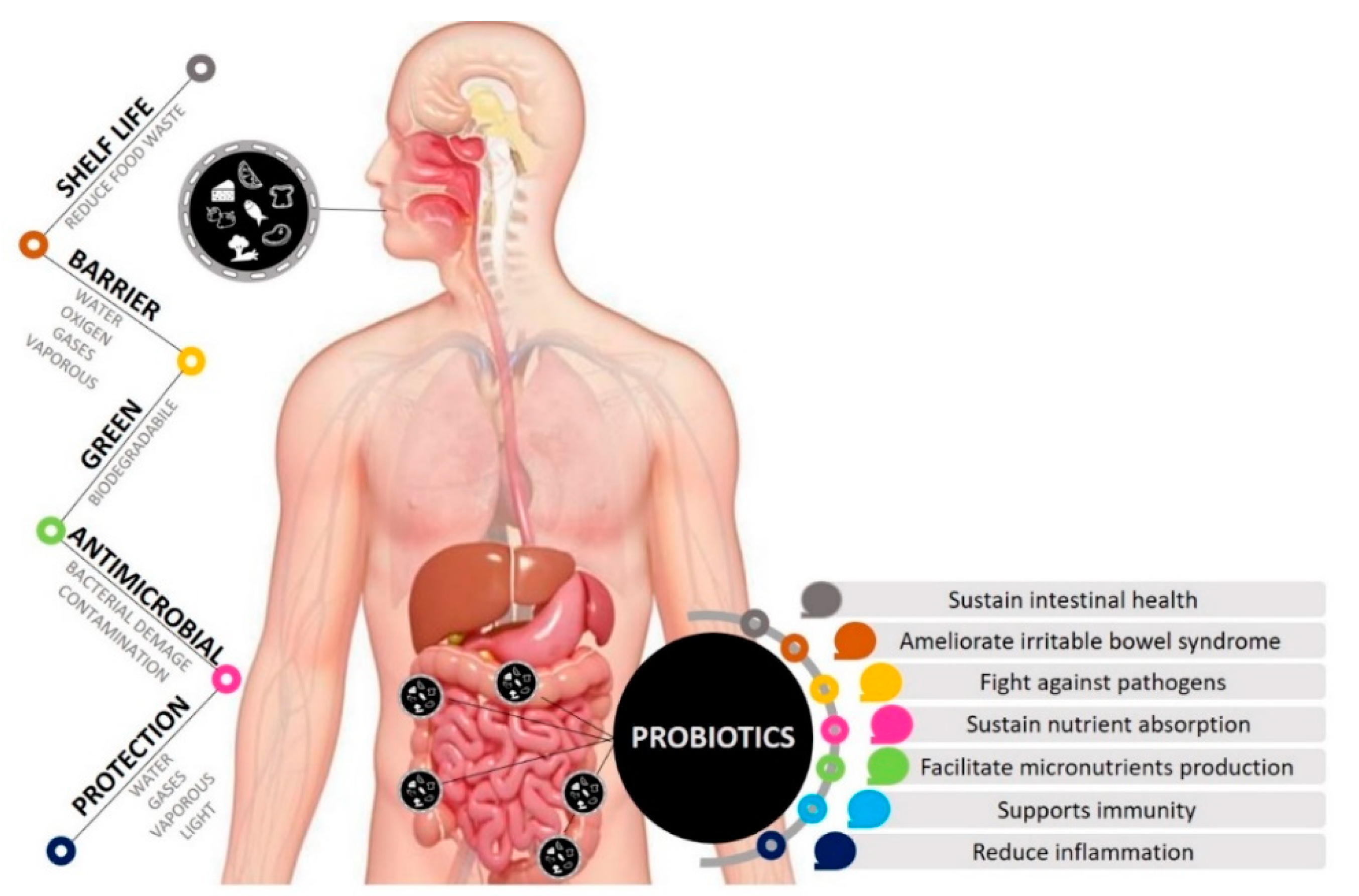
3. Probiotics in Food and Human Health
4. Entrapped Probiotics in EP
5. Materials and Techniques Used for Probiotic EP
6. Probiotics Viability in EP
7. Synbiotics in EP
8. Antimicrobial Effects of Probiotics Incorporated in EP
9. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gere, A.; Radványi, D.; Moskowitz, H. Consumer Perspectives about innovations in traditional foods. In Innovations in Traditional Foods; Galanakis, C.M., Ed.; Woodhead Publishing: Cambridge, UK, 2019; pp. 53–84. [Google Scholar]
- Quirós-Sauceda, A.E.; Ayala-Zavala, J.F.; Olivas, G.I.; González-Aguilar, G.A. Edible coatings as encapsulating matrices for bioactive compounds: A review. J. Food. Sci. Technol. 2014, 51, 1674–1685. [Google Scholar] [CrossRef] [PubMed]
- Salgado, P.R.; Ortiz, C.M.; Musso, Y.S.; Di Giorgio, L.; Mauri, A.N. Edible films and coatings containing bioactives. Curr. Opin. Dood Sci. 2015, 5, 86–92. [Google Scholar] [CrossRef]
- Guilbert, S.; Gontard, N.; Cuq, B. Technology and applications of edible protective films. Packag. Technol. Sci. 1995, 8, 339–346. [Google Scholar] [CrossRef]
- Galus, S.; Kadzińska, J. Food applications of emulsion-based edible films and coatings. Trends Food Sci. Technol. 2015, 45, 273–283. [Google Scholar] [CrossRef]
- Paul, S.K. Edible films and coatings for fruits and vegetables. In Reference Module in Materials Science and Materials Engineering; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Parente Ribeiro Cerqueira, M.Â. Edible packaging. In Encyclopedia of Food Chemistry; Melton, L., Shahidi, F., Varelis, P., Eds.; Academic Press: Oxford, UK, 2019; pp. 173–176. [Google Scholar]
- Mkandawire, M.; Aryee, A.N.A. Resurfacing and modernization of edible packaging material technology. Curr. Opin. Food Sci. 2018, 19, 104–112. [Google Scholar] [CrossRef]
- McMillin, K.W. Advancements in meat packaging. Meat Sci. 2017, 132, 153–162. [Google Scholar] [CrossRef]
- Schumann, B.; Schmid, M. Packaging concepts for fresh and processed meat—Recent progresses. Innov. Food Sci. Emerg. Technol. 2018, 47, 88–100. [Google Scholar] [CrossRef]
- Min, S.; Harris, L.J.; Krochta, J.M. Listeria monocytogenes inhibition by whey protein films and coatings incorporating the lactoperoxidase system. J. Food Sci. 2005, 70, m317–m324. [Google Scholar] [CrossRef]
- Trinetta, V. Edible packaging. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Rodriguez-Turienzo, L.; Cobos, A.; Moreno, V.; Caride, A.; Vieites, J.M.; Diaz, O. Whey protein-based coatings on frozen Atlantic salmon (Salmo salar): Influence of the plasticiser and the moment of coating on quality preservation. Food Chem. 2011, 128, 187–194. [Google Scholar] [CrossRef]
- Valencia-Chamorro, S.A.; Palou, L.; Del Rio, M.A.; Perez-Gago, M.B. Antimicrobial edible films and coatings for fresh and minimally processed fruits and vegetables: A review. Crit. Rev. Food Sci. Nutr. 2011, 51, 872–900. [Google Scholar] [CrossRef]
- Cagri, A.; Ustunol, Z.; Ryser, E.T. Antimicrobial edible films and coatings. J. Food Protect. 2004, 67, 833–848. [Google Scholar] [CrossRef] [PubMed]
- Hassan, B.; Chatha, S.A.S.; Hussain, A.I.; Zia, K.M.; Akhtar, N. Recent advances on polysaccharides, lipids and protein based edible films and coatings: A review. Int. J. Biol. Macromol. 2018, 109, 1095–1107. [Google Scholar] [CrossRef] [PubMed]
- Ehteshami, S.; Abdollahi, F.; Ramezanian, A.; Dastjerdi, A.M.; Rahimzadeh, M. Enhanced chilling tolerance of pomegranate fruit by edible coatings combined with malic and oxalic acid treatments. Sci. Hortic. 2019, 250, 388–398. [Google Scholar] [CrossRef]
- Ares, G.; Giménez, A.; Deliza, R. Methodological approaches for measuring consumer-perceived well-being in a food-related context. In Methods in Consumer Research; Ares, G., Varela, P., Eds.; Woodhead Publishing: Cambridge, UK, 2018; Volume 2, pp. 183–200. [Google Scholar]
- Li, X.-Y.; Du, X.-L.; Liu, Y.; Tong, L.-J.; Wang, Q.; Li, J.-L. Rhubarb extract incorporated into an alginate-based edible coating for peach preservation. Sci. Hortic. 2019, 257, 108685. [Google Scholar] [CrossRef]
- Guo, M.; Yadav, M.P.; Jin, T.Z. Antimicrobial edible coatings and films from micro-emulsions and their food applications. Int. J. Food Microbiol. 2017, 263, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Erginkaya, Z.; Kalkan, S.; Ünal, E. Use of antimicrobial edible films and coatings as packaging materials for food safety. In Food Processing: Strategies for Quality Assessment; Malik, A., Erginkaya, Z., Ahmad, S., Erten, H., Eds.; Springer: New York, NY, USA, 2014; pp. 261–295. [Google Scholar]
- Benbettaïeb, N.; Debeaufort, F.; Karbowiak, T. Bioactive edible films for food applications: Mechanisms of antimicrobial and antioxidant activity. Crit. Rev. Food Sci. Nutr. 2018, 59, 3431–3455. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, P.; Singh, M.; Sehra, N.; Pandey, N.; Sangwan, R.S.; Mishra, B.B. Processing of wet Kinnow mandarin (Citrus reticulata) fruit waste into novel Brønsted acidic ionic liquids and their application in hydrolysis of sucrose. Bioresour. Technol. 2018, 250, 621–624. [Google Scholar] [CrossRef]
- Otoni, C.G.; Avena-Bustillos, R.J.; Azeredo, H.M.C.; Lorevice, M.V.; Moura, M.R.; Mattoso, L.H.C.; McHugh, T.H. Recent advances on edible films based on fruits and vegetables—A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1151–1169. [Google Scholar] [CrossRef]
- Odila Pereira, J.; Soares, J.; Sousa, S.; Madureira, A.R.; Gomes, A.; Pintado, M. Edible films as carrier for lactic acid bacteria. LWT 2016, 73, 543–550. [Google Scholar] [CrossRef]
- Realini, C.E.; Marcos, B. Active and intelligent packaging systems for a modern society. Meat Sci. 2014, 98, 404–419. [Google Scholar] [CrossRef]
- Müller, P.; Schmid, M. Intelligent packaging in the food sector: A brief overview. Foods 2019, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, T. Traditional food & modern lifestyle: Impact of probiotics. Indian J. Med. Res. 2014, 140, 333–335. [Google Scholar] [PubMed]
- Pop, O.L.; Socaci, S.A.; Suharoschi, R.; Vodnar, D.C. Pro and prebiotics foods that modulate human health. In The Role of Alternative and Innovative Food Ingredients and Products in Consumer Wellness; Elesvier: Amsterdam, The Netherlands, 2019; p. 283. [Google Scholar]
- Yahfoufi, N.; Mallet, J.F.; Graham, E.; Matar, C. Role of probiotics and prebiotics in immunomodulation. Curr. Opin. Food Sci. 2018, 20, 82–91. [Google Scholar] [CrossRef]
- Scheid, M.M.A.; Moreno, Y.M.F.; Maróstica Junior, M.R.; Pastore, G.M. Effect of prebiotics on the health of the elderly. Food Res. Int. 2013, 53, 426–432. [Google Scholar] [CrossRef]
- Pop, O.L.; Salanță, L.-C.; Pop, C.R.; Coldea, T.; Socaci, S.A.; Suharoschi, R.; Vodnar, D.C. Prebiotics and dairy applications. In Dietary Fiber: Properties, Recovery, and Applications; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 247–277. [Google Scholar]
- Araya, M.M.L.; Reid, G.; Sanders, M.E.; Stanton, C.; Pineiro, M. Joint FAO/WHO Working Group Report on Drafting Guidelines for the Evaluation of Probiotics in Food; World Health Organization, Food and Agriculture Organization of the United Nations: London, ON, Canada, 2002. [Google Scholar]
- Ogier, J.C.; Serror, P. Safety assessment of dairy microorganisms: The Enterococcus genus. Int. J. Food Microbiol. 2008, 126, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; von Wright, A.; Morelli, L.; Marteau, P.; Brassart, D.; de Vos, W.M.; Fonden, R.; Saxelin, M.; Collins, K.; Mogensen, G.; et al. Demonstration of safety of probiotics—A review. Int. J. Food Microbiol. 1998, 44, 93–106. [Google Scholar] [CrossRef]
- Danneskiold-Samsøe, N.B.; Dias de Freitas Queiroz Barros, H.; Santos, R.; Bicas, J.L.; Cazarin, C.B.B.; Madsen, L.; Kristiansen, K.; Pastore, G.M.; Brix, S.; Maróstica Júnior, M.R. Interplay between food and gut microbiota in health and disease. Food Res. Int. 2019, 115, 23–31. [Google Scholar] [CrossRef]
- Mohanty, D.; Misra, S.; Mohapatra, S.; Sahu, P.S. Prebiotics and synbiotics: Recent concepts in nutrition. Food Biosci. 2018, 26, 152–160. [Google Scholar] [CrossRef]
- Rezazadeh, L.; Gargari, B.P.; Jafarabadi, M.A.; Alipour, B. Effects of probiotic yogurt on glycemic indexes and endothelial dysfunction markers in patients with metabolic syndrome. Nutrition 2019, 62, 162–168. [Google Scholar] [CrossRef]
- Alizadeh Behbahani, B.; Noshad, M.; Falah, F. Inhibition of Escherichia coli adhesion to human intestinal Caco-2 cells by probiotic candidate Lactobacillus plantarum strain L15. Microb. Pathog. 2019, 136, 103677. [Google Scholar] [CrossRef]
- Martin Manuel, P.; Elena, B.; Carolina, M.G.; Gabriela, P. Oral probiotics supplementation can stimulate the immune system in a stress process. J. Nutr. Intermed. Metab. 2017, 8, 29–40. [Google Scholar] [CrossRef]
- Joshi, D.; Roy, S.; Banerjee, S. Prebiotics: A functional food in health and disease. In Natural Products and Drug Discovery; Mandal, S.C., Mandal, V., Konishi, T., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 507–523. [Google Scholar]
- Duncan, S.H.; Flint, H.J. Probiotics and prebiotics and health in ageing populations. Maturitas 2013, 75, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Soukoulis, C.; Behboudi-Jobbehdar, S.; Yonekura, L.; Parmenter, C.; Fisk, I.D. Stability of Lactobacillus rhamnosus GG in prebiotic edible films. Food Chem. 2014, 159, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Majid, I.; Ahmad Nayik, G.; Mohammad Dar, S.; Nanda, V. Novel food packaging technologies: Innovations and future prospective. J. Saudi Soc. Agric. Sci. 2018, 17, 454–462. [Google Scholar] [CrossRef]
- Jantzen, M.; Göpel, A.; Beermann, C. Direct spray drying and microencapsulation of probiotic Lactobacillus reuteri from slurry fermentation with whey. J. Appl. Microbiol. 2013, 115, 1029–1036. [Google Scholar] [PubMed]
- Tapia, M.S.; Rojas-Grau, M.A.; Rodriguez, F.J.; Ramirez, J.; Carmona, A.; Martin-Belloso, O. Alginate- and gellan-based edible films for probiotic coatings on fresh-cut fruits. J. Food Sci. 2007, 72, E190–E196. [Google Scholar] [CrossRef]
- Tahir, H.E.; Xiaobo, Z.; Mahunu, G.K.; Arslan, M.; Abdalhai, M.; Zhihua, L. Recent developments in gum edible coating applications for fruits and vegetables preservation: A review. Carbohydr. Polym. 2019, 224, 115141. [Google Scholar] [CrossRef]
- Altamirano-Fortoul, R.; Moreno-Terrazas, R.; Quezada-Gallo, A.; Rosell, C.M. Viability of some probiotic coatings in bread and its effect on the crust mechanical properties. Food Hydrocoll. 2012, 29, 166–174. [Google Scholar] [CrossRef]
- Ramos, O.L.; Fernandes, J.C.; Silva, S.I.; Pintado, M.E.; Malcata, F.X. Edible films and coatings from whey proteins: A review on formulation, and on mechanical and bioactive properties. Crit. Rev. Food. Sci. Nutr. 2012, 52, 533–552. [Google Scholar] [CrossRef]
- Rojas-Graü, M.A.; Soliva-Fortuny, R.; Martín-Belloso, O. Edible coatings to incorporate active ingredients to fresh-cut fruits: A review. Trends Food Sci. Technol. 2009, 20, 438–447. [Google Scholar] [CrossRef]
- Cerqueira, M.A.; Bourbon, A.I.; Pinheiro, A.C.; Martins, J.T.; Souza, B.W.S.; Teixeira, J.A.; Vicente, A.A. Galactomannans use in the development of edible films/coatings for food applications. Trends Food Sci. Technol. 2011, 22, 662–671. [Google Scholar] [CrossRef]
- Singh, P.; Magalhães, S.; Alves, L.; Antunes, F.; Miguel, M.; Lindman, B.; Medronho, B. Cellulose-based edible films for probiotic entrapment. Food Hydrocoll. 2019, 88, 68–74. [Google Scholar] [CrossRef]
- Tavera-Quiroz, M.J.; Romano, N.; Mobili, P.; Pinotti, A.; Gómez-Zavaglia, A.; Bertola, N. Green apple baked snacks functionalized with edible coatings of methylcellulose containing Lactobacillus plantarum. J. Funct. Food 2015, 16, 164–173. [Google Scholar] [CrossRef]
- Sánchez-González, L.; Quintero Saavedra, J.I.; Chiralt, A. Physical properties and antilisterial activity of bioactive edible films containing Lactobacillus plantarum. Food Hydrocoll. 2013, 33, 92–98. [Google Scholar] [CrossRef]
- Romano, N.; Tavera-Quiroz, M.J.; Bertola, N.; Mobili, P.; Pinotti, A.; Gómez-Zavaglia, A. Edible methylcellulose-based films containing fructo-oligosaccharides as vehicles for lactic acid bacteria. Food Res. Int. 2014, 64, 560–566. [Google Scholar] [CrossRef]
- Tavassoli-Kafrani, E.; Shekarchizadeh, H.; Masoudpour-Behabadi, M. Development of edible films and coatings from alginates and carrageenans. Carbohydr. Polym. 2016, 137, 360–374. [Google Scholar] [CrossRef]
- Tapia, M.S.; Rojas-Graü, M.A.; Carmona, A.; Rodríguez, F.J.; Soliva-Fortuny, R.; Martin-Belloso, O. Use of alginate- and gellan-based coatings for improving barrier, texture and nutritional properties of fresh-cut papaya. Food Hydrocoll. 2008, 22, 1493–1503. [Google Scholar] [CrossRef]
- Varela, P.; Fiszman, S.M. Hydrocolloids in fried foods. A review. Food Hydrocoll. 2011, 25, 1801–1812. [Google Scholar] [CrossRef]
- Elsabee, M.Z.; Abdou, E.S. Chitosan based edible films and coatings: A review. Mater. Sci. Eng. C 2013, 33, 1819–1841. [Google Scholar] [CrossRef]
- Martínez-Camacho, A.P.; Cortez-Rocha, M.O.; Ezquerra-Brauer, J.M.; Graciano-Verdugo, A.Z.; Rodriguez-Félix, F.; Castillo-Ortega, M.M.; Yépiz-Gómez, M.S.; Plascencia-Jatomea, M. Chitosan composite films: Thermal, structural, mechanical and antifungal properties. Carbohydr. Polym. 2010, 82, 305–315. [Google Scholar] [CrossRef]
- Park, S.Y.; Marsh, K.S.; Rhim, J.W. Characteristics of different molecular weight chitosan films affected by the type of organic solvents. J. Food Sci. 2002, 67, 194–197. [Google Scholar] [CrossRef]
- Raafat, D.; Sahl, H.G. Chitosan and its antimicrobial potential—A critical literature survey. Microb. Biotechnol. 2009, 2, 186–201. [Google Scholar] [CrossRef] [PubMed]
- Salama, H.E.; Abdel Aziz, M.S.; Sabaa, M.W. Novel biodegradable and antibacterial edible films based on alginate and chitosan biguanidine hydrochloride. Int. J. Biol. Macromol. 2018, 116, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Galindez, A.; Daza, L.D.; Homez-Jara, A.; Eim, V.S.; Váquiro, H.A. Characterization of ulluco starch and its potential for use in edible films prepared at low drying temperature. Carbohydr. Polym. 2019, 215, 143–150. [Google Scholar] [CrossRef]
- Thakur, R.; Pristijono, P.; Bowyer, M.; Singh, S.P.; Scarlett, C.J.; Stathopoulos, C.E.; Vuong, Q.V. A starch edible surface coating delays banana fruit ripening. LWT 2019, 100, 341–347. [Google Scholar] [CrossRef]
- Arvanitoyannis, I.; Biliaderis, C.G.; Ogawa, H.; Kawasaki, N. Biodegradable films made from low-density polyethylene (LDPE), rice starch and potato starch for food packaging applications: Part 1. Carbohydr. Polym. 1998, 36, 89–104. [Google Scholar] [CrossRef]
- Moalemiyan, M.; Ramaswamy, H.S.; Maftoonazad, N. Pectin-based edible coating for shelf-life extension of ataulfo mango. J. Food Process Eng. 2012, 35, 572–600. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Du, W.-X.; de Avena-Bustillos, R.J.; de Soares, N.F.F.; McHugh, T.H. Edible films from pectin: Physical-mechanical and antimicrobial properties—A review. Food Hydrocoll. 2014, 35, 287–296. [Google Scholar] [CrossRef]
- Chiralt, A.; González-Martínez, C.; Vargas, M.; Atarés, L. 18—Edible films and coatings from proteins. In Proteins in Food Processing, 2nd ed.; Yada, R.Y., Ed.; Woodhead Publishing: Cambridge, UK, 2018; pp. 477–500. [Google Scholar]
- Galanakis, C.M. Preface. In Proteins: Sustainable Source, Processing and Applications; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. ix–xi. [Google Scholar]
- Rhim, J.W.; Shellhammer, T.H. 21—Lipid-based edible films and coatings. In Innovations in Food Packaging; Han, J.H., Ed.; Academic Press: London, UK, 2005; pp. 362–383. [Google Scholar]
- Enujiugha, V.N.; Oyinloye, A.M. Protein-lipid interactions and the formation of edible films and coatings. In Encyclopedia of Food Chemistry; Melton, L., Shahidi, F., Varelis, P., Eds.; Academic Press: Oxford, UK, 2019; pp. 478–482. [Google Scholar]
- Xu, A.; Chen, L.; Wang, J. Functionalized Imidazalium carboxylates for enhancing practical applicability in cellulose processing. Macromolecules 2018, 51, 4158–4166. [Google Scholar] [CrossRef]
- Xu, A.; Guo, X.; Zhang, Y.; Li, Z.; Wang, J. Efficient and sustainable solvents for lignin dissolution: Aqueous choline carboxylate solutions. Green Chem. 2017, 19, 4067–4073. [Google Scholar] [CrossRef]
- Tóth, A.; Halász, K. Characterization of edible biocomposite films directly prepared from psyllium seed husk and husk flour. Food Pack. Shelf Life 2019, 20, 100299. [Google Scholar] [CrossRef]
- Arnon-Rips, H.; Poverenov, E. Improving food products’ quality and storability by using Layer by Layer edible coatings. Trends Food Sci. Technol. 2018, 75, 81–92. [Google Scholar] [CrossRef]
- Lopez-Rubio, A.; Gavara, R.; Lagaron, J.M. Bioactive packaging: Turning foods into healthier foods through biomaterials. Trends Food Sci. Technol. 2006, 17, 567–575. [Google Scholar] [CrossRef]
- Torres-Giner, S.; Perez-Masia, R.; Lagaron, J.M. A review on electrospun polymer nanostructures as advanced bioactive platforms. Polym. Eng. Sci. 2016, 56, 500–527. [Google Scholar] [CrossRef]
- Moreno, O.; Atarés, L.; Chiralt, A.; Cruz-Romero, M.C.; Kerry, J. Starch-gelatin antimicrobial packaging materials to extend the shelf life of chicken breast fillets. LWT 2018, 97, 483–490. [Google Scholar] [CrossRef]
- Fagundes, C.; Palou, L.; Monteiro, A.R.; Pérez-Gago, M.B. Hydroxypropyl methylcellulose-beeswax edible coatings formulated with antifungal food additives to reduce alternaria black spot and maintain postharvest quality of cold-stored cherry tomatoes. Sci. Hortic. 2015, 193, 249–257. [Google Scholar] [CrossRef]
- Guerreiro, A.C.; Gago, C.M.L.; Faleiro, M.L.; Miguel, M.G.C.; Antunes, M.D.C. The effect of alginate-based edible coatings enriched with essential oils constituents on Arbutus unedo L. fresh fruit storage. Postharvest Biol. Technol. 2015, 100, 226–233. [Google Scholar] [CrossRef]
- Synowiec, A.; Gniewosz, M.; Kraśniewska, K.; Przybył, J.L.; Bączek, K.; Węglarz, Z. Antimicrobial and antioxidant properties of pullulan film containing sweet basil extract and an evaluation of coating effectiveness in the prolongation of the shelf life of apples stored in refrigeration conditions. Innov. Food Sci. Emerg. 2014, 23, 171–181. [Google Scholar] [CrossRef]
- Arroyo, B.J.; Bezerra, A.C.; Oliveira, L.L.; Arroyo, S.J.; de Melo, E.A.; Santos, A.M.P. Antimicrobial active edible coating of alginate and chitosan add ZnO nanoparticles applied in guavas (Psidium guajava L.). Food Chem. 2019, 125566. [Google Scholar] [CrossRef]
- Duran, A.; Kahve, H.I. The effect of chitosan coating and vacuum packaging on the microbiological and chemical properties of beef. Meat Sci. 2019, 107961. [Google Scholar] [CrossRef]
- Boyacı, D.; Korel, F.; Yemenicioğlu, A. Development of activate-at-home-type edible antimicrobial films: An example pH-triggering mechanism formed for smoked salmon slices using lysozyme in whey protein films. Food Hydrocoll. 2016, 60, 170–178. [Google Scholar] [CrossRef]
- Kilincceker, O.; Dogan, İ.S.; Kucukoner, E. Effect of edible coatings on the quality of frozen fish fillets. LWT 2009, 42, 868–873. [Google Scholar] [CrossRef]
- Lacroix, M.; Ouattara, B.; Saucier, L.; Giroux, M.; Smoragiewicz, W. Effect of gamma irradiation in presence of ascorbic acid on microbial composition and TBARS concentration of ground beef coated with an edible active coating. Radiat. Phys. Chem. 2004, 71, 73–77. [Google Scholar] [CrossRef]
- Kulawik, P.; Jamróz, E.; Zając, M.; Guzik, P.; Tkaczewska, J. The effect of furcellaran-gelatin edible coatings with green and pu-erh tea extracts on the microbiological, physicochemical and sensory changes of salmon sushi stored at 4 °C. Food Control 2019, 100, 83–91. [Google Scholar] [CrossRef]
- Khan, M.K.I.; Cakmak, H.; Tavman, Ş.; Schutyser, M.; Schroёn, K. Anti-browning and barrier properties of edible coatings prepared with electrospraying. Innov. Food Sci. Emerg. 2014, 25, 9–13. [Google Scholar] [CrossRef]
- Saucedo-Pompa, S.; Rojas-Molina, R.; Aguilera-Carbó, A.F.; Saenz-Galindo, A.; de Garza, H.L.; Jasso-Cantú, D.; Aguilar, C.N. Edible film based on candelilla wax to improve the shelf life and quality of avocado. Food Res. Int. 2009, 42, 511–515. [Google Scholar] [CrossRef]
- Singh, S.; Khemariya, P.; Rai, A.; Rai, A.C.; Koley, T.K.; Singh, B. Carnauba wax-based edible coating enhances shelf-life and retain quality of eggplant (Solanum melongena) fruits. LWT 2016, 74, 420–426. [Google Scholar] [CrossRef]
- Tomás, S.A.; Bosquez-Molina, E.; Stolik, S.; Sánchez, F. Effects of mesquite gum-candelilla wax based edible coatings on the quality of guava fruit (Psidium guajava L.). J. Phys. IV Fr. 2005, 125, 889–892. [Google Scholar] [CrossRef]
- Velickova, E.; Winkelhausen, E.; Kuzmanova, S.; Alves, V.D.; Moldão-Martins, M. Impact of chitosan-beeswax edible coatings on the quality of fresh strawberries (Fragaria ananassa cv Camarosa) under commercial storage conditions. LWT 2013, 52, 80–92. [Google Scholar] [CrossRef]
- Lee, J.Y.; Park, H.J.; Lee, C.Y.; Choi, W.Y. Extending shelf-life of minimally processed apples with edible coatings and antibrowning agents. LWT 2003, 36, 323–329. [Google Scholar] [CrossRef]
- Poverenov, E.; Zaitsev, Y.; Arnon, H.; Granit, R.; Alkalai-Tuvia, S.; Perzelan, Y.; Weinberg, T.; Fallik, E. Effects of a composite chitosan–gelatin edible coating on postharvest quality and storability of red bell peppers. Postharvest Biol. Technol. 2014, 96, 106–109. [Google Scholar] [CrossRef]
- Valencia-Chamorro, S.A.; Pérez-Gago, M.B.; del Río, M.Á.; Palou, L. Effect of antifungal hydroxypropyl methylcellulose (HPMC)–lipid edible composite coatings on postharvest decay development and quality attributes of cold-stored ‘Valencia’ oranges. Postharvest Biol. Technol. 2009, 54, 72–79. [Google Scholar] [CrossRef]
- Khorram, F.; Ramezanian, A.; Hosseini, S.M.H. Shellac, gelatin and Persian gum as alternative coating for orange fruit. Sci. Hortic. 2017, 225, 22–28. [Google Scholar] [CrossRef]
- Guimarães, J.E.R.; de la Fuente, B.; Pérez-Gago, M.B.; Andradas, C.; Carbó, R.; Mattiuz, B.-H.; Palou, L. Antifungal activity of GRAS salts against Lasiodiplodia theobromae in vitro and as ingredients of hydroxypropyl methylcellulose-lipid composite edible coatings to control Diplodia stem-end rot and maintain postharvest quality of citrus fruit. Int. J. Food Microbiol. 2019, 301, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.C. Probiotics as functional foods. Nutr. Clin. Pract. 2003, 18, 497–506. [Google Scholar] [CrossRef]
- Vieira da Silva, B.; Barreira, J.C.M.; Oliveira, M.B.P.P. Natural phytochemicals and probiotics as bioactive ingredients for functional foods: Extraction, biochemistry and protected-delivery technologies. Trends Food Sci. Technol. 2016, 50, 144–158. [Google Scholar] [CrossRef]
- Kanmani, P.; Lim, S.T. Development and characterization of novel probiotic-residing pullulan/starch edible films. Food Chem. 2013, 141, 1041–1049. [Google Scholar] [CrossRef]
- Soukoulis, C.; Singh, P.; Macnaughtan, W.; Parmenter, C.; Fisk, I.D. Compositional and physicochemical factors governing the viability of Lactobacillus rhamnosus GG embedded in starch-protein based edible films. Food Hydrocoll. 2016, 52, 876–887. [Google Scholar] [CrossRef]
- Piermaria, J.; Diosma, G.; Aquino, C.; Garrote, G.; Abraham, A. Edible kefiran films as vehicle for probiotic microorganisms. Innov. Food Sci. Emerg. 2015, 32, 193–199. [Google Scholar] [CrossRef]
- Tang, Y.; Xie, F.; Zhang, D.; Zhu, M.; Liu, L.; Liu, P.; Gu, C. Physical properties and prebiotic activity of maize starch-based functional films. Starch Stärke 2015, 67, 124–131. [Google Scholar] [CrossRef]
- Mattila-Sandholm, T.; Myllärinen, P.; Crittenden, R.; Mogensen, G.; Fondén, R.; Saarela, M. Technological challenges for future probiotic foods. Int. Dairy J. 2002, 12, 173–182. [Google Scholar] [CrossRef]
- Gialamas, H.; Zinoviadou, K.G.; Biliaderis, C.G.; Koutsoumanis, K.P. Development of a novel bioactive packaging based on the incorporation of Lactobacillus sakei into sodium-caseinate films for controlling Listeria monocytogenes in foods. Food Res. Int. 2010, 43, 2402–2408. [Google Scholar] [CrossRef]
- Socaciu, M.-I.; Semeniuc, C.A.; Vodnar, D.C. Edible Films and coatings for fresh fish packaging: focus on quality changes and shelf-life extension. Coatings 2018, 8, 366. [Google Scholar] [CrossRef]
- Lauzon, H.L.; Pérez-Sánchez, T.; Merrifield, D.L.; Ringø, E.; Balcázar, J.L. Probiotic Applications in Cold Water Fish. Species. In Aquaculture Nutrition; John and Wiley and Sons: Hoboken, NJ, USA, 2014; pp. 223–252. [Google Scholar]
- Concha-Meyer, A.; Schöbitz, R.; Brito, C.; Fuentes, R. Lactic acid bacteria in an alginate film inhibit Listeria monocytogenes growth on smoked salmon. Food Control 2011, 22, 485–489. [Google Scholar] [CrossRef]
- López de Lacey, A.M.; López-Caballero, M.E.; Montero, P. Agar films containing green tea extract and probiotic bacteria for extending fish shelf-life. LWT 2014, 55, 559–564. [Google Scholar] [CrossRef]
- Sánchez-González, L.; Quintero Saavedra, J.I.; Chiralt, A. Antilisterial and physical properties of biopolymer films containing lactic acid bacteria. Food Control 2014, 35, 200–206. [Google Scholar] [CrossRef]
| Materials/Methods | Generic Effects | Specific Composition | Type of Food | Specific Effects | Reference | |
|---|---|---|---|---|---|---|
| Polysaccharides | Starch | + colorless + oil-free appearance + reduced caloric content + prolong shelf life + suitable for fruits, vegetables, meat + control oxygen transmission + reduce darkening of the surface - no moisture barrier - hydrophilic nature | Starch-based coatings with D-glucose, silver nitrate. | Chicken Sausages | Antimicrobial activity. | [79] |
| Cellulose and derivatives | Hydroxypropyl methylcellulose (HPMC) and beeswax coatings. | Cherry tomatoes | Prevent weight loss, sustain fruit firmness, improved sensory attributes. | [80] | ||
| Pectin | Pectin and sodium alginate coatings with citral and eugenol essential oils. | Raspberries | Maintain the color, prevent weight loss, trolox equivalent antioxidant capacity, prevent microbial growth. | [81] | ||
| Pullulan | Pullulan-based coatings with sweet basil extract. | Apples | Sustain color, appearance and sensory attributes during hypothermia storage. | [82] | ||
| Alginates | Alginate - chitosan and ZnO nanoparticle | Guavas | Increase the shelf-life of the fruit. | [83] | ||
| Chitosan | Chitosan-based coatings with vacuum packaging. | Beef | Effects on color preservation and lipid oxidation during retail presentation. | [84] | ||
| Proteins | Vegetable-based proteins | + provide mechanical stability + good transparency - not suitable for some diets (vegan) | Whey proteins coatings with lysozyme. | Salmon | Overall quality of salmon. | [85] |
| Gluten and zein coatings with potassium caseinate, rennet casein, xanthan gum, locust bean additives. | Trout Fillets | Sensorial attributes and the physical biochemical qualities. | [86] | |||
| Animal-based proteins | Caseinate-based coatings with ascorbic acid additives. | Beef | Effect of gamma irradiation on microbiological characteristics of ground beef. | [87] | ||
| Furcellaran-gelatin-based edible coating. | Salmon sushi | Exhibit good transparency, mechanical and barrier properties and can be manufactured by extrusion or casting processes. | [88] | |||
| Fats | Oils | + reduce water transmission | Lipid-based (sunflower oil and chocolate) coating with stearic acid, polyglycerol. | Apple slices | Moisture barrier. | [89] |
| Waxes | Candelilla wax coating with ellagic acid. | Avocado | Antifungal characteristics to enhance shelf life. | [90] | ||
| Carnauba wax coating. | Eggplant | Increase in the water vapor resistance and reduction in weight loss. | [91] | |||
| Candelilla wax coatings with mineral oil. | Guava fruit | Weight loss ethylene emission, gloss, retention of the color, firmness. | [92] | |||
| Chitosan-Beeswax coating. | Strawberries | Reduction in weight loss. | [93] | |||
| Multicomponents/Composites | + special tailored for specific characteristics + enhance the permeability or mechanical properties - may get expensive | Composites of carrageenan and whey protein coatings with CMC sodium salt, polyethylene glycol, calcium chloride, glycerol and oxalic acid additives. | Apples | Reduce brownness. | [94] | |
| Composite of chitosan and gelatin coatings. | Red bell peppers | Improve firmness, diminish weight loss, and ethanol concentration. | [95] | |||
| Composite of hydroxypropyl methyl cellulose (HPMC) and lipid coating with potassium sorbate, sodium benzoate, sodium propionate, stearic acid, glycerol additives. | Oranges | Antifungal properties improved during long-term cold storage. | [96] | |||
| Shellac, gelatin and Persian gum. | Orange | Improve permeability characteristics. | [97] | |||
| Hydroxypropyl methylcellulose-lipid composite edible coatings. | Citrus fruits | Maintain postharvest quality. | [98] | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pop, O.L.; Pop, C.R.; Dufrechou, M.; Vodnar, D.C.; Socaci, S.A.; Dulf, F.V.; Minervini, F.; Suharoschi, R. Edible Films and Coatings Functionalization by Probiotic Incorporation: A Review. Polymers 2020, 12, 12. https://doi.org/10.3390/polym12010012
Pop OL, Pop CR, Dufrechou M, Vodnar DC, Socaci SA, Dulf FV, Minervini F, Suharoschi R. Edible Films and Coatings Functionalization by Probiotic Incorporation: A Review. Polymers. 2020; 12(1):12. https://doi.org/10.3390/polym12010012
Chicago/Turabian StylePop, Oana L., Carmen R. Pop, Marie Dufrechou, Dan C. Vodnar, Sonia A. Socaci, Francisc V. Dulf, Fabio Minervini, and Ramona Suharoschi. 2020. "Edible Films and Coatings Functionalization by Probiotic Incorporation: A Review" Polymers 12, no. 1: 12. https://doi.org/10.3390/polym12010012
APA StylePop, O. L., Pop, C. R., Dufrechou, M., Vodnar, D. C., Socaci, S. A., Dulf, F. V., Minervini, F., & Suharoschi, R. (2020). Edible Films and Coatings Functionalization by Probiotic Incorporation: A Review. Polymers, 12(1), 12. https://doi.org/10.3390/polym12010012