Fabrication of Eco-Friendly Solid-State Symmetric Ultracapacitor Device Based on Co-Doped PANI/GO Composite
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of Co-Doped Polyaniline Graphene Oxide Composites
2.3. Fabrication of Symmetric Solid-State Supercapacitor Device
2.4. Characterization
3. Results and Discussion
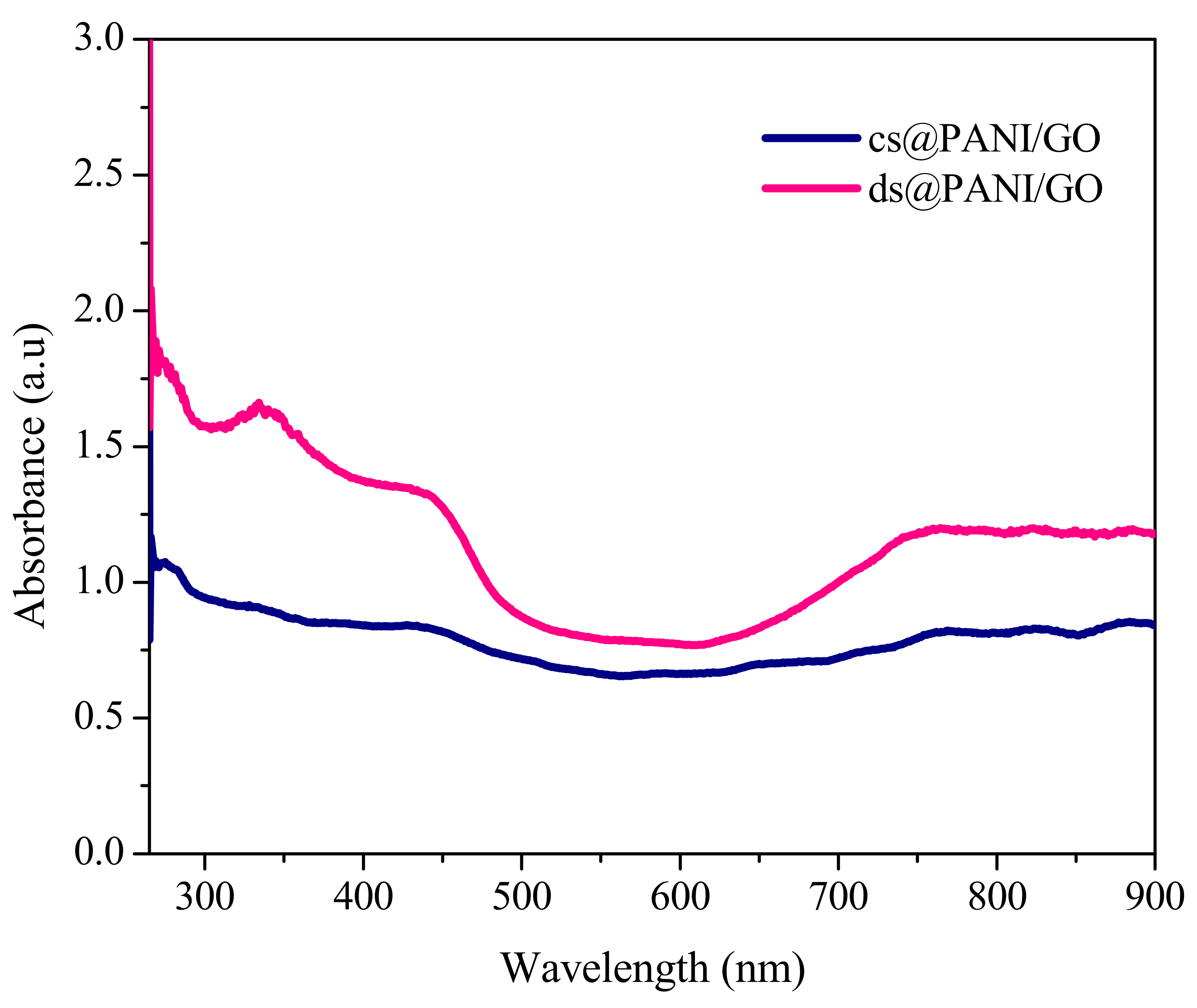
3.1. UV-Visible Spectroscopic Analysis
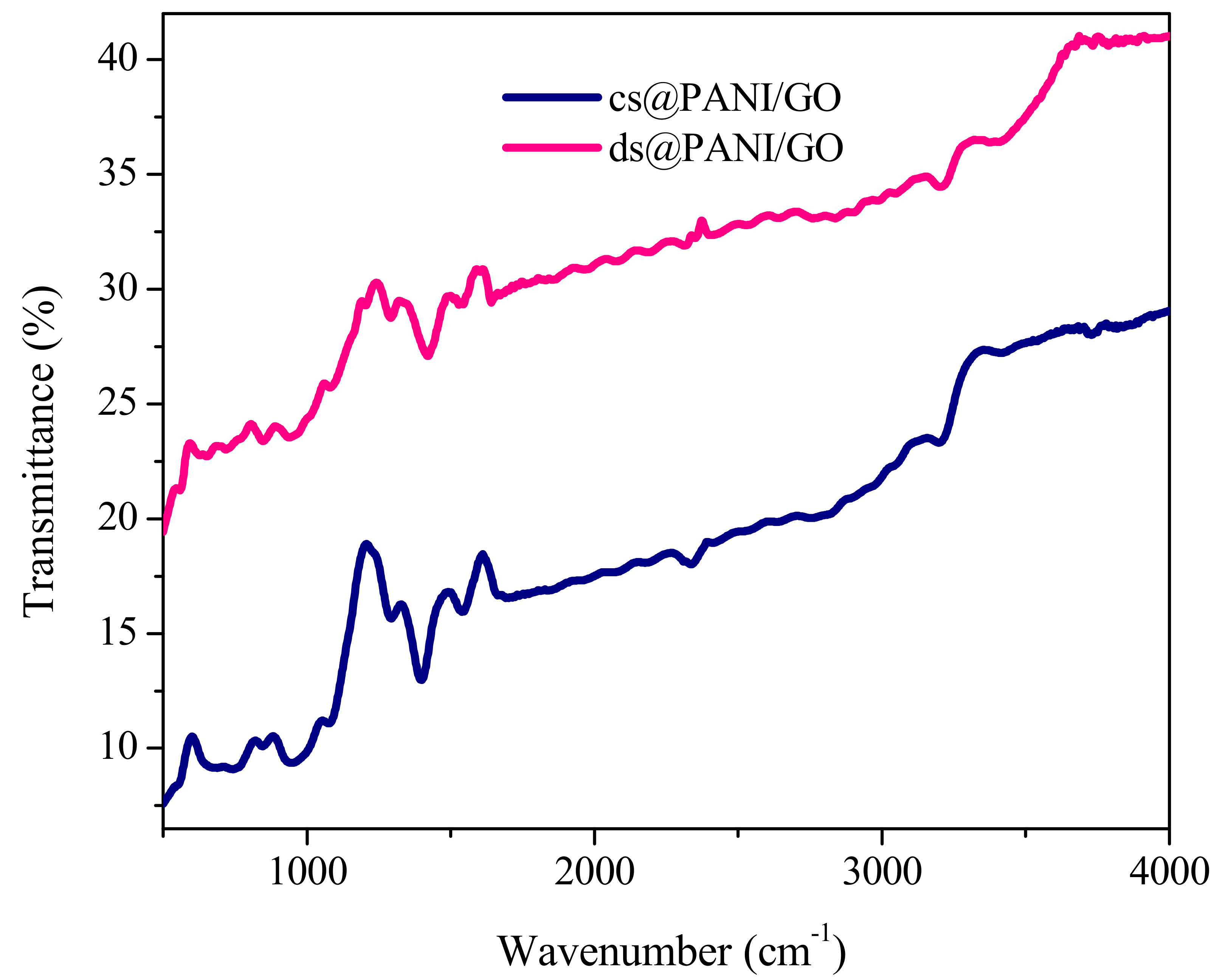
3.2. FTIR Characterization
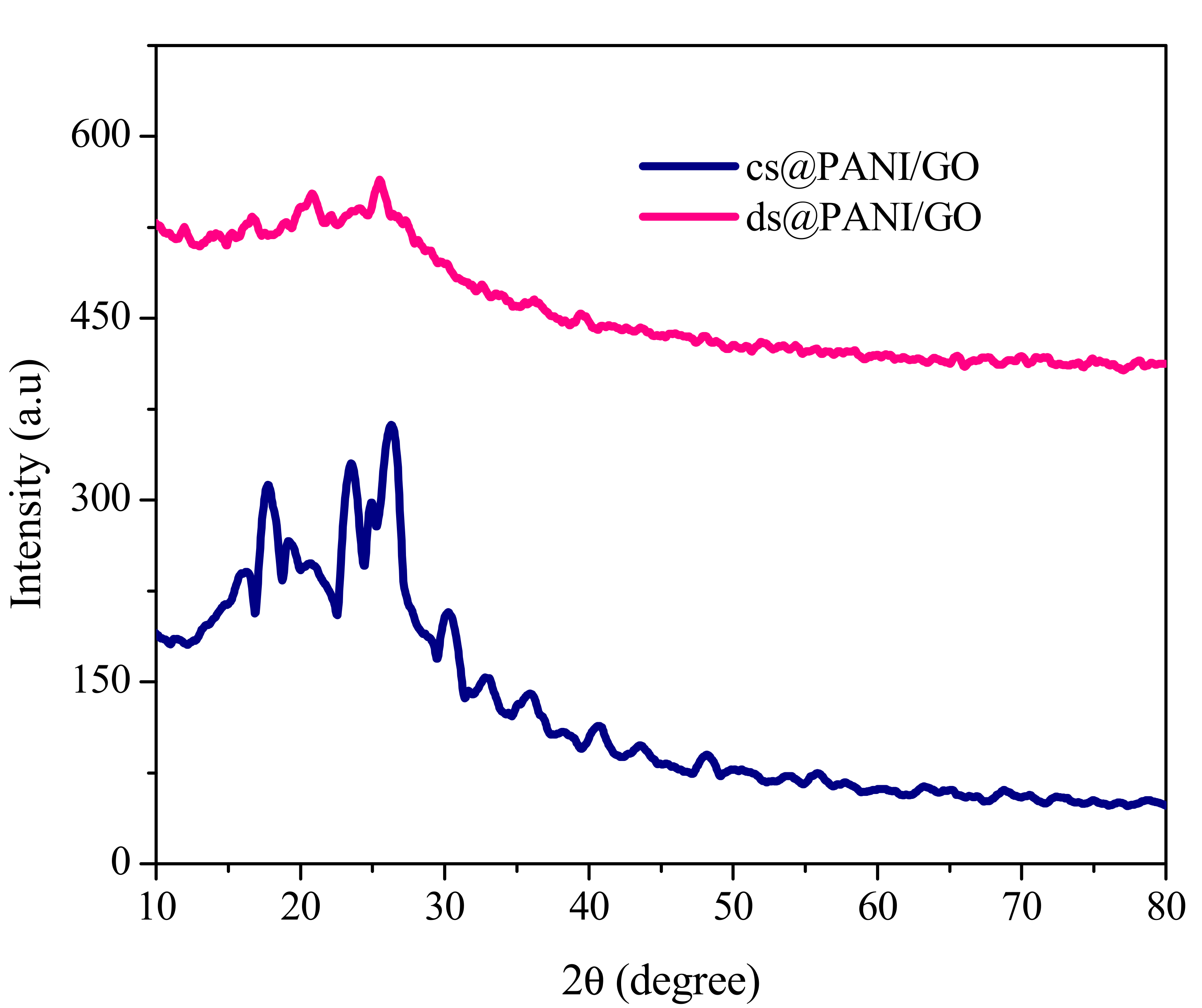
3.3. X-ray Diffraction Analysis
3.4. Morphological Study
3.5. Energy Dispersive X-ray Analyis
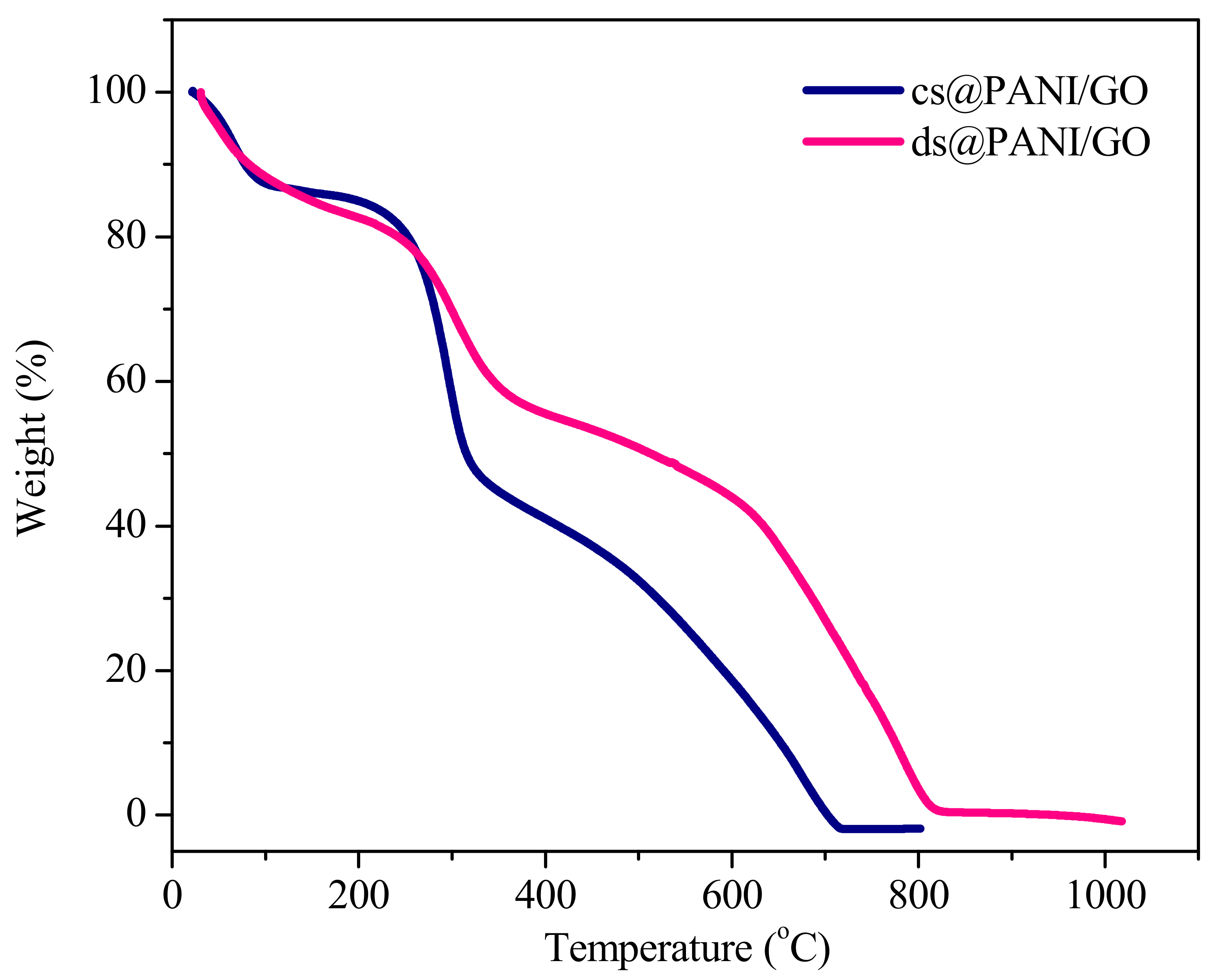
3.6. Thermogravimetry Analysis (TGA)
3.7. Electrochemical Characteristics
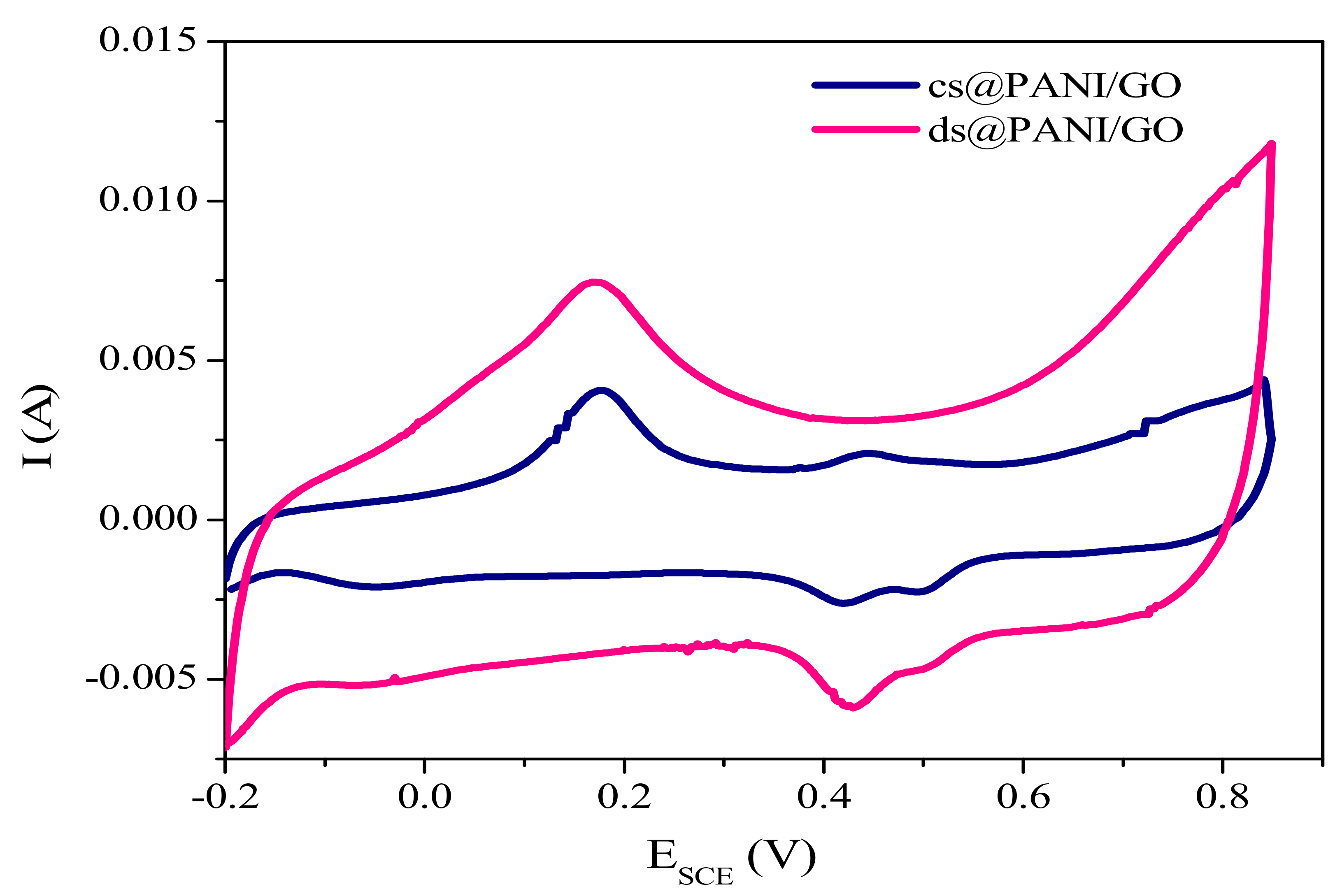
3.7.1. Cyclic Voltammetry (CV)
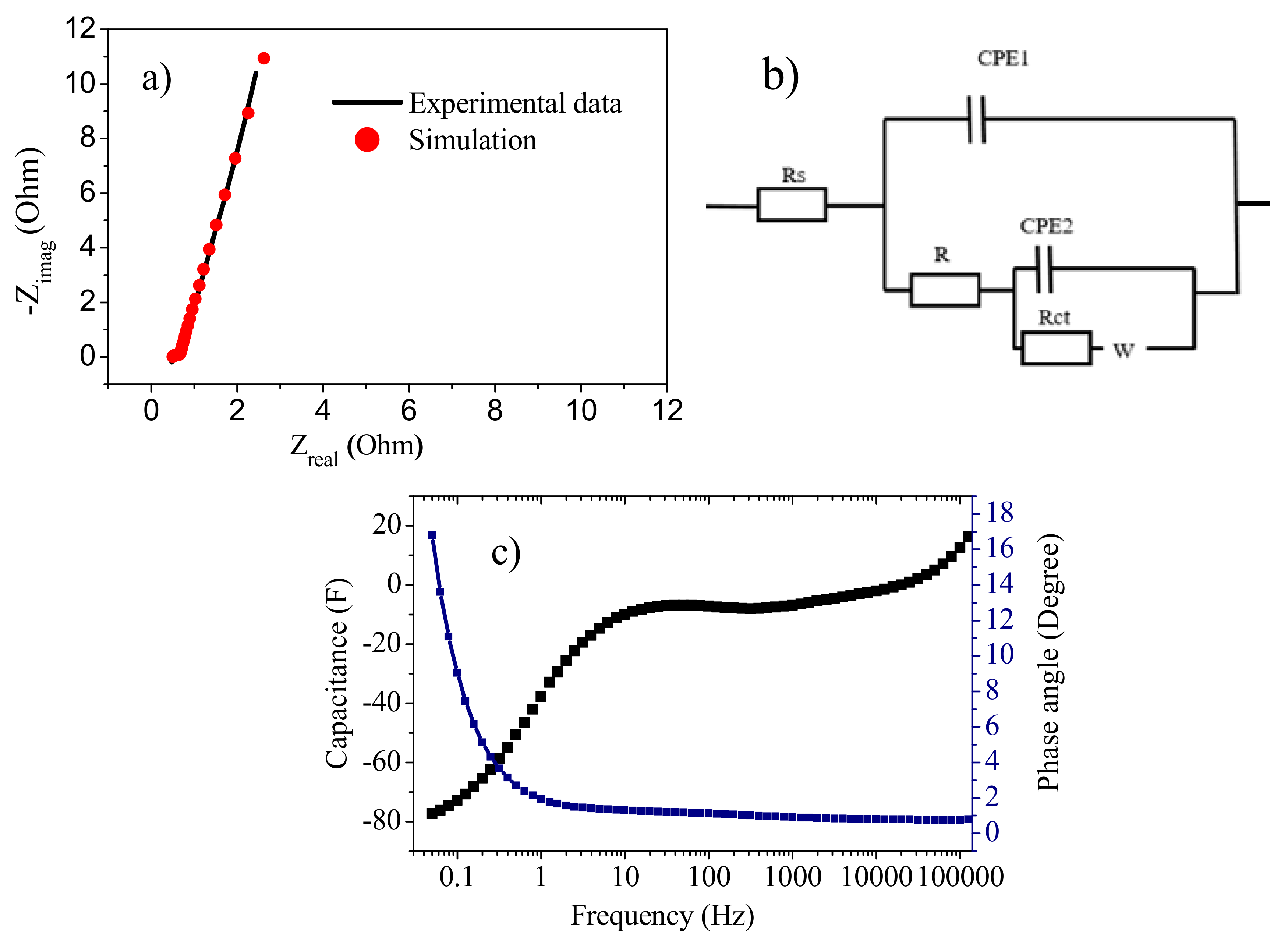
3.7.2. Electrochemical Impedance Spectroscopy (EIS)
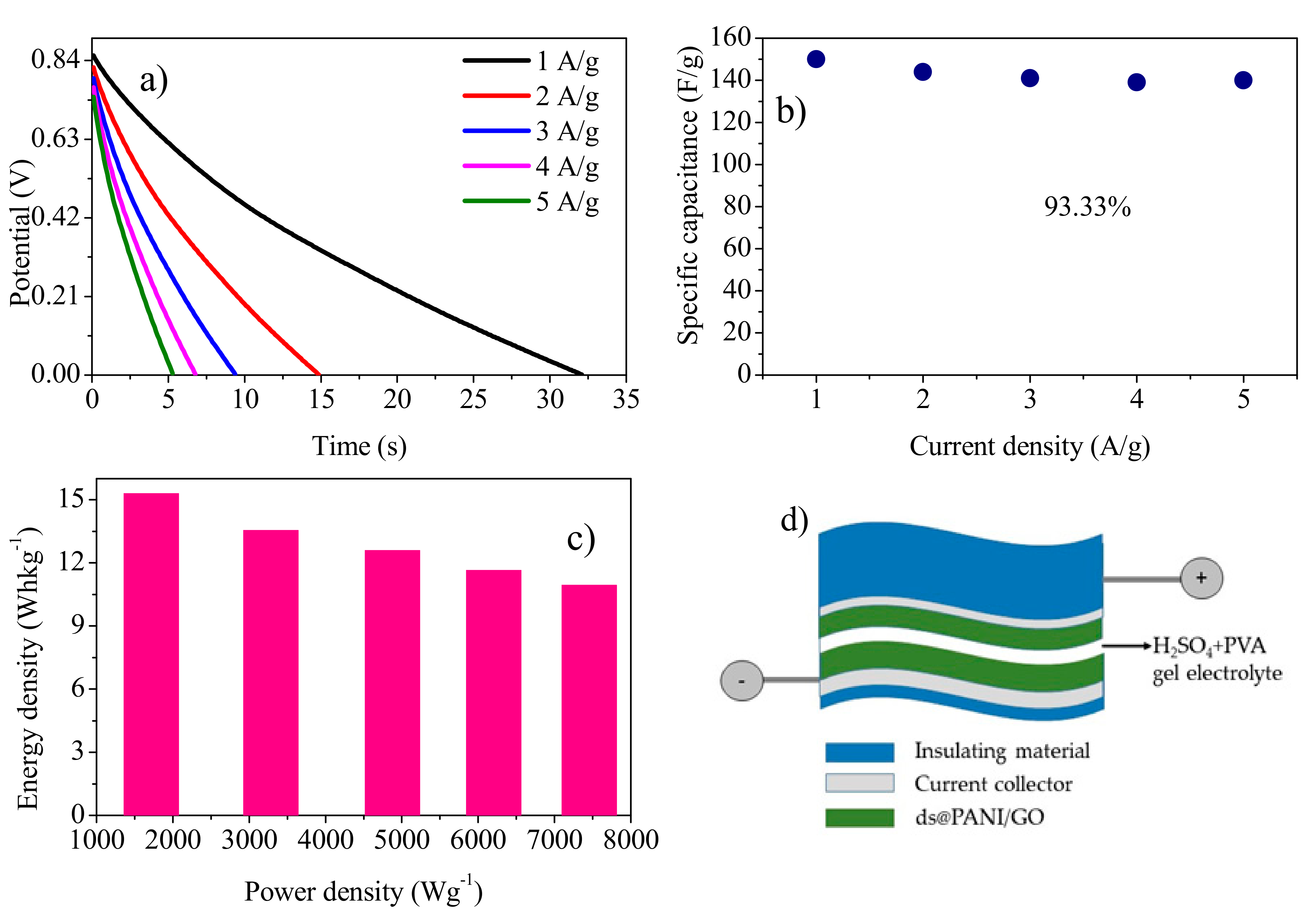
3.8. Solid State Symmetric Device Fabrication with Gold as Current Collector
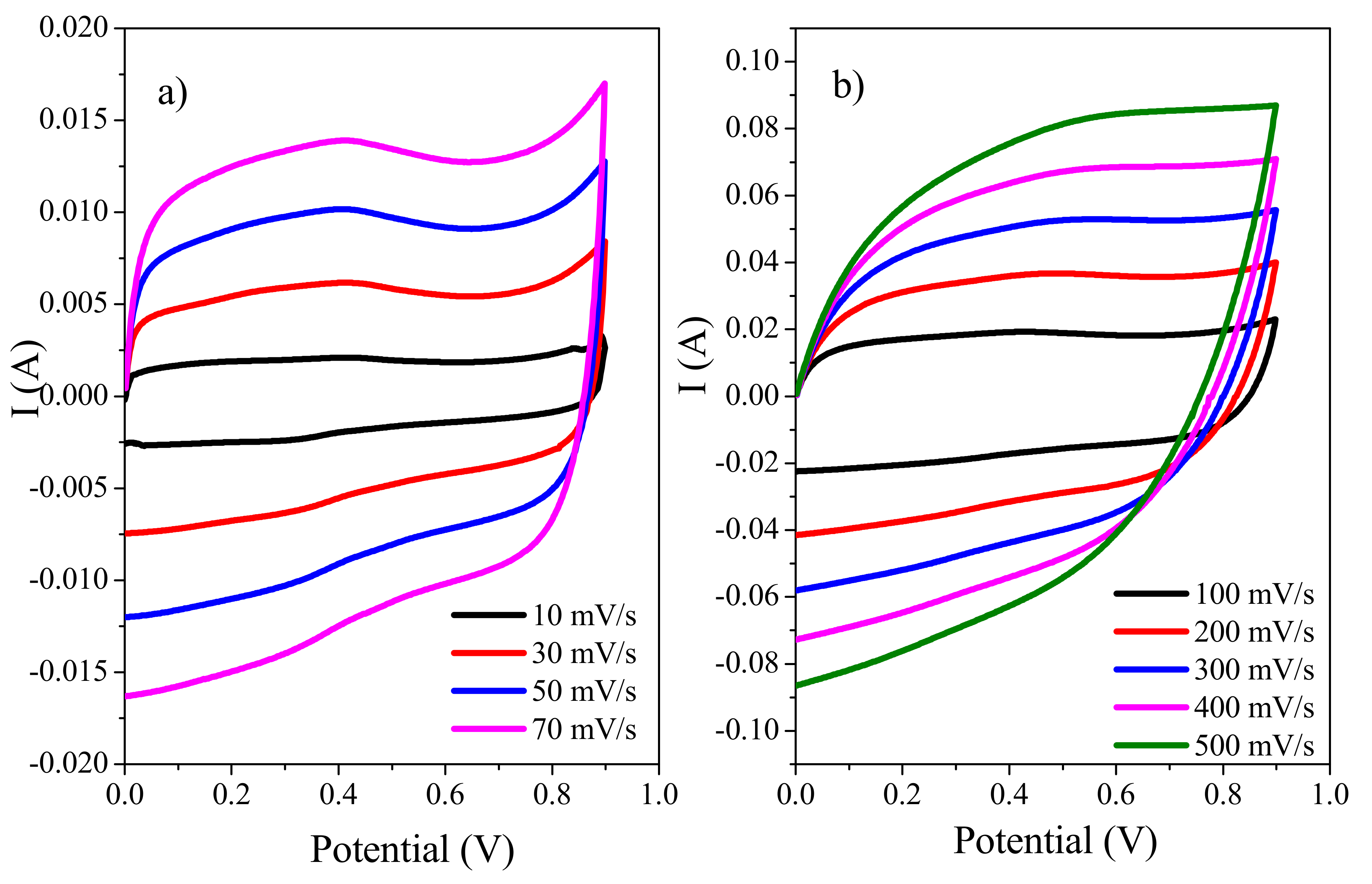
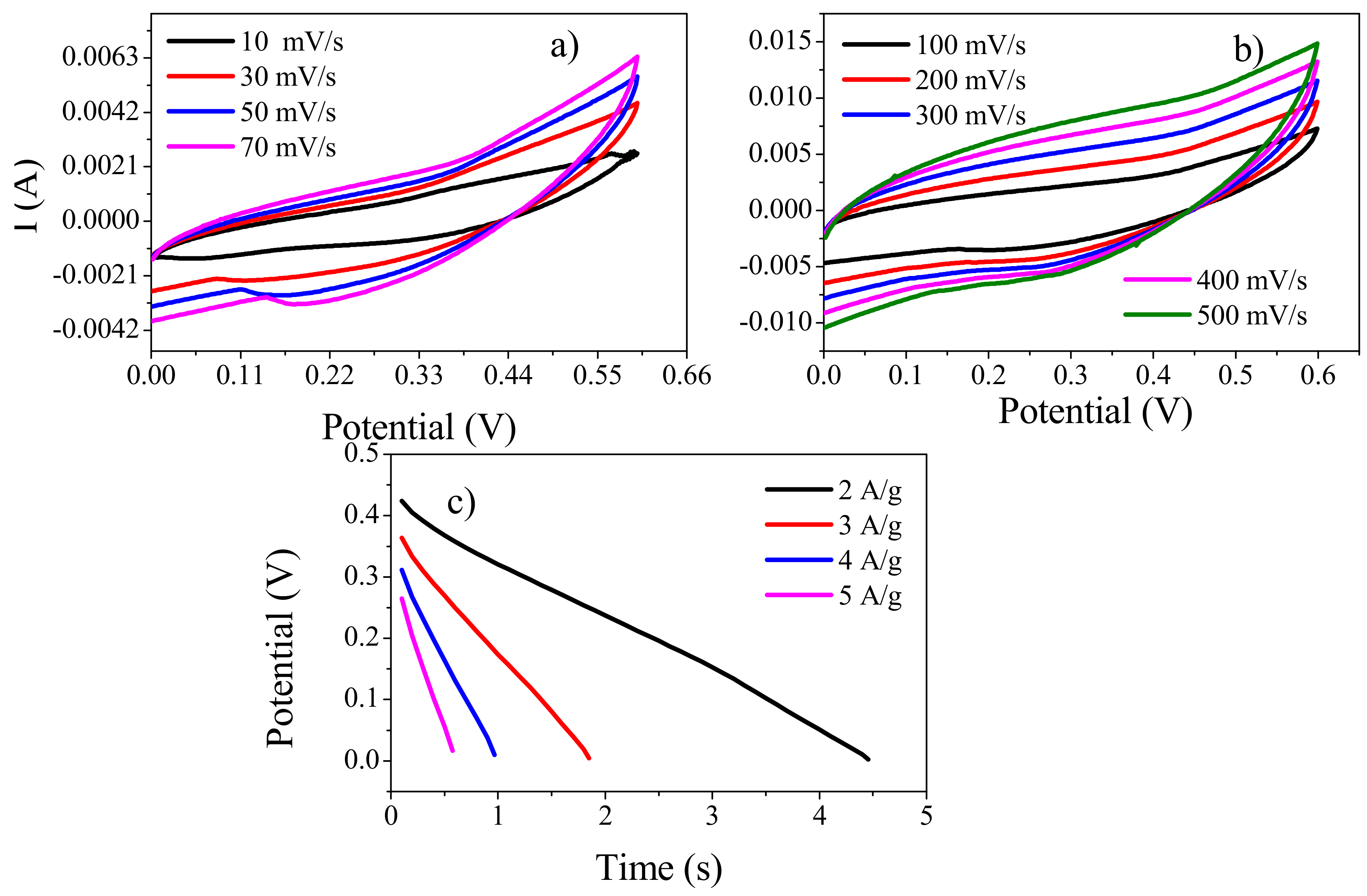
3.8.1. Cyclic Voltammetry
3.8.2. Galvanostatic Charge Discharge Analysis (GCD)
3.8.3. Electrochemical Impedance Spectroscopy (EIS)
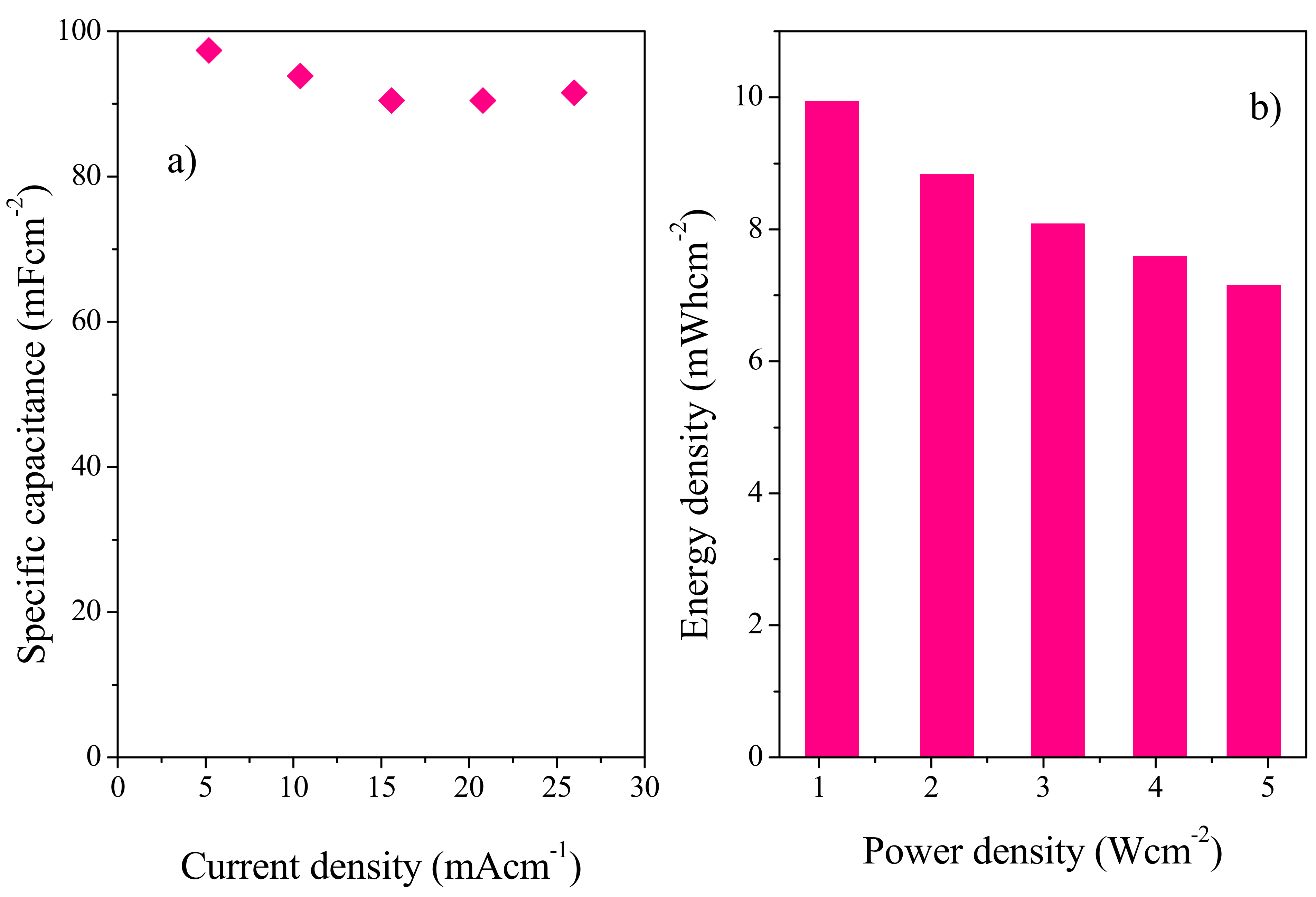
3.9. Solid State Symmetric Device Fabrication with Copper as Current Collector
3.9.1. Cyclic Voltammetry (CV)
3.9.2. Galvanostatic Charge Discharge (GCD) Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Anothumakkool, B.; Torris AT, A.; Bhange, S.N.; Unni, S.M.; Badiger, M.V.; Kurungot, S. Design of A High Performance Thin All-Solid-State Supercapacitor Mimicking the Active Interface of Its Liquid-State Counterpart. ACS Appl. Mater. Interfaces 2013, 5, 13397–13404. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumar, S.T.; Selvan, R.K.; Melo, J.S.; Sanjeeviraja, C. High Performance Solid-State Electric Double Layer Capacitor from Redox Mediated Gel Polymer Electrolyte and Renewable Tamarind Fruit Shell Derived Porous Carbon. ACS Appl. Mater. Interfaces 2013, 5, 10541–10550. [Google Scholar] [CrossRef] [PubMed]
- Yao, B.; Yuan, L.; Xiao, X.; Zhang, J.; Qi, Y.; Zhou, J.; Zhou, J.; Hu, B.; Chen, W. Paper-based Solid-state Supercapacitors with Pencil-Drawing Graphite/Polyaniline Networks Hybrid Electrodes. Nano Energy 2013, 2, 1071–1078. [Google Scholar] [CrossRef]
- Jyothibasu, J.; Lee, R.H. Facile, Scalable, Eco-Friendly Fabrication of High-Performance Flexible All-Solid-State Supercapacitors. Polymers 2018, 10, 1247. [Google Scholar] [CrossRef]
- Hong, X.; Fu, J.; Liu, Y.; Li, S.; Wang, X.; Dong, W.; Yang, S. Recent Progress on Graphene/Polyaniline Composites for High-performance Supercapacitors. Materials 2019, 12, 1451. [Google Scholar] [CrossRef]
- Wang, X.; Wu, D.; Song, X.; Du, W.; Zhao, X.; Zhang, D. Review on Carbon/Polyaniline Hybrids: Design and Synthesis for Supercapacitor. Molecules 2019, 24, 2263. [Google Scholar] [CrossRef]
- Dywili, N.R.; Ntziouni, A.; Ikpo, C.; Ndipingwi, M.; Hlongwa, N.W.; Yonkeu, A.L.; Masikini, M.; Kordatos, K.; Iwuoha, E.I. Graphene Oxide Decorated Nanometal-Poly (Anilino-Dodecylbenzene Sulfonic Acid) for Application in High Performance Supercapacitors. Micromachines 2019, 10, 115. [Google Scholar] [CrossRef]
- Guan, X.; Kong, D.; Huang, Q.; Cao, L.; Zhang, P.; Lin, H.; Lin, Z.; Yuan, H. In Situ Growth of a High-Performance All-Solid-State Electrode for Flexible Supercapacitors Based on a PANI/CNT/EVA Composite. Polymers 2019, 11, 178. [Google Scholar] [CrossRef]
- Cai, X.; Cui, X.; Zu, L.; Zhang, Y.; Gao, X.; Lian, H.; Liu, Y.; Wang, X. Ultra High Electrical Performance of Nano Nickel Oxide and Polyaniline Composite Materials. Polymers 2017, 9, 288. [Google Scholar] [CrossRef]
- Shahabuddin, S.; Sarih, N.; Afzal Kamboh, M.; Rashidi Nodeh, H.; Mohamad, S. Synthesis of Polyaniline-Coated Graphene Oxide@ SrTiO3 Nanocube Nanocomposites for Enhanced Removal of Carcinogenic Dyes from Aqueous Solution. Polymers 2016, 8, 305. [Google Scholar] [CrossRef]
- Hummers, W.S., Jr.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Gul, H.; Shah, A.A.; Krewer, U.; Bilal, S. Fabrication of Energy-Efficient Ultracapacitive Electrodes Based on Conducting Polyaniline-Graphene Oxide Composites. Nanomaterials 2019, 563159, submitted. [Google Scholar]
- Feng, X.M.; Li, R.M.; Ma, Y.W.; Chen, R.F.; Shi, N.E.; Fan, Q.L.; Huang, W. One-Step Electrochemical Synthesis of Graphene/Polyaniline Composite Film and Its Applications. Adv. Funct. Mater. 2011, 21, 2989–2996. [Google Scholar] [CrossRef]
- Bilal, S.; Gul, H.; Gul, S.; Shah, A.H.A. One Pot Synthesis of Highly Thermally Stable Poly (2-Methylaniline) for Corrosion Protection of Stainless Steel. Iran. J. Sci. Technol. A 2018, 42, 1915–1922. [Google Scholar] [CrossRef]
- Tabrizi, A.G.; Arsalani, N.; Namazi, H.; Ahadzadeh, I. Vanadium Oxide Assisted Synthesis of Polyaniline Nanoarrays on Graphene Oxide Sheets and Its Application in Supercapacitors. J. Electroanal. Chem. 2017, 798, 34–41. [Google Scholar] [CrossRef]
- Sovizi, M.R.; Gheshlaghi, Z.F.H. Enhancement in Electrochemical Performances of Li–S Batteries by Electrodeposition of Sulfur on Polyaniline-Dodecyl Benzene Sulfonic Acid-Sulfuric Acid (PANI–DBSA–H2O4) Honeycomb Structure film. New J. Chem. 2018, 42, 2711–2717. [Google Scholar] [CrossRef]
- Hu, F.; Li, W.; Zhang, J.; Meng, W. Effect of Graphene Oxide as A Dopant on the Electrochemical Performance of Graphene Oxide/Polyaniline Composite. J. Mater. Sci. Technol. 2014, 30, 321–327. [Google Scholar] [CrossRef]
- Zhao, Y.; Tang, G.S.; Yu, Z.Z.; Qi, J.S. The Effect of Graphite Oxide on The Thermoelectric Properties of Polyaniline. Carbon 2012, 50, 3064–3073. [Google Scholar] [CrossRef]
- Shao, W.; Jamal, R.; Xu, F.; Ubul, A.; Abdiryim, T. The Effect of a Small Amount of Water on the Structure and Electrochemical Properties of Solid-State Synthesized Polyaniline. Materials 2012, 5, 1811–1825. [Google Scholar] [CrossRef]
- Mousavi, M.F.; Hashemi, M.; Rahmanifar, M.S.; Noori, A. Synergistic Effect Between Redox Additive Electrolyte and PANI–rGO Nanocomposite Electrode for High Energy and High Power Supercapacitor. Electrochim. Acta 2017, 228, 290–298. [Google Scholar] [CrossRef]
- Liu, L.; Zhou, W.; Chen, Y.; Jiao, S.; Huang, P. Pressure-Assisted Synthesis of a Polyaniline-Graphite oxide (PANI–GO) Hybrid and Its Friction Reducing Behavior in Liquid Paraffin (LP). New J. Chem. 2018, 42, 936–942. [Google Scholar] [CrossRef]
- Ramana, G.V.; Padya, B.; Srikanth, V.V.; Jain, P.K.; Padmanabham, G.; Sundararajan, G. Electrically Conductive Carbon Nanopipe-Graphite Nanosheet/Polyaniline Composites. Carbon 2011, 49, 5239–5245. [Google Scholar] [CrossRef]
- Afzali, M.; Mostafavi, A.; Shamspur, T. Square Wave Voltammetric Determination of Anticancer Drug Flutamide Using Carbon Paste Electrode Modified by CuO/GO/PANI Nanocomposite. Arabian J. Chem. 2018. (In press) [CrossRef]
- Fan, X.; Phebus, B.D.; Li, L.; Chen, S. Graphene-Based Composites for Supercapacitor Electrodes. Sci. Adv. Mater. 2015, 7, 1916–1944. [Google Scholar] [CrossRef]
- Gaikwad, P.D.; Shirale, D.J.; Gade, V.K.; Savale, P.A.; Kharat, H.J.; Kakde, K.P.; Hussaini, S.S.; Dhumane, N.R.; Shirsat, M.D. Synthesis of H2SO4 Doped Polyaniline Film by Potentiometric Method. Bull. Mater. Sci. 2006, 29, 169–172. [Google Scholar] [CrossRef]
- Gul, H.; Shah, A.U.H.A.; Gul, S.; Arjomandi, J.; Bilal, S. Study on the Thermal Decomposition Kinetics and Calculation of Activation Energy of Degradation of Poly (o-toluidine) Using Thermogravimetric Analysis. Iran. J. Chem. Chem. Eng. 2018, 37, 193–204. [Google Scholar]
- Wang, L.; Ye, Y.; Lu, X.; Wen, Z.; Li, Z.; Hou, H.; Song, Y. Hierarchical Nanocomposites of Polyaniline Nanowire Arrays on Reduced Graphene Oxide Sheets for Supercapacitors. Sci. Rep. 2013, 3, 3568. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.Z.; Li, W.; Yu, Z.Z.; Rafiee, M.A.; Rafiee, J.; Yavari, F.; Koratkar, N. Enhanced Thermal Stability in Graphene Oxide Covalently Functionalized with 2-Amino-4, 6-Didodecylamino-1, 3, 5-Triazine. Carbon 2011, 49, 1258–1265. [Google Scholar] [CrossRef]
- Luo, J.; Jiang, S.; Wu, Y.; Chen, M.; Liu, X. Synthesis of Stable Aqueous Dispersion of Graphene/Polyaniline Composite Mediated by Polystyrene Sulfonic Acid. J. Polym. Sci. Part. A Polym. Chem. 2011, 50, 4888–4894. [Google Scholar] [CrossRef]
- Wang, J.; Wu, J.; Bai, H. Degradation-Induced Capacitance: A New Insight into the Superior Capacitive Performance of Polyaniline/Graphene Composites. Energy Environ. Sci. 2017, 10, 2372–2382. [Google Scholar]
- Bandyopadhyay, P.; Kuila, T.; Balamurugan, J.; Nguyen, T.T.; Kim, N.H.; Lee, J.H. Facile Synthesis of Novel Sulfonated Polyaniline Functionalized Graphene Using m-aminobenzene Sulfonic Acid for Asymmetric Supercapacitor Application. Chem Eng. J. 2017, 308, 1174–1184. [Google Scholar] [CrossRef]
- Ravi, B.; Rajender, B.; Palaniappan, S. Improving the Electrochemical Performance by Sulfonation of Polyaniline-Graphene-Silica Composite for High Performance Supercapacitor. Int. J. Polym. Mater. Polym. Biomater 2016, 65, 835–840. [Google Scholar] [CrossRef]
- Gui, D.; Liu, C.; Chen, F.; Liu, J. Preparation of Polyaniline/Graphene Oxide Nanocomposite for the Application of Supercapacitor. Appl. Surf. Sci. 2014, 307, 172–177. [Google Scholar] [CrossRef]
- Javed, M.S.; Dai, S.; Wang, M.; Guo, D.; Chen, L.; Wang, X.; Hu, C.; Xi, Y. High Performance Solid State Flexible Supercapacitor Based on Molybdenum Sulfide Hierarchical Nanospheres. J. Power Sources 2015, 285, 63–69. [Google Scholar] [CrossRef]
- Bilal, S.; Fahim, M.; Firdous, I.; Shah, A.H.A. Insight into Capacitive Performance of Polyaniline/Graphene Oxide Composites with Ecofriendly Binder. Appl. Surf. Sci. 2018, 435, 91–101. [Google Scholar] [CrossRef]
- Xu, H.; Tang, J.; Chen, Y.; Liu, J.; Pu, J.; Li, Q. Zn2+–Doped Polyaniline/Graphene Oxide as Electrode Material for Electrochemical Supercapacitors. J. Electron. Mater. 2017, 46, 6150–6157. [Google Scholar] [CrossRef]
- Li, Z.F.; Zhang, H.; Liu, Q.; Liu, Y.; Stanciu, L.; Xie, J. Covalently-Grafted Polyaniline on Graphene Oxide Sheets for High Performance Electrochemical Supercapacitors. Carbon 2014, 71, 257–267. [Google Scholar] [CrossRef]
- Misoon, O.; Seok, K. Effect of Dodecyl Benzene Sulfonic Acid on the Preparation of Polyaniline/Activated Carbon Composites by in Situ Emulsion Polymerization. Electrochim. Acta 2012, 59, 196–201. [Google Scholar] [CrossRef]
- Khalid, M.; Tumelero, M.A.; Pasa, A.A. Asymmetric and Symmetric Solid-State Supercapacitors Based on 3D Interconnected Polyaniline–Carbon Nanotube Framework. RSC Adv. 2015, 5, 62033–62039. [Google Scholar] [CrossRef]
- Qin, G.; Zhang, H.; Liao, H.; Li, Z.; Tian, J.; Lin, Y.; Zhang, D.; Wu, Q. Novel Graphene Nanosheet-Wrapped Polyaniline Rectangular-Like Nanotubes for Flexible All-Solid-State Supercapacitors. J. Mater. Sci. 2017, 52, 10981–10992. [Google Scholar] [CrossRef]
- Zheng, X.; Yu, H.; Xing, R.; Ge, X.; Sun, H.; Li, R.; Zhang, Q. Multi-Growth Site Graphene/Polyaniline Composites with Highly Enhanced Specific Capacitance and Rate Capability for Supercapacitor Application. Electrochim. Acta 2018, 260, 504–513. [Google Scholar] [CrossRef]
- Kurra, N.; Wang, R.; Alshareef, H.N. All Conducting Polymer Electrodes for Asymmetric Solid-State Supercapacitors. J. Mater. Chem. A 2015, 3, 7368–7374. [Google Scholar] [CrossRef]
- Zhao, X.; Gnanaseelan, M.; Jehnichen, D.; Simon, F.; Pionteck, J. Green and Facile Synthesis of Polyaniline/Tannic Acid/rGO Composites for Supercapacitor Purpose. J. Mater. Sci 2019, 54, 10809–10824. [Google Scholar] [CrossRef]
- Ghenaatian, H.R.; Mousavi, M.F.; Rahmanifar, M.S. High Performance Hybrid Supercapacitor Based on Two Nanostructured Conducting Polymers: Self-Doped Polyaniline and Polypyrrole Nanofibers. Electrochim. Acta. 2012, 78, 212–222. [Google Scholar] [CrossRef]
- Bilal, S.; Begum, B.; Gul, S.; Shah, A.H.A. PANI/DBSA/H2SO4: A Promising and Highly Efficient Electrode Material for Aqueous Supercapacitors. Synth. Met. 2018, 235, 1–15. [Google Scholar] [CrossRef]
- Purkait, T.; Singh, G.; Kumar, D.; Singh, M.; Dey, R.S. High-Performance Flexible Supercapacitors Based on Electrochemically Tailored Three-Dimensional Reduced Graphene Oxide Networks. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Li, K.; Liu, J.; Huang, Y.; Bu, F.; Zhao, B.; Xu, Y. Graphene/Polyaniline@carbon Cloth Composite as A High-Performance Flexible Supercapacitor Electrode Prepared by A One-Step Electrochemical Co-Deposition Method. RSC Adv. 2017, 7, 7688–7693. [Google Scholar] [CrossRef]
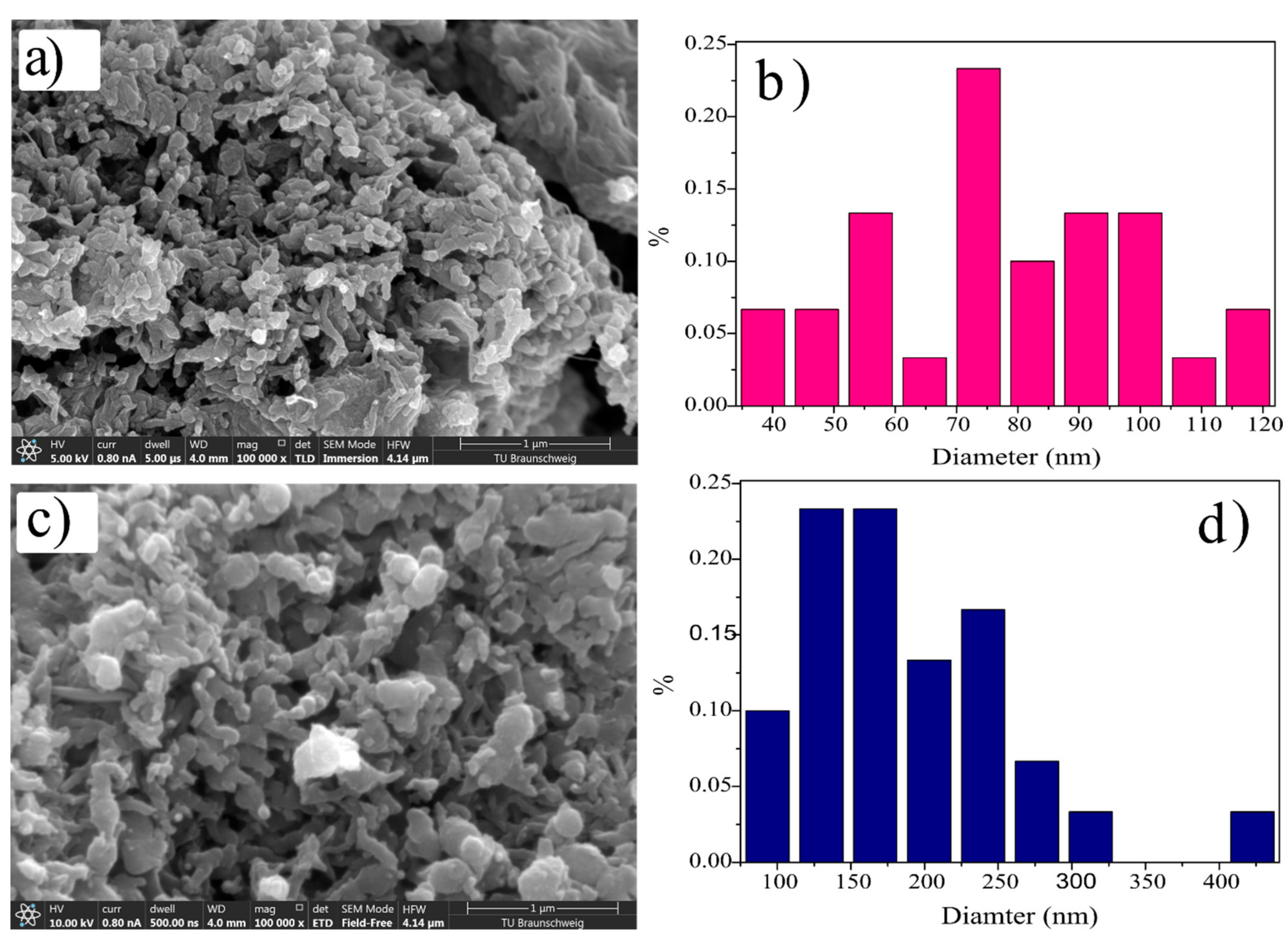
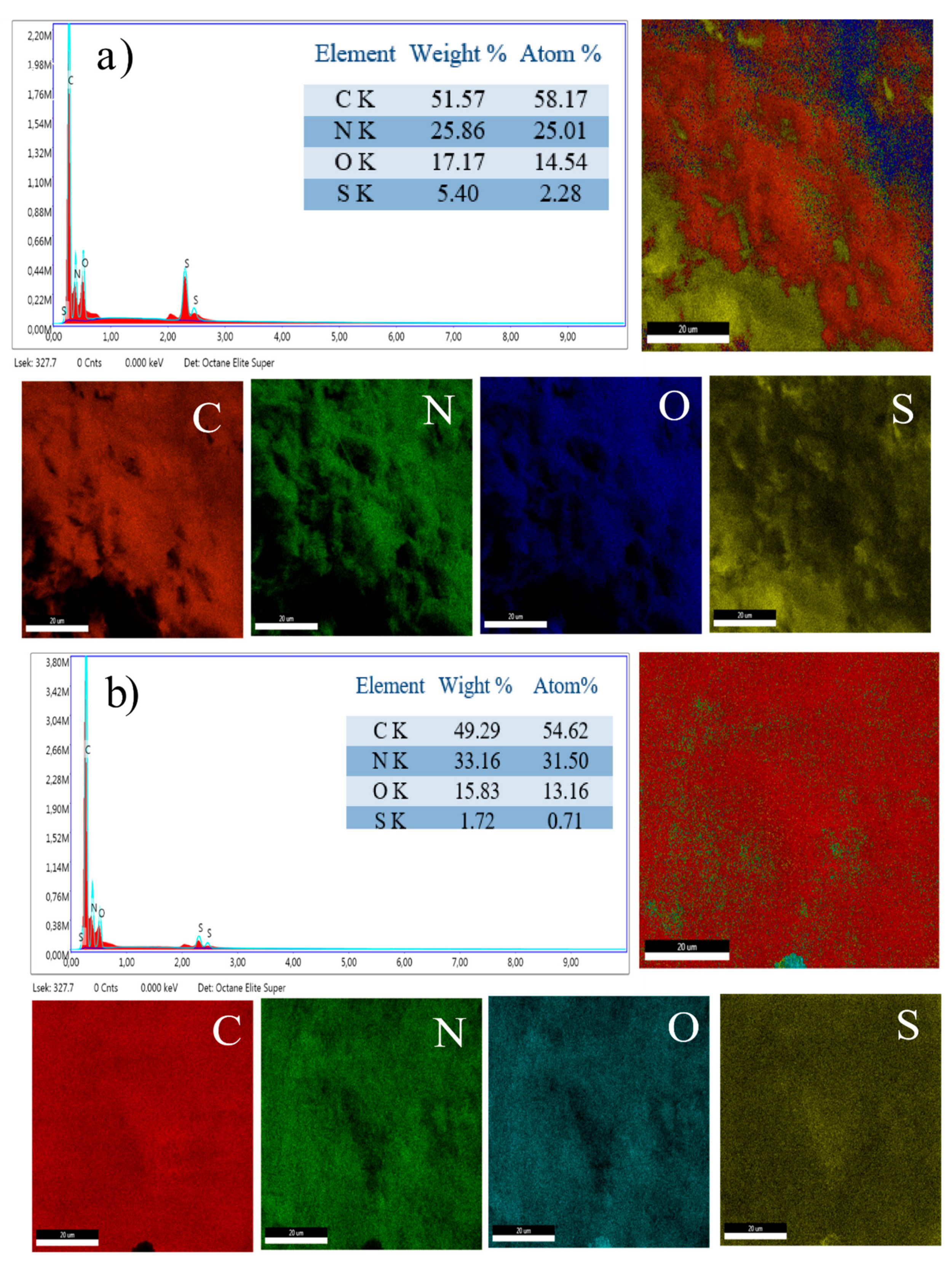

| S. No | Sample Name | Total | Max./nm | Min./nm | Mean/nm |
|---|---|---|---|---|---|
| 1 | ds@PANI/GO composite | 30 | 121.53 | 34.22 | 78.74 |
| 2 | cs@PANI/GO composite | 30 | 440.55 | 75.19 | 190.05 |
| S. No | Electrode Material | Electrolyte | Potential Window (V) | Current Density (A/g) | Specific Capacitance | Reference |
|---|---|---|---|---|---|---|
| 1 | Symmetrical PANI/Au/PEN | PVA/H2SO4 gel | 0–0.8 | 0.15 mA cm−2 | 29 mF cm−2 | [42] |
| 2 | PANI/rGO–TA–24 h | H2SO4–PVA | 0–0.5 | 0.5 A g−1 | 56.9 F g−1 | [43] |
| 3 | PANI/rGO–HH | H2SO4–PVA | 0–0.5 | 0.5 A g−1 | 46.0 F g−1 | [43] |
| 4 | Co-doped ds@PANI/GO | PVA/H2SO4 gel | 0–0.9 | 1 Ag−1 | 150 Fg−1 | Present work |
| 5 | Co-doped ds@PANI/GO | PVA/H2SO4 gel | 0–0.9 | 5 mAcm−2 | 97.38 Fcm−2 | Present work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gul, H.; Shah, A.-u.-H.A.; Bilal, S. Fabrication of Eco-Friendly Solid-State Symmetric Ultracapacitor Device Based on Co-Doped PANI/GO Composite. Polymers 2019, 11, 1315. https://doi.org/10.3390/polym11081315
Gul H, Shah A-u-HA, Bilal S. Fabrication of Eco-Friendly Solid-State Symmetric Ultracapacitor Device Based on Co-Doped PANI/GO Composite. Polymers. 2019; 11(8):1315. https://doi.org/10.3390/polym11081315
Chicago/Turabian StyleGul, Hajera, Anwar-ul-Haq Ali Shah, and Salma Bilal. 2019. "Fabrication of Eco-Friendly Solid-State Symmetric Ultracapacitor Device Based on Co-Doped PANI/GO Composite" Polymers 11, no. 8: 1315. https://doi.org/10.3390/polym11081315
APA StyleGul, H., Shah, A.-u.-H. A., & Bilal, S. (2019). Fabrication of Eco-Friendly Solid-State Symmetric Ultracapacitor Device Based on Co-Doped PANI/GO Composite. Polymers, 11(8), 1315. https://doi.org/10.3390/polym11081315








