Morphology Investigation on Cyclopentane Hydrate Formation/Dissociation in a Sub-Millimeter-Sized Capillary
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
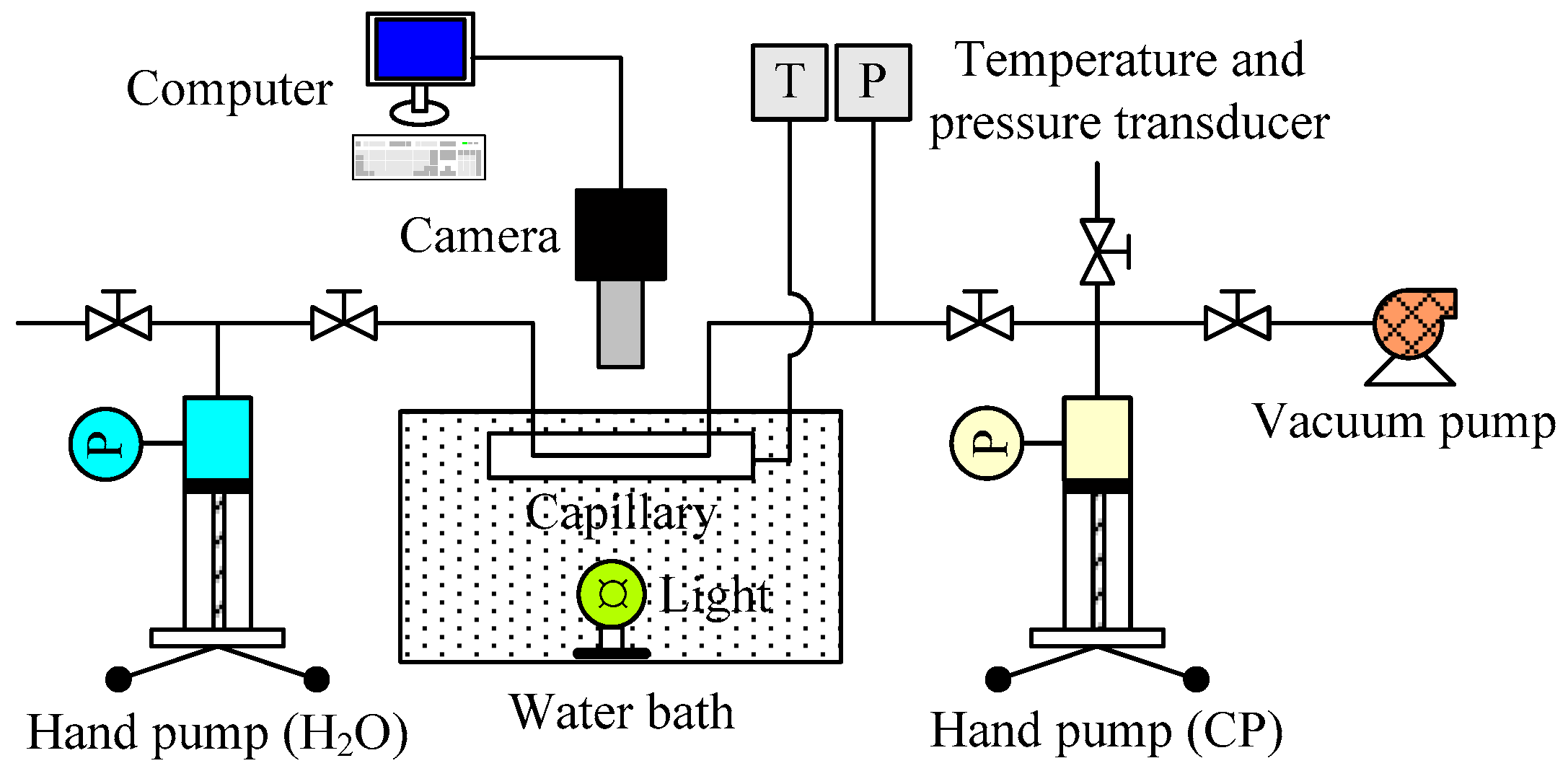
2.2. Apparatus
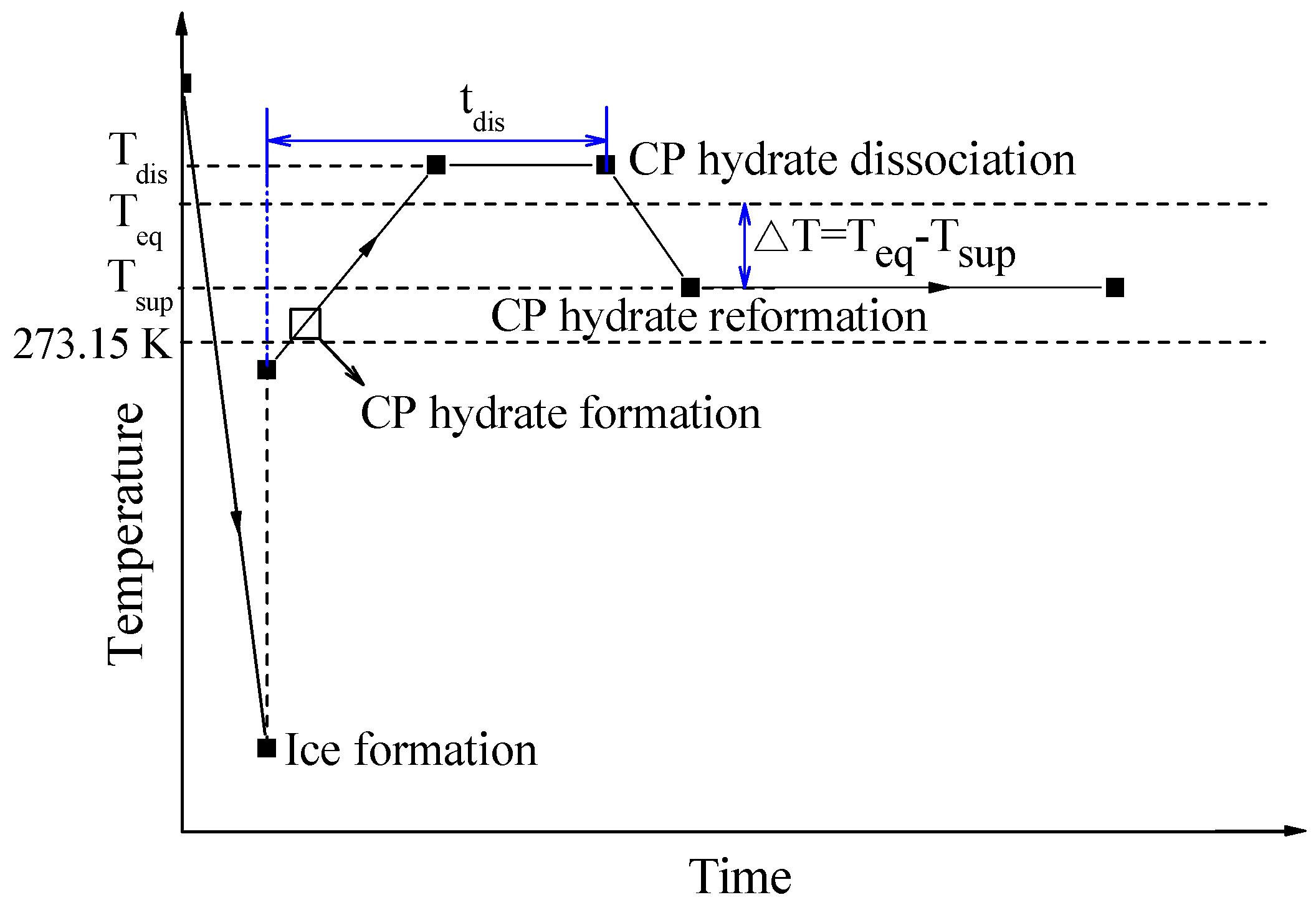
2.3. Procedure
3. Experimental Results and Discussion
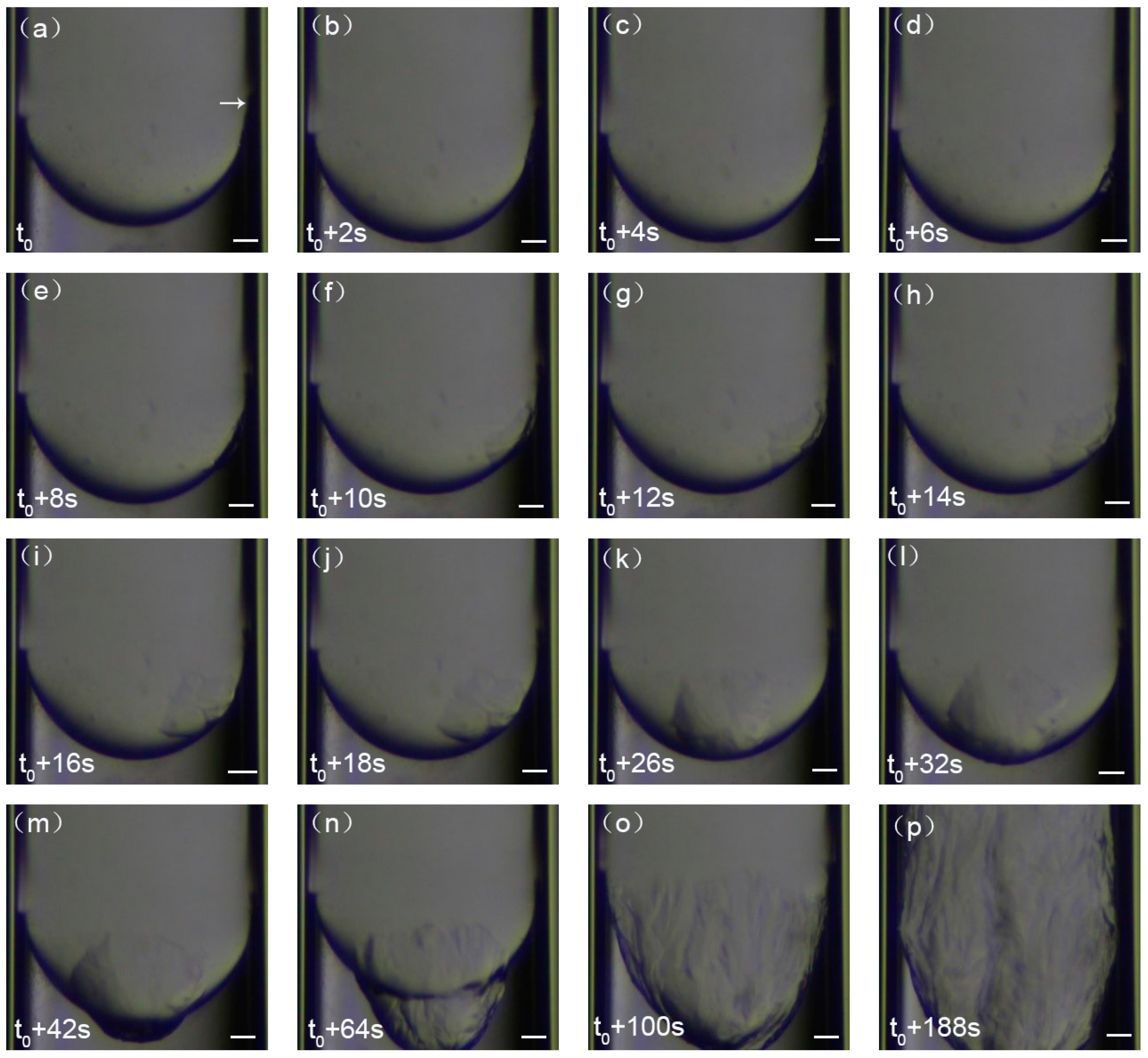
3.1. First Formation of CP Hydrate Crystals in Capillary
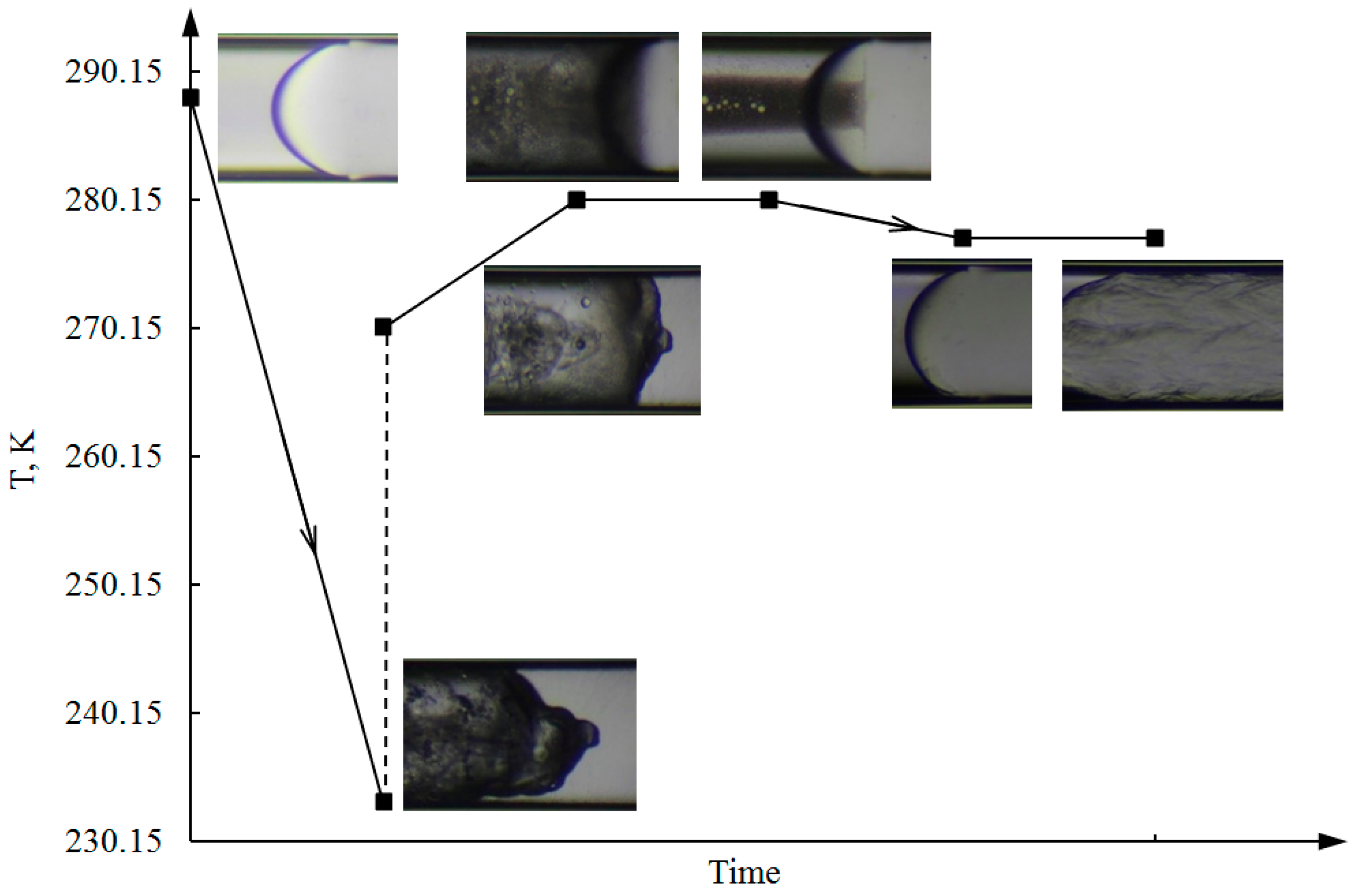
3.2. Melting of Ice, First Formation, and Dissociation of CP Hydrate in the Capillary
3.3. Reformation Process and Appearance of CP Hydrate Crystals in Capillary
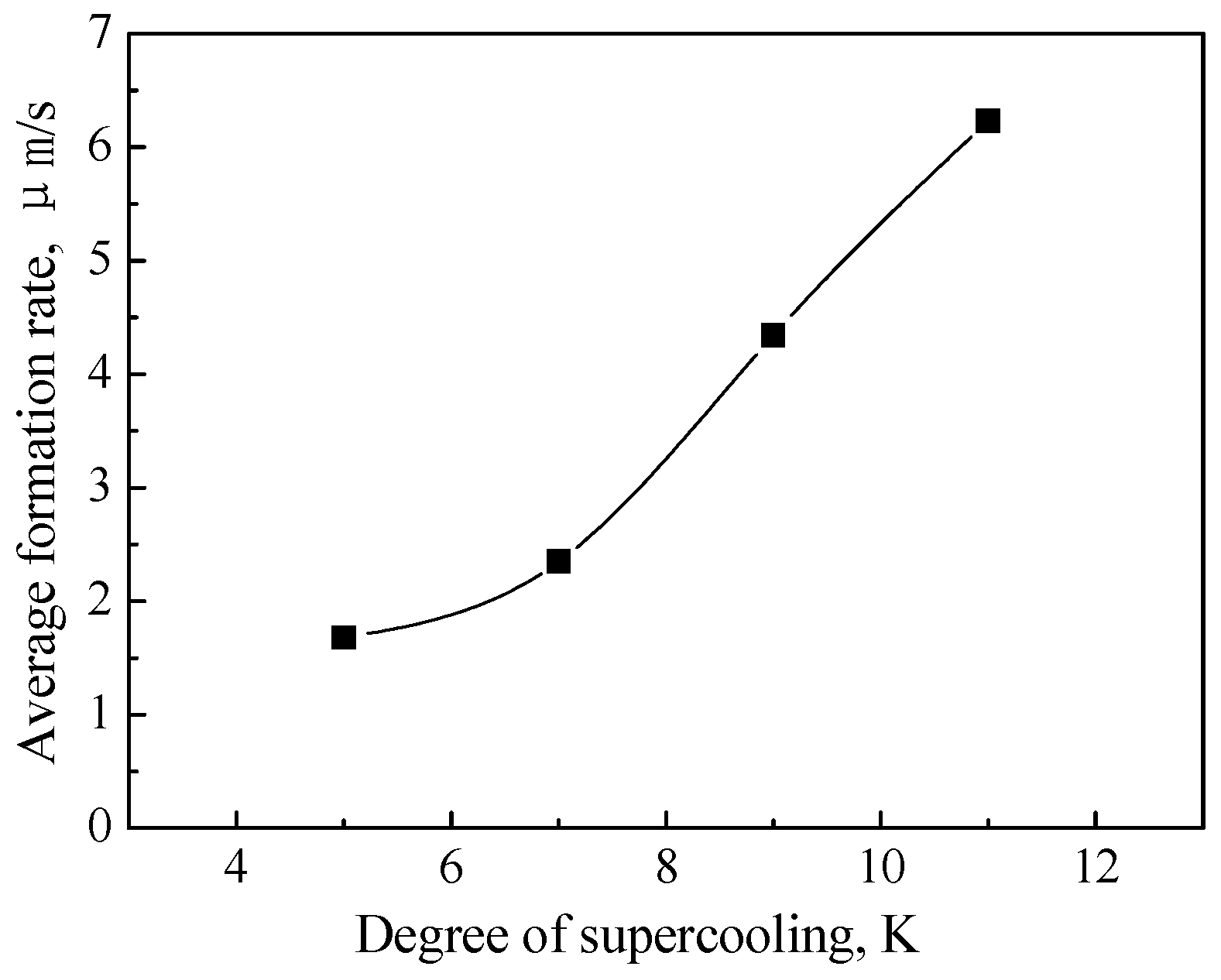
3.4. Influence of Supercooling Degree on CP Hydrate Crystals Reformation
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sloan, E.D.; Koh, C.A. Clathrate Hydrates of Natural Gases, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Englezos, P. Clathrate hydrates. Ind. Eng. Chem. Res. 1997, 32, 1251–1274. [Google Scholar] [CrossRef]
- Khurana, M.; Veluswamy, H.P.; Daraboina, N.; Linga, P. Thermodynamic and kinetic modelling of mixed CH4-THF hydrate for methane storage application. Chem. Eng. J. 2019, 370, 760–771. [Google Scholar] [CrossRef]
- Delroisse, H.; Torré, J.P.; Dicharry, C. Effect of a hydrophilic cationic surfactant on cyclopentane hydrate crystal growth at the water/cyclopentane interface. Cryst. Growth Des. 2017, 17, 5098–5107. [Google Scholar] [CrossRef]
- Aman, Z.M.; Koh, C.A. Interfacial phenomena in gas hydrate systems. Chem. Soc. Rev. 2016, 45, 1678–1690. [Google Scholar] [CrossRef] [PubMed]
- Sloan, E.D. Natural gas hydrates. J. Pet. Technol. 1991, 43, 1414–1417. [Google Scholar] [CrossRef]
- Van der Waals, J.A.; Platteeuw, J.C. Clathrate solutions. Adv. Chem. Phys. 1959, 2, 1–57. [Google Scholar]
- Tse, J.S.; McKinnon, W.R.; Marchi, M. Thermal expansion of structure I ethylene oxide hydrate. J. Phys. Chem. 1987, 91, 4188–4193. [Google Scholar] [CrossRef]
- Ripmeester, J.A.; Ratcliffe, C.I. Xenon-129 NMR studies of clathrate hydrates: New guests for structure II and structure H. J. Phys. Chem. 1990, 94, 876–877. [Google Scholar] [CrossRef]
- Hammerschmidt, E.G. Formation of gas hydrates in natural gas transmission lines. Ind. Eng. Chem. 1934, 26, 851–855. [Google Scholar] [CrossRef]
- Ruffine, L.; Broseta, D.; Desmedt, A. Gas Hydrates 2: Geoscience Issues and Potential Industrial Applications, 1st ed.; ISTE Ltd.: London, UK; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018. [Google Scholar]
- Taheri, Z.; Shabani, M.R.; Nazari, K.; Mehdizaheh, A. Natural gas transportation and storage by hydrate technology: Iran case study. J. Nat. Gas Sci. Eng. 2014, 21, 846–849. [Google Scholar] [CrossRef]
- Skovborg, P.; Rasmussen, P. A mass transport limited model for the growth of methane and ethane gas hydrates. Chem. Eng. Sci. 1994, 49, 1131–1143. [Google Scholar] [CrossRef]
- Naeiji, P.; Mottahedin, M.; Varaminian, F. Separation of methane–ethane gas mixtures via gas hydrate formation. Sep. Purif. Technol. 2014, 123, 139–144. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, J.; Liu, A.; Guo, X.; Yang, L.; Zhang, J. Experiment on the separation of tail gases of ammonia plant via continuous hydrates formation with TBAB. Int. J. Hydrogen Energy 2015, 40, 6358–6364. [Google Scholar] [CrossRef]
- Badu, P.; Linga, P.; Kumar, R.; Englezos, P. A review of the hydrate based gas separation (HBGS) process for carbon dioxide pre-combustion capture. Energy 2015, 85, 261–279. [Google Scholar]
- Tohidi, B.; Yang, J.; Salehabadi, M.; Anderson, R.; Chapoy, A. CO2 Hydrates could provide secondary safety factor in subsurface sequestration of CO2. Environ. Sci. Technol. 2010, 44, 1509–1514. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Rhee, Y.; Kang, S. Investigation of salt removal using cyclopentane hydrate formation and washing treatment for seawater desalination. Desalination 2017, 404, 132–137. [Google Scholar] [CrossRef]
- Lv, Q.; Li, X.; Li, G. Seawater desalination by hydrate formation and pellet production process. Energy Procedia 2019, 158, 5144–5148. [Google Scholar] [CrossRef]
- Adamova, T.P.; Stoporev, A.S.; Manakov, A.Y. Visual studies of methane hydrate formation on the water−oil boundaries. Cryst. Growth Des. 2018, 18, 6713–6722. [Google Scholar] [CrossRef]
- Baek, S.; Ahn, Y.H.; Zhang, J.; Min, J.; Lee, H.; Lee, J.W. Enhanced methane hydrate formation with cyclopentane hydrate seeds. Appl. Energy 2017, 202, 32–41. [Google Scholar] [CrossRef]
- Chen, X.; Espinoza, D.N. Surface area controls gas hydrate dissociation kinetics in porous media. Fuel 2018, 234, 358–363. [Google Scholar] [CrossRef]
- Zhang, B.; Zheng, J.; Yin, Z.; Liu, C.; Wu, Q.; Wu, Q.; Liu, C.; Gao, X.; Zhang, Q. Methane hydrate formation in mixed-size porous media with gas circulation: Effects of sediment properties on gas consumption, hydrate saturation and rate constant. Fuel 2018, 233, 94–102. [Google Scholar] [CrossRef]
- Wang, P.; Yang, M.; Chen, B.; Zhao, Y.; Zhao, J.; Song, Y. Methane hydrate reformation in porous media with methane migration. Chem. Eng. Sci. 2017, 168, 344–351. [Google Scholar] [CrossRef]
- Beltrán, J.G.; Servio, P. Morphological investigations of methane-hydrate films formed on a glass surface. Cryst. Growth Des. 2010, 10, 4339–4347. [Google Scholar] [CrossRef]
- Aifaa, M.; Kodama, T.; Ohmura, R. Crystal growth of clathrate hydrate in a flowing liquid water system with methane gas. Cryst. Growth Des. 2015, 15, 559–563. [Google Scholar] [CrossRef]
- Touil, A.; Broseta, D.; Hobeika, N.; Brown, R. Roles of wettability and supercooling in the spreading of cyclopentane hydrate over a substrate. Langmuir 2017, 33, 10965–10977. [Google Scholar] [CrossRef]
- Martinez de Baňos, M.L.; Carrier, O.; Bouriat, P.; Broseta, D. Droplet-based millifluidics as a new tool to investigate hydrate crystallization: Insights into the memory effect. Chem. Eng. Sci. 2015, 123, 564–572. [Google Scholar] [CrossRef]
- Sefidroodi, H.; Abrahamsen, E.; Kelland, M.A. Investigation into the strength and source of the memory effect for cyclopentane hydrate. Chem. Eng. Sci. 2013, 87, 133–140. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, C.; Yang, M.; Liu, W.; Xu, K.; Liu, Y.; Song, Y. Existence of a memory effect between hydrates with different structures (I, II, and H). J. Nat. Gas Sci. Eng. 2015, 26, 330–335. [Google Scholar] [CrossRef]
- Takeya, S.; Hori, A.; Hondoh, T.; Uchida, T. Freezing-memory effect of water on nucleation of CO2 hydrate crystals. J. Phys. Chem. 2000, 104, 4164–4168. [Google Scholar] [CrossRef]
- Zylyftari, G.; Ahuja, A.; Morris, J.F. Nucleation of cyclopentane hydrate by ice studied by morphology and rheology. Chem. Eng. Sci. 2014, 116, 497–507. [Google Scholar] [CrossRef]
- Karanjkar, P.U.; Lee, J.W.; Morris, J.F. Calorimetric investigation of cyclopentane hydrate formation in an emulsion. Chem. Eng. Sci. 2012, 68, 481–491. [Google Scholar] [CrossRef]
- Martínez de Baños, M.L.; Hobeika, N.; Bouriat, P.; Broseta, D.; Enciso, E.; Clément, F.; Brown, R. How Do gas hydrates spread on a substrate? Cryst. Growth Des. 2016, 16, 4360–4370. [Google Scholar] [CrossRef]
- Hobeika, N.; Martínez de Baños, M.L.; Bouriat, P.; Broseta, D.; Brown, R. High-resolution optical microscopy of gas hydrates. In Gas Hydrates 1: Fundamentals, Characterization and Modeling; ISTE Ltd.: London, UK; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017; Chapter 3. [Google Scholar]




| Materials | Specifications | Suppliers |
|---|---|---|
| CP | 98 wt% | Beijing InnoChem Technology Ltd. (Beijing, China) |
| Liquid nitrogen | 99 mol% | Beijing Yiyangfuli Commercial and Trading Ltd. (Beijing, China) |
| Deionized water | 15 × 106 Ω·cm | Water distillation unit (SZ-93) (Beijing, China) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Q.; Du, M.; Li, X.; Guo, X.; Yang, L. Morphology Investigation on Cyclopentane Hydrate Formation/Dissociation in a Sub-Millimeter-Sized Capillary. Crystals 2019, 9, 307. https://doi.org/10.3390/cryst9060307
Sun Q, Du M, Li X, Guo X, Yang L. Morphology Investigation on Cyclopentane Hydrate Formation/Dissociation in a Sub-Millimeter-Sized Capillary. Crystals. 2019; 9(6):307. https://doi.org/10.3390/cryst9060307
Chicago/Turabian StyleSun, Qiang, Mei Du, Xingxun Li, Xuqiang Guo, and Lanying Yang. 2019. "Morphology Investigation on Cyclopentane Hydrate Formation/Dissociation in a Sub-Millimeter-Sized Capillary" Crystals 9, no. 6: 307. https://doi.org/10.3390/cryst9060307
APA StyleSun, Q., Du, M., Li, X., Guo, X., & Yang, L. (2019). Morphology Investigation on Cyclopentane Hydrate Formation/Dissociation in a Sub-Millimeter-Sized Capillary. Crystals, 9(6), 307. https://doi.org/10.3390/cryst9060307





