Biosynthesis and Characteristics of Aromatic Polyhydroxyalkanoates
Abstract
1. Introduction
2. Biosynthesized PHAs Bearing Aromatic Groups as Side Chains
2.1. Aromatic PHAs Produced from Corresponding Aromatic Compounds
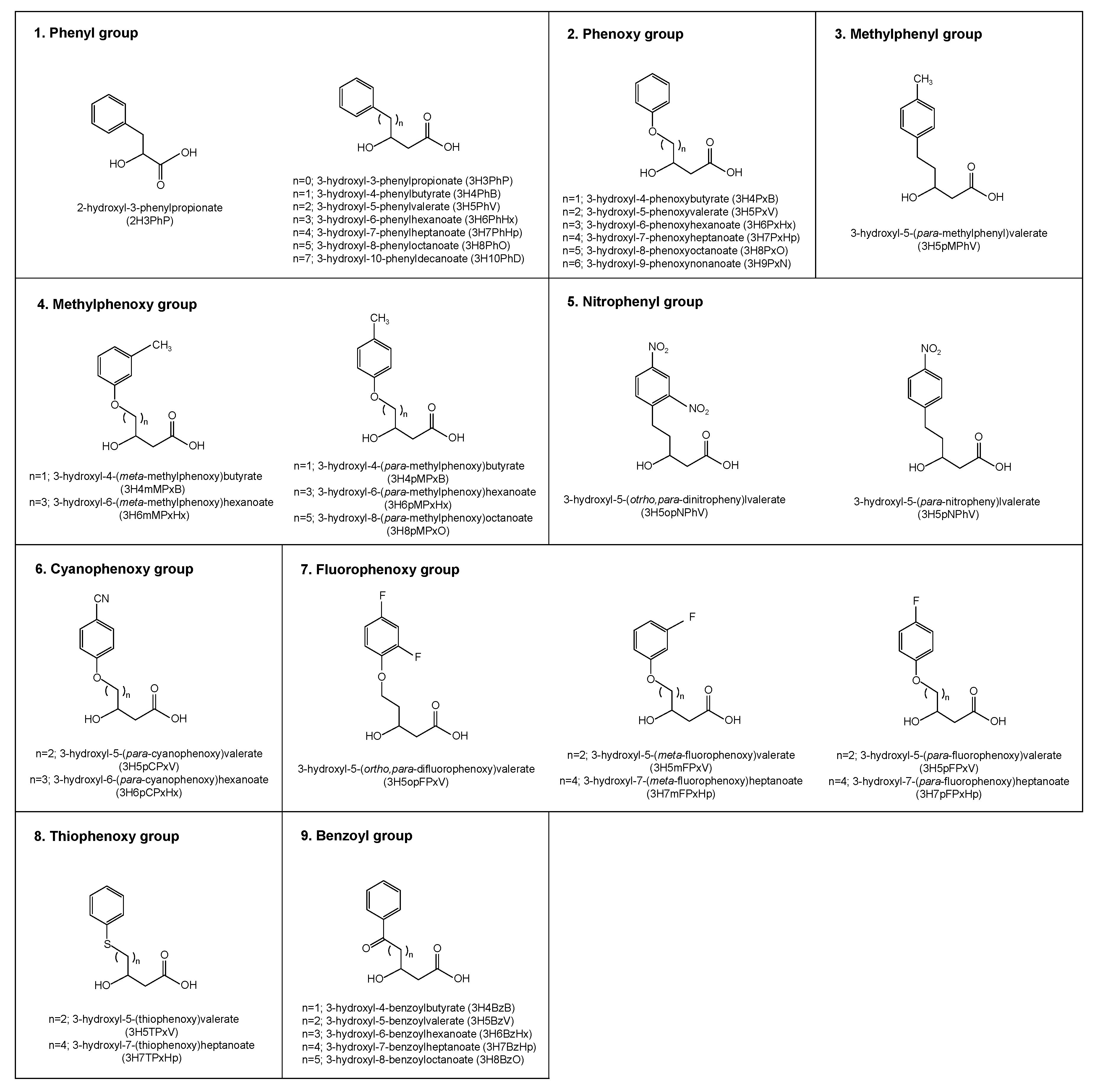
2.1.1. PHAs Containing Phenyl Group
Homopolymer
Copolymer Composed of Monomers Bearing Phenyl Groups with Different Carbon Numbers
Copolymer Containing Aliphatic and Aromatic Monomers
2.1.2. PHAs Containing Phenoxy Group
2.1.3. PHAs Containing Methylphenyl Group
2.1.4. PHAs Containing Methylphenoxy Group
2.1.5. PHAs Containing Nitrophenyl or Nitrophenoxy Group
2.1.6. PHAs Containing Cyanophenoxy Group
2.1.7. PHAs Containing Fluorophenoxy Group
2.1.8. PHAs Containing Thiophenoxy Group
2.1.9. PHAs Containing Benzoyl Group
2.2. Aromatic PHAs Produced through Complete Biosynthesis
3. Physical and Chemical Properties of Aromatic PHAs
3.1. Appearance
3.2. Mechanical Properties
3.3. Surface Properties
3.4. Degradability
3.4.1. Chemical Degradation
3.4.2. Biological Degradation
3.5. Solubility and Solvent Fractionation
3.6. Thermal Properties
3.6.1. Thermal Properties of PHAs Containing Phenyl Groups
Homopolymer
Copolymers Composed of Monomers Bearing Phenyl Groups with Different Carbon Numbers
Copolymers Containing Aliphatic and Aromatic Monomers
3.6.2. Thermal Properties of PHAs Containing Phenoxy Groups
3.6.3. Thermal Properties of PHAs Containing Methylphenyl Groups
3.6.4. Thermal Properties of PHAs Containing Methylphenoxy Groups
3.6.5. Thermal Properties of PHAs Containing Nitrophenyl Groups
3.6.6. Thermal Properties of PHAs Containing Cyanophenoxy Groups
3.6.7. Thermal Properties of PHAs Containing Fluorophenoxy Groups
3.6.8. Thermal Properties of PHAs Containing Thiophenoxy Groups
3.6.9. Thermal Properties of PHAs Containing Benzoyl Groups
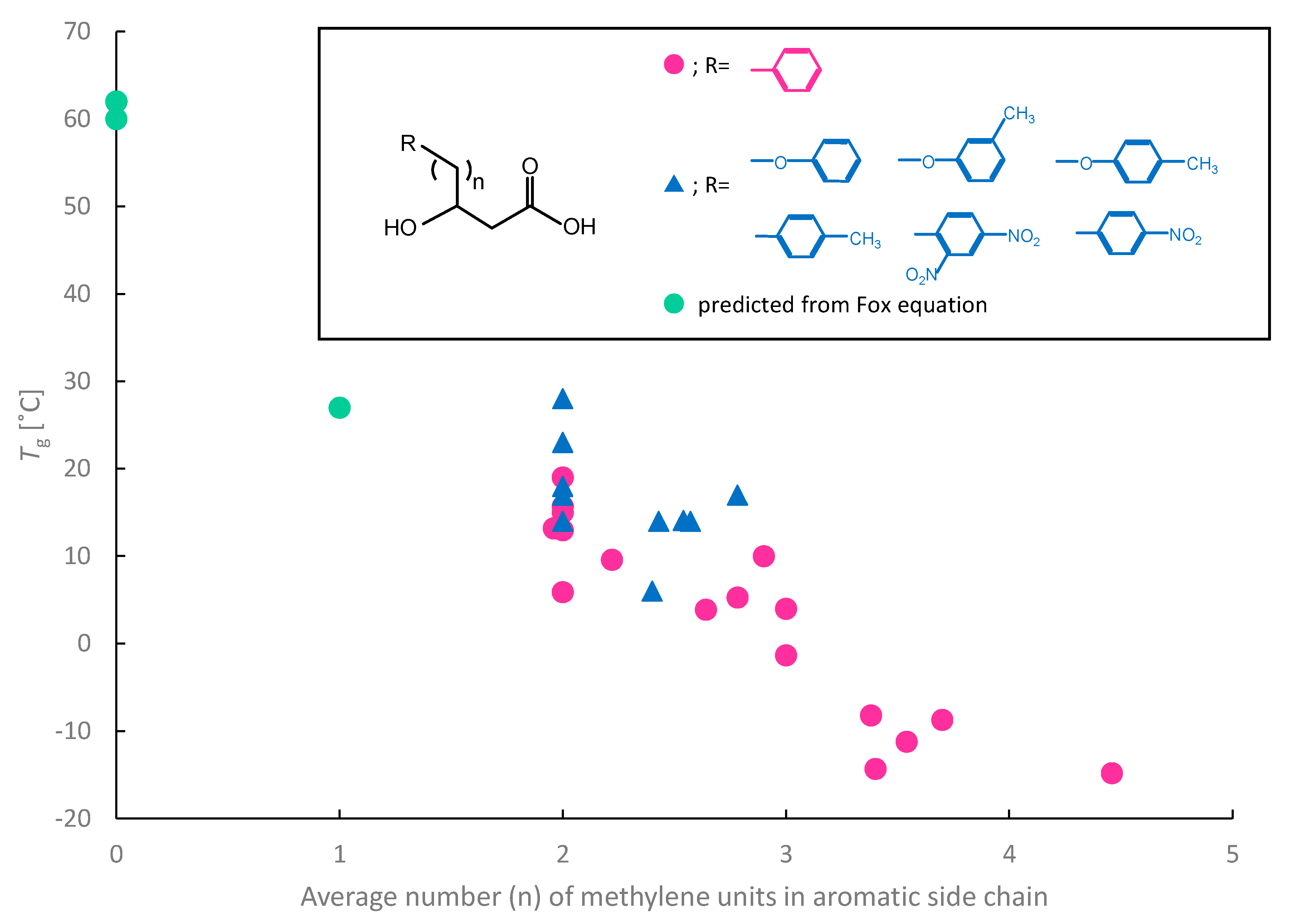
4. Effect of Incorporation of Aromatic Monomer on Tg of PHAs
5. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PHAs | Polyhydroxyalkanoates |
| P(3HB) | poly(3-hydroxybutyrate) |
| mcl-PHA | medium-chain-length PHA |
| 3HA | 3-hydroxyalkanoate |
| 3HE | 3-hydroxy-ω-alkenoate |
| 3HPhA | 3-hydroxy-ω-phenylalkanoate |
| 3HHx | 3-hydroxyhexanoate |
| 3HHp | 3-hydroxyheptanoate |
| 3HO | 3-hydroxyoctanoate |
| 3HN | 3-hydroxynonanoate |
| 3HD | 3-hydroxydecanoate |
| 3HDD | 3-hydroxydodecanoate |
| OA | octanoic acid |
| NA | nonanoic acid |
| 4PhB | 4-phenylbutyric acid |
| 5PhV | 5-phenylvaleric acid |
| 6PhHx | 6-phenylhexanoic acid |
| 7PhHp | 7-phenylheptanoic acid |
| 8PhO | 8-phenyloctanoic acid |
| 10PhD | 10-phenyldecanoic acid |
| 2H3PhP | 2-hydroxy-3-phenylpropionate |
| 2H4PhB | 2-hydroxy-4-phenylbutyrate |
| 3H3PhP | 3-hydroxy-3-phenylpropionate |
| 3H4PhB | 3-hydroxy-4-phenylbutyrate |
| 3H5PhV | 3-hydroxy-5-phenylvalerate |
| 3H6PhHx | 3-hydroxy-6-phenylhexanoate |
| 3H7PhHp | 3-hydroxy-7-phenylheptanoate |
| 3H8PhO | 3-hydroxy-8-phenyloctanoate |
| 3H10PhD | 3-hydroxy-10-phenyldecanoate |
| 3H4PxB | 3-hydroxy-4-phenoxybutyrate |
| 3H5PxV | 3-hydroxy-5-phenoxyvalerate |
| 3H6PxHp | 3-hydroxy-6-phenoxyhexanoate |
| 3H7PxHp | 3-hydroxy-7-phenoxyheptanoate |
| 3H8PxO | 3-hydroxy-8-phenoxyoctanoate |
| 3H9PxN | 3-hydroxy-9-phenoxynonanoate |
| 3H5pMPhV | 3-hydroxy-5-(para-methylphenyl)valerate |
| 3H4pMPxB | 3-hydroxy-4-(para-methylphenoxy)butyrate |
| 3H6pMPxHx | 3-hydroxy-6-(para-methylphenoxy)hexanoate |
| 3H8pMPxO | 3-hydroxy-8-(para-methylphenoxy)octanoate |
| 3H4mMPxB | 3-hydroxy-4-(meta-methylphenoxy)butyrate |
| 3H6mMPxHx | 3-hydroxy-6-(meta-methylphenoxy)hexanoate |
| 3H5pNPhV | 3-hydroxy-5-(para-nitrophenyl)valerate |
| 3H5opNPhV | 3-hydroxy-5-(ortho,para-nitrophenyl)valerate |
| 3H5pCPxV | 3-hydroxy-5-(para-cyanophenoxy)valerate |
| 3H6pCPxHx | 3-hydroxy-6-(para-cyanophenoxy)hexanoate |
| 3H5mFPxV | 3-hydroxy-5-(meta-fluorophenoxy)valerate |
| 3H7mFPxHp | 3-hydroxy-7-(meta-fluorophenoxy)heptanoate |
| 3H5pFPxV | 3-hydroxy-5-(para-fluorophenoxy)valerate |
| 3H7pFPxHp | 3-hydroxy-7-(para-fluorophenoxy)heptanoate |
| 3H5opFPxV | 3-hydroxy-5-(ortho,para-fluorophenoxy)valerate |
| 3H5TPxV | 3-hydroxy-5-thiophenoxyvalerate |
| 3H7TPxHp | 3-hydroxy-7-thiophenoxyheptanoate |
| 3H4BzB | 3-hydroxy-4-benzoylbutyrate |
| 3H5BzV | 3-hydroxy-5-benzoylvalerate |
| 3H6BzHx | 3-hydroxy-6-benzoylhexanoate |
| 3H7BzHp | 3-hydroxy-7-benzoylheptanoate |
| 3H8BzO | 3-hydroxy-8-benzoyloctanoate |
| Tg | glass transition temperature |
| Tm | melting temperature |
| ΔHm | enthalpy of fusion |
| Mn | number-average molecular weight |
| Mw | weight-average molecular weight |
References
- Lee, S.Y. Bacterial polyhydroxyalkanoates. Biotechnol. Bioeng. 1996, 49, 1–14. [Google Scholar] [CrossRef]
- Chen, G.Q.; Patel, M.K. Plastics derived from biological sources: Present and Future: A technical and environmental review. Chem. Rev. 2012, 112, 2082–2099. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Q. A microbial polyhydroxyalkanoates (PHA) based bio- and materials industry. Chem. Soc. Rev. 2009, 38, 2434–2446. [Google Scholar] [CrossRef] [PubMed]
- Pizzoli, M.; Scandola, M.; Ceccorulli, G. Crystallization kinetics and morphology of poly(3-hydroxybutyrate)/cellulose ester blends. Macromolecules 1994, 27, 4755–4761. [Google Scholar] [CrossRef]
- Anderson, A.J.; Dawes, E.A. Occurrence, Metabolism, Metabolic Role, and Industrial Uses of Bacterial Polyhydroxyalkanoates. Microbiol. Rev. 1990, 54, 450–472. [Google Scholar] [PubMed]
- Tsuge, T. Metabolic improvements and use of inexpensive carbon sources in microbial production of polyhydroxyalkanoates. J. Biosci. Bioeng. 2002, 94, 579–584. [Google Scholar] [CrossRef]
- De Koning, G.J.M.; Lemstra, P.J. Crystallization phenomena in bacterial poly[(R)-3-hydroxybutyrate]: 2. Embrittlement and rejuvenation. Polymer 1993, 34, 4089–4094. [Google Scholar] [CrossRef]
- De Koning, G.J.M.; Scheeren, A.H.C.; Lemstra, P.J. Crystallization phenomena in bacterial poly[(R)-3-hydroxybutyrate]: 3. Toughening via texture changes. Polymer 1994, 35, 4598–4605. [Google Scholar] [CrossRef]
- Hazer, B.; Steinbuchel, A. Increased diversification of polyhydroxyalkanoates by modification reactions for industrial and medical applications. Appl. Microbiol. Biotechnol. 2007, 74, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Steinbuchel, A.; Valentin, H.E. Diversity of bacterial polyhydroxyalkanoic acids. FEMS Microbiol. Lett. 1995, 128, 219–228. [Google Scholar] [CrossRef]
- Luengo, J.M.; Garcia, B.; Sandoval, A.; Naharro, G.; Olivera, E.R. Bioplastics from microorganism. Curr. Opin. Biotechnol. 2003, 6, 251–260. [Google Scholar] [CrossRef]
- Tsuge, T.; Saito, Y.; Kikkawa, Y.; Hiraishi, T.; Doi, Y. Biosynthesis and compositional regulation of poly[(3-hydroxybutyrate)-co-(3-hydroxyhexanoate)] in recombinant Ralstonia eutropha expressing mutated polyhydroxyalkanoate synthase genes. Macromol. Biosci. 2004, 4, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Ishii, N.; Sato, S.; Tsuge, T. Thermal properties and crystallization behaviors of medium-chain-length poly(3-hydroxyalkanoate)s. Polymer 2012, 53, 3026–3034. [Google Scholar] [CrossRef]
- Fritzsche, K.; Lenz, R.W. An unusual bacterial polyester with a phenyl pendant group. Macromol. Chem. 1990, 191, 1957–1965. [Google Scholar] [CrossRef]
- Mizuno, S.; Katsumata, S.; Hiroe, A.; Tsuge, T. Biosynthesis and thermal characterization of polyhydroxyalkanoates bearing phenyl and phenylalkyl side groups. Polym. Degrad. Stable 2014, 109, 379–384. [Google Scholar] [CrossRef]
- Mizuno, S.; Enda, Y.; Saika, A.; Hiroe, A.; Tsuge, T. Biosynthesis of polyhydroxyalkanoates containing 2-hydroxy-4-methylvalerate and 2-hydroxy-3-phenylpropionate units from a related or unrelated carbon source. J. Biosci. Bioeng. 2018, 125, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, S.; Hiroe, A.; Fukui, T.; Abe, H.; Tsuge, T. Fractionation and thermal characteristics of biosynthesized polyhydroxyalkanoates bearing aromatic groups as side chains. Polym. J. 2017, 49, 557–565. [Google Scholar] [CrossRef]
- Yang, J.E.; Park, S.J.; Kim, W.J.; Kim, H.J.; Kim, B.J.; Lee, H.; Shin, J.; Lee, S.Y. One-step fermentative production of aromatic polyesters from glucose by metabolically gineered Escherichia coli strains. Nat. Commun. 2018, 9, 79. [Google Scholar] [CrossRef] [PubMed]
- Song, J.J.; Mun, H.C.; Sung, C.Y.; Nam, E.H. Cometabolism of ω-phenylalkanoic acids with butyric acid for efficient production of aromatic polyesters in Pseudomonas putida BM01. J. Microbiol. Biotechnol. 2001, 11, 435–442. [Google Scholar]
- Antoun, S.; Grizzi, I.; Lenz, R.W.; Fuller, C. Production of a Chiral Polyester by Pseudomonas oleovorans Grown with 5-Phenyl-2,4-Pentadienoic Acid. Chirality 1991, 3, 492–494. [Google Scholar] [CrossRef]
- Curley, J.M.; Hazer, B.; Lenz, R.W.; Fuller, R.C. Production of poly(3-hydroxyalkanoates) containing aromatic substituents by Pseudomonas oleovorans. Macromoleculs 1996, 29, 1762–1766. [Google Scholar] [CrossRef]
- Tobin, K.M.; O’Connor, K.E. Polyhydroxyalkanoate accumulating diversity of Pseudomonas species utilising aromatic hydrocarbons. FEMS Microbiol. Lett. 2005, 253, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.G.; Roo, G.; O’Connor, K.E. Accumulation of Polyhydroxyalkanoate from Styrene and Phenylacetic Acid by Pseudomonas putida CA-3. Appl. Environ. Microbiol. 2005, 2046–2052. [Google Scholar] [CrossRef] [PubMed]
- Kim, O.Y.; Gross, R.A.; Rutherford, D.R. Bioengineering of poly(β-hydroxyalkanoates) for advanced material applications: Incorporation of cyano and nitrophenoxy side chain substituents. Can. J. Microbiol. 1995, 41, 32–43. [Google Scholar] [CrossRef]
- Garcia, B.; Olivera, E.R.; Minambres, B.; Fernandez-Valverde, M.; Canedo, L.M.; Prieto, M.A.; Garcia, J.L.; Martinez, M.; Luengo, J.M. Novel Biodegradable Aromatic Plastics from a Bacterial Source. J. Biol. Chem. 1999, 274, 29228–29241. [Google Scholar] [CrossRef] [PubMed]
- Abraham, G.A.; Gallardo, A.; Roman, J.S.; Olivera, E.R.; Jodra, R.; Garcia, B.; Minambres, B.; Garcia, J.L.; Luengo, J.M. Microbial synthesis of poly(β-hydroxyalkanoates) bearing phenyl groups from Pseudomonas putida: Chemical structure and characterization. Biomacromolecules 2001, 2, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.B.; Lenz, R.W.; Fuller, R.C. Preparation and Characterization of Poly(β-hydroxyalkanoates) Obtained from Pseudomonas oleovorans Grown with Mixtures of 5-Phenylvaleric Acid and n-Alkanoic Acids. Macromolecules 1991, 24, 5256–5260. [Google Scholar] [CrossRef]
- Chung, D.M.; Choi, M.H.; Song, J.J.; Yoon, S.C.; Kang, I.K.; Huh, N.E. Intracellular degradation of two structurally different polyhydroxyalkanoic acids accumulated in Pseudomonas putida and Pseudomonas citronellolis from mixtures of octanoic acid and 5-phenylvaleric acid. Int. J. Macromol. 2001, 29, 243–250. [Google Scholar] [CrossRef]
- Hartmann, R.; Hany, R.; Geiger, T.; Egli, T.; Witholt, B.; Zinn, M. Tailored Biosynthesis of Olefinic Medium-Chain-Length Poly[(R)-3-hydroxyalkanoates] in Pseudomonas putida GPo1 with Improved Thermal Properties. Macromolecules 2004, 37, 6780–6785. [Google Scholar] [CrossRef]
- Song, J.J.; Yoon, S.C. Biosynthesis of Novel Aromatic Copolyesters from Insoluble 11-Phenoxyundecanoic Acid by Pseudomonas putida BM01. Appl. Environ. Microbiol. 1996, 62, 536–544. [Google Scholar] [PubMed]
- Kim, Y.B.; Rhee, Y.H.; Han, S.H.; Heo, G.S.; Kim, J.S. Poly-3-hydroxyalkanoates Produced from Pseudomonas oleovorans Grown with ω-Phenoxyalkanoates. Macromolecules 1996, 29, 3432–3435. [Google Scholar] [CrossRef]
- Hazer, B.; Lenz, R.W.; Fuller, R.C. Bacterial production of poly-3-hydroxyalkanoates containing arylalkyl substituent groups. Polymer 1996, 37, 5951–5957. [Google Scholar] [CrossRef]
- Kim, Y.B.; Kim, D.Y.; Rhee, Y.H. PHAs Produced by Pseudomonas putida and Pseudomonas oleovorans Grown with n-Alkanoic Acids Containing Aromatic Groups. Macromolecules 1999, 32, 6058–6064. [Google Scholar] [CrossRef]
- Arostegui, S.M.; Aponte, M.A.; Diaz, E.; Schroder, E. Bacterial Polyesters Produced by Pseudomonas oleovorans Containing Nitrophenyl Groups. Macromolecules 1999, 32, 2889–2895. [Google Scholar] [CrossRef]
- Gross, R.A.; Kim, O. Cyanophenoxy-Containing Microbial Polyesters: Structural Analysis, Thermal Properties, Second Harmonic Generation and In-Vivo Biodegradability. Polym. Int. 1996, 39, 205–213. [Google Scholar] [CrossRef]
- Takagi, Y.; Yasuda, R.; Maehara, A.; Yamane, T. Microbial synthesis and characterization of polyhydroxyalkanoates with fluorinated phenoxy side groups from Pseudomonas putida. Eur. Polym. J. 2004, 40, 1551–1557. [Google Scholar] [CrossRef]
- Takagi, Y.; Hashii, M.; Maehara, A.; Yamane, T. Biosynthesis of Polyhydroxyalkanoate with a Thiophenoxy Side Group Obtained from Pseudomonas putida. Macromolecules 1999, 32, 8315–8318. [Google Scholar] [CrossRef]
- Honma, T.; Imamura, T.; Kenmoku, T.; Kobayashi, S.; Yano, T. Biosynthesis of Novel Poly(3-hydroxyalkanoates) Containing Benzoyl Groups. J. Environ. Biotechnol. 2004, 4, 49–55. [Google Scholar]
- Olivera, E.R.; Carnicero, D.; Garcia, B.; Minambres, B.; Moreno, M.A.; Canedo, L.; DiRusso, C.C.; Naharro, G.; Luengo, J.M. Two different pathways are involved in the β-oxidation of n-alkanoic and n-phenylalkanoic acids in Pseudomonas putida U: Genetic studies and biotechnological applications. Mol. Microbiol. 2001, 39, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Olivera, E.R.; Carnicero, D.; Jodra, R.; Minambres, B.; Garcia, B.; Abraham, G.A.; Gallardo, A.; Roman, J.S.; Garcia, J.L.; Naharro, G.; et al. Genetically engineered Pseudomonas: A factory of new bioplastics with broad applications. Environ. Microbiol. 2001, 3, 612–618. [Google Scholar] [CrossRef] [PubMed]
- Curley, J.M.; Lenz, R.W.; Fuller, R.C. Sequential production of two different polyesters in the inclusion bodies of Pseudomonas oleovorans. Int. J. Biol. Macromol. 1996, 19, 29–34. [Google Scholar] [CrossRef]
- Ward, P.G.; O’Connor, K.E. Bacterial synthesis of polyhydroxyalkanoates containing aromatic and aliphatic monomers by Pseudomonas putida CA-3. Int. J. Biol. Macromol. 2005, 35, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Bao, H.J.; Kang, C.K.; Fukui, T.; Doi, Y. Production of a novel copolyester of 3-hydroxybutyric acid and medium-chain-length 3-hydroxyalkanoic acids by Pseudomonas sp. 61–3 from sugars. Appl. Microbiol. Biotechnol. 1996, 45, 363–370. [Google Scholar] [CrossRef]
- Ritter, H.; von Spee, A.G. Poly(3-hydroxy-5-phenoxypentanoate-co-3-hydroxy-9-phenoxynonanoate) from Pseudomonas oleovorans. Macromol. Chem. Phys. 1996, 195, 1665–1672. [Google Scholar] [CrossRef]
- Kim, D.Y.; Kim, Y.B.; Rhee, Y.H. Evaluation of various carbon substrates for the biosynthesis of polyhydroxyalkanoates bearing functional groups by Pseudomonas putida. Int. J. Biol. Macromol. 2000, 28, 23–29. [Google Scholar] [CrossRef]
- Lenz, R.W.; Regel, W.; Westfelt, L. Cationic polymerization of p-substituted α-methylstyrenes, 1. Tacticity by 1H- and 19F-NMR spectroscopy. Macromol. Chem. 1975, 176, 781–787. [Google Scholar] [CrossRef]
- Lenz, R.W.; Westfelt, L.C. Cationic polymerization of p-substituted α-methylstyrenes. III. Effect of polymerization conditions on tacticity and molecular weight for p-chloro-α-methylstyrene. J. Polym. Sci. 1976, 14, 2147–2153. [Google Scholar] [CrossRef]
- Lenz, R.W.; Sunderland, J.E.; Westfelt, L.C. Cationic polymerization of p-substituted α-methylstyrenes, 2. Crystalline polymers from p-methyl- and p-isopropyl-α-methylstyrene. Macromol. Chem. 1976, 177, 653–662. [Google Scholar] [CrossRef]
- Höpken, J.; Sheiko, S.; Czech, J.; Möller, M. Polymer surface modification by fluorocarbon-hydrocarbon substituents. Polym. Prepr. 1992, 33, 937–938. [Google Scholar]
- Shimizu, T.; Tanaka, Y.; Kutsumizu, S. Ordered structures of poly(1H,1H,2H,2H-perfluorodecyl α-substituted acrylate)s. Macromol. Symp. 1994, 82, 173–184. [Google Scholar] [CrossRef]
- Koketsu, K.; Mitsuhashi, S.; Tabata, K. Identification of homophenylalanine biosynthetic genes from the cyanobacterium Nostoc punctiforme PCC73102 and application to its microbial production by Escherichia coli. Appl. Environ. Microbiol. 2013, 79, 2201–2208. [Google Scholar] [CrossRef] [PubMed]
- Shen, R.; Cai, L.W.; Meng, D.C.; Wu, L.P.; Guo, K.; Dong, G.X.; Liu, L.; Chen, J.C.; Wu, Q.; Chen, G.Q. Benzene containing polyhydroxyalkanoates homo- and copolymers synthesized by genome edited Pseudomonas entomophila. Sci. China Life Sci. 2014, 57, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Sudeh, K.; Abe, H.; Doi, Y. Synthesis, structure and properties of polyhydroxyalkanoates: Biological polyesters. Prog. Polym. Sci. 2000, 25, 1503–1555. [Google Scholar] [CrossRef]
- Witholt, B.; Kessler, B. Perspectives of medium chain length poly(hydroxyalcanoates), a versatile set of bacterial bioplastics. Curr. Opin. Biotechnol. 1999, 10, 279–285. [Google Scholar] [CrossRef]
- Sandval, A.; Arias-Barrau, E.; Bermejo, F.; Canedo, L.; Naharro, G.; Olivera, E.R.; Luengo, J.M. Production of 3-hydroxy-n-phenylalkanoic acids by a genetically engineered strain of Pseudomonas putida. Appl. Microbiol. Biotechnol. 2005, 67, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Foster, L.J.R.; Lenz, R.W.; Fuller, R.C. Intracellular depolymerase activity in isolated inclusion bodies containing polyhydroxyalkanoates with long alkyl and functional substituents in the side chain. Int. J. Biol. Macromol. 1999, 26, 187–192. [Google Scholar] [CrossRef]
- Curley, J.M.; Lenz, R.W.; Fuller, R.C.; Browne, S.E.; Gabriel, C.B.; Panday, S. 13C n.m.r, spectroscopy in living cells of Pseudomonas oleovorans. Polymer 1997, 38, 5313–5319. [Google Scholar] [CrossRef]
- Jenekhe, S.A.; Roberts, M.F. Effects of intermolecular forces on the glass transition of polymers. Macromolecules 1993, 26, 4981–4983. [Google Scholar] [CrossRef]
- Tsuge, T.; Hamada, Y.; Watanabe, Y.; Tomizawa, S.; Yamamoto, T.; Abe, H. Characterization of biosynthesized P(3HB-co-3HA)s swellable in organic solvents. Polym. Degrad. Stable 2010, 95, 1345–1348. [Google Scholar] [CrossRef]
- Tanadchangsaeng, N.; Tsuge, T.; Abe, H. Comonomer compositional distribution, physical properties, and enzymatic degradability of bacterial poly(3-hydroxybutyrate-co-3-hydroxy-4-methylvalerate) copolyesters. Biomacromolecules 2010, 11, 1615–1622. [Google Scholar] [CrossRef] [PubMed]
- Fox, T.G. Influence of diluent and of copolymer composition on the glass temperature of a polymer system. Bull. Am. Phys. Soc. 1956, 1, 123–125. [Google Scholar]
- Yamada, M.; Matsumoto, K.; Uramoto, S.; Motohashi, R.; Abe, H.; Taguchi, S. Lactate fraction dependent mechanical properties of semitransparent poly(lactate-co-3-hydroxybutyrate)s produced by control of lactyl-CoA monomer fluxes in recombinant Escherichia coli. J. Biotechnol. 2011, 154, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Cowie, J.M.G.; Henshall, S.A.E.; McEwen, I.J.; Veličković, J. Glass and sub-glass transitions in the di-alkyl ester series, methyl to hexyl. Polymer 1977, 18, 612–616. [Google Scholar] [CrossRef]
- Olivera, E.R.; Arcos, M.; Naharro, G.; Luengo, J.M. Unusual PHA biosynthesis. Microbiol. Monogr. 2010, 14, 133–186. [Google Scholar] [CrossRef]
- Iwata, T. Strong fibers and films of microbial polyesters. Macromol. Biosci. 2005, 5, 689–701. [Google Scholar] [CrossRef] [PubMed]
- Tsuge, T. Fundamental factors determining the molecular weight of polyhydroxyalkanoate during biosynthesis. Polym. J. 2016, 48, 1051–1057. [Google Scholar] [CrossRef]
- Kumar, P.; Kim, B.S. Valorization of polyhydroxyalkanoates production process by co-synthesis of value-added products. Bioresour. Technol. 2018, 269, 544–556. [Google Scholar] [CrossRef] [PubMed]
| Aromatic Group | Polymer | Bacterial Strain | Carbon Substrate | Reference | |
|---|---|---|---|---|---|
| Phenyl | (homopolymer) | P(3H5PhV) | P. oleovorans | 5PhV | [14] |
| P(3H5PhV) | P. putida BM01 | 5PhV | [19] | ||
| P(3H5PhV) | P. putida KT2440 | 5PhV | [15] | ||
| P(3H5PhV) | P. oleovorans | 5-phenyl-2,4-pentadienoic acid | [20] | ||
| P(3H6PhHx) | P. putida U | 6PhHx | [25] | ||
| (copolymer) | P(3H5PhV-3H7PhHp) | P. putida U | 7PhHp | [26] | |
| P(3H6PhHx-3H8PhO) | P. putida U | 8PhO | [25] | ||
| P(3H6PhHx-3H8PhO-3H10PhD) | P. putida U | 10PhD | [25] | ||
| (copolymer containing aliphatic monomers) | P(3HA-3H5PhV) | P. oleovorans | 5PhV, HA | [27] | |
| P(3HA-3H5PhV) | P. citronellolis | 5PhV, OA | [28] | ||
| P(3HA-HE-3H5PhV) | P. putida GPo1 | 5PhV, OA, 10-undecenoic acid | [29] | ||
| P(3HA-3H4PhB-3H6PhHx) | P. fluorescens B3 | 6PhHx | [22] | ||
| P(3HA-3H4PhB-3H7PhHx) | P. jessenii C8 | 7PhHx | [22] | ||
| P(3HB-3HA-3H3PhP) | R. eutropha PHB-4 expressing PhaC1Ps | 3-hydroxy-3-phenylpropionic acid, fructose | [15] | ||
| P(3HB-3HA-3H3PhP) | R. eutropha PHB-4 expressing PhaC1Ps | cinnamic acid, fructose | [15] | ||
| P(3HB-3HA-3H4PhB) | R. eutropha PHB-4 expressing PhaC1Ps | 4PhB, fructose | [17] | ||
| P(3HB-3HA-2H3PhP) | E. coli expressing mutated PhaC1Ps | phenylalanine, sugars | [16] | ||
| P(3HB-3HA-2H3PhP) | E. coli expressing mutated PhaC1Ps | sugars | [16] | ||
| Phenoxy | P(3H5PxV-3H7PxHp) | P. putida BM01 | 11-phenoxyundecanoic acid | [30] | |
| P(3H4PxB-3H6PxHx-3H8PxO) | P. oleovorans | 8-phenoxyoctanoic acid | [31] | ||
| P(3H5PxV-3H7PxHp-3H9PxN) | P. oleovorans | 11-phenoxyundecanoic acid | [31] | ||
| Methylphenyl | P(3H5pMPhV) | P. oleovorans | 5-(para-methylphenyl)valeric acid | [21] | |
| P(3HA-3H5pMPhV) | P. putida | 9-(para-methylphenyl)nonanoic acid | [32] | ||
| Methylphenoxy | P(3H4pMPxB-3H6pMPxHx) 1 | P. putida | 8-(para-methylphenoxy)octanoic acid | [33] | |
| P(3H4mMPxB-3H6mMPxHx) | P. putida | 8-(meta-methylphenoxy)octanoic acid | [33] | ||
| Nitrophenyl | P(3HA-3H5(pNPh and/or opNPh)V) | P. oleovorans | 5-(ortho,para-dinitrophenyl)valeric acid | [34] | |
| Nitrophenoxy | PHA 2 | P. oleovorans | OA, 6-(para-nitrophenoxy)hexanoic acid | [24] | |
| Cyanophenoxy | P(3HA-3H6pCPxHx) | P. putida KT2440 | OA, 6-(para-cyanophenoxy)hexanoic acid | [35] | |
| P(3HHx-3HO-3H5pCPxV) | P. putida KT2442 | OA, 5-(para-cyanophenoxy)valeric acid | [24] | ||
| Fluorophenoxy | P(3H5mFPxV-3H7mFPxHp) | P. putida | 11-(meta-fluorophenoxy)undecanoic acid | [36] | |
| P(3H5pFPxV-3H7pFPxHp) | P. putida | 11-(para-fluorophenoxy)undecanoic acid | [36] | ||
| P(3H5opFPxV) | P. putida | 11-(ortho,para-difluorophenoxy)undecanoic acid | [36] | ||
| Thiophenoxy | P(3H5TPxV-3H7TPxHp) | P. putida 27N01 | 11-thiophenoxy undecanoic acid | [37] | |
| Benzoyl | P(3HA-3H4BzB) | P. cichorii YN2 | 4-benzoylbutyric acid | [38] | |
| P(3H5BzV-3H7BzHp) | P. cichorii YN3 | 7-benzoylheptanoic acid | [38] | ||
| P(3H5BzV-3H6BzHx-3H8BzO) | P. cichorii YN4 | 8-benzoyloctanoic acid | [38] | ||
| Polymer 1 | Mechanical Properties | Molecular Weight 2 | Reference | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Yield Strength (MPa) | Maximum Tension Strength (MPa) | Elongation at Break (%) | Young’s Modulus (MPa) | Mn (104) | Mw (104) | Mw/Mn | ||||||||||
| P(3HDD) | 5.5 | ± | 0.8 | 5.5 | ± | 0.9 | 60 | ± | 34 | 61.1 | ± | 6.4 | 5.2 | 10.4 | 2.0 | [52] |
| P(3HDD-2.91 mol% 3H5PhV) | 1.53 | ± | 0.65 | 2.05 | ± | 0.51 | 37.38 | ± | 6.28 | 93.91 | ± | 20.52 | 4.1 | 6.56 | 1.6 | [52] |
| P(3HDD-18.7 mol% 3H5PhV) | 3.63 | ± | 0.68 | 4.36 | ± | 0.94 | 86.03 | ± | 39.80 | 94.79 | ± | 34.95 | 4.3 | 7.31 | 1.7 | [52] |
| P(3HDD-31.9 mol% 3H5PhV) | 2.84 | ± | 1.05 | 3.15 | ± | 1.21 | 32.02 | ± | 15.94 | 48.72 | ± | 24.04 | 3.4 | 6.12 | 1.8 | [52] |
| P(3H5PhV) | ― 3 | ― 3 | ― 3 | ― 3 | 2.1 | 4.41 | 2.1 | [52] | ||||||||
| P(3HB) produced by wild-type bacteria | - | 43 | 5 | 3500 | - | 1–300 | approx. 2 | [53] | ||||||||
| Polypropylene | - | 38 | 400 | 1700 | - | - | - | [53] | ||||||||
| Low-density polyethylene | - | 10 | 620 | 200 | - | - | - | [53] | ||||||||
| Aromatic Group | Polymer | Thermal Properties 4 | Molecular Weight 5 | Reference | |||
|---|---|---|---|---|---|---|---|
| Tm (°C) | Tg (°C) | ΔHm (J/g) | Mn (104) | Mw/Mn | |||
| ― | P(3HB) | 162, 178 | 5 | 51 | 32 | 1.9 | [17] |
| mcl-PHA | 45 to 69 | −53 to −28 | - | 17 | 2.1 | [13] | |
| Polypropylene | 176 | −10 | - | - | - | [53] | |
| Low-density polyethylene | 130 | −30 | - | - | - | [53] | |
| Phenyl | P(3H5PhV) | 54 to 69 | 13 | - | 10 | 3.5 | [14] |
| (homopolymer) | P(3H6PhHx) | not detectable | −1.3 | - | 21.6 | 2.2 | [26] |
| P(3H4PhB-95 mol% 3H6PhHx) | not detectable | 10 | - | - | - | [19] | |
| Phenyl | P(3H6PhHx-73 mol% 3H8PhO) | not detectable | −14.8 | - | 8.2 | 2.0 | [26] |
| (copolymer) | P(3H5PhV-77 mol% 3H7PhHp) | not detectable | −11.2 | - | 6.7 | 2.3 | [26] |
| P(3H5PhV-38 mol% 3H6PhHx-50 mol% 3H7PhHp) | not detectable | −8.2 | - | 13.8 | 2.7 | [26] | |
| Phenyl (copolymer containing aliphatic monomers) | P(95.1 mol% 3HB-4.1 mol% 3H3PhP) | 146, 158 | 10.7 | 27.8 | 6.5 | 3.5 | [15] |
| P(89.5 mol% 3HB-8.9 mol% 3H3PhP) | 135, 149 | 14.6 | 7.1 | 9.7 | 3.7 | [15] | |
| P(3HA-98 mol% 3H5PhV) | 51.5 | 13.2 | - | 2.5 | 3.1 | [42] | |
| P(3HA-15 mol% 3H4PhB-83 mol% 3H6PhHx) | 52.1 | 3.9 | - | 9.1 | 3.5 | [42] | |
| P(3HA-85 mol% 3H5PhV-13 mol% 3H7PhHp) | not detectable | 9.6 | - | 6.7 | 3.1 | [42] | |
| P(3HA-7 mol% 3H4PhB-61 mol% 3H6PhHx-30 mol% 3H8PhO) | not detectable | −14.3 | - | 7.2 | 4.1 | [42] | |
| P(3HA-6 mol% 3H4PhB-57 mol% 3H6PhHx-26 mol% 3H8PhO-9 mol% 3H10PhD) | not detectable | −8.7 | - | 5.7 | 2.9 | [42] | |
| P(3HA-45 mol% 3H5PhV) | - | −20 | - | - | - | [28] | |
| P(3HA-40.6 mol% 3H5PhV) 1 | 49 | 5, −31 | 7.2 | 5 | 2.0 | [27] | |
| P(3HA-10 mol% HE-59 mol% 3H5PhV) | not detectable | −6 | - | 12.4 | 2.9 | [29] | |
| (P(3HA-10 mol% 3HE) | 40.1 | −38.7 | - | 8.6 | 2.2 | [29]) | |
| P(3H5PhV) | 50.4 | 5.9 | - | 2.1 | 2.1 | [52] | |
| P(3HDD-31.97 mol% 3H5PhV) | 75.84 | −35.15 | - | 3.4 | 1.8 | [52] | |
| P(3HDD-18.7 mol% 3H5PhV) | 80.13 | −35.81 | - | 4.3 | 1.7 | [52] | |
| P(3HDD-2.91 mol% 3H5PhV) | 81 | −33.35 | - | 4.1 | 1.6 | [52] | |
| (P(3HDD) | 82.4 | −49.3 | - | 5.2 | 2.0 | [52]) | |
| P(3HB-12 mol% 3H3PhP) | 132, 148 | 9 | 4 | 18 | 3.5 | [17] | |
| P(3HB-15 mol% 3H3PhP) | not detectable | 15 | not detectable | 14 | 2.4 | [17] | |
| P(3HB-18 mol% 3H3PhP) | not detectable | 16 | not detectable | 9.2 | 2.1 | [17] | |
| P(3HB-21 mol% 3H3PhP) | not detectable | 20 | not detectable | 3.8 | 2.1 | [17] | |
| P(3HB-4 mol% 3H4PhB) | 138, 151 | 7 | 46 | 7.1 | 2.0 | [17] | |
| P(3HB-8 mol% 3H4PhB) | 126, 136 | 9 | 12 | 4.5 | 2.4 | [17] | |
| P(3HB-15 mol% 3H4PhB) | 105, 119 | 10 | 3 | 1.7 | 2.3 | [17] | |
| Phenoxy | P(3H5PxV-27 mol% 3H7PxHp) | 70 | 14.1 | 2.9 | - | - | [30] |
| P(3H5PxV) | 88 | 23 | - | 7.4 | 2.0 | [38] | |
| Methylphenyl | P(3H5pMPhV) | 95 | 18 | - | - | - | [21] |
| P(3HA-15 mol% 3H5pMPhV) | 60 | 14 | 0.1 | 5 | 2.4 | [32] | |
| Methylphenoxy | P(3H4pMPxB-71.5 mol% 3H6pMPxHx) | 97 | 14 | 33.5 | 2.5 | 2.5 | [33] |
| P(3H5PhV-6.7 mol% 3H4pMPxB-28.3 mol% 3H6pMPxHx) | not detectable | 17 | - | - | - | [33] | |
| P(3H4mMPxB-70 mol% 3H6mMPxHx) | 43 | 6 | 0.2 | - | - | [33] | |
| Nitrophenyl | P(3HA-6.9 mol% 3H5(pNPh and/or opNPh)V) | not detectable | 28.74 | - | - | - | [34] |
| Cyanophenoxy | P(3HA-19.6 mol% 3H6pCPxHx) 1 | 53.5, >64 | −37.5, −21 | 15.1 | - | - | [35] |
| (P(3HA) | 55.5 | −35.4 | 19.3 | - | - | [35] | |
| Fluorophenoxy | P(3H5opFPxV) | 102 | - | - | 1.3 | 2.8 | [36] |
| P(3H5pFPxV-8.7 mol% 3H7pFPxHp) | 52 | - | - | 1.1 | 1.9 | [36] | |
| Thiophenoxy | P(3H5TPxV-3H7TPxHp) 2 | not detectable | 4 | - | 8.1 | 1.8 | [37] |
| Benzoyl | P(3HA-79.8%3H5BzV) 3 | 150 | 36 | - | 33 | 3.9 | [38] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishii-Hyakutake, M.; Mizuno, S.; Tsuge, T. Biosynthesis and Characteristics of Aromatic Polyhydroxyalkanoates. Polymers 2018, 10, 1267. https://doi.org/10.3390/polym10111267
Ishii-Hyakutake M, Mizuno S, Tsuge T. Biosynthesis and Characteristics of Aromatic Polyhydroxyalkanoates. Polymers. 2018; 10(11):1267. https://doi.org/10.3390/polym10111267
Chicago/Turabian StyleIshii-Hyakutake, Manami, Shoji Mizuno, and Takeharu Tsuge. 2018. "Biosynthesis and Characteristics of Aromatic Polyhydroxyalkanoates" Polymers 10, no. 11: 1267. https://doi.org/10.3390/polym10111267
APA StyleIshii-Hyakutake, M., Mizuno, S., & Tsuge, T. (2018). Biosynthesis and Characteristics of Aromatic Polyhydroxyalkanoates. Polymers, 10(11), 1267. https://doi.org/10.3390/polym10111267





