Improvement of Adhesion Properties of Polyamide 6 and Polyoxymethylene-Copolymer by Atmospheric Cold Plasma Treatment
Abstract
1. Introduction
2. Material and Methods
2.1. Materials and Sample Preparation
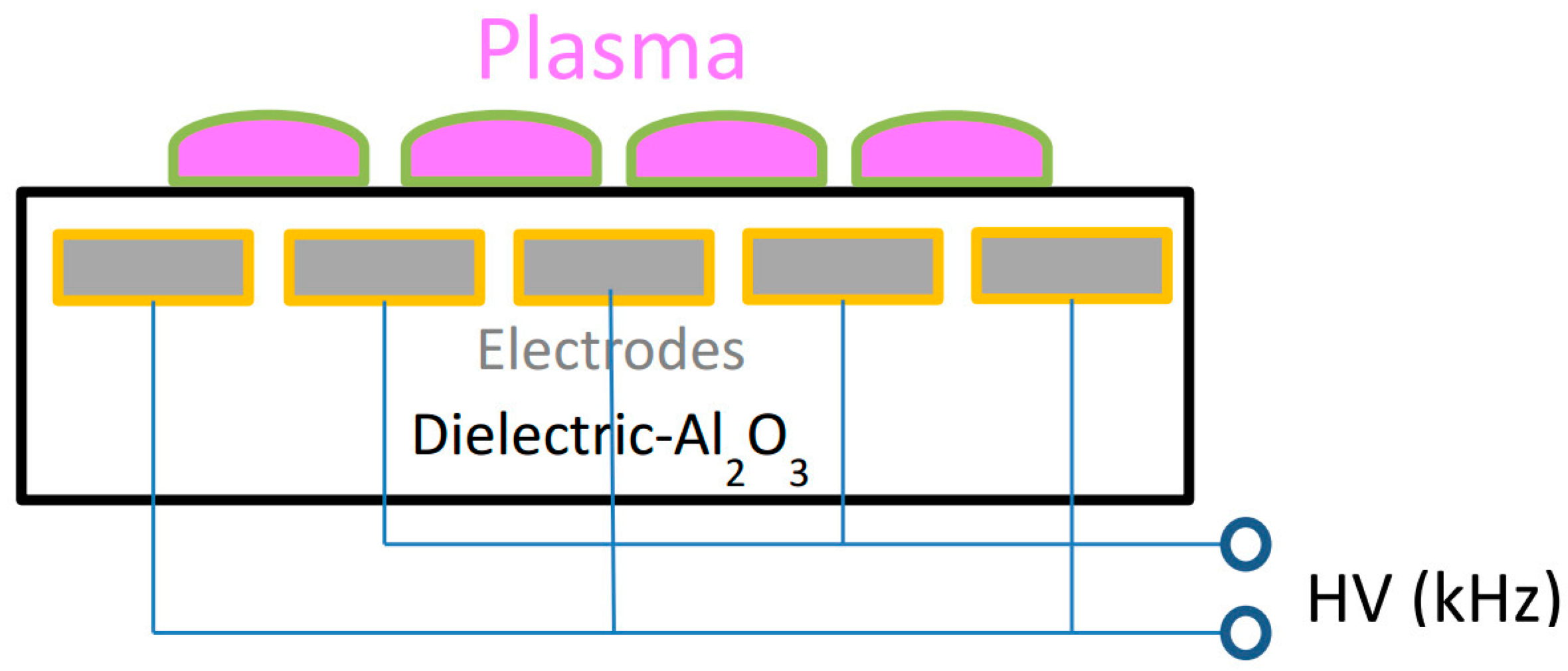
2.2. Plasma Treatment
2.3. Adhesive Testing
2.4. Surface Characterization
3. Results and Discussion
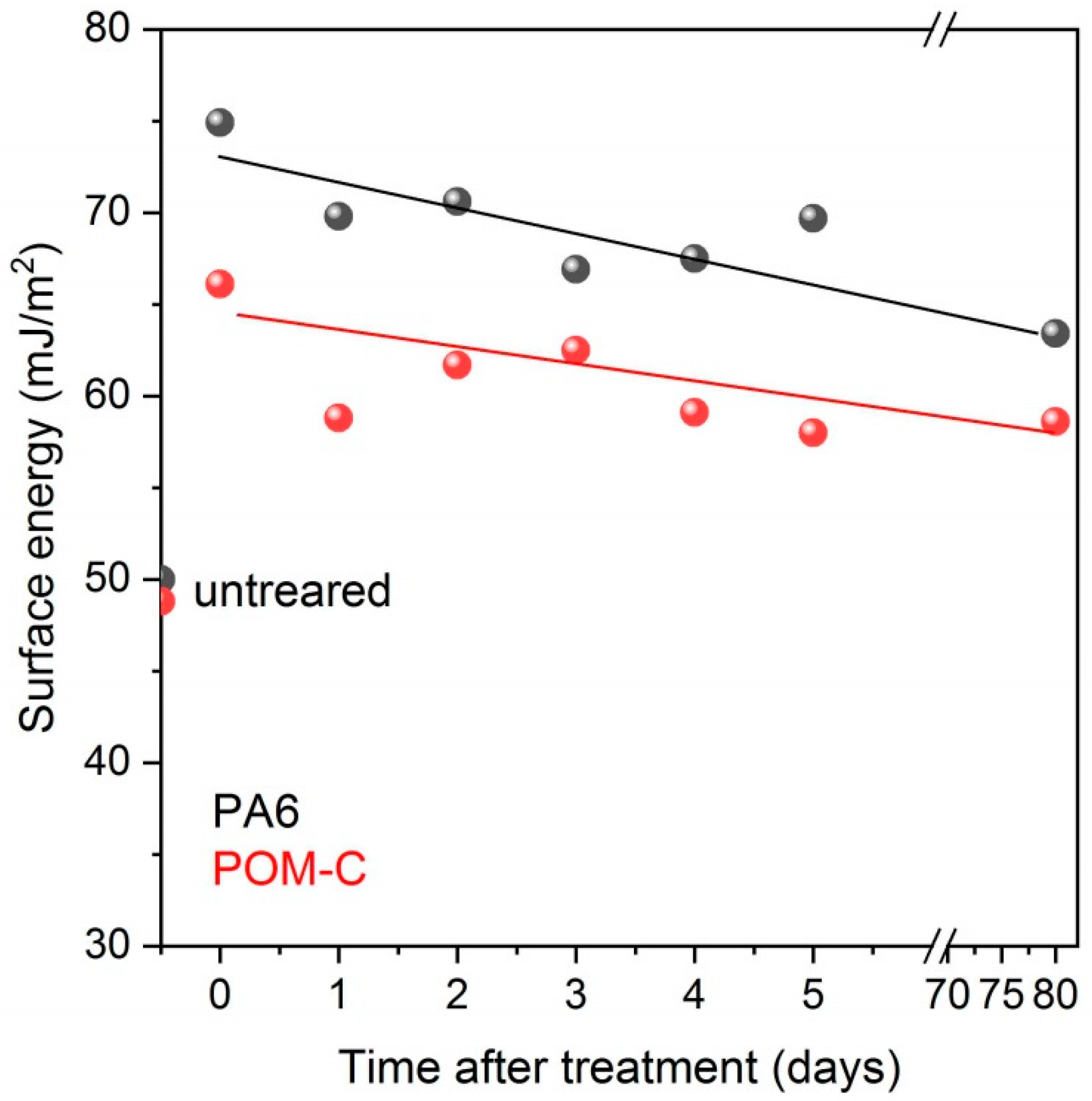
3.1. Contact Angle Measurements
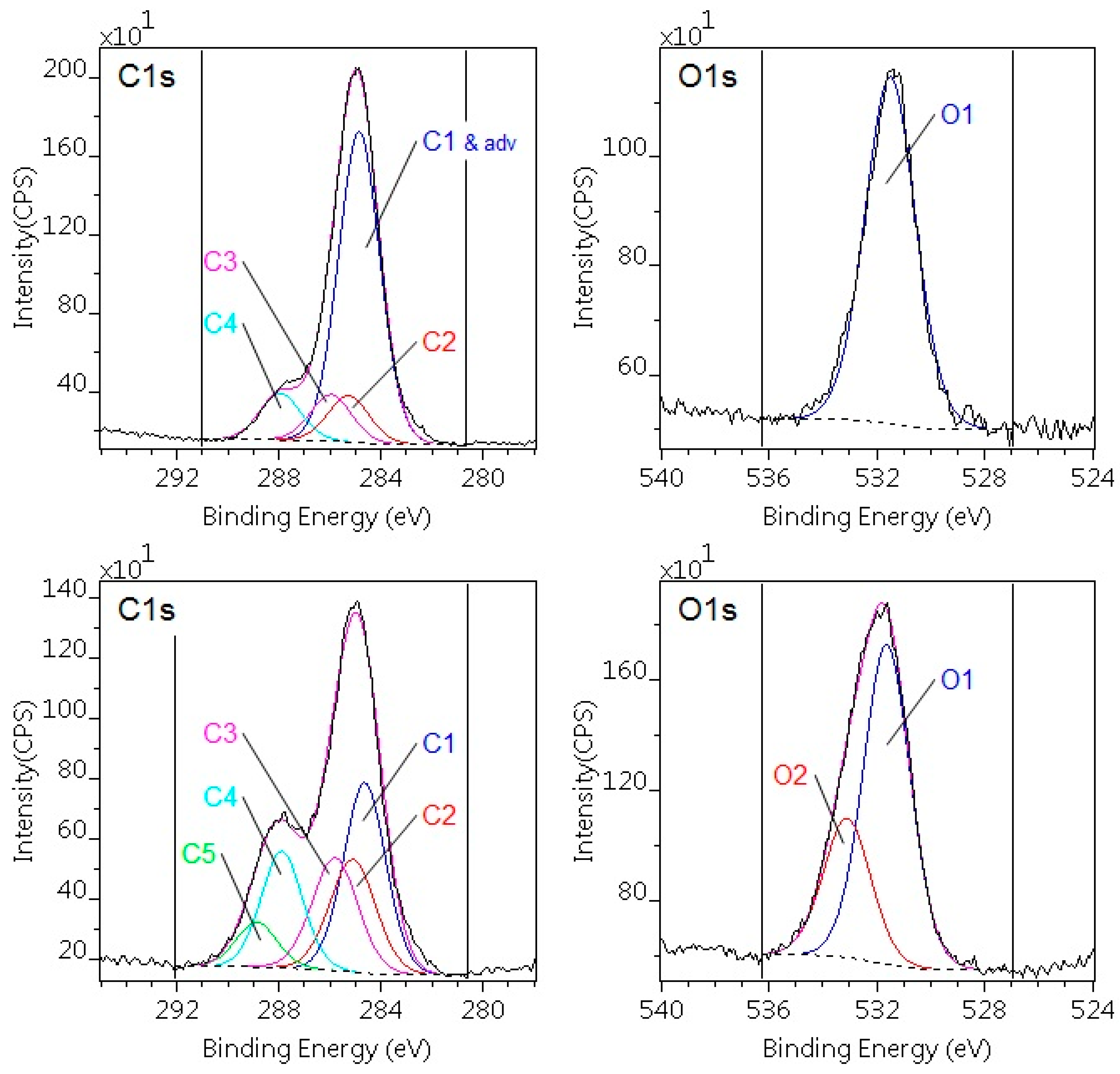
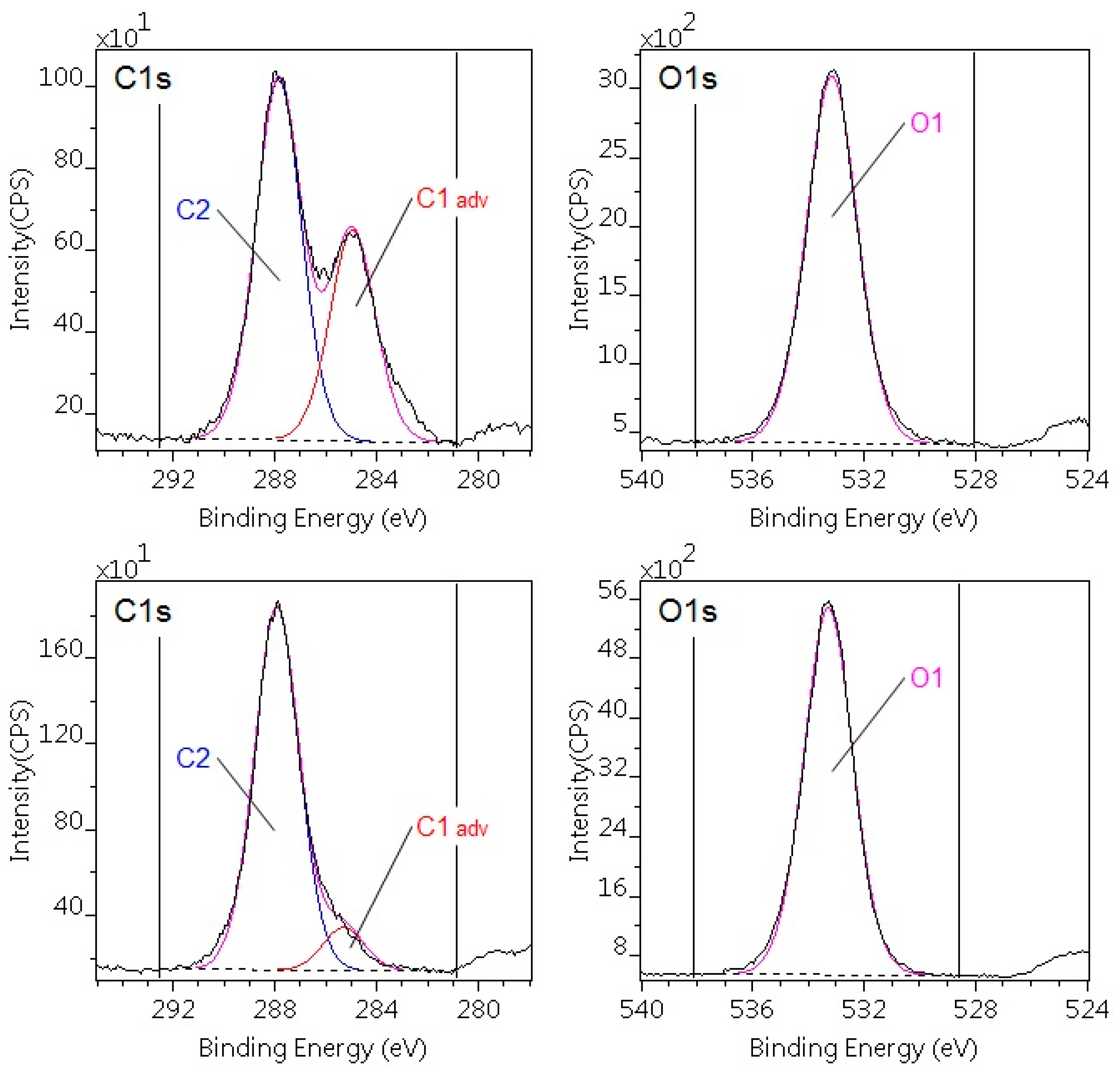
3.2. Analysis of Surface Chemistry after Plasma Treatment
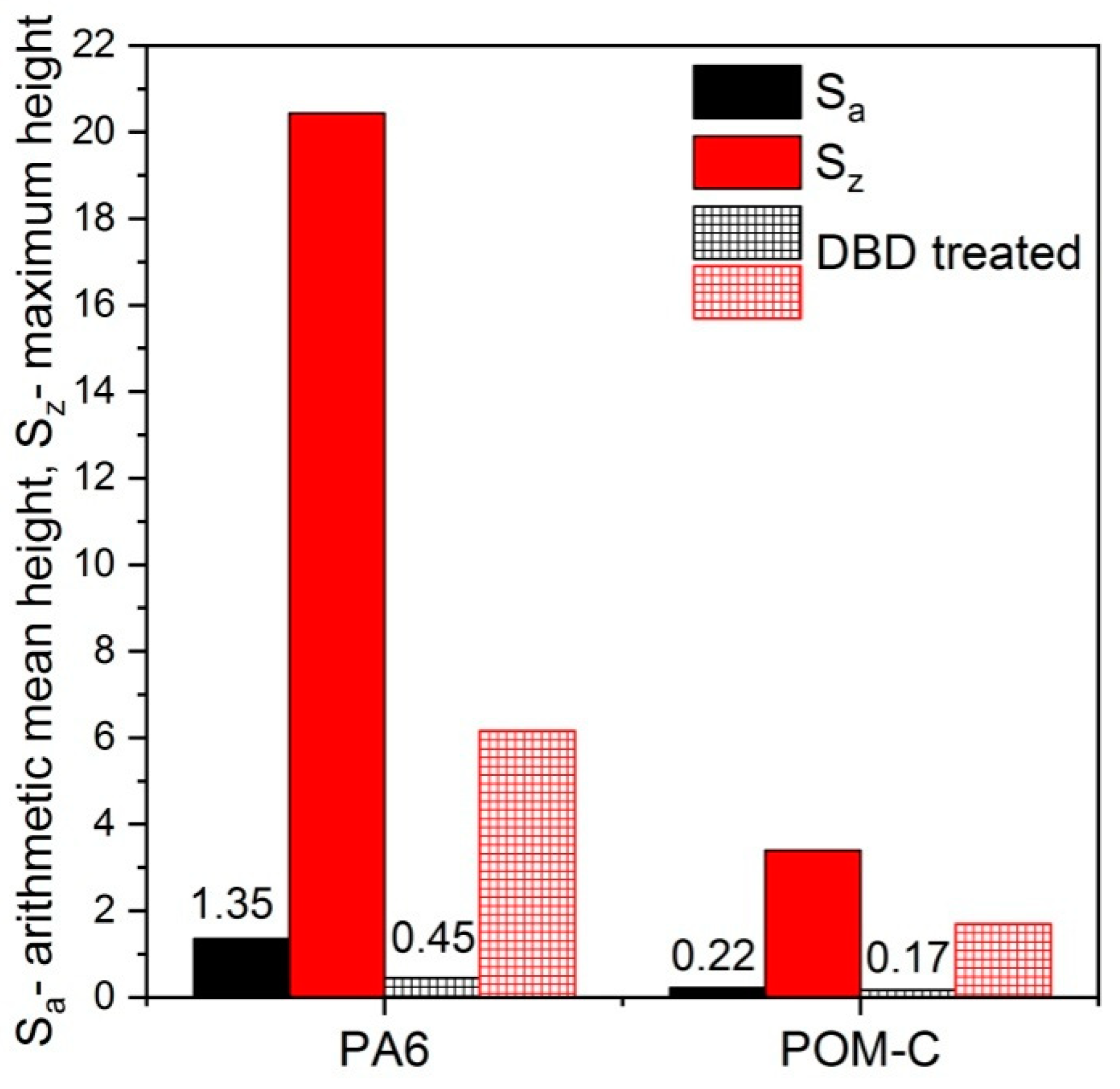
3.3. Surface Topography
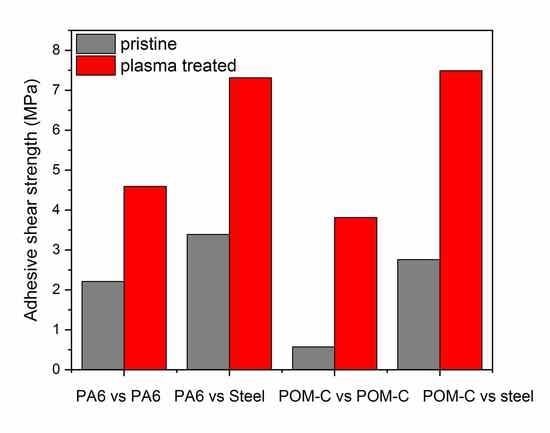
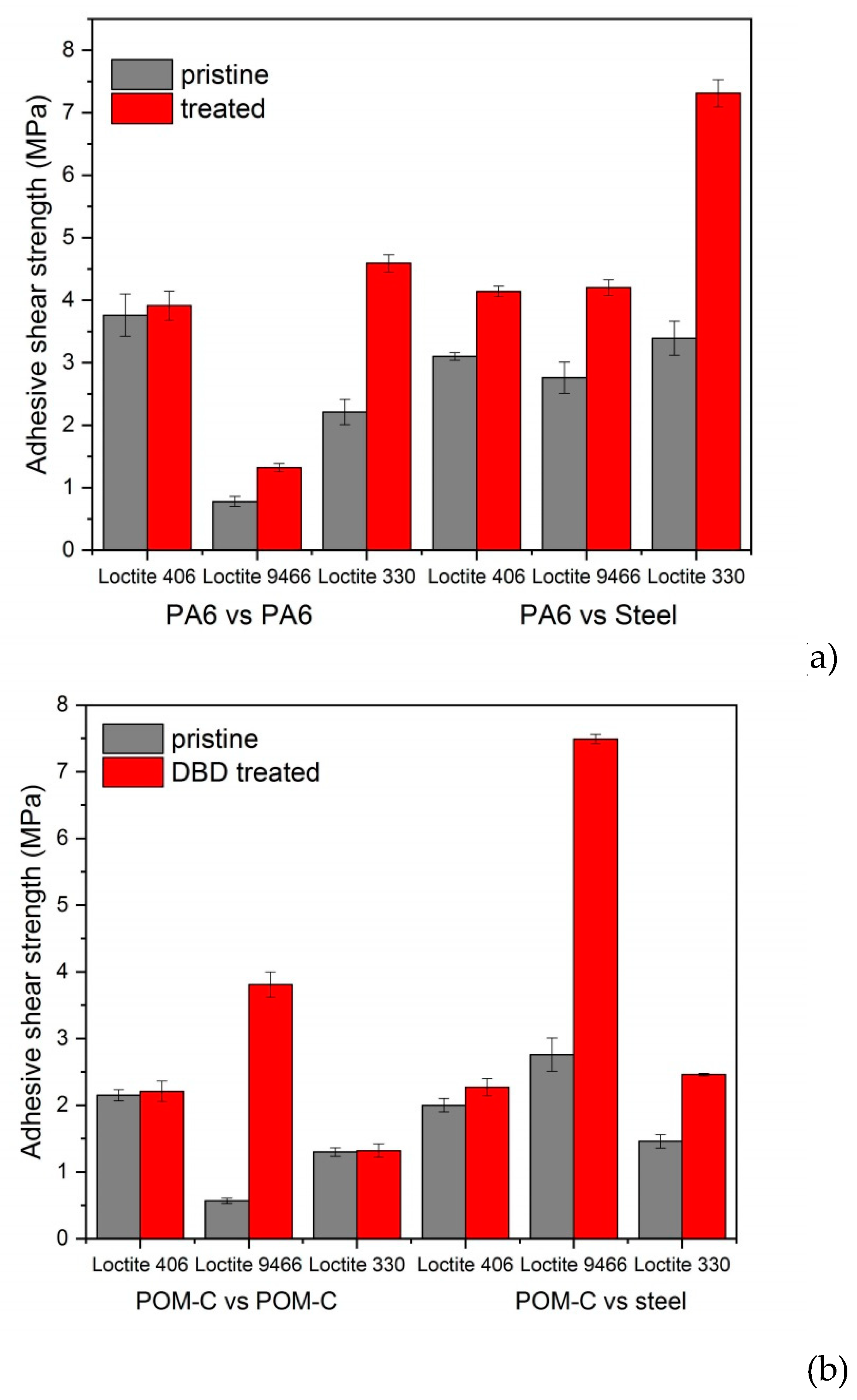
3.4. Adhesive Tests
4. Summary
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Baldan, A. Adhesively-bonded joints in metallic alloys, polymers and composite materials: Mechanical and environmental durability performance. J. Mater. Sci. 2004, 39, 4729–4797. [Google Scholar] [CrossRef]
- Awajaa, F.; Gilbert, M.; Kellya, G.; Foxa, B.; Pigram, P.J. Adhesion of polymers. Prog. Polym. Sci. 2009, 34, 948–968. [Google Scholar] [CrossRef]
- Mandolfino, C.; Lertora, E.; Gambaro, C.; Bruno, M. Improving adhesion performance of polyethylene surfaces by cold plasma treatment. Meccanica 2014, 49, 2299–2306. [Google Scholar] [CrossRef]
- Horakova, M.; Spatenka, P.; Hladik, J.; Hornik, J.; Steidl, J.; Polachova, A. Investigation of adhesion between metal and plasma-modified polyethylene. Plasma Procesess Polym. 2011, 8, 983–988. [Google Scholar] [CrossRef]
- Abdul Samad, M.; Satyanarayana, N.; Sinha, S.K. Tribology of UHMWPE film on air-plasma treated tool steel and the effect of PFPE overcoat. Surf. Coat. Technol. 2010, 204, 1330–1338. [Google Scholar] [CrossRef]
- Van Vrekhem, S.; Cools, P.; Declercq, H.; Van Tongel, A.; Vercruysse, C.; Cornelissen, M.; De Geyter, N.; Morent, R. Application of atmospheric pressure plasma on polyethylene for increased prosthesis adhesion. Thin Solid Films 2015, 596, 256–263. [Google Scholar] [CrossRef]
- Blackman, B.R.K.; Guild, F.J. Forced air plasma treatment for enhanced adhesion of polypropylene and polyethylene. J. Adhes. Sci. Technol. 2013, 27, 2714–2726. [Google Scholar] [CrossRef]
- Favia, P.; Oehr, C.; Wertheimer, M.R. Plasma Processes and Polymers; Wiley-VCH: Weinheim, Germany, 2005. [Google Scholar]
- Kusano, Y. Atmospheric Pressure Plasma Processing for Polymer Adhesion: A Review. J. Adhes. 2014, 90, 755–777. [Google Scholar] [CrossRef]
- Thomas, M.; Mittal, K.L. Atmospheric Pressure Plasma Treatment of Polymers: Relevance to Adhesion; Wiley Blackwell: Weinheim, Germany, 2013. [Google Scholar]
- Bárdos, L.; Baránková, H. Cold atmospheric plasma: Sources, processes, and applications. Thin Solid Films 2010, 518, 6705–6713. [Google Scholar] [CrossRef]
- Kostov, K.G.; Nishime, T.M.C.; Castro, A.H.R.; Toth, A.; Hein, L.R.O. Surface modification of polymeric materials by cold atmospheric plasma jet. Appl. Surf. Sci. 2014, 314, 367–375. [Google Scholar] [CrossRef]
- Shaw, D.; West, A.; Brendin, J.; Wagenaars, E. Mechanisms behind surface modification of polypropylene film using an atmospheric-pressure plasma jet. Plasma Sources Sci. Technol. 2016, 25, 065018. [Google Scholar] [CrossRef]
- Lambare, C.; Tessier, P.-Y.; Poncin-Epaillard, F.; Debarnot, D. Plasma functionalization and etching for enhancing metal adhesion onto polymeric substrates. Rsc Adv. 2015, 5, 62348–62357. [Google Scholar] [CrossRef]
- Kereszturi, K.; Tóth, A.; Mohai, M.; Bertóti, I.; Szépvölgyi, J. Nitrogen plasma-based ion implantation of poly(tetrafluoroethylene): Effect of the main parameters on the surface properties. Appl. Surf. Sci. 2010, 256, 6385–6389. [Google Scholar] [CrossRef]
- Dixon, D.; Meenan, B.J. Atmospheric Dielectric Barrier Discharge Treatments of Polyethylene, Polypropylene, Polystyrene and Poly(ethylene terephthalate) for Enhanced Adhesion. J. Adhes. Sci. Technol. 2012, 26, 2325–2337. [Google Scholar] [CrossRef]
- Bismarck, A.; Brostow, W.; Chiu, R.; Hagg Lobland, H.E.; Ho, K.C.K. Effects of surface plasma treatment on tribology of thermoplastic polymers. Polym. Eng. Sci. 2008, 48, 1971–1976. [Google Scholar] [CrossRef]
- Mandolfino, C.; Lertora, E.; Gambaro, C. Influence of cold plasma treatment parameters on the mechanical properties of polyamide homogeneous bonded joints. Surf. Coat. Technol. 2017, 313, 222–229. [Google Scholar] [CrossRef]
- Tóth, A.; Kereszturi, K.; Mohai, M.; Bertóti, I. Plasma based ion implantation of engineering polymers. Surf. Coat. Technol. 2010, 204, 2898–2908. [Google Scholar] [CrossRef]
- Novák, I.; Popelka, A.; Valentín, M.; Chodák, I.; Špírková, M.; Tóth, A.; Kleinová, A.; Sedliačik, J.; Lehocký, M.; Marônek, M. Surface Behavior of Polyamide 6 Modified by Barrier Plasma in Oxygen and Nitrogen. Int. J. Polym. Anal. Charact. 2014, 19, 31–38. [Google Scholar] [CrossRef]
- Šimor, M.; Ráhel’, J.; Vojtek, P.; Černak, M. Atmospheric-pressure diffuse coplanar surface discharge for surface treatments. Appl. Phys. Lett. 2002, 81, 2716–2718. [Google Scholar] [CrossRef]
- Černák, M.; Černáková, L.; Hudec, I.; Kováčik, D.; Zahoranová, A. Diffuse Coplanar Surface Barrier Discharge and its applications for in-line processing of low-added-value materials. Eur. Phys. J. Appl. Phys. 2009, 47, 22806. [Google Scholar] [CrossRef]
- Owens, D.K.; Wendt, R.C. Estimation of the surface free energy of polymers. J. Appl. Polym. Sci. 1969, 13, 1741–1747. [Google Scholar] [CrossRef]
- Mohai, M. XPS MultiQuant: Multimodel XPS quantification software. Surf. Interface Anal. 2004, 36, 828–832. [Google Scholar] [CrossRef]
- Evans, S.; Pritchard, R.G.; Thomas, J.M. Relative differential subshell photoionisation cross-sections (MgKα) from lithium to uranium. J. Electron Spectrosc. Relat. Phenom. 1978, 14, 341–358. [Google Scholar] [CrossRef]
- Reilman, R.F.; Msezane, A.; Manson, S.T. Relative intensities in photoelectron spectroscopy of atoms and molecules. J. Electron Spectrosc. Relat. Phenom. 1976, 8, 389–394. [Google Scholar] [CrossRef]
- Mohai, M.; Bertóti, I. Correction for Surface Contaminations in XPS: A Practical Approach. In Proceedings of the ECASIA 95 6th European Conference on Applications of Surface and Interface Analysis, Montreux, Switzerland, 9–13 October 1995; Mathieu, H.J., Reihl, B., Briggs, D., Eds.; John Willey & Sons: Chichester, UK, 1996; pp. 675–678. [Google Scholar]
- Kalácska, G.; Zsidai, L.; Keresztes, R.; Tóth, A.; Mohai, M.; Szépvölgyi, J. Effect of nitrogen plasma immersion ion implantation of polyamide-6 on its sliding properties against steel surface. Wear 2012, 290–291, 66–73. [Google Scholar] [CrossRef]
- Yeh, J.T.; Lai, Y.C.; Suen, M.C.; Chen, C.C. An improvement on the adhesion-strength of laminated ultra-high-molecular-weight polyethylene fabrics: Surface-etching/modification using highly effective helium/oxygen/nitrogen plasma treatment. Polym. Adv. Technol. 2011, 22, 1971–1981. [Google Scholar] [CrossRef]
- Tosun, K.; Felekoğlu, B.; Baradan, B. Multiple cracking response of plasma treated polyethylene fiber reinforced cementitious composites under flexural loading. Cem. Concr. Compos. 2012, 34, 508–520. [Google Scholar] [CrossRef]
- Navaneetha Pandiyaraj, K.; Arun Kumar, A.; RamKumar, M.C.; Deshmukh, R.R.; Bendavid, A.; Su, P.G.; Kumar, S.U.; Gopinath, P. Effect of cold atmospheric pressure plasma gas composition on the surface and cyto-compatible properties of low density polyethylene (LDPE) films. Curr. Appl. Phys. 2016, 16, 784–792. [Google Scholar] [CrossRef]
- López, R.; Sanchis, R.; García, D.; Fenollar, O.; Balart, R. Surface characterization of hydrophilic coating obtained by low-pressure CH4–O2 plasma treatment on a polypropylene film. J. Appl. Polym. Sci. 2009, 111, 2992–2997. [Google Scholar] [CrossRef]
- Kostov, K.G.; Nishime, T.M.C.; Hein, L.R.O.; Toth, A. Study of polypropylene surface modification by air dielectric barrier discharge operated at two different frequencies. Surf. Coat. Technol. 2013, 234, 60–66. [Google Scholar] [CrossRef]
| Property | Polyamide 6 (PA6) | Polyoxymethylene (POM-C) |
|---|---|---|
| Density (g/cm3) | 1.14 | 1.4 |
| Yield stress (MPa) | 70 | 65 |
| Elasticity modulus (N/mm2, MPa) | 3100 | 2700 |
| Shore D hardness | 75 | 85 |
| Thermal conductivity (W/mK) | 0.23 | 0.31 |
| Melting temperature (°C) | 255 | 170 |
| Sample | θw (°) | θCH2l2 (°) | γpol (mJ/m2) | γdisp (mJ/m2) | γtot (mJ/m2) |
|---|---|---|---|---|---|
| PA6 pristine | 70 ± 7.2 | 32 ± 2.1 | 6.3 | 43.6 | 50.0 |
| PA6 treated, 30 s | 28 ± 4.1 | 25 ± 2.6 | 26.1 | 46.2 | 72.3 |
| PA6 treated, 60 s | 26 ± 1.0 | 28 ± 2.3 | 27.6 | 45.0 | 72.6 |
| PA6 treated, 180 s | 21 ± 2.4 | 26 ± 2.7 | 29.1 | 45.8 | 74.9 |
| POM-C pristine | 73 ± 4.2 | 32 ± 2.1 | 5.2 | 43.6 | 48.8 |
| POM-C treated, 30 s | 41 ± 4.6 | 16 ± 3.6 | 18.9 | 48.9 | 67.8 |
| POM-C treated, 60 s | 44 ± 5.5 | 20 ± 1.3 | 17.8 | 47.9 | 65.8 |
| POM-C treated, 180 s | 43 ± 4.2 | 20 ± 3.2 | 18.2 | 48.0 | 66.1 |
| Sample | C | C CO | O | N | O/C ratio |
|---|---|---|---|---|---|
| PA6 pristine | 82.6 | - | 8.8 | 8.6 | |
| Corrected | 73.8 | - | 12.8 | 12.5 | 0.17 |
| PA6 treated | 67.3 | - | 20.3 | 13.1 | 0.30 |
| POM-C pristine | 22.5 | 38.4 | 39.0 | - | 1.01 |
| POM-C treated | 5.8 | 47.7 | 46.5 | - | 0.97 |
| C components | Composition (atomic %) | Binding energy (eV) | Chemical states | ||
|---|---|---|---|---|---|
| Pristine | Contamination-corrected pristine | Plasma-treated | |||
| PA6 | |||||
| C1 | 57.1 | 36.6 | 21.1 | 285.0 | C–C, C–H |
| C2 | 8.5 | 12.4 | 13.8 | 285.3 | CH–C=O |
| C3 | 8.5 | 12.4 | 13.8 | 286.0 | C=O, C–N |
| C4 | 8.5 | 12.4 | 16.3 | 288.0 | C=O, N–C=O |
| C5 | 0.0 | 0.0 | 2.4 | 289.3 | O=C–O(H) |
| POM-C | |||||
| C1 | 22.5 | — | 5.8 | 285.0 | C–C, C–H |
| C2 | 38.4 | 47.7 | 47.7 | 287.9 | O–C–O |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Károly, Z.; Kalácska, G.; Zsidai, L.; Mohai, M.; Klébert, S. Improvement of Adhesion Properties of Polyamide 6 and Polyoxymethylene-Copolymer by Atmospheric Cold Plasma Treatment. Polymers 2018, 10, 1380. https://doi.org/10.3390/polym10121380
Károly Z, Kalácska G, Zsidai L, Mohai M, Klébert S. Improvement of Adhesion Properties of Polyamide 6 and Polyoxymethylene-Copolymer by Atmospheric Cold Plasma Treatment. Polymers. 2018; 10(12):1380. https://doi.org/10.3390/polym10121380
Chicago/Turabian StyleKároly, Zoltán, Gábor Kalácska, László Zsidai, Miklós Mohai, and Szilvia Klébert. 2018. "Improvement of Adhesion Properties of Polyamide 6 and Polyoxymethylene-Copolymer by Atmospheric Cold Plasma Treatment" Polymers 10, no. 12: 1380. https://doi.org/10.3390/polym10121380
APA StyleKároly, Z., Kalácska, G., Zsidai, L., Mohai, M., & Klébert, S. (2018). Improvement of Adhesion Properties of Polyamide 6 and Polyoxymethylene-Copolymer by Atmospheric Cold Plasma Treatment. Polymers, 10(12), 1380. https://doi.org/10.3390/polym10121380