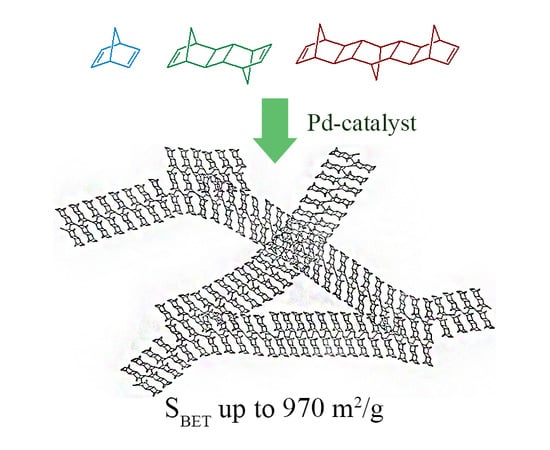
Microporous Materials Based on Norbornadiene-Based Cross-Linked Polymers
Abstract
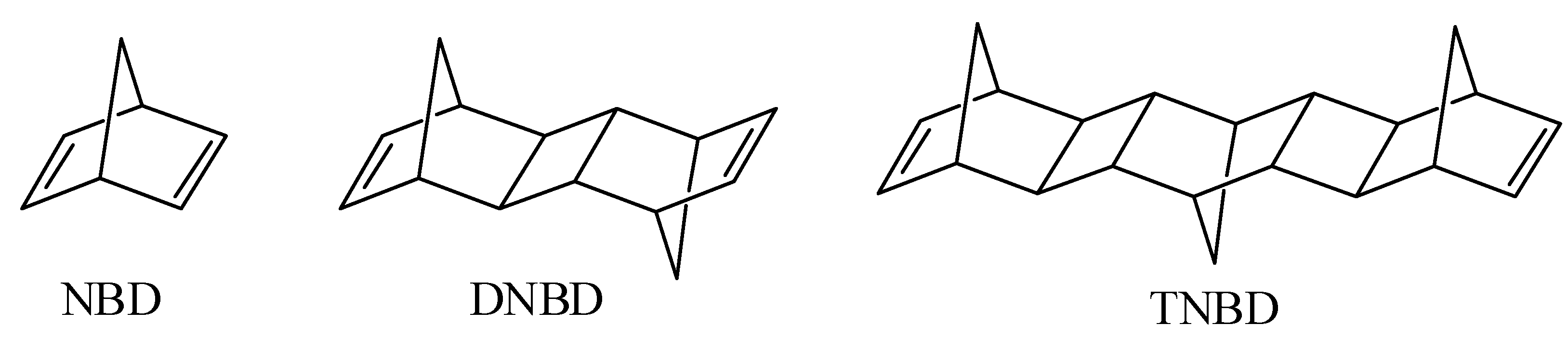
1. Introduction
2. Experimental
2.1. Materials and Methods
2.2. Positron Annihilation Lifetime Spectroscopy (PALS)
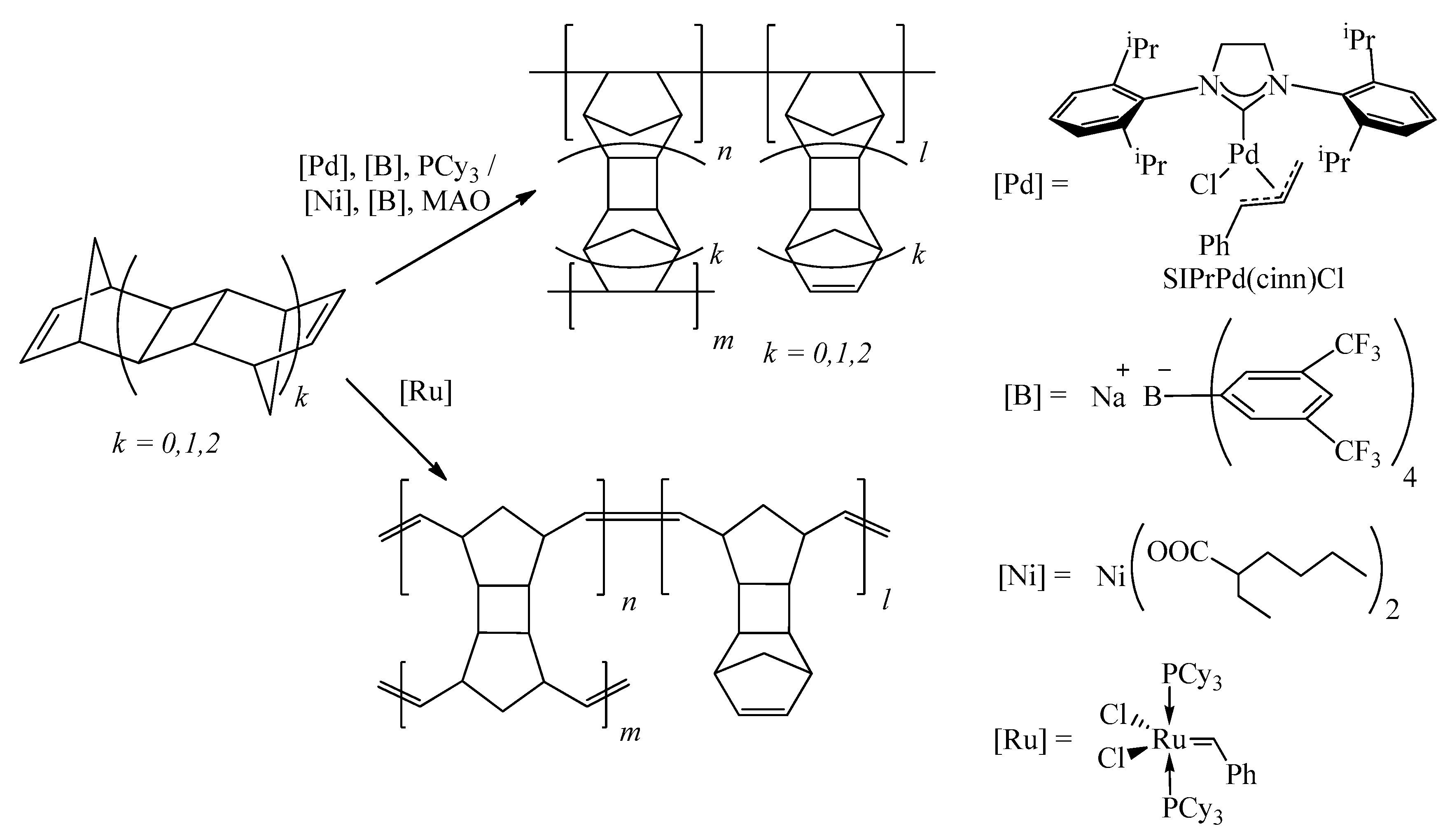
2.3. General Procedure for Addition Homopolymerization Using Pd-Based Catalyst
2.4. Addition Homopolymerization of DNBD Using Ni-Based Catalyst
2.5. Metathesis (ROMP) Homopolymerization of DNBD
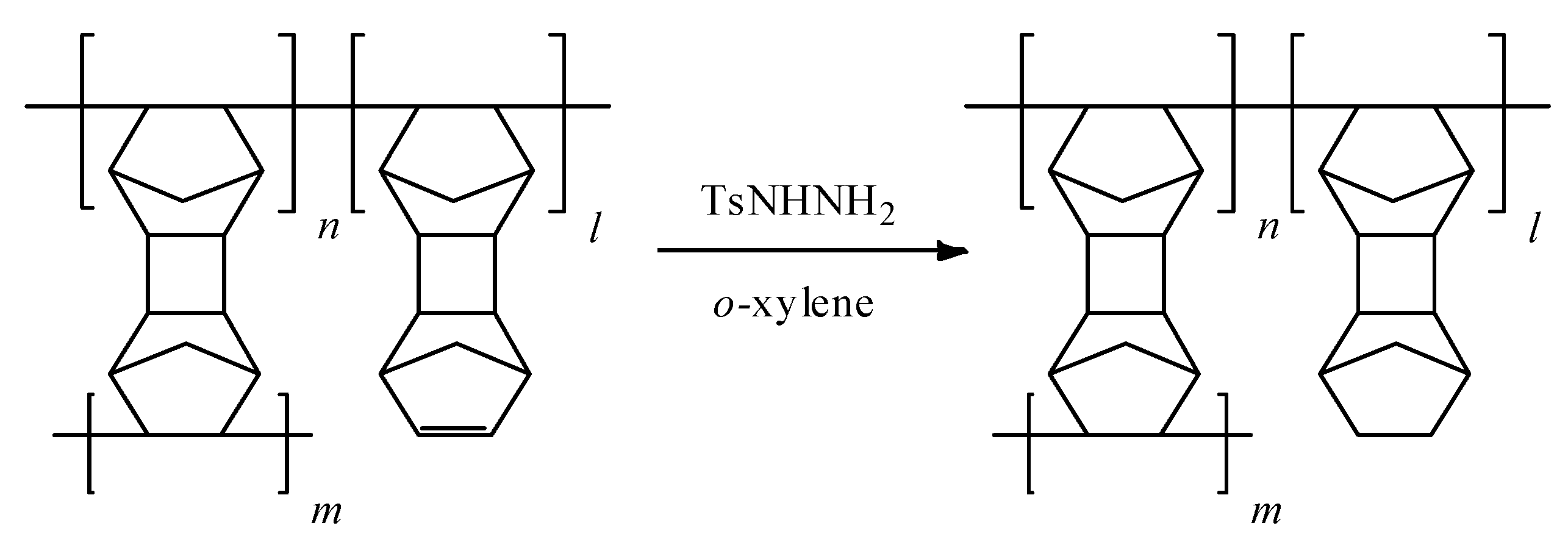
2.6. Hydrogenation of Addition PolyDNBD
3. Results and Discussion
3.1. Synthesis of Polymers
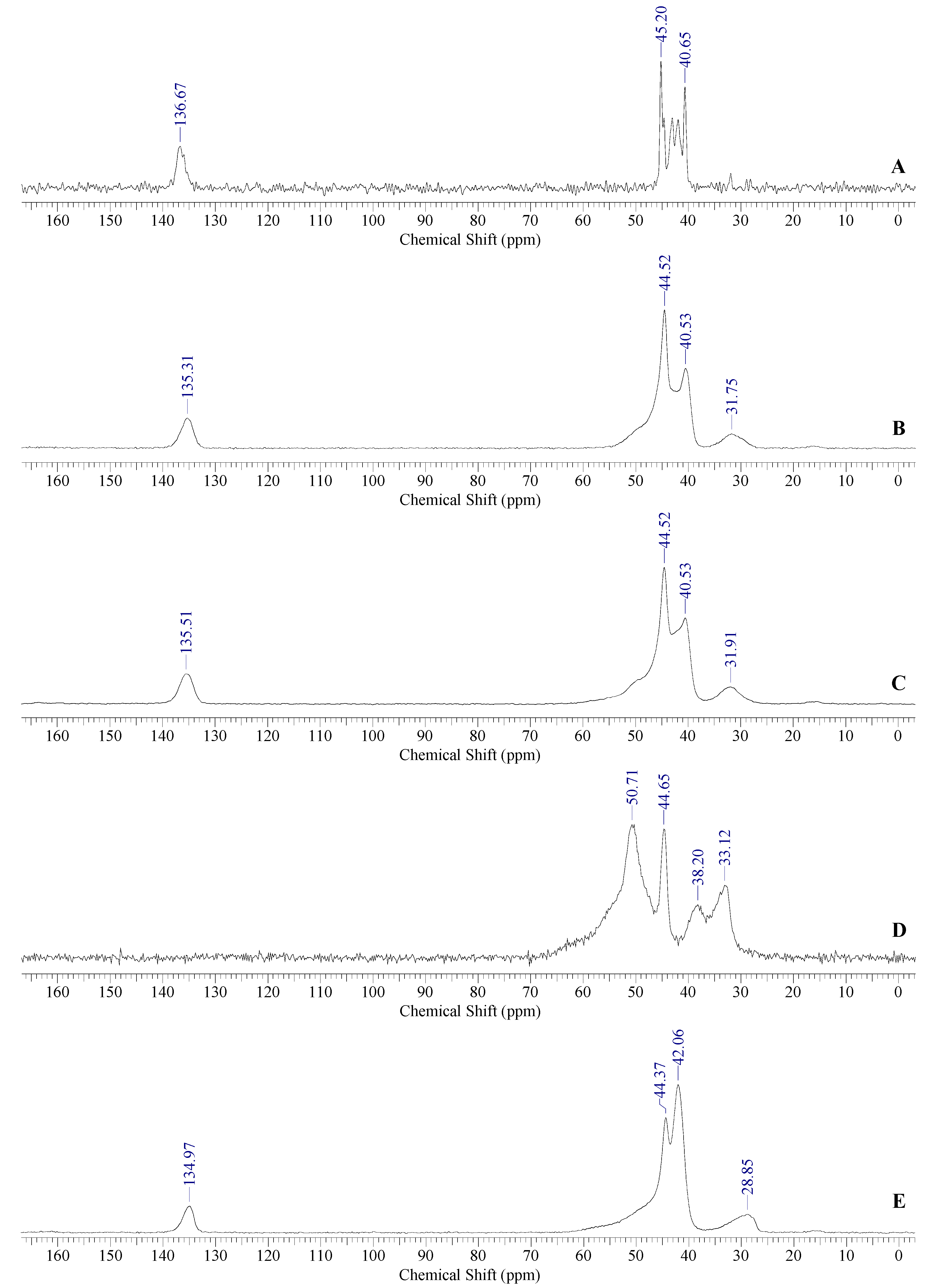
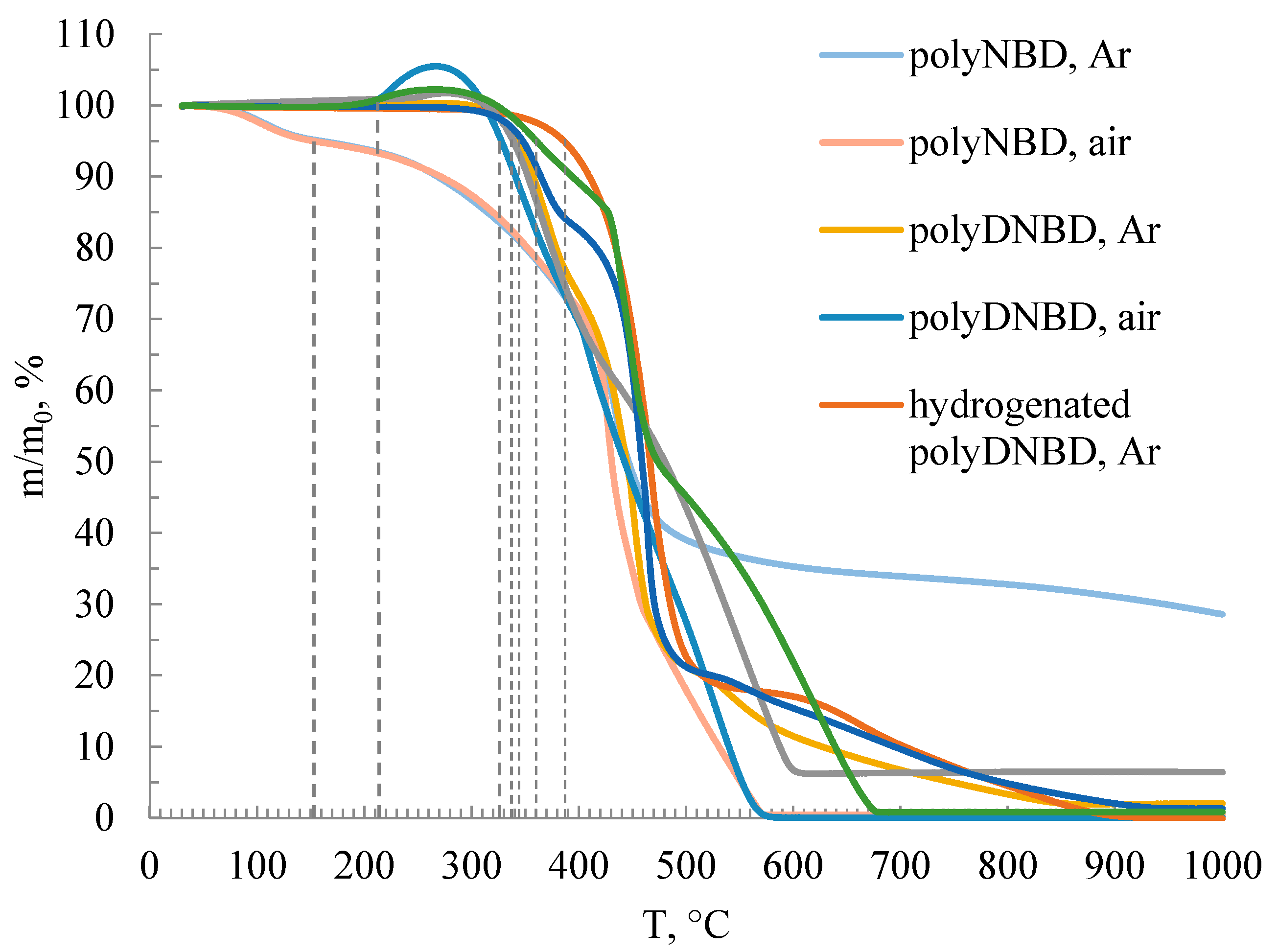
3.2. WAXD and TGA Characterization of the Synthesized Polymers
3.3. Positron Annihilation Lifetime Spectroscopy (PALS) Study
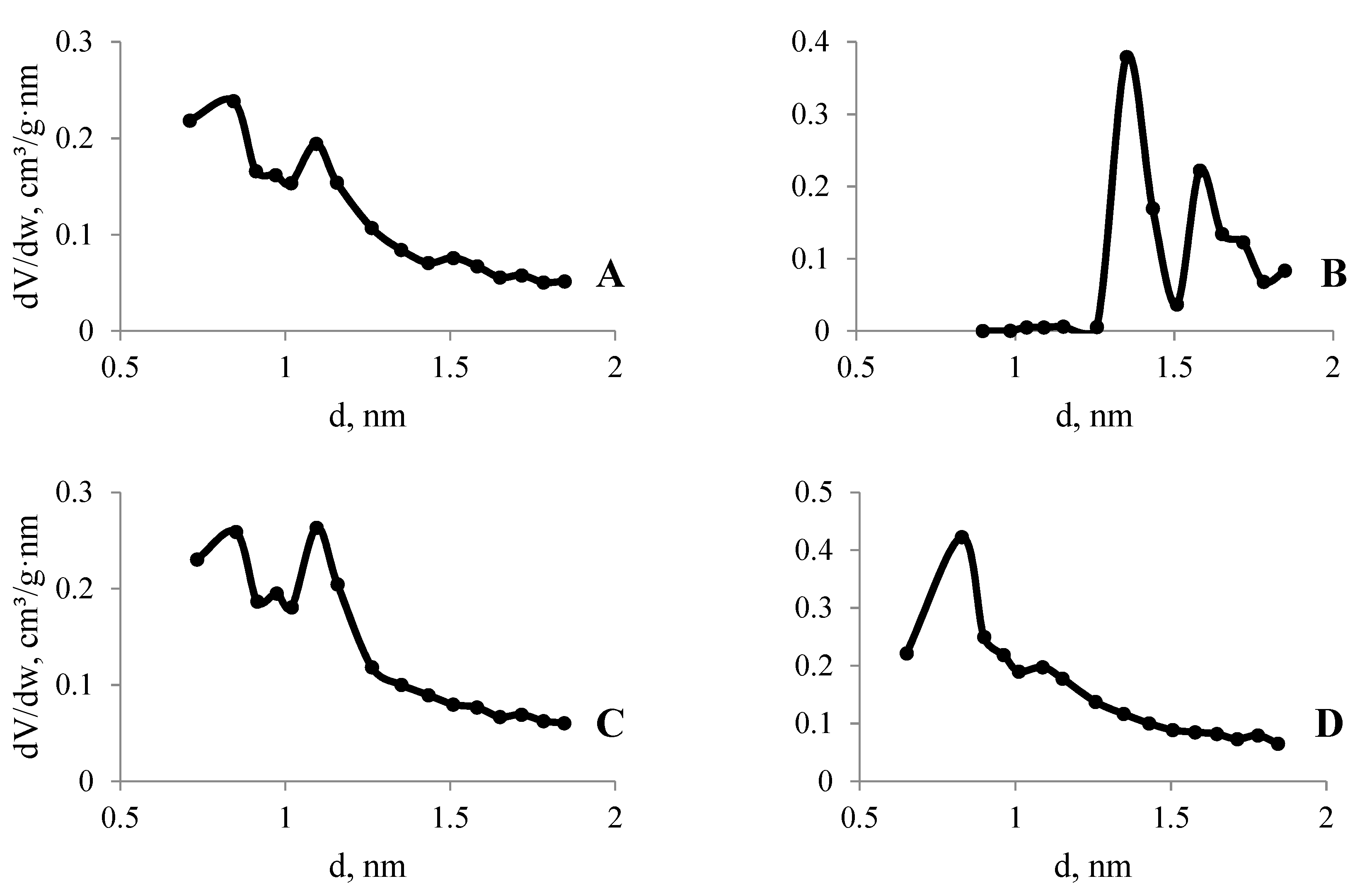
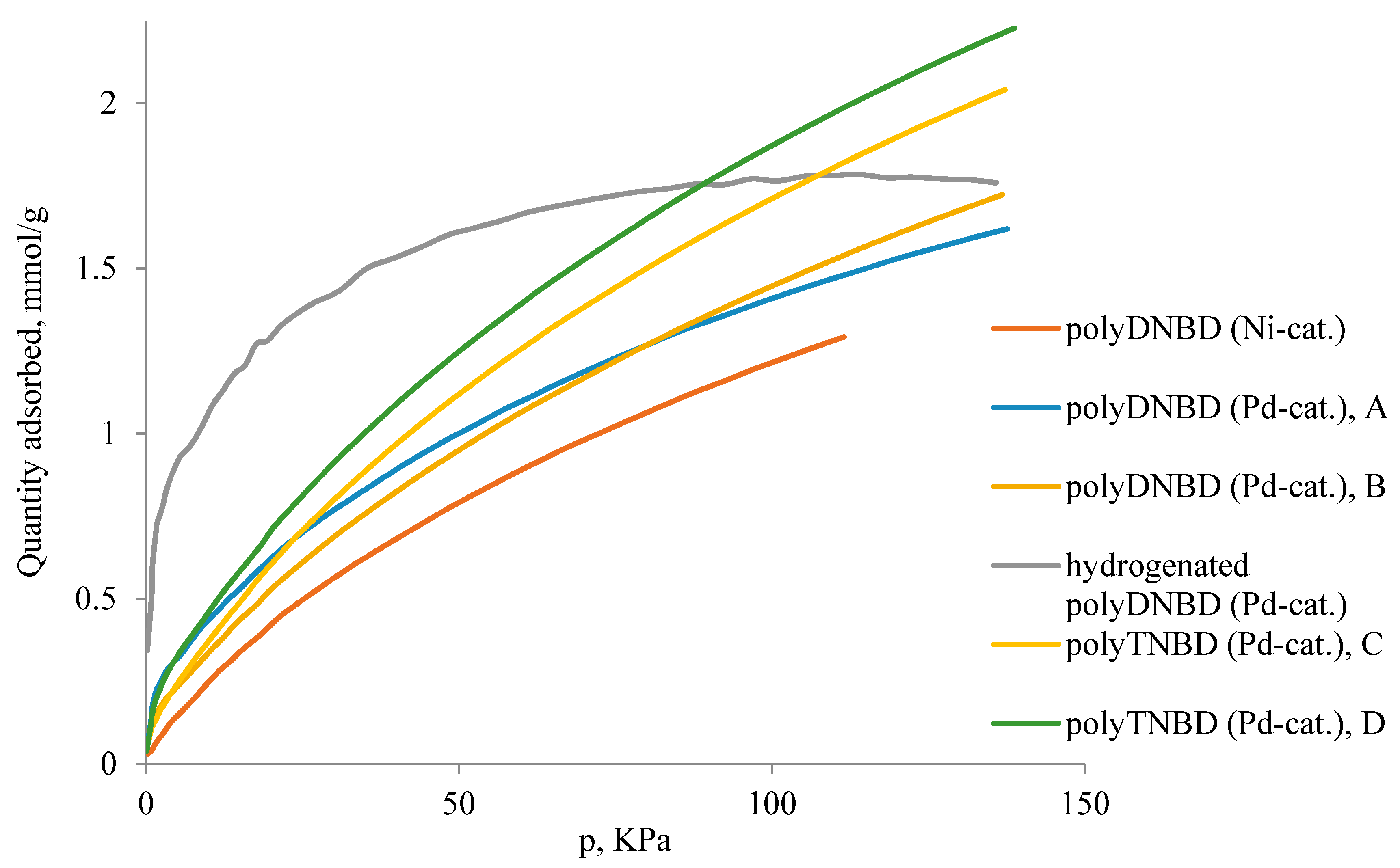
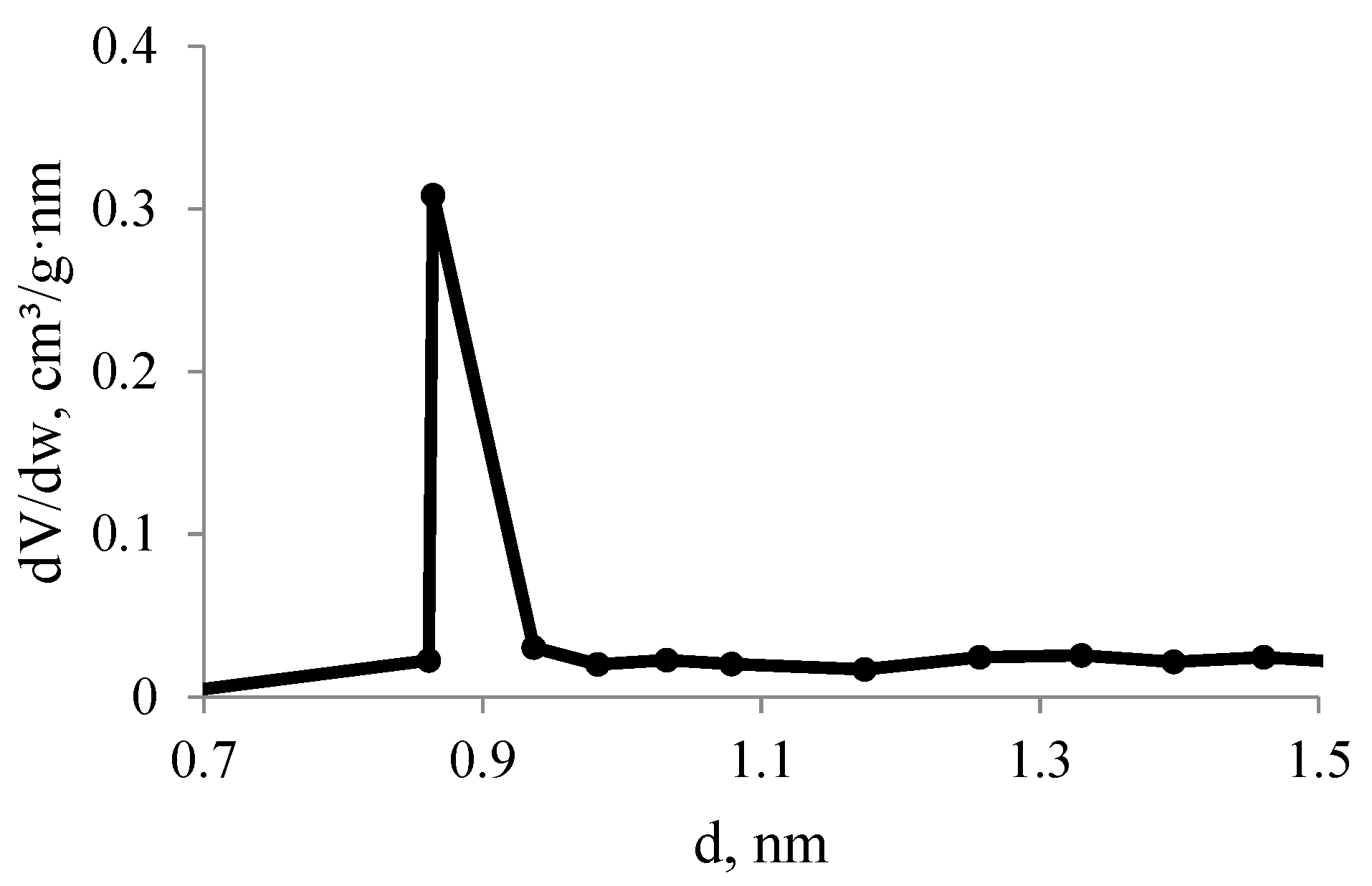
3.4. N2 and CO2 Adsorption Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Morris, R.E.; Wheatley, P.S. Gas Storage in Nanoporous Materials. Angew. Chem. Int. Ed. 2008, 47, 4966–4981. [Google Scholar] [CrossRef] [PubMed]
- Low, Z.-X.; Budd, P.M.; McKeown, N.B.; Patterson, D.A. Gas Permeation Properties, Physical Aging, and Its Mitigation in High Free Volume Glassy Polymers. Chem. Rev. 2018, 118, 5871–5911. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.-C.; Long, J.R.; Yaghi, O.M. Introduction to Metal–Organic Frameworks. Chem. Rev. 2012, 112, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, H.; Yaghi, O.M. Storage of Hydrogen, Methane, and Carbon Dioxide in Highly Porous Covalent Organic Frameworks for Clean Energy Applications. J. Am. Chem. Soc. 2009, 131, 8875–8883. [Google Scholar] [CrossRef] [PubMed]
- Takada, K.; Matsuya, H.; Masuda, T.; Higashimura, T. Gas permeability of polyacetylenes carrying substituents. J. Appl. Polym. Sci. 1985, 30, 1605–1616. [Google Scholar] [CrossRef]
- McKeown, N.B.; Budd, P.M. Polymers of intrinsic microporosity (PIMs): Organic materials for membrane separations, heterogeneous catalysis and hydrogen storage. Chem. Soc. Rev. 2006, 35, 675–683. [Google Scholar] [CrossRef]
- Park, H.B.; Jung, C.H.; Lee, Y.M.; Hill, A.J.; Pas, S.J.; Mudie, S.T.; Wagner, E.V.; Freeman, B.D.; Cookson, D.J. Polymers with Cavities Tuned for Fast Selective Transport of Small Molecules and Ions. Science 2007, 318, 254–258. [Google Scholar] [CrossRef]
- Li, G.; Liu, Q.; Xia, B.; Huang, J.; Li, S.; Guan, Y.; Zhou, H.; Liao, B.; Zhou, Z.; Liu, B. Synthesis of stable metal-containing porous organic polymers for gas storage. Eur. Polym. J. 2017, 91, 242–247. [Google Scholar] [CrossRef]
- Chapala, P.P.; Bermeshev, M.V.; Gavrilova, N.N. Microporous structure of highly permeable additive silicon-containing polytricyclononenes. Polym. Sci. Ser. A 2017, 59, 143–148. [Google Scholar] [CrossRef]
- Wozniak, A.I.; Bermesheva, E.V.; Gavrilova, N.N.; Ilyasov, I.R.; Nechaev, M.S.; Asachenko, A.F.; Topchiy, M.A.; Gribanov, P.S.; Bermeshev, M.V. Addition Homo- and Copolymerizations of Dicyclopentadiene and 5-n-Hexylnorbornene in the Presence of Pd-N-Heterocyclic Carbene Complexes. Macromol. Chem. Phys. 2018, 1800323. [Google Scholar] [CrossRef]
- Du, N.; Dal-Cin, M.M.; Robertson, G.P.; Guiver, M.D. Decarboxylation-Induced Cross-Linking of Polymers of Intrinsic Microporosity (PIMs) for Membrane Gas Separation. Macromolecules 2012, 45, 5134–5139. [Google Scholar] [CrossRef]
- Liu, Q.; Li, G.; Tang, Z.; Chen, L.; Liao, B.; Ou, B.; Zhou, Z.; Zhou, H. Design and synthesis of conjugated polymers of tunable pore size distribution. Mater. Chem. Phys. 2017, 186, 11–18. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Wood, C.D.; Bradshaw, D.; Rosseinsky, M.J.; Cooper, A.I. Hydrogen adsorption in microporous hypercrosslinked polymers. Chem. Commun. 2006, 2670–2672. [Google Scholar] [CrossRef] [PubMed]
- Ivin, K.J.; Mol, J.C. 11—Ring-Opening Metathesis Polymerization: General Aspects. In Olefin Metathesis and Metathesis Polymerization (2); Mol, K.J.I.C., Ed.; Academic Press: London, UK, 1997; pp. 224–259. [Google Scholar]
- Blank, F.; Janiak, C. Metal catalysts for the vinyl/addition polymerization of norbornene. Coord. Chem. Rev. 2009, 253, 827–861. [Google Scholar] [CrossRef]
- Bermeshev, M.V.; Chapala, P.P. Addition polymerization of functionalized norbornenes as a powerful tool for assembling molecular moieties of new polymers with versatile properties. Prog. Polym. Sci. 2018, 84, 1–46. [Google Scholar] [CrossRef]
- Gaylord, N.G.; Deshpande, A.B.; Mandal, B.M.; Martan, M. Poly-2,3- and 2,7-Bicyclo[2.2.1]hept-2-enes: Preparation and Structures of Polynorbornenes. J. Macromol. Sci. A 1977, 11, 1053–1070. [Google Scholar] [CrossRef]
- Bermeshev, M.V.; Bulgakov, B.A.; Genaev, A.M.; Kostina, J.V.; Bondarenko, G.N.; Finkelshtein, E.S. Cationic Polymerization of Norbornene Derivatives in the Presence of Boranes. Macromolecules 2014, 47, 5470–5483. [Google Scholar] [CrossRef]
- Finkelshtein, E.; Gringolts, M.; Bermeshev, M.; Chapala, P.; Rogan, Y. Polynorbornenes. In Membrane Materials for Gas and Vapor Separation; Yampolskii, Y., Finkelshtein, E., Eds.; Wiley: Chichester, UK, 2017; pp. 143–221. [Google Scholar]
- Finkelshtein, E.S.; Bermeshev, M.V.; Gringolts, M.L.; Starannikova, L.E.; Yampolskii, Y.P. Substituted polynorbornenes as promising materials for gas separation membranes. Russ. Chem. Rev. 2011, 80, 341. [Google Scholar] [CrossRef]
- Chapala, P.P.; Bermeshev, M.V.; Starannikova, L.E.; Belov, N.A.; Ryzhikh, V.E.; Shantarovich, V.P.; Lakhtin, V.G.; Gavrilova, N.N.; Yampolskii, Y.P.; Finkelshtein, E.S. A Novel, Highly Gas-Permeable Polymer Representing a New Class of Silicon-Containing Polynorbornens As Efficient Membrane Materials. Macromolecules 2015, 48, 8055–8061. [Google Scholar] [CrossRef]
- Alentiev, D.A.; Bermeshev, M.V.; Starannikova, L.E.; Bermesheva, E.V.; Shantarovich, V.P.; Bekeshev, V.G.; Yampolskii, Y.P.; Finkelshtein, E.S. Stereoselective synthesis and polymerization of Exo-5-trimethylsilylnorbornene. J. Polym. Sci. Part A Polym. Chem. 2018, 56, 1234–1248. [Google Scholar] [CrossRef]
- Alentiev, D.A.; Egorova, E.S.; Bermeshev, M.V.; Starannikova, L.E.; Topchiy, M.A.; Asachenko, A.F.; Gribanov, P.S.; Nechaev, M.S.; Yampolskii, Y.P.; Finkelshtein, E.S. Janus tricyclononene polymers bearing tri(n-alkoxy)silyl side groups for membrane gas separation. J. Mater. Chem. A 2018, 6, 19393–19408. [Google Scholar] [CrossRef]
- Viciu, M.S.; Navarro, O.; Germaneau, R.F.; Kelly, R.A.; Sommer, W.; Marion, N.; Stevens, E.D.; Cavallo, L.; Nolan, S.P. Synthetic and Structural Studies of (NHC)Pd(allyl)Cl Complexes (NHC = N-heterocyclic carbene). Organometallics 2004, 23, 1629–1635. [Google Scholar] [CrossRef]
- Marion, N.; Navarro, O.; Mei, J.; Stevens, E.D.; Scott, N.M.; Nolan, S.P. Modified (NHC)Pd(allyl)Cl (NHC = N-Heterocyclic Carbene) Complexes for Room-Temperature Suzuki−Miyaura and Buchwald−Hartwig Reactions. J. Am. Chem. Soc. 2006, 128, 4101–4111. [Google Scholar] [CrossRef] [PubMed]
- Dzhemilev, U.M.; Popod’ko, N.R.; Kozlova, E.V. Metallokomleksny kataliz v organicheskom sinteze. Alitsiklicheskiye soedineniya; Khimiya: Moscow, Russia, 1999; p. 340. [Google Scholar]
- Lisitsin, D.M.; Poznyak, T.I.; Razumovskii, S.D. Mathematical modeling of the reaction of ozone with some organic compounds in a continuous operation bubbling reactor. Kinet. Katalis 1976, 17, 1049–1056. [Google Scholar]
- Tao, S.J. Positronium Annihilation in Molecular Substances. J. Chem. Phys. 1972, 56, 5499–5510. [Google Scholar] [CrossRef]
- Eldrup, M.; Lightbody, D.; Sherwood, J.N. The temperature dependence of positron lifetimes in solid pivalic acid. Chem. Phys. 1981, 63, 51–58. [Google Scholar] [CrossRef]
- Alentiev, D.A.; Korchagina, S.A.; Finkelshtein, E.S.; Nechaev, M.S.; Asachenko, A.F.; Topchiy, M.A.; Gribanov, P.S.; Bermeshev, M.V. Addition homo- and copolymerization of 3-triethoxysilyltricyclo[4.2.1.02,5]non-7-ene. Russ. Chem. Bull. 2018, 67, 121–126. [Google Scholar] [CrossRef]
- Yevlampieva, N.; Bermeshev, M.; Vezo, O.; Chapala, P.; Il’yasova, Y. Metathesis and additive poly(tricyclononenes) with geminal trimethylsilyl side groups: Chain rigidity, molecular and thin film properties. J. Polym. Res. 2018, 25, 162. [Google Scholar] [CrossRef]
- Yevlampieva, N.P.; Bermeshev, M.V.; Komolkin, A.V.; Vezo, O.S.; Chapala, P.P.; Il’yasova, Y.V. The equilibrium and kinetic rigidity of additive poly(trimethylsilyltricyclononenes) with one and two Si(CH3)3 groups in monomer unit. Polym. Sci. Ser. A 2017, 59, 473–482. [Google Scholar] [CrossRef]
- Budd, P.M.; Ghanem, B.S.; Makhseed, S.; McKeown, N.B.; Msayib, K.J.; Tattershall, C.E. Polymers of intrinsic microporosity (PIMs): Robust, solution-processable, organic nanoporous materials. Chem. Commun. 2004, 230–231. [Google Scholar] [CrossRef]
- Budd, P.M.; Elabas, E.S.; Ghanem, B.S.; Makhseed, S.; McKeown, N.B.; Msayib, K.J.; Tattershall, C.E.; Wang, D. Solution-Processed, Organophilic Membrane Derived from a Polymer of Intrinsic Microporosity. Adv. Mater. 2004, 16, 456–459. [Google Scholar] [CrossRef]
- Thomas, S.; Pinnau, I.; Du, N.; Guiver, M.D. Pure- and mixed-gas permeation properties of a microporous spirobisindane-based ladder polymer (PIM-1). J. Membr. Sci. 2009, 333, 125–131. [Google Scholar] [CrossRef]


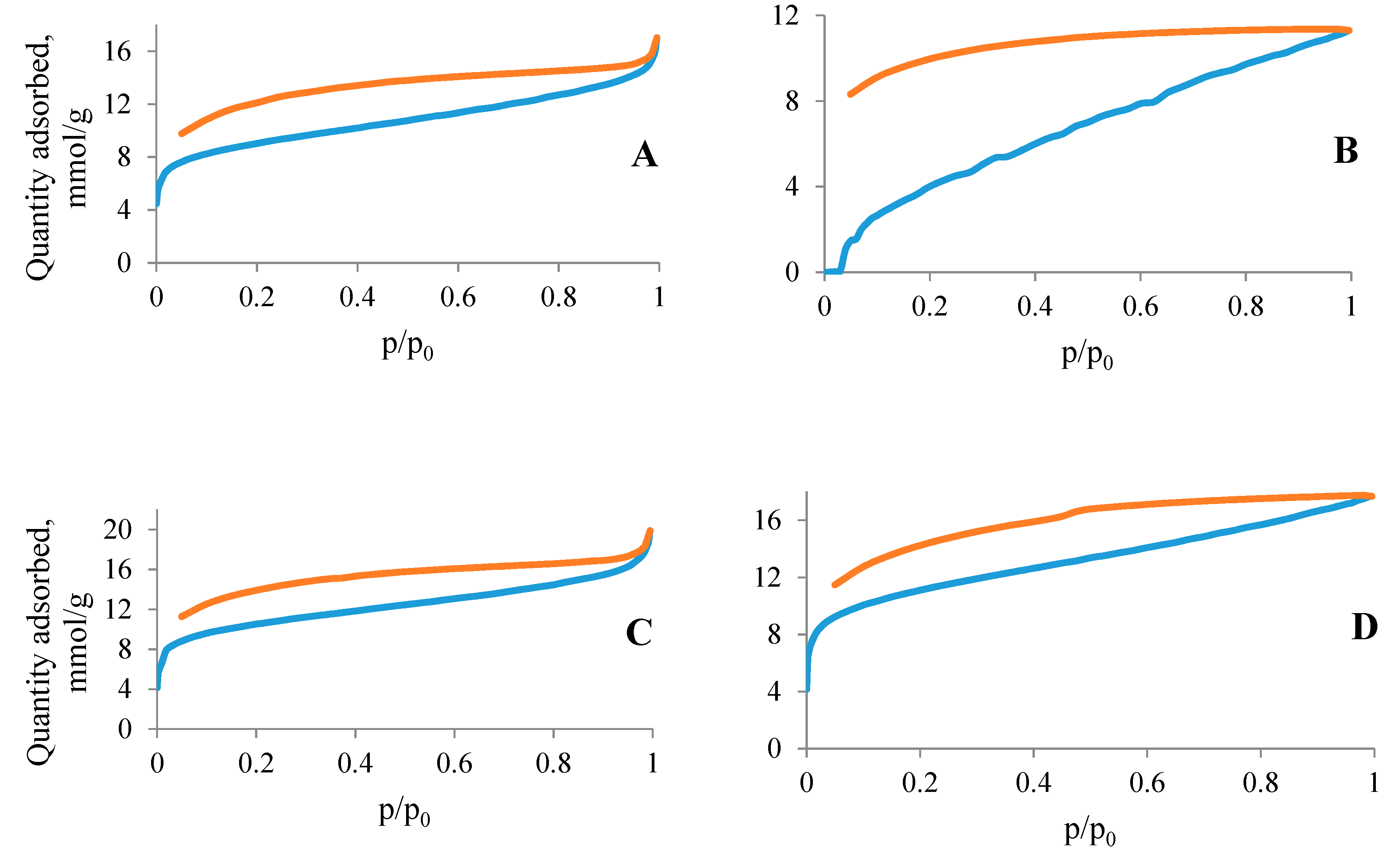
| Monomer | Synthesis Conditions | Yield, % | Double Bonds Content | ||
|---|---|---|---|---|---|
| Catalyst 1 | C, M 2 | Solvent | |||
| Metathesis polymerization | |||||
| DNBD | Ru | 0.26 | 1,2-dichloroethane | 100 | 0.34 3 |
| Addition polymerization | |||||
| NBD | Pd | 0.20 | 1,2-dichloroethane | 53 | 0.067 4 |
| DNBD | Pd | 0.09 | 1,2-dichloroethane | 100 | 0.21 4 |
| DNBD | Pd | 0.35 | 1,2-dichloroethane | 100 | - |
| DNBD | Pd | 0.38 | 1,2-dichloroethane | 100 | 0.60 3 |
| DNBD | Ni | 1.4 | toluene | 100 | 0.46 3 |
| TNBD | Pd | 0.20 | 1,2-dichloroethane + toluene (22 vol.%) | 96 | 0.91 4 |
| TNBD | Pd | 0.04 | 1,2-dichloroethane + toluene (4.4 vol.%) | 84 | - |
| Polymer | τ3, ns | I3, % | τ4, ns | I4, % | R3, Å | R4, Å | |
|---|---|---|---|---|---|---|---|
 | [21] | 3.8 | 11 | 11.7 | 36 | 4.2 | 7.3 |
 | [22] | 2.4 | 12 | 6.3 | 36 | 3.3 | 5.4 |
 (Pd-cat.) | 1.7 | 35 | 8.0 | 4.8 | 2.5 | 6.2 | |
| Polymer | C, M 1 | SBET, m2/g | Total Pore Volume, cm3/g | t-Plot Micropore Area, m2/g | t-Plot External Surface Area, m2/g | t-Plot Micropore Volume, cm3/g | BJH-Adsorption Mesopore Volume, cm3/g | HK-Model 2 | DR-Model 3 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Vmax, cm3/g | d, nm | S, m2/g | Vmon, cm3/g | ||||||||
| addition polyNBD (Pd-cat.) | 0.20 | <10 | - | - | - | - | - | - | - | - | - |
| metathesis polyDNBD | 0.26 | <10 | - | - | - | - | - | - | - | - | - |
| addition polyDNBD (Ni-cat.) | 1.4 | 420 | 0.39 | - | 631 | - | 0.27 | 0.097 | 1.4 | 1245 | 286 |
| addition polyDNBD (Pd-cat.) | 0.09 | 827 | 0.59 | 362 | 465 | 0.15 | 0.24 | 0.34 | 0.77 | 942 | 216 |
| 0.35 | 736 | 0.54 | 355 | 381 | 0.14 | 0.24 | 0.29 | 0.73 | 820 | 288 | |
| 0.38 | 730 | 0.59 | 348 | 383 | 0.15 | 0.28 | 0.29 | 0.71 | 824 | 189 | |
| hydrogenated addition polyDNBD (Pd-cat.) | 0.38 | 814 | 0.72 | 378 | 437 | 0.16 | 0.35 | 0.32 | 0.73 | 953 | 219 |
| addition polyTNBD (Pd-cat.) | 0.20 | 969 | 0.79 | 430 | 538 | 0.18 | 0.39 | 0.38 | 0.72 | 1072 | 246 |
| PIM-1 [33,34] | - | 850–860 | 0.78 | - | - | - | - | - | - | - | - |
| TR-1-450 [7] | - | 510 | - | - | - | - | - | - | - | - | - |
| PTMSP [35] | - | 780 | - | - | - | - | - | - | - | - | - |
| Polymer | C1, M | SL2, m2/g | CO2 Uptake, mmol/g |
|---|---|---|---|
| addition polyDNBD (Pd-cat.) | 0.09 | 314 | 1.46 |
| 0.38 | 254 | 1.42 | |
| addition polyDNBD (Ni-cat.) | 1.4 | 276 | 1.22 |
| hydrogenated addition polyDNBD (Pd-cat.) | 0.38 | 202 | 1.77 |
| addition polyTNBD (Pd-cat.) | 0.20 | 403 | 1.89 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alentiev, D.A.; Dzhaparidze, D.M.; Gavrilova, N.N.; Shantarovich, V.P.; Kiseleva, E.V.; Topchiy, M.A.; Asachenko, A.F.; Gribanov, P.S.; Nechaev, M.S.; Legkov, S.A.; et al. Microporous Materials Based on Norbornadiene-Based Cross-Linked Polymers. Polymers 2018, 10, 1382. https://doi.org/10.3390/polym10121382
Alentiev DA, Dzhaparidze DM, Gavrilova NN, Shantarovich VP, Kiseleva EV, Topchiy MA, Asachenko AF, Gribanov PS, Nechaev MS, Legkov SA, et al. Microporous Materials Based on Norbornadiene-Based Cross-Linked Polymers. Polymers. 2018; 10(12):1382. https://doi.org/10.3390/polym10121382
Chicago/Turabian StyleAlentiev, Dmitry A., Dariya M. Dzhaparidze, Natalia N. Gavrilova, Victor P. Shantarovich, Elena V. Kiseleva, Maxim A. Topchiy, Andrey F. Asachenko, Pavel S. Gribanov, Mikhail S. Nechaev, Sergey A. Legkov, and et al. 2018. "Microporous Materials Based on Norbornadiene-Based Cross-Linked Polymers" Polymers 10, no. 12: 1382. https://doi.org/10.3390/polym10121382
APA StyleAlentiev, D. A., Dzhaparidze, D. M., Gavrilova, N. N., Shantarovich, V. P., Kiseleva, E. V., Topchiy, M. A., Asachenko, A. F., Gribanov, P. S., Nechaev, M. S., Legkov, S. A., Bondarenko, G. N., & Bermeshev, M. V. (2018). Microporous Materials Based on Norbornadiene-Based Cross-Linked Polymers. Polymers, 10(12), 1382. https://doi.org/10.3390/polym10121382