A Study of the Thermal Degradation and Combustion Characteristics of Some Materials Commonly Used in the Construction Sector
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Thermo-Gravimetric Analysis (TGA)
2.2.2. Pyrolysis Combustion Flow Calorimetry (PCFC)
3. Results and Discussion
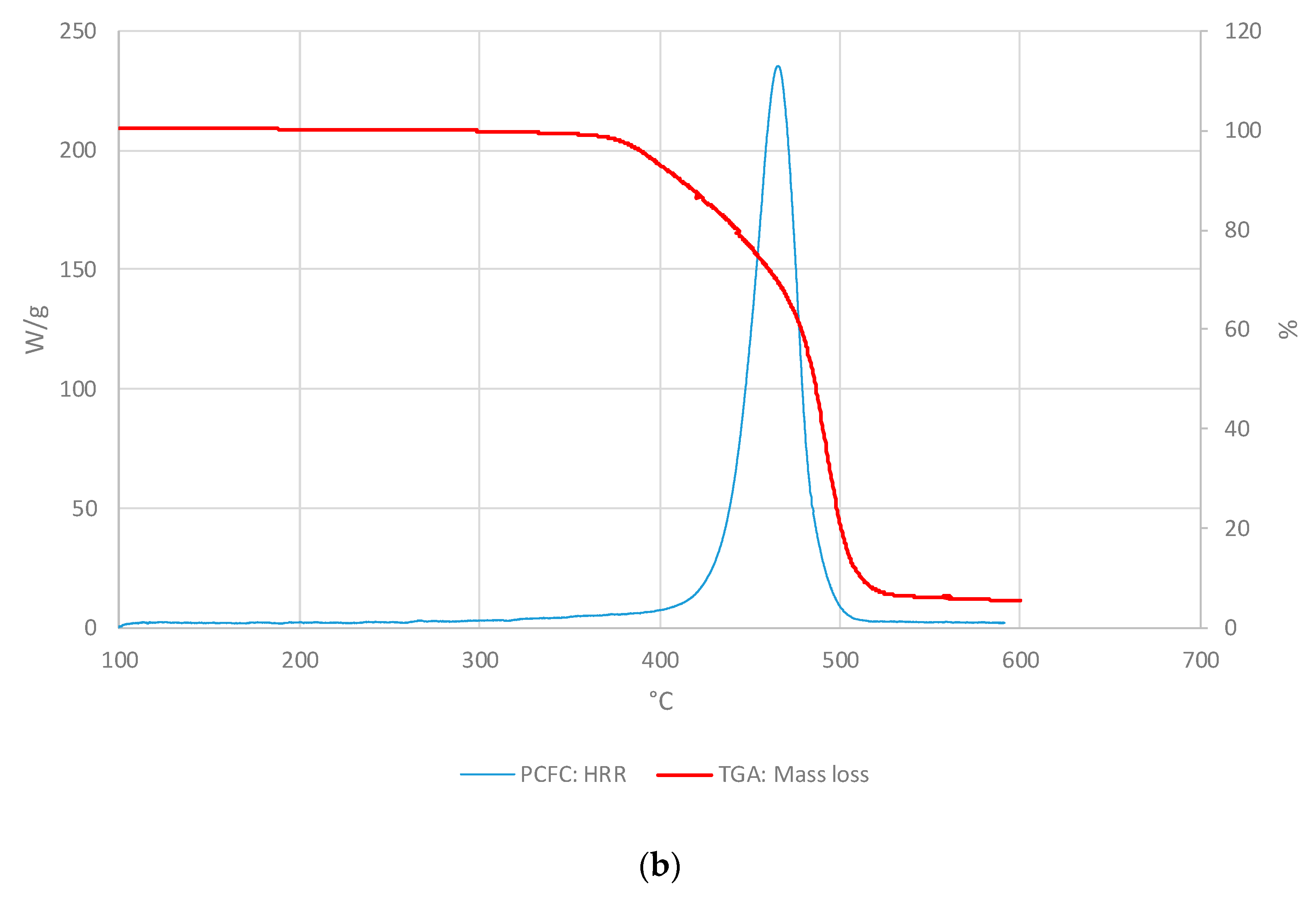
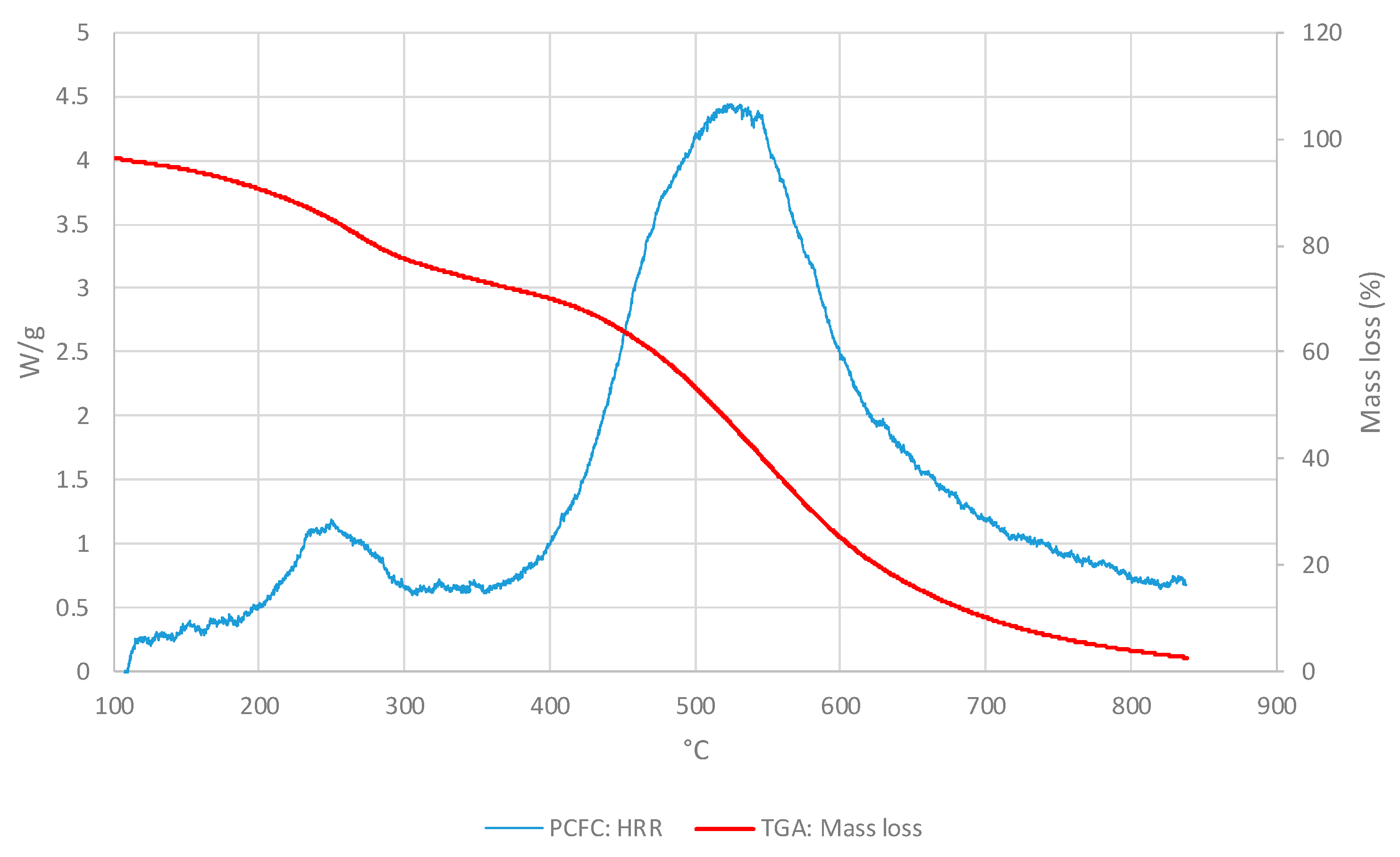
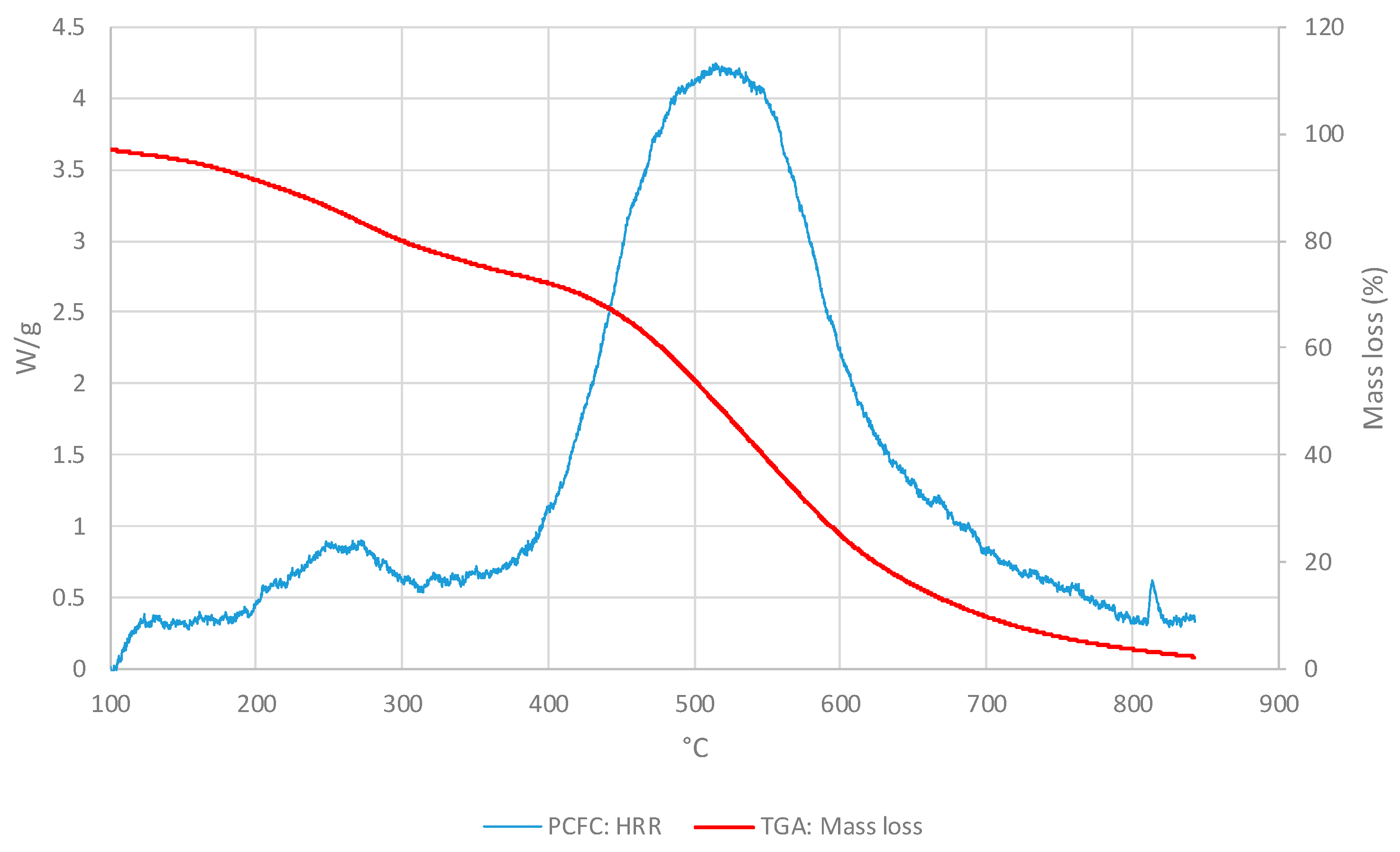
3.1. Polymeric core of the ACP
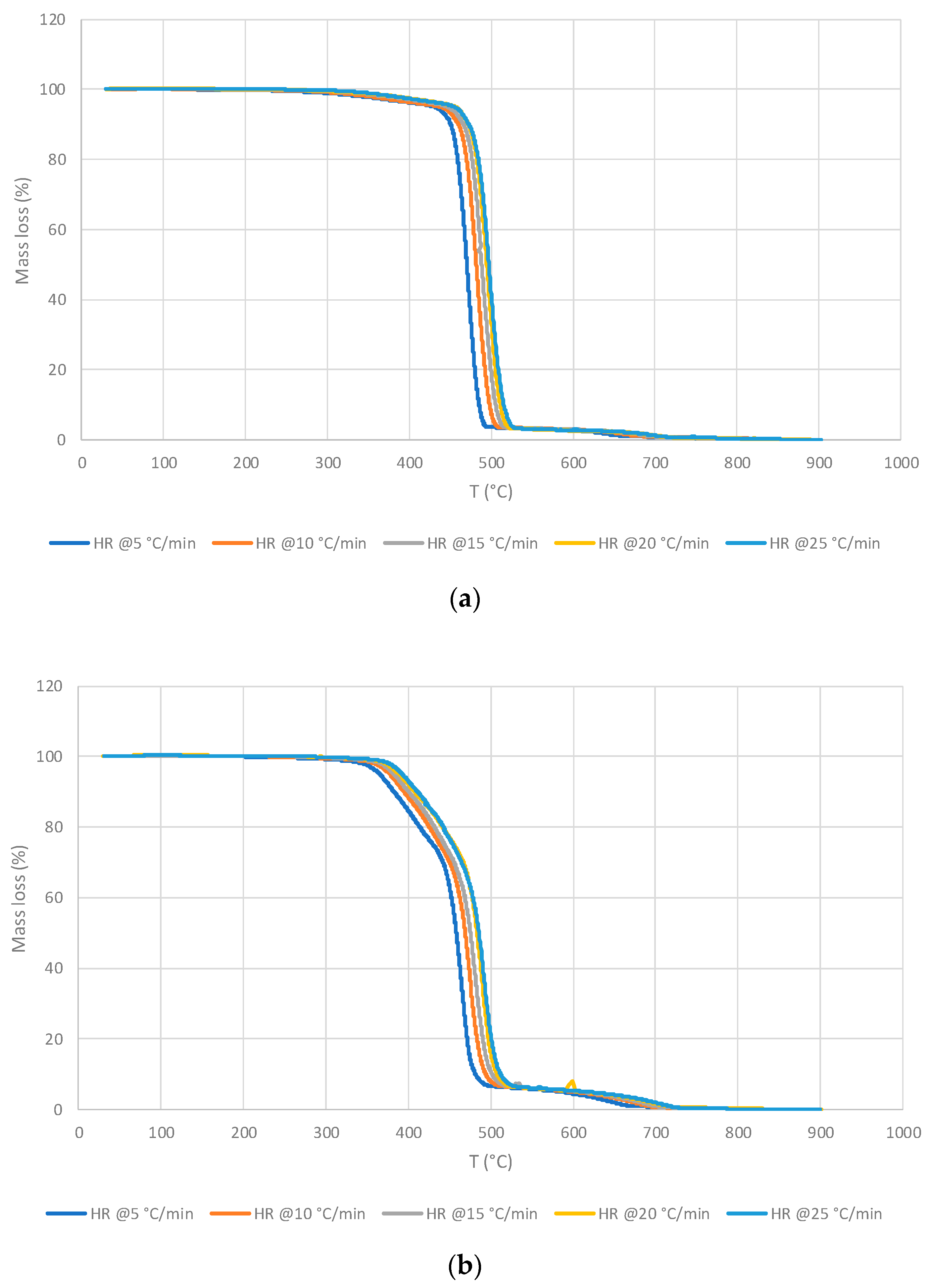
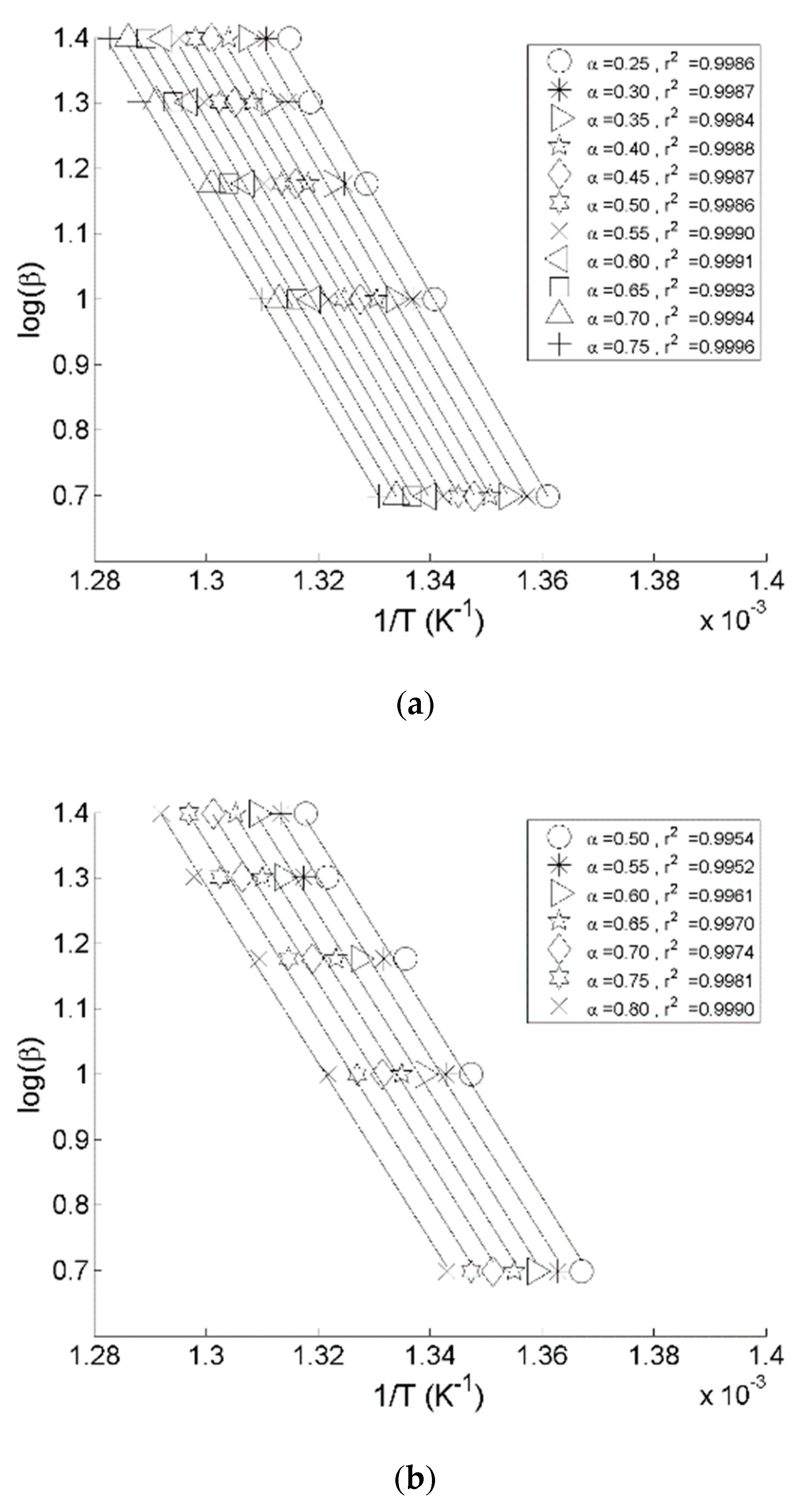
3.1.1. Thermo-gravimetric Analyses
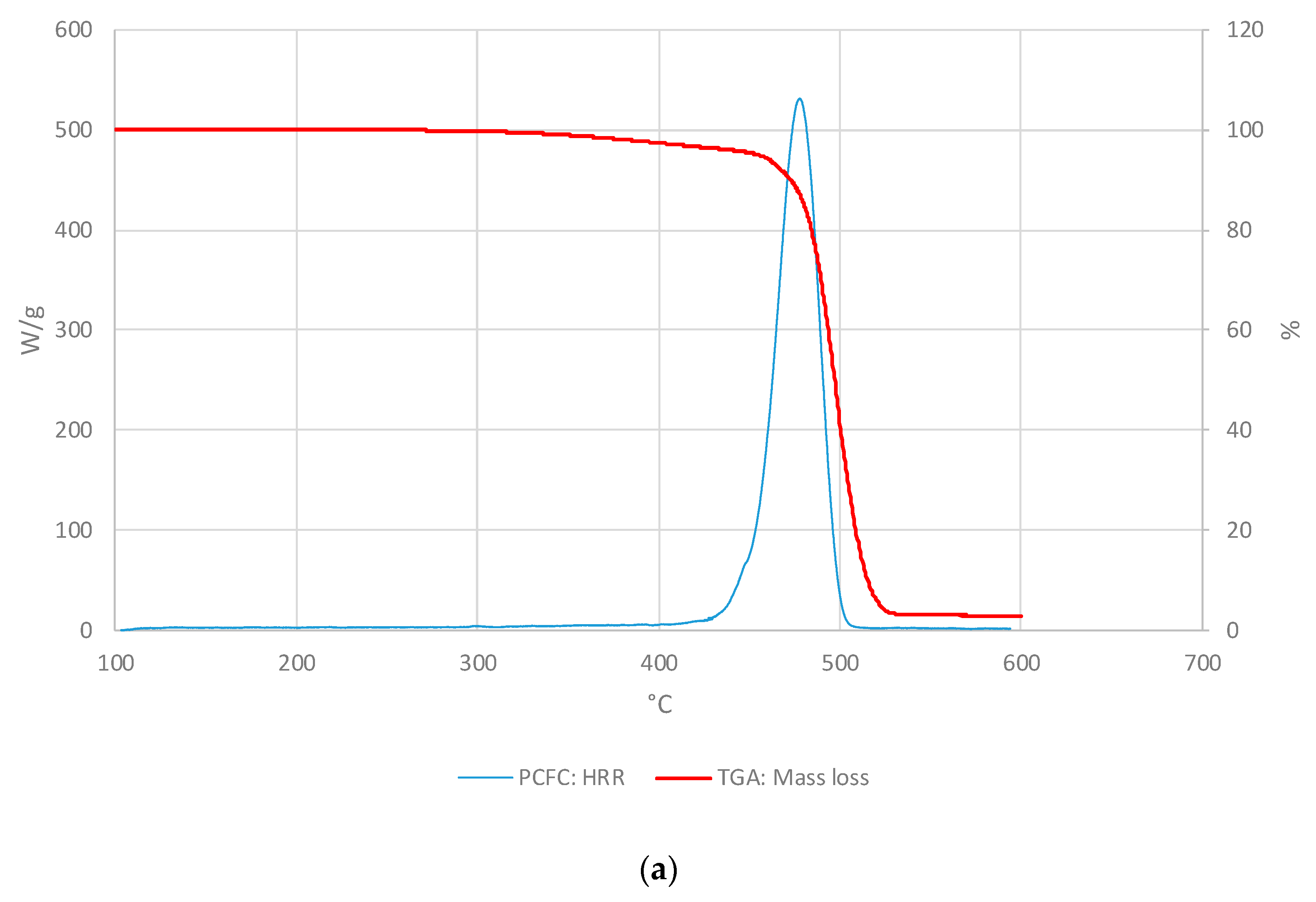
3.1.2. PCFC Studies
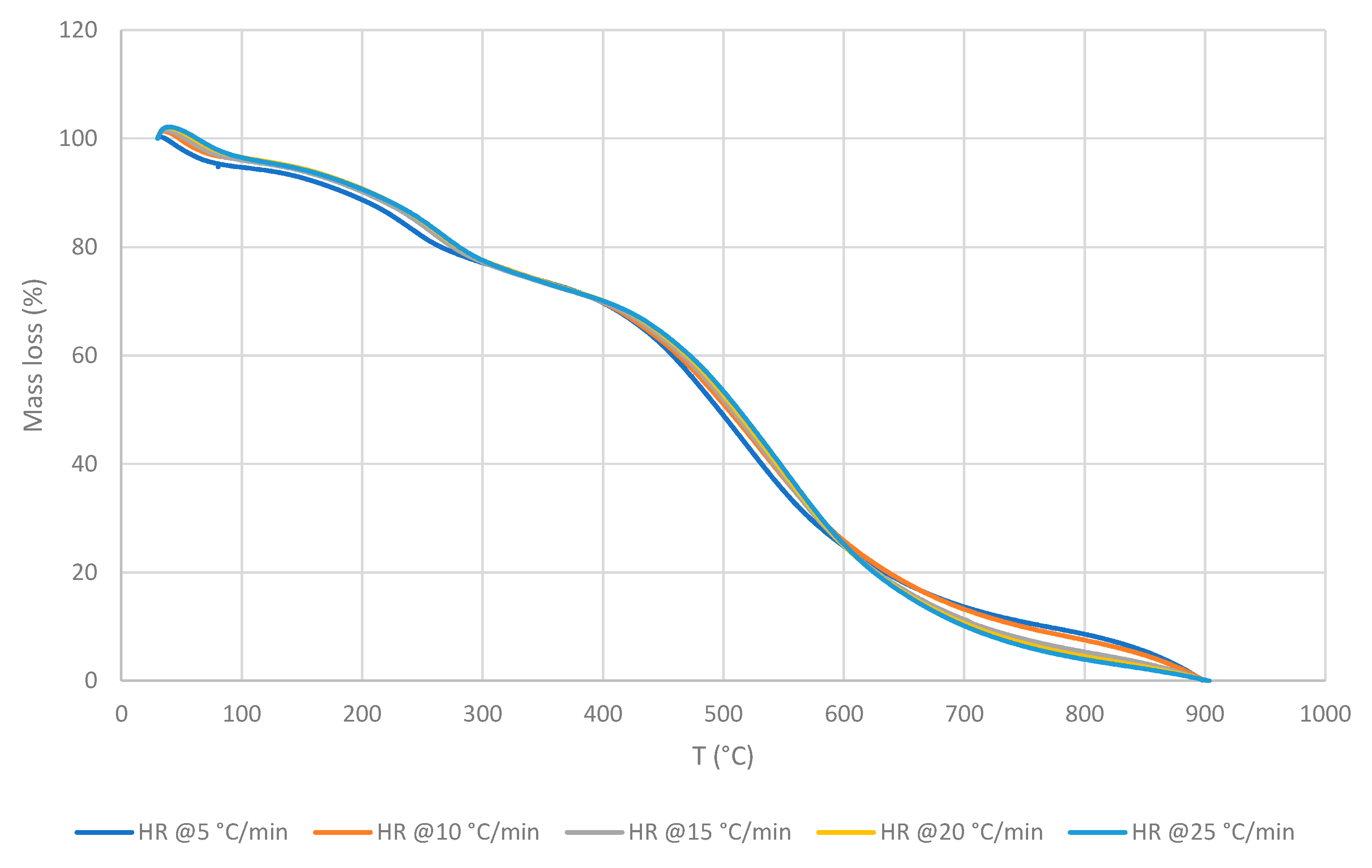
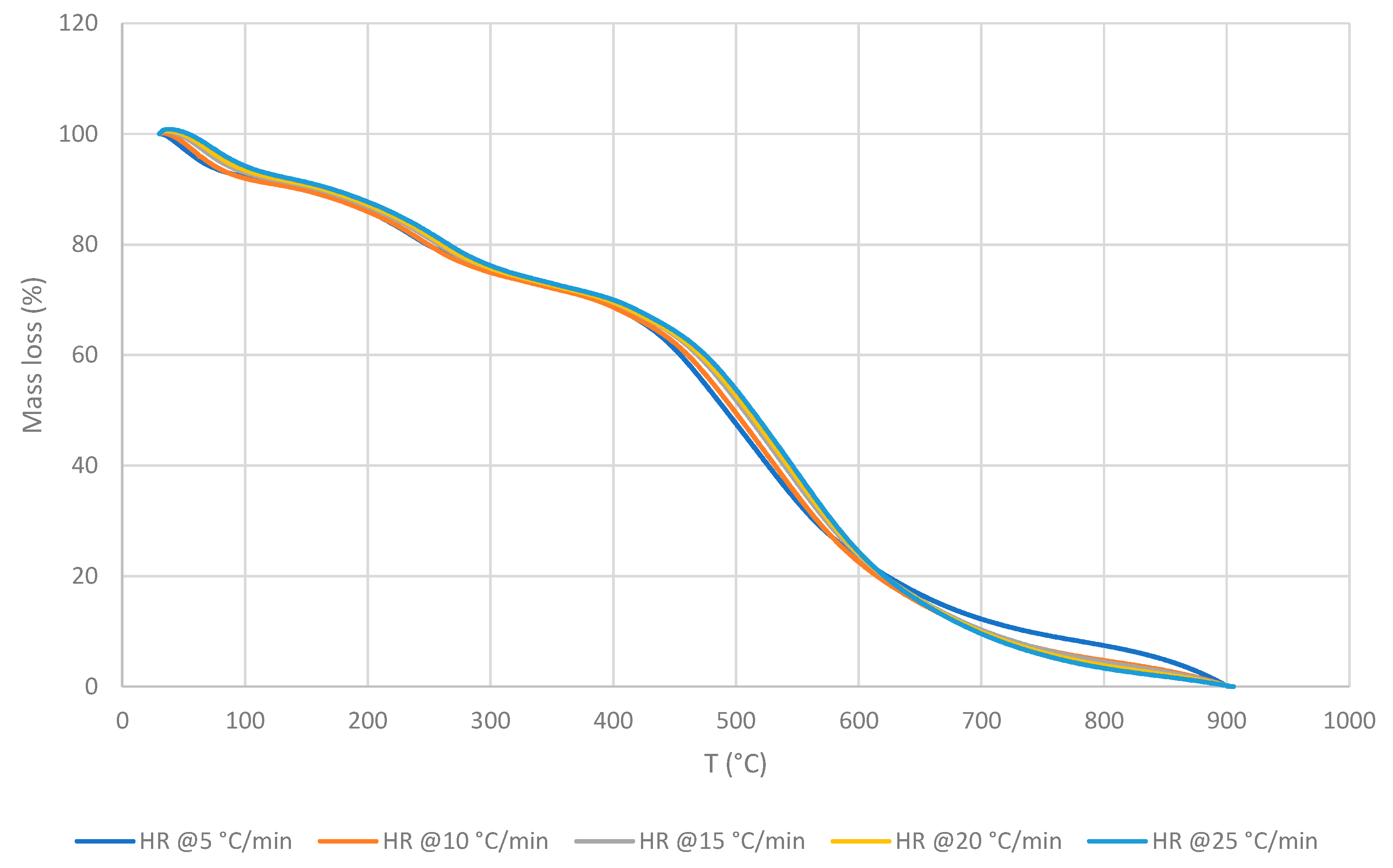
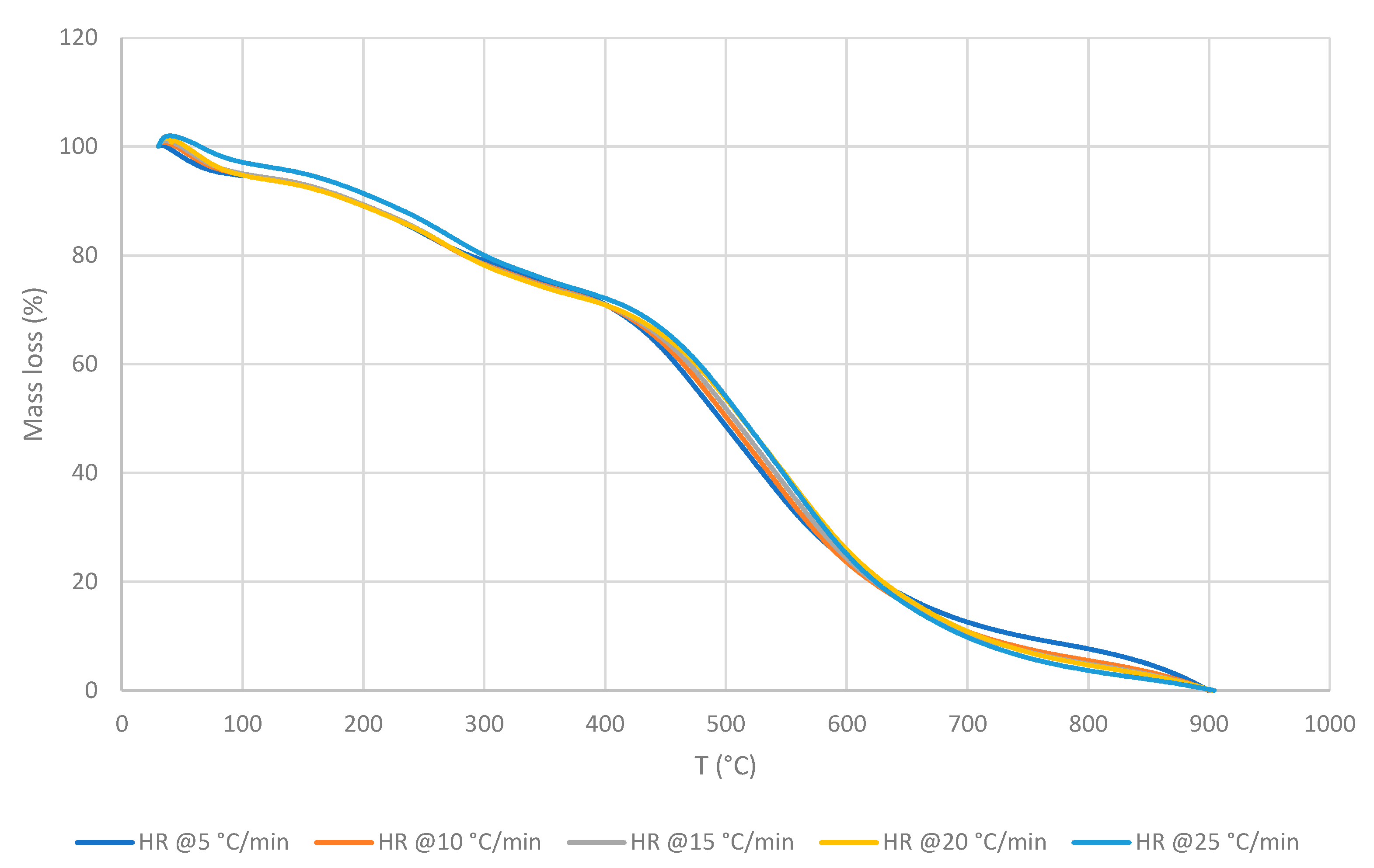
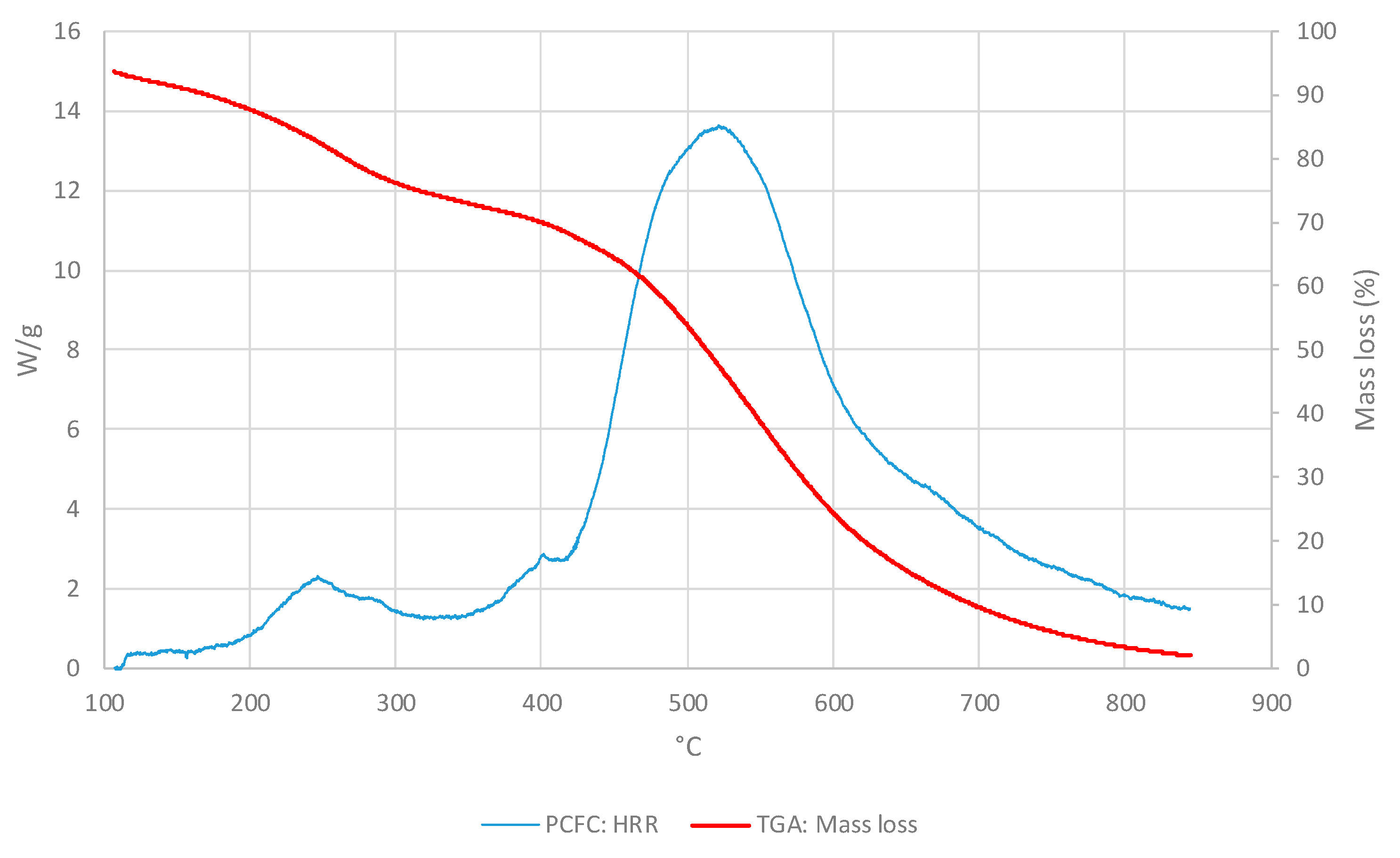
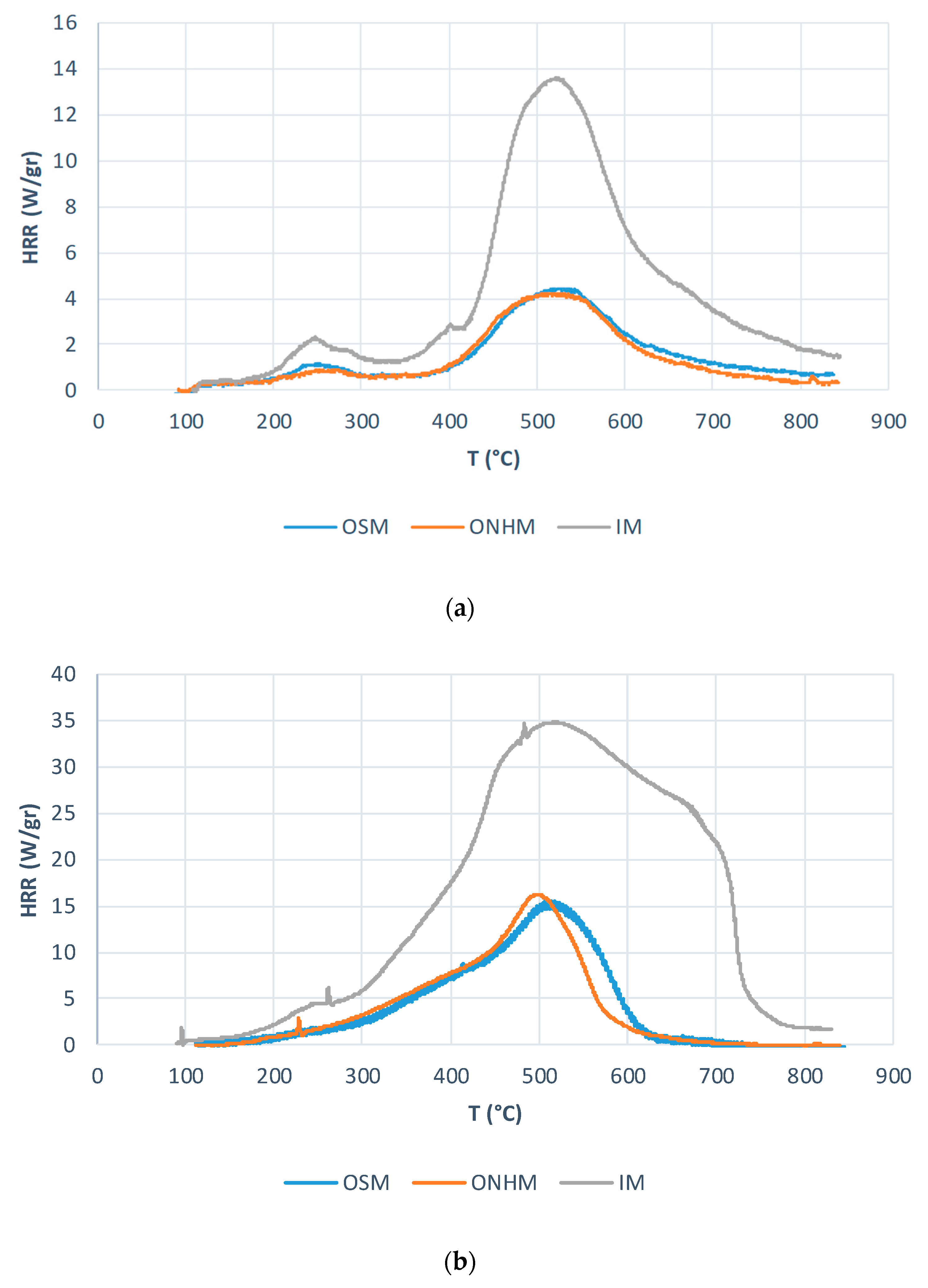
3.2. Cross-Laminated Timber
- Outer squared mesh (OSM)
- Inner material (IM)
- Outer non-homogenous mesh (ONHM)
3.2.1. Thermo-gravimetric Analyses
3.2.2. PCFC Studies
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McKenna, S.T.; Jones, N.; Peck, G.; Dickens, K.; Pawlelec, W.; Oradei, S.; Harris, S.; Stec, A.; Hull, T.R. Fire behaviour of modern façade materials—Understanding the Grenfell Tower fire. J. Hazard. Mater. 2019, 368, 115–123. [Google Scholar] [CrossRef] [PubMed]
- News Corp Australia Network. Thousands of Ticking Time-Bombs: Combustible Building Cladding is Everywhere in Australia. Available online: https://www.news.com.au/national/thousands-of-ticking-timebombs-combustible-building-cladding-is-everywhere-in-australia/news-story/db5f87122c317c64a064216cc5b97cc7 (accessed on 14 August 2019).
- Wadhwani, R.; Sutherland, D.; Moinuddin, K.A.M.; Joseph, P. Kinetics of pyrolysis of litter materials from pine and eucalyptus forests. J. Therm. Anal. Calorim. 2017, 130, 2035–2046. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Fire Tests —Full Scale Room Test for Surface Products; ISO 9705; International Organization for Standardization: Geneva, Switzerland, 1993. [Google Scholar]
- Babrauskas, V. The cone calorimeter. In SFPE Handbook of Fire Protection Engineering, 3rd ed.; DiNenno, P.J., Drysdale, D., Beyler, C.L., Walton, W.D., Custer, R.L.P., Hall, J.R., Jr., Watts, J.M., Jr., Eds.; Society of Fire Protection Engineers: Boston, MA, USA, 2002; Section 3; pp. 3.63–3.81. [Google Scholar]
- TA Instruments; Sauerbrunn, S.; Gill, P. Decomposition Kinetics Using TGA. Available online: http://www.tainstruments.com/pdf/literature/TA075.pdf (accessed on 6 August 2019).
- Lyon, R.E.; Walters, R.N. Pyrolysis combustion flow calorimetry. J. Anal. Appl. Pyrolysis 2004, 71, 27–46. [Google Scholar] [CrossRef]
- Joseph, P.; Tretsiakova-McNally, S.; Zhang, R. Techniques for assessing the combustion behavior of polymeric materials: Some current perspectives and future directions. Macromol. Symp. 2016, 362, 105–118. [Google Scholar] [CrossRef]
- Östman, B.A.; Nussbaum, R.M. Correlation between small-scale rate of heat release and full-scale room flashover for surface linings. Fire Saf. Sci. 1989, 2, 823–832. [Google Scholar] [CrossRef]
- Wickström, U.; Göransson, U. Prediction of heat release rates of surface materials in large-scale fire tests based on cone calorimeter results. J. Test. Eval. 1987, 15, 364–370. [Google Scholar] [CrossRef]
- Dowling, K.C.; Feske, E.F. Predicting the steinner tunnel test performance of rigid polyurethane foams using the cone calorimeter. In UTECH 94: Groundwork for Growth, Proceedings of the International Polyurethane Industry Conference, The Hague, The Netherlands, 22–24 March 1994; Reed, D., Lee, C., Eds.; Crain Communications Ltd.: London, UK, 1994; p. 55. [Google Scholar]
- Weil, E.D.; Patel, N.G.; Said, M.M.; Hirschler, M.M.; Shakir, S. Oxygen index: Correlations to other fire tests. Fire Mater. 1992, 16, 159–167. [Google Scholar] [CrossRef]
- Hirschler, M. Analysis of and potential correlations between fire tests for electrical cables, and how to use this information for fire hazard assessment. Fire Technol. 1997, 33, 291–315. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Komai, T. Correlation between the thermogravimetric analysis and some other flammability tests on covers of rubber conveyor belts. J. Fire Sci. 1990, 8, 455–476. [Google Scholar] [CrossRef]
- Yao, F.; Wu, Q.; Lei, Y.; Guo, W.; Xu, Y. Thermal decomposition kinetics of natural fibers: Activation energy with dynamic thermogravimetric analysis. Polym. Degrad. Stab. 2008, 93, 90–98. [Google Scholar] [CrossRef]
- Malek, J. The kinetic analysis of non-isothermal data. Thermochem. Acta 1992, 200, 257–269. [Google Scholar] [CrossRef]
- Lyon, R.E.; Walters, R.N.; Stoliarov, S.I.; Safronava, N. Principles and Practice of Microscale Combustion Calorimetry; Federal Aviation Administration: Springfield, VA, USA, 2013.
- ASTM International. Standard Test. Method for Determining Flammability Characteristics of Plastics and Other Solid Materials Using Microscale Combustion Calorimetry; ASTM D7309; ASTM International: West Conshohocken, PA, USA, 2013. [Google Scholar]
- Tretsiakova-McNally, S.; Joseph, P. Pyrolysis Combustion Flow Calorimetry studies on some reactively modified polymers. Polymers 2015, 7, 453–467. [Google Scholar] [CrossRef]
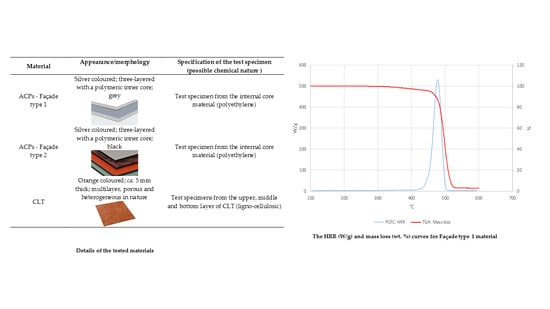
| Material | Appearance/Morphology | Specification of the Test Specimen (Possible Chemical Nature 1) |
|---|---|---|
| ACPs—Façade type 1 | Silver coloured; three-layered with a polymeric inner core; grey | Test specimen from the internal core material (polyethylene) |
| ACPs—Façade type 2 | Silver coloured; three-layered with a polymeric inner core; black | Test specimen from the internal core material (polyethylene) |
| CLT | Orange coloured; ca. 5 mm thick; multilayer, porous and heterogeneous in nature | Test specimens from the upper, middle and bottom layer of CLT (ligno-cellulosic) |
| Sample No. | Method A/Method B | |||||
|---|---|---|---|---|---|---|
| HRC (J∙g−1∙K−1) | pHRR (W∙g−1) | THR (kJ∙g−1) | Char yield (wt.%) | hc (kJ∙kg−1) | ||
| 1 | 1283.00/1886.00 | 528.90/780.90 | 37.60/34.20 | 478/454 | 9.33/4.00 | 41.47/35.63 |
| 2 | 1303.00/1835.00 | 544.00/686.60 | 37.80/35.10 | 479/455 | 8.00/6.67 | 41.09/37.61 |
| 3 | 1289.00/1298.00 | 520.20/612.40 | 36.20/35.10 | 478/451 | 6.67/5.33 | 38.79/37.08 |
| Average | 1291.67/1860.50 | 531.03/693.30 | 37.20/34.80 | 479/453 | 8.00/5.33 | 40.45/36.77 |
| Sample No. | Method A/Method B | |||||
|---|---|---|---|---|---|---|
| HRC (J∙g−1∙K−1) | pHRR (W∙g−1) | THR (kJ∙g−1) | Char yield (wt.%) | hc (kJ∙kg−1) | ||
| 1 | 554.00/1210.00 | 231.00/492.90 | 17.90/18.60 | 465/493 | 60.00/62.67 | 44.75/49.82 |
| 2 | 587.00/1080.00 | 242.90/426.60 | 18.00/19.20 | 471/427 | 60.00/61.33 | 45.00/49.66 |
| 3 | 588.00/1040.00 | 244.40/462.00 | 18.30/18.60 | 470/462 | 60.00/60.00 | 45.75/46.50 |
| Average | 576.33/1110.00 | 239.43/460.50 | 18.07/18.80 | 469/380 | 60.00/61.33 | 45.17/48.66 |
| Sample No. | Method A/Method B | |||||
|---|---|---|---|---|---|---|
| HRC (J∙g−1∙K−1) | pHRR (W∙g−1) | THR (kJ∙g−1) | Char yield (wt.%) | hc (kJ∙kg−1) | ||
| 1 | 9.00/31.00 | 3.36/11.71 | 1.90/6.80 | 484/469 | 87.71/72.71 | 15.4/24.92 |
| 2 | 10.00/39.00 | 3.46/12.66 | 1.80/7.30 | 486/476 | 87.50/72.08 | 14.40/26.15 |
| 3 | 10.00/38.00 | 3.98/12.21 | 1.80/7.40 | 497/477 | 86.88/71.25 | 13.71/35.74 |
| Average | 9.67/36.00 | 3.60/12.19 | 1.83/7.17 | 489/474 | 87.36/72.01 | 14.52/25.60 |
| Sample No. | Method A/Method B | |||||
|---|---|---|---|---|---|---|
| HRC (J∙g−1∙K−1) | pHRR (W∙g−1) | THR (kJ∙g−1) | Char yield (wt.%) | hc (kJ∙kg−1) | ||
| 1 | 30.00/80.00 | 10.57/29.83 | 5.60/24.07 | 478/459 | 64.50/10.25 | 15.77/27.52 |
| 2 | 30.00/81.00 | 10.15/30.08 | 5.60/24.10 | 479/457 | 64.75/9.75 | 15.89/26.70 |
| 3 | 25.00/79.00 | 9.18/29.04 | 4.50/23.40 | 490/453 | 64.75/10.00 | 12.77/26.00 |
| Average | 28.33/80.00 | 9.97/29.65 | 5.23/24.07 | 482/457 | 64.67/10.00 | 14.81/26.74 |
| Sample No. | Method A/Method B | |||||
|---|---|---|---|---|---|---|
| HRC (J∙g−1∙K−1) | pHRR (W∙g−1) | THR (kJ∙g−1) | Char yield (wt.%) | hc (kJ∙kg−1) | ||
| 1 | 9.00/3.80 | 3.19/12.32 | 1.80/6.00 | 468/468 | 87.92/74.79 | 14.90/23.80 |
| 2 | 9.00/3.60 | 3.18/11.78 | 1.80/6.30 | 471/463 | 87.50/73.13 | 14.40/23.44 |
| 3 | 9.00/3.70 | 3.40/12.25 | 2.00/6.20 | 467/465 | 87.50/71.88 | 16.00/22.04 |
| Average | 9.00/3.70 | 3.26/12.12 | 1.87/6.17 | 469/466 | 87.64/73.26 | 15.10/23.10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solorzano, J.A.P.; Moinuddin, K.A.M.; Tretsiakova-McNally, S.; Joseph, P. A Study of the Thermal Degradation and Combustion Characteristics of Some Materials Commonly Used in the Construction Sector. Polymers 2019, 11, 1833. https://doi.org/10.3390/polym11111833
Solorzano JAP, Moinuddin KAM, Tretsiakova-McNally S, Joseph P. A Study of the Thermal Degradation and Combustion Characteristics of Some Materials Commonly Used in the Construction Sector. Polymers. 2019; 11(11):1833. https://doi.org/10.3390/polym11111833
Chicago/Turabian StyleSolorzano, Javier Arturo Piedrahita, Khalid Abu Mohammad Moinuddin, Svetlana Tretsiakova-McNally, and Paul Joseph. 2019. "A Study of the Thermal Degradation and Combustion Characteristics of Some Materials Commonly Used in the Construction Sector" Polymers 11, no. 11: 1833. https://doi.org/10.3390/polym11111833
APA StyleSolorzano, J. A. P., Moinuddin, K. A. M., Tretsiakova-McNally, S., & Joseph, P. (2019). A Study of the Thermal Degradation and Combustion Characteristics of Some Materials Commonly Used in the Construction Sector. Polymers, 11(11), 1833. https://doi.org/10.3390/polym11111833