High-Level Conversion of l-lysine into Cadaverine by Escherichia coli Whole Cell Biocatalyst Expressing Hafnia alvei l-lysine Decarboxylase
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains, Plasmids, Genes, and Chemicals
2.2. Plasmid Construction
2.3. Purification of E. coli and H. alvei l-lysine Decarboxylase for In Vitro Analysis of Enzyme Activities
2.4. Whole Cell Bioconversion of l-Lysine into Cadaverine by the Recombinant E. coli Strains Expressing E. coli and H. alvei l-Lysine Decarboxylase
2.5. Purification of Crude Cadaverine Soluton from the Whole Cell Bioconversion of l-lysine
2.6. Polymerization of Purified Cadaverine for the Synthesis of Bio-Based Polyamide 5,10
2.7. Analytical Procedures
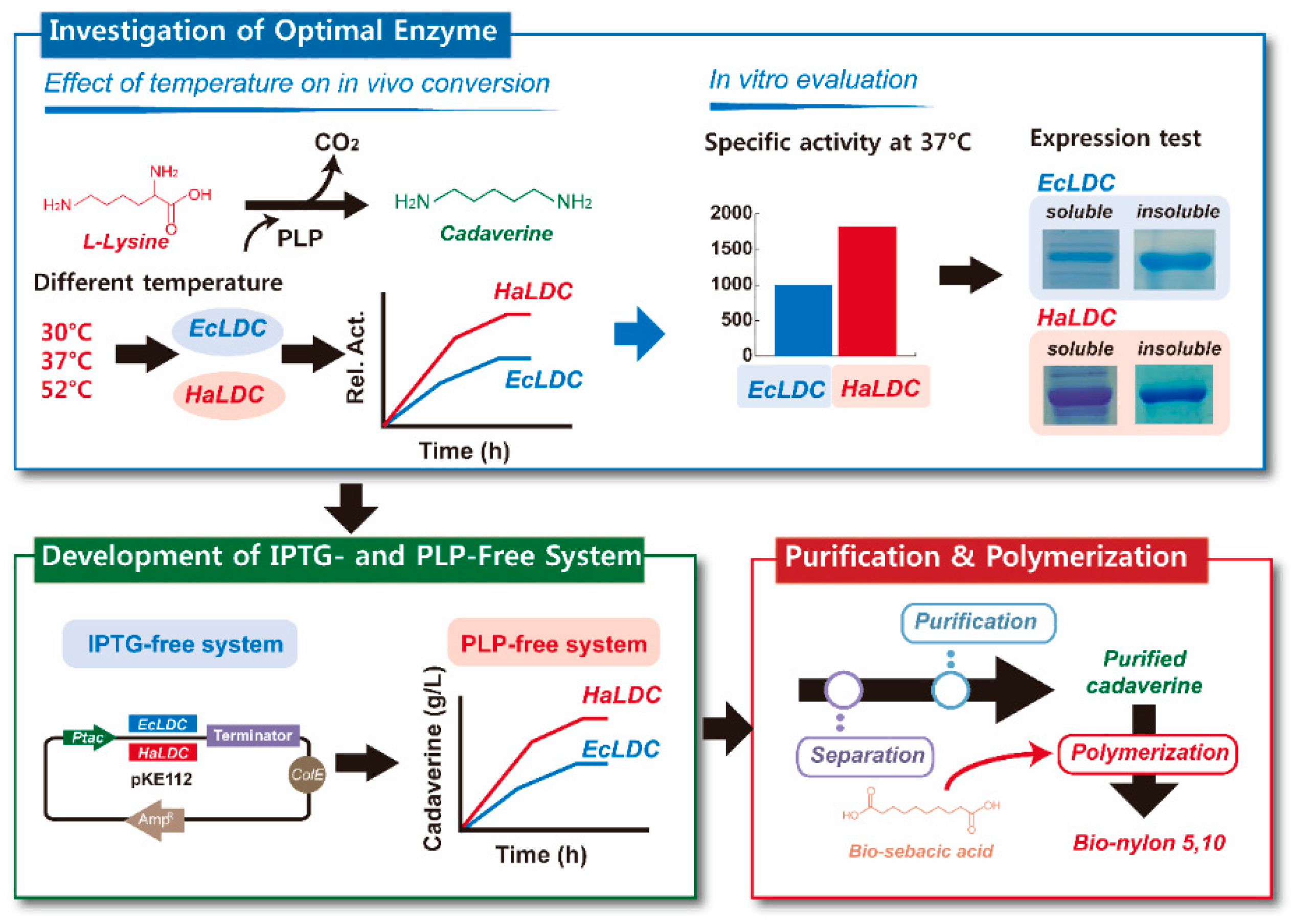
3. Results and Discussion
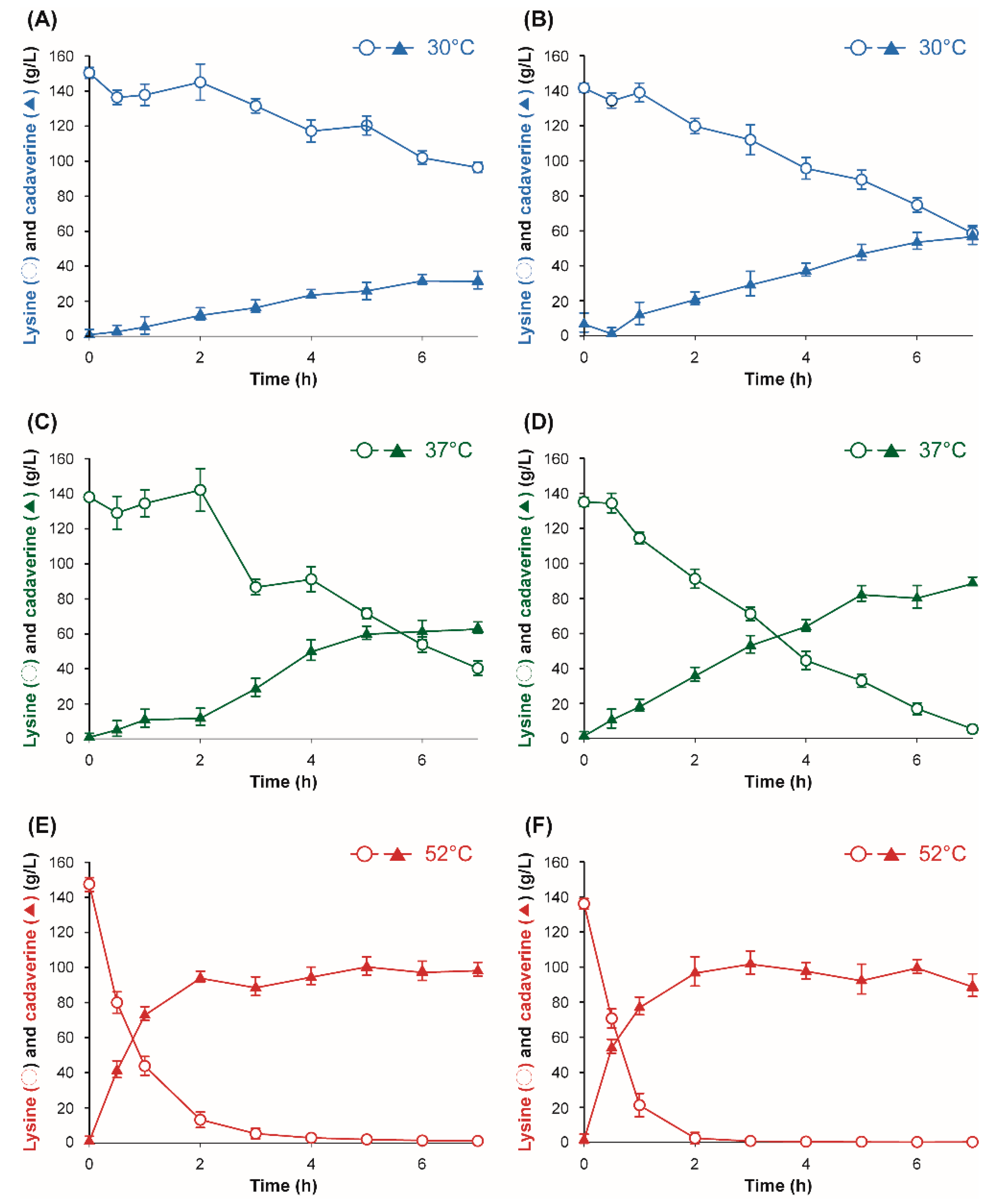
3.1. Characterization of Lysine Decarboxylase from E. coli and H. alvei
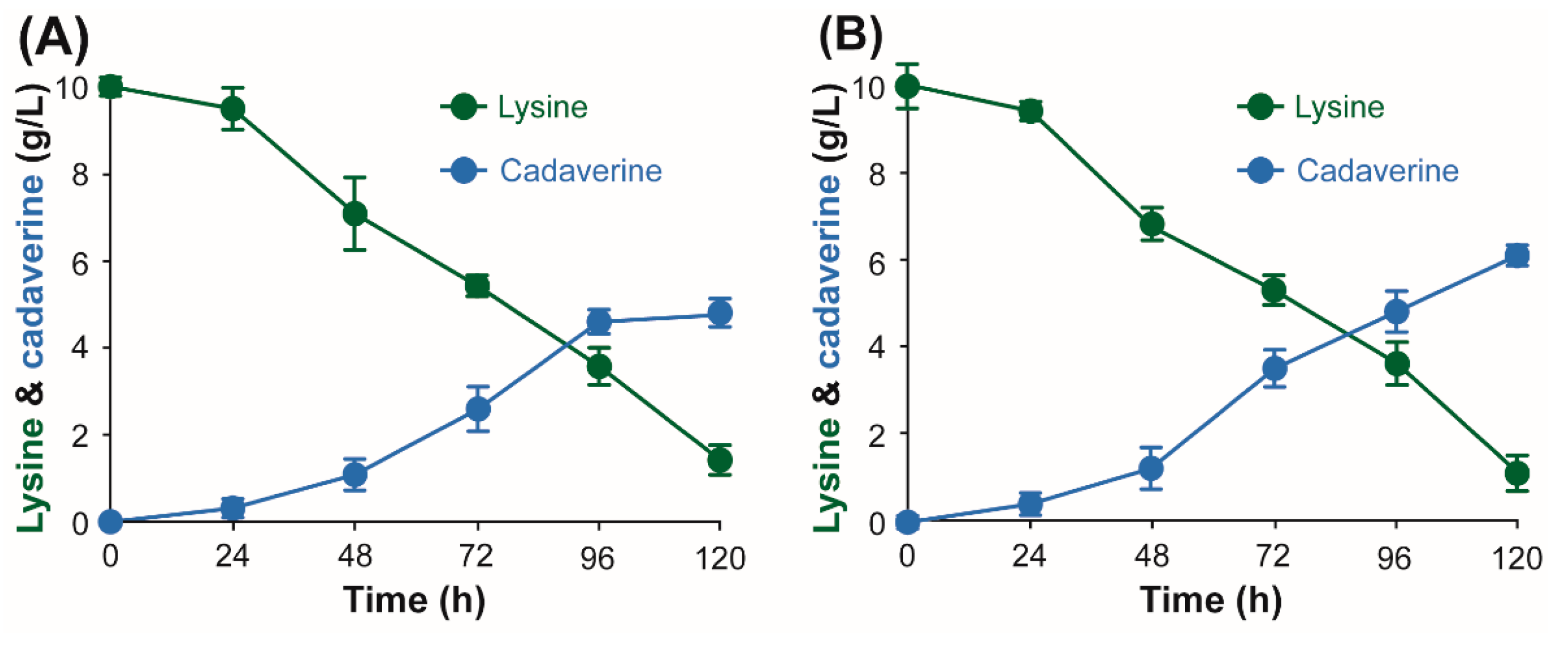
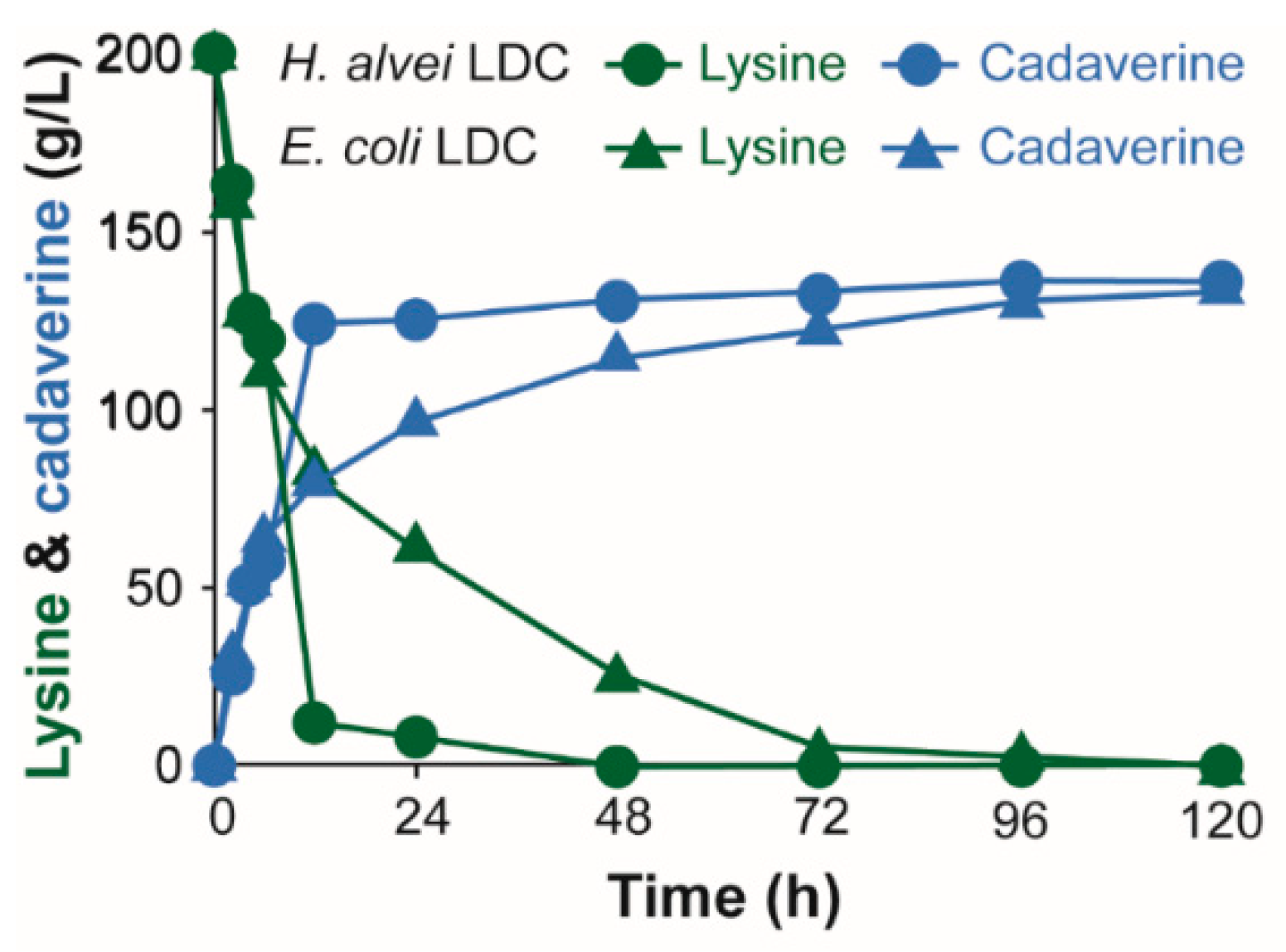
3.2. Development of IPTG-Free System for Whole Cell Bioconversion of l-Lysine into Cadaverine Using Lysine Decarboxylase from H. alvei and E. coli
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Becker, J.; Wittmann, C. Advanced Biotechnology: Metabolically Engineered Cells for the Bio-Based Production of Chemicals and Fuels, Materials, and Health-Care Products. Angew. Chem. Int. Ed. 2015, 54, 2–25. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.H.; Eom, I.Y.; Joo, J.C.; Yu, J.H.; Song, B.G.; Lee, S.H.; Hong, S.H.; Park, S.J. Recent advances in development of biomass pretreatment technologies used in biorefinery for the production of bio-based fuels, chemicals and polymers. Korean J. Chem. Eng. 2015, 32, 1945–1959. [Google Scholar] [CrossRef]
- Lee, J.W.; Kim, H.U.; Choi, S.; Yi, J.H.; Lee, S.Y. Microbial production of building block chemicals and polymers. Curr. Opin. Biotechnol. 2011, 22, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Baritugo, K.; Kim, H.T.; David, Y.; Choi, J.H.; Choi, J.; Kim, T.W.; Park, C.; Hong, S.H.; Na, J.G.; Jeong, K.J.; et al. Recent advances in metabolic engineering of Corynebacterium glutamicum strains as potential platform microorganisms for biorefinery. Biofuel Bioprod. Biorefining 2018, 12, 899–925. [Google Scholar] [CrossRef]
- Baritugo, K.; Kim, H.T.; David, Y.; Choi, J.; Hong, S.H.; Jeong, K.J.; Joo, J.C.; Park, S.J. Metabolic engineering of Corynebacterium glutamicum for fermentative production of chemicals in biorefinery. Appl. Microbiol. Biotechnol. 2018, 102, 3915–3937. [Google Scholar] [CrossRef] [PubMed]
- Joo, J.C.; Khusnutdinova, A.N.; Flick, R.; Kim, T.H.; Bornscheuer, U.T.; Yakunin, A.F.; Mahadevan, R. Alkene hydrogenation activity of enoate reductases for an environmentally benign biosynthesis of adipic acid. Chem. Sci. 2017, 8, 1406–1413. [Google Scholar] [CrossRef] [PubMed]
- Joo, J.C.; Oh, Y.H.; Yu, J.H.; Hyun, S.M.; Khang, T.U.; Kang, K.H.; Song, B.K.; Park, K.; Oh, M.K.; Lee, S.Y.; et al. Production of 5-aminovaleric acid in recombinant Corynebacterium glutamicum strains from a Miscanthus hydrolysate solution prepared by a newly developed Miscanthus hydrolysis process. Bioresour. Technol. 2017, 244, 1692–1700. [Google Scholar] [CrossRef]
- David, Y.; Baylon, M.G.; Sudheer, P.D.V.N.; Baritugo, K.; Chae, C.G.; Kim, Y.J.; Kim, T.W.; Kim, M.; Na, J.G.; Park, S.J. Screening of microorganisms able to degrade low-rank coal in aerobic conditions: Potential coal bio-solubilization mediators from coal to biochemical. Biotechnol. Bioprocess Eng. 2017, 22, 178–185. [Google Scholar] [CrossRef]
- Andin, N.; Longieras, A.; Veronese, T.; Marcato, F.; Molina-Jouve, C. Improving carbon and energy distribution by coupling growth and medium chain length polyhydroxyalkanote production from fatty acids by Pseudomonas putida KT2440. Biotechnol. Bioprocess Eng. 2017, 22, 308–318. [Google Scholar] [CrossRef]
- Rhie, M.N.; Kim, H.T.; Jo, S.Y.; Chu, L.L.; Baritugo, K.; Baylon, M.G.; Lee, J.; Na, J.; Kim, L.H.; Kim, T.W.; et al. Recent advances in the Metabolic Engineering of Klebsiella pneumoniae: A Potential Platform Microorganism for Biorefineries. Biotechnol. Bioprocess Eng. 2019, 24, 48–64. [Google Scholar] [CrossRef]
- Kim, H.S.; Oh, Y.H.; Jang, Y.; Kang, K.H.; David, Y.; Yu, J.H.; Song, B.K.; Choi, J.; Chang, Y.K.; Joo, J.C.; et al. Recombinant Ralstonia eutropha engineered to utilize xylose and its use for the production of poly(3-hydroxybutyrate) from sunflower stalk hydrolysate solution. Microb. Cell Factories 2016, 15, 95. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Jang, Y.A.; Noh, W.; Oh, Y.H.; Lee, H.; David, Y.; Baylon, M.G.; Shin, J.; Yang, J.E.; Choi, S.Y.; et al. Metabolic engineering of Ralstonia eutropha for the production of polyhydroxyalkanoates from sucrose. Biotechnol. Bioeng. 2015, 112, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Chae, T.U.; Kim, W.J.; Choi, S.; Park, S.J.; Lee, S.Y. Metabolic engineering of Escherichia coli for the production of 1,3-diaminopropane, a three-carbon diamine. Sci. Rep. 2015, 5, 13040. [Google Scholar] [CrossRef] [PubMed]
- Sudheer, P.D.V.N.; David, Y.; Chae, C.G.; Kim, Y.J.; Baylon, M.G.; Baritugo, K.A.; Kim, T.W.; Kim, M.S.; Na, J.G.; Park, S.J. Advances in biological treatment of coal for synthetic natural gas and chemicals. Korean J. Chem. Eng. 2016, 33, 2788–2801. [Google Scholar] [CrossRef]
- Ghasemlou, M.; Daver, F.; Ivanova, E.P.; Adhikari, B. Bio-based routes to synthesize cyclic carbonates and polyamines precursors of non–isocyanate polyurethanes: A review. Eur. Polym. J. 2019. [Google Scholar] [CrossRef]
- Choi, S.Y.; Parks, S.J.; Kim, W.J.; Yang, J.E.; Lee, H.; Shin, J.; Lee, S.Y. One-step fermentative production of poly(lactate-co-glycolate) from carbohydrates in Escherichia coli. Nat. Biotechnol. 2016, 34, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Chae, C.G.; Kim, Y.J.; Lee, S.J.; Oh, Y.H.; Yang, J.E.; Joo, J.C.; Kang, K.H.; Jang, Y.A.; Lee, H.; Park, A.R.; et al. Biosynthesis of poly(2-hydroxybutyrate-co-lactate) in metabolically engineered Escherichia coli. Biotechnol. Bioprocess Eng. 2016, 21, 169–174. [Google Scholar] [CrossRef]
- Yang, J.E.; Park, S.J.; Kim, W.J.; Kim, H.J.; Kim, B.; Lee, H.; Shin, J.; Lee, S.Y. One-step fermentative production of aromatic polyesters from glucose by metabolically engineered Escherichia coli strains. Nat. Commun. 2018, 9, 79. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Lama, S.; Kim, J.R.; Park, S.H. Production of 1,3-Propanediol from Glucose by Recombinant Escherichia coli BL21(DE3). Biotechnol. Bioprocess Eng. 2018, 23, 250–258. [Google Scholar] [CrossRef]
- Li, J.; Feng, R.; Wen, Z.; Zhang, A. Overexpression of ARO10 in pdc5Δmutant resulted in higher isobutanol titers in Saccharomyces cerevisiae. Biotechnol. Bioprocess Eng. 2017, 22, 382–389. [Google Scholar] [CrossRef]
- Zong, H.; Liu, X.; Chen, W.; Zhuge, B.; Sun, J. Construction of glycerol synthesis pathway in Klebsiella pneumoniae for bioconversion of glucose into 1,3-propanediol. Biotechnol. Bioprocess Eng. 2017, 22, 549–555. [Google Scholar] [CrossRef]
- Ravikumar, S.; David, Y.; Park, S.J.; Choi, J. A chimeric two-component regulatory system-based Escherichia coli biosensor engineered to detect glutamate. Appl. Biochem. Biotechnol. 2018, 186, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.; Wendisch, V.F. Biotechnological production of polyamines by Bacteria: Recent achievements and future perspectives. Appl. Microbiol. Biotechnol. 2011, 91, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.H.; Park, S.H.; Oh, Y.H.; Choi, J.W.; Lee, M.H.; Cho, J.S.; Jeong, K.J.; Joo, J.C.; Yu, J.; Park, S.J.; et al. Metabolic engineering of Corynebacterium glutamicum for enhanced production of 5-aminovaleric acid. Microb. Cell Fact. 2016, 15, 174. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.T.; Kang, T.U.; Baritugo, K.; Hyun, S.M.; Kang, K.H.; Jung, S.H.; Song, B.K.; Park, K.; Oh, M.; Kim, G.B.; et al. Metabolic engineering of Corynebacterium glutamicum for the production of glutaric acid, a C5 dicarboxylic acid platform chemical. Metab. Eng. 2018, 51, 99–109. [Google Scholar] [CrossRef]
- Choi, J.W.; Yim, S.S.; Lee, S.H.; Kang, T.J.; Park, S.J.; Jeong, K.J. Enhanced production of gamma-aminobutyrate (GABA) in recombinant Corynebacterium glutamicum by expressing glutamate decarboxylase active in expanded pH range. Microb. Cell Factories 2015, 14, 21. [Google Scholar] [CrossRef]
- Baritugo, K.; Kim, H.T.; David, Y.; Khang, T.U.; Hyun, S.M.; Kang, K.H.; Yu, J.H.; Choi, J.H.; Song, J.J.; Joo, J.C.; et al. Enhanced production of gamma-aminobutyrate (GABA) in recombinant Corynebacterium glutamicum strains from empty fruit bunch biosugar solution. Microb. Cell Factories 2018, 17, 129. [Google Scholar] [CrossRef]
- Baritugo, K.; Kim, H.T.; Rhie, M.N.; Jo, S.Y.; Khang, T.U.; Kang, K.H.; Sog, B.K.; Lee, B.; Song, J.J.; Choi, J.H.; et al. Construction of a Vitreoscilla Hemoglobin Promoter-based Tunable Expression System for Corynebacterium glutamicum. Catalysts 2018, 8, 561. [Google Scholar] [CrossRef]
- Oh, Y.H.; Choi, J.W.; Kim, E.Y.; Song, B.K.; Jeong, K.J.; Park, K.; Kim, I.; Woo, H.M.; Lee, S.H.; Park, S.J. Construction of Synthetic Promoter-Based Expression Cassettes for the production of Cadaverine in Recombinant Corynebacterium glutamicum. Appl. Biochem. Biotech. 2015, 176, 2065–2075. [Google Scholar] [CrossRef]
- Kind, S.; Neubauer, S.; Becker, J.; Yamamoto, M.; Völkert, M.; Abendroth, G.; Zelder, O.; Wittmann, C. From zero to hero-production of bio-based nylon from renewable re-sources using engineered Corynebacterium glutamicum. Metab. Eng. 2014, 25, 113–123. [Google Scholar] [CrossRef]
- Kind, S.; Wittmann, C. Bio-based production of the platform chemical 1, 5-diaminopentane. Appl. Microbiol. Biotechnol. 2011, 91, 1287–1296. [Google Scholar] [CrossRef] [PubMed]
- Kind, S.; Jeong, W.K.; Schroder, H.; Wittmann, C. Systems-wide metabolic pathway engineering in Corynebacterium glutamicum for bio-based production of diaminopentane. Metab. Eng. 2010, 12, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Mimitsuka, T.; Sawai, H.; Hatsu, M.; Yamada, K. Metabolic engineering of Corynebacterium glutamicum for cadaverine fermentation. Biosci. Biotechnol. Biochem. 2007, 71, 2130–2135. [Google Scholar] [CrossRef] [PubMed]
- Tateno, T.; Okada, Y.; Tsuchidate, T.; Tanaka, T.; Fukuda, H.; Kondo, A. Direct production of cadaverine from soluble starch using Corynebacterium glutamicum coexpressing α-amylase and L-lysine decarboxylase. Appl. Microbiol. Biotechnol. 2009, 82, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.T.; Baritugo, K.A.; Oh, Y.H.; Hyun, S.M.; Khang, T.U.; Kang, K.H.; Jung, S.H.; Song, B.K.; Park, K.; Kim, I.K.; et al. Metabolic engineering of Corynebacterium glutamicum for the high-level production of cadaverine that can be used for synthesis of biopolyamide 510. ACS Sustain. Chem. Eng. 2018, 6, 5296–5305. [Google Scholar] [CrossRef]
- Qian, Z.G.; Xia, X.; Lee, S.Y. Metabolic engineering of Escherichia coli for the production of cadaverine: A five carbon diamine. Biotechnol. Bioeng. 2011, 108, 93–103. [Google Scholar] [CrossRef]
- Park, S.J.; Kim, E.Y.; Noh, W.; Park, H.M.; Oh, Y.H.; Lee, S.H.; Song, B.K.; Jegal, J.; Lee, S.Y. Metabolic engineering of Escherichia coli for the production of 5-aminovalerate and glutarate as C5 platform chemicals. Metab. Eng. 2013, 16, 42–47. [Google Scholar] [CrossRef]
- Park, S.J.; Oh, Y.H.; Noh, W.; Kim, H.Y.; Shin, J.H.; Lee, E.G.; Lee, S.; David, Y.; Baylon, M.G.; Song, B.K.; et al. High-level conversion of L-lysine into 5-aminovalerate that can be used for nylon 6,5 synthesis. Biotechnol. J. 2014, 9, 1322–1328. [Google Scholar] [CrossRef]
- Park, S.J.; Kim, E.Y.; Won, N.; Oh, Y.H.; Kim, H.Y.; Song, B.K.; Cho, K.M.; Hong, S.H.; Lee, S.H.; Jegal, J. Synthesis of nylon 4 from gamma-aminobutyrate (GABA) produced by recombinant Escherichia coli. Bioprocess Biosyst. Eng. 2013, 36, 885–892. [Google Scholar] [CrossRef]
- Oh, Y.H.; Kang, K.H.; Kwon, M.J.; Choi, J.W.; Joo, J.C.; Lee, S.H.; Yang, Y.H.; Song, B.K.; Kim, I.K.; Yoon, K.H.; et al. Development of engineered Escherichia coli whole-cell biocatalysts for high-level conversion of l-L-lysine into cadaverine. J. Ind. Microbiol. Biotechnol. 2015, 42, 1481–1491. [Google Scholar] [CrossRef]
- Nishi, K.; Endo, S.; Mori, Y.; Totsuka, K.; Hirao, Y. Method for producing cadaverine dicarboxylate and its use for the production of nylon. Eur. Pat. 2006, EP1482055. [Google Scholar]
- Ma, W.; Cao, W.; Zhang, H.; Chen, K.; Li, Y.; Ouyang, P. Enhanced cadaverine production from L-lysine using recombinant Escherichia coli co-overexpressing CadA and CadB. Biotechnol. Lett. 2015, 37, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Joo, J.C.; Lee, E.; Hyun, S.M.; Kim, H.J.; Park, S.J.; Yang, Y.H.; Park, K. Characterization of a whole-cell biotransformation using a constitutive lysine decarboxylase from Escherichia coli for the high-level production of cadaverine from industrial grade L-lysine. Appl. Biochem. Biotechnol. 2018, 185, 909–924. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Zhuang, X.; Tang, Z.; Chen, X. Polylactic acid (PLA): Research, development and industrialization. Biotechnol. J. 2010, 5, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Guo, B.H. Poly (butylene succinate) and its copolymers: Research, development and industrialization. Biotechnol. J. 2010, 5, 1149–1163. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zhang, K.; Zhongjun, C.; Cai, H.; Honggui, W.; Ouyang, P. Directed Evolution and Mutagenesis of L-lysine Decarboxylase from Hafnia alvei AS1.1009 to improve its activity toward efficient cadaverine production. Biotechnol. Bioprocess. Eng. 2015, 20, 439–446. [Google Scholar] [CrossRef]
- Beier, H.; Fecker, L.F.; Berlin, J. Lysine decarboxylase from Hafnia alvei: Purification, molecular data and preparation of polyclonal antibodies. Z. Nat. C 1987, 42, 1307–1312. [Google Scholar] [CrossRef]
- Hong, E.Y.; Lee, S.G.; Park, B.J.; Lee, J.M.; Yun, H.; Kim, B.G. Simultaneously Enhancing the Stability and Catalytic Activity of Multimeric Lysine Decarboxylase CadA by Engineering Interface Regions for Enzymatic Production of Cadaverine at High Concentration of Lysine. Biotechnol. J. 2017, 12, 1700278. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Miwa, Y.; Miyoshi, K.; Furuyama, J.; Ohmori, H. The Escherichia coli ldcC gene encodes another L-lysine decarboxylase, probably a constitutive enzyme. Genes Genet. Syst. 1997, 72, 167–172. [Google Scholar] [CrossRef]
- Fecker, L.; Beier, H.; Berlin, J. Cloning and characterization of a L-lysine decarboxylase gene from Hafnia alvei. Mol. Gen. Genet. 1986, 203, 177–184. [Google Scholar] [CrossRef]
| Strain/Plasmid | Relevant Characteristics | Ref. |
|---|---|---|
| Strain | ||
| XL1-Blue | recA1 endA1 gyrA96 thi-1 hsdR17 supE44 relA1 lac [FA1proAB lacIqZΔacI Tn10 (TetR)] | [40] |
| BL21(DE3) | F- dcm ompT hsdS(rBmB-) gal λ(DE3) | [40] |
| XBHaLDC | E. coli XL1-Blue with pKE112-HaLdcC | This study |
| XBEcLDC | E. coli XL1-Blue with pKE112-EcLdcC | This study |
| BL21HaLDC | E. coli BL21(DE3) with pET22b-HaLdcC | This study |
| BL21EcLDC | E. coli BL21(DE3) with pET22b-EcLdcC | This study |
| Plasmids | ||
| pKE112-MCS | Expression vector; tac promoter, Ralstonia eutropha PHA biosynthesis genes transcription terminator; Apr | [40] |
| pKE112-EcLdcC | pKE112-MCS derivative, tac promoter, the E. coli ldcC gene, Apr | This study |
| pKE112-HaLdcC | pKE112-MCS derivative, tac promoter, the H. alvei ldc gene, Apr | This study |
| pET22b-EcLdcC | pET22b-MCS derivative, T7 promoter, the E. coli ldc gene, Apr | This study |
| pET22b-HaLdcC | pET22b-MCS derivative, T7 promoter, the H. alvei ldcC gene, Apr | This study |
| pET24ma-EcLdcC | pET24ma-MCS derivative, T7 promoter, the E. coli ldcC gene, Kmr | This study |
| pET24ma-HaLdcC | pET24ma-MCS derivative, T7 promoter, the H. alvei ldc gene, Kmr | This study |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.T.; Baritugo, K.-A.; Oh, Y.H.; Kang, K.-H.; Jung, Y.J.; Jang, S.; Song, B.K.; Kim, I.-K.; Lee, M.O.; Hwang, Y.T.; et al. High-Level Conversion of l-lysine into Cadaverine by Escherichia coli Whole Cell Biocatalyst Expressing Hafnia alvei l-lysine Decarboxylase. Polymers 2019, 11, 1184. https://doi.org/10.3390/polym11071184
Kim HT, Baritugo K-A, Oh YH, Kang K-H, Jung YJ, Jang S, Song BK, Kim I-K, Lee MO, Hwang YT, et al. High-Level Conversion of l-lysine into Cadaverine by Escherichia coli Whole Cell Biocatalyst Expressing Hafnia alvei l-lysine Decarboxylase. Polymers. 2019; 11(7):1184. https://doi.org/10.3390/polym11071184
Chicago/Turabian StyleKim, Hee Taek, Kei-Anne Baritugo, Young Hoon Oh, Kyoung-Hee Kang, Ye Jean Jung, Seyoung Jang, Bong Keun Song, Il-Kwon Kim, Myung Ock Lee, Yong Taek Hwang, and et al. 2019. "High-Level Conversion of l-lysine into Cadaverine by Escherichia coli Whole Cell Biocatalyst Expressing Hafnia alvei l-lysine Decarboxylase" Polymers 11, no. 7: 1184. https://doi.org/10.3390/polym11071184
APA StyleKim, H. T., Baritugo, K.-A., Oh, Y. H., Kang, K.-H., Jung, Y. J., Jang, S., Song, B. K., Kim, I.-K., Lee, M. O., Hwang, Y. T., Park, K., Park, S. J., & Joo, J. C. (2019). High-Level Conversion of l-lysine into Cadaverine by Escherichia coli Whole Cell Biocatalyst Expressing Hafnia alvei l-lysine Decarboxylase. Polymers, 11(7), 1184. https://doi.org/10.3390/polym11071184








