The Influence of bFGF on the Fabrication of Microencapsulated Cartilage Cells under Different Shaking Modes
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
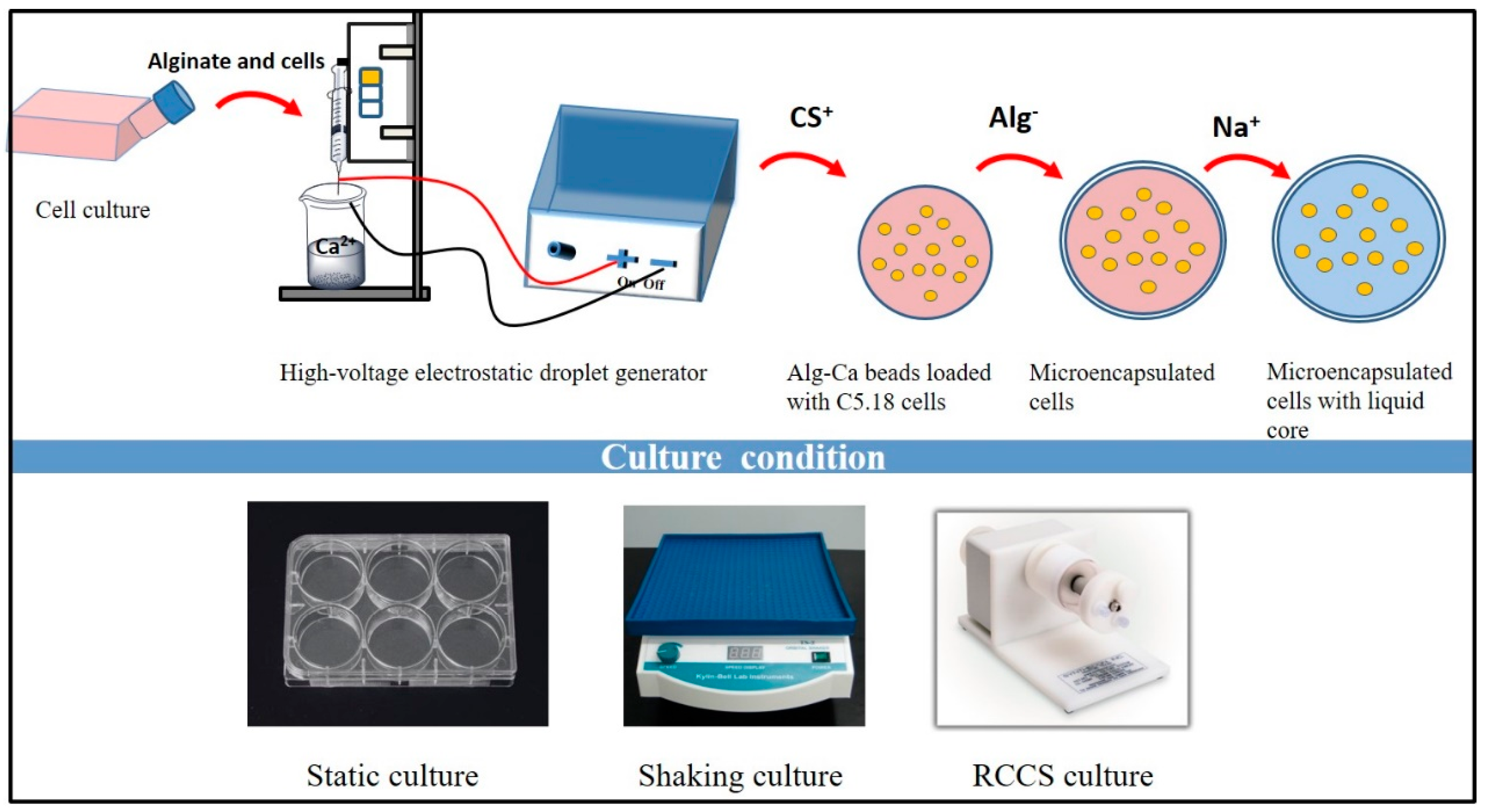
2.2. Preparation of Blank microcapsules and Microencapsulated C5.18 Cells (Artificial Cells)
2.3. Characterization of Morphology
2.4. Cell Proliferation Assay
2.5. AO/EB (Acridine Orange/Ethidium Bromide) Staining
2.6. Histological Staining
2.6.1. Hematoxylin-Eosin Staining of Cell Section
2.6.2. Alcian Blue Stain
2.6.3. Safranin-O
2.7. Immunohistochemistry (IHC) Staining
2.8. Glycosaminoglycan (GAG) Release [21]
2.9. Type II Collagen Secretion
2.10. Statistical Analysis
3. Results and Discussion
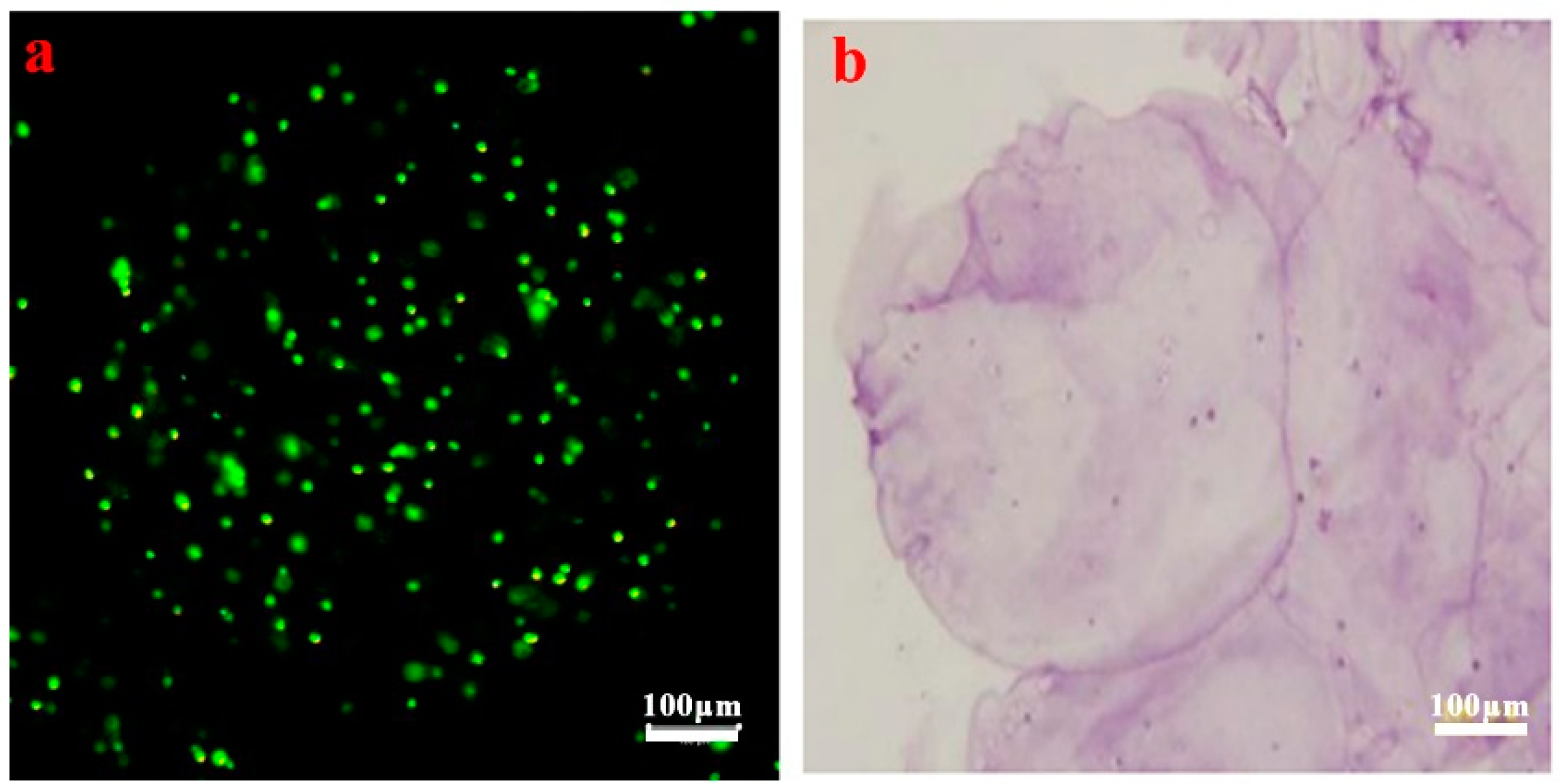
3.1. Morphology of Microcapsules and Artificial Cells
3.2. Cell Viability
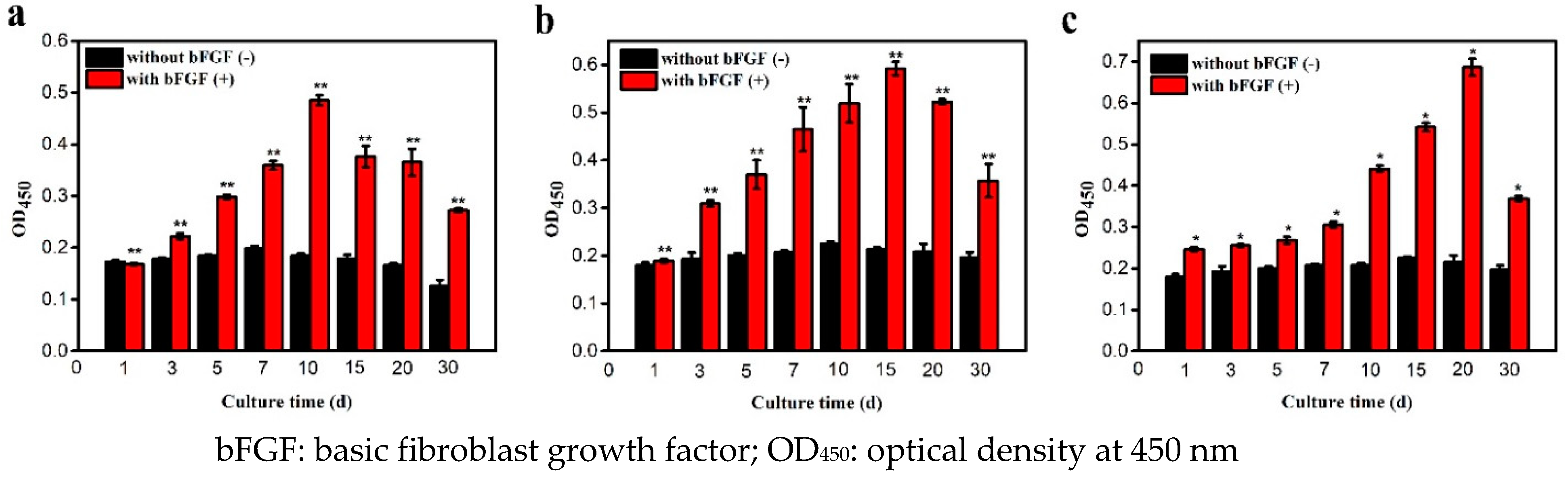
3.3. Cell Proliferation Assay
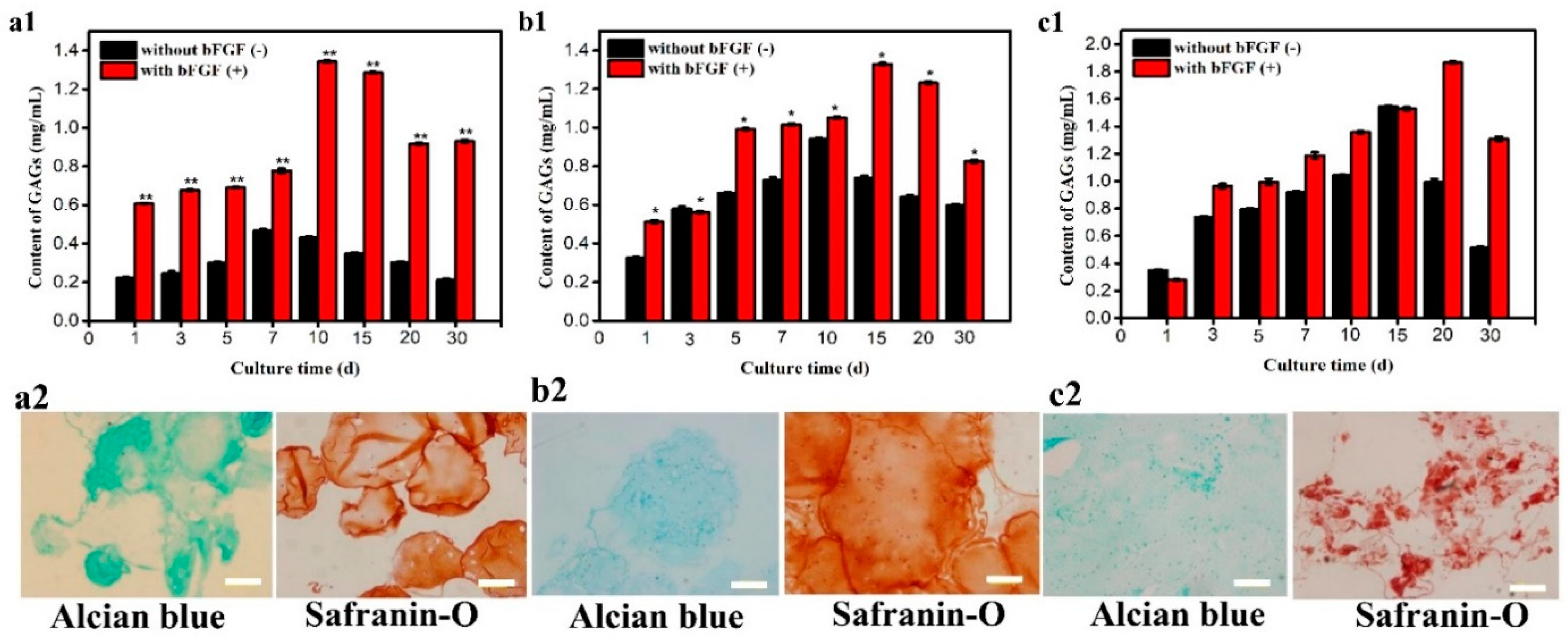
3.4. Concentration of GAG
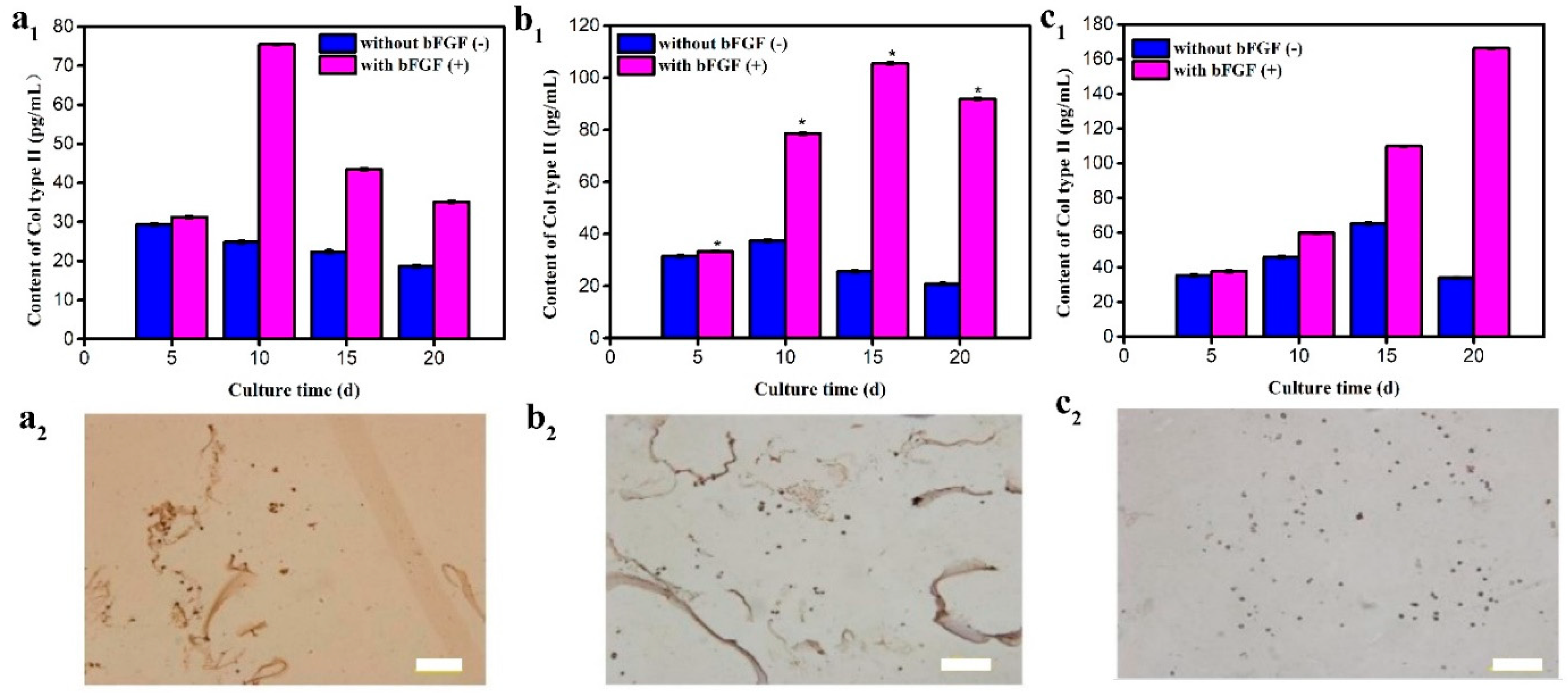
3.5. Type II Collagen Secretion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Huang, X.; Huang, X.; Jiang, X.-H.; Hu, F.-Q.; Du, Y.-Z.; Zhu, Q.-F.; Jin, C.-S. In vitro antitumour activity of stearic acid-g-chitosan oligosaccharide polymeric micelles loading podophyllotoxin. J. Microencapsul. 2012, 29, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hã, J.M.; Sedlarik, K.M. Advantages of alginate bandages for coverage of extensive and poorly healing wounds. Rozhledy v Chirurgii: Mesicnik Ceskoslovenske Chirurgicke Spolecnosti 1992, 71, 152–154. [Google Scholar]
- Chang, S.J.; Kuo, S.M.; Manousakas, I.; Niu, G.C.-C.; Chen, J.P. Preparation and characterization of hyaluronan/collagen ii microspheres under an electrostatic field system with disc electrodes. Acta Biomater. 2009, 5, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Koyano, T.; Minoura, N.; Nagura, M.; Kobayashi, K. Attachment and growth of cultured fibroblast cells on pva/chitosan-blended hydrogels. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 39, 486–490. [Google Scholar] [CrossRef]
- Chan, B.P.; Hui, T.Y.; Yeung, C.W.; Li, J.; Mo, I.; Chan, G.C.F. Self-assembled collagen-human mesenchymal stem cell microspheres for regenerative medicine. Biomaterials 2007, 28, 4652–4666. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Choi, J.K.; Huang, H.; Zhao, S.; Dumbleton, J.; Li, J.; He, X. A biomimetic core-shell platform for miniaturized 3d cell and tissue engineering. Part. Part. Syst. Charact. 2015, 32, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.J.; He, X.M. Microencapsulating and banking living cells for cell-based medicine. J. Healthc. Eng. 2011, 2, 427–446. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zhang, X.; Wang, X.; Wang, C.; Tang, B. Microenvironment of alginate-based microcapsules for cell culture and tissue engineering. J. Biosci. Bioeng. 2012, 114, 1–8. [Google Scholar] [CrossRef]
- Darling, E.M.; Athanasiou, K.A. Articular cartilage bioreactors and bioprocesses. Tissue Eng. 2003, 9, 9–26. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, X.; Wang, S.; Chen, A.; Wu, W.; Long, R. A novel cartilage tissue construction based on artificial cells and matrix-shaping. Mater. Lett. 2015, 159, 24–27. [Google Scholar] [CrossRef]
- Komura, M.; Komura, H.; Komuro, H.; Ikebukuro, K.; Hikita, A.; Hoshi, K.; Takato, T. Tracheal cartilage growth by intratracheal injection of basic fibroblast growth factor. J. Pediatr. Surg. 2017, 52, 235–238. [Google Scholar] [CrossRef]
- Losi, P.; Briganti, E.; Errico, C.; Lisella, A.; Sanguinetti, E.; Chiellini, F.; Soldani, G. Fibrin-based scaffold incorporating vegf- and bfgf-loaded nanoparticles stimulates wound healing in diabetic mice. Acta Biomater. 2013, 9, 7814–7821. [Google Scholar] [CrossRef]
- Li, X.; Su, G.; Wang, J.; Zhou, Z.; Li, L.; Liu, L.; Guan, M.; Zhang, Q.; Wang, H. Exogenous bfgf promotes articular cartilage repair via up-regulation of multiple growth factors. Osteoarthr. Cartil. 2013, 21, 1567–1575. [Google Scholar] [CrossRef]
- Bourget, L.; Chang, T.M. Phenylalanine ammonia-lyase immobilized in semipermeable microcapsules for enzyme replacement in phenylketonuria. Febs Lett. 1985, 180, 5–8. [Google Scholar] [CrossRef]
- Aubin, J.E.; Heersche, J.N.; Merrilees, M.J.; Sodek, J. Isolation of bone cell clones with differences in growth, hormone responses, and extracellular matrix production. J. Cell Biol. 1982, 92, 452–461. [Google Scholar] [CrossRef]
- Mandrycky, C.; Wang, Z.; Kim, K.; Kim, D.-H. 3d bioprinting for engineering complex tissues. Biotechnol. Adv. 2016, 34, 422–434. [Google Scholar] [CrossRef]
- Zhou, X.; Wu, K.; Long, R.; Kankala, R.K.; Wang, S.; Liu, Y. Preparation of a mvl-ca-alg/cs mems system with add-on effect for type 2 diabetes treatment. Int. J. Polym. Mater. Polym. 2017, 67, 1–7. [Google Scholar] [CrossRef]
- Zhang, L.; He, A.J.; Yin, Z.Q.; Yu, Z.Y.; Luo, X.S.; Liu, W.; Zhang, W.J.; Cao, Y.L.; Liu, Y.; Zhou, G.D. Regeneration of human-ear-shaped cartilage by co-culturing human microtia chondrocytes with bmscs. Biomaterials 2014, 35, 4878–4887. [Google Scholar] [CrossRef]
- Zhao, D.; Zhang, X.Y. Selenium antagonizes the lead-induced apoptosis of chicken splenic lymphocytes in vitro by activating the pi3k/akt pathway. Biol. Trace Elem. Res. 2018, 182, 119–129. [Google Scholar] [CrossRef]
- Fischer, A.H.; Jacobson, K.A.; Rose, J.; Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. CSH Protoc. 2008, 2008, pdb.prot4986. [Google Scholar] [CrossRef]
- Enobakhare, B.O.; Bader, D.L.; Lee, D.A. Quantification of sulfated glycosaminoglycans in chondrocyte/alginate cultures, by use of 1,9-dimethylmethylene blue. Anal. Biochem. 1996, 243, 189–191. [Google Scholar] [CrossRef]
- Du, M.; Zhu, Y.; Yuan, L.; Liang, H.; Mou, C.; Li, X.; Sun, J.; Zhuang, Y.; Zhang, W.; Shi, Q.; et al. Assembled 3d cell niches in chitosan hydrogel network to mimic extracellular matrix. Colloids Surf. A-Physicochem. Eng. Asp. 2013, 434, 78–87. [Google Scholar] [CrossRef]
- Farndale, R.W.; Buttle, D.J.; Barrett, A.J. Improved quantitation and discrimination of sulphated glycosaminoglycans by use of dimethylmethylene blue. Biochim. Biophys. Acta 1986, 883, 173–177. [Google Scholar] [CrossRef]
- Strand, B.L.; Gaserod, O.; Kulseng, B.; Espevik, T.; Skjak-Braek, G. Alginate-polylysine-alginate microcapsules: Effect of size reduction on capsule properties. J. Microencapsul. 2002, 19, 615–630. [Google Scholar] [CrossRef]
- Lu, D.B.; Zhang, Y.; Niu, S.Q.; Wang, L.T.; Lin, S.X.; Wang, C.M.; Ye, W.C.; Yan, C.L. Study of phenol biodegradation using bacillus amyloliquefaciens strain wjdb-1 immobilized in alginate-chitosan-alginate (aca) microcapsules by electrochemical method. Biodegradation 2012, 23, 209–219. [Google Scholar] [CrossRef]
- Shimoaka, T.; Ogasawara, T.; Yonamine, A.; Chikazu, D.; Kawano, H.; Nakamura, K.; Itoh, N.; Kawaguchi, H. Regulation of osteoblast, chondrocyte, and osteoclast functions by fibroblast growth factor (fgf)-18 in comparison with fgf-2 and fgf-10. J. Biol. Chem. 2002, 277, 7493–7500. [Google Scholar] [CrossRef]
- Wroblewski, J.; Edwall-Arvidsson, C. Inhibitory effects of basic fibroblast growth factor on chondrocyte differentiation. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1995, 10, 735–742. [Google Scholar] [CrossRef]
- Snyder, J.; Son, A.R.; Hamid, Q.; Wu, H.; Sun, W. Hetero-cellular prototyping by synchronized multi-material bioprinting for rotary cell culture system. Biofabrication 2016, 8, 015002. [Google Scholar] [CrossRef]
- Chen, J.-L.; Duan, L.; Zhu, W.; Xiong, J.; Wang, D. Extracellular matrix production in vitro in cartilage tissue engineering. J. Transl. Med. 2014, 12, 88. [Google Scholar] [CrossRef]
- Sharma, G.; Saxena, R.K.; Mishra, P. Differential effects of cyclic and static pressure on biochemical and morphological properties of chondrocytes from articular cartilage. Clin. Biomech. 2007, 22, 248–255. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, X.; Tang, X.; Long, R.; Wang, S.; Wang, P.; Cai, D.; Liu, Y. The Influence of bFGF on the Fabrication of Microencapsulated Cartilage Cells under Different Shaking Modes. Polymers 2019, 11, 471. https://doi.org/10.3390/polym11030471
Zhou X, Tang X, Long R, Wang S, Wang P, Cai D, Liu Y. The Influence of bFGF on the Fabrication of Microencapsulated Cartilage Cells under Different Shaking Modes. Polymers. 2019; 11(3):471. https://doi.org/10.3390/polym11030471
Chicago/Turabian StyleZhou, Xia, Xiaolin Tang, Ruimin Long, Shibin Wang, Pei Wang, Duanhua Cai, and Yuangang Liu. 2019. "The Influence of bFGF on the Fabrication of Microencapsulated Cartilage Cells under Different Shaking Modes" Polymers 11, no. 3: 471. https://doi.org/10.3390/polym11030471
APA StyleZhou, X., Tang, X., Long, R., Wang, S., Wang, P., Cai, D., & Liu, Y. (2019). The Influence of bFGF on the Fabrication of Microencapsulated Cartilage Cells under Different Shaking Modes. Polymers, 11(3), 471. https://doi.org/10.3390/polym11030471





