Characterization of Polydioxanone in Near-Field Electrospinning
Abstract
1. Introduction
2. Materials and Methods
2.1. NFES Apparatus
2.2. Fiber Diameter Characterization
2.2.1. Setup and Materials
2.2.2. Fiber Diameter Processing Parameters
2.2.3. Fiber Variability Processing Parameters
2.2.4. Fiber Imaging and Diameter Analysis
2.3. 3D NFES Templates
2.3.1. Setup and Materials
2.3.2. Template Imaging and Fiber Diameter Analysis
2.4. Fiber Crystalinity
2.4.1. Fiber Processing Parameters
2.4.2. Differential Scanning Calorimetry (DSC)
2.5. Template-Driven Cell Alignment
2.5.1. Template Preparation Processing Parameters
2.5.2. In Vitro Cell Culture
2.5.3. Fluorescent Microscopy and Analysis
2.6. Statistics
3. Results
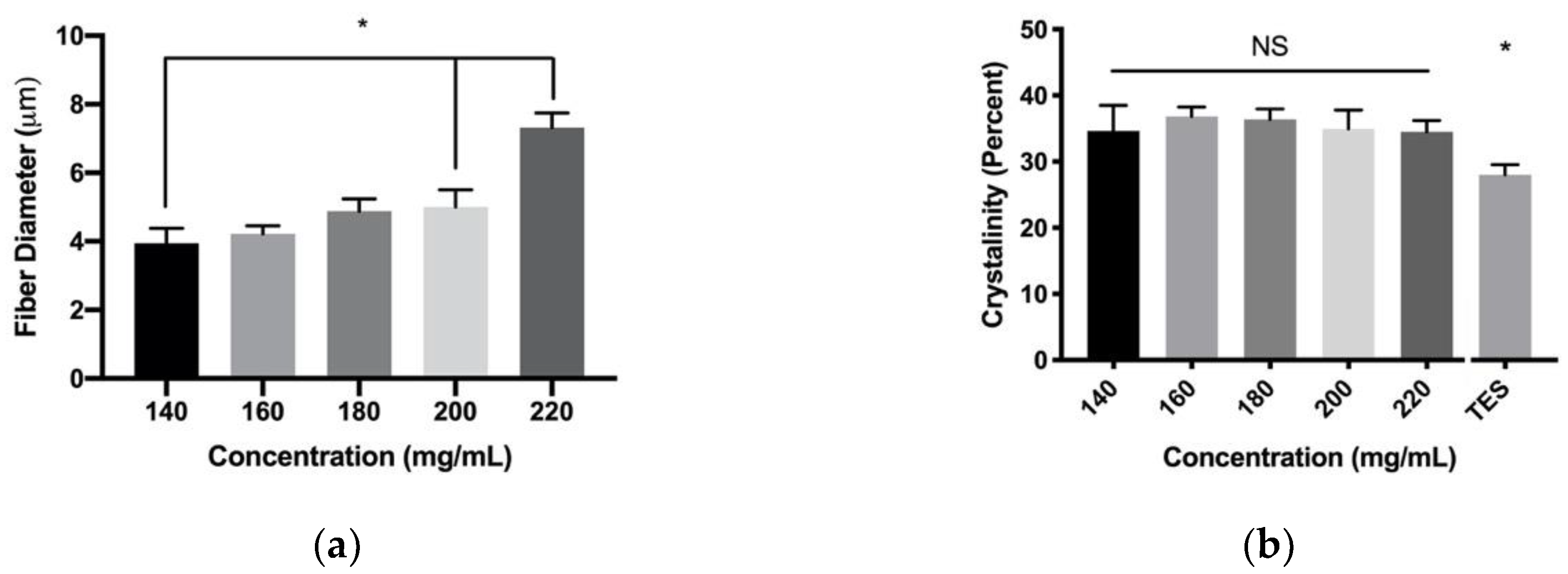
3.1. PDO Fiber Characterization
3.1.1. Processing Parameters
3.1.2. Fiber Variability
3.1.3. Fiber Stacking
3.2. Fiber Crystalinity
3.3. In Vitro Cell Culture
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Formhals, A. Process and Apparatus for Preparing Artificial Threads. U.S. Patent 1975504A, 2 October 1934. [Google Scholar]
- Doshi, J.; Reneker, D.H. Electrospinning process and apllications of electrospun fibers. J. Electrost. 1995, 35, 151–160. [Google Scholar] [CrossRef]
- Taylor, G. Disintegration of water drops in an electric field. Proc. R. Soc. Lond. Ser. A Math. Phys. 1964, 280, 383–397. [Google Scholar]
- Cloupeau, M.; Prunet-Fock, B. Electrostatic spraying of liquids in cone-jet mode. J. Electrost. 1989, 22, 135–159. [Google Scholar] [CrossRef]
- Greiner, A.; Wendorff, J.H. Electrospinning: A fascinating method for the preparation of ultrathin fibers. Angew. Chem. Int. Ed. Engl. 2007, 46, 5670–5703. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Zhang, J.; Li, J.; Li, D.; Xiao, C.; Xiao, H.; Yang, H.; Zhuang, X.; Chen, X. Electrospun polymer biomaterials. Prog. Polym. Sci. 2019, 90, 1–34. [Google Scholar] [CrossRef]
- Yang, D.; Lu, B.; Zhao, Y.; Jiang, X. Fabrication of aligned fibrous arrays by magnetic electrospinning. Adv. Mater. 2007, 19, 3702–3706. [Google Scholar] [CrossRef]
- Boland, E.D.; Coleman, B.D.; Barnes, C.P.; Simpson, D.G.; Wnek, G.E.; Bowlin, G.L. Electrospinning polydioxanone for biomedical applications. Acta Biomater. 2005, 1, 115–123. [Google Scholar] [CrossRef]
- Wang, C.; Wang, J.; Zeng, L.; Qiao, Z.; Liu, X.; Liu, H.; Zhang, J.; Ding, J. Fabrication of electrospun polymer nanofibers with diverse morphologies. Molecules 2019, 24, 834. [Google Scholar] [CrossRef]
- Kameoka, J.; Orth, R.; Yang, Y.; Czaplewski, D.; Mathers, R.; Coates, G.W.; Craighead, H.G. A scanning tip electrospinning source for deposition of oriented nanofibres. Nanotechnology 2003, 14, 1124–1129. [Google Scholar] [CrossRef]
- Sun, D.; Chang, C.; Li, S.; Lin, L. Near-field electrospinning. Nano Lett. 2006, 6, 839–842. [Google Scholar] [CrossRef]
- He, X.-X.; Zheng, J.; Yu, G.-F.; You, M.-H.; Yu, M.; Ning, X.; Long, Y.-Z. Near-field electrospinning: Progress and applications. J. Phys. Chem. C 2017, 121, 8663–8678. [Google Scholar] [CrossRef]
- Fattahi, P.; Dover, J.T.; Brown, J.L. 3d near-field electrospinning of biomaterial microfibers with potential for blended microfiber-cell-loaded gel composite structures. Adv. Healthc. Mater. 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Bezwada, R.; Jamiolkowski, D.; Cooper, K. Poly-dioxanone and its copolymers. In Handbook of Biodegradable Polymers; Domb, A.J., Kost, J., Wiseman, D.M., Eds.; Hardwood Academic Publishers: Amsterdam, The Netherlands, 1998; pp. 29–62. [Google Scholar]
- Yang, K.; Wang, X.; Wang, Y. Poly(p-dioxanone) and its copolymers. J. Macromol. Sci. 2002, C42, 373–398. [Google Scholar] [CrossRef]
- Fetz, A.E.; Neeli, I.; Buddington, K.K.; Read, R.W.; Smeltzer, M.P.; Radic, M.Z.; Bowlin, G.L. Localized delivery of cl-amidine from electrospun polydioxanone templates to regulate acute neutrophil netosis: A preliminary evaluation of the pad4 inhibitor for tissue engineering. Front. Pharmacol. 2018, 9, 289. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.J.; McClure, M.J.; Sell, S.A.; Barnes, C.P.; Walpoth, B.H.; Simpson, D.G.; Bowlin, G.L. Suture-reinforced electrospun polydioxanone-elastin small-diameter tubes for use in vascular tissue engineering: A feasibility study. Acta Biomater. 2008, 4, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Selders, G.S.; Fetz, A.E.; Spear, S.L.; Bowlin, G.L. Fabrication and characterization of air-impedance electrospun polydioxanone templates. Electrospinning 2016, 1. [Google Scholar] [CrossRef]
- Minden-Birkenmaier, B.A.; Selders, G.S.; Fetz, A.E.; Gehrmann, C.J.; Bowlin, G.L. Electrospun systems for drug delivery. Electrospun Mater. Tissue Eng. Biomed. Appl. 2017, 117–145. [Google Scholar] [CrossRef]
- Chen, M.; Patra, P.K.; Warner, S.B.; Bhowmick, S. Role of fiber diameter in adhesion and proliferation of nih 3t3 fibroblast on electrospun polycaprolactone scaffolds. Tissue Eng. 2007, 13, 579–587. [Google Scholar] [CrossRef]
- Abhari, R.E.; Mouthuy, P.A.; Zargar, N.; Brown, C.; Carr, A. Effect of annealing on the mechanical properties and the degradation of electrospun polydioxanone filaments. J. Mech. Behav. Biomed. Mater. 2017, 67, 127–134. [Google Scholar] [CrossRef]
- Ramdhanie, L.I.; Aubuchon, S.R.; Boland, E.D.; Knapp, D.C.; Barnes, C.P.; Simpson, D.G.; Wnek, G.E.; Bowlin, G.L. Thermal and mechanical characterization of electrospun blends of poly(lactic acid) and poly(glycolic acid). Polym. J. 2006, 38, 1137–1145. [Google Scholar] [CrossRef]
- McQuin, C.; Goodman, A.; Chernyshev, V.; Kamentsky, L.; Cimini, B.A.; Karhohs, K.W.; Doan, M.; Ding, L.; Rafelski, S.M.; Thirstrup, D.; et al. Cellprofiler 3.0: Next-generation image processing for biology. PLoS Biol. 2018, 16, e2005970. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Limkrailassiri, K.; Lin, L. Continuous near-field electrospinning for large area deposition of orderly nanofiber patterns. Appl. Phys. Lett. 2008, 93. [Google Scholar] [CrossRef]
- Fuh, Y.K.; Wu, Y.C.; He, Z.Y.; Huang, Z.M.; Hu, W.W. The control of cell orientation using biodegradable alginate fibers fabricated by near-field electrospinning. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 62, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Bisht, G.S.; Canton, G.; Mirsepassi, A.; Kulinsky, L.; Oh, S.; Dunn-Rankin, D.; Madou, M.J. Controlled continuous patterning of polymeric nanofibers on three-dimensional substrates using low-voltage near-field electrospinning. Nano Lett. 2011, 11, 1831–1837. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Malheiro, A.; van Blitterswijk, C.; Mota, C.; Wieringa, P.A.; Moroni, L. Direct writing electrospinning of scaffolds with multidimensional fiber architecture for hierarchical tissue engineering. ACS Appl. Mater. Interfaces 2017, 9, 38187–38200. [Google Scholar] [CrossRef]
- Luo, G.; Teh, K.S.; Liu, Y.; Zang, X.; Wen, Z.; Lin, L. Direct-write, self-aligned electrospinning on paper for controllable fabrication of three-dimensional structures. ACS Appl. Mater. Interfaces 2015, 7, 27765–27770. [Google Scholar] [CrossRef]
- He, F.-L.; He, J.; Deng, X.; Li, D.-W.; Ahmad, F.; Liu, Y.-Y.; Liu, Y.-L.; Ye, Y.-J.; Zhang, C.-Y.; Yin, D.-C. Investigation of the effects of melt electrospinning parameters on the direct-writing fiber size using orthogonal design. J. Phys. D Appl. Phys. 2017, 50, 1–16. [Google Scholar] [CrossRef]
- Brown, T.D.; Edin, F.; Detta, N.; Skelton, A.D.; Hutmacher, D.W.; Dalton, P.D. Melt electrospinning of poly(epsilon-caprolactone) scaffolds: Phenomenological observations associated with collection and direct writing. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 45, 698–708. [Google Scholar] [CrossRef]
- Thompson, C.J.; Chase, G.G.; Yarin, A.L.; Reneker, D.H. Effect of parameters on nanofiber diameter determined from electrospinning model. Polymer 2007, 48, 6913–6922. [Google Scholar] [CrossRef]
- Deitzel, J.M.; Kleinmeyer, J.; Harris, D.; Beck Tan, N.C. The effect of processing variables on the morphology of electrospun nanofibers and textiles. Polymer 2001, 42, 261–272. [Google Scholar] [CrossRef]
- Gil-Castell, O.; Badia, J.D.; Bou, J.; Ribes-Greus, A. Performance of polyester-based electrospun scaffolds under in vitro hydrolytic conditions: From short-term to long-term applications. Nanomaterials 2019, 9, 786. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.L.; Chu, C.C.; Grubb, D. Hydrolytic degradation and morphologic study of poly-p-dioxanone. J. Biomed. Mater. Res. 1993, 27, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Ero-Phillips, O.; Jenkins, M.; Stamboulis, A. Tailoring crystallinity of electrospun plla fibres by control of electrospinning parameters. Polymers 2012, 4, 1331–1348. [Google Scholar] [CrossRef]
- Dias, J.C.; Ribeiro, C.; Sencadas, V.; Botelho, G.; Ribelles, J.L.G.; Lanceros-Mendez, S. Influence of fiber diameter and crystallinity on the stability of electrospun poly(l-lactic acid) membranes to hydrolytic degradation. Polym. Test. 2012, 31, 770–776. [Google Scholar] [CrossRef]
- Zheng, G.; Li, W.; Wang, X.; Wu, D.; Sun, D.; Lin, L. Precision deposition of a nanofibre by near-field electrospinning. J. Phys. D Appl. Phys. 2010, 43, 1–6. [Google Scholar] [CrossRef]
- Fetz, A.E.; Neeli, I.; Rodriguez, I.A.; Radic, M.Z.; Bowlin, G.L. Electrospun template architecture and composition regulate neutrophil netosis in vitro and in vivo. Tissue Eng. Part A 2017, 23, 1054–1063. [Google Scholar] [CrossRef] [PubMed]












| Dependent Parameter | Range, (Increment) | Independent Parameters | Value |
|---|---|---|---|
| Air Gap | 1.8–3.0 mm, (0.2) | Polymer Concentration: | 180 mg/mL |
| Needle Gauge: | 23 gauge, 2″ length | ||
| Translational Velocity: | 30 mm/s | ||
| Applied Voltage: | −1.3 kV | ||
| Flow Rate: | 15 μL/h | ||
| Polymer Concentration | 140–220 mg/mL, (20) | Air Gap: | 2.5 mm |
| Needle Gauge: | 23 gauge, 2″ length | ||
| Translational Velocity: | 30 mm/s | ||
| Applied Voltage: | −1.2 kV | ||
| Flow Rate: | 15 μL/h | ||
| Needle Gauge | 18,21, and 23 gauge, 2″ length | Air Gap: | 1.8 mm |
| Polymer Concentration: | 180 mg/mL | ||
| Translational Velocity: | 30 mm/s | ||
| Applied Voltage: | −1.2 kV | ||
| Flow Rate: | 23 gauge: 15 μL/h 1 | ||
| 21 gauge: 33.7 μL/h 1 | |||
| 18 gauge: 91.6 μL/h 1 | |||
| Translational Velocity | 10–50 mm/s, (10) And 100 mm/s | Air Gap: | 1.8 mm |
| Polymer Concentration: | 180 mg/mL | ||
| Needle Gauge: | 23 gauge, 2″ length | ||
| Applied Voltage: | −1.2 kV | ||
| Flow Rate: | 10 μL/h | ||
| Applied Voltage | 1.1–1.6 kV, (0.1) | Air Gap: | 1.8 mm |
| Polymer Concentration: | 160 mg/mL | ||
| Needle Gauge: | 23 gauge, 2″ length | ||
| Translational Velocity: | 30 mm/s | ||
| Flow Rate: | 15 μL/h |
| Dependent Parameter | Range (Increment) | Independent Parameters | Value |
|---|---|---|---|
| Polymer Concentration | 140–220 mg/mL, (20) | Air Gap: | 2.5 mm |
| Needle Gauge: | 23 gauge, 2″ length | ||
| Translational Velocity: | 30 mm/s | ||
| Applied Voltage: | −1.1 kV | ||
| Flow Rate: | 15 μL/h | ||
| Translational Velocity | 10–50 mm/s, (10) and 100 mm/s | Air Gap: | 1.8 mm |
| Polymer Concentration: | 180 mg/mL | ||
| Needle Gauge: | 23 gauge, 2″ length | ||
| Applied Voltage: | −1.1 kV | ||
| Flow Rate: | 10 μL/h |
| Dependent Parameter | Range and Increment | Independent Parameters | Value |
|---|---|---|---|
| Polymer Concentration | 140–220 mg/mL, (20) | Air Gap: | 1.8 mm |
| Needle Gauge: | 23 gauge, 2″ length | ||
| Translational Velocity: | 30 mm/s | ||
| Applied Voltage: | −1.3 kV | ||
| Flow Rate: | 15 μL/h | ||
| Translational Velocity | 10–50 mm/s, (10) | Air Gap: | 1.8 mm |
| Polymer Concentration: | 180 mg/mL | ||
| Needle Gauge: | 23 gauge, 2″ length | ||
| Applied Voltage: | −1.2 kV | ||
| Flow Rate: | 15 μL/h |
| Sample | Melt Enthalpy (J/g) | Crystallinity | Theoretical Melt Enthalpy for 100% Crystallization (J/g) |
|---|---|---|---|
| Stock PDO | 84.2 | 42.5% | 198.5 |
| 140 mg/mL PDO | 68.7 | 34.6% | − |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
E. King, W., III; Gillespie, Y.; Gilbert, K.; L. Bowlin, G. Characterization of Polydioxanone in Near-Field Electrospinning. Polymers 2020, 12, 1. https://doi.org/10.3390/polym12010001
E. King W III, Gillespie Y, Gilbert K, L. Bowlin G. Characterization of Polydioxanone in Near-Field Electrospinning. Polymers. 2020; 12(1):1. https://doi.org/10.3390/polym12010001
Chicago/Turabian StyleE. King, William, III, Yvonne Gillespie, Keaton Gilbert, and Gary L. Bowlin. 2020. "Characterization of Polydioxanone in Near-Field Electrospinning" Polymers 12, no. 1: 1. https://doi.org/10.3390/polym12010001
APA StyleE. King, W., III, Gillespie, Y., Gilbert, K., & L. Bowlin, G. (2020). Characterization of Polydioxanone in Near-Field Electrospinning. Polymers, 12(1), 1. https://doi.org/10.3390/polym12010001






