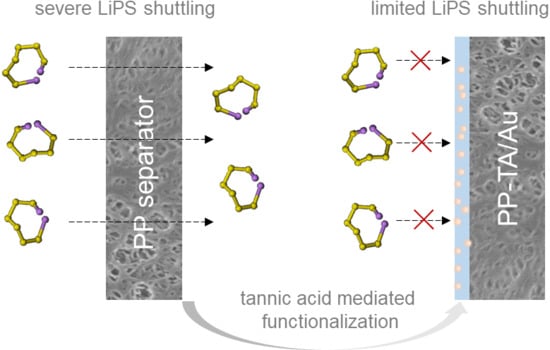
A Bioinspired Functionalization of Polypropylene Separator for Lithium-Sulfur Battery
Abstract
:1. Introduction
2. Materials and Methods
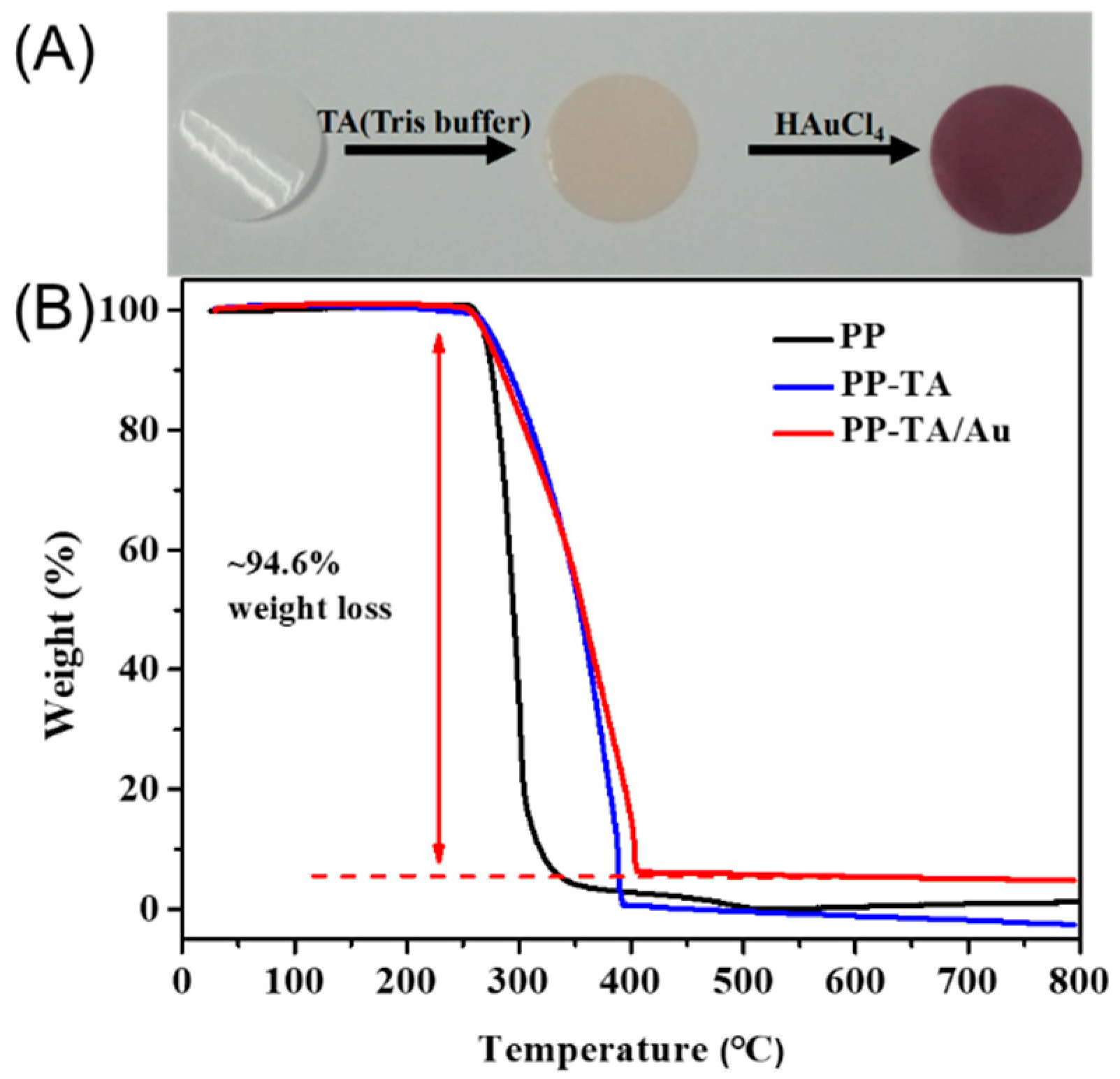
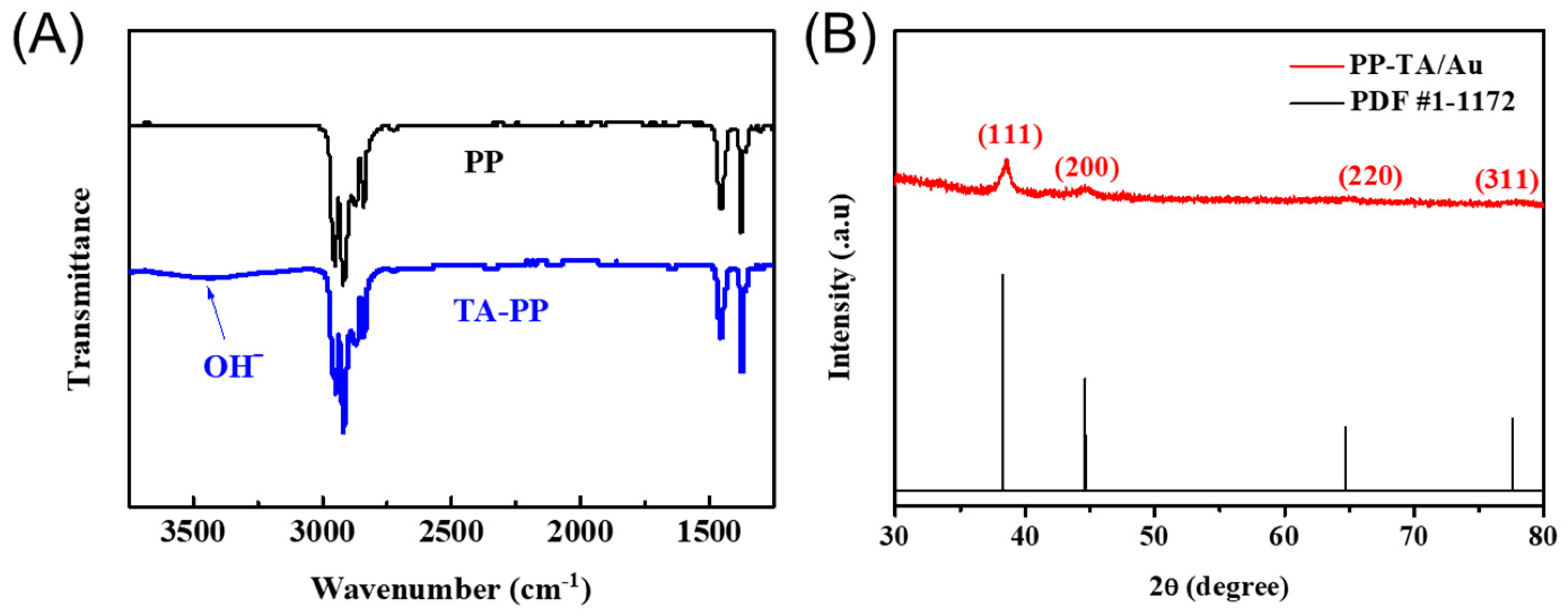
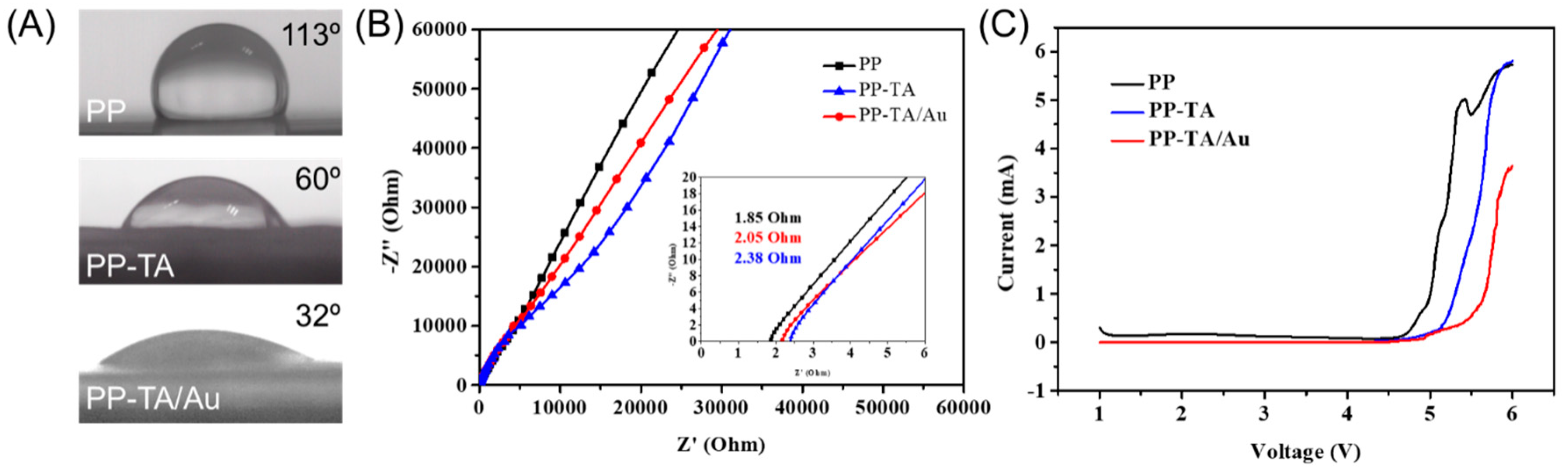
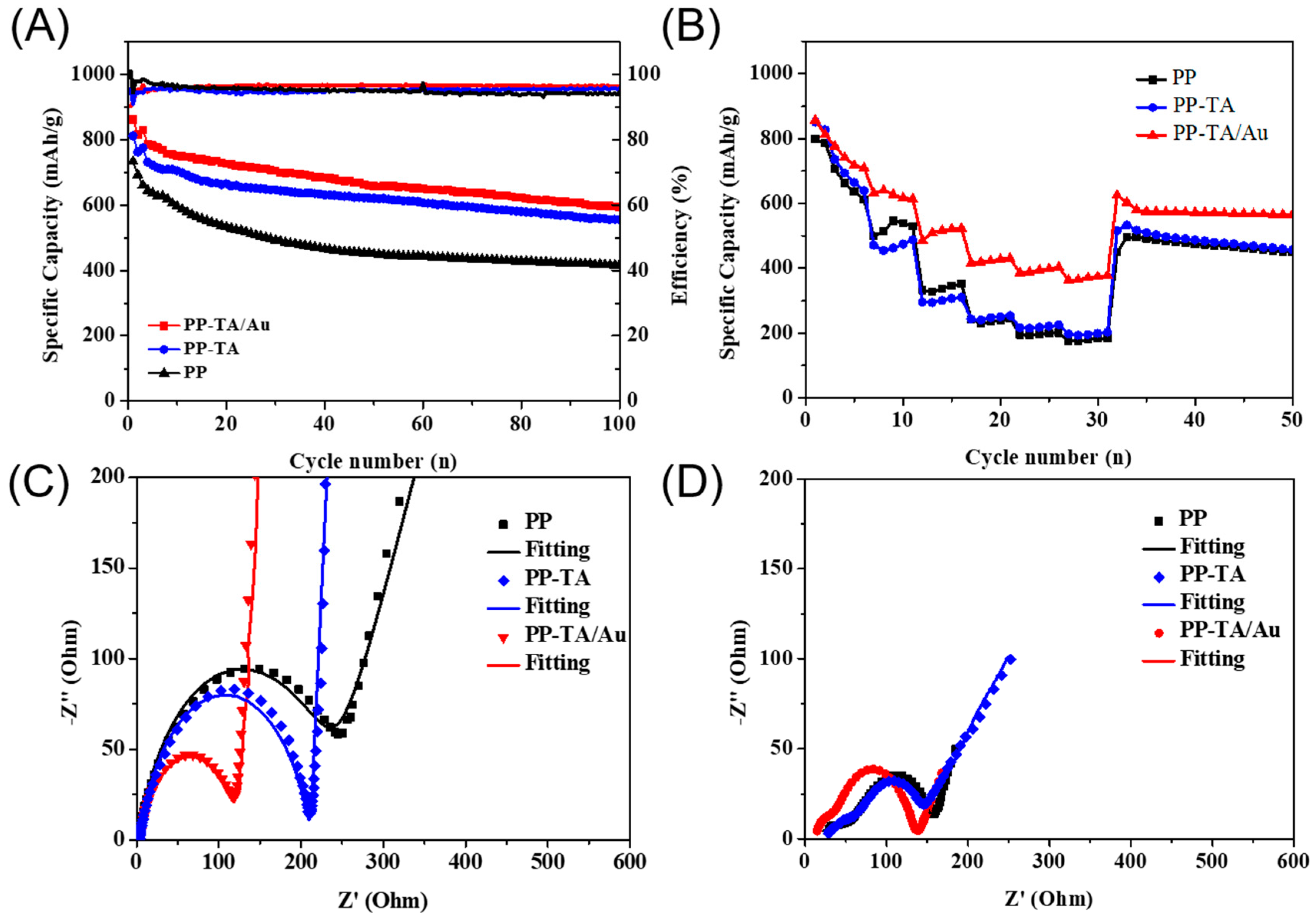
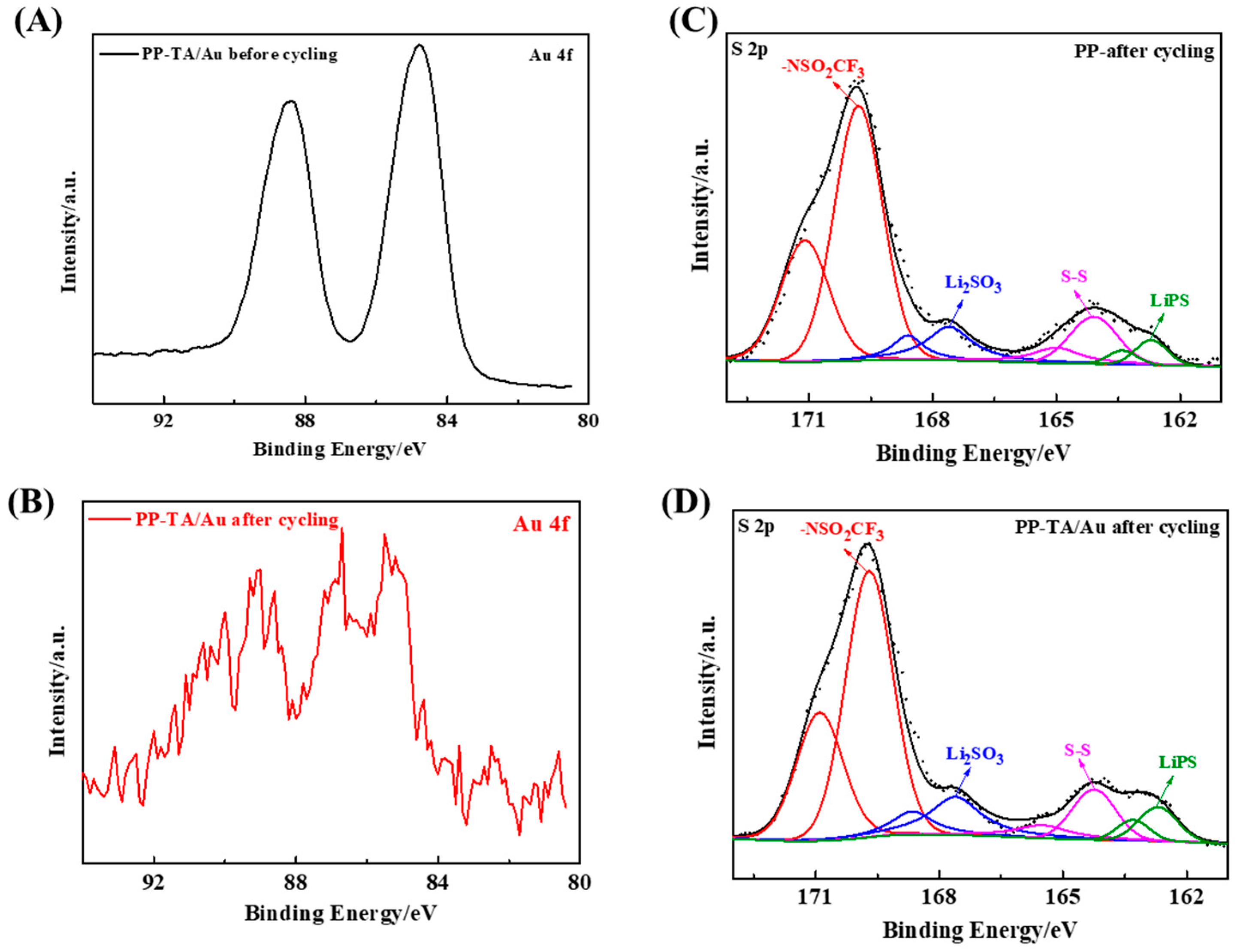
3. Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tao, T.; Lu, S.G.; Fan, Y.; Lei, W.W.; Huang, S.M.; Chen, Y. Anode Improvement in Rechargeable Lithium-Sulfur Batteries. Adv. Mater. 2017, 29, 1700542. [Google Scholar] [CrossRef]
- Peng, H.J.; Huang, J.Q.; Cheng, X.B.; Zhang, Q. Review on High-Loading and High-Energy Lithium-Sulfur Batteries. Adv. Energy Mater. 2017, 7, 1700260. [Google Scholar] [CrossRef]
- Liu, X.; Huang, J.Q.; Zhang, Q.; Mai, L.Q. Nanostructured Metal Oxides and Sulfides for Lithium-Sulfur Batteries. Adv. Mater. 2017, 29, 1601759. [Google Scholar] [CrossRef] [PubMed]
- Fang, R.P.; Zhao, S.Y.; Sun, Z.H.; Wang, W.; Cheng, H.M.; Li, F. More Reliable Lithium-Sulfur Batteries: Status, Solutions and Prospects. Adv. Mater. 2017, 29, 1606823. [Google Scholar] [CrossRef]
- Li, C.X.; Xi, Z.C.; Guo, D.X.; Chen, X.J.; Yin, L.W. Chemical Immobilization Effect on Lithium Polysulfides for Lithium-Sulfur Batteries. Small 2018, 14, 1701986. [Google Scholar] [CrossRef]
- Xiang, Y.; Li, J.; Lei, J.; Liu, D.; Xie, Z.; Qu, D.; Li, K.; Deng, T.; Tang, H. Advanced separators for lithium-ion and lithium-sulfur batteries: A review of recent progress. ChemSusChem 2016, 9, 3023–3039. [Google Scholar] [CrossRef]
- Qiu, W.; An, C.; Yan, Y.; Xu, J.; Zhang, Z.; Guo, W.; Wang, Z.; Zheng, Z.; Wang, Z.; Deng, Q.; et al. Suppressed polysulfide shuttling and improved Li+ transport in LiS batteries enabled by NbN modified PP separator. J. Power Sources 2019, 423, 98–105. [Google Scholar] [CrossRef]
- Zhang, L.; Wan, F.; Wang, X.; Cao, H.; Dai, X.; Niu, Z.; Wang, Y.; Chen, J. Dual-Functional Graphene Carbon as Polysulfide Trapper for High Performance Lithium Sulfur Batteries. ACS Appl. Mat. Interfaces 2018, 10, 5594–5602. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, Q.; Wei, F. Multi-functional separator/interlayer system for high-stable lithium-sulfur batteries: Progress and prospects. Energ. Storage Mater. 2015, 1, 127–145. [Google Scholar] [CrossRef]
- Chung, S.; Manthiram, A. Bifunctional Separator with a Light-Weight Carbon-Coating for Dynamically and Statically Stable Lithium-Sulfur Batteries. Adv. Mater. 2014, 24, 5299–5306. [Google Scholar] [CrossRef]
- Chung, S.; Manthiram, A. A Polyethylene Glycol-Supported Microporous Carbon Coating as a Polysulfide Trap for Utilizing Pure Sulfur Cathodes in Lithium–Sulfur Batteries. Adv. Mater. 2014, 26, 7352–7357. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Sun, M.; Liang, H.; Ying, W.; Zeng, X.; Ying, Y.; Zhou, S.; Liang, C.; Lin, Z.; Peng, X. Blocking Polysulfides and Facilitating Lithium-Ion Transport: Polystyrene Sulfonate@HKUST-1 Membrane for Lithium-Sulfur Batteries. ACS Appl. Mat. Interfaces 2018, 10, 30451–30459. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Fan, L.; Qiu, Y.; Wang, M.; Cheng, J.; Guan, B.; Guo, Z.; Zhang, N.; Sun, K. Ion-Selective Prussian-Blue-Modified Celgard Separator for High-Performance Lithium-Sulfur Battery. Chemsuschem 2018, 11, 3345–3351. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Chen, Z.; Chen, F.; Hu, G.; Wang, S.; Sun, Z.; Sun, X.; Li, F. From interlayer to lightweight capping layer: Rational design of mesoporous TiO2 threaded with CNTs for advanced Li-S batteries. Carbon 2019, 143, 523–530. [Google Scholar] [CrossRef]
- Zheng, B.; Yu, L.; Zhao, Y.; Xi, J. Ultralight carbon flakes modified separator as an effective polysulfide barrier for lithium-sulfur batteries. Electrochim. Acta 2019, 295, 910–917. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, Y.; Zhang, Y.; Xiang, M.; Wu, H.; Liu, H.; Dou, S. Interwoven V2O5 nanowire/graphene nanoscroll hybrid assembled as efficient polysulfide-trapping-conversion interlayer for long-life lithium-sulfur batteries. J. Mater. Chem. A 2018, 6, 19358–19370. [Google Scholar] [CrossRef]
- Xiang, Y.; Wang, Z.; Qiu, W.; Guo, Z.; Liu, D.; Qu, D.; Xie, Z.; Tang, H.; Li, J. Interfacing soluble polysulfides with a SnO2 functionalized separator: An efficient approach for improving performance of Li-S battery. J Membrane Sci 2018, 563, 380–387. [Google Scholar] [CrossRef]
- Yuan, H.; Peng, H.-J.; Li, B.-Q.; Xie, J.; Kong, L.; Zhao, M.; Chen, X.; Huang, J.-Q.; Zhang, Q. Conductive and Catalytic Triple-Phase Interfaces Enabling Uniform Nucleation in High-Rate Lithium-Sulfur Batteries. Adv. Energy Mater. 2019, 9, 1802768. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, C.; Zhou, G.; Lv, W.; Ling, G.; Zhi, L.; Yang, Q. Catalytic effects in lithium-sulfur batteries: Promoted sulfur transformation and reduced shuttle effect. Adv. Sci. 2018, 5, 1700270. [Google Scholar] [CrossRef]
- Zhang, B.-W.; Sheng, T.; Liu, Y.-D.; Wang, Y.-X.; Zhang, L.; Lai, W.-H.; Wang, L.; Yang, J.; Gu, Q.-F.; Chou, S.-L.; et al. Atomic cobalt as an efficient electrocatalyst in sulfur cathodes for superior room-temperature sodium-sulfur batteries. Nat. Commun. 2018, 9, 4082. [Google Scholar] [CrossRef]
- Imtiaz, S.; Zafar, Z.A.; Razaq, R.; Sun, D.; Xin, Y.; Li, Q.; Zhang, Z.; Zheng, L.; Huang, Y.; Anderson, J.A. Electrocatalysis on Separator Modified by Molybdenum Trioxide Nanobelts for Lithium-Sulfur Batteries. Advanced Materials Interfaces 2018, 5, 1800243. [Google Scholar] [CrossRef]
- Ali, S.; Waqas, M.; Jing, X.; Chen, N.; Chen, D.; Xiong, J.; He, W. Carbon-Tungsten Disulfide Composite Bilayer Separator for High Performance Lithium-Sulfur Batteries. ACS Appl. Mat. Interfaces 2018, 10, 39417–39421. [Google Scholar] [CrossRef]
- Hu, N.; Lv, X.; Dai, Y.; Fan, L.; Xiong, D.; Li, X. SnO2/Reduced Graphene Oxide Interlayer Mitigating the Shuttle Effect of Li-S Batteries. ACS Appl. Mat. Interfaces 2018, 10, 18665–18674. [Google Scholar] [CrossRef]
- Jiang, S.; Chen, M.; Wang, X.; Wu, Z.; Zeng, P.; Huang, C.; Wang, Y. MoS2-Coated N-doped Mesoporous Carbon Spherical Composite Cathode and CNT/Chitosan Modified Separator for Advanced Lithium Sulfur Batteries. ACS Sustainable Chem. Eng. 2018, 6, 16828–16837. [Google Scholar] [CrossRef]
- Wu, J.; Zeng, H.; Li, X.; Xiang, X.; Liao, Y.; Xue, Z.; Ye, Y.; Xie, X. Ultralight Layer-by-Layer Self-Assembled MoS2-Polymer Modified Separator for Simultaneously Trapping Polysulfides and Suppressing Lithium Dendrites. Adv. Energy Mater. 2018, 8. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, H.; Wang, S.; Deng, Y.; Qin, X.; Qin, X.; Chen, G. N-doped carbon-coated hollow carbon nanofibers with interspersed TiO2 for integrated separator of Li-S batteries. Electrochim. Acta 2019, 297, 641–649. [Google Scholar] [CrossRef]
- Ejima, H.; Richardson, J.J.; Liang, K.; Best, J.P.; van Koeverden, M.P.; Such, G.K.; Cui, J.; Caruso, F. One-Step Assembly of Coordination Complexes for Versatile Film and Particle Engineering. Science 2013, 341, 154–157. [Google Scholar] [CrossRef] [PubMed]
- Sedó, J.; Saiz-Poseu, J.; Busqué, F.; Ruiz-Molina, D. Catechol-Based Biomimetic Functional Materials. Adv. Mater. 2013, 5, 653–701. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.Y.; Zhu, W.Y.; Guo, W.; Lou, F.G.; Deng, B.H.; Liu, D.; Xie, Z.Z.; Li, J.S.; Qu, D.Y.; Tang, H.L. Controlled carbon coating of Fe2O3 nanotube with tannic acid: A bio-inspired approach toward high performance lithium-ion battery anode. J. Alloy. Compd. 2017, 719, 347–352. [Google Scholar] [CrossRef]
- Lin, Z.; Li, X.; Huang, W.; Zhu, X.; Wang, Y.; Shan, Z. Active Platinum Nanoparticles as a Bifunctional Promoter for Lithium Sulfur Batteries. Chem. Electro. Chem. 2017, 4, 1–7. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Li, X.; Yan, Y.; Zhu, W.; Shao, L.-H.; Li, J. A Bioinspired Functionalization of Polypropylene Separator for Lithium-Sulfur Battery. Polymers 2019, 11, 728. https://doi.org/10.3390/polym11040728
Zhang Z, Li X, Yan Y, Zhu W, Shao L-H, Li J. A Bioinspired Functionalization of Polypropylene Separator for Lithium-Sulfur Battery. Polymers. 2019; 11(4):728. https://doi.org/10.3390/polym11040728
Chicago/Turabian StyleZhang, Zhijia, Xuequan Li, Yawen Yan, Wenyi Zhu, Li-Hua Shao, and Junsheng Li. 2019. "A Bioinspired Functionalization of Polypropylene Separator for Lithium-Sulfur Battery" Polymers 11, no. 4: 728. https://doi.org/10.3390/polym11040728
APA StyleZhang, Z., Li, X., Yan, Y., Zhu, W., Shao, L.-H., & Li, J. (2019). A Bioinspired Functionalization of Polypropylene Separator for Lithium-Sulfur Battery. Polymers, 11(4), 728. https://doi.org/10.3390/polym11040728