Mechanical Properties and Wear Resistance of Sulfonated Graphene/Waterborne Polyurethane Composites Prepared by In Situ Method
Abstract
:1. Introduction
2. Experimental
2.1. Materials
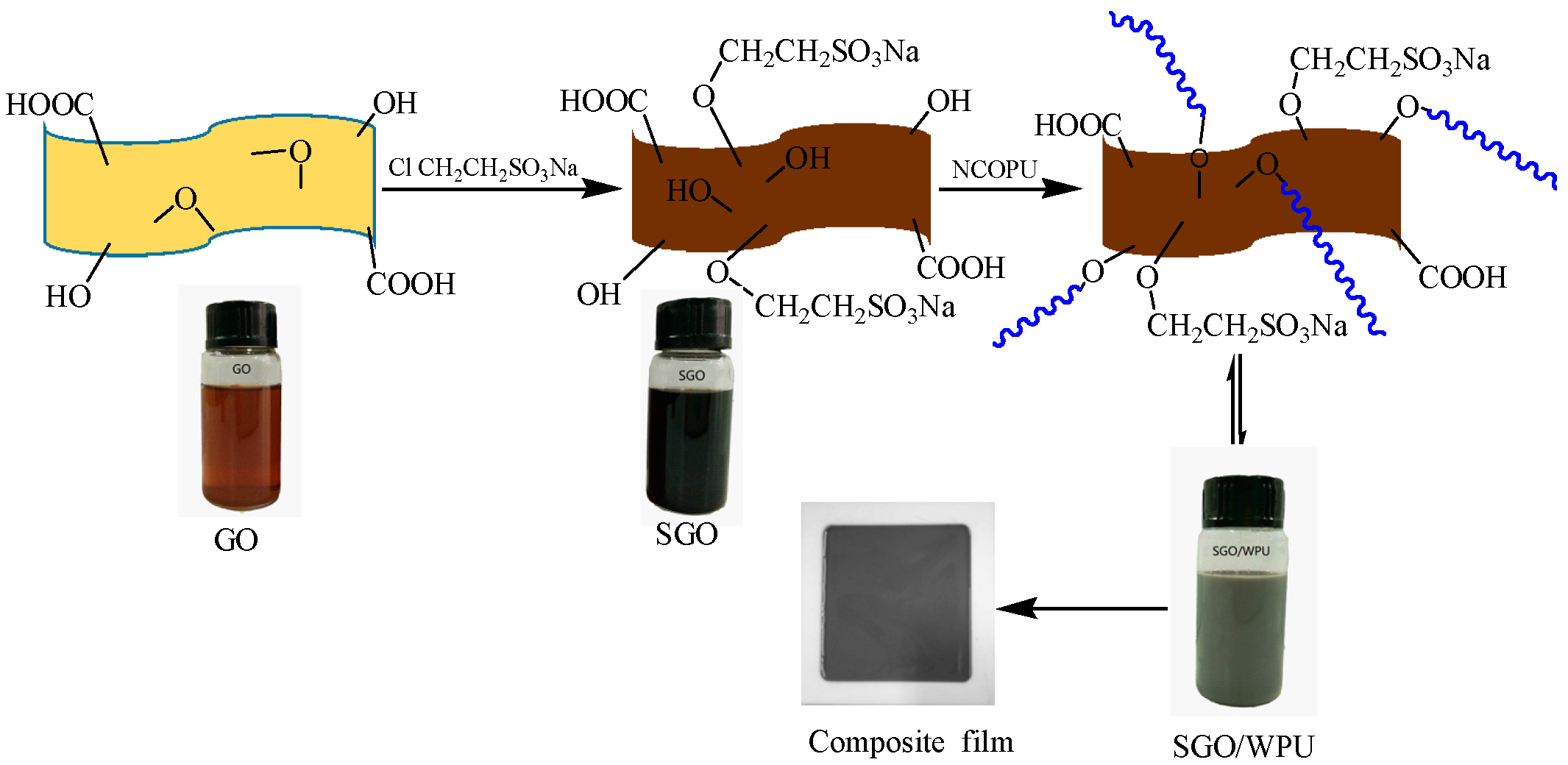
2.2. Preparation of SGO
2.3. Preparation of SGO/WPU
2.4. Preparation of SGO/WPU Composite Film
2.5. Characterization
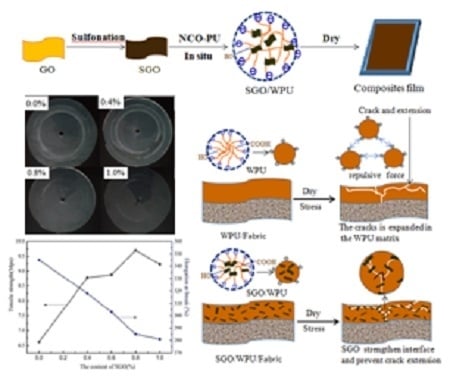
3. Results and Discussion
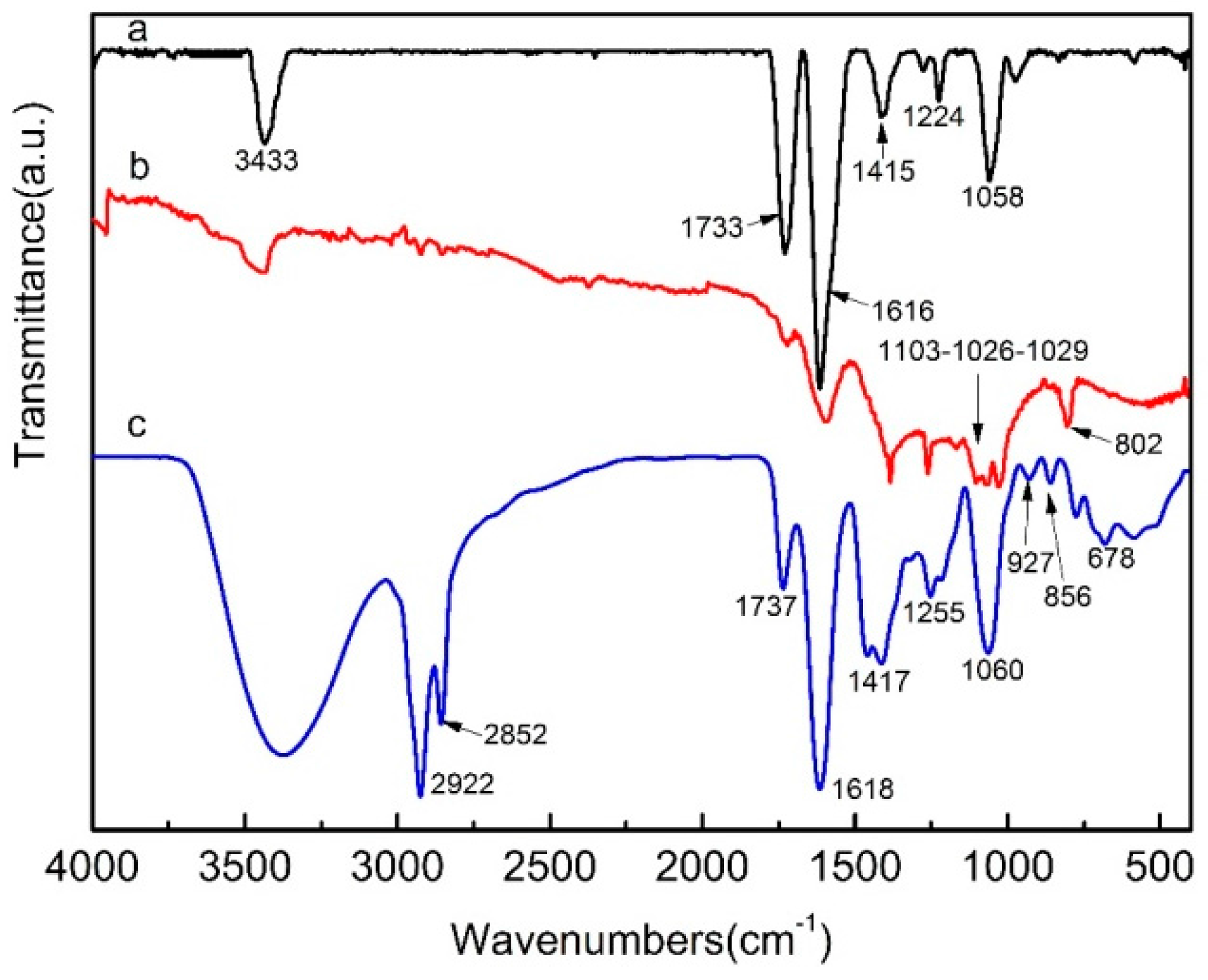
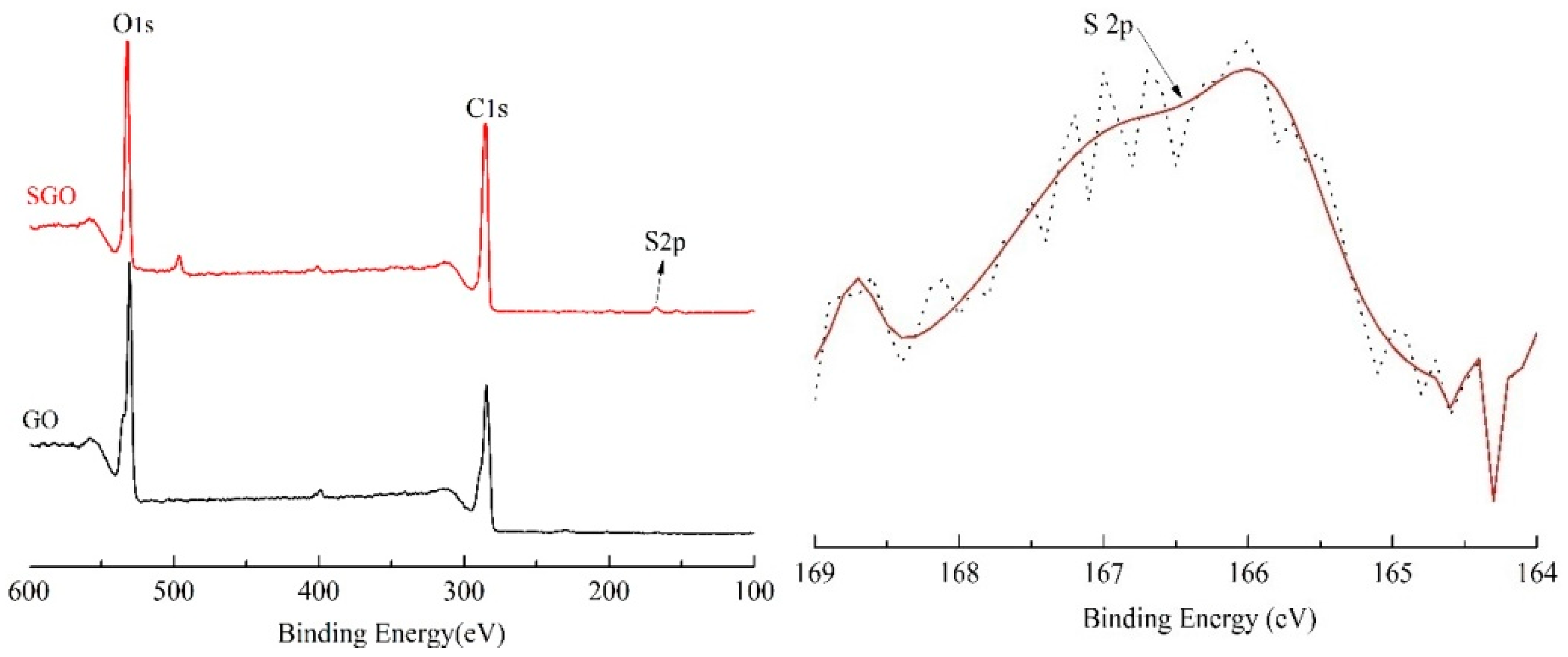
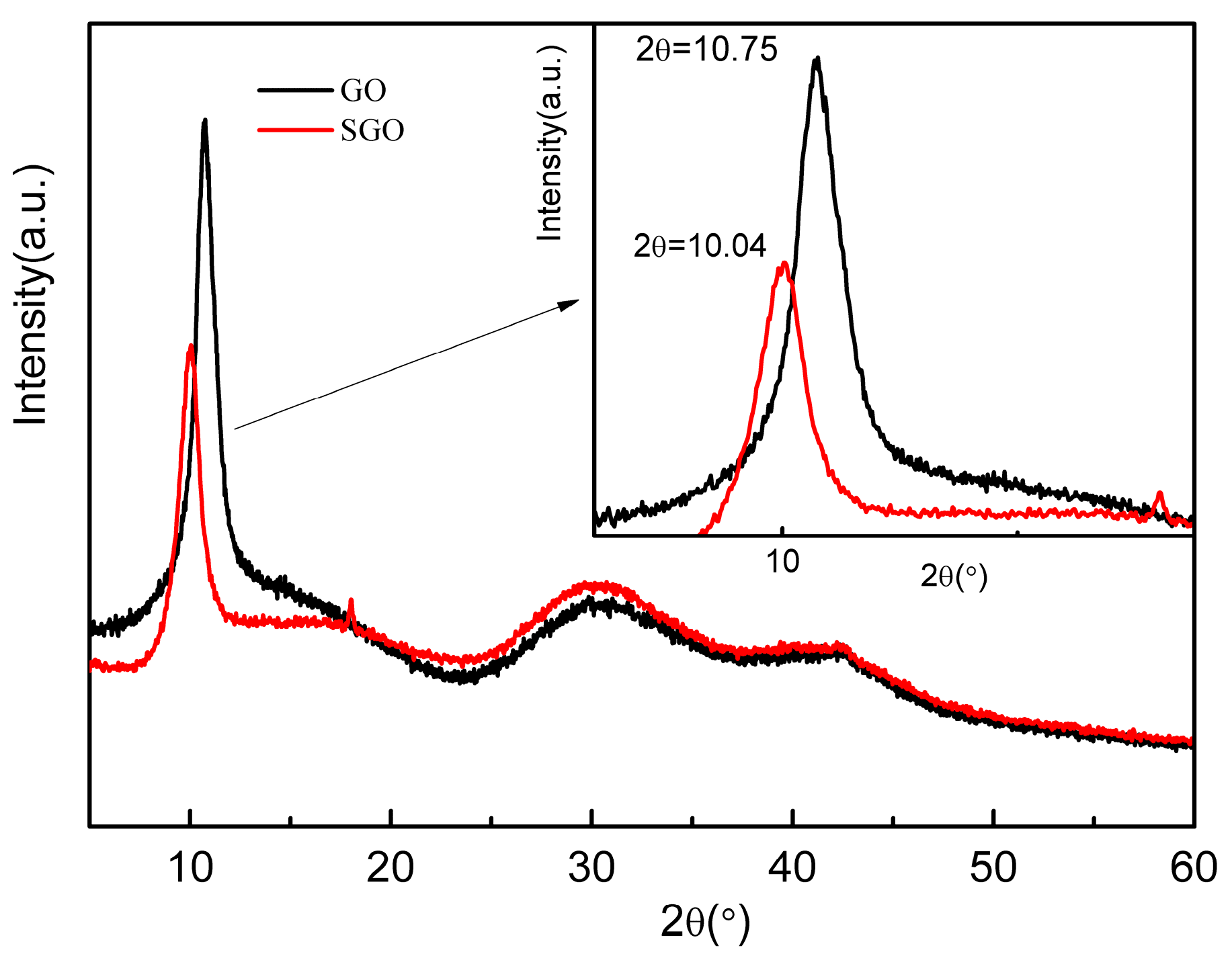
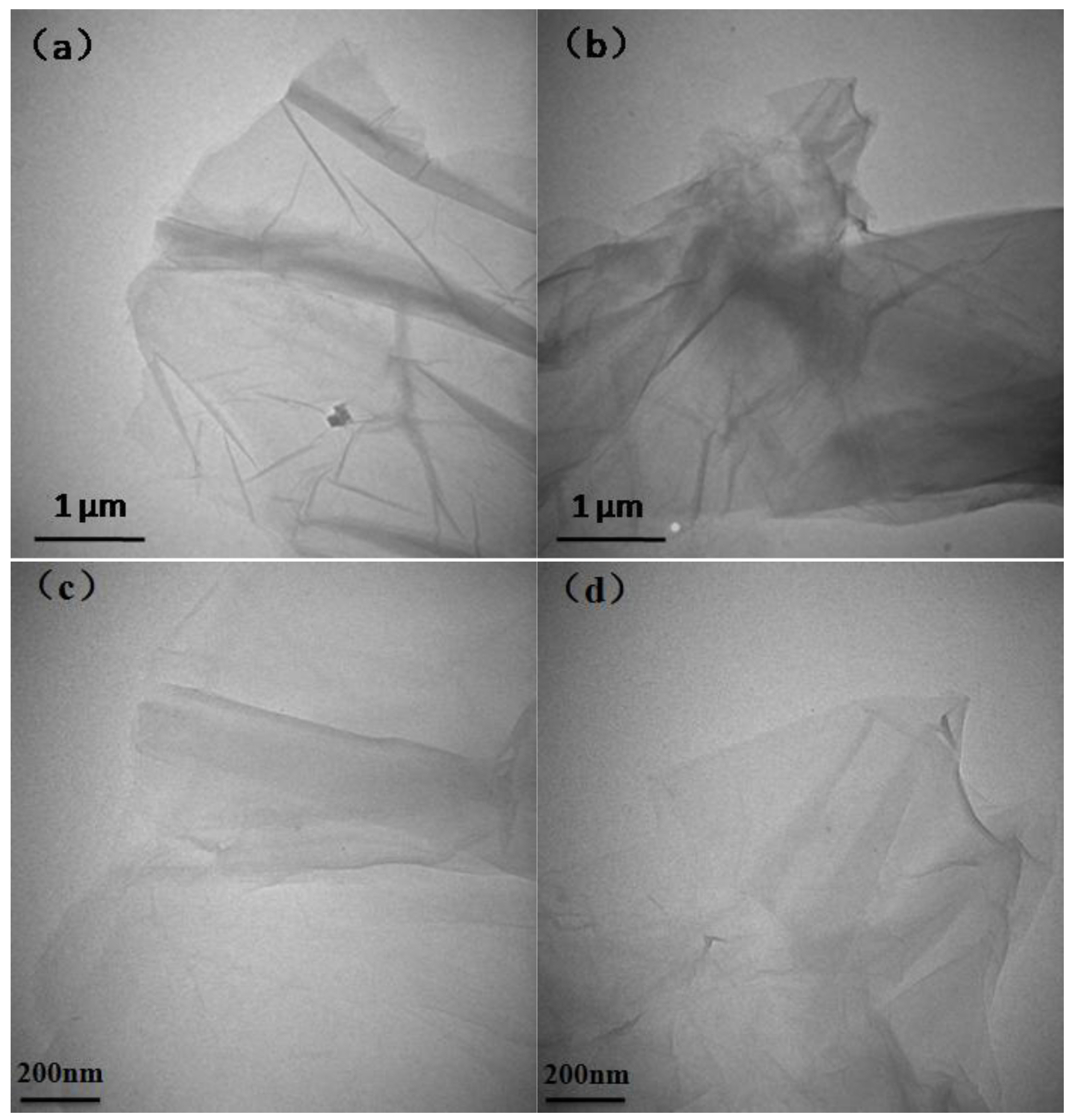
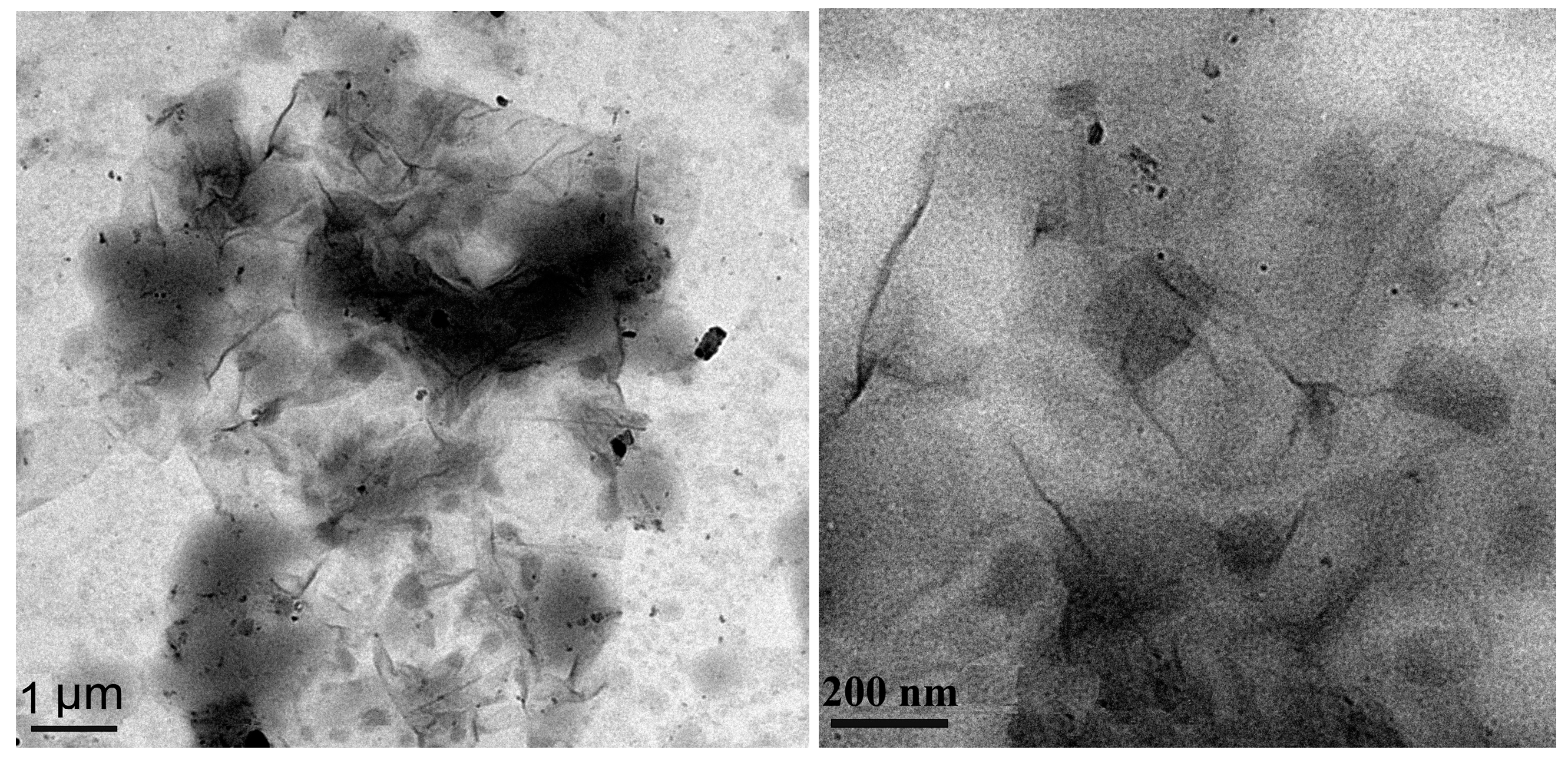
3.1. The Structure and Morphology of GO and SGO
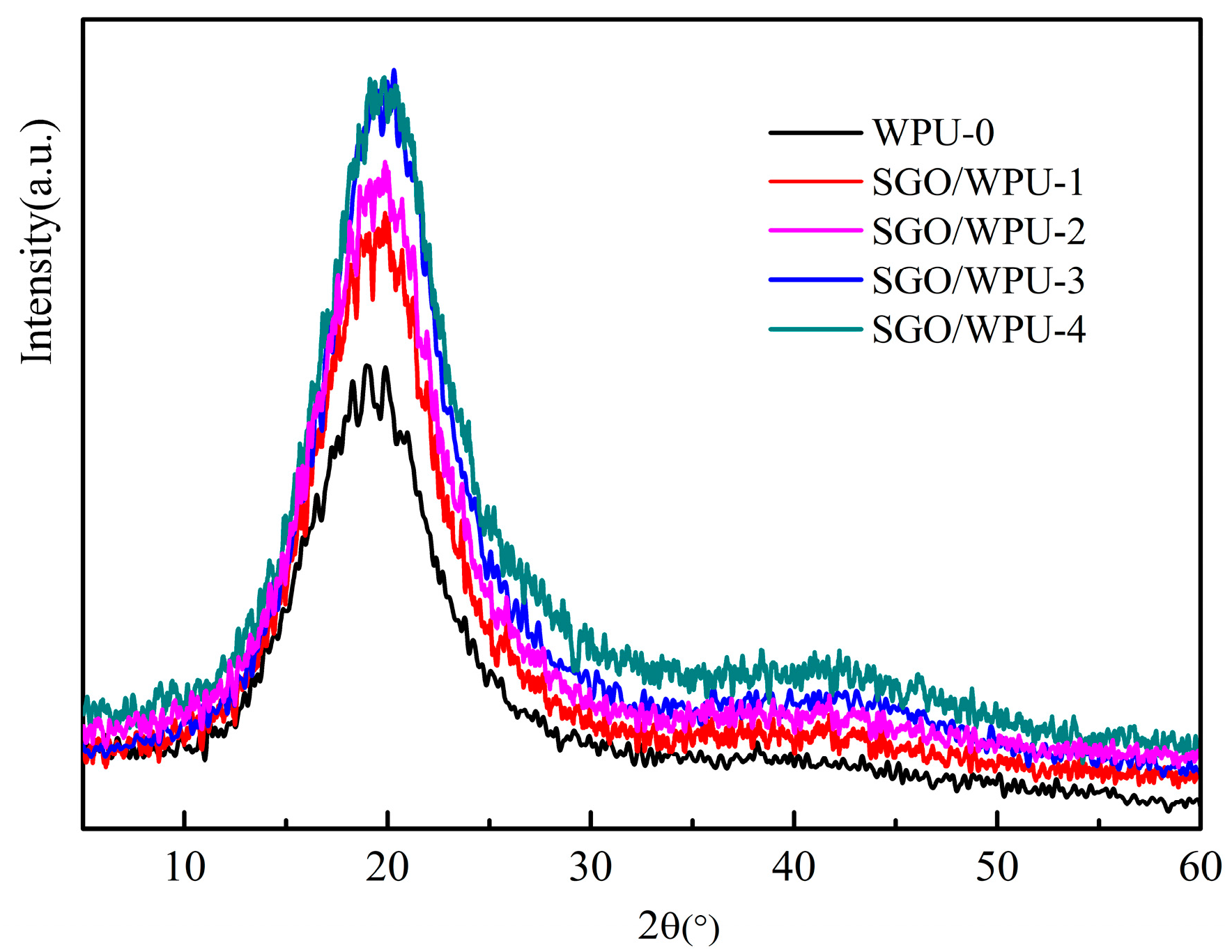
3.2. The Structure and Morphology of SGO/WPU
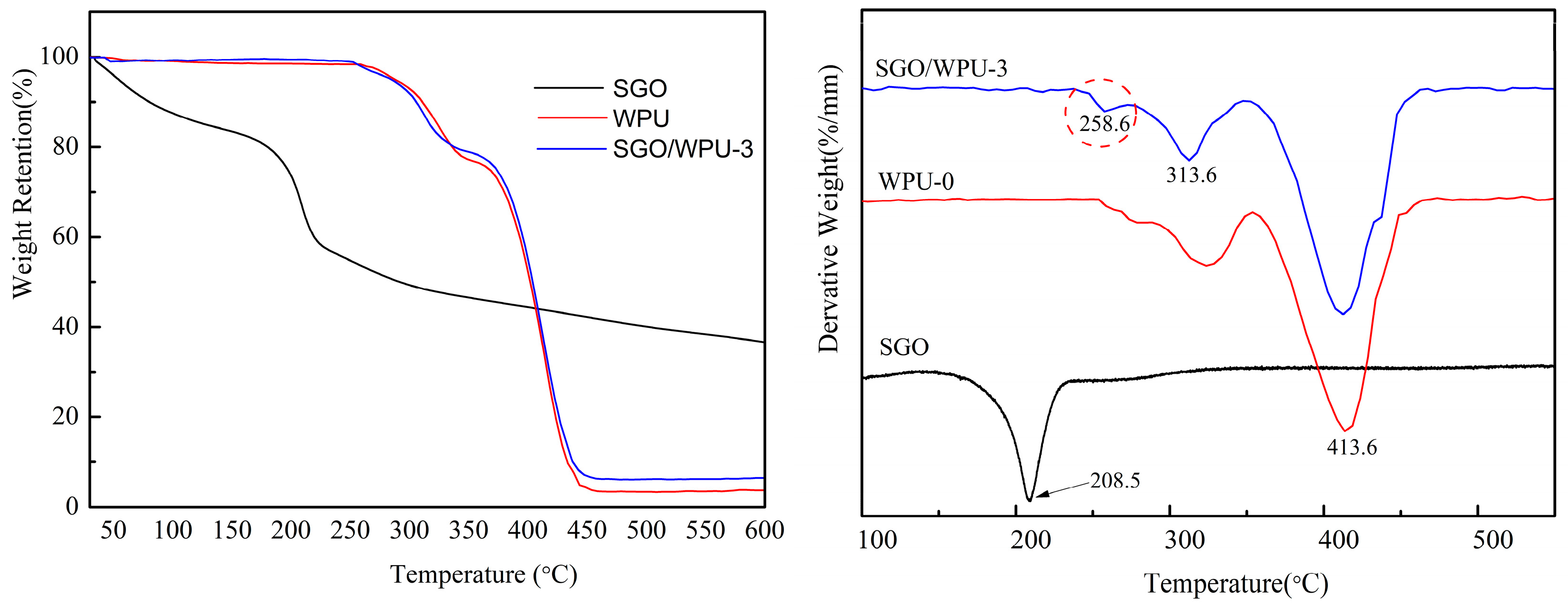
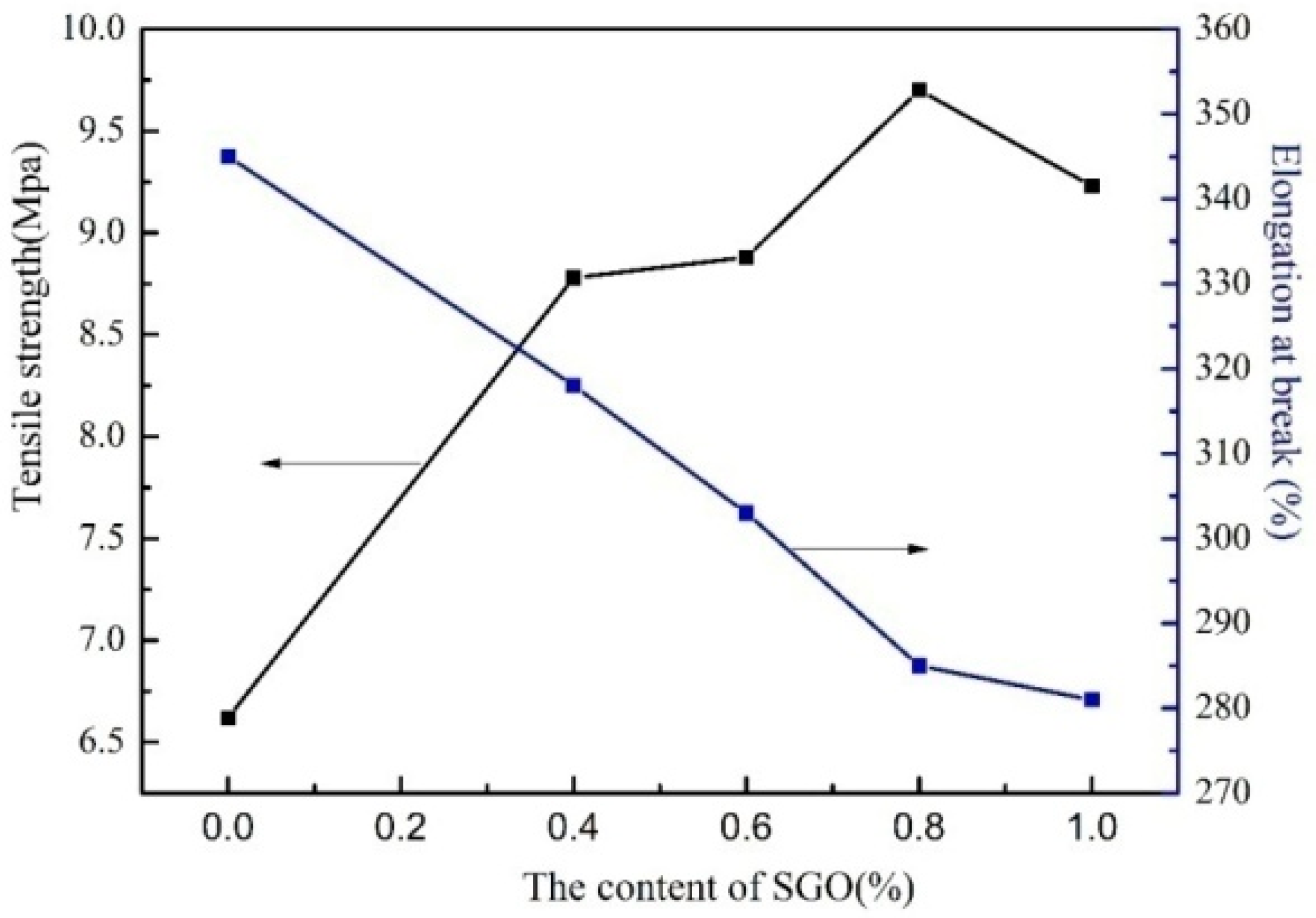
3.3. Mechanical Properties of SGO/WPU Composites
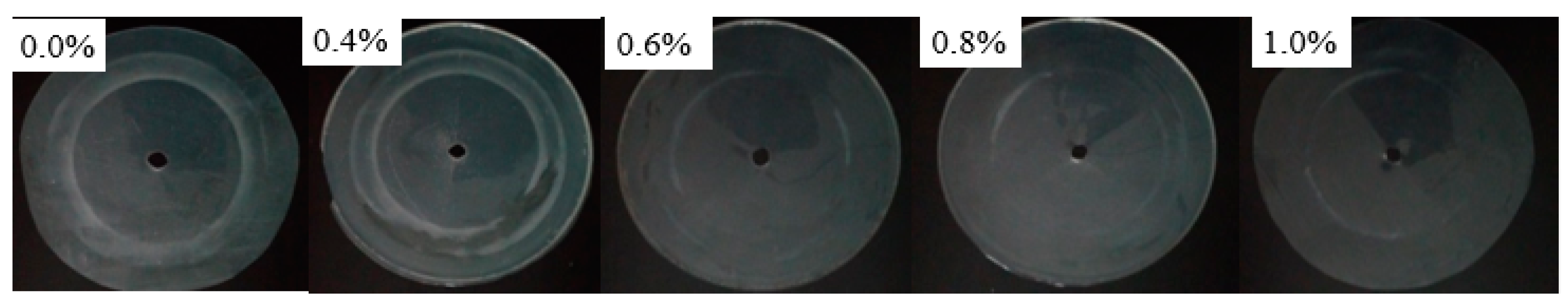
3.4. Abrasion Resistance Test
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Geim, A.K. Graphene: Status and prospects. Science 2009, 324, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Wang, S.J.; Ki, J.K. Preparation of graphite nanoplatelets and graphene sheets. J. Colloid Interface Sci. 2009, 336, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Yan, J.; She, X.J.; Xu, L.; Xia, J.X.; Xu, Y.G.; Song, Y.H.; Huang, L.Y.; Li, H.M. Graphene-analogue carbon nitride: Novel exfoliation synthesis and its application in photocatalysis and photoelectrochemical selective detection of trace amount of Cu2+. Nanoscale 2014, 6, 1406–1415. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Kulkarni, D.D.; Choi, I.; Tsukruk, V.V. Graphene-polymer nanocomposites for structural and functional applications. Prog. Polym. Sci. 2014, 39, 1934–1972. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, R.; Chen, W. Graphene-supported nanoelectrocatalysts for fuel cells: Synthesis, properties, and applications. Chem. Rev. 2014, 114, 5117–5160. [Google Scholar] [CrossRef] [PubMed]
- Stankovich, S.; Piner, R.D.; Chen, X.Q.; Wu, N.Q.; Nguyen, S.T.; Ruoff, R.S. Stable aqueous dispersions of graphitic nanoplatelets via thereduction of exfoliated graphite oxide in the presence of poly(sodium4-styrenesulfonate). J. Mater. Chem. 2006, 16, 155–158. [Google Scholar] [CrossRef]
- Rafiee, M.A. Graphene-Based Composite Materials; Rensselaer Polytechnic Institute: Troy, NY, USA, 2011. [Google Scholar]
- Kim, H.; Abdala, A.A.; Macosko, C.W. Graphene/polymer nanocomposites. Macromolecules 2010, 43, 6515–6530. [Google Scholar] [CrossRef]
- Gao, W.; Majumder, M.; Alemany, L.B.; Narayanan, T.N.; Ibarra, M.A.; Pradhan, B.K.; Ajayan, P.M. Engineered graphite oxide materials for application in water purification. ACS Appl. Mater. Interfaces 2011, 3, 1821–1826. [Google Scholar] [CrossRef] [PubMed]
- Georgakilas, V.; Tiwari, J.N.; Kemp, K.C.; Perman, J.A.; Bourlinos, A.B.; Kim, S.W.; Zboril, R. Noncovalent functionalization of graphene and graphene oxide for energy materials, biosensing, catalytic, and biomedical applications. Chem. Rev. 2016, 116, 5464–5519. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Mark, J.E.; Zhu, Y.; Ruoff, R.S.; Schaefer, D.W. Mechanical properties of polybutadiene reinforced with octadecylamine modified graphene oxide. Polymer 2014, 55, 5389–5395. [Google Scholar] [CrossRef]
- Mallakpour, S.; Abdolmaleki, A.; Borandeh, S. Covalently functionalized graphene sheets with biocompatible natural amino acids. Appl. Surf. Sci. 2014, 307, 533–542. [Google Scholar] [CrossRef]
- Gao, P.; Ng, K.; Sun, D.D. Sulfonatedgraphene oxide-ZnO-Ag photocatalyst for fast photodegradation and disinfection under visible light. J. Hazard. Mater. 2013, 262, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.Y. Emulsion Polymerization, Dispersion Polymerization, Stabilized by Sulfonated Grapheme, Sulfonated Carbon Nanotubes. Ph.D. Thesis, Hefei University of Technology, Hefei, China, 2014. [Google Scholar]
- Zhou, X.; Li, Y.; Fang, C.; Cheng, Y.C.; Meng, W.L. Recent advances in synthesis of waterborne polyurethane and their application in water-based ink: A review. J. Mater. Sci. Technol. 2015, 31, 708–722. [Google Scholar] [CrossRef]
- Li, J.; Zheng, W.; Zeng, W.; Zhang, D.Q.; Peng, X.H. Structure, properties and application of a novel low-glossed waterborne polyurethane. Appl. Surf. Sci. 2014, 307, 255–262. [Google Scholar] [CrossRef]
- Asif, A.; Hu, L.; Shi, W. Synthesis, rheological, and thermal properties of waterborne hyperbranched polyurethane acrylate dispersions for UV curable coatings. Colloid Polym. Sci. 2009, 287, 1041–1049. [Google Scholar] [CrossRef]
- Luo, X.; Liu, R.; Zhang, P.; Ge, B.H.; Cao, M. Study on polymerizable nano-SiO2 modifying UV-curing waterborne polyurethane film performance. J. Funct. Mater. 2015, 2, 02109–02114. [Google Scholar]
- Yousefi, N.; Sun, X.Y.; Lin, X.Y.; Shen, X.; Jia, J.J.; Zhang, B.; Tang, B.Z.; Chan, M.S.; Kim, J.K. Highly aligned graphene/polymer nanocomposites with excellent dielectric properties for high-performance electromagnetic interference shielding. Adv. Mater. 2014, 26, 5480–5487. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.C.; Ma, C.C.M.; Hsiao, S.T.; Wang, Y.S.; Yang, C.Y.; Liao, W.H.; Li, S.M.; Wang, J.A.; Cheng, T.Y.; Lin, C.W.; et al. Electromagnetic interference shielding performance of waterborne polyurethane composites filled with silver nanoparticles deposited on functionalized graphene. Appl. Surf. Sci. 2016, 385, 436–444. [Google Scholar] [CrossRef]
- Kuan, H.C.; Ma, C.C.M.; Chang, W.P.; Yuen, S.M.; Wu, H.H.; Lee, T.M. Synthesis, thermal, mechanical and rheological properties of multiwall carbon nanotube/waterborne polyurethane nanocomposite. Compos. Sci. Technol. 2005, 65, 1703–1710. [Google Scholar] [CrossRef]
- Zeng, Z.H.; Chen, M.J.; Jin, H.; Li, W.W.; Xue, X.; Zhou, L.C.; Pei, Y.M.; Zhang, H.; Zhang, Z. Thin and flexible multi-walled carbon nanotube/waterborne polyurethane composites with high-performance electromagnetic interference shielding. Carbon 2016, 96, 768–777. [Google Scholar] [CrossRef]
- Hou, Y.N.; Wu, M.H.; Yu, D.Y. Preparation and properties of functionalized graphene modified waterborne polyurethane. Text. Res. J. 2015, 36, 80–85. [Google Scholar]
- Hsiao, S.T.; Ma, C.C.M.; Liao, W.H.; Wang, Y.S.; Huang, S.M.; Huang, Y.C.; Yang, R.B.; Liang, W.F. Lightweight and flexible reduced graphene oxide/water-borne polyurethane composites with high electrical conductivity and excellent electromagnetic interference shielding performance. ACS Appl. Mater. Interfaces. 2014, 6, 10667–10678. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.J.; Gai, J.; Chen, Y.; Yang, T.; Yan, J. Application of graphehe in leather and synthetic leather manufacture. China Leather. 2016, 45, 19–24. [Google Scholar]
- Choi, S.H.; Kim, D.H.; Raghu, A.V.; Reddy, K.R.; Lee, H.I.; Yoon, K.S.; Jeong, H.M.; Kim, B.K. Properties of graphene/waterborne polyurethane nanocomposites cast from colloidal dispersion mixtures. J. Macromol. Sci. B. 2012, 51, 197–207. [Google Scholar] [CrossRef]
- Sun, Z.; He, Y.Z.; Fan, H.J.; Yan, J. The preparation and properties of graphene/polyurethane composites for leather finishing. Leather Sci. Eng. 2016, 26, 10–15. [Google Scholar]
- Suen, M.C.; Gu, J.H.; Lee, H.T.; Wu, C.L.; Liao, C.S.; Yang, J.J. In situ polymerisation and characteristic properties of the waterborne graphene oxide/poly(siloxane-urethane)s nanocomposites. Polym. Bull. 2017, 74, 4921–4942. [Google Scholar] [CrossRef]
- Lin, J.; Dong, J.; Chen, D.; Chen, G. Synthesis of hydrophilic sulfonatedgraphene based ondiazonium salt of 4-aminobenzenesulfonic acid functionalized graphene. Sci. Sin. 2016, 46, 791–799. [Google Scholar]


| Samples | m(NCOPU)/g | V(SGO Acetone Solution)/mL | SGO/wt % |
|---|---|---|---|
| WPU-0 | 20 | 0 | 0.0 |
| SGO/WPU-1 | 20 | 40 | 0.4 |
| SGO/WPU-2 | 20 | 60 | 0.6 |
| SGO/WPU-3 | 20 | 80 | 0.8 |
| SGO/WPU-4 | 20 | 100 | 1.0 |
| Sample | Elongation at Break/% | Tensile Strength/MPa | Young Modulus/MPa |
|---|---|---|---|
| WPU-0 | 355 | 6.62 | 0.18 |
| SGO/WPU-1 | 318 | 8.78 | 0.22 |
| SGO/WPU-2 | 303 | 8.89 | 0.23 |
| SGO/WPU-3 | 287 | 9.70 | 0.25 |
| SGO/WPU-4 | 285 | 9.33 | 0.24 |
| Sample | Grinding Wheel Rotation Number/N | Mass Loss/mg |
|---|---|---|
| WPU-0 | 200 | 2.5 |
| SGO/WPU-1 | 200 | 1.6 |
| SGO/WPU-2 | 200 | 1.1 |
| SGO/WPU-3 | 200 | 0.3 |
| SGO/WPU-4 | 200 | 0.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, J.; Wang, X.; Guo, P.; Wang, Y.; Luo, X. Mechanical Properties and Wear Resistance of Sulfonated Graphene/Waterborne Polyurethane Composites Prepared by In Situ Method. Polymers 2018, 10, 75. https://doi.org/10.3390/polym10010075
Feng J, Wang X, Guo P, Wang Y, Luo X. Mechanical Properties and Wear Resistance of Sulfonated Graphene/Waterborne Polyurethane Composites Prepared by In Situ Method. Polymers. 2018; 10(1):75. https://doi.org/10.3390/polym10010075
Chicago/Turabian StyleFeng, Jianyan, Xuechuan Wang, Peiying Guo, Yujie Wang, and Xiaomin Luo. 2018. "Mechanical Properties and Wear Resistance of Sulfonated Graphene/Waterborne Polyurethane Composites Prepared by In Situ Method" Polymers 10, no. 1: 75. https://doi.org/10.3390/polym10010075
APA StyleFeng, J., Wang, X., Guo, P., Wang, Y., & Luo, X. (2018). Mechanical Properties and Wear Resistance of Sulfonated Graphene/Waterborne Polyurethane Composites Prepared by In Situ Method. Polymers, 10(1), 75. https://doi.org/10.3390/polym10010075