Self-Assembled, Adjuvant/Antigen-Based Nanovaccine Mediates Anti-Tumor Immune Response against Melanoma Tumor
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
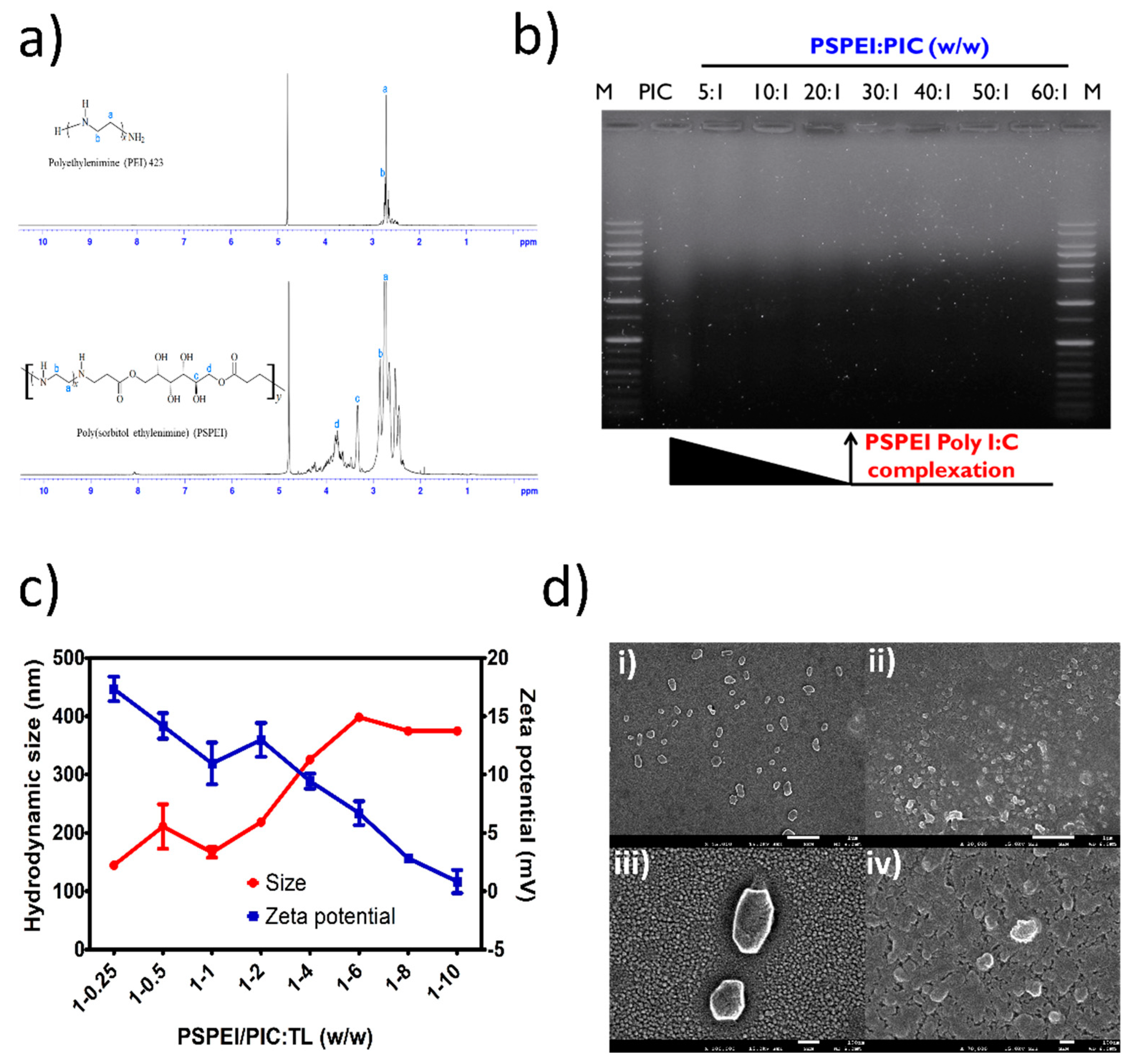
2.2. Synthesis of PSPEI
2.3. Tumor Cell Lysate
2.4. Formulation of PSPEI Complexed PIC and TCL (PSPEI-PAA)
2.5. Characterization of PSPEI-PAA
2.6. Scanning Electron Microscope Imaging
2.7. Isolation of Bone Marrow Derived Dendritic Cells (BMDCs)
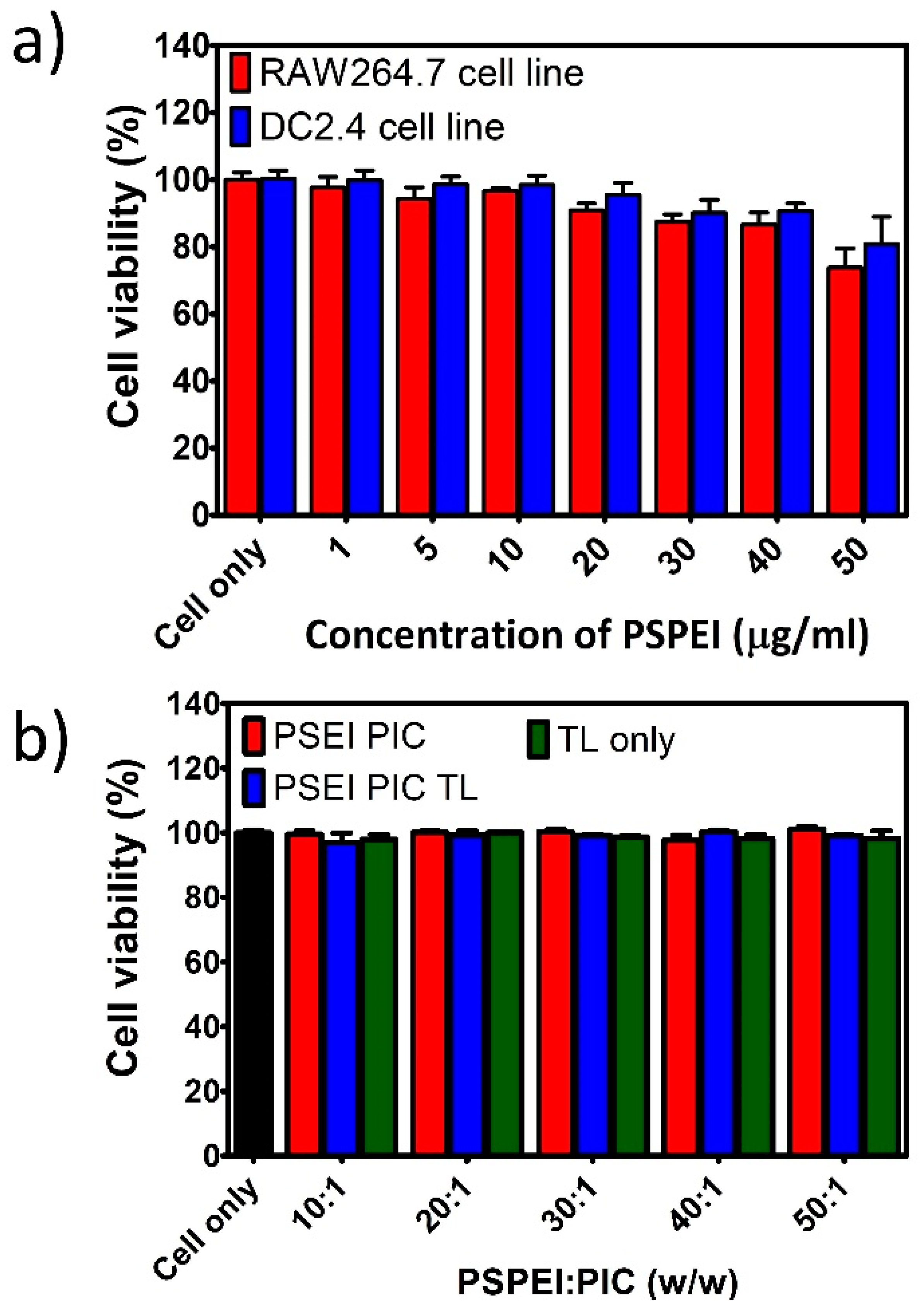
2.8. In Vitro Cell Viability Assay Using MTS Reagent
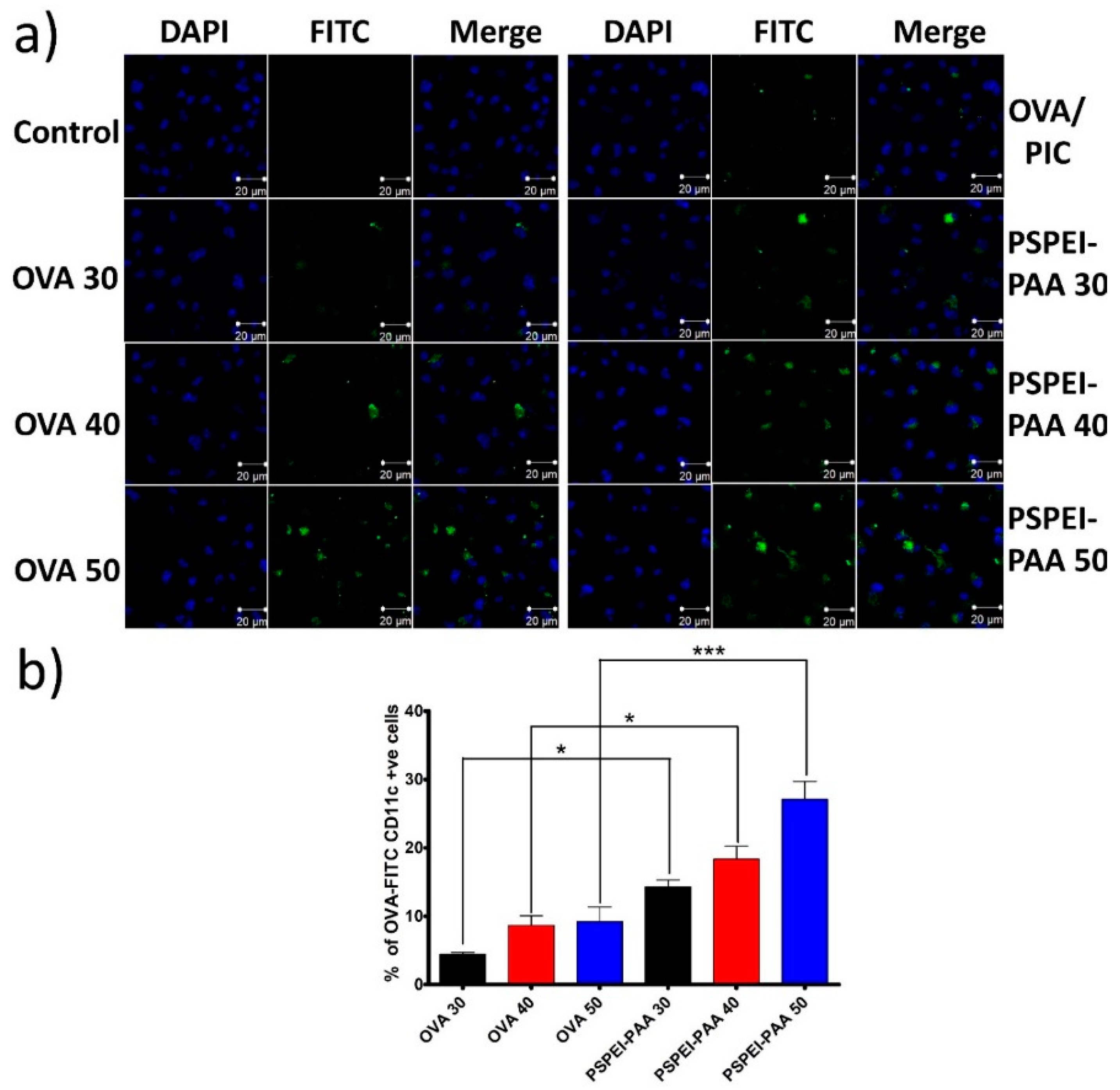
2.9. Confocal Microscope Imaging
2.10. Fluorescence-Activated Cell Sorter (FACS) Analysis
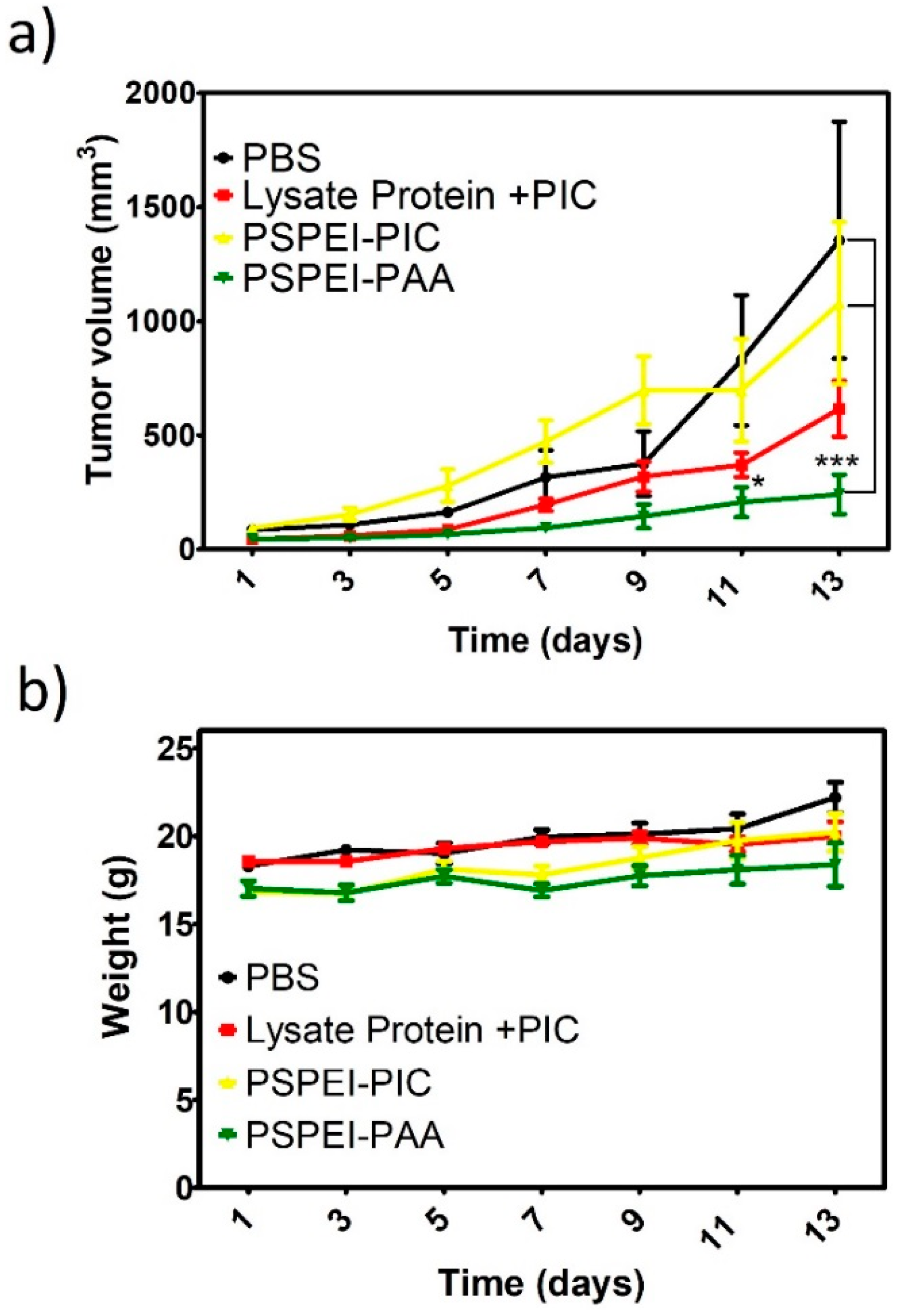
2.11. B1610 Tumor Model, Animal Grouping and Immunization
2.12. Isolation of Immune Cells from Primary Tumor and Tumor Draining Lymph Node
2.13. Splenocyte Proliferation Assay
2.14. Statistical Analysis
3. Results
3.1. Physiochemical Characterization of Polysorbital-Polyethylenimine (PSPEI) Complexed with Poly I:C (PIC) and Tumor Lysate (TL) (PSPEI-PAA) Nanocomplex
3.2. Viability of Immune Cells Treated with PSPEI-PAA Nanocomplex
3.3. Intracellular Uptake of PSPEI-PAA Nanocomplex in Immature Dendritic Cells
3.4. Bone Marrow Derived Dendritic Cell Maturation by PSPEI-PAA Nanocomplex
3.5. Antitumor Activity of PSPEI-PAA Nanocomplex in B16F10 Tumor Model
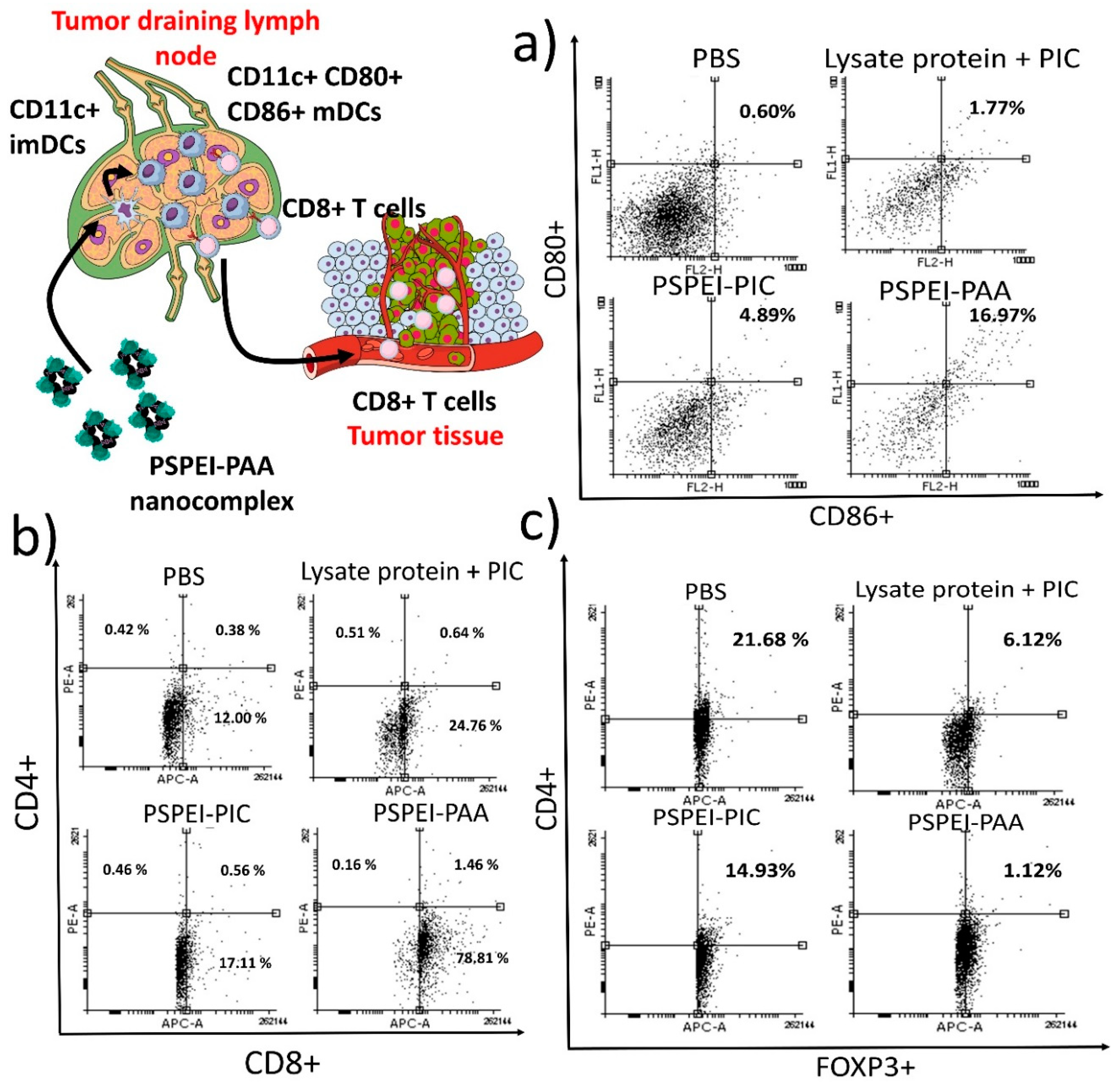
3.6. Characterization of Immune Cells from Tumor Draining Lymph Node and Tumor
3.7. Antitumor Immune Response of PSPEI-PAA Nanocomplex Activated Splenocytes
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Sanlorenzo, M.; Vujic, I.; Posch, C.; Dajee, A.; Yen, A.; Kim, S.; Ashworth, M.; Rosenblum, M.D.; Algazi, A.; Osella-Abate, S.; et al. Melanoma immunotherapy. Cancer Biol. Ther. 2014, 15, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Ozao-Choy, J.; Lee, D.J.; Faries, M.B. Melanoma vaccines: Mixed past, promising future. Surg. Clin. N. Am. 2014, 94, 1017–1030. [Google Scholar] [CrossRef] [PubMed]
- Yushak, M.L.; Chiang, V.L.; Kluger, H.M. Clinical trials in melanoma patients with brain metastases. Pigment Cell Melanoma Res. 2015, 28, 741–743. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, J.L.; Sondel, P.M. Enhancing cancer immunotherapy via activation of innate immunity. Semin. Oncol. 2015, 42, 562–572. [Google Scholar] [CrossRef] [PubMed]
- Takakura, K.; Kajihara, M.; Ito, Z.; Ohkusa, T.; Gong, J.; Koido, S. Dendritic-tumor fusion cells in cancer immunotherapy. Discov. Med. 2015, 19, 169–174. [Google Scholar] [PubMed]
- Maus, M.V.; Fraietta, J.A.; Levine, B.L.; Kalos, M.; Zhao, Y.; June, C.H. Adoptive immunotherapy for cancer or viruses. Annu. Rev. Immunol. 2014, 32, 189–225. [Google Scholar] [CrossRef]
- Zhang, X.; Kang, Z.; Li, Q.; Zhang, J.; Cheng, S.; Chang, H.; Wang, S.; Cao, S.; Li, T.; Li, J.; et al. Antigen-adjuvant effects of icariin in enhancing tumor-specific immunity in mastocytoma-bearing dba/2j mice. Biomed. Pharmacother. 2018, 99, 810–816. [Google Scholar] [CrossRef]
- Seya, T.; Shime, H.; Takeda, Y.; Tatematsu, M.; Takashima, K.; Matsumoto, M. Adjuvant for vaccine immunotherapy of cancer--focusing on toll-like receptor 2 and 3 agonists for safely enhancing antitumor immunity. Cancer Sci. 2015, 106, 1659–1668. [Google Scholar] [CrossRef]
- Schijns, V.; Tartour, E.; Michalek, J.; Stathopoulos, A.; Dobrovolskiene, N.T.; Strioga, M.M. Immune adjuvants as critical guides directing immunity triggered by therapeutic cancer vaccines. Cytotherapy 2014, 16, 427–439. [Google Scholar] [CrossRef]
- Yuan, S.; Shi, C.; Liu, L.; Han, W. Muc1-based recombinant bacillus calmette-guerin vaccines as candidates for breast cancer immunotherapy. Expert Opin. Biol. Ther. 2010, 10, 1037–1048. [Google Scholar] [CrossRef] [PubMed]
- O’Hagan, D.T.; Friedland, L.R.; Hanon, E.; Didierlaurent, A.M. Towards an evidence based approach for the development of adjuvanted vaccines. Curr. Opin. Immunol. 2017, 47, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Bowen, W.S.; Svrivastava, A.K.; Batra, L.; Barsoumian, H.; Shirwan, H. Current challenges for cancer vaccine adjuvant development. Expert Rev. Vaccines 2018, 17, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Shao, K.; Singha, S.; Clemente-Casares, X.; Tsai, S.; Yang, Y.; Santamaria, P. Nanoparticle-based immunotherapy for cancer. ACS Nano 2015, 9, 16–30. [Google Scholar] [CrossRef] [PubMed]
- Zang, X.; Zhao, X.; Hu, H.; Qiao, M.; Deng, Y.; Chen, D. Nanoparticles for tumor immunotherapy. Eur. J. Pharm. Biopharm. 2017, 115, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, C.H.; Perry, J.L.; Tian, S.; Luft, J.C.; DeSimone, J.M. Nanoparticulate immunotherapy for cancer. J. Control. Release 2015, 219, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Musetti, S.N.; Huang, L. Nanomaterials for cancer immunotherapy. Biomaterials 2017, 148, 16–30. [Google Scholar] [CrossRef]
- Saleh, T.; Shojaosadati, S.A. Multifunctional nanoparticles for cancer immunotherapy. Hum. Vaccinnes Immunother. 2016, 12, 1863–1875. [Google Scholar] [CrossRef]
- Fang, R.H.; Kroll, A.V.; Zhang, L. Nanoparticle-based manipulation of antigen-presenting cells for cancer immunotherapy. Small 2015, 11, 5483–5496. [Google Scholar] [CrossRef]
- Firdous, J.; Islam, M.A.; Park, S.M.; Cheon, I.S.; Shim, B.S.; Yoon, H.S.; Song, M.; Chang, J.; Choi, Y.J.; Park, Y.M.; et al. Induction of long-term immunity against respiratory syncytial virus glycoprotein by an osmotic polymeric nanocarrier. Acta Biomater. 2014, 10, 4606–4617. [Google Scholar] [CrossRef]
- Nguyen, K.C.; Muthiah, M.; Islam, M.A.; Kalash, R.S.; Cho, C.S.; Park, H.; Lee, I.K.; Kim, H.J.; Park, I.K.; Cho, K.A. Selective transfection with osmotically active sorbitol modified pei nanoparticles for enhanced anti-cancer gene therapy. Colloids Surf. B Biointerfaces 2014, 119, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Lu, H. Tlr agonists for cancer immunotherapy: Tipping the balance between the immune stimulatory and inhibitory effects. Front. Immunol. 2014, 5, 83. [Google Scholar] [CrossRef]
- Stier, S.; Maletzki, C.; Klier, U.; Linnebacher, M. Combinations of TLR ligands: A promising approach in cancer immunotherapy. Clin. Dev. Immunol. 2013, 2013, 271246. [Google Scholar] [CrossRef] [PubMed]
- Ayari, C.; Besancon, M.; Bergeron, A.; LaRue, H.; Bussieres, V.; Fradet, Y. Poly(I:C) potentiates bacillus calmette-guerin immunotherapy for bladder cancer. Cancer Immunol. Immunother. 2016, 65, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Forghani, P.; Waller, E.K. Poly (I:C) modulates the immunosuppressive activity of myeloid-derived suppressor cells in a murine model of breast cancer. Breast Cancer Res. Treat. 2015, 153, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Hou, Z.; Han, Q.; Zhang, C.; Tian, Z.; Zhang, J. Poly(i:C) exhibits an anti-cancer effect in human gastric adenocarcinoma cells which is dependent on rlrs. Int. Immunopharmacol. 2013, 17, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.K.; Yadav, P.K.; Tiwari, A.K.; Gandham, R.K.; Sahoo, A.P. Poly (I:C) enhances the anti-tumor activity of canine parvovirus ns1 protein by inducing a potent anti-tumor immune response. Tumour. Biol. 2016, 37, 12089–12102. [Google Scholar] [CrossRef] [PubMed]
- Salazar, A.M.; Erlich, R.B.; Mark, A.; Bhardwaj, N.; Herberman, R.B. Therapeutic in situ autovaccination against solid cancers with intratumoral poly-iclc: Case report, hypothesis, and clinical trial. Cancer Immunol. Res. 2014, 2, 720–724. [Google Scholar] [CrossRef] [PubMed]
- Pitcovski, J.; Shahar, E.; Aizenshtein, E.; Gorodetsky, R. Melanoma antigens and related immunological markers. Crit. Rev. Oncol. Hematol. 2017, 115, 36–49. [Google Scholar] [CrossRef]
- Andrews, M.C.; Woods, K.; Cebon, J.; Behren, A. Evolving role of tumor antigens for future melanoma therapies. Future Oncol. 2014, 10, 1457–1468. [Google Scholar] [CrossRef]
- Pichon, C.; Midoux, P. Mannosylated and histidylated lpr technology for vaccination with tumor antigen mrna. Methods Mol. Biol. 2013, 969, 247–274. [Google Scholar] [PubMed]
- Liu, L.N.; Shivakumar, R.; Allen, C.; Fratantoni, J.C. Delivery of whole tumor lysate into dendritic cells for cancer vaccination. Methods Mol. Biol. 2008, 423, 139–153. [Google Scholar] [PubMed]
- Cho, K.-H.; Singh, B.; Maharjan, S.; Jang, Y.; Choi, Y.-J.; Cho, C.-S. Local delivery of ctgf sirna with poly(sorbitol-co-pei) reduces scar contraction in cutaneous wound healing. Tissue Eng. Regen. Med. 2017, 14, 211–220. [Google Scholar] [CrossRef]
- Luu, Q.P.; Shin, J.Y.; Kim, Y.K.; Islam, M.A.; Kang, S.K.; Cho, M.H.; Choi, Y.J.; Cho, C.S. High gene transfer by the osmotic polysorbitol-mediated transporter through the selective caveolae endocytic pathway. Mol. Pharm. 2012, 9, 2206–2218. [Google Scholar] [CrossRef] [PubMed]
- Schaffert, D.; Kiss, M.; Rodl, W.; Shir, A.; Levitzki, A.; Ogris, M.; Wagner, E. Poly(I:C)-mediated tumor growth suppression in egf-receptor overexpressing tumors using egf-polyethylene glycol-linear polyethylenimine as carrier. Pharm. Res. 2011, 28, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Syga, M.I.; Nicoli, E.; Kohler, E.; Shastri, V.P. Albumin incorporation in polyethylenimine-DNA polyplexes influences transfection efficiency. Biomacromolecules 2016, 17, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Cho, W.Y.; Hong, S.H.; Singh, B.; Islam, M.A.; Lee, S.; Lee, A.Y.; Gankhuyag, N.; Kim, J.E.; Yu, K.N.; Kim, K.H.; et al. Suppression of tumor growth in lung cancer xenograft model mice by poly(sorbitol-co-pei)-mediated delivery of osteopontin sirna. Eur. J. Pharm. Biopharm. 2015, 94, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Chou, M.J.; Yu, H.Y.; Hsia, J.C.; Chen, Y.H.; Hung, T.T.; Chao, H.M.; Chern, E.; Huang, Y.Y. Highly efficient intracellular protein delivery by cationic polyethyleneimine-modified gelatin nanoparticles. Materials 2018, 11, 301. [Google Scholar] [CrossRef]
- Menon, J.U.; Ravikumar, P.; Pise, A.; Gyawali, D.; Hsia, C.C.; Nguyen, K.T. Polymeric nanoparticles for pulmonary protein and DNA delivery. Acta Biomater. 2014, 10, 2643–2652. [Google Scholar] [CrossRef]
- Kafil, V.; Omidi, Y. Cytotoxic impacts of linear and branched polyethylenimine nanostructures in a431 cells. Bioimpacts 2011, 1, 23–30. [Google Scholar]
- Han, H.D.; Byeon, Y.; Jang, J.H.; Jeon, H.N.; Kim, G.H.; Kim, M.G.; Pack, C.G.; Kang, T.H.; Jung, I.D.; Lim, Y.T.; et al. In vivo stepwise immunomodulation using chitosan nanoparticles as a platform nanotechnology for cancer immunotherapy. Sci. Rep. 2016, 6, 38348. [Google Scholar] [CrossRef] [PubMed]
- Salem, M.L.; Diaz-Montero, C.M.; El-Naggar, S.A.; Chen, Y.; Moussa, O.; Cole, D.J. The tlr3 agonist poly(I:C) targets cd8+ t cells and augments their antigen-specific responses upon their adoptive transfer into naive recipient mice. Vaccine 2009, 27, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Pulko, V.; Liu, X.; Krco, C.J.; Harris, K.J.; Frigola, X.; Kwon, E.D.; Dong, H. Tlr3-stimulated dendritic cells up-regulate b7-h1 expression and influence the magnitude of cd8 t cell responses to tumor vaccination. J. Immunol. 2009, 183, 3634–3641. [Google Scholar] [CrossRef] [PubMed]
- Chandran, S.S.; Paria, B.C.; Srivastava, A.K.; Rothermel, L.D.; Stephens, D.J.; Kammula, U.S. Tumor-specific effector cd8+ t cells that can establish immunological memory in humans after adoptive transfer are marked by expression of il7 receptor and c-myc. Cancer Res. 2015, 75, 3216–3226. [Google Scholar] [CrossRef] [PubMed]
- Sckisel, G.D.; Mirsoian, A.; Minnar, C.M.; Crittenden, M.; Curti, B.; Chen, J.Q.; Blazar, B.R.; Borowsky, A.D.; Monjazeb, A.M.; Murphy, W.J. Differential phenotypes of memory cd4 and cd8 t cells in the spleen and peripheral tissues following immunostimulatory therapy. J. Immunother. Cancer 2017, 5, 33. [Google Scholar] [CrossRef] [PubMed]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajendrakumar, S.K.; Mohapatra, A.; Singh, B.; Revuri, V.; Lee, Y.-K.; Kim, C.S.; Cho, C.-S.; Park, I.-K. Self-Assembled, Adjuvant/Antigen-Based Nanovaccine Mediates Anti-Tumor Immune Response against Melanoma Tumor. Polymers 2018, 10, 1063. https://doi.org/10.3390/polym10101063
Rajendrakumar SK, Mohapatra A, Singh B, Revuri V, Lee Y-K, Kim CS, Cho C-S, Park I-K. Self-Assembled, Adjuvant/Antigen-Based Nanovaccine Mediates Anti-Tumor Immune Response against Melanoma Tumor. Polymers. 2018; 10(10):1063. https://doi.org/10.3390/polym10101063
Chicago/Turabian StyleRajendrakumar, Santhosh Kalash, Adityanarayan Mohapatra, Bijay Singh, Vishnu Revuri, Yong-Kyu Lee, Chang Seong Kim, Chong-Su Cho, and In-Kyu Park. 2018. "Self-Assembled, Adjuvant/Antigen-Based Nanovaccine Mediates Anti-Tumor Immune Response against Melanoma Tumor" Polymers 10, no. 10: 1063. https://doi.org/10.3390/polym10101063
APA StyleRajendrakumar, S. K., Mohapatra, A., Singh, B., Revuri, V., Lee, Y.-K., Kim, C. S., Cho, C.-S., & Park, I.-K. (2018). Self-Assembled, Adjuvant/Antigen-Based Nanovaccine Mediates Anti-Tumor Immune Response against Melanoma Tumor. Polymers, 10(10), 1063. https://doi.org/10.3390/polym10101063





