Optical and Thermomechanical Properties of Doped Polyfunctional Acrylate Copolymers
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
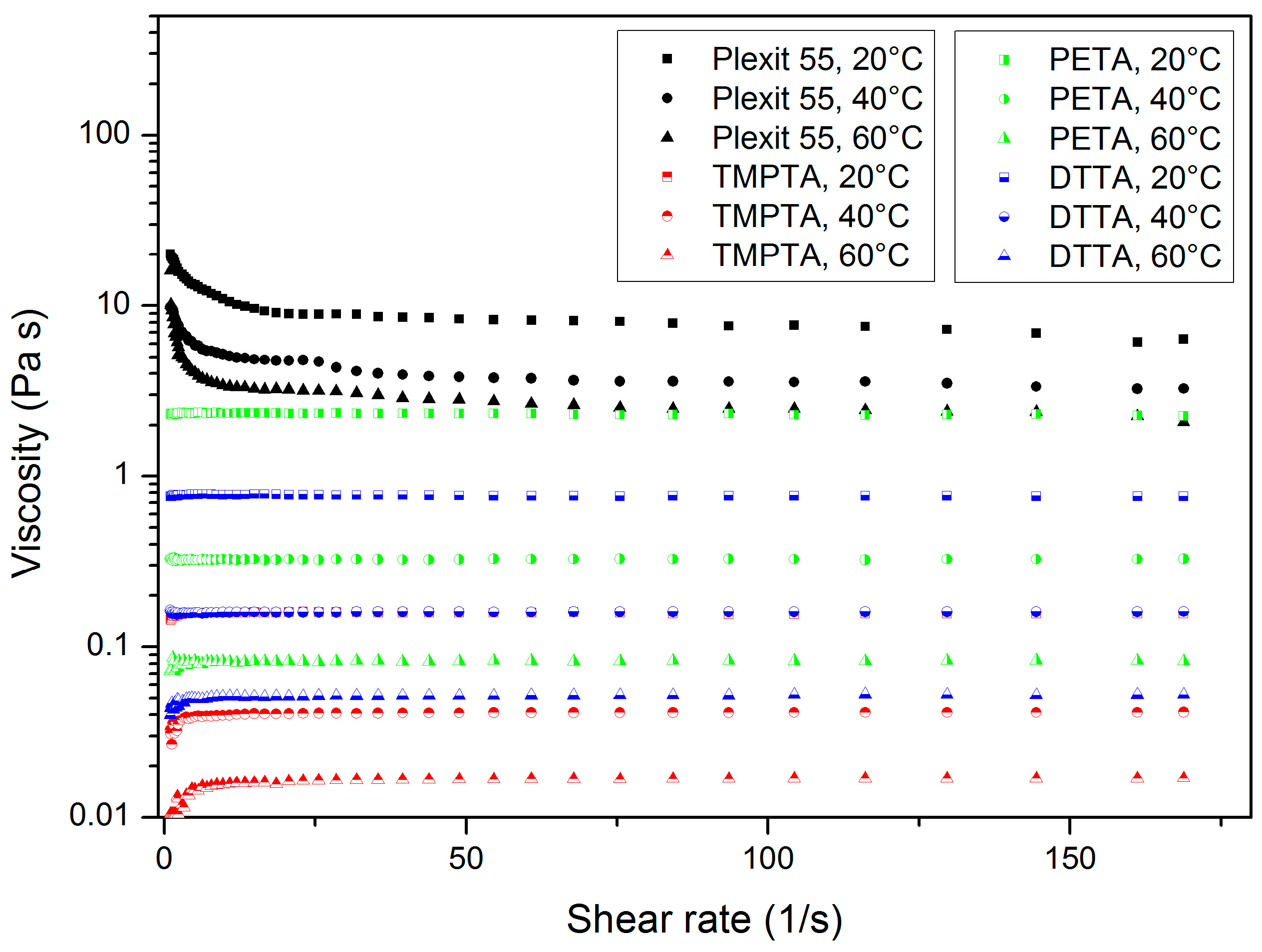
3.1. Shear Rate and Temperature-Dependent Viscosity of Uncured Mixtures
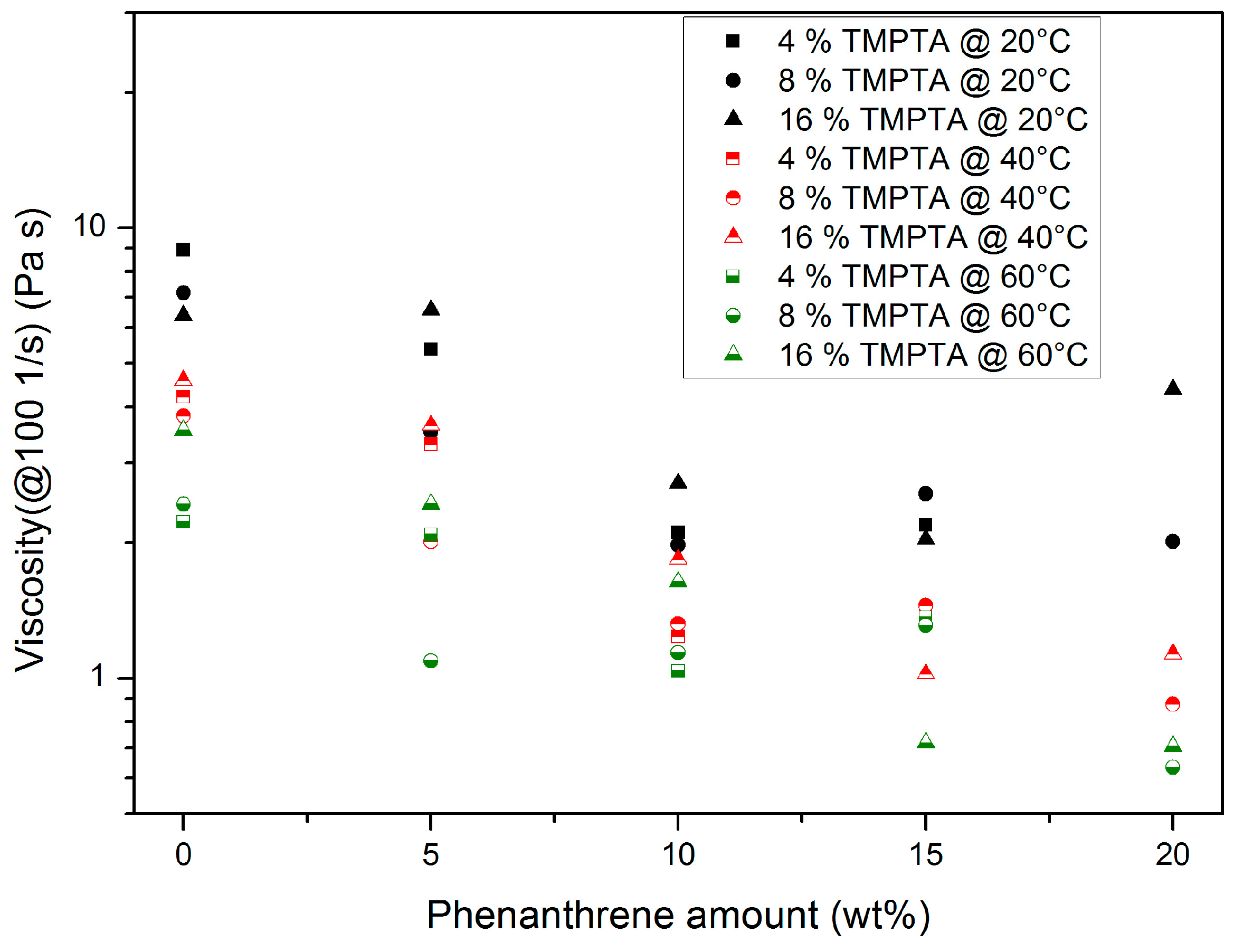
3.1.1. Trimethylolpropantriacrylate (TMPTA)-Containing Systems
- A temperature increase causes a viscosity drop: this is a common behaviour for all organic liquids and can be described by the Andrade–Eyring relation [25].
- Increasing TMPTA amounts cause a viscosity drop due to simple mixing rules.
- A total of 20 wt % phenanthrene exceeds the solubility limit at a TMPTA content of 4 wt %.
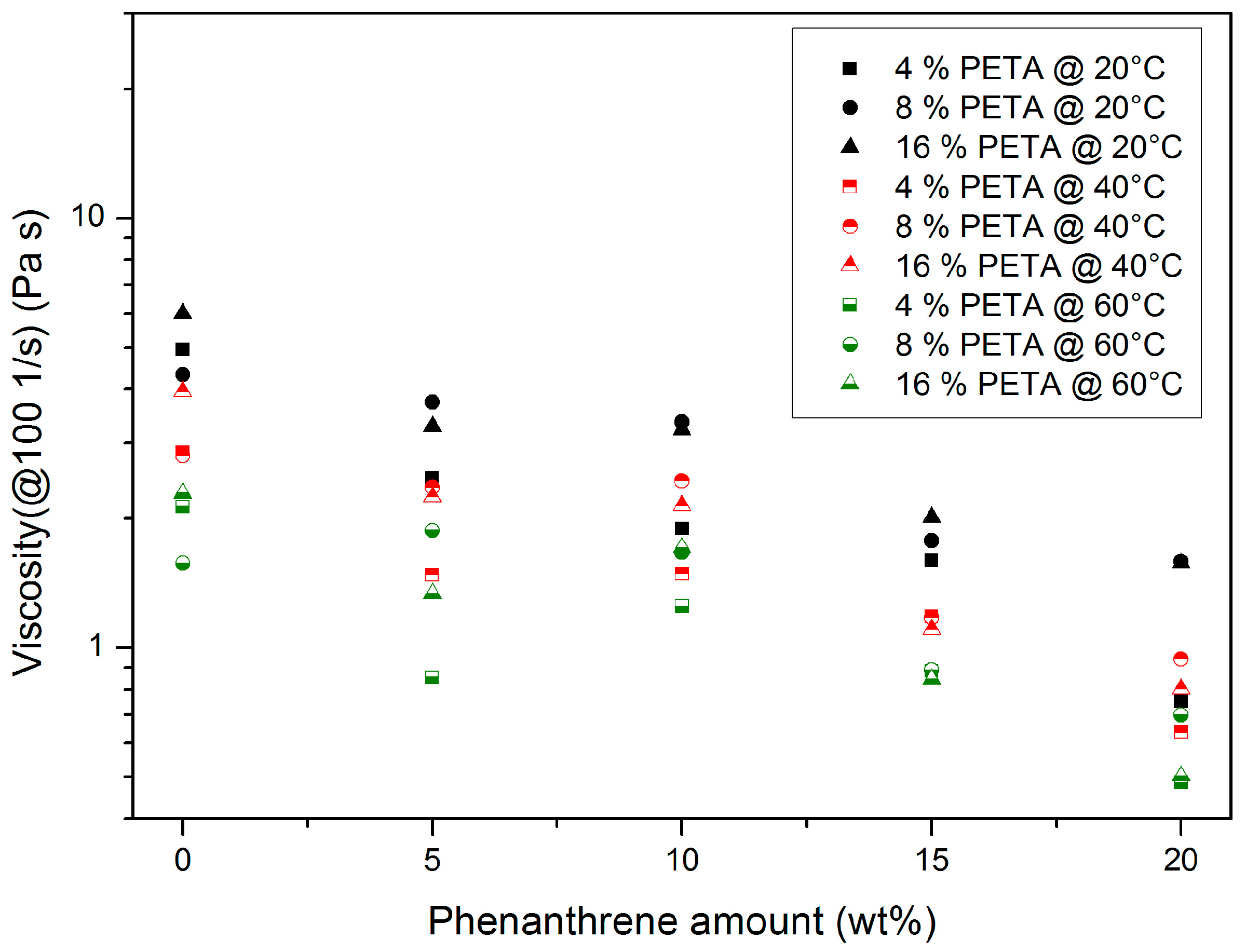
3.1.2. Pentaerythritol Triacrylate (PETA)-Containing Systems
- At a constant PETA content, a temperature increase causes a viscosity drop.
- Increasing phenanthrene amounts cause a principal viscosity drop due to plasticizing.
- Within the investigated concentration range, phenanthrene is completely soluble up to 20 wt %.
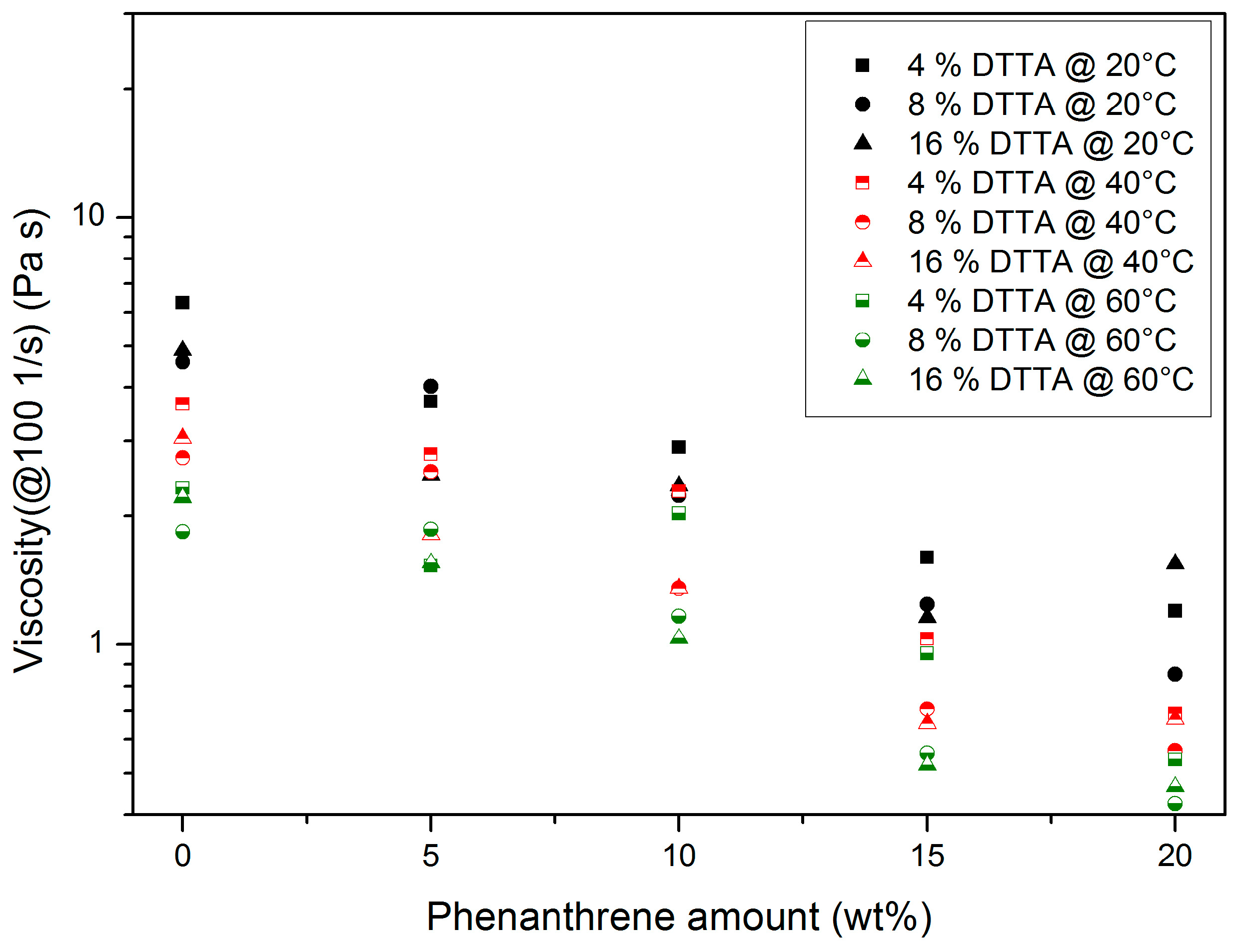
3.1.3. Di(trimethylolpropane) Tetraacrylate (DTTA)-Containing Systems
- At constant DTTA content, a temperature increase causes a viscosity drop.
- Increasing phenanthrene amounts cause a principal viscosity drop due to plasticizing.
- A total of 20 wt % phenanthrene exceeds the solubility limit at a DTTA content of 16 wt %.
3.2. Optical Properties of Uncured and Cured Systems
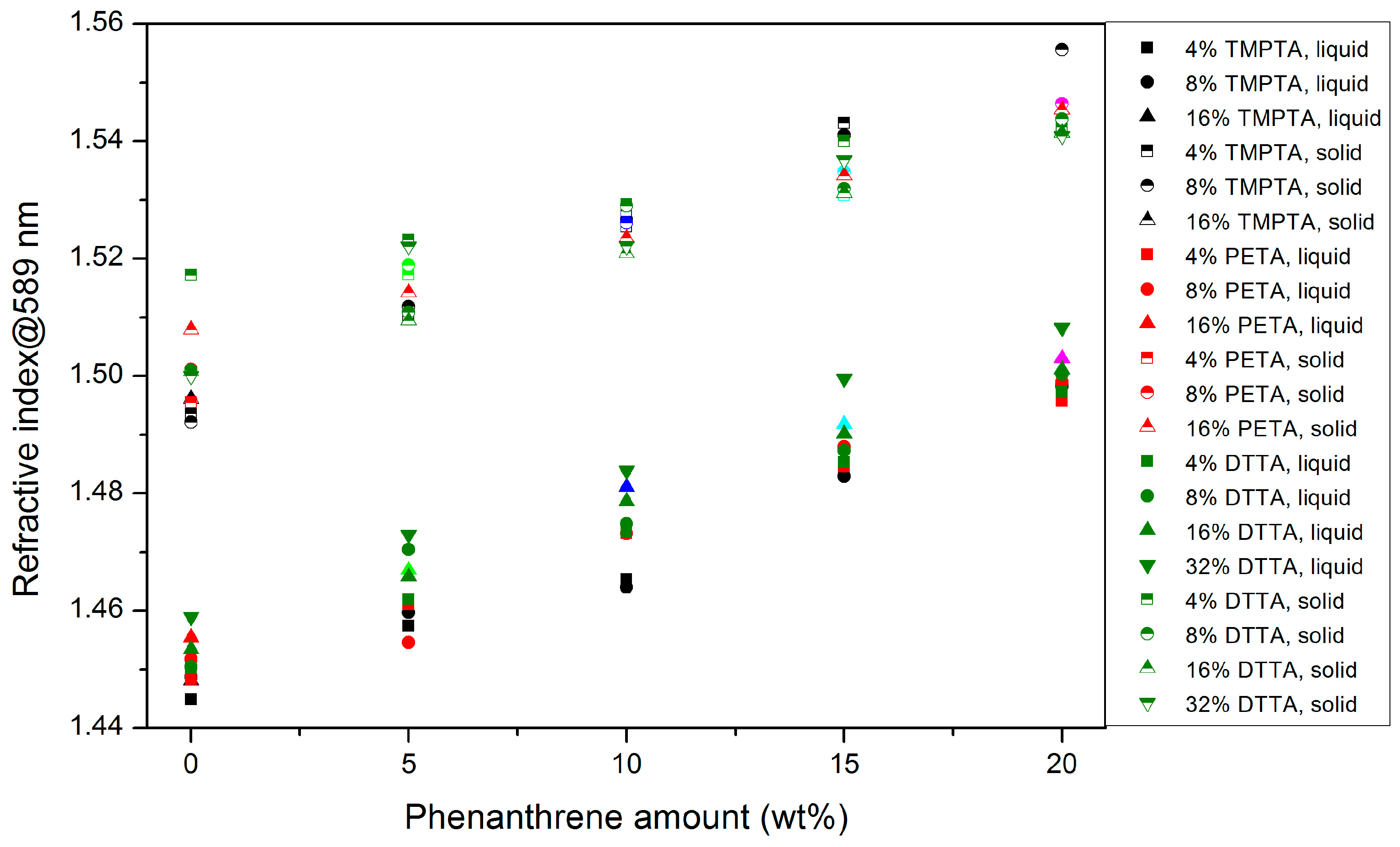
3.2.1. Refractive Index
- Due to the lower density of the liquid monomer mixtures, the refractive index is lower in general than the related ones in the solidified state.
- All measured values are close together, the rise in the refractive index can be attributed mainly to the increasing phenanthrene moiety.
- In the case of DTTA-based systems, the use of larger DTTA amounts are favorable in the liquid state.
- A clear correlation of the polyfunctional acrylate monomer structure with the refractive index values cannot be found.
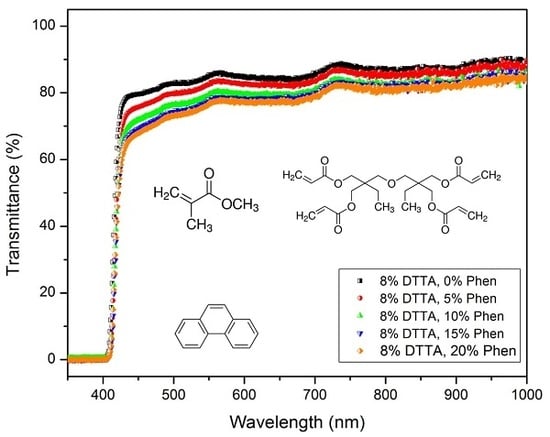
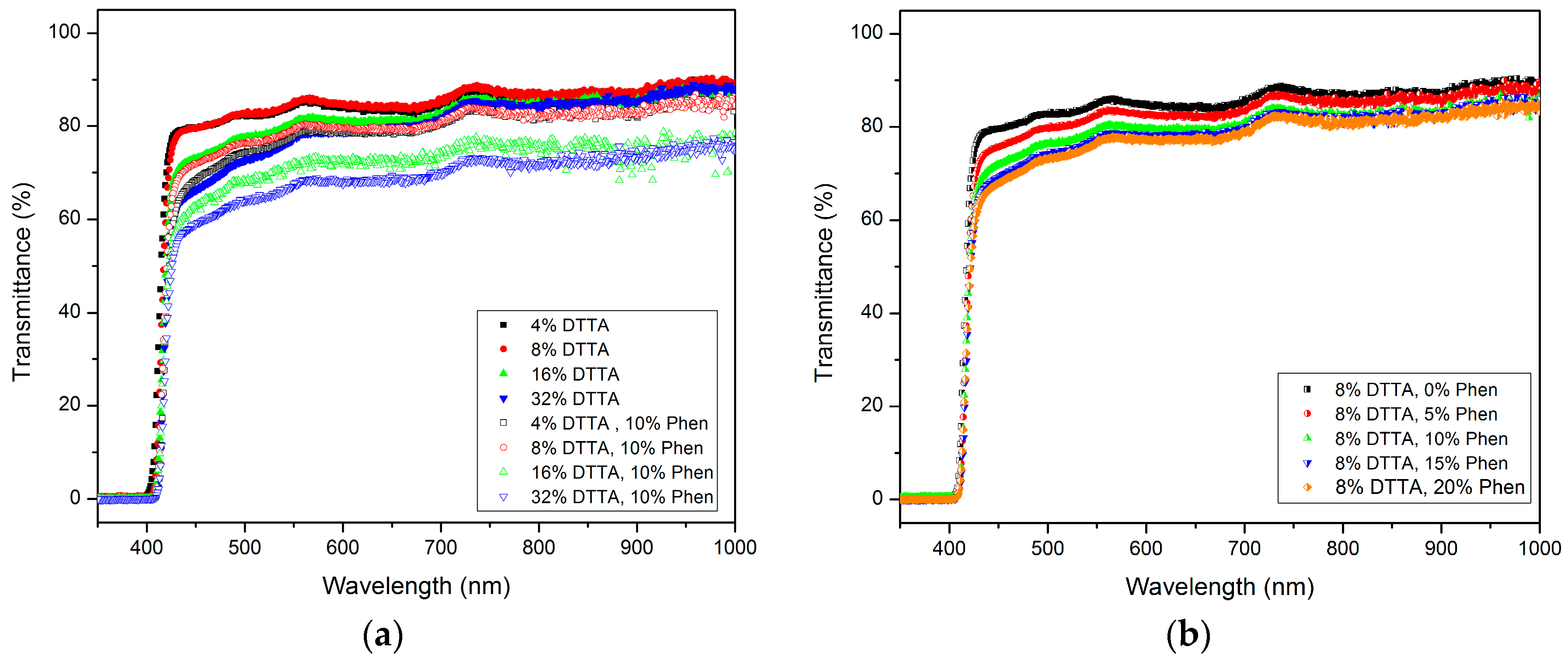
3.2.2. Optical Transmittance in the Visible Range
3.3. Photoinitiated Curing Behaviour
- Occurrence of an induction period prior to the chemical reaction;
- Reduction of the polymerization rate and final conversion;
- Reduction of the chain length and conversion rate;
- Formation of a sticky surface appearance due to oligomer formation.
- UV-curing in an inert atmosphere;
- Preventing oxygen diffusion by protecting films or transparent foils;
- Selection of highly effective light sources like vapor pressure arc lamps;
- Use of suitable additives trapping oxygen-like triphenylphosphine [31];
- Use of monomers with huge chemical reactivity.
3.4. Thermomechnical Properties
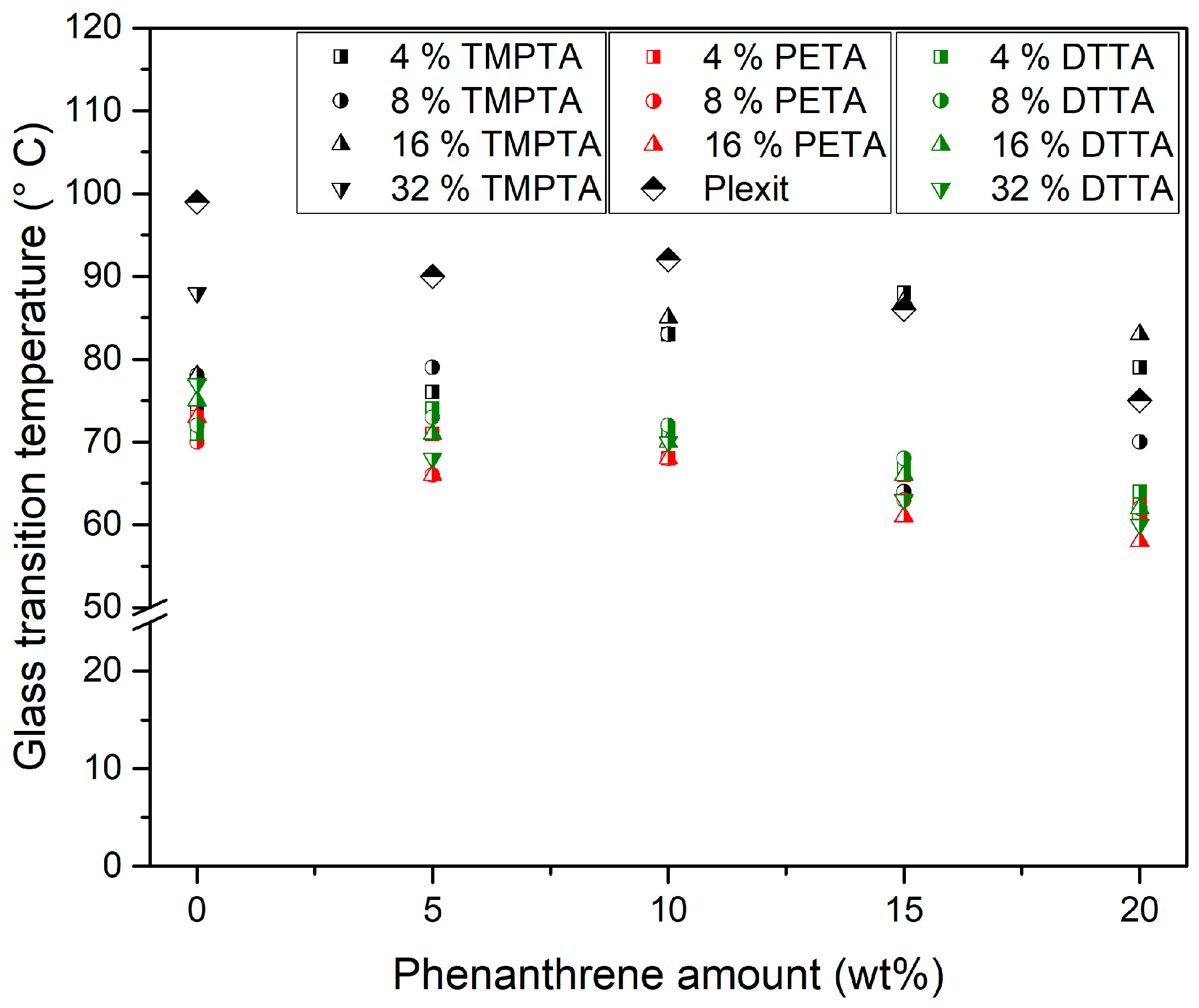
3.4.1. Glass Transition Temperature
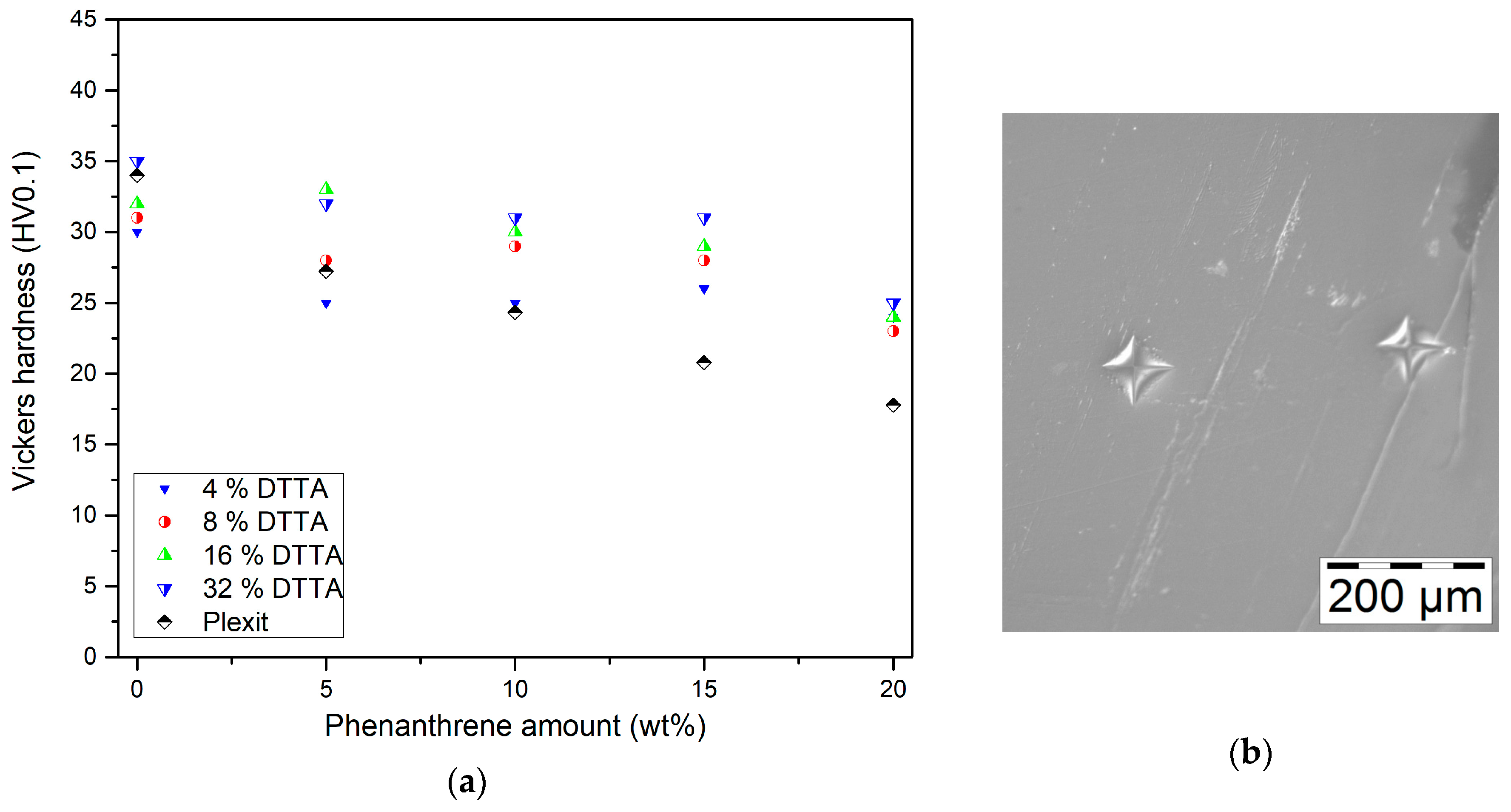
3.4.2. Vickers Hardness
4. Conclusions
- The resulting mixture’s viscosity can be adjusted by the addition of the polyfunctional acrylate comonomer without a pronounced impact on the optical properties.
- The presence of the polyfunctional acrylate monomer supports the photopolymerization and eliminates the negative oxygen inhibition effect.
- The polyfunctional acrylates have no significant relevant effect on the refractive index and the optical transmittance in the visible.
- Phenanthrene can be used for a pronounced increase of the refractive index.
- Increasing amounts of the polyfunctional acrylate in the copolymer can compensate the plasticizing effect of the dopant phenanthrene, enabling a higher concentration of the dopant in the guest–host system and therefore larger refractive index values.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fréchet, J.M.J. Functional polymers: From plastic electronics to polymer-assisted therapeutics. Prog. Polym. Sci. 2005, 30, 844–857. [Google Scholar] [CrossRef]
- Gale, M.T.; Gimkiewicz, C.; Obi, S.; Schnieper, M.; Söchtig, J.; Thiele, H.; Westenhöfer, S. Replication technology for optical microsystems. Opt. Lasers Eng. 2005, 43, 373–386. [Google Scholar] [CrossRef]
- Szabo, D.V.; Hanemann, T. Polymer Nanocomposites for optical applications. In Advances in Poymer NanoComposites—Types and Applications; Gao, F., Ed.; Woodhead Publishing Ltd.: Oxford, UK, 2012; ISBN 978-1-84569-672-6. [Google Scholar]
- Ferreira, R.A.S.; André, P.S.; Carlos, L.D. Organic-inorganic hybrid materials towards passive and active architectures for the next generation of optical networks. Opt. Mater. 2010, 32, 1397–1409. [Google Scholar] [CrossRef]
- Oh, M.-C.; Chu, W.-S.; Shin, J.-S.; Kim, J.-W.; Kim, K.-J.; Seo, J.-K.; Lee, H.-K.; Noh, Y.-O.; Lee, H.-J. Polymeric optical waveguide devices exploiting special properties of polymer materials. Opt. Commun. 2016, 362, 3–12. [Google Scholar] [CrossRef]
- Overmeyer, L.; Wolfer, T.; Wang, Y.; Schwenke, A.; Sajti, L.; Roth, B.; Dikty, S. Polymer Based Planar Optronic Systems. In Proceedings of the LAMP2013—The 6th International Congress on Laser Advanced Materials Processing, Niigata, Japan, 23–26 July 2013. [Google Scholar]
- Baeumer, S. Handbook of Plastic Optics; Wiley-VCH: Weinheim, UK, 2005; ISBN 3-527-40424-4. [Google Scholar]
- Worgull, M. Hot Embossing—Theory and Technology of Microreplication; Elsevier: Oxford, UK, 2009; ISBN 978-0-8155-1579-1. [Google Scholar]
- Hanemann, T.; Haußelt, J.; Ritzhaupt-Kleissl, E. Compounding, micro injection moulding and characterisation of polycarbonate-nanosized alumina-composites for application in microoptics. Microsyst. Technol. 2009, 15, 421–427. [Google Scholar] [CrossRef]
- Heptagon Technology. Available online: http://hptg.com/technology/wli/ (accessed on 12 January 2018).
- Bollgruen, P.; Wolfer, T.; Gleissner, U.; Mager, D.; Megnin, C.; Overmeyer, L.; Hanemann, T.; Korvink, J.G. Ink-jet printed optical waveguides. Flex. Print. Electron. 2017, 2, 045003. [Google Scholar] [CrossRef]
- Eiselt, T.; Preinfalk, J.; Bittkau, K.; Gomard, G.; Hanemann, T.; Lemmer, U. Inkjet-printed internal light extraction layers for organic light emitting diodes. Flex. Print. Electron. 2018. [Google Scholar] [CrossRef]
- Hofmann, M.; Xiao, Y.; Sherman, S.; Bollgrün, P.; Schmidt, T.; Gleissner, U.; Zappe, H. Inkjet printed single-mode waveguides on hot-embossed foils. In Proceedings of the SPIE Volume 9628 Optical Systems Design 2015: Optical Fabrication, Testing, and Metrology, Jena, Germany, 24 September 2015; Duparré, A., Geyl, R., Eds.; SPIE: Bellingham, WA, USA, 2015; pp. 96281R-1–96281R-6. [Google Scholar] [CrossRef]
- Hanemann, T.; Szabó, D.V. Polymer-Nanoparticle Composites: From Synthesis to Modern Applications. Materials 2010, 3, 3468–3517. [Google Scholar] [CrossRef]
- Schlabach, S.; Ochs, R.; Hanemann, T.; Szabó, D.V. Nanoparticles in polymer-matrix composites. Microsyst. Technol. 2010, 17, 183–193. [Google Scholar] [CrossRef]
- Tao, P.; Viswanath, A.; Li, Y.; Siegel, R.W.; Benicewicz, B.C.; Schadler, L.S. Bulk transparent epoxy nanocomposites filled with poly(glycidyl methacrylate) brush-grafted TiO2 nanoparticles. Polymer 2013, 54, 1639–1646. [Google Scholar] [CrossRef]
- Hanemann, T.; Honnef, K. Tailoring the optical and thermomechanical properties of polymer host-guest systems. J. Appl. Polym. Sci. 2011, 122, 3514–3519. [Google Scholar] [CrossRef]
- Hanemann, T.; Honnef, K. Viscosity and Refractive Index Tailored Methacrylate-Based Polymers. J. Appl. Polym. Sci. 2014, 131, 40194. [Google Scholar] [CrossRef]
- Hanemann, T.; Böhm, J.; Honnef, K.; Ritzhaupt-Kleissl, E.; Haußelt, J. Polymer/Phenanthrene-Derivative Host-Guest Systems: Rheological, Optical and Thermal Properties. Macromol. Mater. Eng. 2007, 292, 285–294. [Google Scholar] [CrossRef]
- Hanemann, T.; Honnef, K. Viscosity and refractive index adjustment of poly(methyl methacrylate-co-ethyleneglycol dimethacrylate) for application in microoptics. Polym. Adv. Technol. 2015, 26, 294–299. [Google Scholar] [CrossRef]
- Yang, L.; Wu, W.; Xu, B.-H.; Jia, P.-Z.; Nie, J. Study of UV-curable composite resin of transfer tray for orthodontics. Front. Mater. Sci. China 2008, 2, 430–436. [Google Scholar] [CrossRef]
- Bretterbauer, K.; Holzmann, C.; Rubatscher, E.; Schwarzinger, C.; Roessler, A.; Paulik, C. UV-curable coatings of highly crosslinked trimethylmelamine based acrylates and methacrylates. Eur. Polym. J. 2013, 49, 4141–4148. [Google Scholar] [CrossRef]
- Magdassi, S. The Chemistry of Inkjet Inks; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2010. [Google Scholar]
- Gleissner, U.; Hanemann, T. Tailoring the optical and rheological properties of an epoxy acrylate based host-guest system. Opt. Eng. 2014, 53. [Google Scholar] [CrossRef]
- Hanemann, T.; Honnef, K. Rheological investigations on the flow behavior of polymer-microsized iron powder composites. Polym. Compos. 2009, 30, 1114–1118. [Google Scholar] [CrossRef]
- Zappe, H. Fundamentals of Micro-Optics; Cambridge University Press: Cambridge, UK, 2010; ISBN -13. [Google Scholar]
- Fouassier, J.P.; Laleveé, J. Photoinitiators for Polymer Synthesis; Wiley-VCH: Weinheim, Germany, 2012; ISBN 978-3-527-33210-6. [Google Scholar]
- Hagemann, H.J.; Jansen, L.G.J. Photoinitiators and photoinitiation, 9. Photoinitiators for radical polymerization which counter oxygen-inhibition. Makromol. Chem. 1988, 189, 2781–2795. [Google Scholar] [CrossRef]
- Ligon, S.C.; Husar, B.; Wutzel, H.; Holman, R.; Liska, R. Strategies to reduce oxygen inhibition in photoinduced polymerization. Chem. Rev. 2014, 114, 557–589. [Google Scholar] [CrossRef] [PubMed]
- Husar, B.; Ligon, S.C.; Wutzel, H.; Hoffmann, H.; Liska, R. Experimental comparison of various anti-oxygen inhibition strategies in LED curing. In Proceedings of the Radtech UV & EB Technical Conference, Bethesda, MD, USA, 12–14 May 2014. [Google Scholar]
- Belon, C.; Allonas, X.; Croutxé-barghorn, C.; Lalevée, J. Overcoming the oxygen inhibition in the photopolymerization of acrylates: A study of the beneficial effect of triphenylphosphine. J. Polym. Sci. Part A Polym. Chem. 2010, 48, 2462–2469. [Google Scholar] [CrossRef]
- Gleißner, U.; Sherman, S.; Megnin, C.; Zappe, H.; Hanemann, T. Polymers with Customizable Optical and Rheological Properties for Printable Single-mode Waveguides. Procedia Eng. 2015, 120, 3–6. [Google Scholar] [CrossRef][Green Version]
- Bruno, P.; Malucelli, G.; Tylkowski, B.; Ferré, J.; Giamberini, M. Acrylic microspheres as drug-delivery systems: Synthesis through in situ microemulsion photoinduced polymerization and characterization. Polym. Int. 2013, 62, 304–309. [Google Scholar] [CrossRef]
- Kaczmarek, H.; Vukovic-Kwiatkowska, I. Preparation and characterization of n interpenetrating networks based on polyacrylates and poly(lactic acid). Express Polym. Lett. 2012, 6, 78–94. [Google Scholar] [CrossRef]
| Series Denotation | Composition [wt %] | ||
|---|---|---|---|
| Plexit55 | Polyfunctional Acrylate | Phenanthrene | |
| 0 wt % series | 98 | 0 | 0 |
| 93 | 0 | 5 | |
| 88 | 0 | 10 | |
| 83 | 0 | 15 | |
| 78 | 0 | 20 | |
| 4 wt % series | 94 | 4 | 0 |
| 89 | 4 | 5 | |
| 84 | 4 | 10 | |
| 80 | 3 | 15 | |
| 75 | 3 | 20 | |
| 8 wt % series | 90 | 8 | 0 |
| 85 | 8 | 5 | |
| 81 | 7 | 10 | |
| 76 | 7 | 15 | |
| 72 | 6 | 20 | |
| 16 wt % series | 82 | 16 | 0 |
| 78 | 15 | 5 | |
| 74 | 14 | 10 | |
| 69 | 14 | 15 | |
| 65 | 13 | 20 | |
| 32 wt % series | 66 | 32 | 0 |
| 63 | 30 | 5 | |
| 59 | 29 | 10 | |
| 56 | 27 | 15 | |
| 53 | 25 | 20 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanemann, T.; Honnef, K. Optical and Thermomechanical Properties of Doped Polyfunctional Acrylate Copolymers. Polymers 2018, 10, 337. https://doi.org/10.3390/polym10030337
Hanemann T, Honnef K. Optical and Thermomechanical Properties of Doped Polyfunctional Acrylate Copolymers. Polymers. 2018; 10(3):337. https://doi.org/10.3390/polym10030337
Chicago/Turabian StyleHanemann, Thomas, and Kirsten Honnef. 2018. "Optical and Thermomechanical Properties of Doped Polyfunctional Acrylate Copolymers" Polymers 10, no. 3: 337. https://doi.org/10.3390/polym10030337
APA StyleHanemann, T., & Honnef, K. (2018). Optical and Thermomechanical Properties of Doped Polyfunctional Acrylate Copolymers. Polymers, 10(3), 337. https://doi.org/10.3390/polym10030337