POSS Compounds as Modifiers for Rigid Polyurethane Foams (Composites)
Abstract
1. Introduction
2. Experimental Section
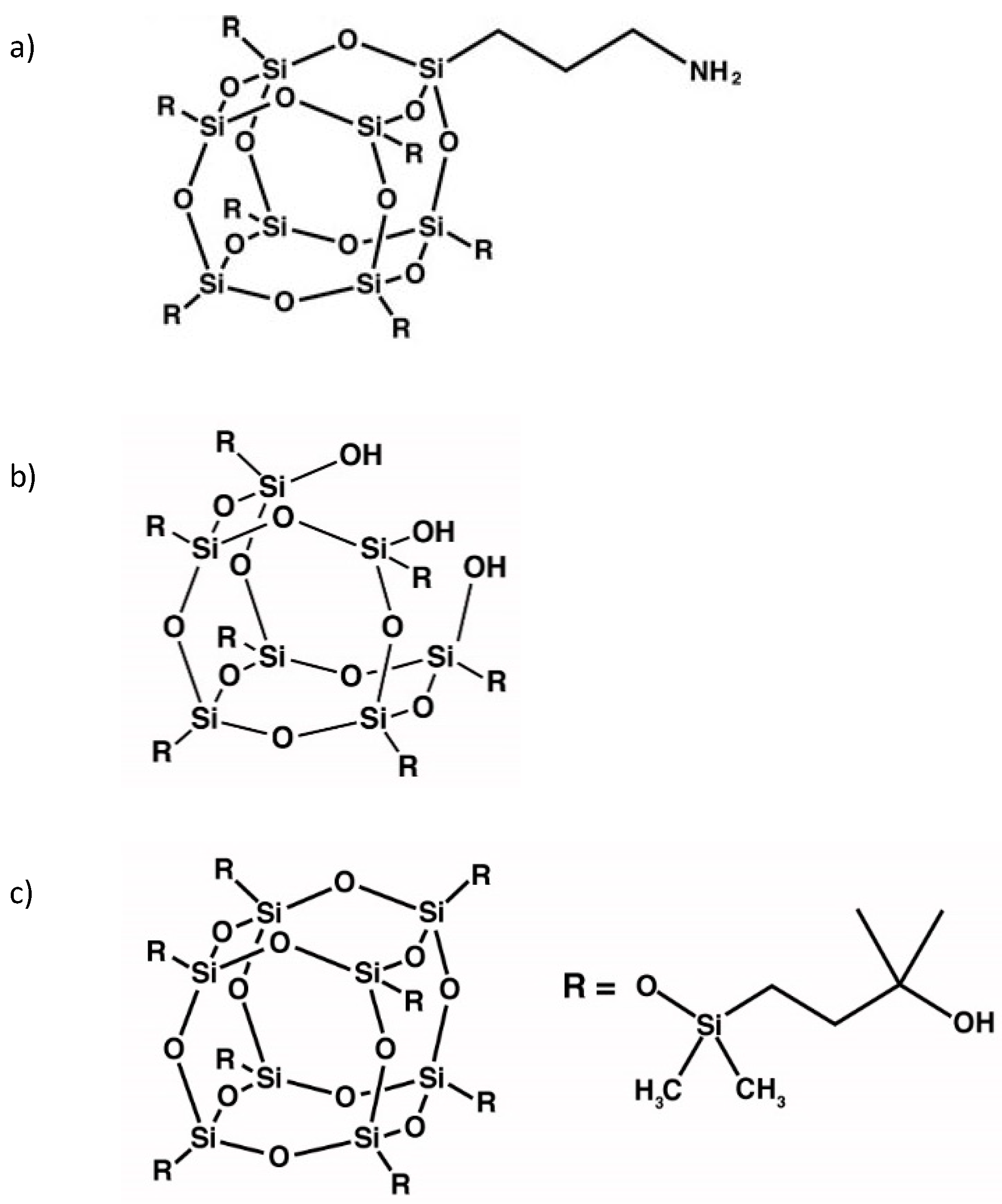
2.1. Materials
2.2. Manufacturing of RPUFs
2.3. Characterization Techniques
3. Results and Discussion
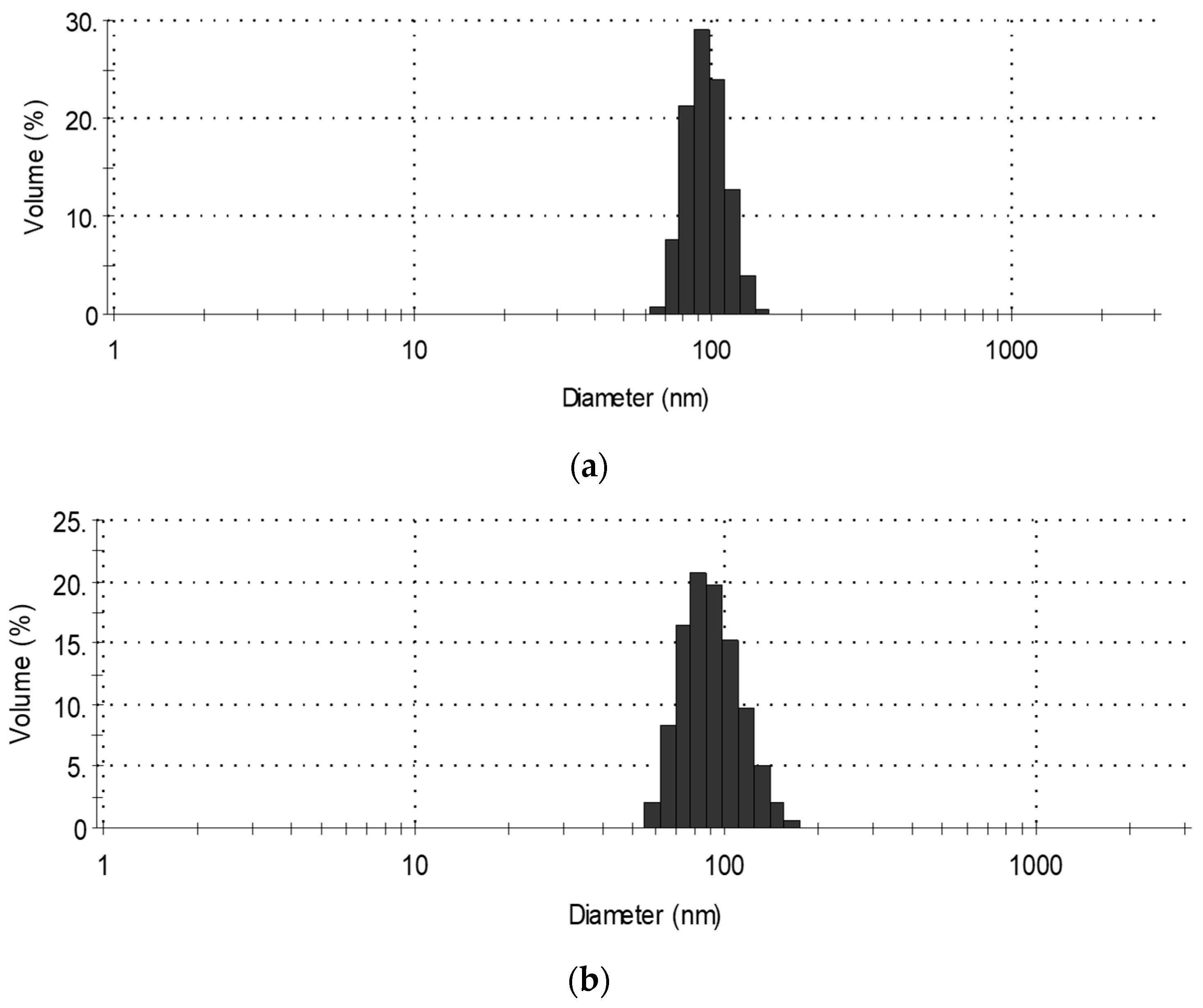
3.1. Average Size of POSS Powder Particles and the Dispersion of POSS-Modified Polyol Premixes
3.2. Impact of POSS on PU Mixture Viscosity
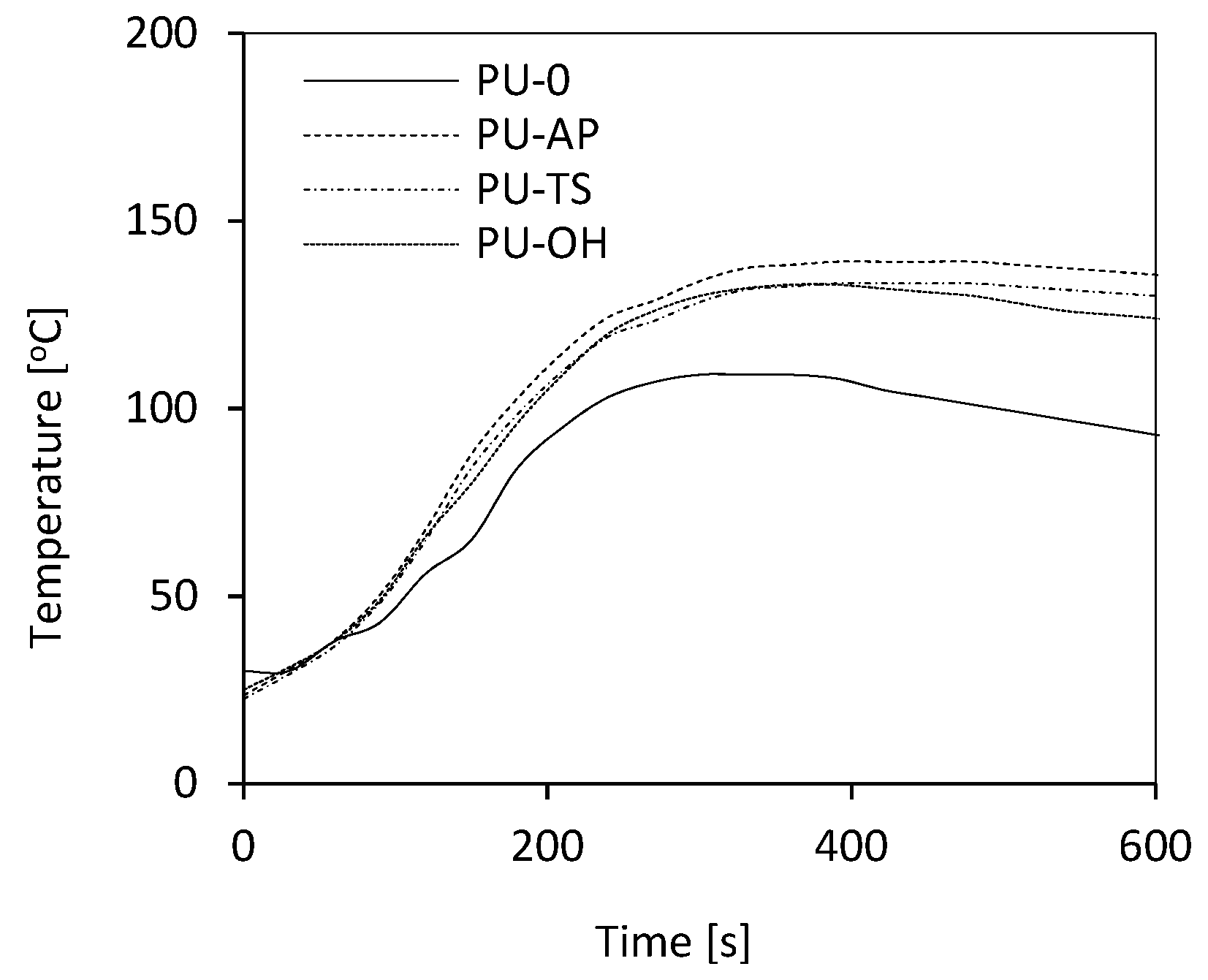
3.3. The Influence of POSS on the Maximum Temperature (Tmax) of the Reaction Mixture during the Foaming Process
3.4. Foaming Kinetic of RPUFs
3.5. Density of RPUFs
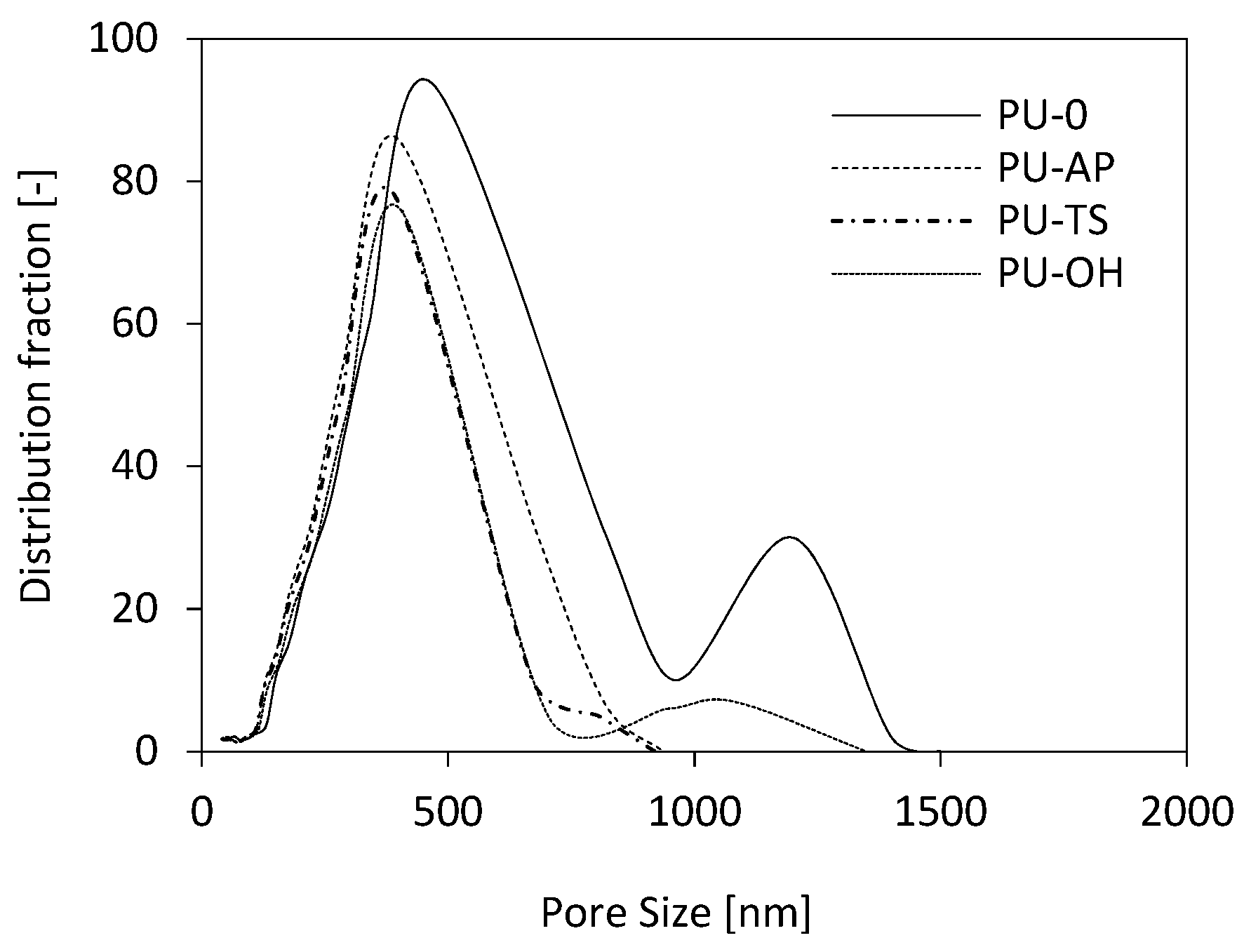
3.6. Morphology of RPUFs
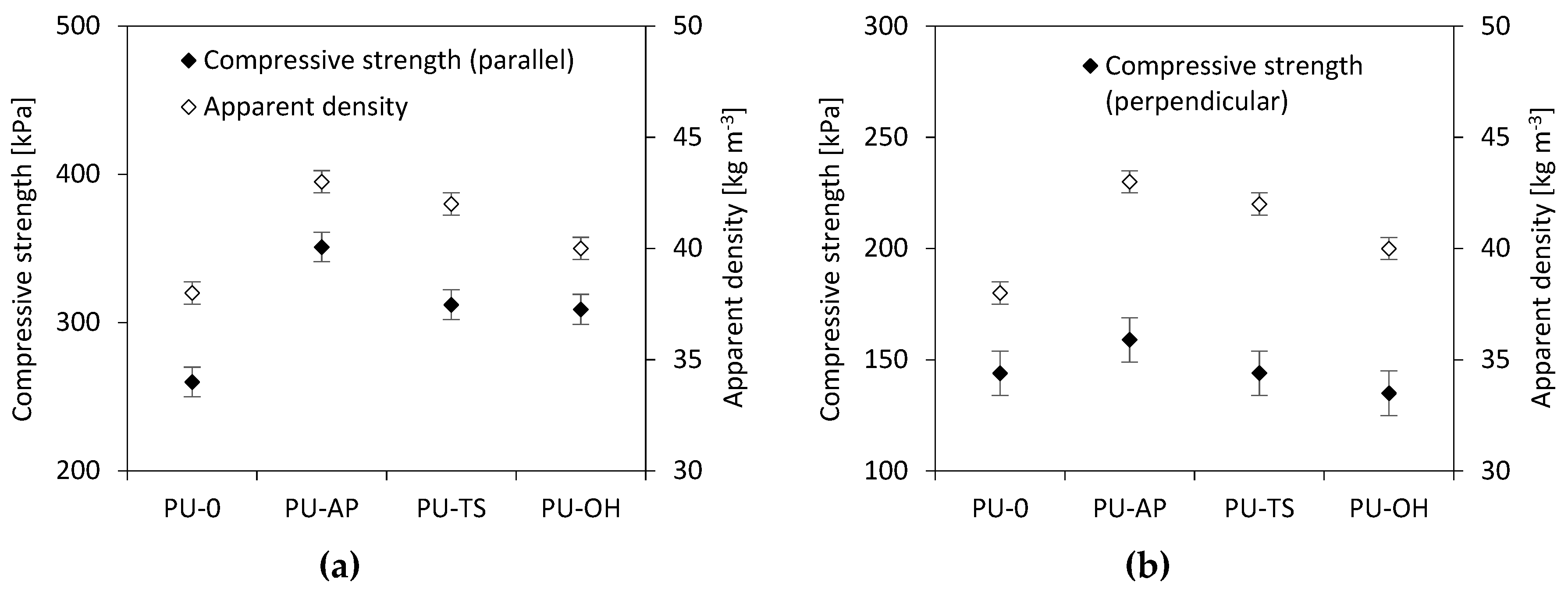
3.7. Compressive Strength of RPUFs
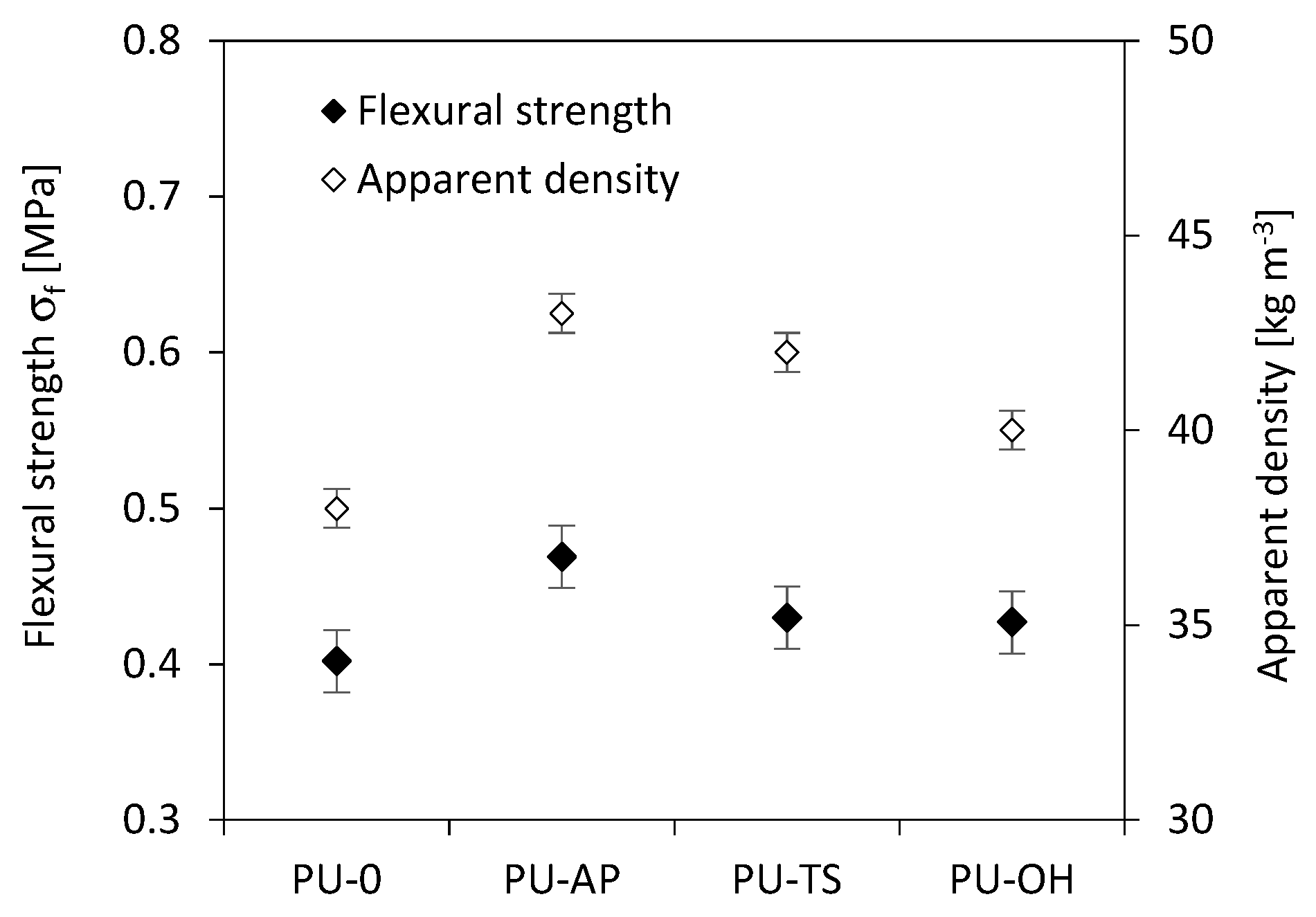
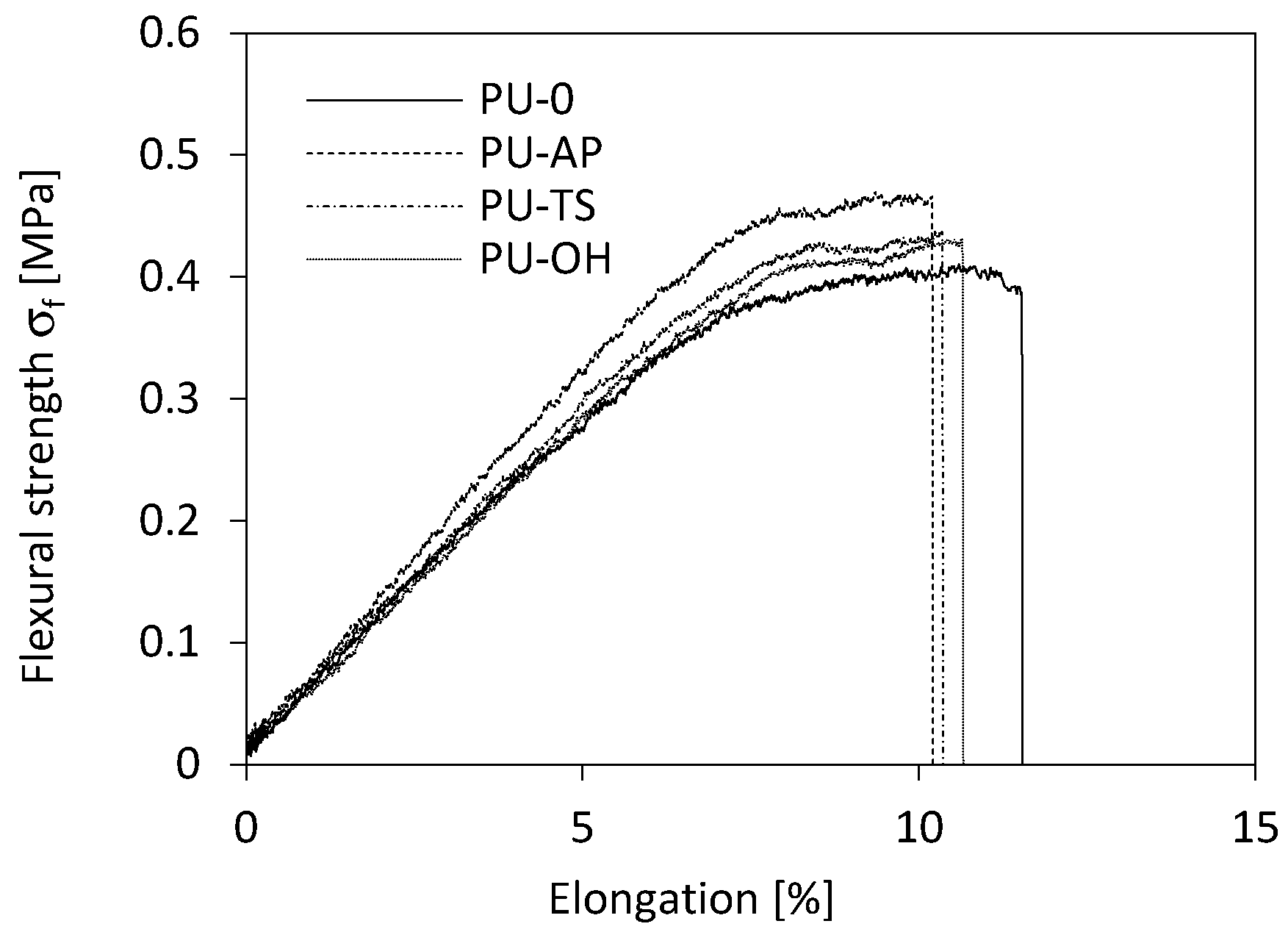
3.8. Flexural Strength of RPUFs
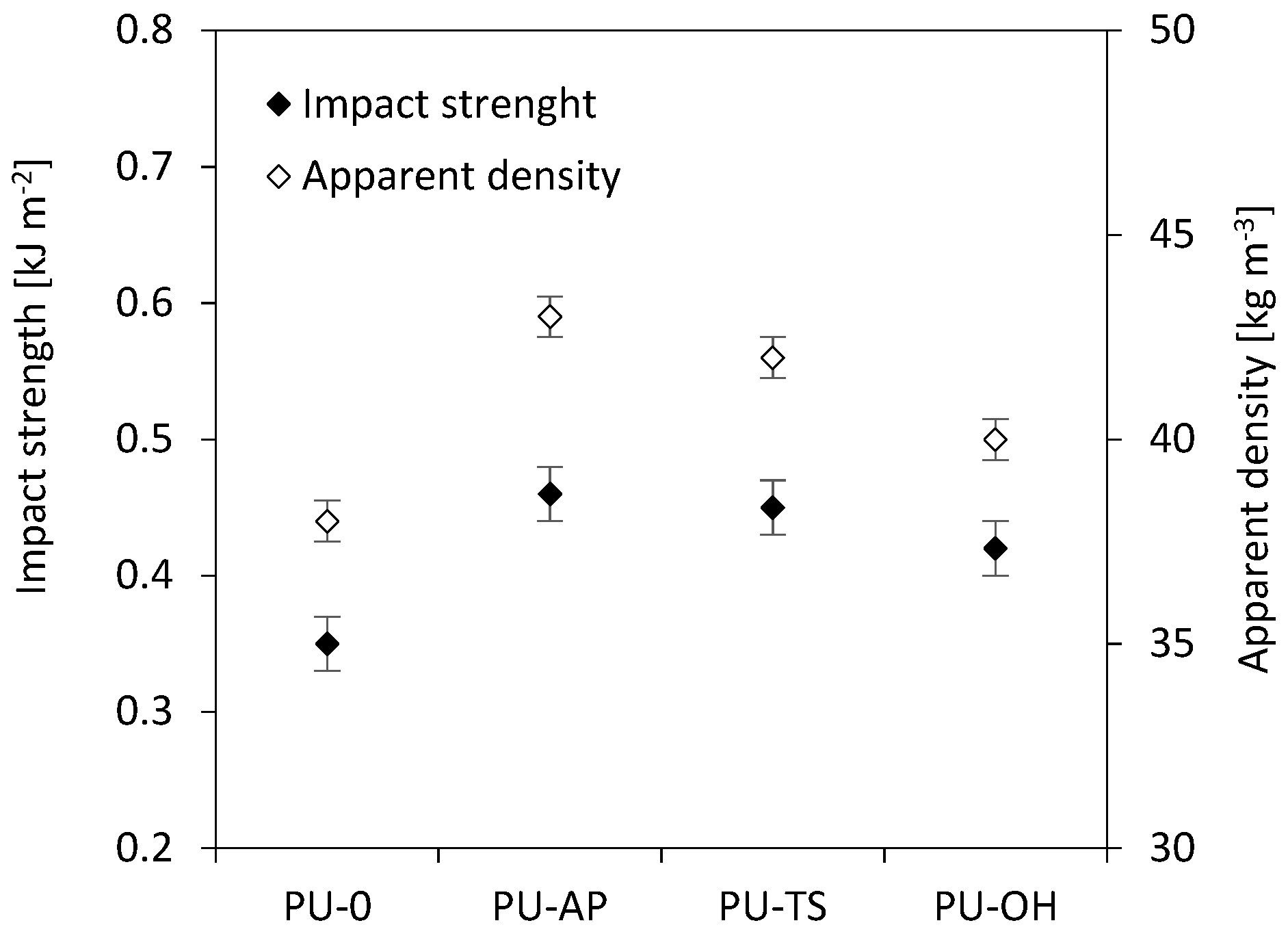
3.9. Impact Strength of RPUFs
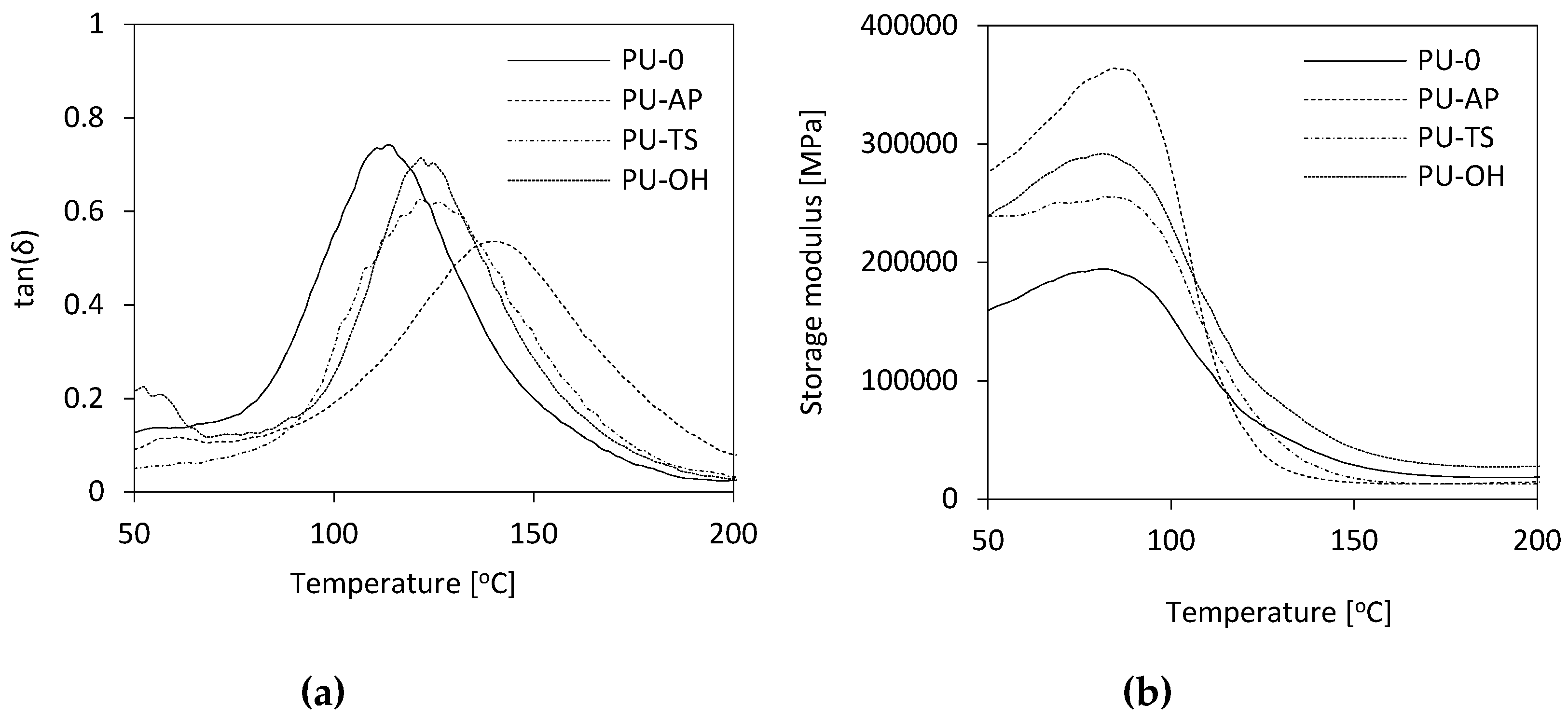
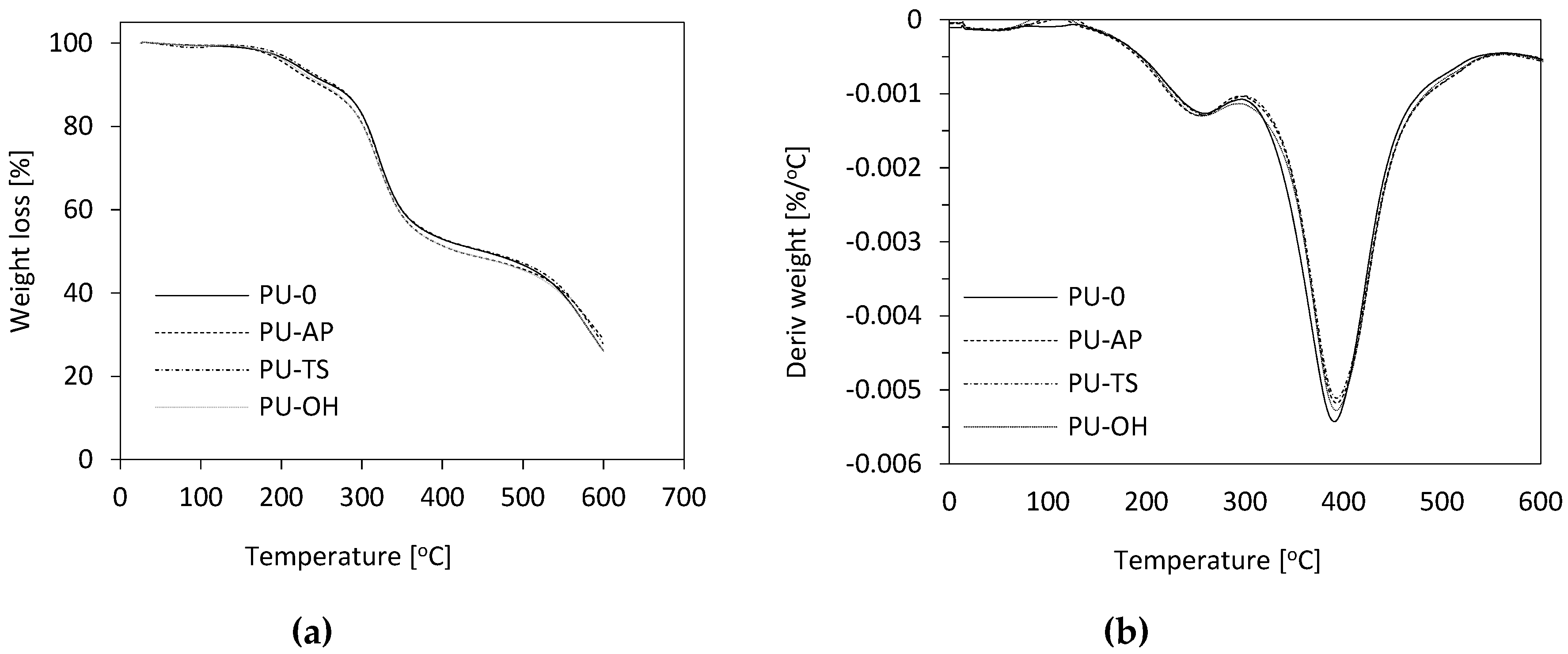
3.10. Dynamic Mechanical Analysis (DMA) and Thermogravimetric Analysis (TGA)
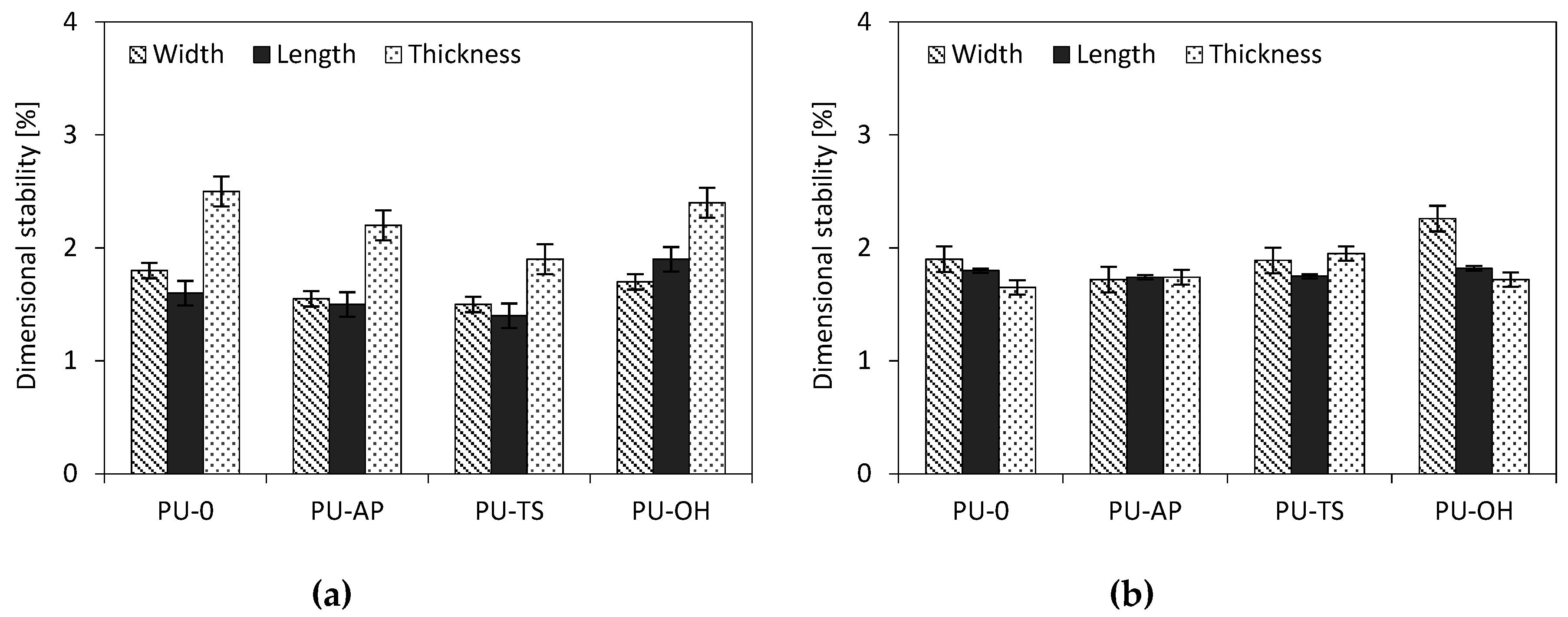
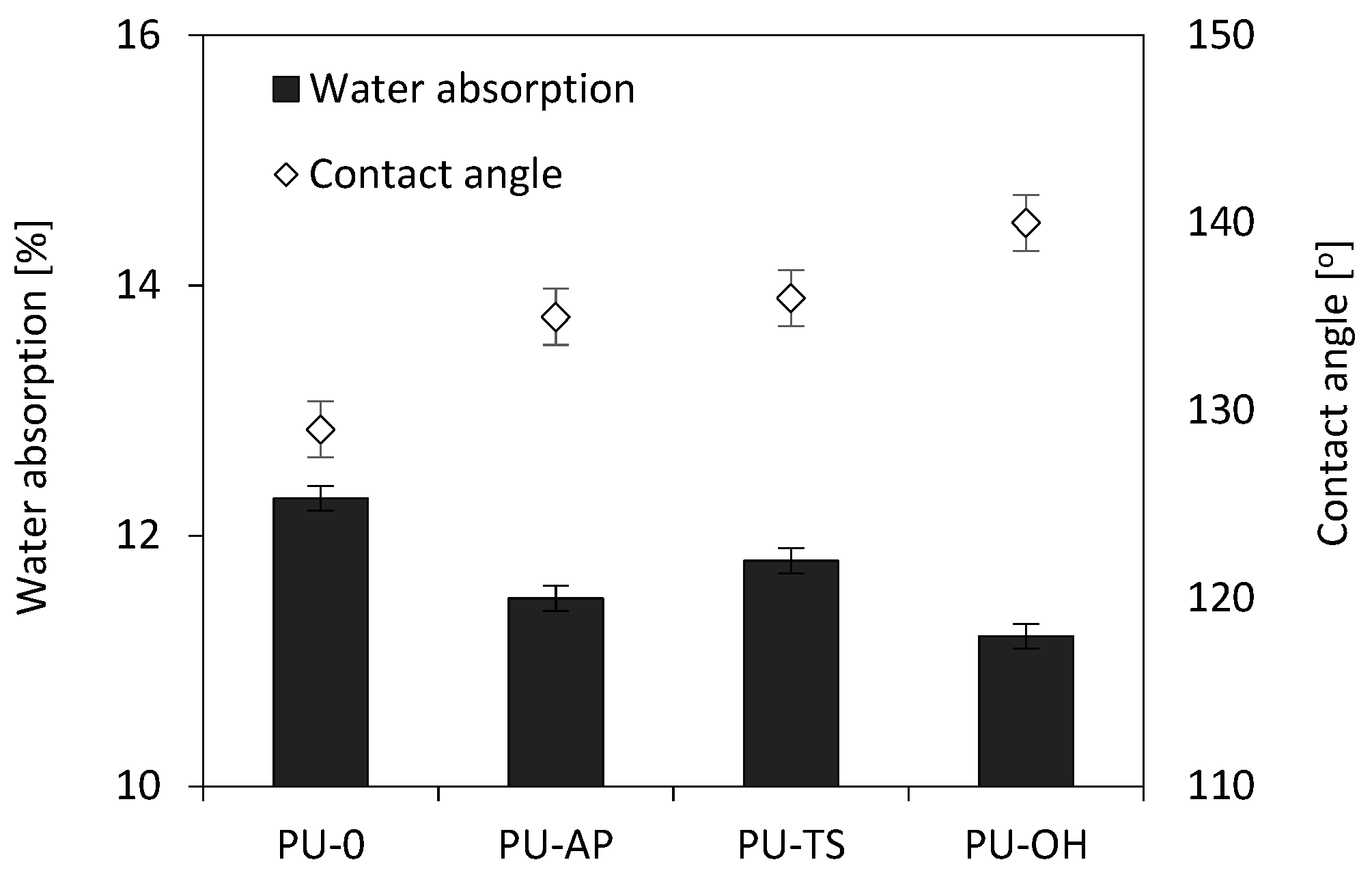
3.11. Dimensional Stability, Contact Angle and Water Absorption
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nikje, M.M.A.; Noruzian, M.; Moghaddam, S.T. Investigation of Fe3O4/AEAP supermagnetic nanoparticles on the morphological, thermal and magnetite behavior of polyurethane rigid foam nanocomposites. Polimery 2015, 60, 26–32. [Google Scholar] [CrossRef]
- Xie, H.; Yang, W.; Yuen, A.C.Y.; Xie, C.; Xie, J.; Lu, H.; Yeoh, G.H. Study on flame retarded flexible polyurethane foam/alumina aerogel composites with improved fire safety. Chem. Eng. J. 2017, 311, 310–317. [Google Scholar] [CrossRef]
- Yang, C.; Fischer, L.; Maranda, S.; Worlitschek, J. Rigid polyurethane foams incorporated with phase change materials: A state-of-the-art review and future research pathways. Energy Build. 2015, 87, 25–36. [Google Scholar] [CrossRef]
- Tan, S.; Abraham, T.; Ference, D.; Macosko, C.W. Rigid polyurethane foams from a soybean oil-based Polyol. Polymers 2011, 52, 2840–2846. [Google Scholar] [CrossRef]
- Rigid Polyurethane Foam Market - Global Industry Analysis, Size, Share, Growth Trends & Forecasts 2017–2025, PR Newswire, New York. 2017. Available online: https://search.proquest.com/docview/1966284809?rfr_id=info%3Axri%2Fsid%3Aprimo (accessed on 27 May 2018).
- Global Polyurethane (PU) Foam Market Trends, Analysis & Forecasts 2016–2024, NASDAQ OMX’s News Release Distrib. Channel, New York. 2016. Available online: https://search.proquest.com/docview/1793434616?rfr_id=info%3Axri%2Fsid%3Aprimo (accessed on 27 May 2018).
- Mahajan, N.; Gupta, P. New insights into the microbial degradation of polyurethanes. RSC Adv. 2015, 5, 41839–41854. [Google Scholar] [CrossRef]
- Yang, H.; Wang, X.; Song, L.; Yu, B.; Yuan, Y.; Hu, Y.; Yuen, K.K.R. Aluminum hypophosphite in combination with expandable graphite as a novel flame retardant system for rigid polyurethane foams. Polym. Adv. Technol. 2014, 25, 1034–1043. [Google Scholar] [CrossRef]
- Hu, X.-M.; Wang, D.-M. Enhanced fire behavior of rigid polyurethane foam by intumescent flame retardants. J. Appl. Polym. Sci. 2013, 129, 238–246. [Google Scholar] [CrossRef]
- Shi, Y.; Yuan, Y.; Yang, H.; Yu, B.; Wang, W.; Song, L.; Hu, Y.; Zhang, Y. Phosphorus and Nitrogen-Containing Polyols: Synergistic Effect on the Thermal Property and Flame Retardancy of Rigid Polyurethane Foam Composites. Ind. Eng. Chem. Res. 2016, 55, 10813–10822. [Google Scholar]
- Yang, H.; Liu, H.; Jiang, Y.; Chen, M.; Wan, C. Density Effect on Flame Retardancy, Thermal Degradation, and Combustibility of Rigid Polyurethane Foam Modified by Expandable Graphite or Ammonium Polyphosphate. Polymers 2019, 11, 668. [Google Scholar] [CrossRef] [PubMed]
- Kurańska, M.; Prociak, A. The influence of rapeseed oil-based polyols on the foaming process of rigid polyurethane foams. Ind. Crop. Prod. 2016, 89, 182–187. [Google Scholar] [CrossRef]
- Sobolewski, M.; Błażejczak, A. Izolacyjność cieplna wysokoprężnej pianki poliuretanowej w aerozolu. Cz. 1. Właściwości i zastosowanie pianek poliuretanowych, Izolacje. R. 2014, 19, pp. 11–12. Available online: http://yadda.icm.edu.pl/baztech/element/bwmeta1.element.baztech-5d8e9b3a-a1e9-4bf7-85dd-3adb61863dd1 (accessed on 1 June 2019).
- Paciorek-Sadowska, J.; Czupryński, B.; Liszkowska, J.; Jaskółowski, W. Nowy poliol boroorganiczny do produkcji sztywnych pianek poliuretanowo-poliizocyjanurowych. Cz. II. Otrzymywanie sztywnych pianek poliuretanowo-poliizocyjanurowych z zastosowaniem nowego poliolu boroorganicznego, Polimery. T. 2010, 55, pp. 99–105. Available online: http://yadda.icm.edu.pl/baztech/element/bwmeta1.element.baztech-article-BATB-0001-0012 (accessed on 1 June 2019).
- Fan, H.; Tekeei, A.; Suppes, G.J.; Hsieh, F.-H. Rigid polyurethane foams made from high viscosity soy-polyols. J. Appl. Polym. Sci. 2013, 127, 1623–1629. [Google Scholar] [CrossRef]
- Członka, S.; Bertino, M.F.; Strzelec, K. Rigid polyurethane foams reinforced with industrial potato protein. Polym. Test. 2018, 68, 135–145. [Google Scholar] [CrossRef]
- Formela, K.; Hejna, A.; Zedler, Ł.; Przybysz, M.; Ryl, J.; Saeb, M.R.; Piszczyk, Ł. Structural, thermal and physico-mechanical properties of polyurethane/brewers’ spent grain composite foams modified with ground tire rubber. Ind. Crop. Prod. 2017, 108, 844–852. [Google Scholar] [CrossRef]
- Silva, M.C.; Takahashi, J.A.; Chaussy, D.; Belgacem, M.N.; Silva, G.G. Composites of rigid polyurethane foam and cellulose fiber residue. J. Appl. Polym. Sci. 2010. [Google Scholar] [CrossRef]
- Shi, X.; Yang, P.; Peng, X.; Huang, C.; Qian, Q.; Wang, B.; He, J.; Liu, X.; Li, Y.; Kuang, T. Bi-phase fire-resistant polyethylenimine/graphene oxide/melanin coatings using layer by layer assembly technique: Smoke suppression and thermal stability of flexible polyurethane foams. Polymers 2019, 170, 65–75. [Google Scholar] [CrossRef]
- Cho, J.H.; Vasagar, V.; Shanmuganathan, K.; Jones, A.R.; Nazarenko, S.; Ellison, C.J. Bioinspired Catecholic Flame Retardant Nanocoating for Flexible Polyurethane Foams. Chem. Mater. 2015, 27, 6784–6790. [Google Scholar] [CrossRef]
- Roberts, B.; Jones, A.; Ezekoye, O.; Ellison, C.; Webber, M.; Roberts, B. Development of kinetic parameters for polyurethane thermal degradation modeling featuring a bioinspired catecholic flame retardant. Combust. Flame 2017, 177, 184–192. [Google Scholar] [CrossRef]
- Septevani, A.A.; Evans, D.A.; Annamalai, P.K.; Martin, D.J. The use of cellulose nanocrystals to enhance the thermal insulation properties and sustainability of rigid polyurethane foam. Ind. Crop. Prod. 2017, 107, 114–121. [Google Scholar] [CrossRef]
- Song, Z.-L.; Ma, L.-Q.; Wu, Z.-J.; He, D.-P. Effects of viscosity on cellular structure of foamed aluminum in foaming process. J. Mater. Sci. 2000, 35, 15–20. [Google Scholar] [CrossRef]
- Dolomanova, V.; Rauhe, J.C.M.; Jensen, L.R.; Pyrz, R.; Timmons, A.B. Mechanical properties and morphology of nano-reinforced rigid PU foam. J. Cell. Plast. 2011, 47, 81–93. [Google Scholar] [CrossRef]
- Niyogi, D.; Kumar, R.; Gandhi, K.S. Water blown free rise polyurethane foams. Polym. Eng. Sci. 1999, 39, 199–209. [Google Scholar] [CrossRef]
- Sung, G.; Kim, J.H. Influence of filler surface characteristics on morphological, physical, acoustic properties of polyurethane composite foams filled with inorganic fillers. Compos. Sci. Technol. 2017, 146, 147–154. [Google Scholar] [CrossRef]
- Gu, R.; Khazabi, M.; Sain, M. Fiber reinforced soy-based polyurethane spray foam insulation. Part 2: Thermal and mechanical properties. BioResources 2011, 6, 3775–3790. [Google Scholar]
- Luo, X.; Mohanty, A.; Misra, M. Lignin as a reactive reinforcing filler for water-blown rigid biofoam composites from soy oil-based polyurethane. Ind. Crop. Prod. 2013, 47, 13–19. [Google Scholar] [CrossRef]
- Michałowski, S.; Hebda, E.; Pielichowski, K. Thermal stability and flammability of polyurethane foams chemically reinforced with POSS. J. Therm. Anal. Calorim. 2017, 130, 155–163. [Google Scholar] [CrossRef]
- Michałowski, S.; Pielichowski, K. 1,2-Propanediolizobutyl POSS as a co-flame retardant for rigid polyurethane foams. J. Therm. Anal. Calorim. 2018, 134, 1351–1358. [Google Scholar] [CrossRef]
- Hebda, E.; Ozimek, J.; Raftopoulos, K.N.; Michałowski, S.; Pielichowski, J.; Jancia, M.; Pielichowski, K. Synthesis and morphology of rigid polyurethane foams with POSS as pendant groups or chemical crosslinks. Polym. Adv. Technol. 2015, 26, 932–940. [Google Scholar] [CrossRef]
- Cordes, D.B.; Lickiss, P.D.; Rataboul, F. Recent Developments in the Chemistry of Cubic Polyhedral Oligosilsesquioxanes. Chem. Rev. 2010, 110, 2081–2173. [Google Scholar] [CrossRef]
- Hosaka, N.; Otsuka, H.; Hino, M.; Takahara, A. Control of Dispersion State of Silsesquioxane Nanofillers for Stabilization of Polystyrene Thin Films. Langmuir 2008, 24, 5766–5772. [Google Scholar] [CrossRef]
- Fina, A.; Monticelli, O.; Camino, G. POSS-based hybrids by melt/reactive blending. J. Mater. Chem. 2010, 20, 9297. [Google Scholar] [CrossRef]
- Adnan, S.; Tuan Ismail, T.N.M.; Mohd Noor, N.; Din, N.M.; Mariam, N.S.; Hanzah, N.A.; Shoot Kian, Y.; Abu Hassan, H. Development of Flexible Polyurethane Nanostructured Biocomposite Foams Derived from Palm Olein-Based Polyol. Adv. Mater. Sci. Eng. 2016, 2016, 1–12. [Google Scholar] [CrossRef]
- Pawlak, T.; Kowalewska, A.; Zgardzińska, B.; Potrzebowski, M.J. Structure, Dynamics, and Host–Guest Interactions in POSS Functionalized Cross-Linked Nanoporous Hybrid Organic–Inorganic Polymers. J. Phys. Chem. C 2015, 119, 26575–26587. [Google Scholar] [CrossRef]
- Blanco, I. The Rediscovery of POSS: A Molecule Rather than a Filler. Polymers 2018, 10, 904. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tseng, M.; Fangchiang, M. Polymerization and nanocomposites properties of multifunctional methylmethacrylate POSS. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 5157–5166. [Google Scholar] [CrossRef]
- Rashid, E.S.A.; Ariffin, K.; Kooi, C.C.; Akil, H.M. Preparation and properties of POSS/epoxy composites for electronic packaging applications. Mater. Des. 2009, 30, 1–8. [Google Scholar] [CrossRef]
- Fan, X.; Cao, M.; Zhang, X.; Li, Z. Synthesis of star-like hybrid POSS-(PDMAEMA-b-PDLA)8 copolymer and its stereocomplex properties with PLLA. Mater. Sci. Eng. C 2017, 76, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Blanco, I.; Bottino, F.A.; Cicala, G.; Cozzo, G.; Latteri, A.; Recca, A. Synthesis and thermal characterization of new dumbbell shaped POSS/PS nanocomposites: Influence of the symmetrical structure of the nanoparticles on the dispersion/aggregation in the polymer matrix. Polym. Compos. 2015, 36, 1394–1400. [Google Scholar] [CrossRef]
- Phillips, S.H.; Haddad, T.S.; Tomczak, S.J. Developments in nanoscience: Polyhedral oligomeric silsesquioxane (POSS)-polymers. Curr. Opin. Solid State Mater. Sci. 2004, 8, 21–29. [Google Scholar] [CrossRef]
- Pielichowski, K.; Njuguna, J.; Janowski, B.; Pielichowski, J. Polyhedral Oligomeric Silsesquioxanes (POSS)-Containing Nanohybrid Polymers; Springer: Berlin/Heidelberg, Germany, 2006; Vol. 201, pp. 225–296. [Google Scholar]
- Marcinkowska, A.; Przadka, D.; Dudziec, B.; Szczesniak, K.; Andrzejewska, E. Anchor Effect in Polymerization Kinetics: Case of Monofunctionalized POSS. Polymers 2019, 11, 515. [Google Scholar] [CrossRef]
- Liu, L.; Tian, M.; Zhang, W.; Zhang, L.; Mark, J.E. Crystallization and morphology study of polyhedral oligomeric silsesquioxane (POSS)/polysiloxane elastomer composites prepared by melt blending. Polymers 2007, 48, 3201–3212. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, Y.; Liu, L. Effects of TriSilanolIsobutyl-POSS on thermal stability of methylsilicone resin. Polym. Degrad. Stab. 2006, 91, 2731–2738. [Google Scholar] [CrossRef]
- Song, X.; Zhang, X.; Li, T.; Li, Z.; Chi, H. Mechanically Robust Hybrid POSS Thermoplastic Polyurethanes with Enhanced Surface Hydrophobicity. Polymers 2019, 11, 373. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.; Najwa, K. Cellulose Nanocrystals Reinforced Thermoplastic Polyurethane Nanocomposites. Ph.D. Thesis, The University of Queensland, Queensland, Australia, 2016. [Google Scholar] [CrossRef]
- KAIRYTĖ, A.; Vaitkus, S.; Vėjelis, S.; Girskas, G.; Balčiūnas, G. Rapeseed-based polyols and paper production waste sludge in polyurethane foam: Physical properties and their prediction models. Ind. Crop. Prod. 2018, 112, 119–129. [Google Scholar] [CrossRef]
- Michalowski, S.; Cabulis, U.; Kirpluks, M.; Prociak, A.; Kuranska, M. Microcellulose as a natural filler in polyurethane foams based on the biopolyol from rapeseed oil. Polimery 2016, 61, 625–632. [Google Scholar]
- Yan, D.-X.; Xu, L.; Chen, C.; Tang, J.; Ji, X.; Li, Z. Enhanced mechanical and thermal properties of rigid polyurethane foam composites containing graphene nanosheets and carbon nanotubes. Polym. Int. 2012, 61, 1107–1114. [Google Scholar] [CrossRef]
- Ghoreishi, R.; Al-Moameri, H.; Zhao, Y.; Suppes, G.J. Simulation Blowing Agent Performance, Cell Morphology, and Cell Pressure in Rigid Polyurethane Foams. Ind. Eng. Chem. Res. 2016, 55, 2336–2344. [Google Scholar]
- Członka, S.; Sienkiewicz, N.; Strąkowska, A.; Strzelec, K. Keratin feathers as a filler for rigid polyurethane foams on the basis of soybean oil polyol. Polym. Test. 2018, 72, 32–45. [Google Scholar] [CrossRef]
- Członka, S.; Bertino, M.F.; Strzelec, K.; Strąkowska, A.; Masłowski, M. Rigid polyurethane foams reinforced with solid waste generated in leather industry. Polym. Test. 2018, 69, 225–237. [Google Scholar] [CrossRef]
- Wolska, A.; Goździkiewicz, M.; Ryszkowska, J. Thermal and mechanical behaviour of flexible polyurethane foams modified with graphite and phosphorous fillers. J. Mater. Sci. 2012, 47, 5627–5634. [Google Scholar] [CrossRef]
- Guo, C.; Zhou, L.; Lv, J. Effects of Expandable Graphite and Modified Ammonium Polyphosphate on the Flame-Retardant and Mechanical Properties of Wood Flour-Polypropylene Composites. Polym. Polym. Compos. 2013, 21, 449–456. [Google Scholar] [CrossRef]
- Ciecierska, E.; Jurczyk-Kowalska, M.; Bazarnik, P.; Gloc, M.; Kulesza, M.; Krauze, S.; Lewandowska, M.; Kowalski, M. Flammability, mechanical properties and structure of rigid polyurethane foams with different types of carbon reinforcing materials. Compos. Struct. 2016, 140, 67–76. [Google Scholar] [CrossRef]
- Kim, J.M.; Han, M.S. Thermal, Morphological and Rheological Properties of Rigid Polyurethane Foams as Thermal Insulating Materials. AIP Conference Proceedings 2008, 1027, 905–907. [Google Scholar]
- Chang, L.-C. Improving the Mechanical Performance of Wood Fiber Reinforced Bio-based Polyurethane Foam. Ph.D. Thesis, University of Toronto, Toronto, Canada, 2014. [Google Scholar]
- Kurańska, M.; Prociak, A. Porous polyurethane composites with natural fibres. Compos. Sci. Technol. 2012, 72, 299–304. [Google Scholar] [CrossRef]
- Kurańska, M.; Aleksander, P.; Mikelis, K.; Ugis, C. Porous polyurethane composites based on bio-components. Compos. Sci. Technol. 2013, 75, 70–76. [Google Scholar] [CrossRef]
- Mosiewicki, M.; Dell’Arciprete, G.; Aranguren, M.; Marcovich, N. Polyurethane Foams Obtained from Castor Oil-based Polyol and Filled with Wood Flour. J. Compos. Mater. 2009, 43, 3057–3072. [Google Scholar] [CrossRef]
- Finlay, K.A.; Gawryla, M.D.; Schiraldi, D.A. Effects of Fiber Reinforcement on Clay Aerogel Composites. Materials 2015, 8, 5440–5451. [Google Scholar] [CrossRef]
- Hamilton, A.R.; Thomsen, O.T.; Madaleno, L.A.; Jensen, L.R.; Rauhe, J.C.M.; Pyrz, R. Evaluation of the anisotropic mechanical properties of reinforced polyurethane foams. Compos. Sci. Technol. 2013, 87, 210–217. [Google Scholar] [CrossRef]
- Marcovich, N.E.; Kurańska, M.; Prociak, A.; Malewska, E.; Kulpa, K. Open cell semi-rigid polyurethane foams synthesized using palm oil-based bio-polyol. Ind. Crops Prod. 2017, 102, 88–96. [Google Scholar] [CrossRef]
- Wu, L.; Van Gemert, J.; Camargo, R.E. Rheology Study in Polyurethane Rigid Foams. Auburn Hills, MI, USA, 2012. Available online: http://www.huntsman.com/polyurethanes/Media%20Library/a_MC1CD1F5AB7BB1738E040EBCD2B6B01F1/Products_MC1CD1F5AB8081738E040EBCD2B6B01F1/Construction_MC1CD1F5AEF051738E040EBCD2B6B01F1/Technical%20presentati_MC1CD1F5AF6F41738E040EBCD2B6B01F1/files/cpi_08_lifengwu_revised.pdf (accessed on 1 June 2019).
- Hatakeyama, H.; Kosugi, R.; Hatakeyama, T. Thermal properties of lignin-and molasses-based polyurethane foams. J. Therm. Anal. Calorim. 2008, 92, 419–424. [Google Scholar] [CrossRef]
- Ye, L.; Meng, X.-Y.; Ji, X.; Li, Z.-M.; Tang, J.-H. Synthesis and characterization of expandable graphite–poly(methyl methacrylate) composite particles and their application to flame retardation of rigid polyurethane foams. Polym. Degrad. Stab. 2009, 94, 971–979. [Google Scholar] [CrossRef]
- Gama, N.V.; Silva, R.; Mohseni, F.; Davarpanah, A.; Amaral, V.; Ferreira, A.; Barros-Timmons, A. Enhancement of physical and reaction to fire properties of crude glycerol polyurethane foams filled with expanded graphite. Polym. Test. 2018, 69, 199–207. [Google Scholar] [CrossRef]
- Jiao, L.; Xiao, H.; Wang, Q.; Sun, J. Thermal degradation characteristics of rigid polyurethane foam and the volatile products analysis with TG-FTIR-MS. Polym. Degrad. Stab. 2013, 98, 2687–2696. [Google Scholar] [CrossRef]
- Levchik, S.V.; Weil, E.D. Thermal decomposition, combustion and fire-retardancy of polyurethanes—A review of the recent literature. Polym. Int. 2004, 53, 1585–1610. [Google Scholar] [CrossRef]
- Septevani, A.A.; Evans, D.A.; Chaleat, C.; Martin, D.J.; Annamalai, P.K.; Evans, D.A.C. A systematic study substituting polyether polyol with palm kernel oil based polyester polyol in rigid polyurethane foam. Ind. Crop. Prod. 2015, 66, 16–26. [Google Scholar] [CrossRef]
- Pagacz, J.; Hebda, E.; Michałowski, S.; Ozimek, J.; Sternik, D.; Pielichowski, K. Polyurethane foams chemically reinforced with POSS—Thermal degradation studies. Thermochim. Acta 2016, 642, 95–104. [Google Scholar] [CrossRef]
- Chattopadhyay, D.; Webster, D.C. Thermal stability and flame retardancy of polyurethanes. Prog. Polym. Sci. 2009, 34, 1068–1133. [Google Scholar] [CrossRef]

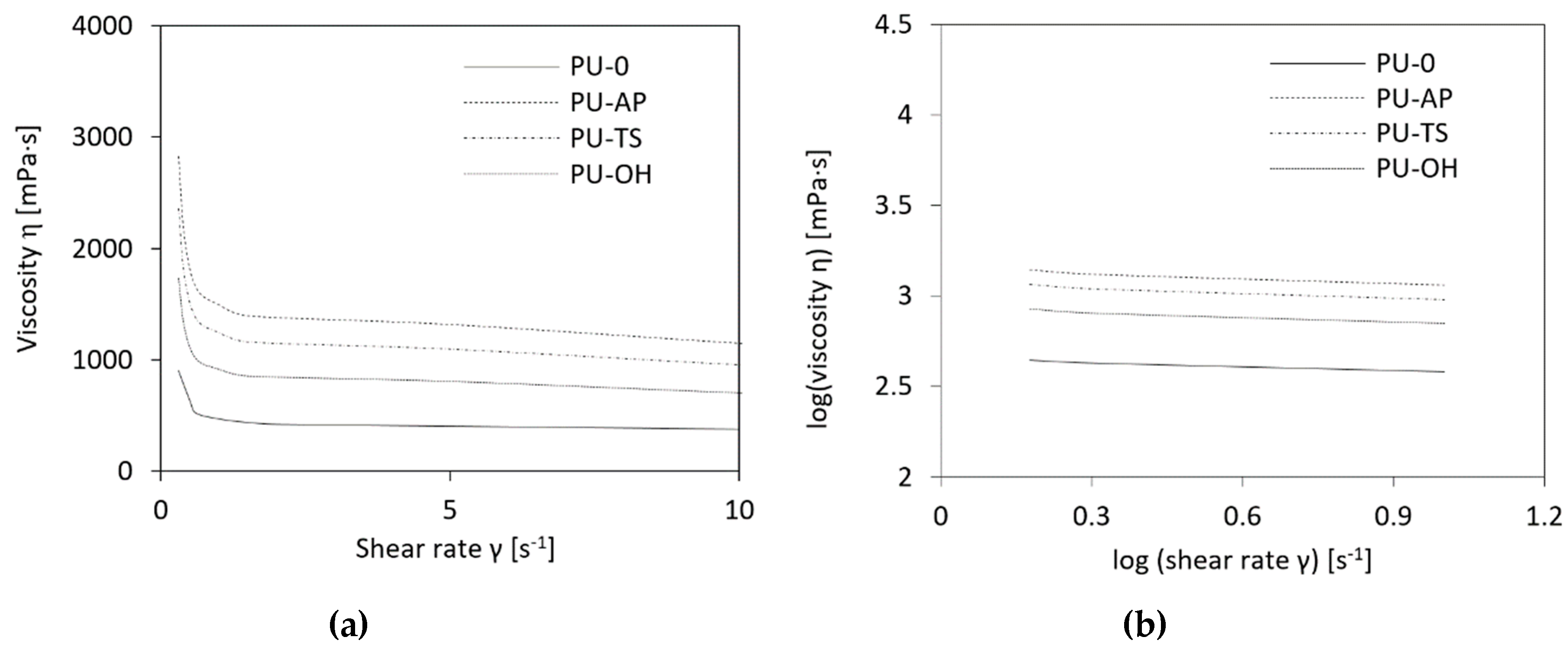



| Sample Code | Dynamic Viscosity η [mPa·s] | Fitting Equation | Power Law Index (n) | R2 | ||
|---|---|---|---|---|---|---|
| 0.5 RPM | 5 RPM | 10 RPM | ||||
| PU-0 | 628 | 424 | 380 | y = −0.058 + 0.335 | 0.335 | 0.982 |
| PU-AP | 2110 | 1313 | 1149 | y = −0.060 + 0.318 | 0.298 | 0.978 |
| PU-TS | 1317 | 1098 | 957 | y = −0.060 + 0.315 | 0.315 | 0.978 |
| PU-OH | 1149 | 805 | 702 | y = −0.059 + 0.298 | 0.318 | 0.979 |
| Sample Code | Temperature [°C] | Cream Time [s] | Extension Time [s] | Tack-free Time [s] | Cell Size [µm] | Wall Thickness [µm] | Apparent Density [kg m−3] |
|---|---|---|---|---|---|---|---|
| PU-0 | 110 | 43 ± 4 | 277 ± 10 | 341 ± 14 | 472 ± 10 | 62 ± 4 | 38 |
| PU-AP | 135 | 49 ± 2 | 512 ± 11 | 376 ± 12 | 390 ± 8 | 68 ± 2 | 43 |
| PU-TS | 136 | 47 ± 2 | 504 ± 8 | 370 ± 12 | 402 ± 6 | 66 ± 3 | 42 |
| PU-OH | 134 | 46 ± 2 | 428 ± 9 | 320 ± 10 | 410 ± 8 | 66 ± 2 | 40 |
| Sample Code | Compressive Strength (Parallel) σ10 [kPa] | Compressive Strength (Perpendicular) σ10 [kPa] | Young Modulus [MPa] | Flexural Strength σf [MPa] | Elongation [%] | Impact Strength [kJ m−2] |
|---|---|---|---|---|---|---|
| PU-0 | 260 | 144 | 5 | 0.402 | 11.2 | 0.35 |
| PU-AP | 351 | 159 | 6.1 | 0.469 | 10.2 | 0.46 |
| PU-TS | 312 | 144 | 5.4 | 0.430 | 10.4 | 0.45 |
| PU-OH | 309 | 135 | 5.2 | 0.427 | 10.8 | 0.42 |
| Sample Code | Tg | T5% | T10% | T50% | T70% | Char Residue |
|---|---|---|---|---|---|---|
| [°C] | [°C] | [°C] | [°C] | [°C] | [%] | |
| AP-POSS | - | 267 | 280 | 342 | 531 | 21.6 |
| TS-POSS | - | 260 | 287 | 350 | 449 | 18.4 |
| OH-POSS | - | n.d. | n.d. | n.d. | n.d. | n.d. |
| PU-0 | 112 | 220 | 265 | 454 | 591 | 27.9 |
| PU-AP | 137 | 205 | 245 | 418 | 595 | 29.0 |
| PU-TS | 127 | 216 | 261 | 451 | 586 | 28.7 |
| PU-OH | 121 | 210 | 251 | 449 | 585 | 28.6 |
| Sample Code | Dimensional Stability (+70°C) [%] | Dimensional Stability (−20°C) [%] | Water Absorption [%] | Contact Angle [°] | ||||
|---|---|---|---|---|---|---|---|---|
| Width | Length | Thickness | Width | Length | Thickness | |||
| PU-0 | 1.80 | 1.6 | 2.5 | 1.90 | 1.8 | 1.65 | 12.3 | 129 |
| PU-AP | 1.55 | 1.5 | 2.2 | 1.72 | 1.74 | 1.74 | 11.5 | 135 |
| PU-TS | 1.5 | 1.4 | 1.90 | 1.89 | 1.75 | 1.95 | 11.8 | 136 |
| PU-OH | 1.7 | 1.9 | 2.4 | 2.26 | 1.82 | 1.72 | 11.2 | 140 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strąkowska, A.; Członka, S.; Strzelec, K. POSS Compounds as Modifiers for Rigid Polyurethane Foams (Composites). Polymers 2019, 11, 1092. https://doi.org/10.3390/polym11071092
Strąkowska A, Członka S, Strzelec K. POSS Compounds as Modifiers for Rigid Polyurethane Foams (Composites). Polymers. 2019; 11(7):1092. https://doi.org/10.3390/polym11071092
Chicago/Turabian StyleStrąkowska, Anna, Sylwia Członka, and Krzysztof Strzelec. 2019. "POSS Compounds as Modifiers for Rigid Polyurethane Foams (Composites)" Polymers 11, no. 7: 1092. https://doi.org/10.3390/polym11071092
APA StyleStrąkowska, A., Członka, S., & Strzelec, K. (2019). POSS Compounds as Modifiers for Rigid Polyurethane Foams (Composites). Polymers, 11(7), 1092. https://doi.org/10.3390/polym11071092







