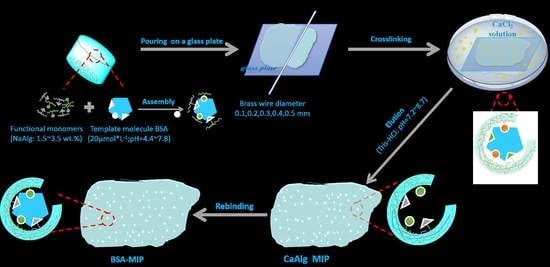
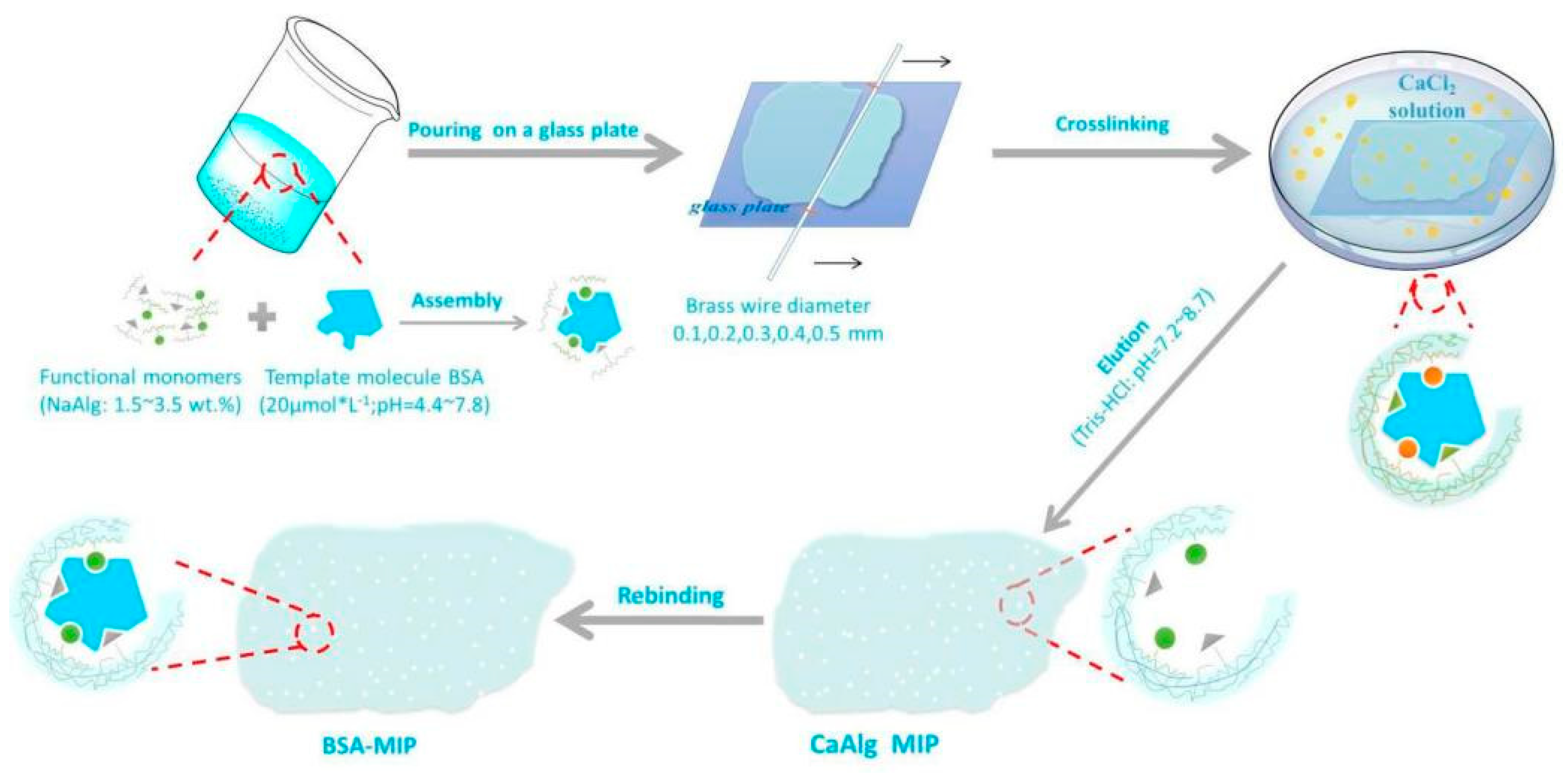
Adsorption and Electrochemical Detection of Bovine Serum Albumin Imprinted Calcium Alginate Hydrogel Membrane
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of CaAlg Hydrogel Membranes with Different NaAlg Concentrations
2.3. Preparation of BSA-Imprinted and Non-Imprinted CaAlg Hydrogel Membrane
2.4. Preparation of MIP and NIP CaAlg Hydrogel Membrane Electrochemical Sensor
2.5. Characterizations
2.5.1. Fluorescence Microscope
2.5.2. Circular Dichromatic Spectrum (CD) Analysis
2.6. Swelling Properties of CaAlg Hydrogel Membrane
2.7. BSA Rebinding of MIP and NIP CaAlg Membrane
2.8. Electrochemical Detection
3. Results and Discussion
3.1. The Fluorescence Microscope Images of MIP Hydrogel Membrane
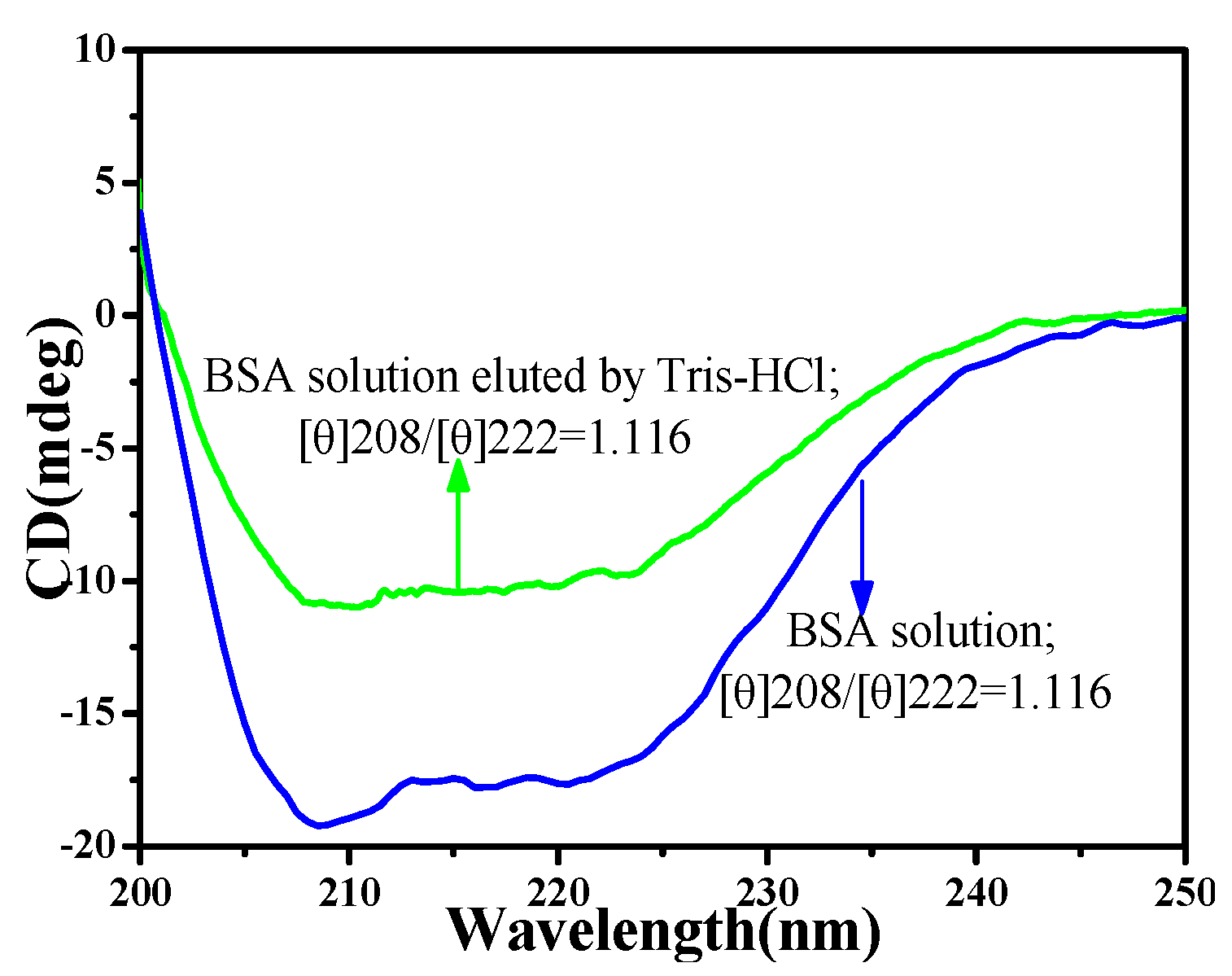
3.2. The Circular Dichromatic Spectrum (CD) of BSA before and After Being Eluted
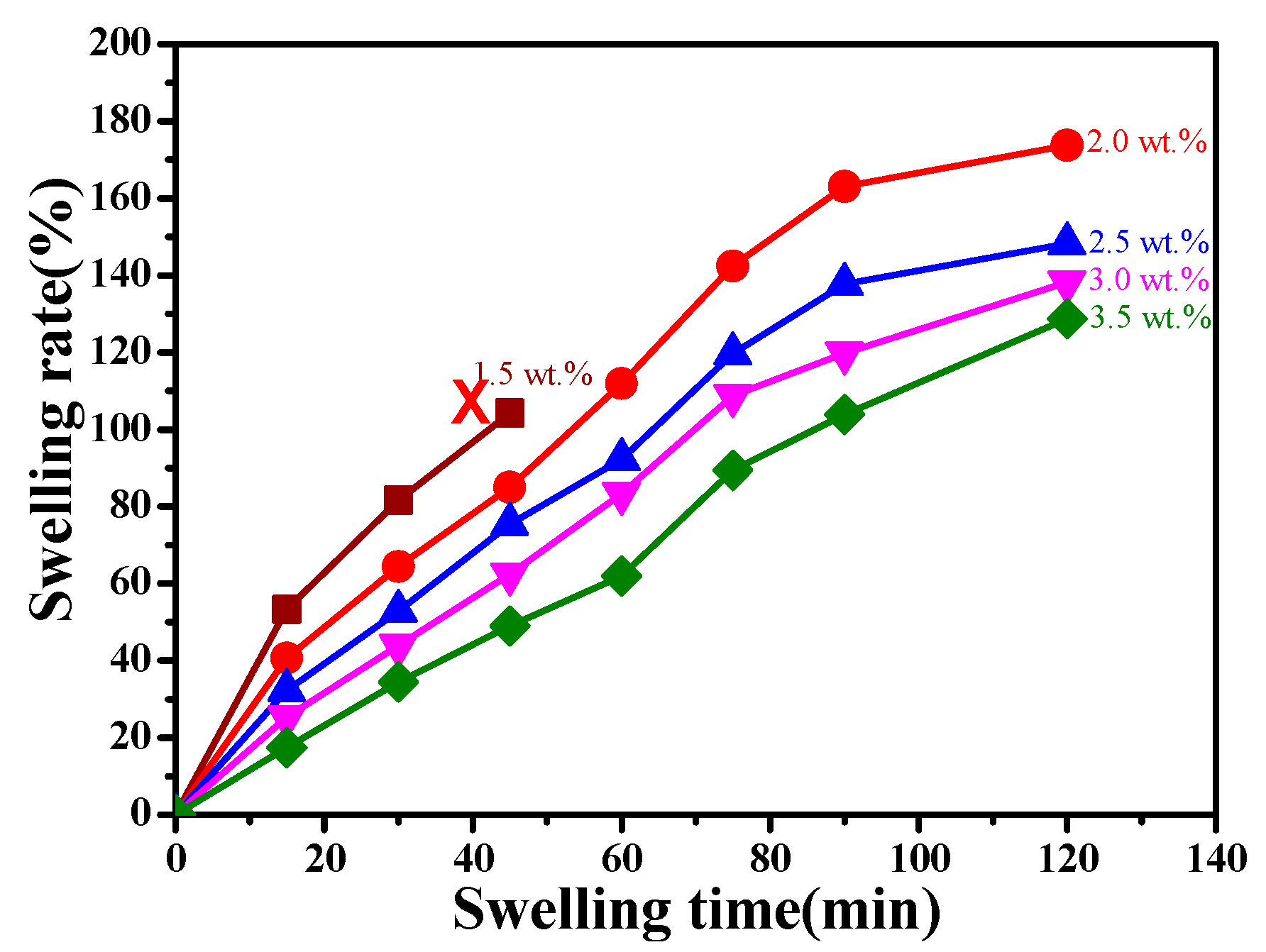
3.3. Swelling Properties of CaAlg Hydrogels Prepared with Different NaAlg Concentrations
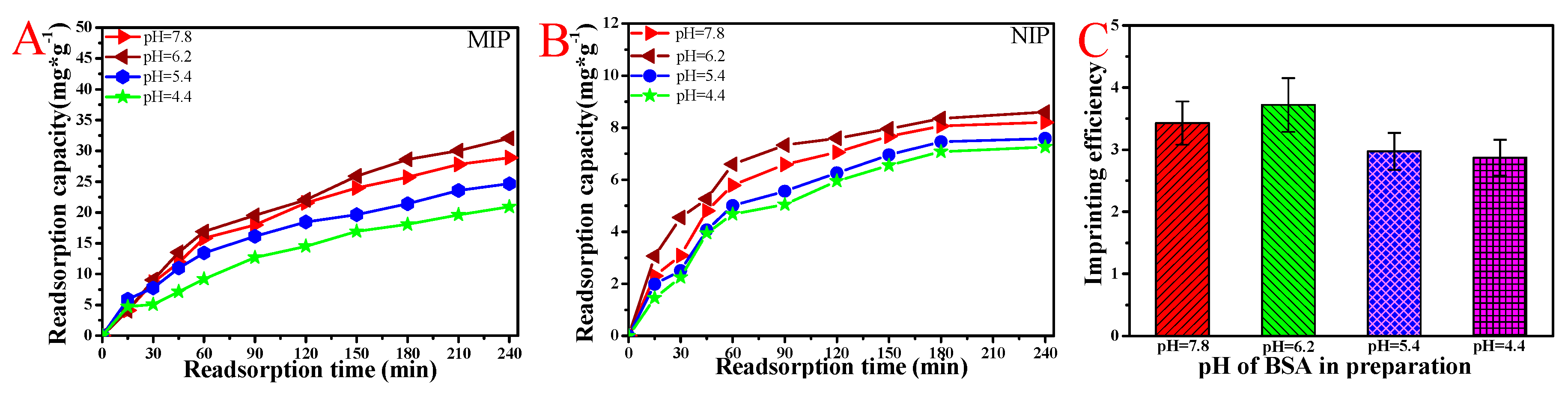
3.4. BSA Rebinding of MIP and NIP Hydrogel Prepared under Different pH Values of BSA
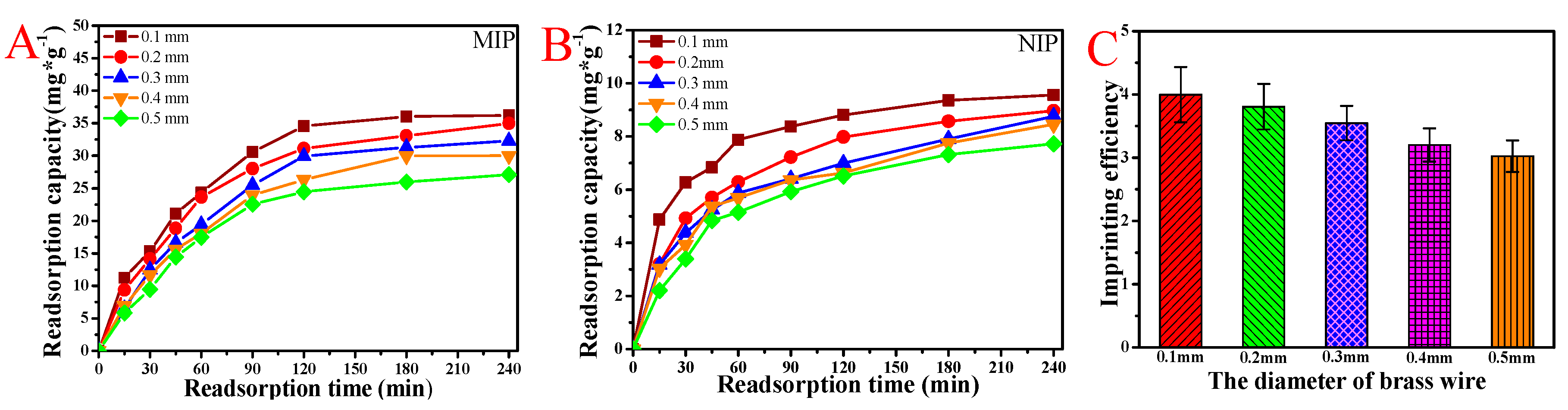
3.5. BSA Rebinding of the MIP and NIP Membranes Scraped with Different Diameter Brass Wire
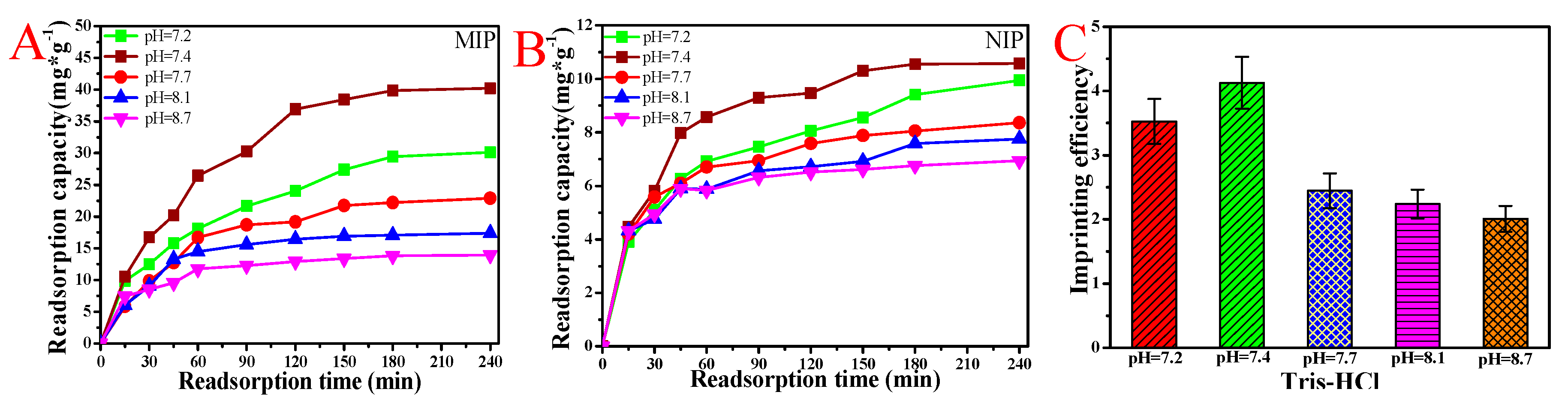
3.6. BSA Rebinding of MIP and NIP Hydrogel Eluted by Tris-HCl at Different pH Values
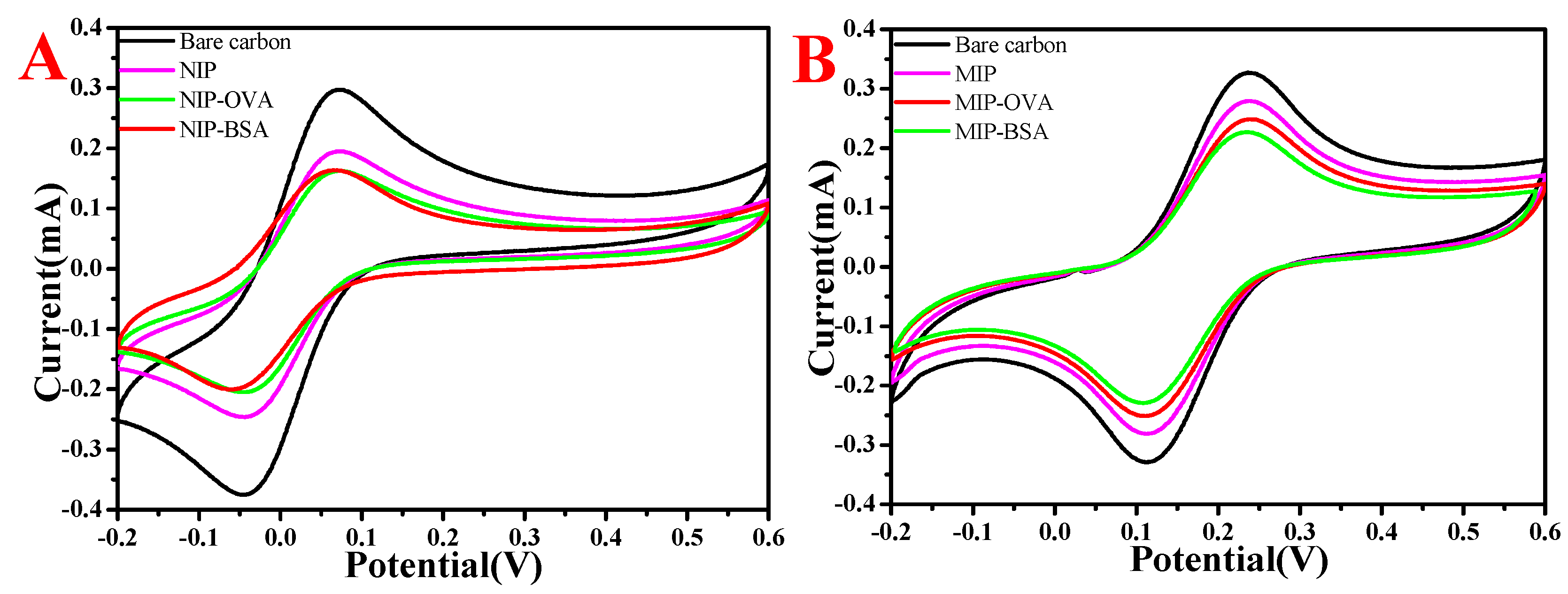
3.7. Electrochemical Characterization of MIP and NIP CaAlg Hydrogel Membrane Electrochemical Sensor
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Liu, L.; Chen, C.; Chen, C.; Kang, X.; Zhang, H.; Tao, Y.; Xie, Q.; Yao, S. Poly(noradrenalin) based bi-enzyme biosensor for ultrasensitive multi-analyte determination. Talanta 2018, 194, 343–349. [Google Scholar] [CrossRef]
- Pakchin, P.; Ghanbari, H.; Saber, R.; Omidi, Y. Electrochemical immunosensor based on chitosan/gold nanoparticle-carbon nanotube as platform and lactate oxidase as the label for the detection of CA125. Biosens. Bioelectron. 2018, 122, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Kumari, P.; Adeloju, S.B. Fabrication of a novel DNA affinity biosensor based on hybridisation induced current by electrostatic repulsion of silicotungstic acid as a redox indicator. Talanta 2019, 194, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Saeed, A.A.; Sánchez, J.L.A.; O’Sullivan, C.K.; Abbas, M.N. DNA biosensors based on gold nanoparticles-modified graphene oxide for the detection of breast cancer biomarkers for early diagnosis. Bioelectrochemistry 2017, 118, 91–99. [Google Scholar] [CrossRef]
- Gandhi, M.S.A.; Babu, P.R.; Senthilnathan, K.; Li, Q. High sensitivity photonic crystal fiber-based refractive index microbiosensor. Opt. Fiber Technol. 2018, 46, 88–94. [Google Scholar] [CrossRef]
- Gui, R.; Jin, H.; Guo, H.; Wang, Z. Recent advances and future prospects in molecularly imprinted polymers-based electrochemical biosensors. Biosens. Bioelectron. 2018, 100, 56–70. [Google Scholar] [CrossRef]
- Wei, Y.; Zeng, Q.; Hu, Q.; Wang, M.; Tao, J.; Wang, L. Self-cleaned electrochemical protein imprinting biosensor basing on a thermo-responsive memory hydrogel. Biosens. Bioelectron. 2018, 99, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Huynh, T.-P.; Sharma, P.S.; Sosnowska, M.; D’ Souza, F.; Kutner, W. Functionalized polythiophenes: Recognition materials for chemosensors and biosensors of superior sensitivity, selectivity, and detectability. Prog. Polym. Sci. 2015, 47, 1–25. [Google Scholar] [CrossRef]
- Zhao, K.; Feng, L.; Lin, H.; Fu, Y.; Lin, B.; Cui, W. Adsorption and photocatalytic degradation of methyl orange imprinted composite membranes using TiO2/calcium alginate hydrogel as matrix. Catal. Today 2014, 236, 127–134. [Google Scholar] [CrossRef]
- Ayadi, H.; Mekhalif, T.; Salmi, Z.; Carbonnier, B.; Djazi, F.; Chehimi, M.M. Molecularly imprinted PVC beads for the recognition of proteins. J. Appl. Polym. Sci. 2016, 5, 133. [Google Scholar] [CrossRef]
- Sunayama, H.; Kitayama, Y.; Takeuchi, T. Regulation of protein-binding activities of molecularly imprinted polymers via post-imprinting modifications to exchange functional groups within the imprinted cavity. J. Mol. Recognit. 2017, 31, e2633. [Google Scholar] [CrossRef]
- Kamon, Y.; Takeuchi, T. Molecularly Imprinted Nanocavities Capable of Ligand-Binding Domain and Size/Shape Recognition for Selective Discrimination of Vascular Endothelial Growth Factor Isoforms. ACS Sens. 2018, 3, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Sulc, R.; Szekely, G.; Shinde, S.; Wierzbicka, C.; Vilela, F.; Bauer, D.; Sellergren, B. Phospholipid imprinted polymers as selective endotoxin scavengers. Sci. Rep. 2017, 7, 44299. [Google Scholar] [CrossRef]
- Pan, J.; Chen, W.; Ma, Y.; Pan, G. Molecularly imprinted polymers as receptor mimics for selective cell recognition. Chem. Soc. Rev. 2018, 47, 5574–5587. [Google Scholar] [CrossRef]
- Schirhagl, R.; Ren, K.; Zare, R.N. Surface-imprinted polymers in microfluidic devices. Sci. Chi. Chem. 2012, 55, 469–483. [Google Scholar] [CrossRef]
- Griffete, N.; Fresnais, J.; Espinosa, A.; Wilhelm, C.; Be, A.; Menager, C. Design of magnetic molecularly imprinted polymer nanoparticles for controlled release of doxorubicin under an alternative magnetic field in athermal conditions. Nanoscale 2015, 7, 18891–18896. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, L.; Qin, W. An Sci-based bibliometris study on molecular imprinting technique. Mater. Rev. 2015, 5, 105–109. [Google Scholar]
- Li, S.; Cao, S.; Whitcombe, M.J.; Piletsky, S.A. Size matters: Challenges in imprinting macromolecules. Prog. Polym. Sci. 2014, 39, 145–163. [Google Scholar] [CrossRef]
- Takeuchi, T.; Mori, T.; Kuwahara, A.; Ohta, T.; Oshita, A.; Sunayama, H.; Ooya, T. Conjugated-Protein Mimics with Molecularly Imprinted Reconstructible and Transformable Regions that are Assembled Using Space-Filling Prosthetic Groups. Angew. Chem. 2014, 126, 12979–12984. [Google Scholar] [CrossRef]
- Wang, X.; Yu, S.; Liu, W.; Fu, L.; Wang, Y.; Li, J.; Chen, L. Molecular imprinting based hybrid ratiometric fluorescence sensor for the visual determination of bovine hemoglobin. ACS Sens. 2018, 3, 378–385. [Google Scholar] [CrossRef]
- Xing, R.; Ma, Y.; Wang, Y.; Wen, Y.; Liu, Z. Specific recognition of proteins and peptides via controllable oriented surface imprinting of boronate affinity-anchored epitopes. Chem. Sci. 2019, 10, 1831–1835. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Lv, Z.; Tang, Q.; Gong, C.-B.; Lam, M.H.W.; Ma, X.-B.; Chow, C.-F. Photoresponsive molecularly imprinted hydrogel casting membrane for the determination of trace tetracycline in milk. J. Mol. Recognit. 2015, 29, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-H.; Zhang, S.-Q.; Yang, W.; Chen, X.-L.; Zhuang, Z.-X.; Xu, J.-G.; Wang, X.-R. Molecularly Imprinted Sol-Gel Nanotubes Membrane for Biochemical Separations. J. Am. Chem. Soc. 2004, 126, 4054–4055. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.W.; Chen, Z.; Zhao, K.; Liu, L.X.; Dong, X.; Wang, H.; Zhang, C.; Leng, X.G.; Zhang, L.H. Polypropylene non-woven supported fibronectin molecular imprinted calcium alginate/ polyacrylamide hydrogel membrane for cell adhesion. Chin. Chem. Lett. 2015, 26, 807–810. [Google Scholar] [CrossRef]
- Verheyen, E.; Schillemans, J.P.; van Wijk, M.; Demeniex, M.A.; Hennink, W.E.; van Nostrum, C.F. Challenges for the effective molecular imprinting of proteins. Biomaterials 2011, 32, 3008–3020. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Huang, J.; Ying, X.; Cheng, G. Macromolecularly imprinted calcium phosphate/alginate hybrid polymer microspheres with the surface imprinting of bovine serum albumin in inverse-phase suspension. J. Appl. Polym. Sci. 2008, 109, 2687–2693. [Google Scholar] [CrossRef]
- Liu, D.; Zhao, K.; Qi, M.; Li, S.; Xu, G.; Wei, J.; He, X. Preparation of protein molecular-imprinted polysiloxane membrane using calcium alginate membrane as matrix and its application for cell culture. Polymers 2018, 10, 170. [Google Scholar] [CrossRef]
- Kan, B.; Lin, B.; Zhao, K.; Zhang, X.; Feng, L.; Wei, J.; Fan, Y. Imprinting of bovine serum albumin in nonwoven polypropylene membrane supported polyacrylamide/calcium alginate interpenetrating polymer network hydrogel. RSC Adv. 2014, 4, 55846–55852. [Google Scholar] [CrossRef]
- Shiomi, T.; Matsui, M.; Mizukami, F.; Sakaguchi, K. A method for the molecular imprinting of hemoglobin on silica surfaces using silanes. Biomaterials 2005, 26, 5564–5571. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Chen, Z.; Zhao, K.; Kan, B.; Li, H.; Zhang, X.; Lin, B.; Zhang, L. Adsorption and sustained release of haemoglobin imprinted polysiloxane using a calcium alginate membrane as a matrix. RSC Adv. 2015, 5, 26977–26984. [Google Scholar] [CrossRef]
- Zhao, K.; Lin, B.; Cui, W.; Feng, L.; Chen, T.; Wei, J. Preparation and adsorption of bovine serum albumin-imprinted polyacrylamide hydrogel membrane grafted on non-woven polypropylene. Talanta 2014, 121, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Bayer, C.L.; Herrero, É.P.; Peppas, N.A. Alginate membranes as macromolecular imprinted matrices. J. Biomater. Sci. Polym. Ed. 2011, 22, 1523–1534. [Google Scholar] [CrossRef] [PubMed]
- Edgar, P.; Herrero, E.M.; Del, V.; Nicholas, A.P. Protein imprinting by means of alginate based polymer microcapsules. Ind. Eng. Chem. Res. 2010, 49, 9811–9814. [Google Scholar]
- Song, H.; Zhao, K.; Li, S.; Wei, M.; Zhang, Z.; Sun, P.; Zhuge, F.; Jiao, R. Preparation and characterization of protein molecular imprinted calcium alginate hydrogel film with controllable thickness. Sci. Sin. Technol. 2016, 46, 931–939. (In Chinese) [Google Scholar]
- Xie, J.; Ng, W.; Lee, L.Y.; Wang, C.-H. Encapsulation of protein drugs in biodegradable microparticles by co-axial electrospray. Colloid Interface Sci. 2008, 317, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Liza, N.; Sven, F.; Jens, B. Probing the mechanism of insulin fibril formation with insulin mutants. Biochemistry 2001, 40, 8397–8409. [Google Scholar]
- Kupai, J.; Razali, M.; Buyuktiryaki, S.; Kecili, R.; Szekely, G. Long-term stability and reusability of molecularly imprinted polymers. Polym. Chem. 2017, 8, 666–673. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, M.; Zhao, K.; Bao, Q.; Pan, P.; Zhao, Y.; Yang, Z.; Wang, H.; Wei, J. Adsorption and Electrochemical Detection of Bovine Serum Albumin Imprinted Calcium Alginate Hydrogel Membrane. Polymers 2019, 11, 622. https://doi.org/10.3390/polym11040622
Qi M, Zhao K, Bao Q, Pan P, Zhao Y, Yang Z, Wang H, Wei J. Adsorption and Electrochemical Detection of Bovine Serum Albumin Imprinted Calcium Alginate Hydrogel Membrane. Polymers. 2019; 11(4):622. https://doi.org/10.3390/polym11040622
Chicago/Turabian StyleQi, Meng, Kongyin Zhao, Qiwen Bao, Peng Pan, Yuwei Zhao, Zhengchun Yang, Huiquan Wang, and Junfu Wei. 2019. "Adsorption and Electrochemical Detection of Bovine Serum Albumin Imprinted Calcium Alginate Hydrogel Membrane" Polymers 11, no. 4: 622. https://doi.org/10.3390/polym11040622
APA StyleQi, M., Zhao, K., Bao, Q., Pan, P., Zhao, Y., Yang, Z., Wang, H., & Wei, J. (2019). Adsorption and Electrochemical Detection of Bovine Serum Albumin Imprinted Calcium Alginate Hydrogel Membrane. Polymers, 11(4), 622. https://doi.org/10.3390/polym11040622