Silk Composite with a Fluoropolymer as a Water-Resistant Protein-Based Material
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Silk Powder
2.2. Film Preparation
2.3. Tensile Tests
2.4. Thermal Analysis
2.5. Scanning Electron Microscopy (SEM) Observations
2.6. Advancing Contact Angle of Water
2.7. Water Vapor Barrier Test
2.8. Shrinkage Test
2.9. Biodegradation Test
3. Results and Discussion
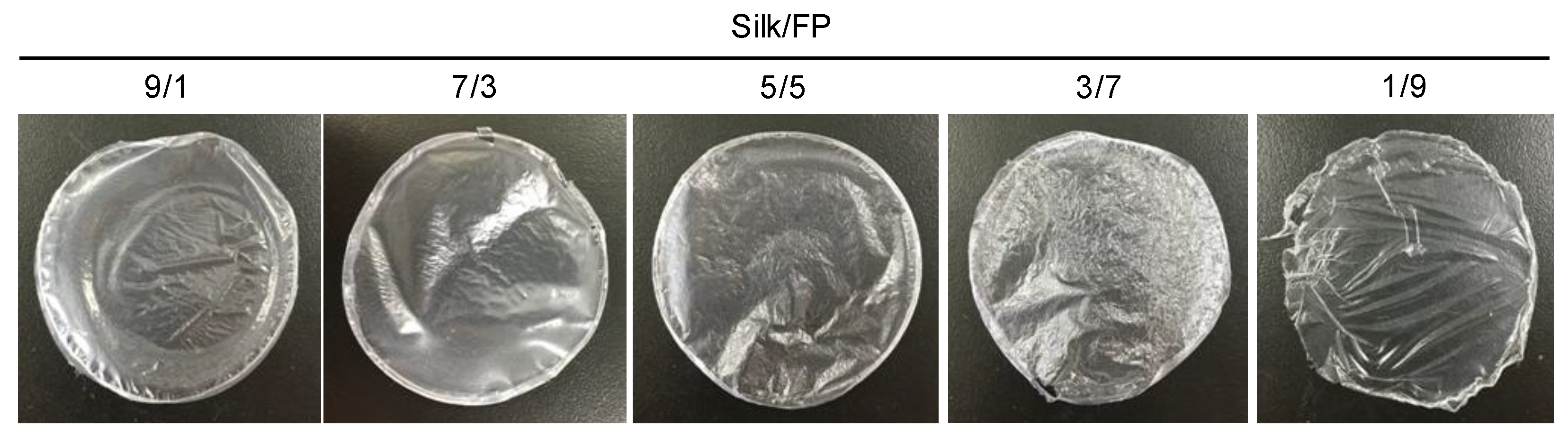
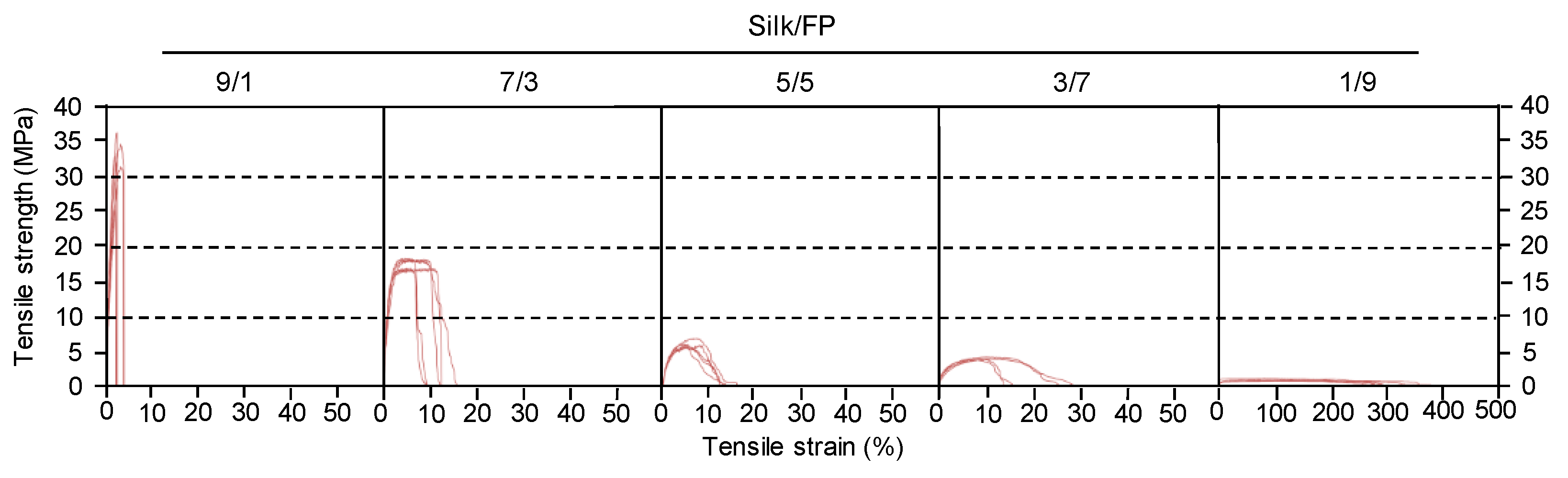
3.1. Basic Properties of Silk/Fluoropolymer Blend Films
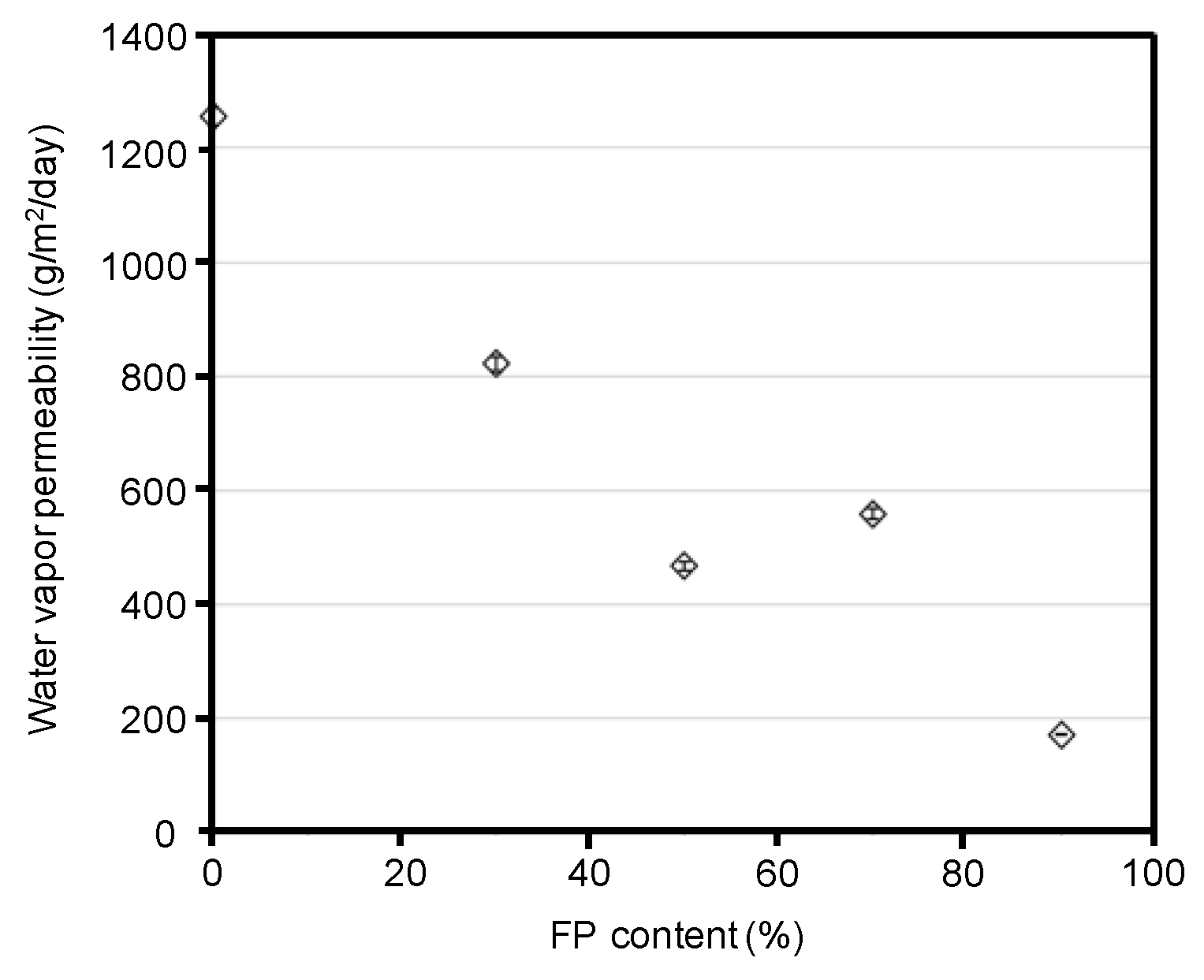
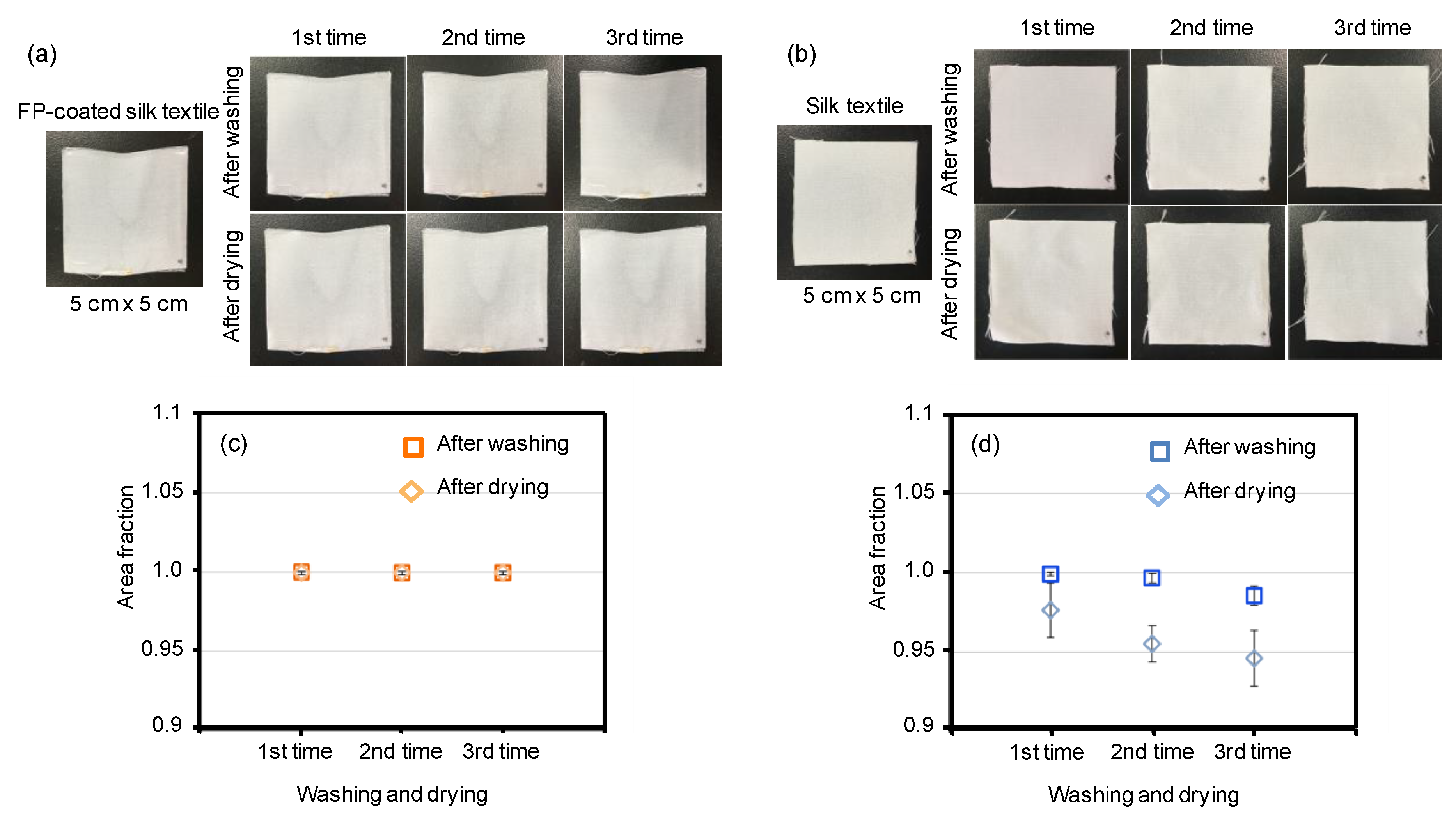
3.2. Fluoropolymer Coating with Silk Materials
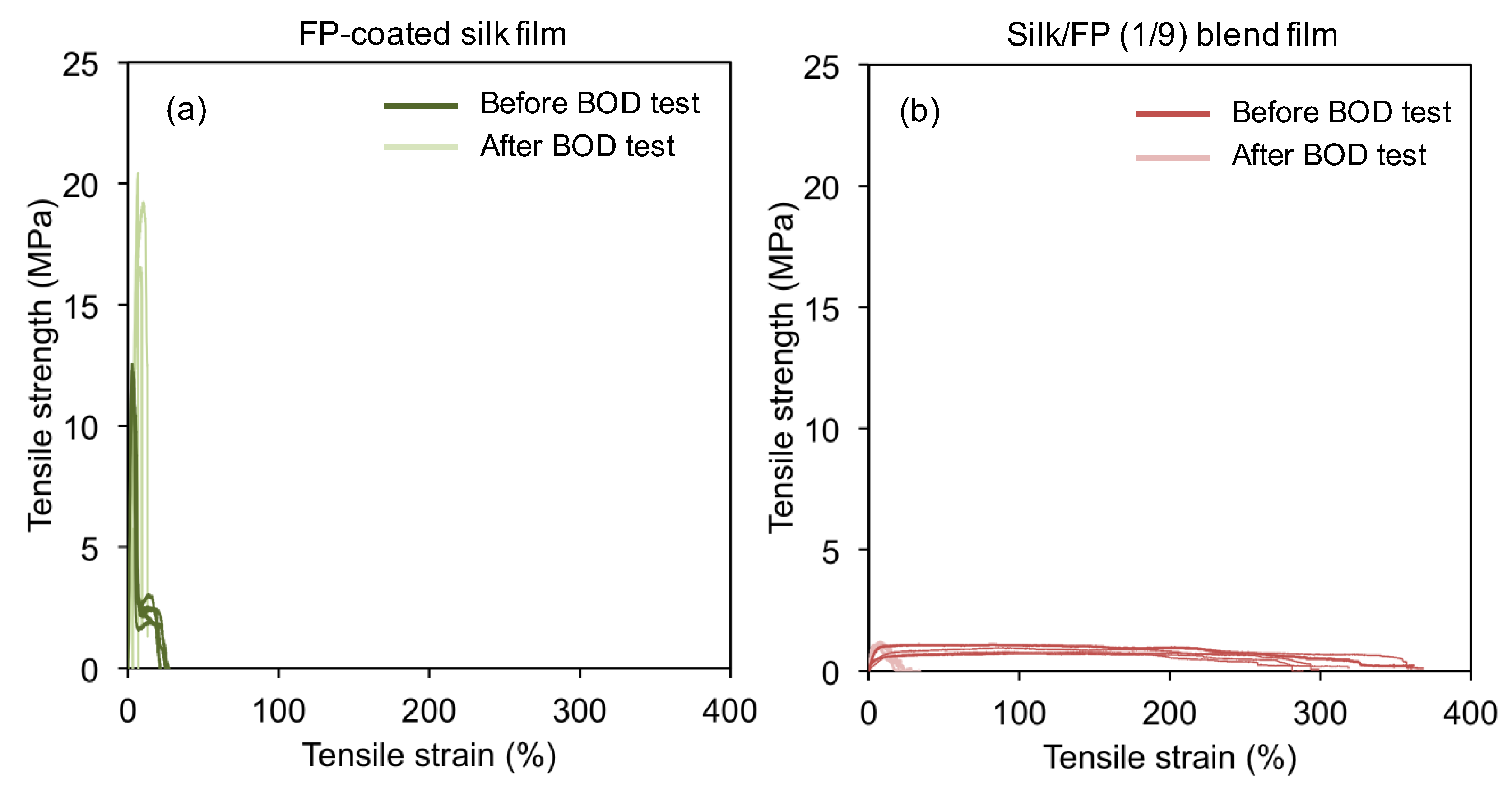
3.3. Biodegradability of Silk, Fluoropolymer-Coated Silk, and Silk/Fluoropolymer Blend
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| RH | relative humidity |
| DSC | differential scanning calorimetry |
| NMR | nuclear magnetic resonance |
| HFIP | hexafluoro-2-propanol |
| SEM | scanning electron microscopy |
| θadv | advancing contact angle |
| FP | fluoropolymer |
| Tg | glass transition temperature |
| BOD | biochemical oxygen demand |
References
- Kaplan, D.L.; Mello, C.M.; Arcidiacono, S.; Fossey, S.; Senecal, K.; Muller, W. Silk. In Protein-Based Materials; Springer: Berlin, Germany, 1997; pp. 103–131. [Google Scholar]
- Pritchard, E.M.; Kaplan, D.L. Silk fibroin biomaterials for controlled release drug delivery. Expert Opin. Drug Deliv. 2011, 8, 797–811. [Google Scholar] [CrossRef] [PubMed]
- Numata, K.; Cebe, P.; Kaplan, D.L. Mechanism of enzymatic degradation of beta-sheet crystals. Biomaterials 2010, 31, 2926–2933. [Google Scholar] [CrossRef] [PubMed]
- Vollrath, F.; Porter, D. Spider silk as a model biomaterial. Appl. Phys. A Mater. 2006, 82, 205–212. [Google Scholar] [CrossRef]
- Altman, G.H.; Diaz, F.; Jakuba, C.; Calabro, T.; Horan, R.L.; Chen, J.; Lu, H.; Richmond, J.; Kaplan, D.L. Silk-based biomaterials. Biomaterials 2003, 24, 401–416. [Google Scholar] [CrossRef]
- Numata, K.; Kaplan, D.L. Silk-based delivery systems of bioactive molecules. Adv. Drug Deliv. Rev. 2010, 62, 1497–1508. [Google Scholar] [CrossRef] [PubMed]
- Rockwood, D.N.; Preda, R.C.; Yucel, T.; Wang, X.; Lovett, M.L.; Kaplan, D.L. Materials fabrication from bombyx mori silk fibroin. Nat. Protoc. 2011, 6, 1612–1631. [Google Scholar] [CrossRef] [PubMed]
- Numata, K. Poly(amino acid)s/polypeptides as potential functional and structural materials. Polym. J. 2015, 47, 537–545. [Google Scholar] [CrossRef]
- Numata, K.; Masunaga, H.; Hikima, T.; Sasaki, S.; Sekiyama, K.; Takata, M. Use of extension-deformation-based crystallisation of silk fibres to differentiate their functions in nature. Soft Matter 2015, 11, 6335–6342. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Dong, L.; Hickner, M.A.; Glass, T.E.; Webb, V.; McGrath, J.E. State of water in disulfonated poly(arylene ether sulfone) copolymers and a perfluorosulfonic acid copolymer (nafion) and its effect on physical and electrochemical properties. Macromolecules 2003, 36, 6281–6285. [Google Scholar] [CrossRef]
- Kuntz, I.D. Hydration of macromolecules. III. Hydration of polypeptides. J. Am. Chem. Soc. 1971, 93, 514–516. [Google Scholar] [CrossRef]
- Lee, K.Y.; Ha, W.S. DSC studies on bound water in silk fibroin/s-carboxymethyl kerateine blend films. Polymer 1999, 40, 4131–4134. [Google Scholar] [CrossRef]
- Numata, K.; Katashima, T.; Sakai, T. State of water, molecular structure, and cytotoxicity of silk hydrogels. Biomacromolecules 2011, 12, 2137–2144. [Google Scholar] [CrossRef] [PubMed]
- Ishida, M.; Asakura, T.; Yokoi, M.; Saito, H. Solvent-induced and mechanical-treatment-induced conformational transition of silk fibroins studied by high-resolution solid-state c-13 NMR-spectroscopy. Macromolecules 1990, 23, 88–94. [Google Scholar] [CrossRef]
- Asakura, T.; Demura, M.; Watanabe, Y.; Sato, K. H-1 pulsed nmr-study of bombyx-mori silk fibroin—Dynamics of fibroin and of absorbed water. J. Polym. Sci. Polym. Phys. 1992, 30, 693–699. [Google Scholar] [CrossRef]
- Yoshimizu, H.; Asakura, T. The structure of bombyx-mori silk fibroin membrane swollen by water studied with ESR, C-13-NMR, and FT-IR spectroscopies. J. Appl. Polym. Sci. 1990, 40, 1745–1756. [Google Scholar] [CrossRef]
- Asakura, T.; Isobe, K.; Aoki, A.; Kametani, S. Macromolecules, in press.
- Hu, X.; Kaplan, D.; Cebe, P. Effect of water on the thermal properties of silk fibroin. Thermochim. Acta 2007, 461, 137–144. [Google Scholar] [CrossRef]
- Hu, X.; Kaplan, D.; Cebe, P. Dynamic protein−water relationships during β-sheet formation. Macromolecules 2008, 41, 3939–3948. [Google Scholar] [CrossRef]
- Mo, C.; Wu, P.; Chen, X.; Shao, Z. The effect of water on the conformation transition of bombyx mori silk fibroin. Vib. Spectrosc. 2009, 51, 105–109. [Google Scholar] [CrossRef]
- Guan, J.; Porter, D.; Vollrath, F. Thermally induced changes in dynamic mechanical properties of native silks. Biomacromolecules 2013, 14, 930–937. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Q.; Yao, J.; Huang, L.; Chen, X.; Shao, Z. Correlation between structural and dynamic mechanical transitions of regenerated silk fibroin. Polymer 2010, 51, 6278–6283. [Google Scholar] [CrossRef]
- Fu, C.; Porter, D.; Shao, Z. Moisture effects on antheraea pernyi silk’s mechanical property. Macromolecules 2009, 42, 7877–7880. [Google Scholar] [CrossRef]
- Plaza, G.R.; Guinea, G.V.; Pérez-Rigueiro, J.; Elices, M. Thermo-hygro-mechanical behavior of spider dragline silk: Glassy and rubbery states. J. Polym. Sci. Part B Polym. Phys. 2006, 44, 994–999. [Google Scholar] [CrossRef]
- Agarwal, N.; Hoagland, D.A.; Farris, R.J. Effect of moisture absorption on the thermal properties of bombyx mori silk fibroin films. J. Appl. Polym. Sci. 1997, 63, 401–410. [Google Scholar] [CrossRef]
- Hu, X.; Shmelev, K.; Sun, L.; Gil, E.S.; Park, S.H.; Cebe, P.; Kaplan, D.L. Regulation of silk material structure by temperature-controlled water vapor annealing. Biomacromolecules 2011, 12, 1686–1696. [Google Scholar] [CrossRef] [PubMed]
- Numata, K.; Ifuku, N.; Masunaga, H.; Hikima, T.; Sakai, T. Silk resin with hydrated dual chemical-physical cross-links achieves high strength and toughness. Biomacromolecules 2017, 18, 1937–1946. [Google Scholar] [CrossRef] [PubMed]
- Yazawa, K.; Ishida, K.; Masunaga, H.; Hikima, T.; Numata, K. Influence of water content on the beta-sheet formation, thermal stability, water removal, and mechanical properties of silk materials. Biomacromolecules 2016, 17, 1057–1066. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.J.; Kaplan, D.L. Mechanism of silk processing in insects and spiders. Nature 2003, 424, 1057–1061. [Google Scholar] [CrossRef] [PubMed]
- Malay, A.D.; Sato, R.; Yazawa, K.; Watanabe, H.; Ifuku, N.; Masunaga, H.; Hikima, T.; Guan, J.; Mandal, B.B.; Damrongsakkul, S.; et al. Relationships between physical properties and sequence in silkworm silks. Sci. Rep. 2016, 6, 27573. [Google Scholar] [CrossRef] [PubMed]
- Numata, K.; Srivastava, R.K.; Finne-Wistrand, A.; Albertsson, A.-C.; Doi, Y.; Abe, H. Branched poly(lactide) synthesized by enzymatic polymerization: Effects of molecular branches and stereochernistry on enzymatic degradation and alkaline hydrolysis. Biomacromolecules 2007, 8, 3115–3125. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Fukuzumi, H.; Saito, T.; Isogai, A.; Zhang, L. Transparent cellulose films with high gas barrier properties fabricated from aqueous alkali/urea solutions. Biomacromolecules 2011, 12, 2766–2771. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, K.; Urano, Y.; Numata, K. Biodegradability of nylon 4 film in a marine environment. Polym. Degrad. Stab. 2013, 98, 1847–1851. [Google Scholar] [CrossRef]







© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Numata, K.; Ifuku, N.; Isogai, A. Silk Composite with a Fluoropolymer as a Water-Resistant Protein-Based Material. Polymers 2018, 10, 459. https://doi.org/10.3390/polym10040459
Numata K, Ifuku N, Isogai A. Silk Composite with a Fluoropolymer as a Water-Resistant Protein-Based Material. Polymers. 2018; 10(4):459. https://doi.org/10.3390/polym10040459
Chicago/Turabian StyleNumata, Keiji, Nao Ifuku, and Akira Isogai. 2018. "Silk Composite with a Fluoropolymer as a Water-Resistant Protein-Based Material" Polymers 10, no. 4: 459. https://doi.org/10.3390/polym10040459
APA StyleNumata, K., Ifuku, N., & Isogai, A. (2018). Silk Composite with a Fluoropolymer as a Water-Resistant Protein-Based Material. Polymers, 10(4), 459. https://doi.org/10.3390/polym10040459





