Ammonium Lignosulfonate Adhesives for Particleboards with pMDI and Furfuryl Alcohol as Crosslinkers
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Adhesive Formulations
2.2. Differential Scanning Calorimetry (DSC)
2.3. Shear Bond Strength
2.4. Fourier Transform Infrared (FTIR) Spectroscopy
2.5. Adhesive Penetration
2.6. Particleboard Manufacturing and Testing
2.7. Formaldehyde Emission Testing
2.8. Statistical Analysis
3. Results
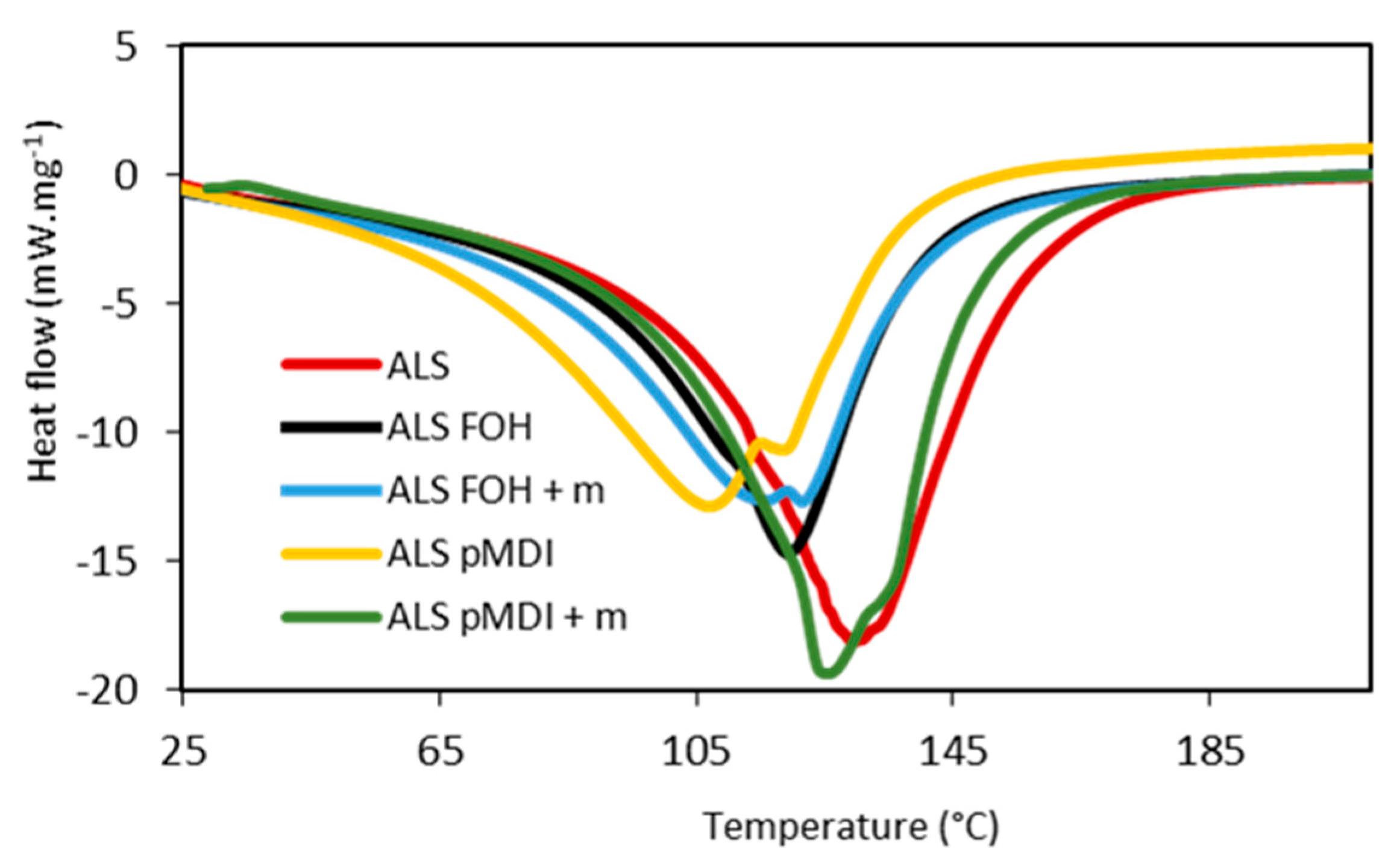
3.1. Curing Behavior of the Adhesives
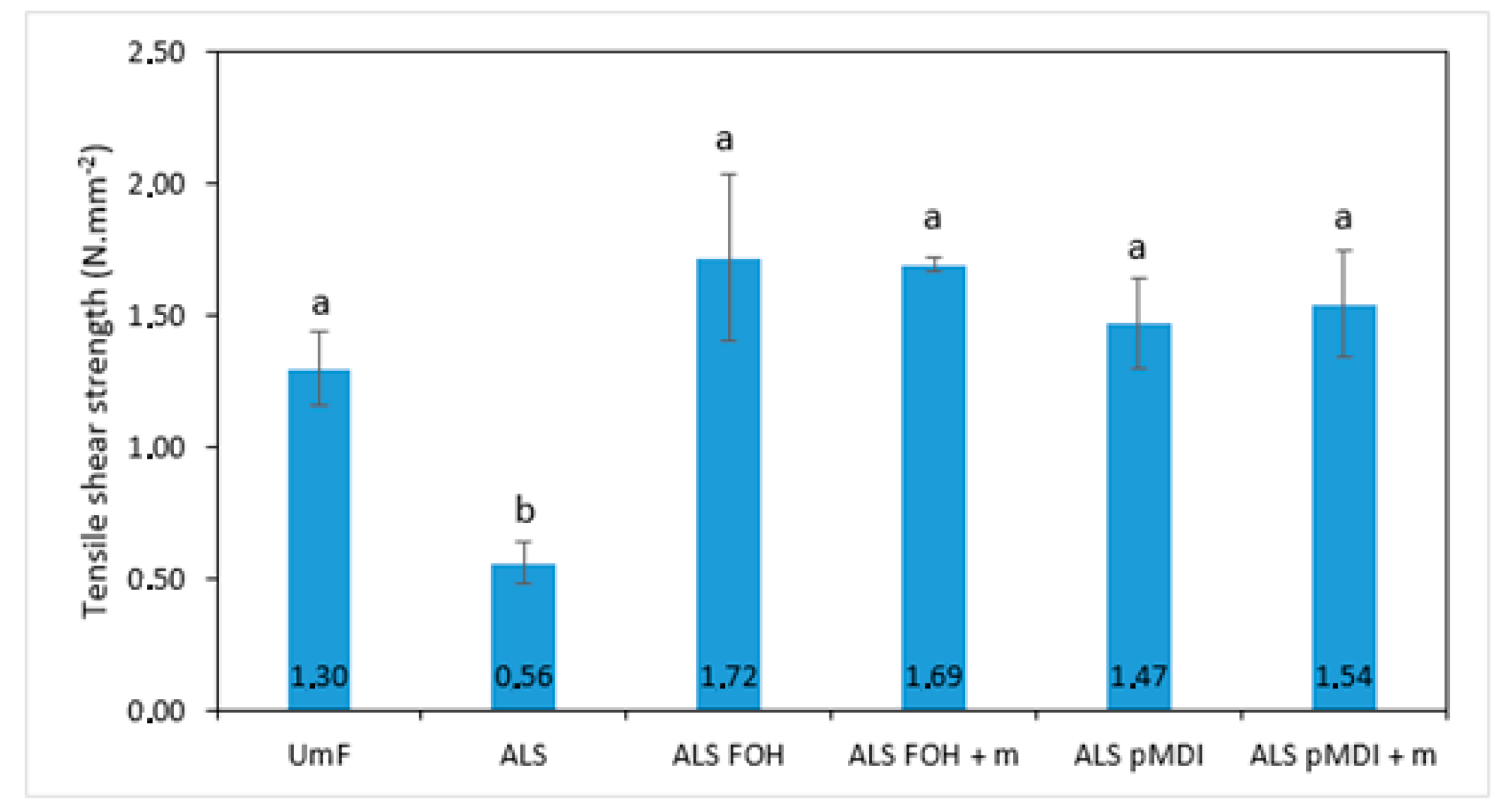
3.2. Tensile Shear Strength and Adhesive Penetration
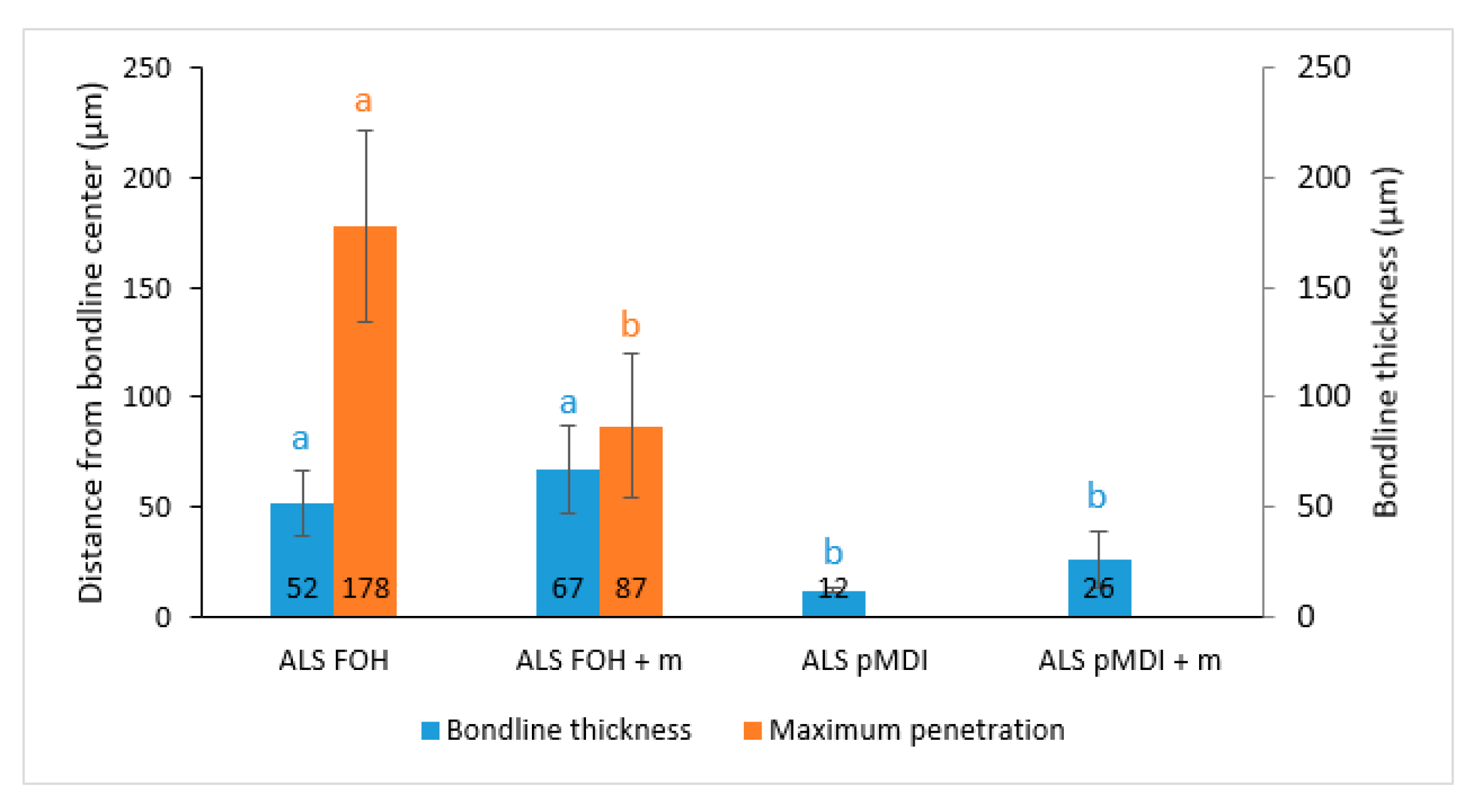
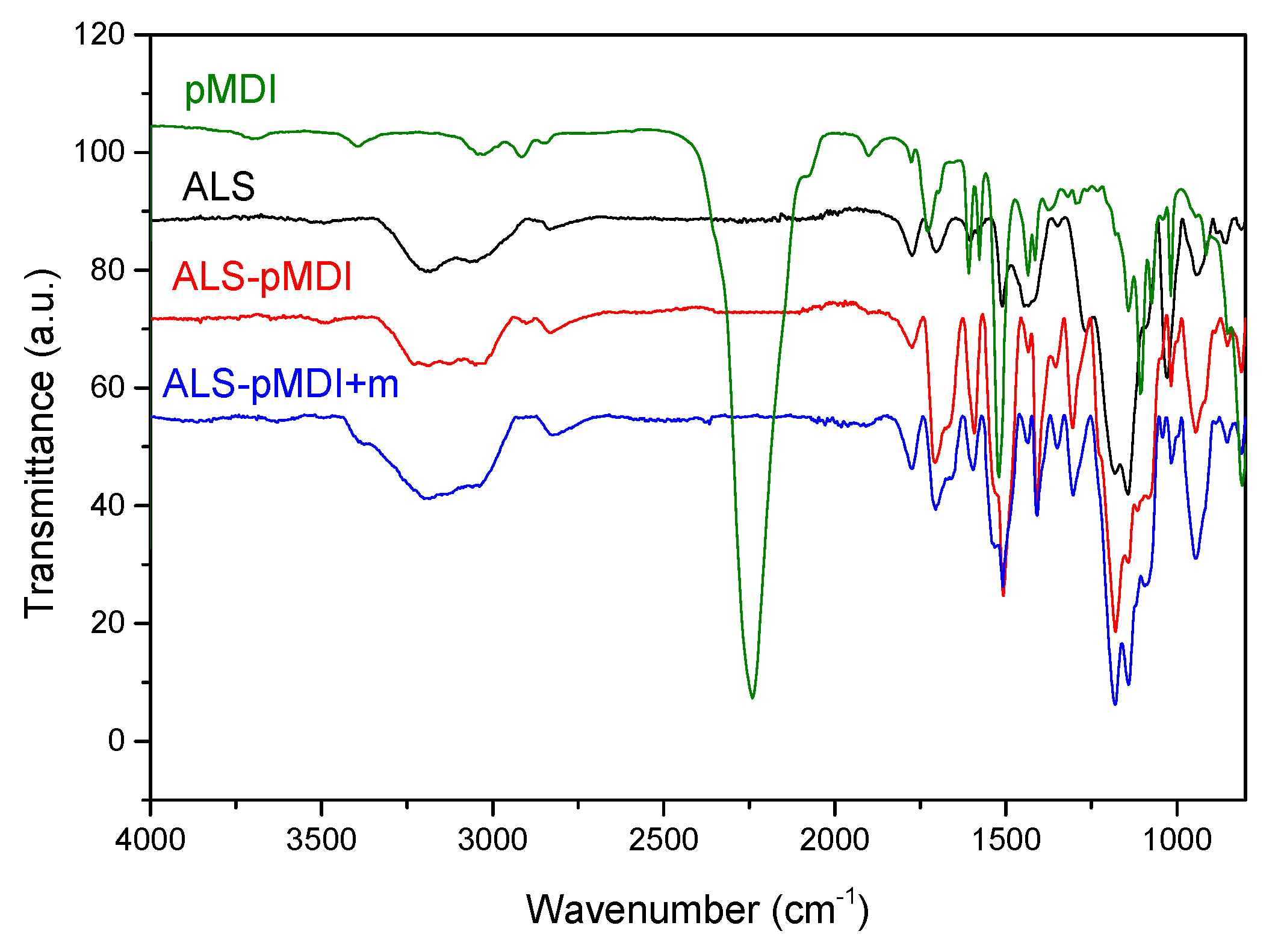
3.3. Chemical Characterization of Adhesive Bondline
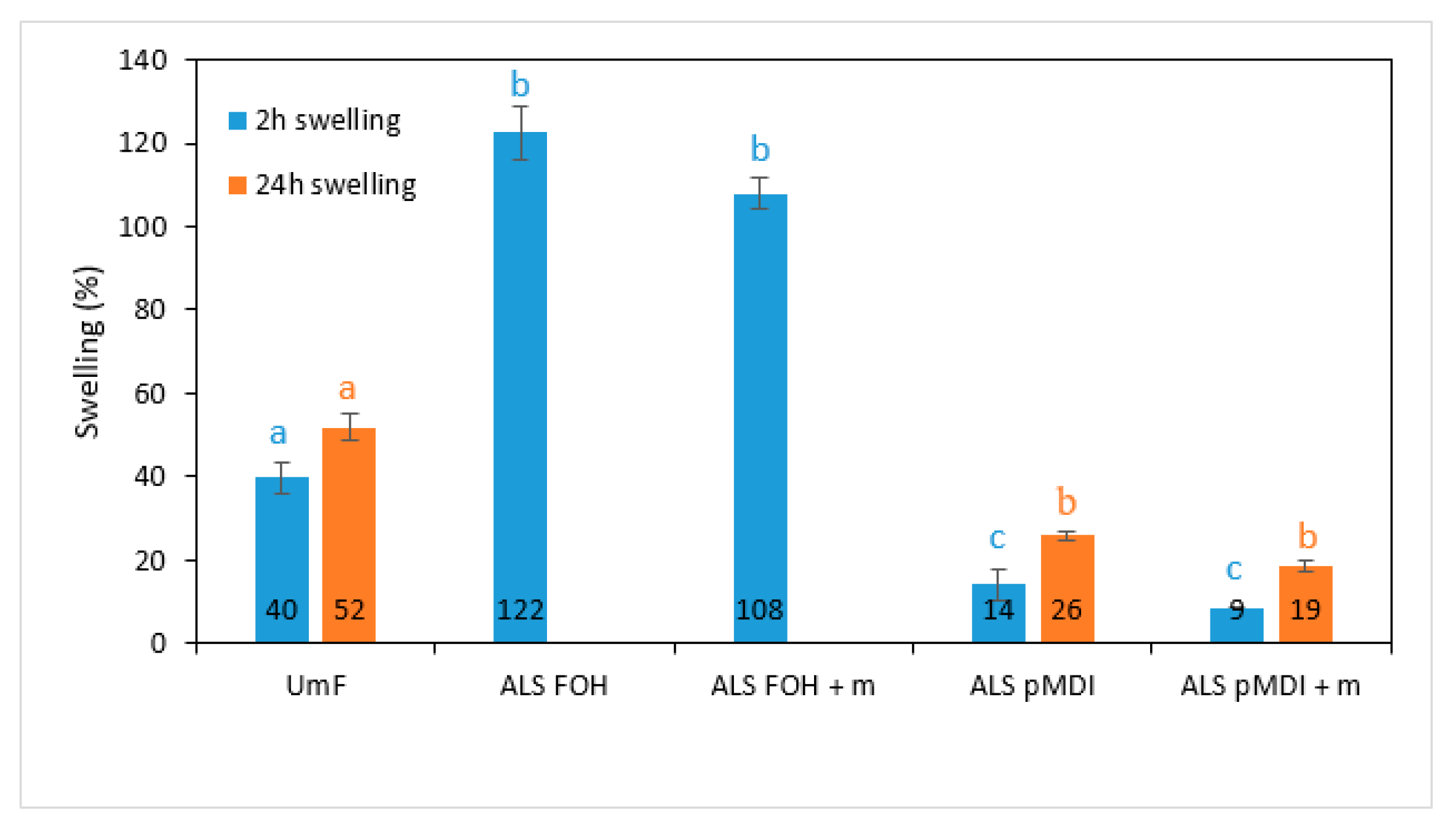
3.4. Mechanical and Physical Properties of Particleboards
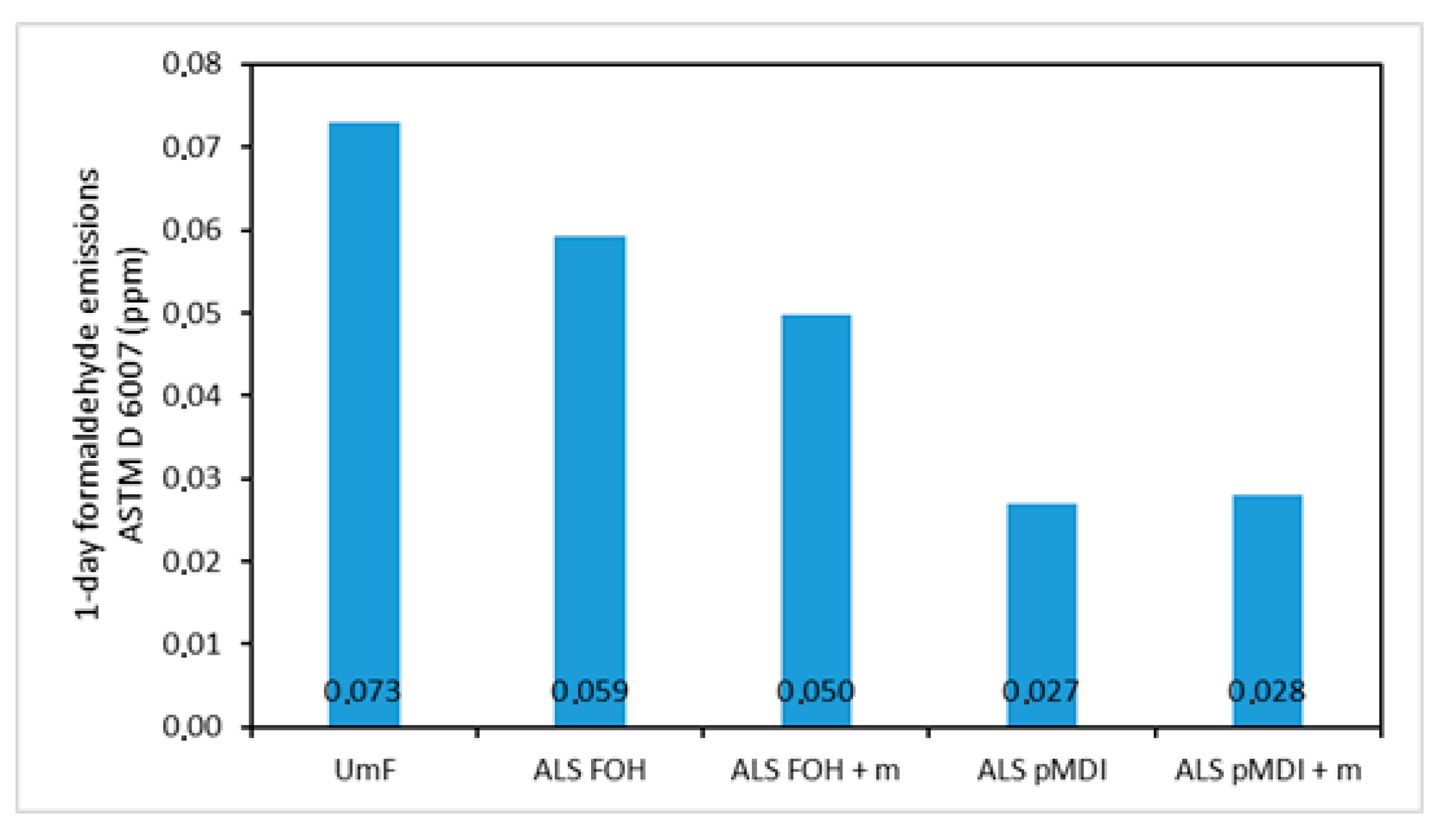
3.5. Formaldehyde Emissions
4. Conclusions
- All samples with crosslinkers had lower curing temperature and curing heat than the un-crosslinked ALS sample. The ALS-pMDI sample cured at the lowest temperature (107 °C) and had the lowest curing heat (123 J/g).
- For those formulations crosslinked with pMDI, FTIR results pointed out for chemical reaction of ALS with pMDI in the cured adhesives, i.e., the formation of urethane linkages.
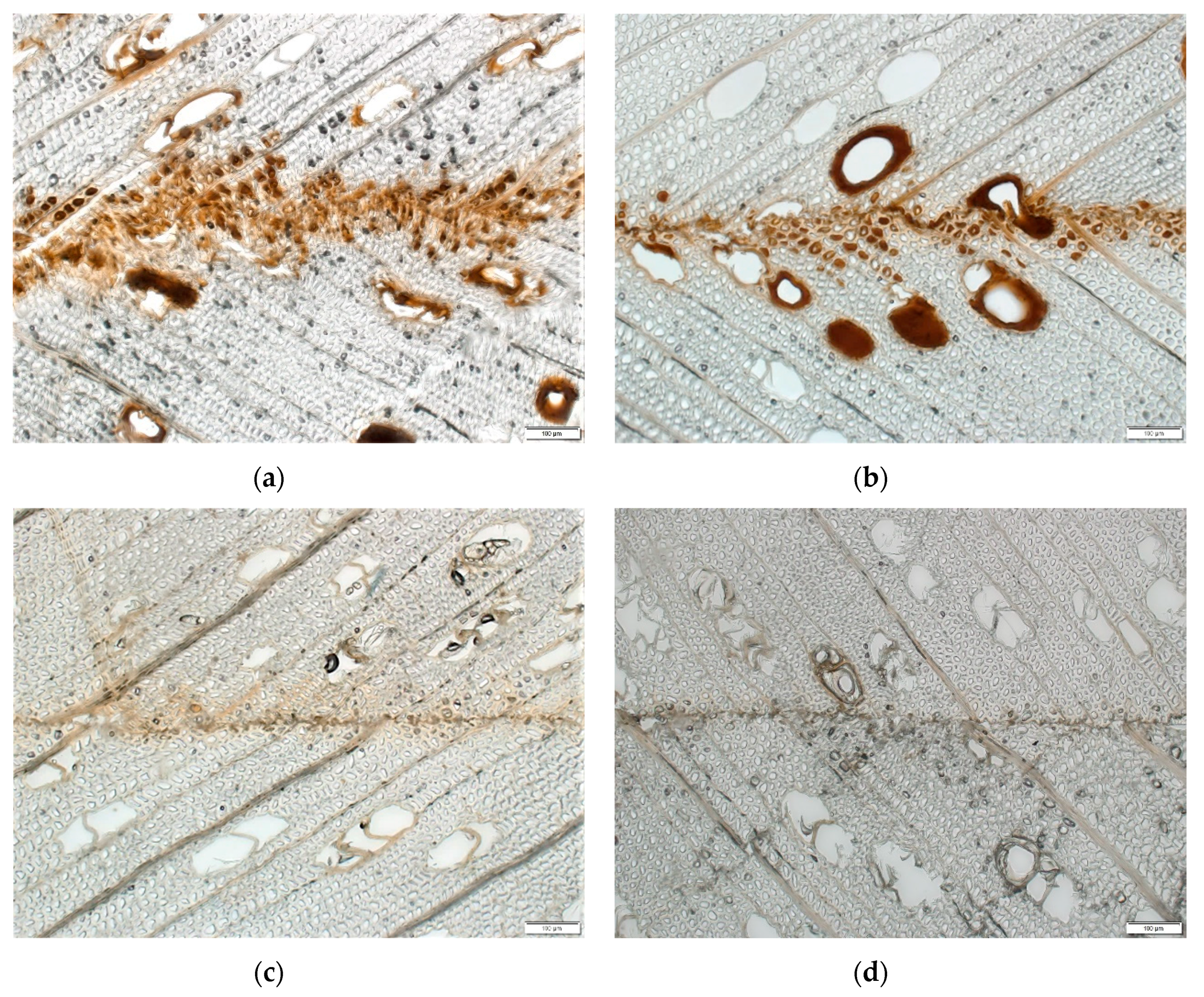
- The FOH crosslinked ALS samples had thick dark bondlines, while the pMDI cured ALS samples had thin light bondlines, possibly due to the higher molecular weight of pMDI and vapor pressure created by the isocyanate crosslinking.
- The penetration of the FOH crosslinked ALS occurred mainly through vessels with some filling of the fiber lumens. The FOH crosslinked ALS without tannin had deeper penetration than the one with tannin, suggesting that tannin prohibits penetration, allowing more glue to remain near the bondline.
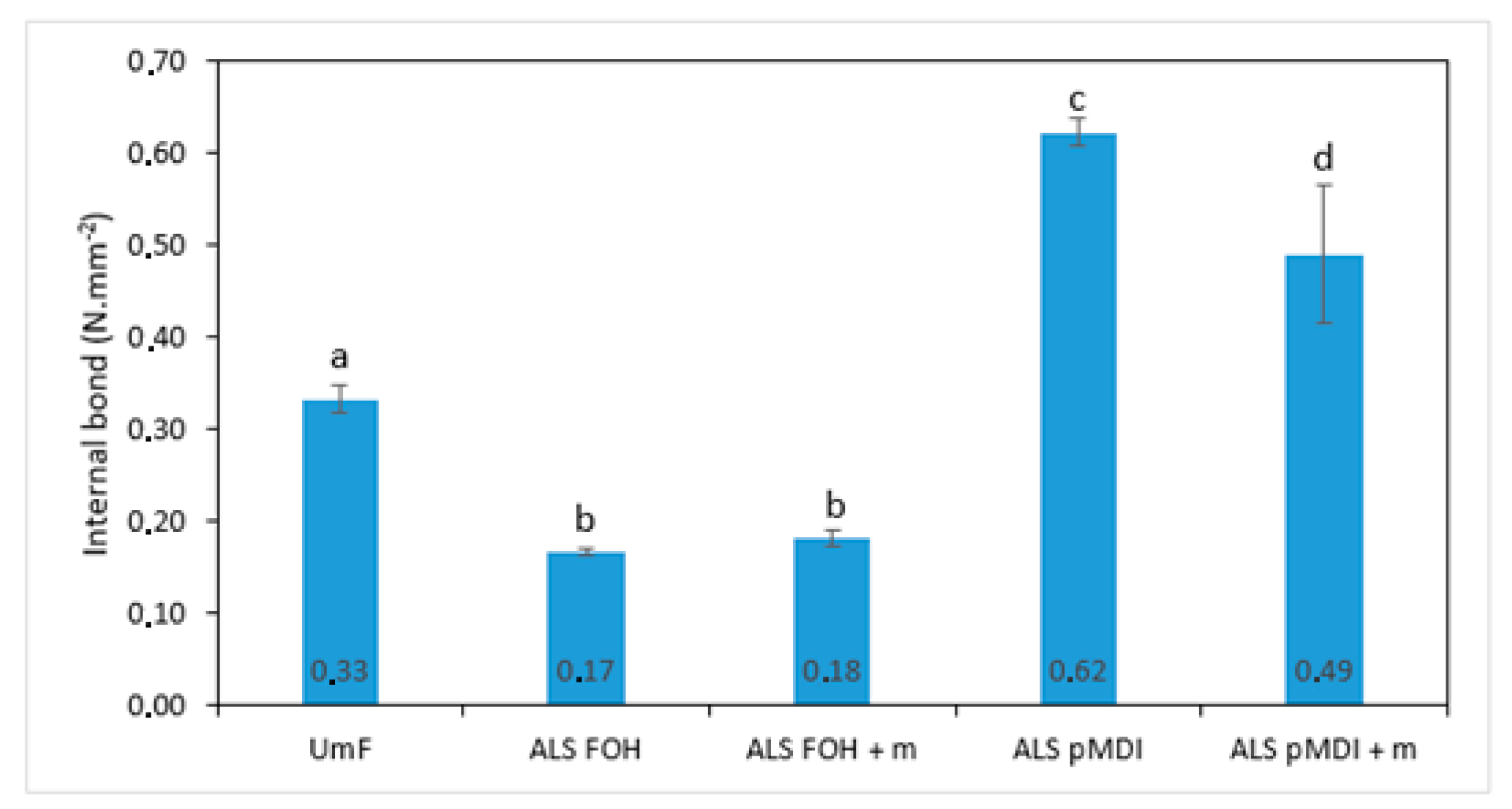
- The mechanical properties of the particleboards produced using FOH crosslinked ALS, with and without tannin (IB: 0.17 N/mm2 and 0.18 N/mm2, respectively), were inferior to those produced using pMDI crosslinked ALS, with and without tannin (IB: 0.62 N/mm2 and 0.49 N/mm2, respectively). A similar trend could be seen in thickness swelling where the FOH crosslinked ALS samples disintegrated after 2 h. The contribution of ALS to the final strength of the ALS pMDI samples needs to be further evaluated.
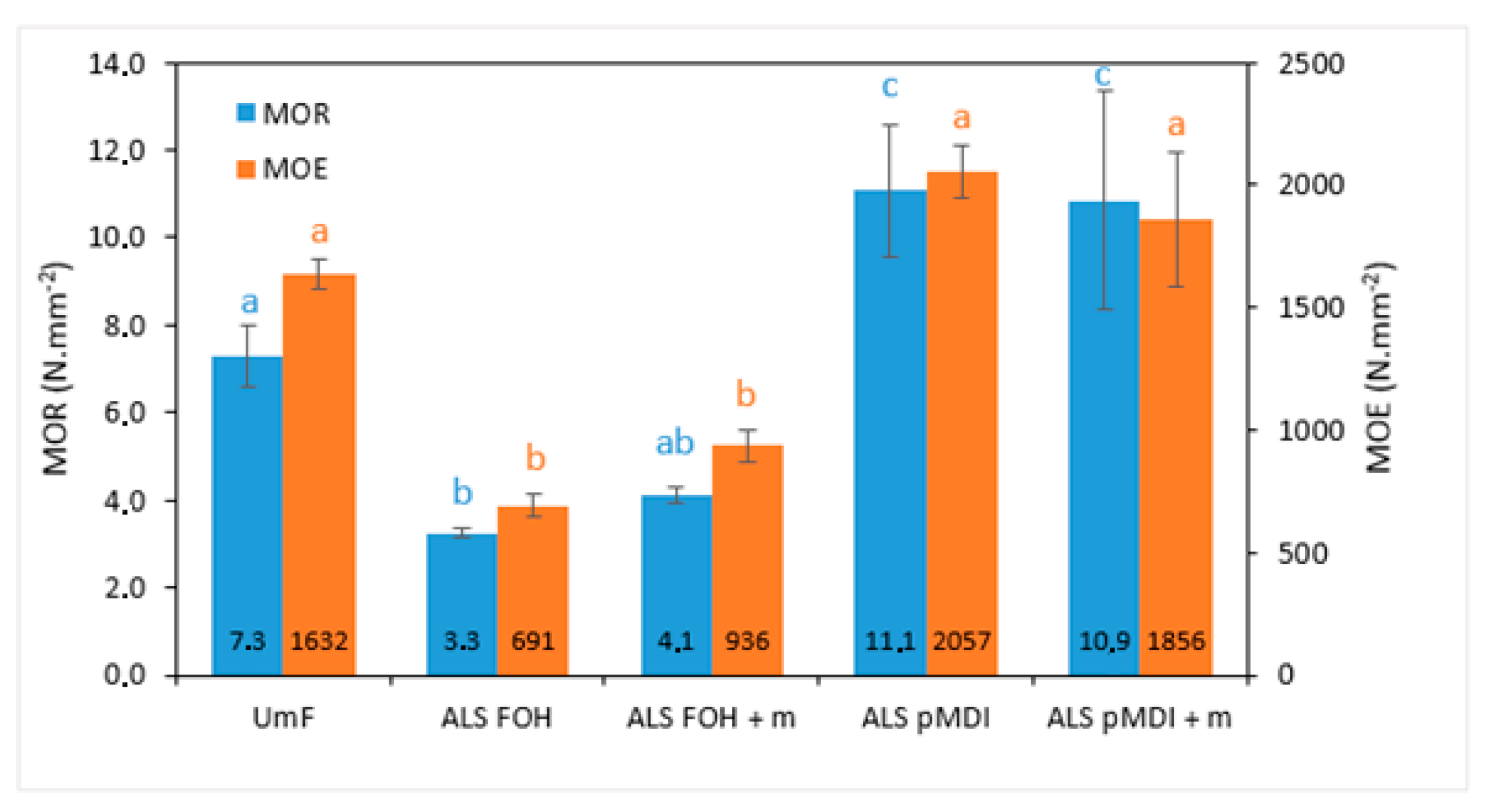
- Although particleboard properties were worse for the FOH crosslinked samples, the tensile shear strengths of both FOH and pMDI crosslinked ALS were at the same level as the UmF reference for the 2-layered veneer samples. The poor performance of FOH crosslinked ALS samples in particleboards could be due to the shorter pre-polymerization time and low FOH and total glue amount.
- The addition of mimosa tannin (10% to ALS amount) had no effect on any of the mechanical properties for both particleboard and 2-layered veneer samples. However, it lowered the emissions of FOH crosslinked ALS samples from 0.059 ppm of ALS FOH to 0.050 ppm of ALS FOH + m.
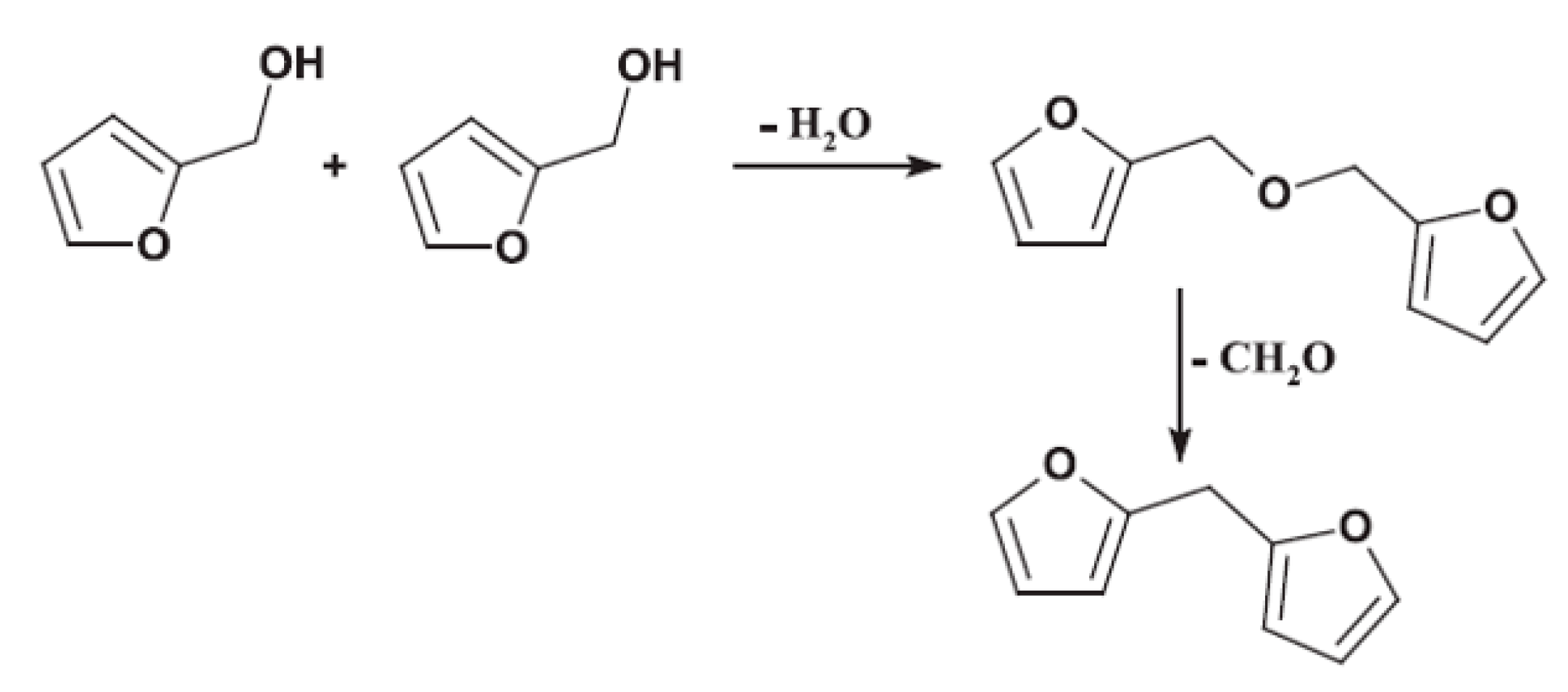
- The formaldehyde emissions of pMDI crosslinked particleboards were at the level of natural wood (0.027 ppm and 0.028 ppm). Interestingly, the FOH crosslinked boards emitted beyond the level of natural wood (0.059 ppm and 0.050 ppm). This can be due to one of the polycondensation mechanisms of FOH, where two methylol groups of two FOH molecules react, leading to ether bridge that can transform into methylene bridge by the release of formaldehyde.
Author Contributions
Funding
Conflicts of Interest
References
- International Agency for Research on Cancer (IARC). IARC Classifies Formaldehyde as Carcinogenic to Humans; International Agency for Research on Cancer (IARC): Lyon, Franc, 2004; Press Release June 15, No. 153. [Google Scholar]
- Bundesministerium für Umwelt Naturschutz und nukleare Sicherheit, Bekanntmachung analytischer Verfahren für Probenahmen und Untersuching für die in Anlage 1 der Chemikalien-Verbotsverordnung genannten Stoffe und Stoffgruppen. 2018, Bundesanzeiger, BAnz AT 26.11.2018 B2. Available online: https://www.umwelt-online.de/recht/gefstoff/chemverb.vo/chvvanalyt18.htm (accessed on 9 October 2019).
- Aydin, I.; Demirkir, C.; Colak, S.; Colakoglu, G. Utilization of bark flours as additive in plywood manufacturing. Eur. J. Wood Wood Prod. 2017, 75, 63–69. [Google Scholar] [CrossRef]
- Réh, R.; Igaz, R.; Krišťák, Ľ.; Ružiak, I.; Gajtanska, M.; Božíková, M.; Kučerka, M. Functionality of beech bark in adhesive mixtures used in plywood and Its effect on the stability associated with material systems. Materials 2019, 12, 1298. [Google Scholar]
- Gu, K.; Li, K. Preparation and evealuation of particleboard with a soy flour-polyethylenimine-maleic anhydride adhesive. J. Am. Oil Chem. Soc. 2011, 88, 673–679. [Google Scholar] [CrossRef]
- Da Silva, C.G.; Grelier, S.; Pichavant, F.; Frollini, E.; Castellan, A. Adding value to lignins isolated from sugarcane bagasse and Miscanthus. Ind. Crops Prod. 2013, 42, 87–95. [Google Scholar] [CrossRef]
- El Mansouri, N.E.; Pizzi, A.; Salvadó, J. Lignin-based wood panel adhesives without formaldehyde. Holz Roh- Werkst. (1937–2008) 2006, 65, 65. [Google Scholar] [CrossRef]
- Tan, H.; Zhang, Y.; Weng, X. Preparation of the Plywood Using Starch-based Adhesives Modified with blocked isocyanates. Procedia Eng. 2011, 15, 1171–1175. [Google Scholar] [CrossRef]
- Hemmilä, V.; Adamopoulos, S.; Karlsson, O.; Kumar, A. Development of sustainable bio-adhesives for engineered wood panels: A Review. RSC Adv. 2017, 7, 38604–38630. [Google Scholar] [CrossRef]
- Frihart, C.; Birkeland, M. Soy Properties and Soy Wood Adhesives; American Chemical Society: Midland, MI, USA, 2014; pp. 167–192. [Google Scholar]
- Pizzi, A. Recent developments in eco-efficient bio-based adhesives for wood bonding: Opportunities and issues. J. Adhes. Sci. Technol. 2006, 20, 829–846. [Google Scholar] [CrossRef]
- Norström, E.; Demircan, D.; Fogelström, L.; Khabbaz, F.; Malmström, E. Chapter 4: Green Binders for Wood Adhesives. In Applied Adhesive Bonding Science and Technology; Ozer, H., Ed.; InTech Open: Rijeka, Croatia, 2018. [Google Scholar]
- Ferdosian, F.; Pan, Z.; Gao, G.; Zhao, B. Bio-based adhesives and evaluation for wood composites application. Polymers 2017, 9, 70. [Google Scholar] [CrossRef]
- Costello, H. Lignin Products Global Market Size, Sales Data 2017–2022 & Applications in Animal Feed Industry 2017: (Market Analysis No. 188450); Orbis Research: Dallas, TX, USA, 2017. [Google Scholar]
- Farag, S.; Kouisni, L.; Chaouki, J. Lumped approach in kinetic modeling of microwave pyrolysis of Kraft lignin. Energy Fuels 2014, 28, 1406–1417. [Google Scholar] [CrossRef]
- Fatehi, P.; Chen, J. Extraction of Technical Lignins from Pulping Spent Liquors, Challenges and Opportunities. In Production of Biofuels and Chemicals from Lignin; Fang, Z., Smith, J.R.L., Eds.; Springer: Singapore, 2016; pp. 35–54. [Google Scholar]
- Ragauskas, A.J.; Beckham, G.T.; Biddy, M.J.; Chandra, R.; Chen, F.; Davis, M.F.; Davison, B.H.; Dixon, R.A.; Gilna, P.; Keller, M.; et al. Lignin valorization: Improving lignin processing in the biorefinery. Science 2014, 344, 1246843. [Google Scholar] [CrossRef] [PubMed]
- Azadi, P.; Inderwildi, O.R.; Farnood, R.; King, D.A. Liquid fuels, hydrogen and chemicals from lignin: A critical review. Renewable Sustainable Energy Rev. 2013, 21, 506–523. [Google Scholar] [CrossRef]
- Chakar, F.S.; Ragauskas, A.J. Review of current and future softwood kraft lignin process chemistry. Ind. Crops Prod. 2004, 20, 131–141. [Google Scholar] [CrossRef]
- Danielson, B.; Simonson, R. Kraft lignin in phenol formaldehyde resin. Part 1. Partial replacement of phenol by kraft lignin in phenol formaldehyde adhesives for plywood. J. Adhes. Sci. Technol. 1998, 12, 923. [Google Scholar] [CrossRef]
- Vázquez, G.; González, J.; Freire, S.; Antorrena, G. Effect of chemical modification of lignin on the gluebond performance of lignin-phenolic resins. Bioresour. Technol. 1997, 60, 191–198. [Google Scholar] [CrossRef]
- Choura, M.; Belgacem, N.; Gandini, A. Acid-Catalyzed Polycondensation of Furfuryl Alcohol: Mechanisms of Chromophore Formation and Cross-Linking. Macromolecules 1996, 29, 3839–3850. [Google Scholar] [CrossRef]
- Bertarione, S.; Bonino, F.; Cesano, F.; Jain, S.; Zanetti, M.; Scarano, D.; Zecchina, A. Micro-FTIR and Micro-Raman Studies of a Carbon Film Prepared from Furfuryl Alcohol Polymerization. J. Phys. Chem. 2009, 113, 10571–10574. [Google Scholar] [CrossRef]
- Bertarione, S.; Bonino, F.; Cesano, F.; Damin, A.; Scarano, D.; Zecchina, A. Furfuryl Alcohol Polymerization in H-Y Confined Spaces: Reaction Mechanism and Structure of Carbocationic Intermediates. J. Phys. Chem. 2008, 112, 2580–2589. [Google Scholar] [CrossRef]
- Abdullah, U.H.B.; Pizzi, A. Tannin-furfuryl alcohol wood panel adhesives without formaldehyde. Eur. J. Wood Wood Prod. 2013, 71, 131–132. [Google Scholar] [CrossRef]
- Guigo, N.; Mija, A.; Vincent, L.; Sbirrazzuoli, N. Eco-friendly composite resins based on renewable biomass resources: Polyfurfuryl alcohol/lignin thermosets. Eur. Polym. J. 2010, 46, 1016–1023. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, W.; Zhou, X.; Liang, J.; Du, G.; Wu, Z. Lignin-based adhesive crosslinked by furfuryl alcohol–glyoxal and epoxy resins. Nord. Pulp Pap. Res. J. 2019, 34, 228–238. [Google Scholar] [CrossRef]
- Dongre, P.; Driscoll, M.; Amidon, T.; Bujanovic, B. Lignin-Furfural Based Adhesives. Energies 2015, 8, 7897–7914. [Google Scholar] [CrossRef]
- Papadopoulos, A.; Hill, C.; Traboulay, E.R.B.; Hague, J. Isocyanate resins for particleboard: PMDI vs EMDI. Holz Roh- Werkst. (1937-2008) 2002, 60, 81–83. [Google Scholar] [CrossRef]
- Solt, P.; Konnerth, J.; Gindl-Altmutter, W.; Kantner, W.; Moser, J.; Mitter, R.; van Herwijnen, H.W.G. Technological performance of formaldehyde-free adhesive alternatives for particleboard industry. Int. J. Adhes. Adhes. 2019, 94, 99–131. [Google Scholar] [CrossRef]
- Kutnar, A.; Burnard, M.D. The past, present, and future of EU wood adhesive research and market. In Proceedings of the International Conference on Wood Adhesives, Toronto, ON, Canada, 9–11 October 2014; pp. 22–35. [Google Scholar]
- Amaral-Labat, G.A.; Pizzi, A.; Gonçalves, A.R.; Celzard, A.; Rigolet, S.; Rocha, G.J.M. Environment-friendly soy flour-based resins without formaldehyde. J. Appl. Polym. Sci. 2008, 108, 624–632. [Google Scholar] [CrossRef]
- Lei, H.; Pizzi, A.; Du, G. Environmentally friendly mixed tannin/lignin wood resins. J. Appl. Polym. Sci. 2008, 107, 203–209. [Google Scholar] [CrossRef]
- Ballerini, A.; Despres, A.; Pizzi, A. Non-toxic, zero emission tannin-glyoxal adhesives for wood panels. Holz Roh- Werkst. (1937-2008) 2005, 63, 477–478. [Google Scholar] [CrossRef]
- Hemmilä, V.; Hosseinpourpia, R.; Adamopoulos, S.; Eceiza, A. Characterization of Wood-based Industrial Biorefinery Lignosulfonates and Supercritical Water Hydrolysis Lignin for Value-Added Applications. Under review for Waste and Biomass Valorization. 2019. [Google Scholar]
- Hosseinpourpia, R.; Echart, A.S.; Adamopoulos, S.; Gabilondo, N.; Eceiza, A. Modification of Pea Starch and Dextrin Polymers with Isocyanate Functional Groups. Polymers 2018, 10, 939. [Google Scholar] [CrossRef]
- EN 205, Adhesives. Wood adhesives for non-structural applications. In Determination of Tensile Shear Strength of Lap Joints; European Committee for Standardization: Brussels, Belgium, 2016; Available online: http://www.rivacommerce.com/images/EN205-204.pdf (accessed on 9 October 2019).
- Hosseinpourpia, R.; Adamopoulos, S.; Parsland, C. Utilization of different tall oils for improving the water resistance of cellulosic fibers. J. Appl. Polym. Sci. 2018, 136, 47303. [Google Scholar] [CrossRef]
- Sernek, M.; Resnik, J.; Kamke, F.A. Penetration of liquid urea-formaldehyde adhesive into beech wood. Wood Fiber Sci. 1999, 41–48. [Google Scholar]
- Bastani, A.; Adamopoulos, S.; Militz, H. Effect of open assembly time and equilibrium moisture content on the penetration of polyurethane adhesive into thermally modified wood. J. Adhes. 2017, 93, 575–583. [Google Scholar] [CrossRef]
- EN 319, Particleboards and fibreboards—Determination of tensile strength perpendicular to the plane of the board. 1993, European Standard. Available online: https://apawood-europe.org/official-guidelines/european-standards/individual-standards/en-319/ (accessed on 9 October 2019).
- EN 310, Wood-based panels—Determination of modulus of elasticity in bending and of bending strength. 1993, European Standard. Available online: https://www.iso.org/standard/32836.html (accessed on 9 October 2019).
- EN 317, Particleboards and fibreboards—Determination of swelling in thickness after immersion in water. 1993, European Standard. Available online: https://www.scienceopen.com/document?vid=38d0d5f2-5ac7-45ff-89ff-e33e6831ea7f (accessed on 9 October 2019).
- ASTM D 6007, Standard test method for determining formaldehyde concentrations in air from wood products using a small-scale chamber. 2014, ASTM International. Available online: https://www.astm.org/Standards/D6007.htm (accessed on 9 October 2019).
- Hemmilä, V.; Zabka, M.; Adamopoulos, S. Evaluation of Dynamic Microchamber as a Quick Factory Formaldehyde Emission Control Method for Industrial Particleboards. Adv. Mater. Sci. Eng. 2018, 1–9. [Google Scholar] [CrossRef]
- Hemmilä, V.; Meyer, B.; Larsen, A.; Schwab, H.; Adamopoulos, S. Influencing factors, repeatability and correlation of chamber methods in measuring formaldehyde emissions from fiber- and particleboards. Int. J. Adhes. Adhes. 2019, 95, 102420. [Google Scholar] [CrossRef]
- Solt, P.; Jääskeläinen, A.-S.; Lingenfelter, P.; Konnerth, J.; van Herwijnen, H. Impact of Molecular Weight of Kraft Lignin on Adhesive Performance of Lignin-Based Phenol-Formaldehyde Resins. For. Prod. J. 2018, 68, 365–371. [Google Scholar]
- Szczurek, A.; Fierro, V.; Thébault, M.; Pizzi, A.; Celzard, A. Structure and properties of poly(furfuryl alcohol)-tannin polyHIPEs. Eur. Polym. J. 2016, 78, 195–212. [Google Scholar] [CrossRef]
- Kalami, S.; Arefmanesh, M.; Master, E.; Nejad, M. Replacing 100% of phenol in phenolic adhesive formulations with lignin. J. Appl. Polym. Sci. 2017, 134, 45124. [Google Scholar] [CrossRef]
- Wang, M.; Leitch, M.; Xu, C. Synthesis of phenol–formaldehyde resol resins using organosolv pine lignins. Eur. Polym. J. 2009, 45, 3380–3388. [Google Scholar] [CrossRef]
- Ferdosian, F.; Yuan, Z.; Anderson, M.; Xu, C. Sustainable lignin-based epoxy resins cured with aromatic and aliphatic amine curing agents: Curing kinetics and thermal properties. Thermochim. Acta 2015, 618, 48–55. [Google Scholar] [CrossRef]
- Watkins, D.; Nuruddin, M.; Hosur, M.; Tcherbi-Narteh, A.; Jeelani, S. Extraction and characterization of lignin from different biomass resources. J. Mater. Res. Technol. 2015, 4, 26–32. [Google Scholar] [CrossRef]
- Domínguez, J.C.; Grivel, J.C.; Madsen, B. Study on the non-isothermal curing kinetics of a polyfurfuryl alcohol bioresin by DSC using different amounts of catalyst. Thermochim. Acta 2012, 529, 29–35. [Google Scholar] [CrossRef]
- Chen, H.; Yan, N. Application of Western red cedar (Thuja plicata) tree bark as a functional filler in pMDI wood adhesives. Ind. Crops Prod. 2018, 113, 1–9. [Google Scholar] [CrossRef]
- Gavrilovic-Grmusa, I.; Dunky, M.; Miljkovic, J.; Djiporovic-Momcilovic, M. Influence of the viscosity of UF resins on the radial and tangential penetration into poplar wood and on the shear strength of adhesive joints. Holzforschung 2012, 66, 849–856. [Google Scholar] [CrossRef]
- Bastani, A.; Adamopoulos, S.; Koddenberg, T.; Militz, H. Study of adhesive bondlines in modified wood with fluorescence microscopy and X-ray micro-computed tomography. Int. J. Adhes. Adhes. 2016, 68, 351–358. [Google Scholar] [CrossRef]
- Kamke, F.A.; Lee, J.N. Adhesive penetration in wood−a review. Wood Fiber Sci. 2007, 39, 205–220. [Google Scholar]
- Kariz, M.; Sernek, M. Bonding of Heat-Treated Spruce with Phenol-Formaldehyde Adhesive. J. Adhes. Sci. Technol. 2010, 24, 1703–1716. [Google Scholar] [CrossRef]
- Gómez-Fernández, S.; Ugarte, L.; Calvo-Correas, T.; Peña-Rodríguez, C.; Corcuera, M.A.; Eceiza, A. Properties of flexible polyurethane foams containing isocyanate functionalized kraft lignin. Ind. Crops Prod. 2017, 100, 51–64. [Google Scholar] [CrossRef]
- Duong, L.D.; Nam, G.-Y.; Oh, J.-S.; Park, I.-K.; Luong, N.D.; Yoon, H.-K.; Lee, S.-H.; Lee, Y.; Yun, J.-H.; Lee, C.-G.; et al. High Molecular-Weight Thermoplastic Polymerization of Kraft Lignin Macromers with Diisocyanate. BioResources 2014, 9, 13. [Google Scholar] [CrossRef]
- EN 312:2010 Particleboards: Specifications. 2010, Brussels, European standard. Available online: https://infostore.saiglobal.com/en-au/standards/une-en-312-2010-16459_SAIG_AENOR_AENOR_36532/ (accessed on 9 October 2019).
- Luckeneder, P.; Gavino, J.; Kuchernig, R.; Petutschnigg, A.; Tondi, G. Sustainable phenolic fractions as basis for furfuryl alcohol-based co-polymers and their use as wood adhesives. Polymers 2016, 8, 396. [Google Scholar] [CrossRef]
- Dao, L.T.; Zavarin, E. Chemically activated furfuryl alcohol-based wood adhesives. 1. The role of furfuryl alcohol. Holzforschung 1996, 470–476. [Google Scholar] [CrossRef]
- Hosseinpourpia, R.; Adamopoulos, S.; Mai, C.; Taghiyari, H.R. Properties of medium-density fibreboards bonded with dextrin-based wood adhesive. Wood Res. 2019, 64, 185–194. [Google Scholar]
- Sonnenschein, M.F. Introduction to polyurethane chemistry. In Polyurethanes: Science, Technology, Markets, and Trends; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015. [Google Scholar]
- Boran, S.; Usta, M.; Ondaral, S.; Gümüşkaya, E. The efficiency of tannin as a formaldehyde scavenger chemical in medium density fiberboard. Composites B 2012, 43, 2487–2491. [Google Scholar] [CrossRef]
| Adhesive ID | Base 1: Lignin to Tannin Ratio | Crosslinker Amount in Relation to Base 1 (%) | Application Amount for 2-Layered Veneer (g solids/m2) | Application Amount for Particleboard (wt % to dry particles) | Final Viscosity for Veneer/Particleboard (mPa·s) |
|---|---|---|---|---|---|
| UmF 2 | 100 | 12 | 370/370 | ||
| ALS | 10:0 | - | 100 | 12 | 80/90 |
| ALS-FOH | 10:0 | 25 | 100 | 8 | 2100/210 |
| ALS-FOH + m | 9:1 | 25 | 100 | 8 | 2350/190 |
| ALS-pMDI | 10:0 | 25 | 100 | 8 | 120/110 |
| ALS-pMDI + m | 9:1 | 25 | 100 | 8 | 110/100 |
| Adhesive identification | Onset (°C) | Tmax1 (°C) | Tmax2 (°C) | Curing Heat (J/g) |
|---|---|---|---|---|
| ALS | 104 | 129 | - | 172 |
| ALS-FOH | 98 | 119 | - | 117 |
| ALS-FOH + m | 84 | 120 | 115 | 126 |
| ALS-pMDI | 75 | 107 | 118 | 123 |
| ALS-pMDI + m | 113 | 125 | 135 | 167 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hemmilä, V.; Adamopoulos, S.; Hosseinpourpia, R.; Ahmed, S.A. Ammonium Lignosulfonate Adhesives for Particleboards with pMDI and Furfuryl Alcohol as Crosslinkers. Polymers 2019, 11, 1633. https://doi.org/10.3390/polym11101633
Hemmilä V, Adamopoulos S, Hosseinpourpia R, Ahmed SA. Ammonium Lignosulfonate Adhesives for Particleboards with pMDI and Furfuryl Alcohol as Crosslinkers. Polymers. 2019; 11(10):1633. https://doi.org/10.3390/polym11101633
Chicago/Turabian StyleHemmilä, Venla, Stergios Adamopoulos, Reza Hosseinpourpia, and Sheikh Ali Ahmed. 2019. "Ammonium Lignosulfonate Adhesives for Particleboards with pMDI and Furfuryl Alcohol as Crosslinkers" Polymers 11, no. 10: 1633. https://doi.org/10.3390/polym11101633
APA StyleHemmilä, V., Adamopoulos, S., Hosseinpourpia, R., & Ahmed, S. A. (2019). Ammonium Lignosulfonate Adhesives for Particleboards with pMDI and Furfuryl Alcohol as Crosslinkers. Polymers, 11(10), 1633. https://doi.org/10.3390/polym11101633