A Mild Method for Surface-Grafting PEG Onto Segmented Poly(Ester-Urethane) Film with High Grafting Density for Biomedical Purpose
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of SPEU and SPEU Films
2.3. Grafting of PEG on the SPEU Film Surface
2.4. Instruments and Characterization
3. Results and Discussion
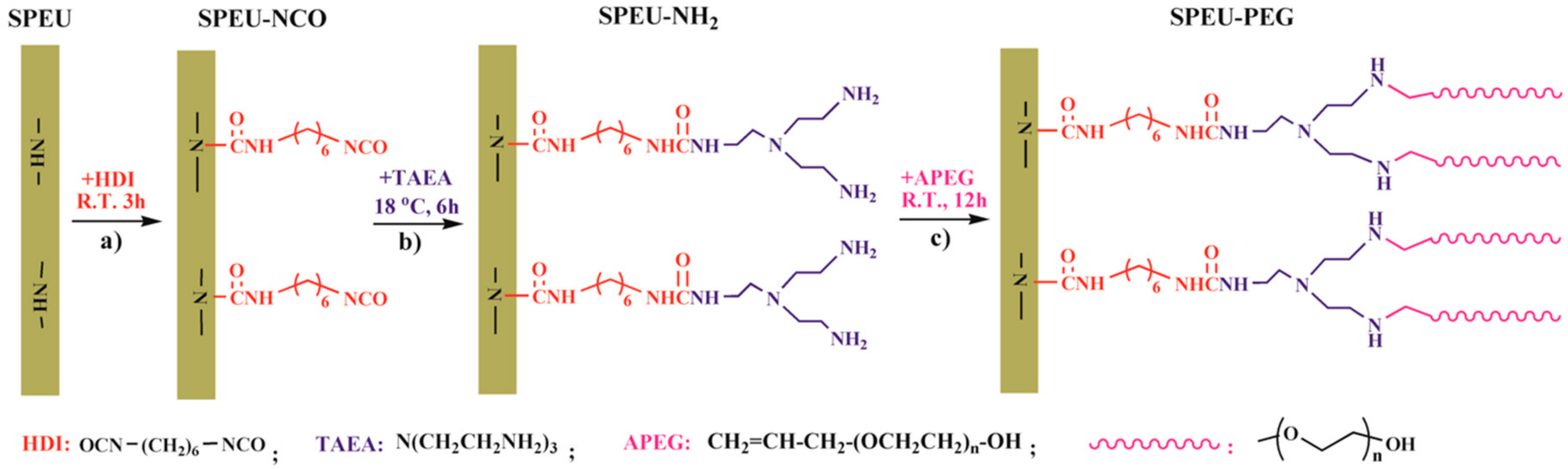
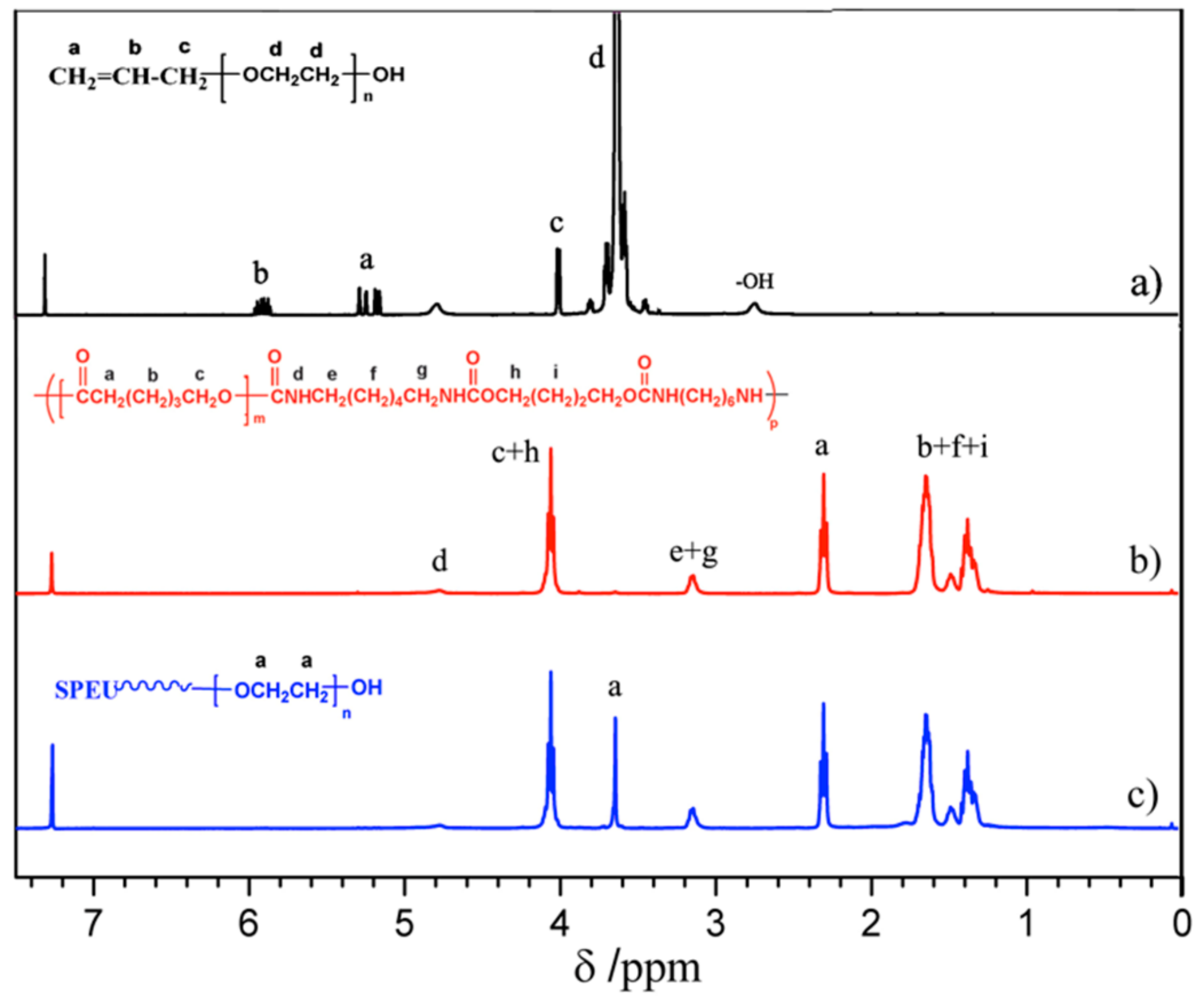
3.1. Preparation and Characterization
3.1.1. Activating SPEU Surface with HDI (SPEU-NCO)
3.1.2. Introducing -NH2 Group onto SPEU-NCO Film (SPEU-NH2)
3.1.3. Grafting of PEG onto SPEU-NH2 Film (SPEU-PEG)
3.2. XPS Analysis
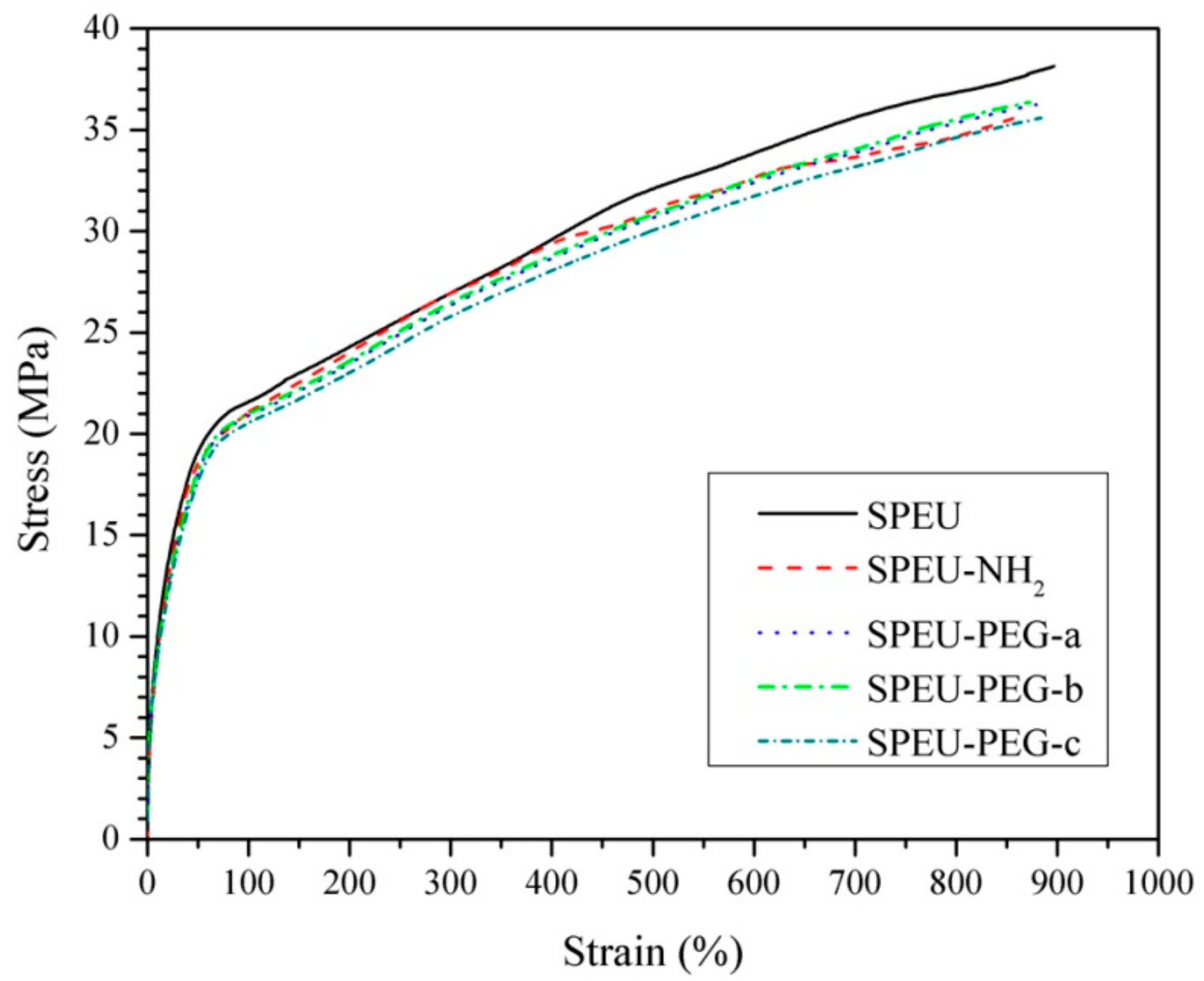
3.3. Mechanical Properties
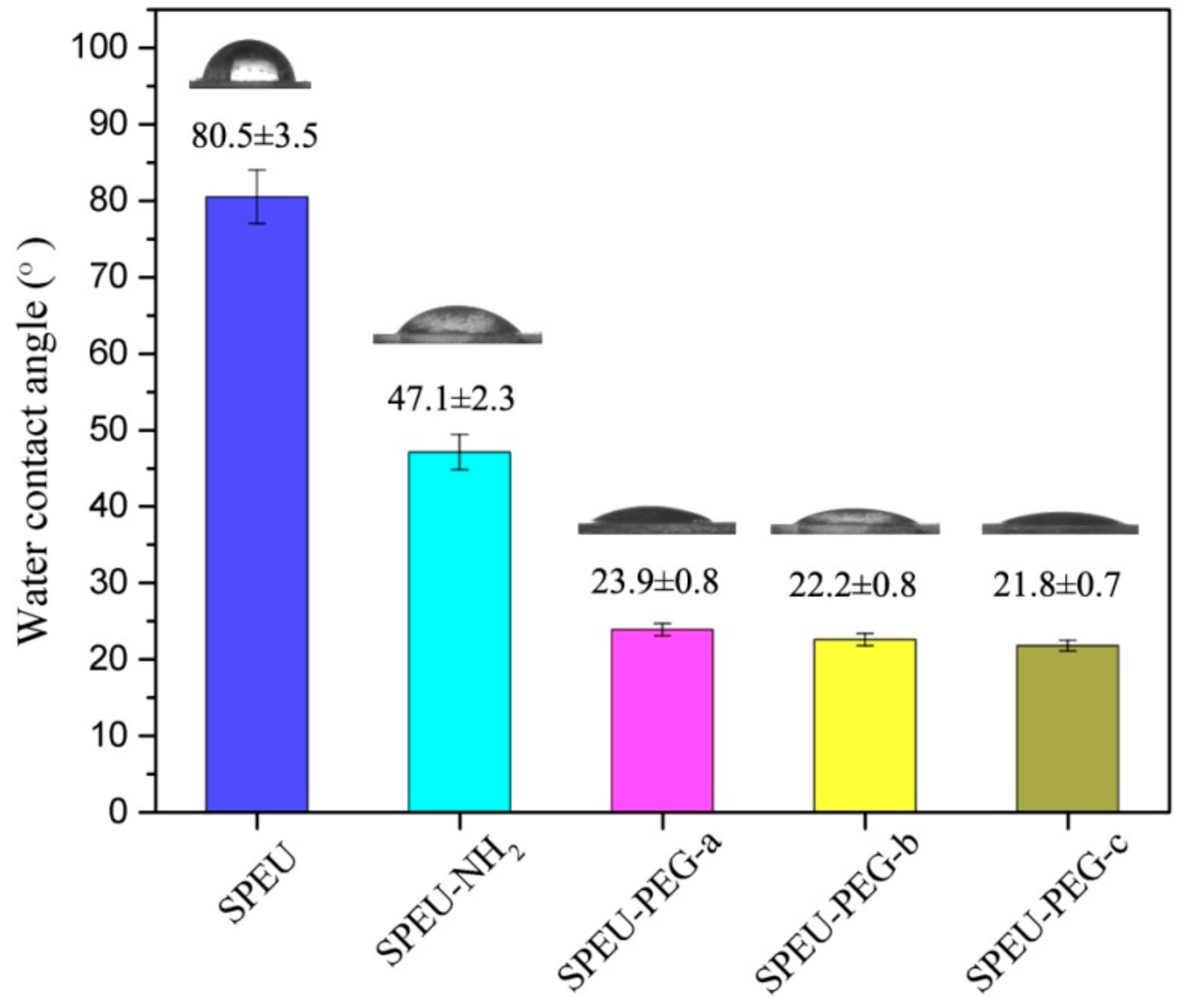
3.4. Surface Hydrophilicity and Swellability
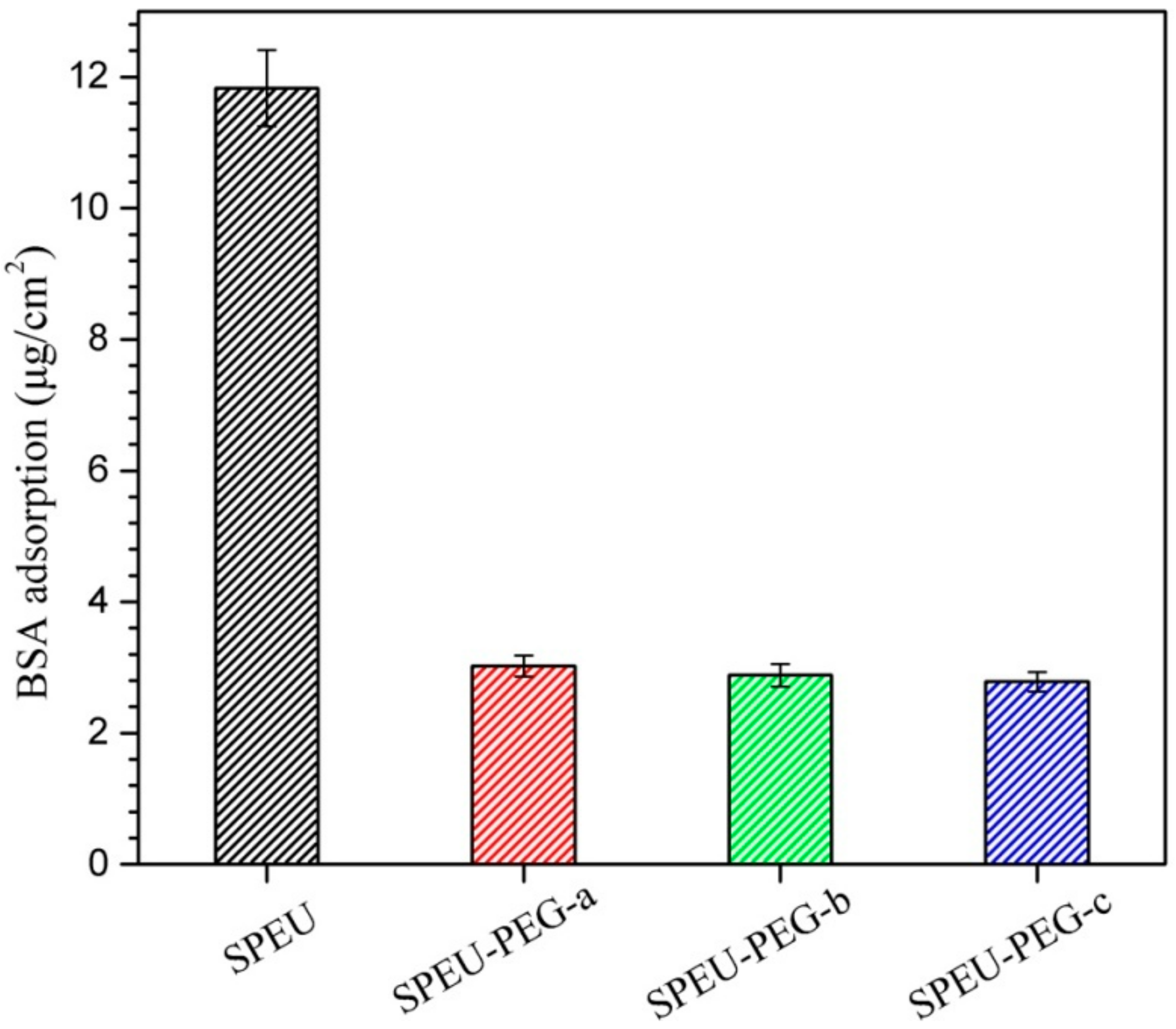
3.5. Protein Adsorption
3.6. Platelet Adhesion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ghanbari, H.; Viatge, H.; Kidane, A.G.; Burriesci, G.; Tavakoli, M.; Seifalian, A.M. Polymeric heart valves: New materials, emerging hopes. Trends Biotechnol. 2009, 27, 359–367. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Hu, Z.; Xu, A.; Liu, R.; Yin, H.; Wang, J.; Wang, S. The preparation and performance of a new polyurethane vascular prosthesis. Cell Biochem. Biophys. 2013, 66, 855–866. [Google Scholar] [CrossRef] [PubMed]
- Jozwiak, A.B.; Kielty, C.M.; Black, R.A. Surface functionalization of polyurethane for the immobilization of bioactive moieties on tissue scaffolds. J. Mater. Chem. 2018, 18, 2240–2248. [Google Scholar] [CrossRef]
- Qu, W.Q.; Xia, Y.R.; Jiang, L.J.; Zhang, L.W.; Hou, Z.S. Synthesis and characterization of a new biodegradable polyurethanes with good mechanical properties. Chin. Chem. Lett. 2016, 27, 135–138. [Google Scholar] [CrossRef]
- Tenorio-Alfonso, A.; Sánchez, M.C.; Franco, J.M. Preparation, characterization and mechanical properties of bio-based polyurethane adhesives from isocyanate-functionalized cellulose acetate and castor oil for bonding wood. Polymers 2017, 9, 132. [Google Scholar] [CrossRef]
- Hsu, S.; Chen, C.W.; Hung, K.C.; Tsai, Y.C. Li, S. Thermo-responsive polyurethane hydrogels based on poly(ε-caprolactone) diol and amphiphilic polylactide-poly(ethylene glycol) block copolymers. Polymers 2016, 8, 252. [Google Scholar] [CrossRef]
- Puskas, J.E.; Chen, Y. Biomedical application of commercial polymers and novel polyisobutylene-based thermoplastic elastomers for soft tissue replacement. Biomacromolecule 2004, 5, 1141–1154. [Google Scholar] [CrossRef] [PubMed]
- Bochynska, A.I.; Hammink, G.; Grijpma, D.W.; Buma, P. Tissue adhesives for meniscus tear repair: An overview of current advances and prospects for future clinical solutions. J. Mater. Sci. Mater. Med. 2016, 27, 85–102. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Topham, N.; Anderson, J.M.; Hiltner, A.; Lodoen, G.; Payer, C.R. Foreign-body giant cells and polyurethane biostability: In vivo correlation of cell adhesion and surface cracking. J. Biomed. Mater. Res. 1991, 25, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.H.; McNally, A.K.; Rubin, K.R.; Renier, M.; Wu, Y.; Rose-Caprara, V.; Anderson, J.M.; Hiltner, A.; Urbanski, P.; Stokes, K. Human plasma a 2-macroglobulin promotes in vitro oxidation stress cracking of Pellethane 2363-80A: In vivo and in vitro correlations. J. Biomed. Mater. Res. 1993, 27, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Lv, J.; Gao, B.; Zhang, L.; Yang, D.; Shi, C.; Guo, J. Modification of polycarbonateurethane surface with poly (ethylene glycol) monoacrylate and phosphorylcholine glyceraldehyde for anti-platelet adhesion. Front. Chem. Sci. Eng. 2014, 8, 188–196. [Google Scholar] [CrossRef]
- Kara, F.; Aksoy, E.A.; Yuksekdag, Z.; Hasirci, N.; Aksoy, S. Synthesis and surface modification of polyurethanes with chitosan for antibacterial properties. Carbohydr. Polym. 2014, 112, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Chen, H.; Glenn, M.W.; Brash, J.L. Lysine-PEG-modified polyurethane as a fibrinolytic surface: Effect of PEG chain length on protein interactions, platelet interactions and clot lysis. Acta Biomater. 2009, 5, 1864–1871. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Lee, H.B.; Andrade, J.D. Blood compatibility of polyethylene oxide surfaces. Prog. Polym. Sci. 1995, 20, 1043–1079. [Google Scholar] [CrossRef]
- Han, D.K.; Park, K.D.; Ryu, G.H.; Kim, U.K.; Min, B.G.; Kim, Y.H. Plasma protein adsorption to sulfonated poly(ethylene oxide)-grafted polyurethane surface. J. Biomed. Mater. Res. 1996, 30, 23–30. [Google Scholar] [CrossRef]
- Vert, M.; Domurado, D. Poly(ethylene glycol): Protein repulsiveor albumin-compatible. J. Biomater. Sci. Polym. Ed. 2000, 11, 1307–1317. [Google Scholar] [CrossRef] [PubMed]
- Najafabadi, S.A.A.; Keshvari, H.; Ganji, Y.; Tahriri, M.; Ashuri, M. Chitosan/heparin surface modified polyacrylic acid grafted polyurethane film by two step plasma treatment. Surf. Eng. 2012, 28, 710–714. [Google Scholar] [CrossRef]
- Irving, M.; Murphy, M.F.; Lilley, F.; French, P.W.; Burton, D.R.; Dixon, S.; Sharp, M.C. The use of abrasive polishing and laser processing for developing polyurethane surfaces for controlling fibroblast cell behavior. Mater. Sci. Eng. C 2017, 71, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Deng, H.; Li, Z.; Xiu, H.; Qi, X.; Zhang, Q.; Wang, K.; Chen, F.; Fu, Q. Graphene/thermoplastic polyurethane nanocomposites: Surface modification of graphene through oxidation, polyvinyl pyrrolidone coating and reduction. Compos. Part A Appl. Sci. Manuf. 2015, 68, 264–275. [Google Scholar] [CrossRef]
- Gao, B.; Feng, Y.; Lu, J.; Zhang, L.; Zhao, M.; Shi, C.; Khan, M.; Guo, J. Grafting of phosphorylcholine functional groups on polycarbonate urethane surface for resisting platelet adhesion. Mater. Sci. Eng. C 2013, 33, 2871–2878. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Miller, A.L., II; Fundora, K.A.; Yaszemski, M.J.; Lu, L. Poly(ε-caprolactone) dendrimer cross-Linked via metal-free click chemistry: Injectable hydrophobic platform for tissue engineering. ACS Macro Lett. 2016, 5, 1261–1265. [Google Scholar] [CrossRef]
- Zhang, C.; Luo, N.; Hirt, D.E. Surface grafting polyethylene glycol (PEG) onto poly(ethylene-co-acrylic acid) films. Langmuir 2006, 22, 6851–6857. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Hu, X.; Zhang, Y.; Li, D.; Wu, Z.; Zhang, T. Effect of chain density and conformation on protein adsorption at PEG-grafted polyurethane surfaces. Colloids Surf. B 2008, 61, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Alves, P.; Coelho, J.F.J.; Haack, J.; Rota, A.; Bruinink, A.; Gil, M.H. Surface modification and characterization of thermoplastic polyurethane. Eur. Polym. J. 2009, 45, 1412–1419. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Xia, Y.; Liu, L.; Zhang, D.; Hou, Z. Synthesis of a novel biomedical poly(ester urethane) based on aliphatic uniform-size diisocyanate and the blood compatibility of PEG-grafted surfaces. J. Biomater. Appl. 2018, 32, 1329–1342. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, Z.; Khan, A.S.; Roohpour, N.; Glogauer, M.; Rehman, I.U. Protein adsorption capability on polyurethane and modified-polyurethane membrane for periodontal guided tissue regeneration applications. Mater. Sci. Eng. C 2016, 68, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Roohpour, N.; Wasikiewicz, J.M.; Moshaverinia, A.; Paul, D.; Grahn, M.F.; Rehman, I.U.; Vadgama, P. Polyurethane membranes modified with isopropyl myristate as a potential candidate for encapsulating electronic implants: A study of biocompatibility and water permeability. Polymers 2010, 2, 102–119. [Google Scholar] [CrossRef]
- Xu, W.; Xiao, M.; Yuan, L.; Zhang, J.; Hou, Z. Preparation, physicochemical properties and hemocompatibility of biodegradable chitooligosaccharide-based polyurethane. Polymers 2018, 10, 580. [Google Scholar] [CrossRef]
- Zhang, N.; Yin, S.; Hou, Z.; Xu, W.; Zhang, J.; Xiao, M.; Zhang, Q. Preparation, physicochemical properties and biocompatibility of biodegradable poly(ether-ester-urethane) and chitosan oligosaccharide composites. J. Polym. Res. 2018, 25, 212. [Google Scholar] [CrossRef]
- Hou, Z.; Qu, W.; Kan, C. Synthesis and properties of triethoxysilane-terminated anionic polyurethane and its waterborne dispersions. J. Polym. Res. 2015, 22, 111. [Google Scholar] [CrossRef]
- Park, K.D.; Kim, Y.S.; Han, D.K.; Kim, Y.H.; Lee, E.H.B.; Suh, H.; Choi, K.S. Bacterial adhesion on PEG modified polyurethane surfaces. Biomaterials 1998, 19, 851–859. [Google Scholar] [CrossRef]
- Lee, K.W.; Kowalczyk, S.P.; Shaw, J.M. Surface modification of BPDA-PDA polyimide. Langmuir 1991, 7, 2450–2453. [Google Scholar] [CrossRef]
- Gao, W.; Feng, Y.; Lu, J.; Khan, M.; Guo, J. Biomimetic surface modification of polycarbonateurethane film via phosphorylcholine-graft for resisting platelet adhesion. Macromol. Res. 2012, 20, 1063–1069. [Google Scholar] [CrossRef]
- Zhou, Y.; Nie, W.; Zhao, J.; Yuan, X. Rapidly in situ forming adhesive hydrogel based on a PEG-maleimide modified polypeptide through Michael addition. J. Mater. Sci. Mater. Med. 2013, 24, 2277–2286. [Google Scholar] [CrossRef] [PubMed]
- Caracciolo, P.C.; Queiroz, A.A.D.; Higa, Q.Z.; Buffa, F.; Abraham, G.A. Segmented poly(esterurethane urea)s from novel urea-diol chain extenders: Synthesis, characterization and in vitro biological properties. Acta Biomater. 2008, 4, 976–988. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Xia, Y.; Jia, Q.; Hou, Z.; Zhang, N. Preparation and properties of biomedical segmented polyurethanes based on poly(ether ester) and uniform-size diurethane diisocyanates. J. Biomater. Sci. Polym. Ed. 2017, 28, 119–138. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.J.; Bae, J.H.; Kim, J.J.; Na, K.; Lee, E.S. Long acting porous microparticle for pulmonary protein delivery. Int. J. Pharm. 2007, 333, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.; Jacobs, H.A.; Kim, S.W. Surface adsorption and fibrinogen interactions with hirudin-thrombin complex. J. Biomed. Mater. Res. 1996, 32, 11–18. [Google Scholar] [CrossRef]
- Ren, Z.; Chen, G.; Wei, Z.; Sang, L.; Qi, M. Hemocompatibility evaluation of polyurethane film with surface-grafted poly(ethylene glycol) and carboxymethyl-chitosan. J. Appl. Polym. Sci. 2013, 127, 308–315. [Google Scholar] [CrossRef]
- Malmsten, M.; Emoto, K.; Alstine, J.M.V. Effect of chain density on inhibition of protein adsorption by poly(ethylene glycol) based coatings. J. Colloid Interface Sci. 1998, 202, 507–517. [Google Scholar] [CrossRef]

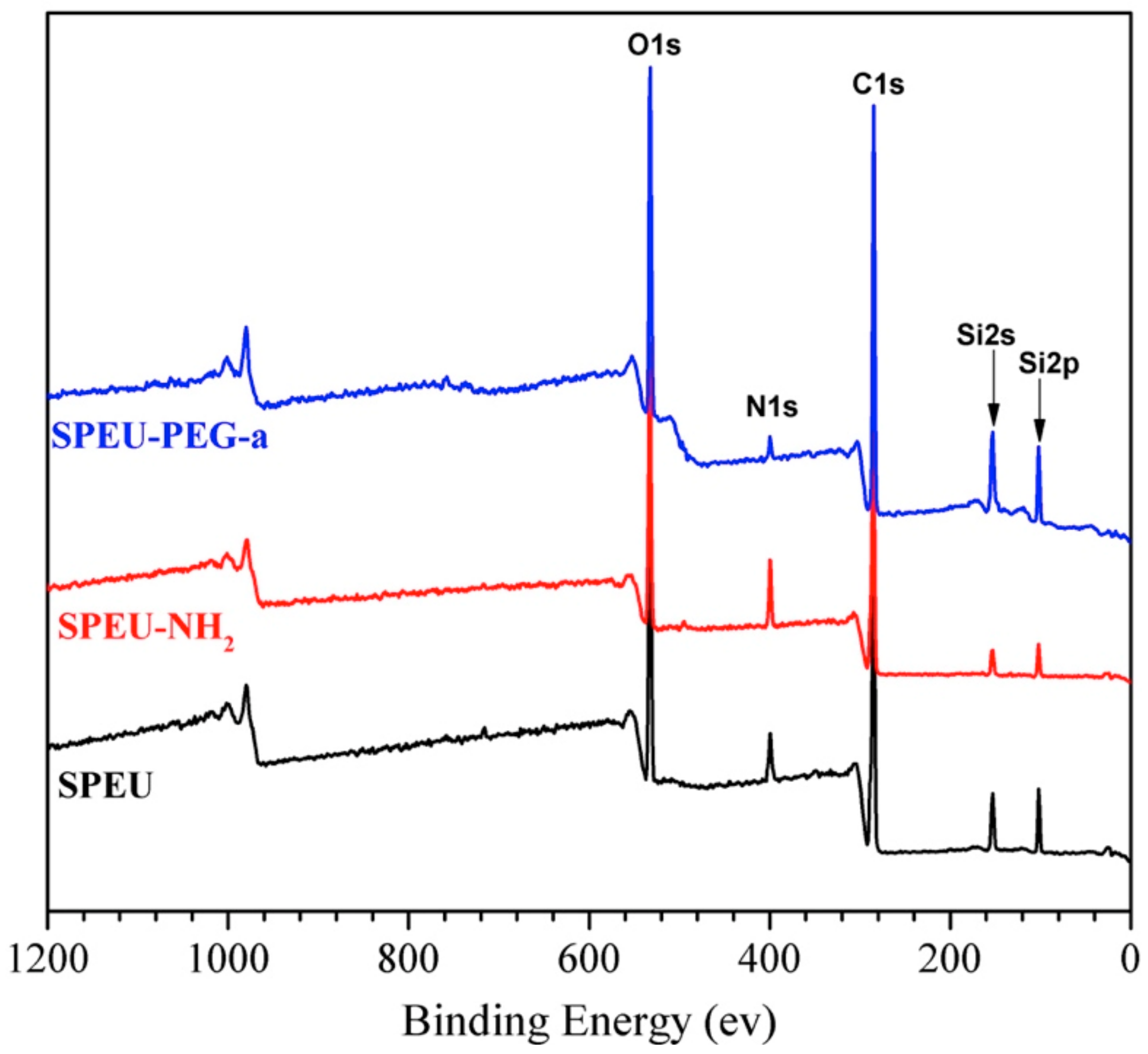

| Films | Atomic Content/% | ||
|---|---|---|---|
| C1s | O1s | N1s | |
| SPEU | 78.2 | 18.7 | 3.1 |
| SPEU-NH2 | 74.8 | 14.9 | 10.3 |
| SPEU-PEG-a | 72.1 | 25.7 | 2.2 |
| SPEU-PEG-b | 71.3 | 27.1 | 1.6 |
| SPEU-PEG-c | 70.2 | 28.4 | 1.4 |
| Films | Strain at Break (%) | Ultimate Stress (MPa) | Yield Strain (%) | Yield Stress (MPa) | Initial Modulus (MPa) |
|---|---|---|---|---|---|
| SPEU | 896 ± 25 | 38.1 ± 2.1 | 46.7 ± 2.6 | 20.2 ± 1.2 | 43.3 |
| SPEU-NH2 | 866 ± 16 | 35.7 ± 1.8 | 45.8 ± 2.1 | 19.3 ± 1.1 | 42.1 |
| SPEU-PEG-a | 873 ± 18 | 36.1 ± 1.4 | 52.7 ± 2.2 | 19.8 ± 0.9 | 37.6 |
| SPEU-PEG-b | 879 ± 21 | 36.4 ± 1.9 | 56.8 ± 2.0 | 20.1 ± 1.1 | 35.4 |
| SPEU-PEG-c | 884 ± 20 | 35.5 ± 1.7 | 58.7 ± 1.9 | 19.9 ± 1.3 | 34.0 |
| Films | SPEU | SPEU-PEG-a | SPEU-PEG-b | SPEU-PEG-c |
|---|---|---|---|---|
| Quantity of adhered platelets (per mm2) | 20,702 ± 880 | 781 ± 56 | 731 ± 57 | 697 ± 52 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Gao, Y.; Zhao, J.; Yuan, L.; Li, C.; Liu, Z.; Hou, Z. A Mild Method for Surface-Grafting PEG Onto Segmented Poly(Ester-Urethane) Film with High Grafting Density for Biomedical Purpose. Polymers 2018, 10, 1125. https://doi.org/10.3390/polym10101125
Liu L, Gao Y, Zhao J, Yuan L, Li C, Liu Z, Hou Z. A Mild Method for Surface-Grafting PEG Onto Segmented Poly(Ester-Urethane) Film with High Grafting Density for Biomedical Purpose. Polymers. 2018; 10(10):1125. https://doi.org/10.3390/polym10101125
Chicago/Turabian StyleLiu, Lulu, Yuanyuan Gao, Juan Zhao, Litong Yuan, Chenglin Li, Zhaojun Liu, and Zhaosheng Hou. 2018. "A Mild Method for Surface-Grafting PEG Onto Segmented Poly(Ester-Urethane) Film with High Grafting Density for Biomedical Purpose" Polymers 10, no. 10: 1125. https://doi.org/10.3390/polym10101125
APA StyleLiu, L., Gao, Y., Zhao, J., Yuan, L., Li, C., Liu, Z., & Hou, Z. (2018). A Mild Method for Surface-Grafting PEG Onto Segmented Poly(Ester-Urethane) Film with High Grafting Density for Biomedical Purpose. Polymers, 10(10), 1125. https://doi.org/10.3390/polym10101125






