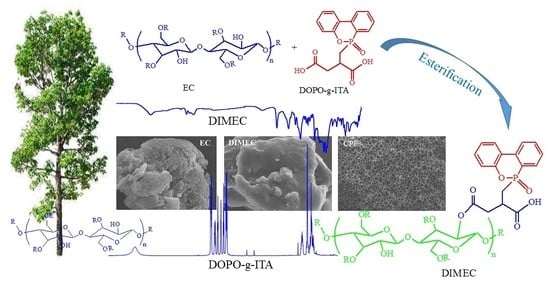
Preparation and Characterization of DOPO-ITA Modified Ethyl Cellulose and Its Application in Phenolic Foams
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Synthesis of DOPO-ITA
2.2.2. Preparation of DOPO-ITA Modified EC (DIMEC)
2.2.3. Preparation of Composite PFs
2.3. Characterizations
3. Results and Discussion
3.1. The Properties of DOPO-ITA
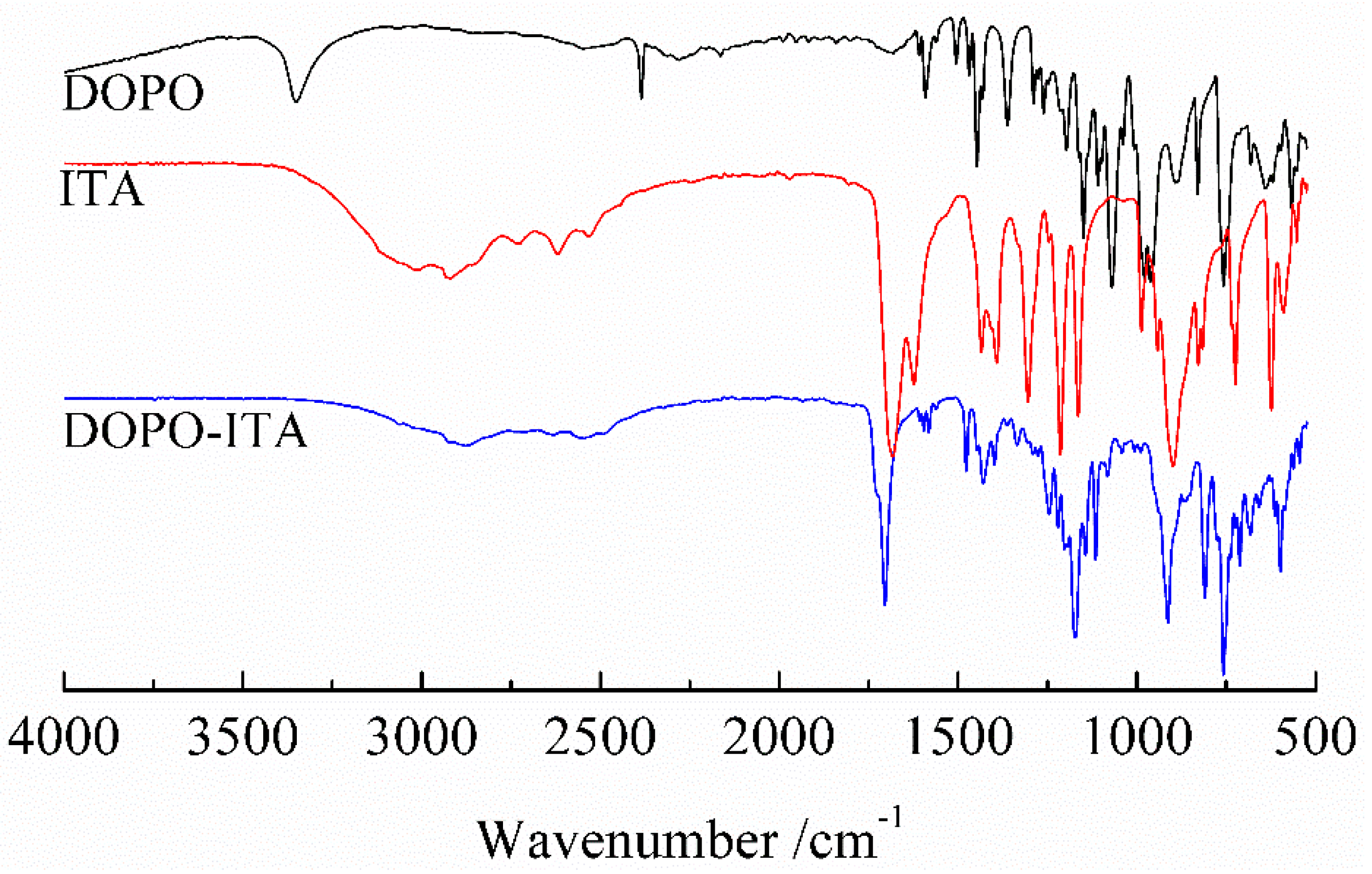
3.1.1. FT-IR of DOPO-ITA
3.1.2. 1H NMR of DOPO-ITA
3.2. The Properties of DOPO-ITA Modified EC (DIMEC)
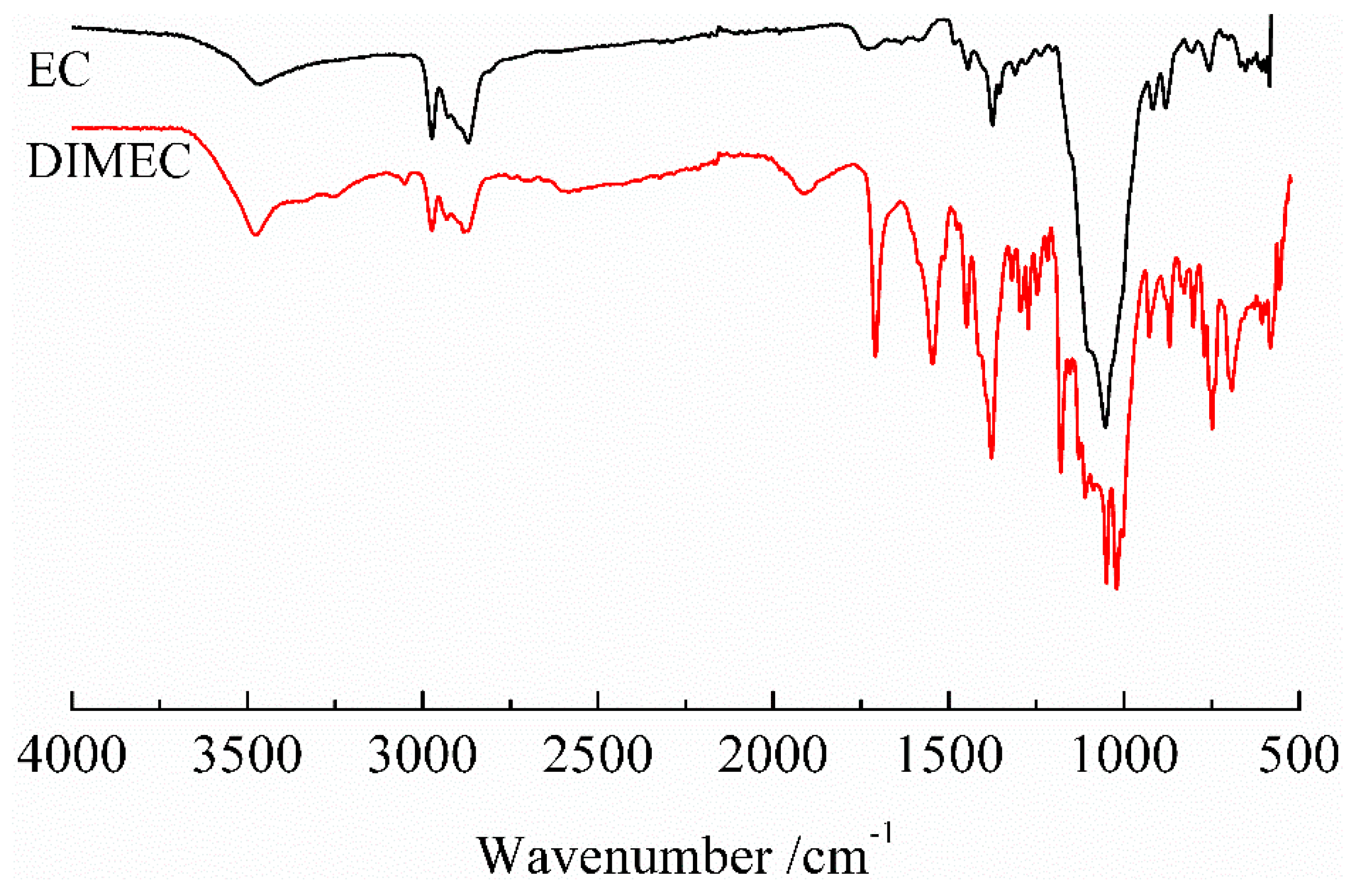
3.2.1. FT-IR of DIMEC
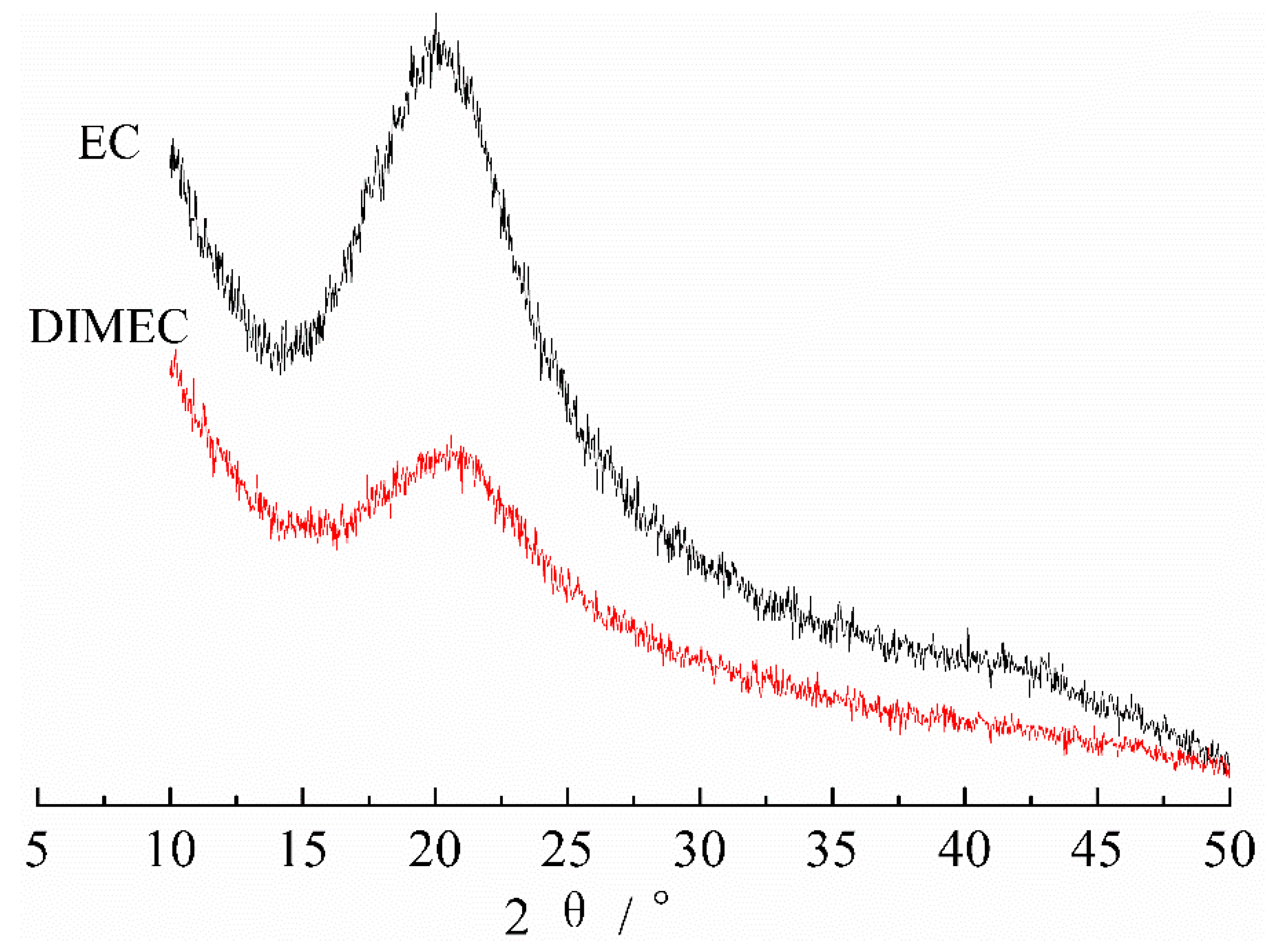
3.2.2. XRD of DIMEC
3.2.3. SEM Micrographs of DIMEC
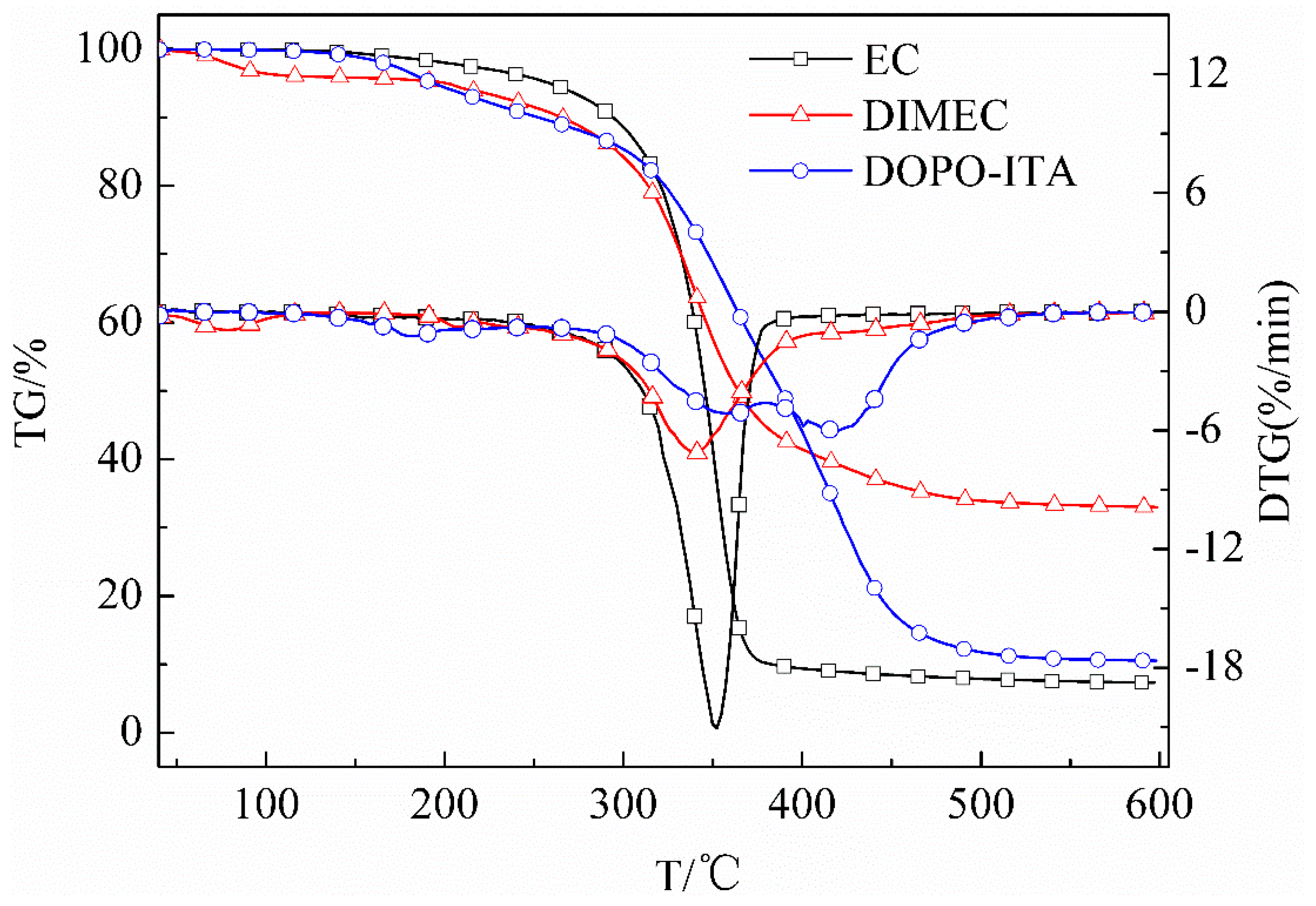
3.2.4. TG and DTG of DIMEC and EC
3.3. The Curing Kinetics of DCPRs
3.4. The Properties of Composite PFs (CPFs)
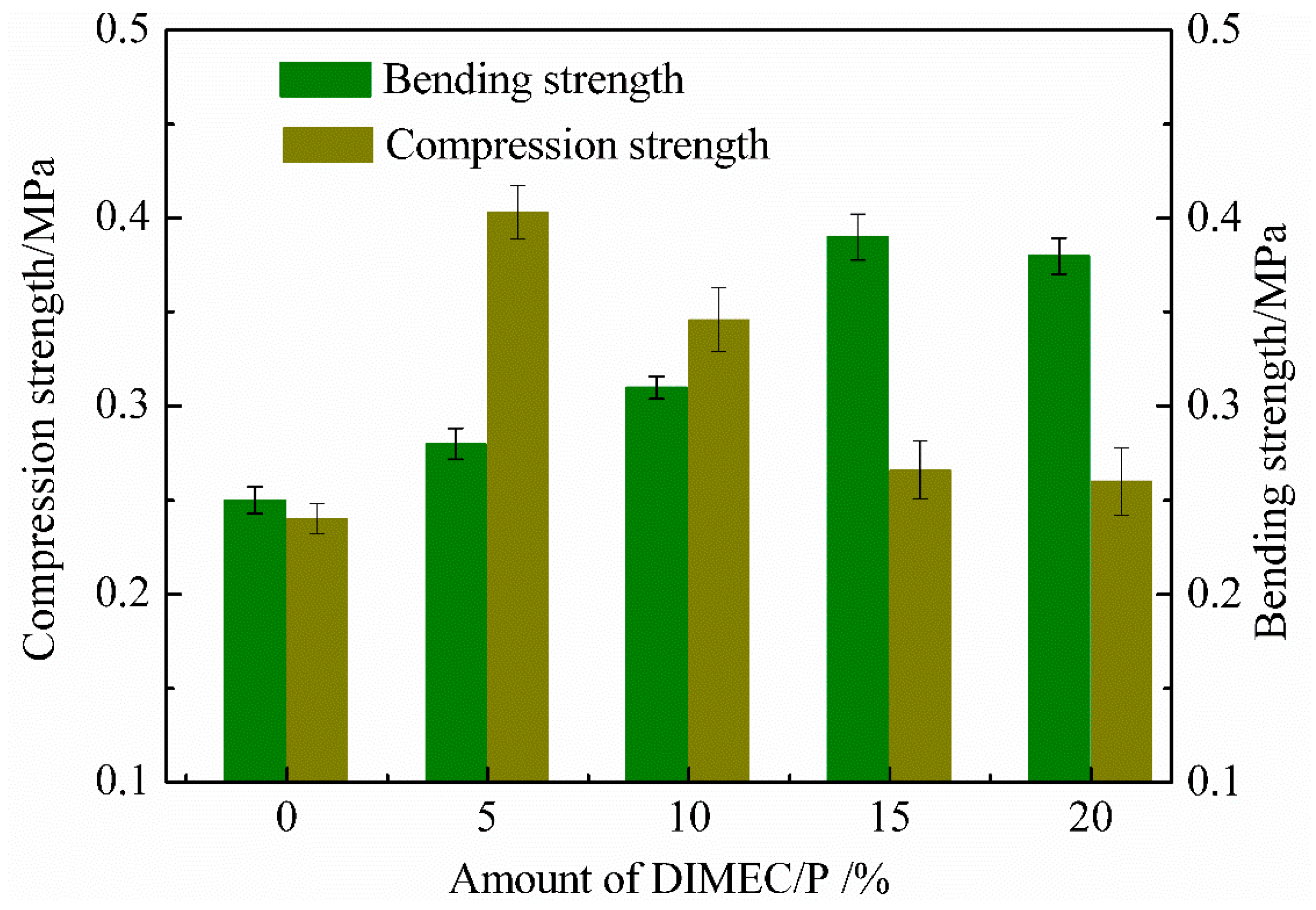
3.4.1. Compression and Bending Strength
3.4.2. Tensile Strength
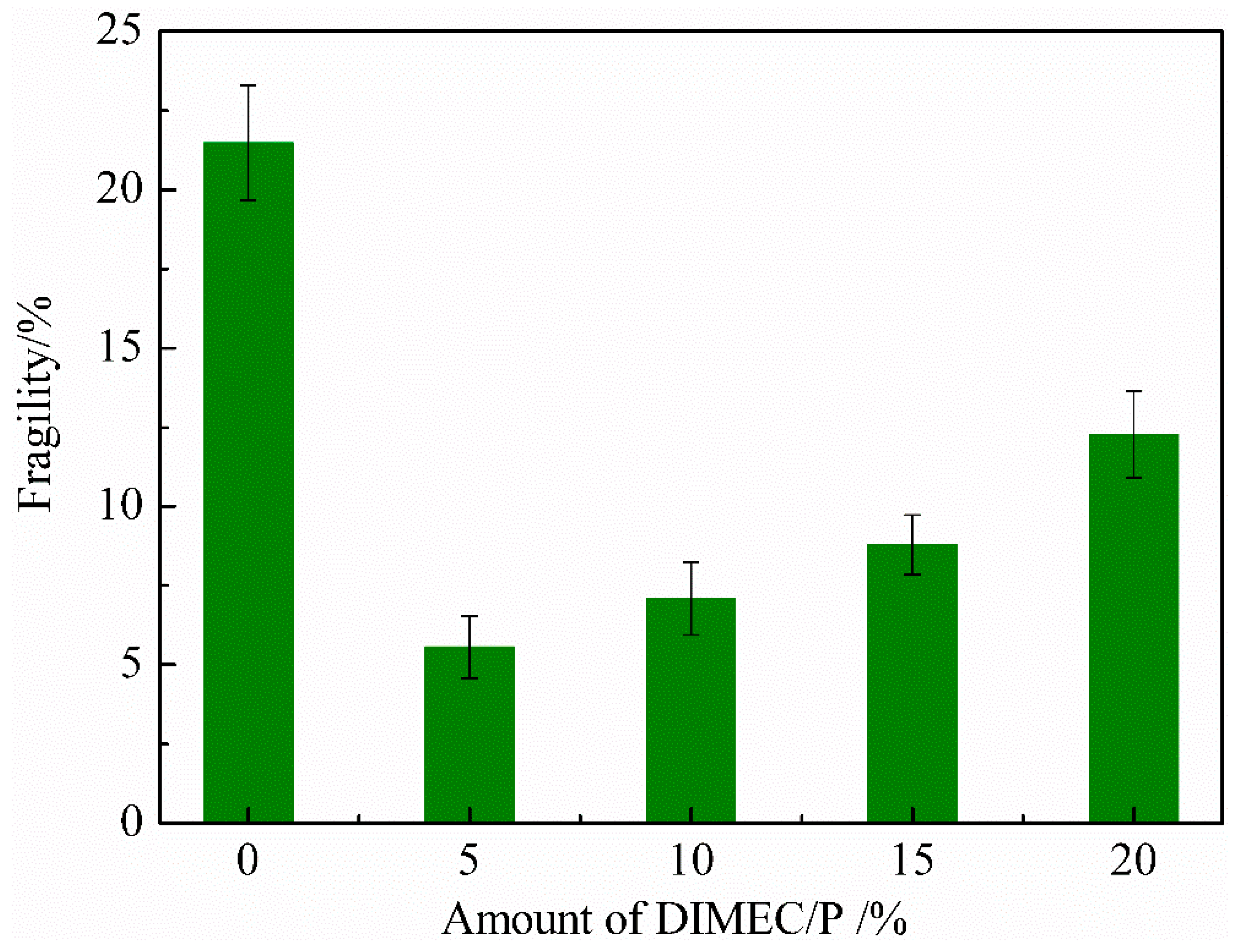
3.4.3. Fragility
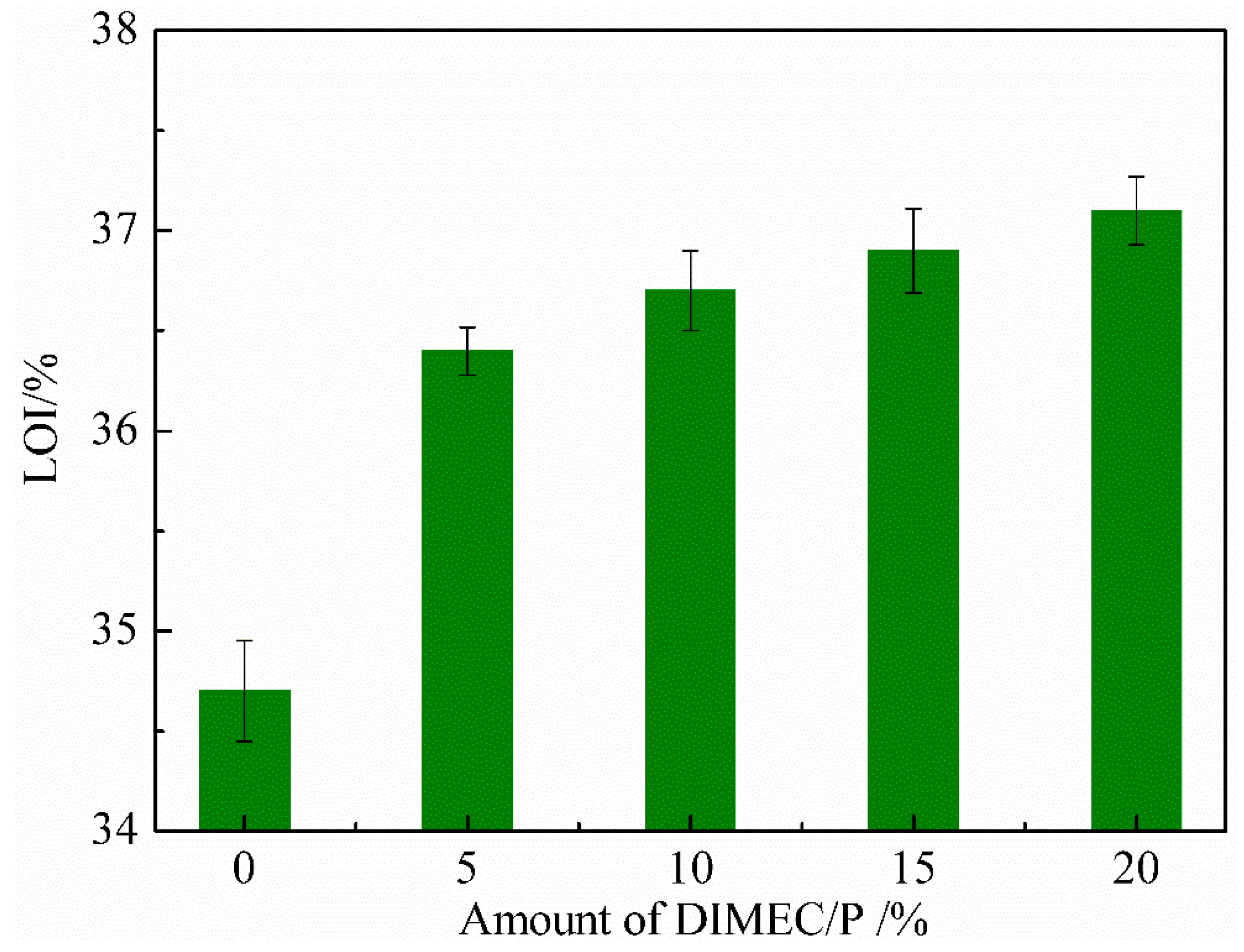
3.4.4. Limited Oxygen Index (LOI)
3.4.5. TG and DTG
3.4.6. SEM
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yang, H.; Wang, X.; Yuan, H.; Song, L.; Hu, Y.; Yuen, R.K.K. Fire performance and mechanical properties of phenolic foams modified by phosphorus-containing polyethers. J. Polym. Res. 2012, 19, 9831. [Google Scholar] [CrossRef]
- Lei, S.; Guo, Q.; Zhang, D.; Shi, J.; Liu, L.; Wei, X. Preparation and properties of the phenolic foams with controllable nanometer pore structure. J. Appl. Polym. Sci. 2010, 117, 3545–3550. [Google Scholar] [CrossRef]
- Auad, M.L.; Zhao, L.; Shen, H.; Nutt, S.R.; Sorathia, U. Flammability properties and mechanical performance of epoxy modified phenolic foams. J. Appl. Polym. Sci. 2010, 104, 1399–1407. [Google Scholar] [CrossRef]
- Rangari, V.K.; Hassan, T.A.; Zhou, Y.; Mahfuz, H.; Jeelani, S.; Prorok, B.C. Cloisite clay-infused phenolic foam nanocomposites. J. Appl. Polym. Sci. 2010, 103, 308–314. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, C.; Chu, F. Effects of fiber surface treatments on the properties of wood fiber-phenolic foam composites. Bioresources 2017, 12, 4722–4736. [Google Scholar] [CrossRef]
- Shen, H.; Nutt, S. Mechanical characterization of short fiber reinforced phenolic foam. Compos. Part A Appl. Sci. Manuf. 2003, 34, 899–906. [Google Scholar] [CrossRef]
- Shen, H.; Lavoie, A.J.; Nutt, S.R. Enhanced peel resistance of fiber reinforced phenolic foams. Compos. Part A Appl. Sci. Manuf. 2003, 34, 941–948. [Google Scholar] [CrossRef]
- Zhou, J.; Yao, Z.; Chen, Y.; Wei, D.; Wu, Y. Thermomechanical analyses of phenolic foam reinforced with glass fiber mat. Mater. Des. 2013, 51, 131–135. [Google Scholar] [CrossRef]
- Yang, H.; Wang, X.; Yu, B.; Yuan, H.; Song, L.; Hu, Y.; Yuen, R.K.K.; Guan, H.Y. A novel polyurethane prepolymer as toughening agent: Preparation, characterization, and its influence on mechanical and flame retardant properties of phenolic foam. J. Appl. Polym. Sci. 2013, 128, 2720–2728. [Google Scholar] [CrossRef]
- Jing, S.; Li, T.; Li, X.; Xu, Q.; Hu, J.; Li, R. Phenolic foams modified by cardanol through bisphenol modification. J. Appl. Polym. Sci. 2013, 131, 1001–1007. [Google Scholar] [CrossRef]
- Mulinari, D.R.; Voorwald, H.J.; Cioffi, M.O.; Silva, M.L.D. Cellulose fiber-reinforced high-density polyethylene composites-mechanical and thermal properties. J. Compos. Mater. 2016, 51, 1807–1815. [Google Scholar] [CrossRef]
- Luo, Q.; Li, Y.; Pan, L.; Song, L.; Yang, J.; Wu, L.; Lu, S. Effective reinforcement of epoxy composites with hyperbranched liquid crystals grafted on microcrystalline cellulose fibers. J. Mater. Sci. 2016, 51, 8888–8899. [Google Scholar] [CrossRef]
- Pérez-Fonseca, A.A.; Robledo-Ortíz, J.R.; González-Núñez, R.; Rodrigue, D. Effect of thermal annealing on the mechanical and thermal properties of polylactic acid-cellulosic fiber biocomposites. J. Appl. Polym. Sci. 2016, 133, 43750. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, J.; Wang, W.; Wang, Q. Research of fiber reinforced wood-plastic composites: A review. Sci. Silvae Sin. 2016, 52, 130–139. [Google Scholar] [CrossRef]
- Klemm, D.; Heublein, B.; Fink, H.P.; Bohn, A. Cellulose: Fascinating biopolymer and sustainable raw material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- Bruno, L.; Kasapis, S.; Heng, P.W.S. Effect of polymer molecular weight on the structural properties of non aqueous ethyl cellulose gels intended for topical drug delivery. Carbohydr. Polym. 2012, 88, 382–388. [Google Scholar] [CrossRef]
- Kang, H.; Liu, W.; He, B.; Shen, D.; Lin, M.; Yong, H. Synthesis of amphiphilic ethyl cellulose grafting poly(acrylic acid) copolymers and their self-assembly morphologies in water. Polymer 2006, 47, 7927–7934. [Google Scholar] [CrossRef]
- Shen, D.; Yu, H.; Huang, Y. Densely grafting copolymers of ethyl cellulose through atom transfer radical polymerization. J. Polym. Sci. Part A Polym. Chem. 2005, 43, 4099–4108. [Google Scholar] [CrossRef]
- Yu, Y.L.; Zhang, M.J.; Xie, R.; Ju, X.J.; Wang, J.Y.; Pi, S.W.; Chu, L.Y. Thermo-responsive monodisperse core-shell microspheres with pnipam core and biocompatible porous ethyl cellulose shell embedded with pnipam gates. J. Colloid Interface Sci. 2012, 376, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Zhang, J.; Zou, H.; Shen, T.; Ren, J. Amphiphilic ethyl cellulose brush polymers with mono and dual side chains: Facile synthesis, self-assembly, and tunable temperature-pH responsivities. Polymer 2012, 53, 956–966. [Google Scholar] [CrossRef]
- Bai, Y.; Jiang, C.; Wang, Q.; Wang, T. A novel high mechanical strength shape memory polymer based on ethyl cellulose and polycaprolactone. Carbohydr. Polym. 2013, 96, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Qian, L.; Qiu, Y.; Dong, Y. Synergistic flame-retardant effect and mechanisms of boron/phosphorus compounds on epoxy resins. Polym. Adv. Technol. 2018, 29, 641–648. [Google Scholar] [CrossRef]
- Zhang, W.; Li, X.; Yang, R. Novel flame retardancy effects of DOPO-POSS on epoxy resins. Polym. Degrad. Stab. 2011, 96, 2167–2173. [Google Scholar] [CrossRef]
- Zang, L.; Wagner, S.; Ciesielski, M.; Müller, P.; Döring, M. Novel star-shaped and hyperbranched phosphorus-containing flame retardants in epoxy resins. Polym. Adv. Technol. 2011, 22, 1182–1191. [Google Scholar] [CrossRef] [Green Version]
- Dumitrascu, A. Flame retardant polymeric materials achieved by incorporation of styrene monomers containing both nitrogen and phosphorus. Polym. Degrad. Stab. 2012, 97, 2611–2618. [Google Scholar] [CrossRef]
- Sun, D.; Yao, Y. Synthesis of three novel phosphorus-containing flame retardants and their application in epoxy resins. Polym. Degrad. Stab. 2011, 96, 1720–1724. [Google Scholar] [CrossRef]
- Wang, P.; Cai, Z. Highly efficient flame-retardant epoxy resin with a novel DOPO-based triazole compound: Thermal stability, flame retardancy and mechanism. Polym. Degrad. Stab. 2017, 137, 138–150. [Google Scholar] [CrossRef]
- Perret, B.; Schartela, B.; Ciesielski, M.; Diederichs, J.; Döring, M.; Krämer, J.; Altstädt, V. Novel DOPO-based flame retardants in high-performance carbon fibre epoxy composites for aviation. Eur. Polym. J. 2011, 47, 1081–1089. [Google Scholar] [CrossRef]
- Li, Z.; Zhuang, Z.L.; Wei-Bing, W.U.; Dai, H.Q. Preparation of cellulose-g-PNIPAAM copolymers by atom transfer radical polymerization. Trans. China Pulp Paper 2016, 31, 41–46. [Google Scholar]
- Dai, J.; Ma, S.; Wu, Y.; Han, L.; Zhang, L.; Zhu, J.; Liu, X. Polyesters derived from itaconic acid for the properties and bio-based content enhancement of soybean oil-based thermosets. Green Chem. 2015, 17, 2383–2392. [Google Scholar] [CrossRef]
- Winkler, M.; Lacerda, T.M.; Mack, F.; Meier, M.A.R. Renewable polymers from itaconic acid by polycondensation and ring-opening-metathesis polymerization. Macromolecules 2015, 48, 1398–1403. [Google Scholar] [CrossRef]
- Kamzolova, S.V.; Allayarov, R.K.; Lunina, J.N.; Morgunov, I.G. The effect of oxalic and itaconic acids on threo-Ds-isocitric acid production from rapeseed oil by Yarrowia lipolytica. Bioresour. Technol. 2016, 206, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Carja, I.D.; Serbezeanu, D.; Vladbubulac, T.; Hamciuc, C.; Coroaba, A.; Lisa, G.; López, C.G.; Soriano, M.F.; Pérez, V.F.; Sánchez, M.D.R. A straightforward, eco-friendly and cost-effective approach towards flame retardant epoxy resins. J. Mater. Chem. A 2014, 2, 16230–16241. [Google Scholar] [CrossRef]
- Dong, Q.; Ding, Y.; Wen, B.; Wang, F.; Dong, H.; Zhang, S.; Wang, T.; Yang, M. Improvement of thermal stability of polypropylene using DOPO-immobilized silica nanoparticles. Colloid Polym. Sci. 2012, 290, 1371–1380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.; Wang, C.; Chu, F. The structure and properties of eucalyptus fiber/phenolic foam composites under n-β(aminoethyl)-γ-aminopropyl trimethoxy silane pretreatments. Polish J. Chem. Technol. 2017, 19. [Google Scholar] [CrossRef]
- Shan, G.; Jia, L.; Zhao, T.; Jin, C.; Liu, R.; Xiao, Y. A novel DDPSI-FR flame retardant treatment and its effects on the properties of wool fabrics. Fibers Polym. 2017, 18, 2196–2203. [Google Scholar] [CrossRef]
- Tang, C.; Yan, H.; Li, M.; Lv, Q. A novel phosphorus-containing polysiloxane for fabricating high performance electronic material with excellent dielectric and thermal properties. J. Mater. Sci. Mater. Electron. 2018, 29, 195–204. [Google Scholar] [CrossRef]
- Fang, Y.; Zhou, X.; Xing, Z.; Wu, Y. An effective flame retardant for poly(ethylene terephthalate) synthesized by phosphaphenanthrene and cyclotriphosphazene. J. Appl. Polym. Sci. 2017, 134, 45246. [Google Scholar] [CrossRef]
- Peng, H.; Yang, C.Q.; Wang, X.; Wang, S. The combination of itaconic acid and sodium hypophosphite as a new cross-linking system for cotton. Ind. Eng. Chem. Res. 2012, 51, 11301–11311. [Google Scholar] [CrossRef]
- Lin, C.H.; Wu, C.Y.; Wang, C.S. Synthesis and properties of phosphorus-containing advanced epoxy resins. Ii. J. Appl. Polym. Sci. 2015, 78, 228–235. [Google Scholar] [CrossRef]
- Zhang, C.; Huang, J.Y.; Liu, S.M.; Zhao, J.Q. The synthesis and properties of a reactive flame-retardant unsaturated polyester resin from a phosphorus-containing diacid. Polym. Adv. Technol. 2011, 22, 1768–1777. [Google Scholar] [CrossRef]
- Songqi, M.A.; Liu, X.Q.; Jiang, Y.H.; Fan, L.B.; Feng, J.X.; Jin, Z. Synthesis and properties of phosphorus-containing bio-based epoxy resin from itaconic acid. Sci. China Chem. 2014, 57, 379–388. [Google Scholar] [CrossRef]
- Xiao-Qiang, X.V.; Yu-Min, W.U.; Ning, Z.G.; Wang, C.S. Synthesis and properties of halogen-free flame-retarded poly(butylene terephthalate) composite. Liaoning Chem. Ind. 2015. [Google Scholar] [CrossRef]
- Li, X.G.; Kresse, I.; Springer, J.; Nissen, J.; Yang, Y.L. Morphology and gas permselectivity of blend membranes of polyvinylpyridine with ethylcellulose. Polymer 2001, 42, 6859–6869. [Google Scholar] [CrossRef]
- Chen, T.; Chen, X.; Wang, M.; Hou, P.; Jie, C.; Li, J.; Xu, Y.; Zeng, B.; Dai, L. A novel halogen-free co-curing agent with linear multi-aromatic rigid structure as flame-retardant modifier in epoxy resin. Polym. Adv. Technol. 2017. [Google Scholar] [CrossRef]
- Kissinger, H.E. Reaction kinetics in differential thermal analysis. Anal. Chem. 1957, 29, 1702–1706. [Google Scholar] [CrossRef]
- Ozawa, T. Estimation of activation energy by isoconversion methods. Thermochim. Acta 1992, 203, 159–165. [Google Scholar] [CrossRef]
- Starink, M.J. A new method for the derivation of activation energies from experiments performed at constant heating rate. Thermochim. Acta 1996, 288, 97–104. [Google Scholar] [CrossRef]
- Crane, L.W.; Dynes, P.J.; Kaelble, D.H. Analysis of curing kinetics in polymer composites. J. Polym. Sci. Polym. Lett. Ed. 1973, 11, 533–540. [Google Scholar] [CrossRef]
- He, G.; Riedl, B. Phenol-urea-formaldehyde cocondensed resol resins: Their synthesis, curing kinetics, and network properties. J. Polym. Sci. Part B Polym. Phys. 2010, 41, 1929–1938. [Google Scholar] [CrossRef]
- He, G.; Riedl, B. Curing kinetics of phenol formaldehyde resin and wood-resin interactions in the presence of wood substrates. Wood Sci. Technol. 2004, 38, 69–81. [Google Scholar] [CrossRef]
- Wang, C.; Xu, G. Research on hard-segment flame-retardant modification of waterborne polyurethane. China Coat. 2010, 25, 57–60. [Google Scholar] [CrossRef]
- Ma, Y.; Geng, X.; Zhang, X.; Wang, C.; Chu, F. Synthesis of DOPO-g-GPTS modified wood fiber and its effects on the properties of composite phenolic foams. J. Appl. Polym. Sci. 2018, 46917. [Google Scholar] [CrossRef]




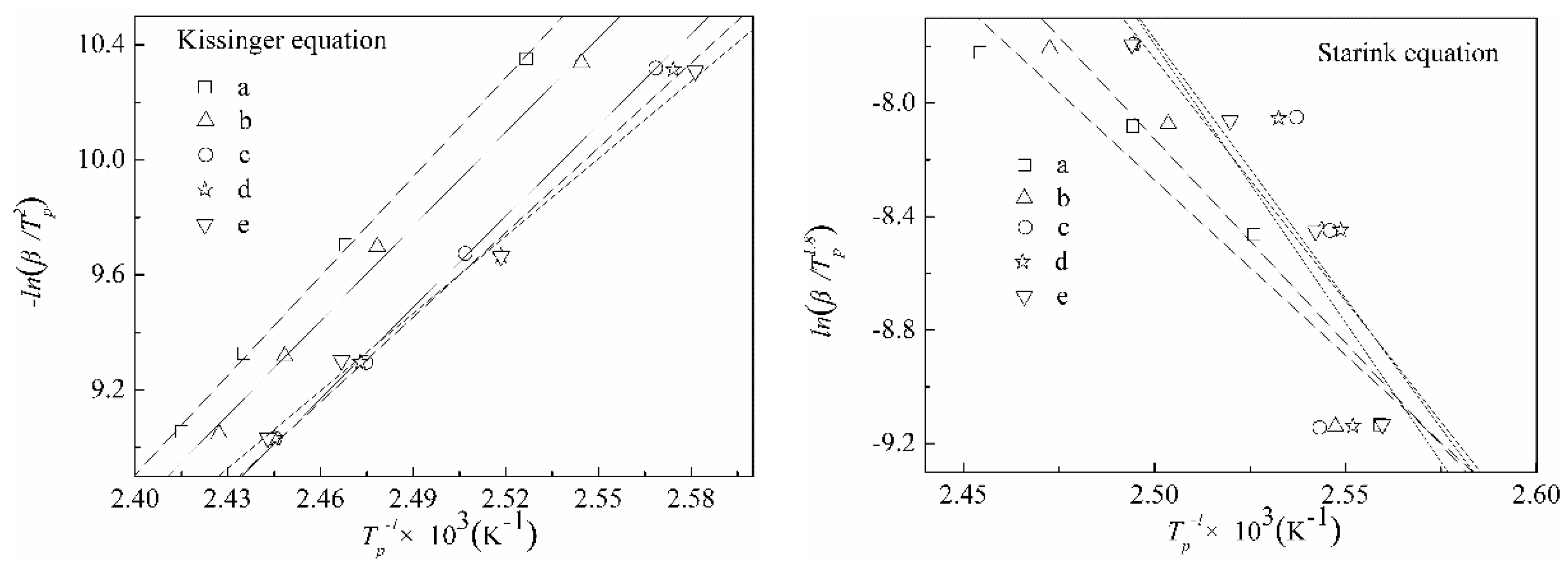
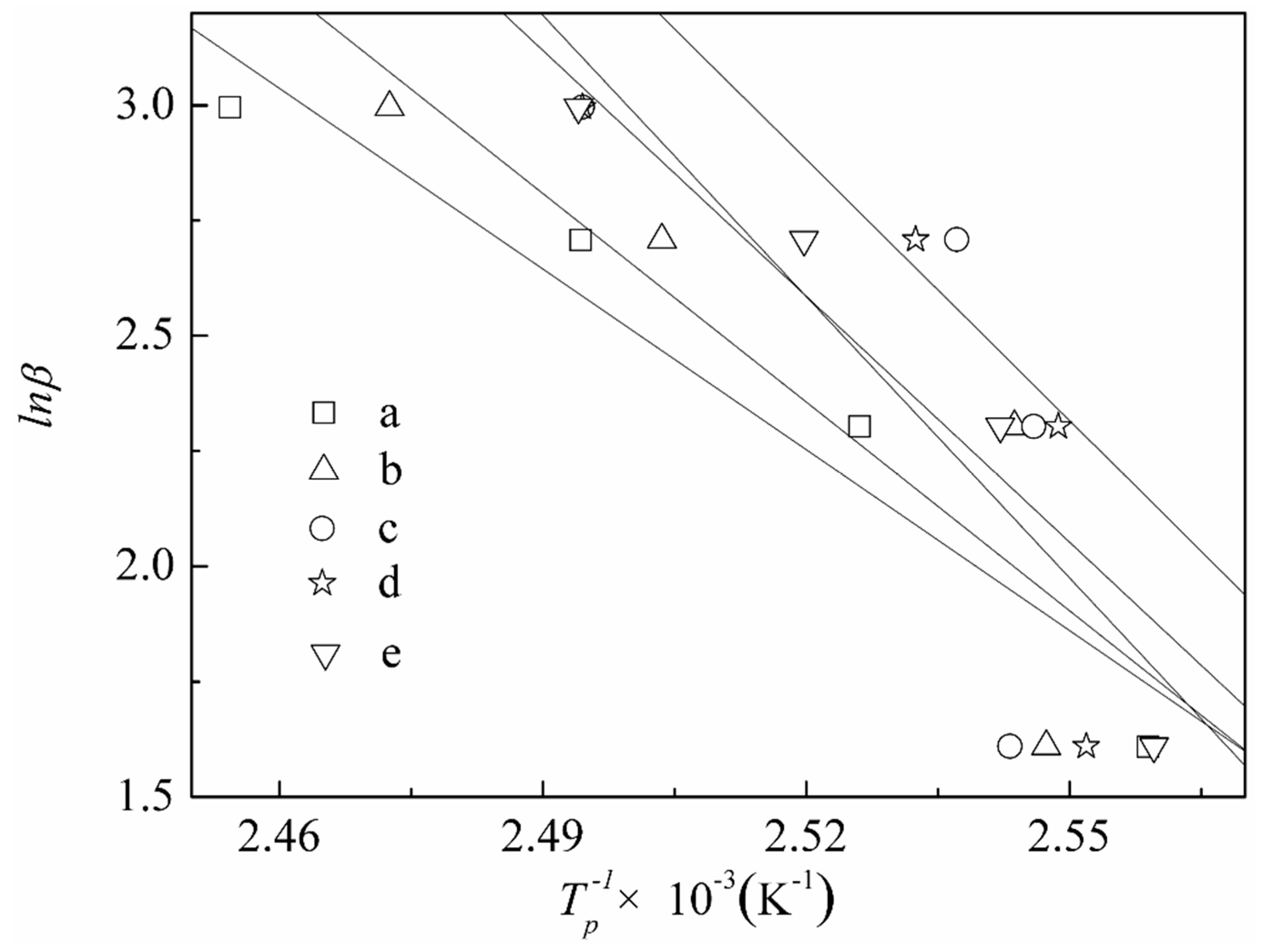




| Content of DIMEC/P | β (k·min−1) | Tp (°C) | Kissinger Equation [46] | Starink Equation [48] | n | ||
|---|---|---|---|---|---|---|---|
| Ea (kJ·mol−1) | r | Ea (kJ·mol−1) | r | ||||
| 0 | 5 | 117.64 | 102.00 | 0.970 | 102.66 | 0.9673 | 0.94 |
| 10 | 122.72 | ||||||
| 15 | 127.76 | ||||||
| 20 | 134.29 | ||||||
| 5% | 5 | 119.42 | 118.80 | 08821 | 119.02 | 0.8831 | 0.94 |
| 10 | 119.98 | ||||||
| 15 | 126.29 | ||||||
| 20 | 131.30 | ||||||
| 10% | 5 | 120.06 | 141.28 | 0.6963 | 141.41 | 0.6980 | 0.95 |
| 10 | 119.64 | ||||||
| 15 | 121.00 | ||||||
| 20 | 127.74 | ||||||
| 15% | 5 | 118.72 | 151.35 | 0.8215 | 154.81 | 0.8787 | 0.94 |
| 10 | 119.21 | ||||||
| 15 | 121.73 | ||||||
| 20 | 127.74 | ||||||
| 20% | 5 | 117.54 | 162.56 | 0.9571 | 162.61 | 0.9574 | 0.97 |
| 10 | 120.23 | ||||||
| 15 | 123.72 | ||||||
| 20 | 127.81 | ||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Y.; Gong, X.; Liao, C.; Geng, X.; Wang, C.; Chu, F. Preparation and Characterization of DOPO-ITA Modified Ethyl Cellulose and Its Application in Phenolic Foams. Polymers 2018, 10, 1049. https://doi.org/10.3390/polym10101049
Ma Y, Gong X, Liao C, Geng X, Wang C, Chu F. Preparation and Characterization of DOPO-ITA Modified Ethyl Cellulose and Its Application in Phenolic Foams. Polymers. 2018; 10(10):1049. https://doi.org/10.3390/polym10101049
Chicago/Turabian StyleMa, Yufeng, Xuanang Gong, Chuhao Liao, Xiang Geng, Chunpeng Wang, and Fuxiang Chu. 2018. "Preparation and Characterization of DOPO-ITA Modified Ethyl Cellulose and Its Application in Phenolic Foams" Polymers 10, no. 10: 1049. https://doi.org/10.3390/polym10101049