Polarization Dependence of Low-Frequency Vibrations from Multiple Faces in an Organic Single Crystal
Abstract
1. Introduction
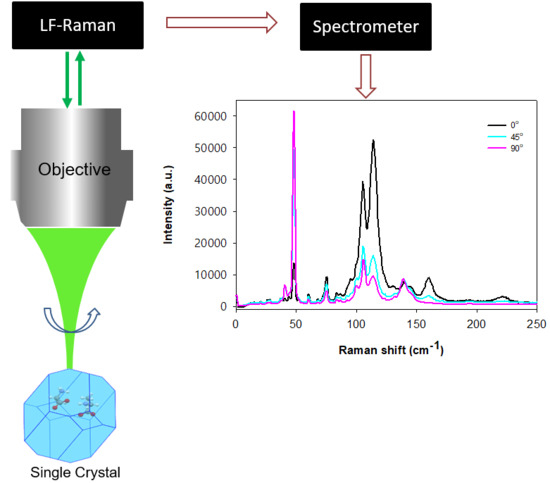
2. Materials and Methods
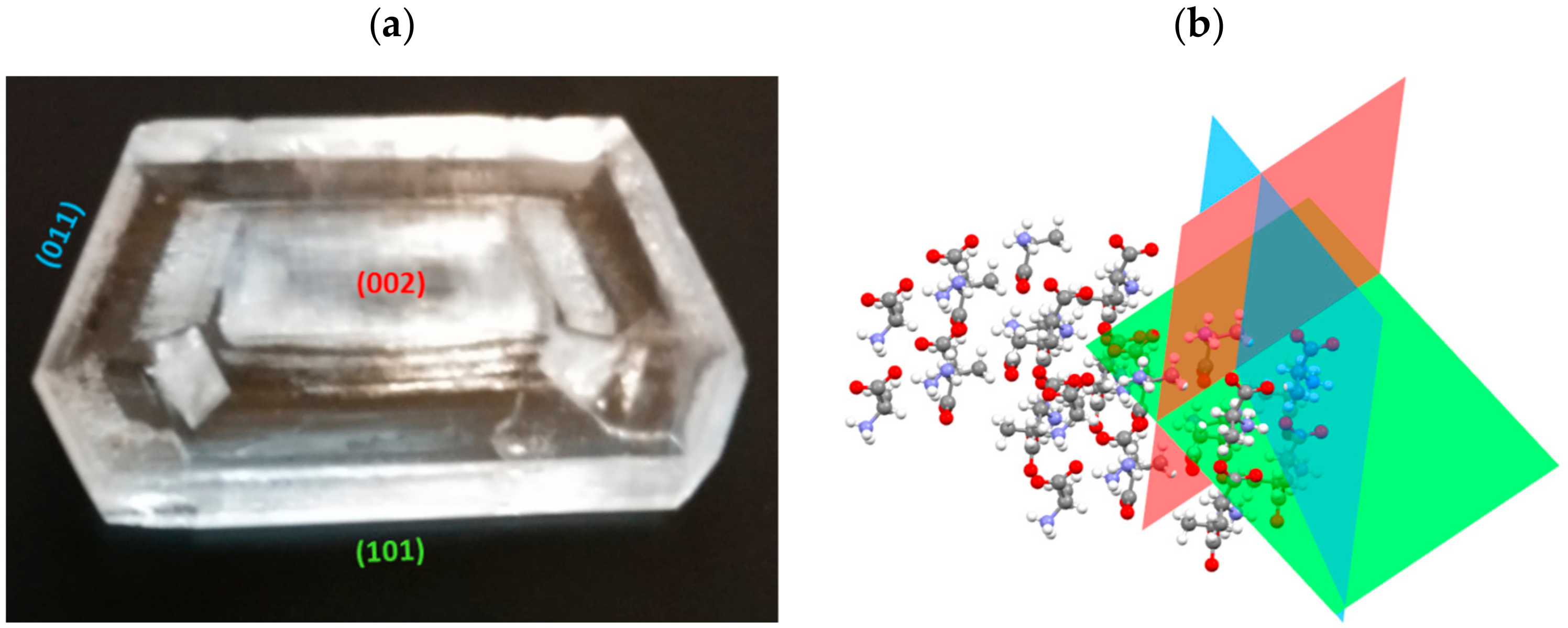
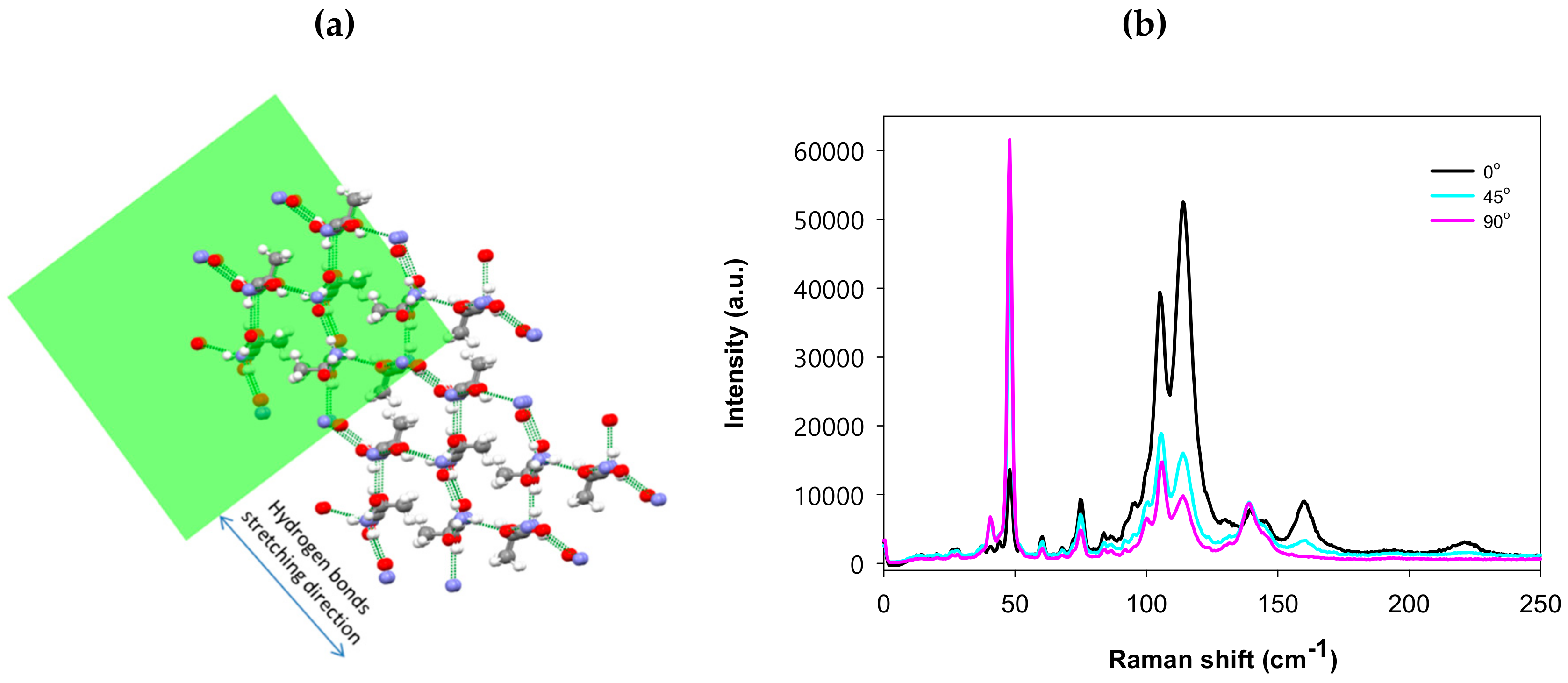
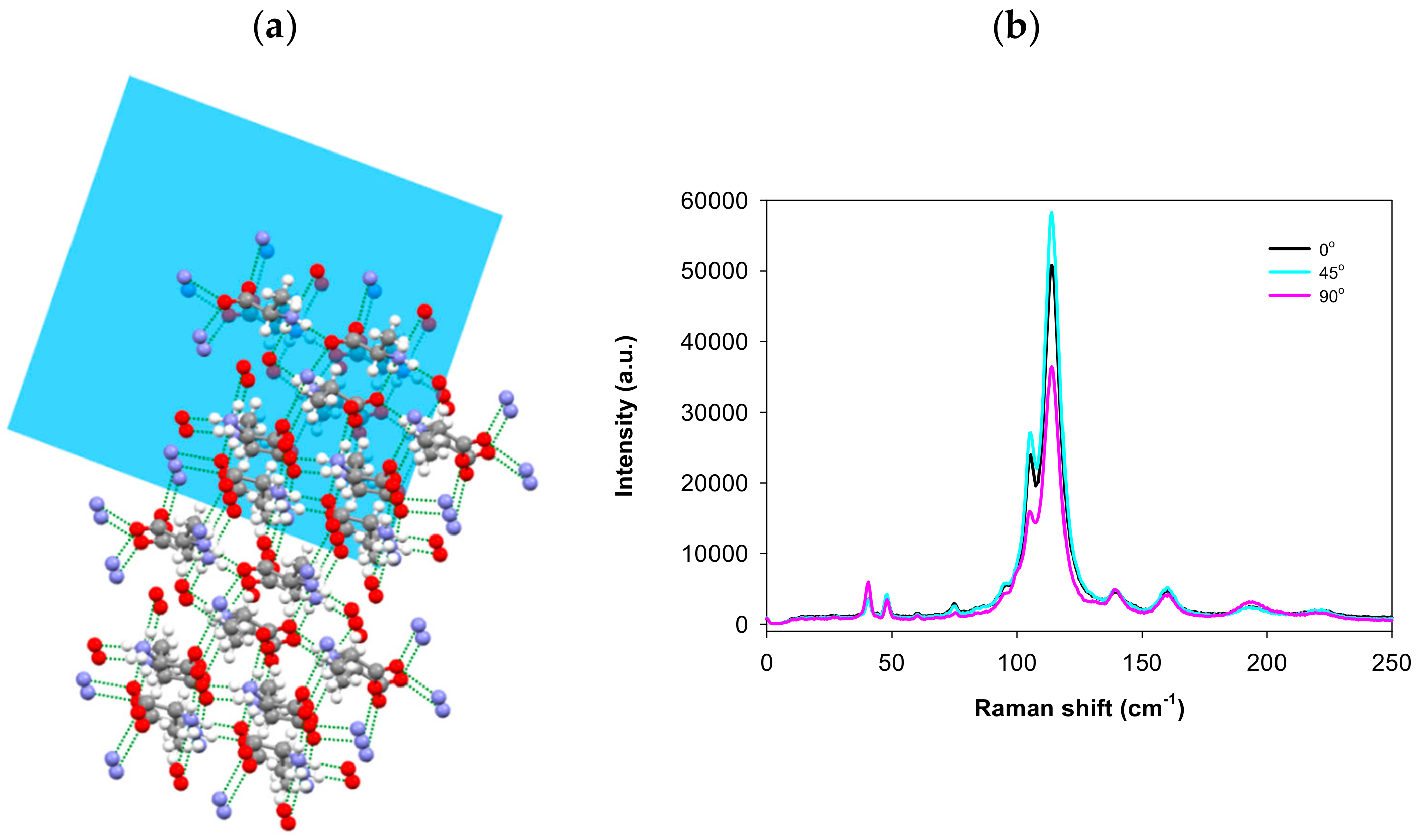
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, J.; Sarma, B.; Evans, J.M.B.; Myerson, A.S. Pharmaceutical crystallization. Cryst. Growth Des. 2011, 11, 887–895. [Google Scholar] [CrossRef]
- Morissette, S.L.; Almarsson, Ö.; Peterson, M.L.; Remenar, J.F.; Read, M.J.; Lemmo, A.V.; Ellis, S.; Cima, M.J.; Gardner, C.R. High-throughput crystallization: Polymorphs, salts, co-crystals and solvates of pharmaceutical solids. Adv. Drug Deliv. Rev. 2004, 56, 275–300. [Google Scholar] [CrossRef] [PubMed]
- Gues-Hornedo, N.R. Denette Murphy Significance of Controlling Crystallization Mechanisms and Kinetics in Pharmaceutical Systems. J. Pharm. Sci. 1999, 88, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, I.; Arbiol, J.; Dezanneau, G.; Cornet, A.; Morante, J.R. Crystalline structure, defects and gas sensor response to NO2 and H2S of tungsten trioxide nanopowders. Sens. Actuators B Chem. 2003, 93, 475–485. [Google Scholar] [CrossRef]
- Tabi, T.; Sajó, I.E.; Szabó, F.; Luyt, A.S.; Kovács, J.G. Crystalline structure of annealed polylactic acid and its relation to processing. Express Polym. Lett. 2010, 4, 659–668. [Google Scholar] [CrossRef]
- Deng, H.; Li, X.; Peng, Q.; Wang, X.; Chen, J.; Li, Y. Monodisperse magnetic single-crystal ferrite microspheres. Angew. Chem. 2005, 117, 2842–2845. [Google Scholar] [CrossRef]
- Aviv, H.; Nemtsov, I.; Mastai, Y.; Tischler, Y.R. Characterization of Crystal Chirality in Amino Acids Using Low-Frequency Raman Spectroscopy. J. Phys. Chem. A 2017, 121, 7882–7888. [Google Scholar] [CrossRef] [PubMed]
- Nemtsov, I.; Mastai, Y.; Tischler, Y.R.; Aviv, H. Chiral Purity of Crystals Using Low-Frequency Raman Spectroscopy. ChemPhysChem 2018, 19, 3116–3121. [Google Scholar] [CrossRef]
- Crasto, D.; Malola, S.; Brosofsky, G.; Dass, A.; Hakkinen, H. Single crystal XRD structure and theoretical analysis of the chiral Au30S (S-t-Bu) 18 cluster. J. Am. Chem. Soc. 2014, 136, 5000–5005. [Google Scholar] [CrossRef]
- Dong, Q.; Fang, Y.; Shao, Y.; Mulligan, P.; Qiu, J.; Cao, L.; Huang, J. Electron-hole diffusion lengths >175 μm in solution grown CH3NH3PbI3 single crystals. Science 2015, 347, 967–971. [Google Scholar] [CrossRef]
- Bianconi, A. Surface X-Ray absorption spectroscopy: surface EXAFS and surface XANES. Appl. Surf. Sci. 1980, 6, 392–418. [Google Scholar] [CrossRef]
- Yano, J.; Yachandra, V.K. X-ray absorption spectroscopy. Photosynth. Res. 2009, 102, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Schiff, D.; Aviv, H.; Rosenbaum, E.; Tischler, Y.R. Spectroscopic Method for Fast and Accurate Group A Streptococcus Bacteria Detection. Anal. Chem. 2016, 88, 2164–2169. [Google Scholar] [CrossRef] [PubMed]
- Aviv, H.; Berezin, S.; Agai, O.; Sinwani, M.; Tischler, Y.R. Deposition and Characterization of Roughened Surfaces. Langmuir 2017, 33, 1810–1815. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Zhu, X.; Fan, Q.; Wan, X. Raman spectra of amino acids and their aqueous solutions. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 78, 1187–1195. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.R.C.; Aschaffenburg, D.J.; Ofori-Okai, B.K.; Schmuttenmaer, C.A. Intermolecular vibrations in hydrophobic amino acid crystals: Experiments and calculations. J. Phys. Chem. B 2013, 117, 10444–10461. [Google Scholar] [CrossRef] [PubMed]
- Niehues, G.; Heyden, M.; Schmidt, D.A.; Havenith, M.; Berzofsky, J.A.; DeLisi, C.; Haller, E.E.; Havenith, M. Exploring hydrophobicity by THz absorption spectroscopy of solvated amino acids. Faraday Discuss. 2011, 150, 193–207. [Google Scholar] [CrossRef]
- Kalanoor, B.S.; Ronen, M.; Oren, Z.; Gerber, D.; Tischler, Y.R. New Method to Study the Vibrational Modes of Biomolecules in the Terahertz Range Based on a Single-Stage Raman Spectrometer. ACS Omega 2017, 2, 1232–1240. [Google Scholar] [CrossRef]
- Woods, K.N.; Pfeffer, J.; Dutta, A.; Klein-Seetharaman, J.; Haak, J.R. Vibrational resonance, allostery, and activation in rhodopsin-like G protein-coupled receptors. Sci. Rep. 2016, 6, 37290–37306. [Google Scholar] [CrossRef]
- Takahashi, M.; Okamura, N.; Ding, X.; Shirakawa, H.; Minamide, H. Intermolecular hydrogen bond stretching vibrations observed in terahertz spectra of crystalline vitamins. CrystEngComm 2018, 20, 1960–1969. [Google Scholar] [CrossRef]
- Hariharan, P.; Oreb, B.F.; Brown, N. A digital phase-measurement system for real-time holographic interferometry. Opt. Commun. 1982, 41, 393–396. [Google Scholar] [CrossRef]
- Marguí, E.; Van Meel, K.; Van Grieken, R.; Buendía, A.; Fontàs, C.; Hidalgo, M.; Queralt, I. Method for the determination of Pd-catalyst residues in active pharmaceutical ingredients by means of high-energy polarized-beam energy dispersive X-ray fluorescence. Anal. Chem. 2009, 81, 1404–1410. [Google Scholar] [CrossRef] [PubMed]
- De Boer, J.F.; Milner, T.E.; van Gemert, M.J.C.; Nelson, J.S. Two-dimensional birefringence imaging in biological tissue by polarization-sensitive optical coherence tomography. Opt. Lett. 1997, 22, 934–936. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-C.; Kim, E.-S. A novel configuration of LCD projectors for efficient orthogonal polarization of two projected views. Opt. Commun. 2006, 266, 55–66. [Google Scholar] [CrossRef]
- Iizuka, K. Welcome to the wonderful world of 3D: introduction, principles and history. Opt. Photonics News 2006, 17, 42–51. [Google Scholar] [CrossRef]
- Padgett, M.; Allen, L. Light with a twist in its tail. Contemp. Phys. 2000, 41, 275–285. [Google Scholar] [CrossRef]
- Duesberg, G.S.; Loa, I.; Burghard, M.; Syassen, K.; Roth, S. polarized Raman. Phys. Rev. Lett. 2000, 85, 5436–5439. [Google Scholar] [CrossRef]
- Haesler, J.; Schindelholz, I.; Riguet, E.; Bochet, C.G.; Hug, W. Absolute configuration of chirally deuterated neopentane. Nature 2007, 446, 526. [Google Scholar] [CrossRef]
- Barron, L.D.; Zhu, F.; Hecht, L.; Tranter, G.E.; Isaacs, N.W. Raman optical activity: An incisive probe of molecular chirality and biomolecular structure. J. Mol. Struct. 2007, 834, 7–16. [Google Scholar] [CrossRef]
- Gorelik, V.S.; Kaminskii, A.A.; Melnik, N.N.; Sverbil, P.P.; Voinov, Y.P.; Zavaritskaya, T.N.; Zlobina, L.I. Spontaneous Raman scattering spectra of ADP and DADP crystals in different polarization schemes. J. Russ. Laser Res. 2008, 29, 357–363. [Google Scholar] [CrossRef]
- Maeda, Y.; Udono, H.; Terai, Y. Raman spectra for β-FeSi2 bulk crystals. Thin Solid Films 2004, 461, 165–170. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.U.; Lee, J.; Park, H.J.; Lee, Z.; Lee, C.; Cheong, H. Anomalous polarization dependence of Raman scattering and crystallographic orientation of black phosphorus. Nanoscale 2015, 7, 18708–18715. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.H.; Storms, R.D. Raman Study of Hydrogen Bonding and Long-Wavelength Lattice Modes in an L-Alanine Single Crystal. J. Chem. Phys. 1971, 55, 5110–5119. [Google Scholar] [CrossRef]
- Razzetti, C.; Ardoino, M.; Zanotti, L.; Zha, M.; Paorici, C. Solution growth and characterisation of L-alanine single crystals. Cryst. Res. Technol. 2002, 37, 456–465. [Google Scholar] [CrossRef]
- Zhao, Y.; Luo, X.; Li, H.; Zhang, J.; Araujo, P.T.; Gan, C.K.; Wu, J.; Zhang, H.; Quek, S.Y.; Dresselhaus, M.S.; et al. Interlayer breathing and shear modes in few-trilayer MoS2 and WSe2. Nano Lett. 2013, 13, 1007–1015. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Men, Z.; Li, S.; Wang, S.; Li, Z.; Sun, C. Study of hydrogen bonding in ethanol-water binary solutions by Raman spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 189, 621–624. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nemtsov, I.; Aviv, H.; Mastai, Y.; Tischler, Y.R. Polarization Dependence of Low-Frequency Vibrations from Multiple Faces in an Organic Single Crystal. Crystals 2019, 9, 425. https://doi.org/10.3390/cryst9080425
Nemtsov I, Aviv H, Mastai Y, Tischler YR. Polarization Dependence of Low-Frequency Vibrations from Multiple Faces in an Organic Single Crystal. Crystals. 2019; 9(8):425. https://doi.org/10.3390/cryst9080425
Chicago/Turabian StyleNemtsov, Irena, Hagit Aviv, Yitzhak Mastai, and Yaakov R. Tischler. 2019. "Polarization Dependence of Low-Frequency Vibrations from Multiple Faces in an Organic Single Crystal" Crystals 9, no. 8: 425. https://doi.org/10.3390/cryst9080425
APA StyleNemtsov, I., Aviv, H., Mastai, Y., & Tischler, Y. R. (2019). Polarization Dependence of Low-Frequency Vibrations from Multiple Faces in an Organic Single Crystal. Crystals, 9(8), 425. https://doi.org/10.3390/cryst9080425