Poly(furfuryl alcohol)-Polycaprolactone Blends
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Film Preparation
2.2.2. Atomic Force Microscopy (AFM Characterization)
2.2.3. Attenuated Total Reflection-Fourier Transform Infrared (ATR-FTIR) Characterization
2.2.4. Thermal Analysis
2.2.5. Determination of Sample Crystallinity
2.2.6. Mechanical Analysis
2.2.7. Biocompatibility Study
2.2.8. Determination of Antioxidant Activity
2.2.9. Oxygen Transmission Rate Measurements
3. Results
3.1. PCL/PFA Films Morphology and Surface Topography
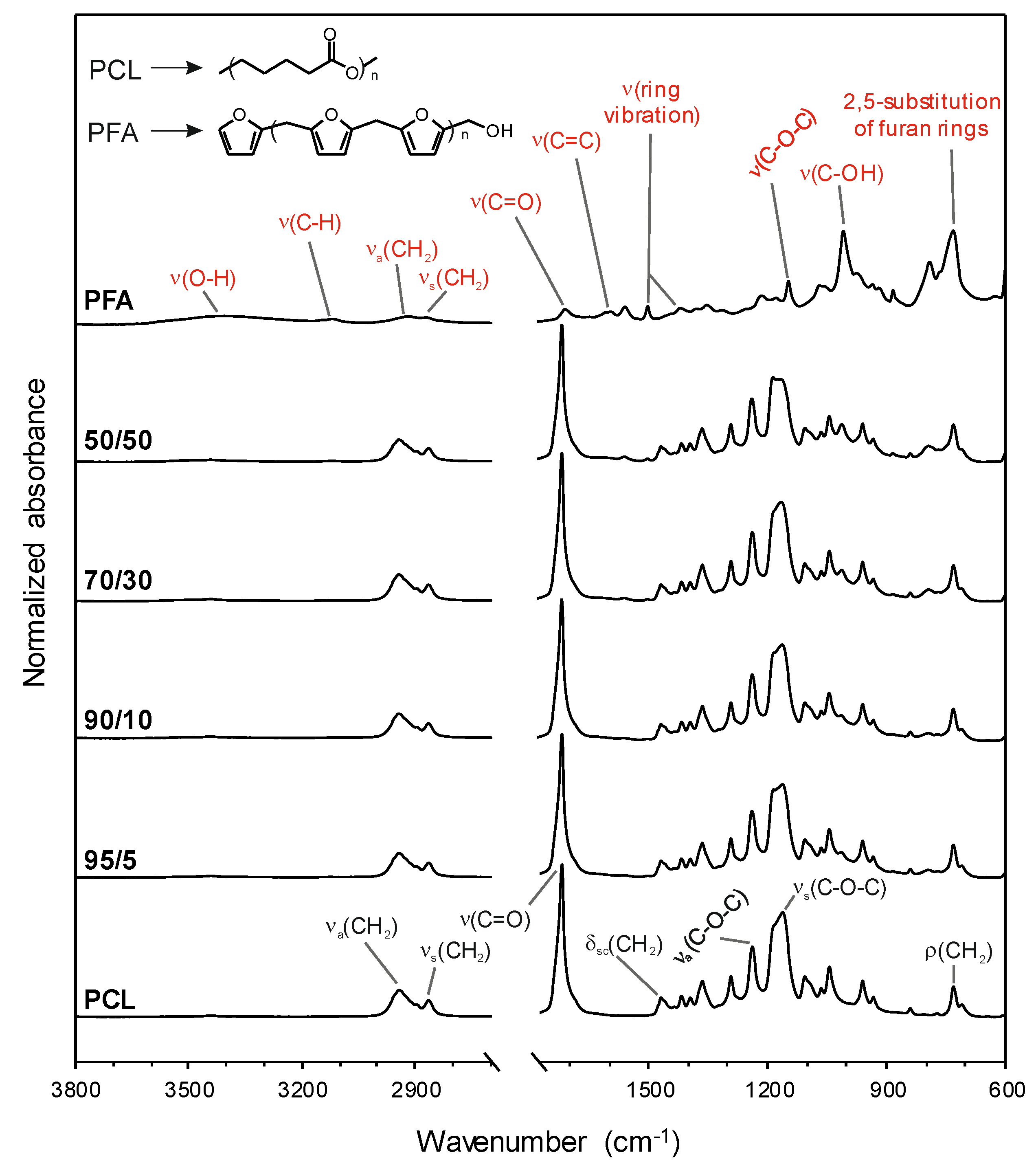
3.2. Chemical Analysis
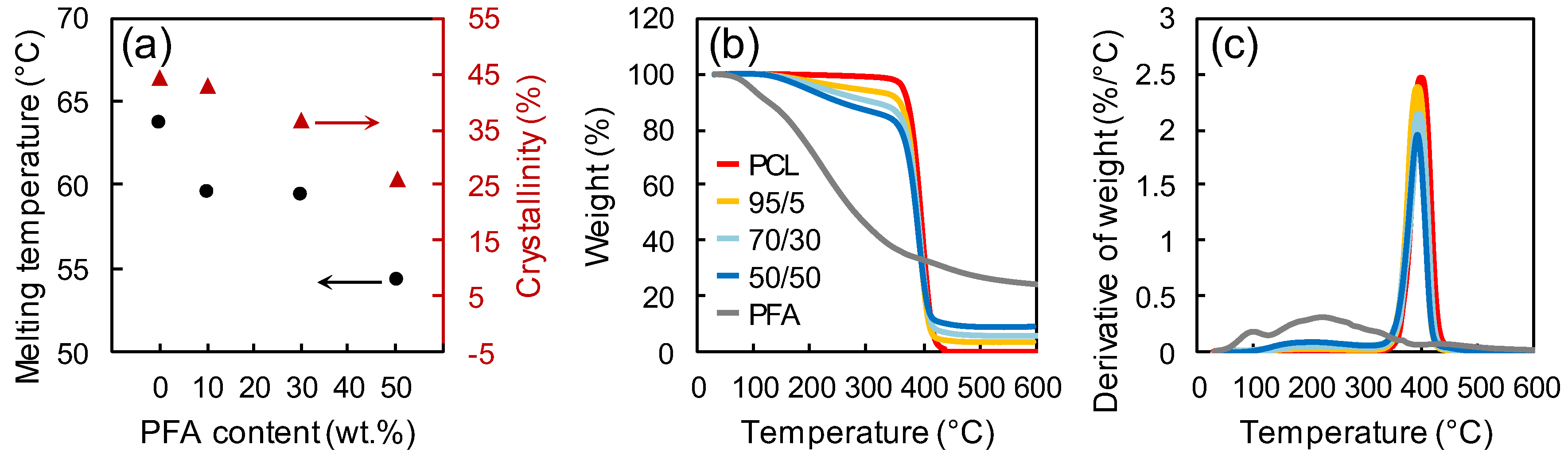
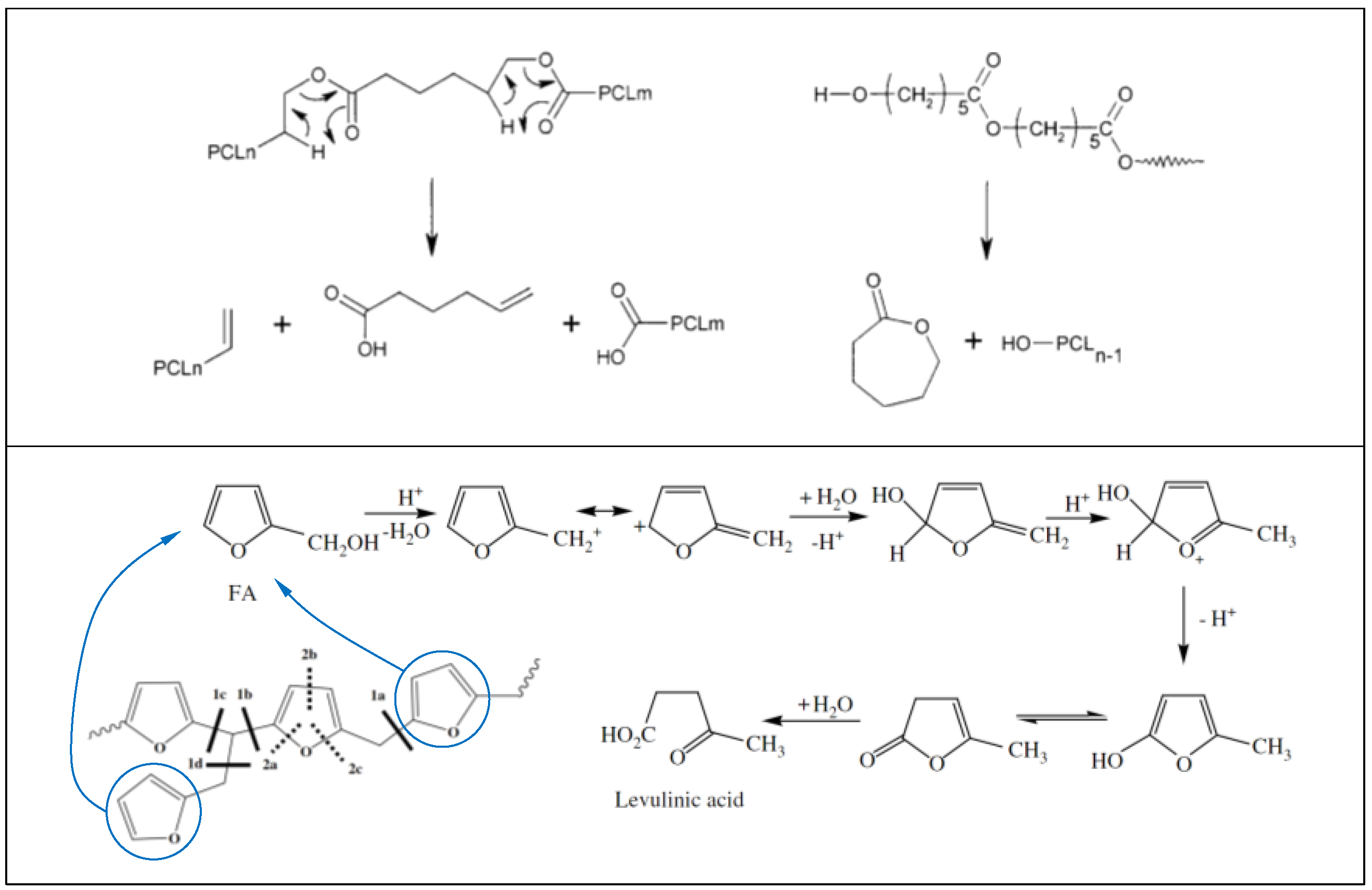
3.3. Thermal Properties and Crystallinity
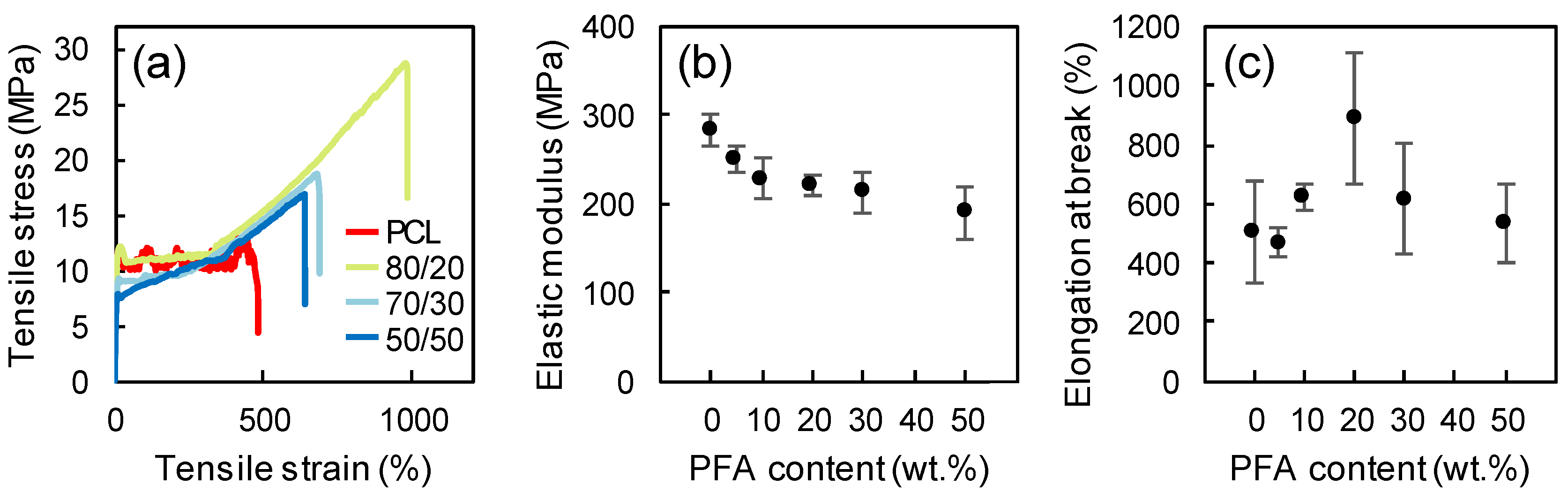
3.4. Mechanical Properties
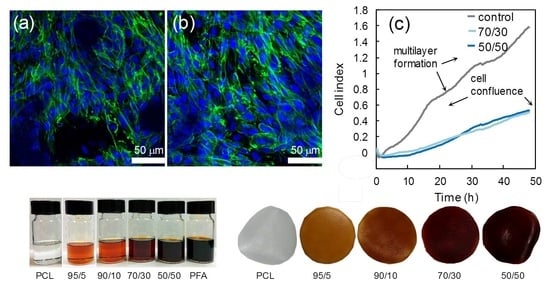
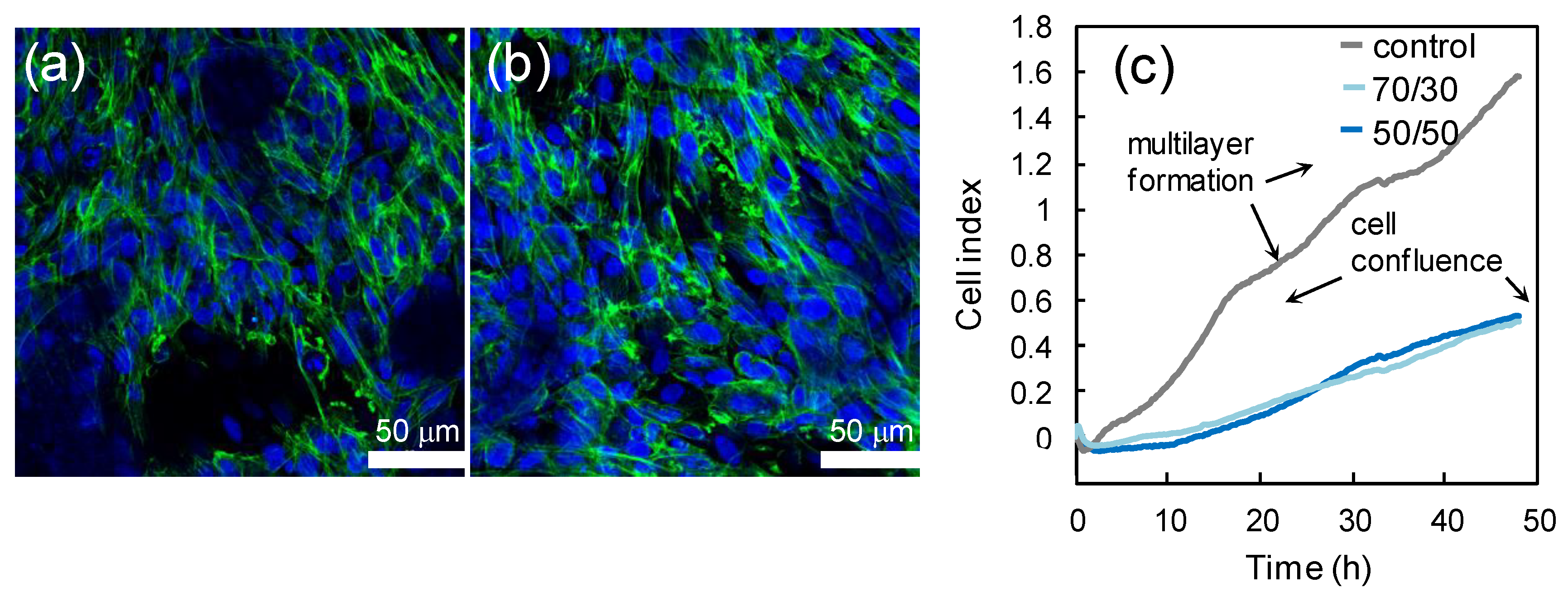
3.5. Biocompatibility Study
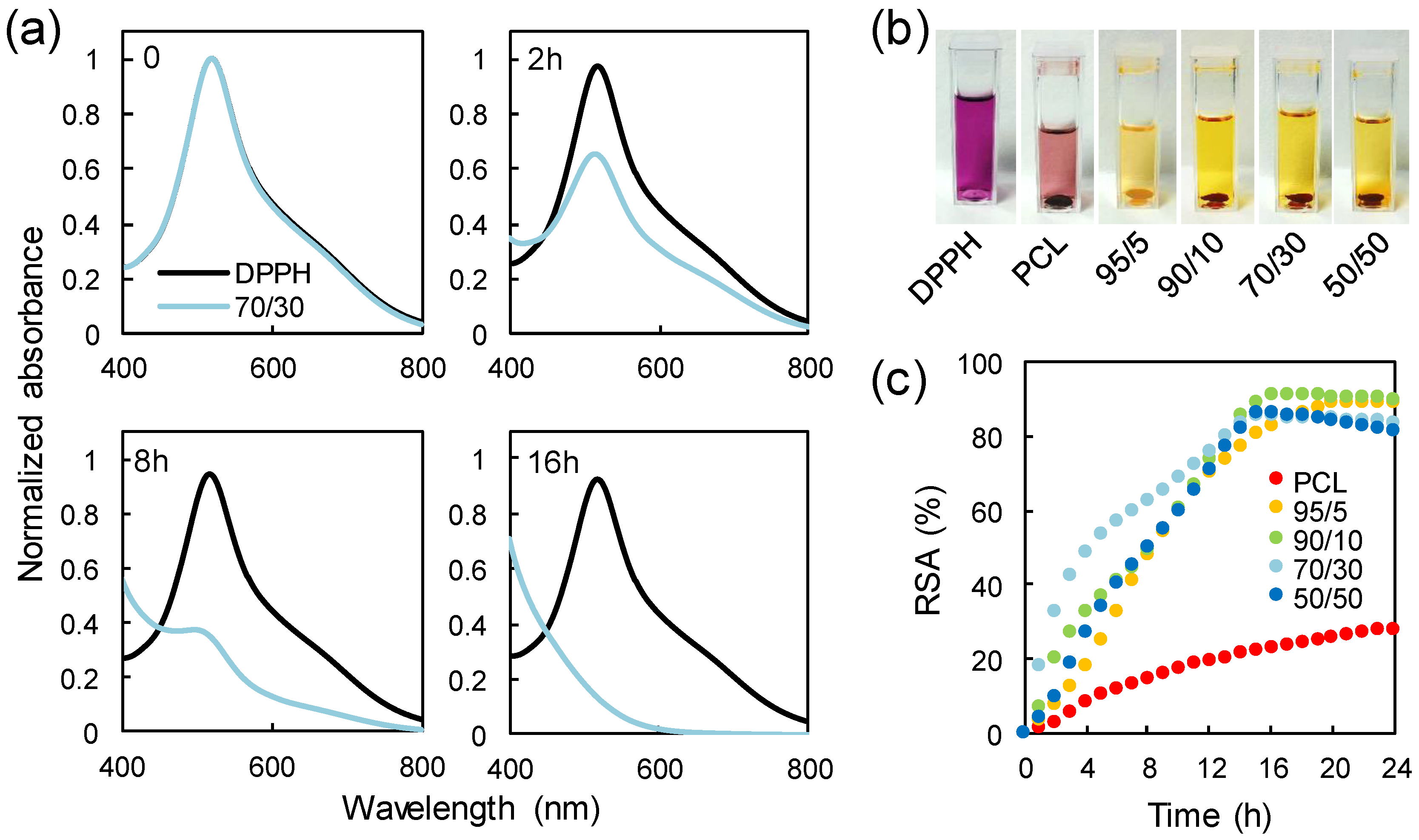
3.6. Antioxidant Properties
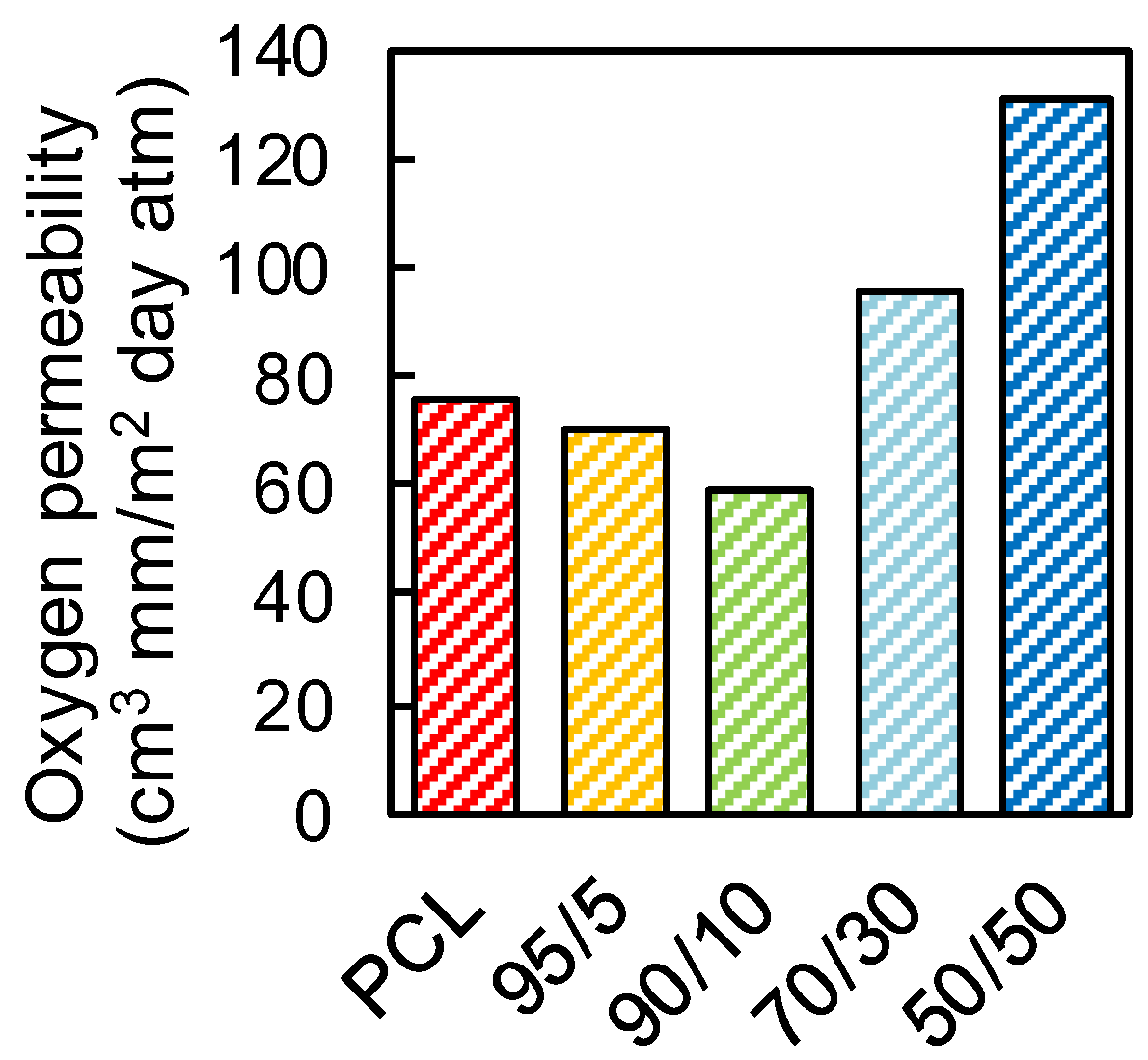
3.7. Gas Barrier Properties
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Mariscal, R.; Maireles-Torres, P.; Ojeda, M.; Sádaba, I.; López Granados, M. Furfural: A renewable and versatile platform molecule for the synthesis of chemicals and fuels. Energy Environ. Sci. 2016, 9, 1144–1189. [Google Scholar] [CrossRef]
- Chan, X.; Yang, P.; Ooi, C.; Cen, J.; Orlov, A.; Kim, T. Separation and purification of furfuryl alcohol monomer and oligomers using a two-phase extracting process. ACS Sustain. Chem. Eng. 2016, 4, 4084–4088. [Google Scholar] [CrossRef]
- Binder, J.; Blank, J.J.; Cefali, A.V.; Raines, R.T. Synthesis of furfural from xylose and xylan. ChemSusChem 2010, 3, 1268–1272. [Google Scholar] [CrossRef]
- Luckeneder, P.; Gavino, J.; Kuchernig, R.; Petutschnigg, A.; Tondi, G. Sustainable phenolic fractions as basis for furfuryl alcohol-based co-polymers and their use as wood adhesives. Polymers 2016, 8, 396. [Google Scholar] [CrossRef] [PubMed]
- Gardikes, J.J.; Young, D.K. Composition Containing Furfuryl Alcohol and Use Thereof in Foundry Binders. U.S. Patent 4,371,648, 1 February 1983. [Google Scholar]
- Wang, H.; Yao, J. Use of Poly(furfuryl alcohol) in the fabrication of nanostructured carbons and nanocomposites. Ind. Eng. Chem. Res. 2006, 45, 6393–6404. [Google Scholar] [CrossRef]
- Burket, C.L.; Rajagopalan, R.; Marencic, A.P.; Dronvajjala, K.; Foley, H.C. Genesis of porosity in polyfurfuryl alcohol derived nanoporous carbon. Carbon 2006, 44, 2957–2963. [Google Scholar] [CrossRef]
- Fitzer, E.; Schaefer, W.; Yamada, S. The formation of glasslike carbon by pyrolysis of polyfurfuryl alcohol and phenolic resin. Carbon 1969, 7, 643–646. [Google Scholar] [CrossRef]
- Pranger, L.A.; Nunnery, G.A.; Tannenbaum, R. Mechanism of the nanoparticle-catalyzed polymerization of furfuryl alcohol and the thermal and mechanical properties of the resulting nanocomposites. Compos. Part B Eng. 2012, 43, 1139–1146. [Google Scholar] [CrossRef]
- He, L.; Li, D.; Zhang, G.; Webley, P.A.; Zhao, D.; Wang, H. Synthesis of carbonaceous poly(furfuryl alcohol) membrane for water desalination. Ind. Eng. Chem. Res. 2010, 49, 4175–4180. [Google Scholar] [CrossRef]
- Guo, C.; Zhou, L.; Lv, J. Effects of expandable graphite and modified ammonium polyphosphate on the flame-retardant and mechanical properties of wood flour-polypropylene composites. Polym. Polym. Compos. 2013, 21, 449–456. [Google Scholar] [CrossRef]
- Jin, L.; Huanting, W.; Shaoan, C.; Kwong-Yu, C. Nafion-polyfurfuryl alcohol nanocomposite membranes for direct methanol fuel cells. J. Memb. Sci. 2005, 246, 95–101. [Google Scholar]
- Deka, H.; Misra, M.; Mohanty, A. Renewable resource based “all green composites” from kenaf biofiber and poly(furfuryl alcohol) bioresin. Ind. Crops Prod. 2013, 41, 94–101. [Google Scholar] [CrossRef]
- Toriz, G.; Arvidsson, R.; Westin, M.; Gatenholm, P. Novel cellulose ester-poly (furfuryl alcohol)-flax fiber biocomposites. J. Appl. Polym. Sci. 2003, 88, 337–345. [Google Scholar] [CrossRef]
- Guigo, N.; Mija, A.; Vincent, L.; Sbirrazzuoli, N. Eco-friendly composite resins based on renewable biomass resources: Polyfurfuryl alcohol/lignin thermosets. Eur. Polym. J. 2010, 46, 1016–1023. [Google Scholar] [CrossRef]
- Deka, H.; Mohanty, A.; Misra, M. Renewable-resource-based green blends from poly(furfuryl alcohol) bioresin and lignin. Macromol. Mater. Eng. 2014, 299, 552–559. [Google Scholar] [CrossRef]
- Pranger, L.; Tannenbaum, R. Biobased nanocomposites prepared by in situ polymerization of furfuryl alcohol with cellulose whiskers or montmorillonite clay. Macromolecules 2008, 41, 8682–8687. [Google Scholar] [CrossRef]
- Sharib, M.; Kumar, R.; Kumar, K.D. Polylactic acid incorporated polyfurfuryl alcohol bioplastics: Thermal, mechanical and curing studies. J. Therm. Anal. Calorim. 2018, 132, 1593–1600. [Google Scholar] [CrossRef]
- Gao, S.; Tang, G.; Hua, D.; Xiong, R.; Han, J.; Jiang, S.; Zhang, Q.; Huang, C. Stimuli-responsive bio-based polymeric systems and their applications. J. Mater. Chem. B 2019, 7, 709–729. [Google Scholar] [CrossRef]
- Hutmacher, D.W.; Schantz, T.; Zein, I.; Ng, K.W.; Teoh, S.H.; Tan, K.C. Mechanical properties and cell cultural response of polycaprolactone scaffolds designed and fabricated via fused deposition modeling. J. Biomed. Mater. Res. 2001, 55, 203–216. [Google Scholar] [CrossRef]
- Jenkins, M.J.; Harrison, K.L.; Silva, M.M.C.G.; Whitaker, M.J.; Shakesheff, K.M.; Howdle, S.M. Characterisation of microcellular foams produced from semi-crystalline PCL using supercritical carbon dioxide. Eur. Polym. J. 2006, 42, 3145–3151. [Google Scholar] [CrossRef]
- Peña, J.; Corrales, T.; Izquierdo-Barba, I.; Doadrio, A.L.; Vallet-Regí, M. Long term degradation of poly(ε-caprolactone) films in biologically related fluids. Polym. Degrad. Stab. 2006, 91, 1424–1432. [Google Scholar] [CrossRef]
- Sinha, V.R.; Bansal, K.; Kaushik, R.; Kumria, R.; Trehan, A. Poly-ϵ-caprolactone microspheres and nanospheres: An overview. Int. J. Pharm. 2004, 278, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Ikada, Y.; Tsuji, H. Biodegradable polyesters for medical and ecological applications. Macromol. Rapid Commun. 2000, 21, 117–132. [Google Scholar] [CrossRef]
- Labet, M.; Thielemans, W. Synthesis of polycaprolactone: A review. Chem. Soc. Rev. 2009, 38, 3484–3504. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.; Chang, F.C. Temperature-dependent phase behavior in poly (ϵ-caprolactone)-epoxy blends. Polymer 2001, 42, 2193–2199. [Google Scholar] [CrossRef]
- Fejos, M.; Molnár, K.; Karger-Kocsis, J. Epoxy/polycaprolactone systems with triple-shape memory effect: Electrospun nanoweb with and without graphene versus co-continuous morphology. Materials 2013, 6, 4489–4504. [Google Scholar] [CrossRef] [PubMed]
- Keshavan, S.; Oropesa-Nuñez, R.; Diaspro, A.; Canale, C.; Dante, S. Adhesion and migration of CHO cells on micropatterned single layer graphene. 2D Mater. 2017, 4, 025022. [Google Scholar] [CrossRef]
- Ke, N.; Wang, X.B.; Xu, X.; Abassi, Y.A. Mammalian Cell Viability: Methods and Protocols; Stoddart, M.J., Ed.; Humana Press: Totowa, NJ, USA, 2011. [Google Scholar]
- Bellamy, L.J. The Infrared Spectra of Complex Molecules; Chapman and Hall: New York, NY, USA, 1975. [Google Scholar]
- Elzein, T.; Nasser-Eddine, M.; Delaite, C.; Bistac, S.; Dumas, P. FTIR study of polycaprolactone chain organization at interfaces. J. Colloid Interface Sci. 2004, 273, 381–387. [Google Scholar] [CrossRef]
- Choura, M.; Belgacem, N.M.; Gandini, A. Acid-catalyzed polycondensation of furfuryl alcohol: Mechanisms of chromophore formation and cross-linking. Macromolecules 1996, 29, 3839–3850. [Google Scholar] [CrossRef]
- Wang, Y.; Rodriguez-Perez, M.A.; Reis, R.L.; Mano, J.F. Thermal and thermomechanical behaviour of polycaprolactone and starch/polycaprolactone blends for biomedical applications. Macromol. Mater. Eng. 2005, 290, 792–801. [Google Scholar] [CrossRef]
- Ray, S.S. Environmentally Friendly Polymer Nanocomposites: Types, Processing and Properties; Woodhead Publishing: Cambridge, UK, 2013. [Google Scholar]
- Guigo, N.; Mija, A.; Zavaglia, R.; Vincent, L.; Sbirrazzuoli, N. New insights on the thermal degradation pathways of neat poly(furfuryl alcohol) and poly(furfuryl alcohol)/SiO2 hybrid materials. Polym. Degrad. Stab. 2009, 94, 908–913. [Google Scholar] [CrossRef]
- Persenaire, O.; Alexandre, M.; Degée, P.; Dubois, P. Mechanisms and kinetics of thermal degradation of poly(ε-caprolactone). Biomacromolecules 2001, 2, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Vieira, M.G.A.; Da Silva, M.A.; Dos Santos, L.O.; Beppu, M.M. Natural-based plasticizers and biopolymer films: A review. Eur. Polym. J. 2011, 47, 254–263. [Google Scholar] [CrossRef]
- Farah, S.; Anderson, D.G.; Langer, R. Physical and mechanical properties of PLA, and their functions in widespread applications-A comprehensive review. Adv. Drug Deliv. Rev. 2016, 107, 367–392. [Google Scholar] [CrossRef] [PubMed]
- Fuster, M.D.; Mitchell, A.E.; Ochi, H.; Shibamoto, T. Antioxidative activities of heterocyclic compounds formed in brewed coffee. J. Agric. Food Chem. 2000, 3, 5600–5603. [Google Scholar] [CrossRef]
- Wendlinger, C.; Vetter, W. High concentrations of furan fatty acids in organic butter samples from the German market. J. Agric. Food Chem. 2014, 62, 8740–8744. [Google Scholar] [CrossRef]
- Goldberg, D.M.; Hoffman, B.; Yang, J.; Soleas, G.J. Phenolic constituents, furans, and total antioxidant status of distilled spirits. J. Agric. Food Chem. 1999, 47, 3978–3985. [Google Scholar] [CrossRef]
- Tran, T.N.; Heredia-Guerrero, J.A.; Mai, B.T.; Ceseracciu, L.; Marini, L.; Athanassiou, A.; Bayer, I.S. Bioelastomers based on cocoa shell waste with antioxidant ability. Adv. Sustain. Syst. 2017, 1, 1700002. [Google Scholar] [CrossRef]
- Siparsky, G.L.; Voorhees, K.J.; Miao, F. Hydrolysis of polylactic acid (PLA) and polycaprolactone (PCL) in aqueous acetonitrile solutions: Autocatalysis. J. Environ. Polym. Degrad. 1998, 6, 31–41. [Google Scholar] [CrossRef]
- Reano, A.F.; Pion, F.; Domenek, S.; Ducrot, P.H.; Allais, F. Chemo-enzymatic preparation and characterization of renewable oligomers with bisguaiacol moieties: Promising sustainable antiradical/antioxidant additives. Green Chem. 2016, 18, 3334–3345. [Google Scholar] [CrossRef]
- Kai, D.; Ren, W.; Tian, L.; Chee, P.L.; Liu, Y.; Ramakrishna, S.; Loh, X.J. Engineering poly(lactide)-lignin nanofibers with antioxidant activity for biomedical application. ACS Sustain. Chem. Eng. 2016, 4, 5268–5276. [Google Scholar] [CrossRef]
- Aytac, Z.; Uyar, T. Antioxidant activity and photostability of α-tocopherol/β-cyclodextrin inclusion complex encapsulated electrospun polycaprolactone nanofibers. Eur. Polym. J. 2016, 79, 140–149. [Google Scholar] [CrossRef]
- Xie, J.; Schaich, K.M. Re-evaluation of the 2,2-diphenyl-1-picrylhydrazyl free radical (DPPH) assay for antioxidant activity. J. Agric. Food Chem. 2014, 62, 4251–4260. [Google Scholar] [CrossRef] [PubMed]
- Schaich, K.M.; Tian, X.; Xie, J. Hurdles and pitfalls in measuring antioxidant efficacy: A critical evaluation of ABTS, DPPH, and ORAC assays. J. Funct. Foods 2015, 18, 782–796. [Google Scholar] [CrossRef]
- Siracusa, V. Food packaging permeability behaviour: A report. Int. J. Polym. Sci. 2012, 2012, 302029. [Google Scholar] [CrossRef]

| Polymer | Oxygen Permeability (cm3 mm)/(m2 day atm) |
|---|---|
| Poly(ethylene terephthalate) (PET) | 1–5 |
| Polypropylene (PP) | 50–100 |
| Polyethylene (PE) | 50–200 |
| Polystyrene (PS) | 100–150 |
| Poly(vinyl chloride) (PVC) | 2–8 |
| Poly(ethylene naphthalate) (PEN) | 0.5 |
| Polyamide (PA) | 0.1–1 (dry) |
| Poly(vinyl alcohol) (PVAL) | 0.02 (dry) |
| Ethylene vinyl alcohol (EVOH) | 0.001–0.01 (dry) |
| Poly(vinylidene chloride) (PVDC) | 0.01–0.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nanni, G.; Heredia-Guerrero, J.A.; Paul, U.C.; Dante, S.; Caputo, G.; Canale, C.; Athanassiou, A.; Fragouli, D.; Bayer, I.S. Poly(furfuryl alcohol)-Polycaprolactone Blends. Polymers 2019, 11, 1069. https://doi.org/10.3390/polym11061069
Nanni G, Heredia-Guerrero JA, Paul UC, Dante S, Caputo G, Canale C, Athanassiou A, Fragouli D, Bayer IS. Poly(furfuryl alcohol)-Polycaprolactone Blends. Polymers. 2019; 11(6):1069. https://doi.org/10.3390/polym11061069
Chicago/Turabian StyleNanni, Gabriele, José A. Heredia-Guerrero, Uttam C. Paul, Silvia Dante, Gianvito Caputo, Claudio Canale, Athanassia Athanassiou, Despina Fragouli, and Ilker S. Bayer. 2019. "Poly(furfuryl alcohol)-Polycaprolactone Blends" Polymers 11, no. 6: 1069. https://doi.org/10.3390/polym11061069
APA StyleNanni, G., Heredia-Guerrero, J. A., Paul, U. C., Dante, S., Caputo, G., Canale, C., Athanassiou, A., Fragouli, D., & Bayer, I. S. (2019). Poly(furfuryl alcohol)-Polycaprolactone Blends. Polymers, 11(6), 1069. https://doi.org/10.3390/polym11061069