1. Introduction
Zinc oxide, particularly in the form of nanostructures, has been studied enormously for its advantageous properties such as a direct wide band-gap (band-gap energy 3.37 eV), high exciton binding energy (60 meV), high electron mobility, and high piezoelectric constants [
1]. These properties open space for a broad range of applications including UV photodetectors and light-emitting devices, piezoelectric nanogenerators, solar cells, and chemical sensors [
2]. Zinc oxide typically crystalizes in the most thermodynamically stable wurtzite structure in a hexagonal crystal system that exhibits asymmetry, which is caused by the absence of a center of inversion along the c-direction [
3]. This structure consists of alternating planes of Zn and O atoms, where each Zn atom is tetrahedrally coordinated to four O atoms and vice versa. Due to the nocentrosymmetric nature of the wurtzite structure, ZnO exhibits a nonzero dipole moment along the c-axis, which results in spontaneous polarization. One dimensional ZnO nanostructures such as nanowires and nanorods grow along the c-axis. As a result, the top facets are formed by polar c-planes while the sidewalls are formed by nonpolar m-planes. This morphology is driven by thermodynamics, where polar faces have a higher surface energy than nonpolar faces; however, the aspect ratio can be controlled by the growth kinetics [
4].
The broad range of possible applications has led to the development of many routes of synthesis of nanostructured ZnO from both the solutions and from the gas phase. While the gas phase methods are usually expensive and require sophisticated equipment [
5], the solution phase synthesis, often called chemical bath deposition (CBD), is considered a simple, low-cost, and scalable method. As the synthesis is carried out in aqueous solution, the growth temperature is less than the boiling point of water [
6]. The family of “wet chemistry” CBD methods includes precipitation from saturated solutions of zinc salts [
7], hydrothermal synthesis [
6], sol–gel [
8,
9,
10,
11,
12,
13], and emulsion methods [
14] or spray pyrolysis [
15].
The simplicity of CBD methods makes them very popular. A control of the structural and morphological uniformity of these nanostructures is essential for their implementation into nanoscale devices and heterostructures. The morphology and properties of ZnO nanostructures can be controlled by the adjustment of the growth conditions (time, temperature) and chemical speciation in the solution (pH, nature of chemical precursors, etc.) [
10,
16,
17]. Concerning the arrays of ZnO nanorods, their structural uniformity and vertical alignment strongly depend on the properties of the substrate. High-quality ZnO nanorods can be grown by selective area homoepitaxy on lattice-matched ZnO substrates [
18] or by heteroepitaxy on GaN templates [
19]. However, these substrates are expensive and require lithographic techniques to achieve control over the nucleation of individual nanorods. The nucleation of the nanorods can also be achieved via the deposition of a suitable seed layer, which is usually formed by densely packed nanoparticles or nanocrystals. It has been shown that the ZnO nanorods grown by CBD homoepitaxially nucleate on top of ZnO nanocrystals oriented along the polar c-axis [
10]. When there is no requirement for the vertical orientation of the nanorod arrays, the seed layer can consist of randomly oriented nanoparticles deposited from colloidal solutions by dip coating or by electrophoretic deposition [
20,
21]. A great advantage of this approach is related to the fact that a heat treatment is not required. This allows for the deposition of the seed layer on virtually any substrate including textiles or plastics [
22]. For the arrays of vertically oriented nanorods, the seed layer is required to have a strong c-axis texture, low mosaicity, porosity, and surface roughness [
23].
The sol–gel process has become popular for the preparation of ZnO seed layers because it enables the control of the orientation of the crystallites [
11,
12,
23,
24,
25,
26]. During the sol–gel process a zinc salt is transformed from a solution to a solid deposit by using a sol or a gel as an intermediate step at a low temperature. The sol–gel method comprises several chemical processes: hydrolysis of molecular precursors; polymerization via addition of bimolecular ions, formation of oxo-, hydroxyl- and aqua-bridges; condensation by dehydration; and nucleation and growth [
27].
The precursor solution consists of three principal components: a zinc precursor, a solvent, and an additive. The precursor acts as a source of zinc ions. Many chemical forms of the zinc salt can serve as a precursor, either as inorganic salts such as nitrates, chlorides, and perchlorites, or organic salts such as acetate and acetylacetonates. The main drawback of inorganic precursors is the difficulty in removing anionic species in the final product. Therefore, the use of zinc acetate prevails, because the decomposition of by-products occurs under the heat treatment of the xerogel (dried gel). In addition, zinc acetate plays an important role in sol formation by complexing the zinc ions in competition with the additive [
28]. The solvent is necessary to dissolve the zinc salt by the in situ formation of alkoxides or alkoxy-complexes. It is required that the solvent has a relatively high dielectric constant and appropriately high boiling point. The most frequently used solvents are alcohols with a low-carbon number such as methanol, ethanol, 1-propanol, and 2-methoxyethanol. During the aging time of the sol, the zinc complexes undergo hydrolysis and inorganic polymerization and form zinc oxy-acetate Zn
4O(CH
3COO)
6 nanoparticles [
23,
29]. The last component, the additive, acts as a base and a complexing agent. It must have at least one functional group, which enhances its role as a chelating agent. The additive, in some cases, also facilitates the dissolution of the zinc salt in the alcoholic media. Plenty of additives including mono-, di-, tri-ethanolamine, and some inorganic bases (LiOH, NaOH) have been reported [
17,
30]; nevertheless, the use of monoethanolamine (MEA) prevails. The additive component also helps to form a stable sol and avoid the rapid precipitation of zinc hydroxide during the aging time of the sol [
28].
After an appropriate aging time, typically ranging from hours to days, the sol is deposited by dip coating or spin coating, which results in the formation of a xerogel on the substrate. The xerogel film further undergoes a heat treatment to obtain the ZnO seed layer with the desired properties. The heat treatment is in most cases a two-step process consisting of a low temperature preheating and a high temperature annealing. The preheating follows each deposition cycle and is one of the most important factors in controlling the seed layer properties. It governs both the orientation of the crystallites during solvent evaporation and the removal of organic compounds. The preheating temperature significantly varies from case to case, depending on the composition of the precursor solution. A clear rule is that the temperature should be higher than the boiling points of the solvent and of the additive, but also of the organic by-products. The preheating temperatures reported in the literature vary across a broad range from 150 °C to 450 °C [
10,
12,
13,
24,
26,
31]. The preferential orientation along the c-axis increases with increasing temperature; however, too high temperature results in simultaneous crystallization of ZnO, which can prevent the decomposition of the organic species and can lead to the rapid evaporation of the solvent and of the by-products during the high temperature annealing. This can result in the formation of cracks or punctures in the layer, which deteriorates the degree of orientation of the crystallites [
25,
31,
32]. Typically reported annealing temperatures lie in the range of 500–600 °C in ambient atmosphere [
8,
12,
18,
33]. Occasionally annealing in an inert atmosphere (argon, nitrogen) has also been investigated [
23,
34] where the inert atmosphere in comparison to ambient conditions allows for a high temperature annealing, which has been reported to enhance the texture [
23] though at the expense of the uniformity and reproducibility. During the annealing process, the crystallization of ZnO occurs and small ZnO crystallites are sintered to form larger ones.
Despite the fact that many articles have been devoted to the study of the heat treatment during the preparation of ZnO seed layers [
12,
23,
24,
25,
26,
31], there are still plenty of open questions in the fabrication process of high quality seed layers for the growth of vertically aligned ZnO nanorods. One of the crucial parameters of the process is the preheating temperature, the influence of which is, however, often underestimated.
One of the first in-depth studies from Ohyama et al. [
12] was focused on a very similar chemical system to the one reported in this paper; however, by that time, there was no interest in using the layers for the growth of ZnO nanorods. Moreover, no aging time of the precursor solution was employed, even though the aging time can play a significant role in the chemical reactions and polymerization of the sol [
17]. The preheating temperature of 300 °C was reported as the most appropriate to evaporate and decompose the organic species. Another important conclusion was that the preheating temperature has to be lower than the temperature at which crystallization of ZnO occurs, otherwise the crystallization process can prevent the decomposition of the organic compounds. This conclusion has been further supported by other studies [
24,
25,
26]. The question that remains unanswered is whether the chosen temperature is high enough to decompose all the organic species. There have been several studies on the thermogravimetric analysis of zinc acetate or of the deposited xerogel [
24,
25,
26,
31]. While Greene et al. [
31] used a one-step heat treatment and suggested that the temperature of 350 °C was sufficient to crystalize ZnO from a highly diluted solution of zinc acetate in absolute ethanol (c = 0.005 M), Kim et al. [
25] claimed that the evaporation and decomposition occurred only up to 250 °C if isopropanol was used as a solvent. On the other hand, Wang et al. [
26] suggested a higher temperature of preheating (up to 450 °C) because their analyses confirmed the fact that the zinc copolymers Zn
4O(CH
3COO)
6 and ZnNH(C
2H
4O)
2 were present (Fourier-transform infrared spectroscopy) in the precursor solution and started to decompose at T = 392 °C (thermogravimetric analysis). One of the recent studies from Demes et al. [
24] based on thermogravimetric analysis showed that the decomposition of the organic species took place up to 330 °C. A table summarizing the parameters used in the cited articles is shown in the
Supplementary Materials (Table S1 in Supplementary Materials).
In this study, we focused on the preparation of ZnO seed layers by the sol–gel method deposited by dip coating on silicon substrates for the synthesis of vertically aligned zinc oxide nanorods by the CBD method. We fixed the chemistry and the parameters of the dip coating and particularly investigated the influence of the heat treatment process including both the temperatures and the atmosphere. We varied the preheating temperature and the annealing conditions and thereby affected the crystallinity and morphology of the seed layer, which influences the vertical alignment of ZnO nanorods grown by CBD. Apart from conventional annealing in the air, annealing in Ar was investigated, as it enables the application of higher annealing temperatures without introducing oxygen-related defects into ZnO nanostructures.
2. Materials and Methods
The ZnO seed layers were deposited on a silicon substrate by dip coating. The chemical precursor solution was obtained by the sol–gel synthesis, where the reactants were zinc acetate dihydrate (ZAD, Zn(CH
3COO)
2.2 H
2O) from Sigma-Aldrich and monoethanolamine (MEA, C
2H
7NO) in 2-methoxyethanol from Carl Roth in equimolar concentration (0.375 M). The solution was heated and stirred to dissolve ZAD and to initialize the hydrolysis of the sol. The aging time was set to 24 hours to continue polymerization of the sol. The deposition was carried out by three times repeated dip coating with the preheating step after each cycle. The preheating took place at 300 °C, 350 °C, or 400 °C for 10 min in a laboratory muffle furnace in air to evaporate the solvent and decompose and remove the residual organic compounds. The last step was annealing, to ensure the ZnO recrystallization. Moreover, the annealing decreased the number of structural defects [
26,
35]. The seed layers were annealed either in Ar at 800 °C in a tubular furnace or in the air at 600 °C in a muffle furnace for 1 hour. For comparison, the ZnO seed layers were also deposited by electrophoretic deposition of a commercially available solution of ZnO nanoparticles dispersed in ethanol (supplied by Sigma-Aldrich). Full details of the electrophoretic deposition are described in our previous work [
21]. The ZnO nanorods were grown on the seeded substrates by CBD synthesis in equimolar solution consisting of zinc nitrate hexahydrate (Zn(NO
3)
2.6 H
2O) and hexamethylenetetramine (C
6H
12N
4) from Sigma-Aldrich dissolved in deionized water. Both 0.05 M solutions were mixed together and preheated to 41 °C. Then, the seeded substrate was immersed upside down into this solution. The reaction temperature was 95 °C and the growth time was 2 hours.
The seed layers and the ZnO nanorods were characterized by scanning electron microscopy (SEM; Prague, Czech Republic), x-ray diffraction (XRD; Prague, Czech Republic), atomic force microscopy (AFM; Prague, Czech Republic), transmission electron microscopy (TEM; Prague, Czech Republic), and photoluminescence spectroscopy (PL; Prague, Czech Republic). SEM images were collected with Tescan Lyra 3 GM using the in-beam SE detector. The sizes of the seed layer crystallites and the diameters of the nanorods were obtained from SEM images using ImageJ software [
36] by averaging a high number of measurements (≈ 100). The XRD data were collected with an X’Pert PRO θ-θ powder diffractometer in Bragg-Brentano geometry using Cu Kα radiation (λ = 1.5418 Å, U = 40 kV, I = 30 mA). The data were scanned with an ultrafast detector X’Celerator over the angular range 30–80° (2θ), using the step size 0.0167° and a counting time 20.32 s per step. The AFM analysis was conducted by a JPK NanoWizard 3 Nanooptics atomic force microscope. AC non-contact mode was used in combination with TESPA-V2 probes by BRUKER with the resonance frequency of 320 kHz. The TEM, high resolution TEM (HRTEM), and automated crystalline orientation and phase mapping (ACOM) [
37] measurements of the ZnO seed layers were carried out using a JEOL JEM-2200FS system. The microscope was a FEG instrument operating at 200 kV, equipped with a Nanomegas Astar beam precession and scanning system. The precession angle in our ACOM experiments was 0.63° and the step size was 2.5 nm. The TEM lamellas were prepared by the lift-out method with the Tescan Lyra 3 FIB-SEM. The PL spectra were measured with a set-up comprising a HeCd laser (325 nm) as an excitation source, a grating monochromator Jobin Yvon THR 1000, and a GaAs photomultiplier (R943-02, Hamamatsu).
3. Results and Discussion
The seed layers and the CBD grown nanorods on these seed layers are shown in the SEM images in
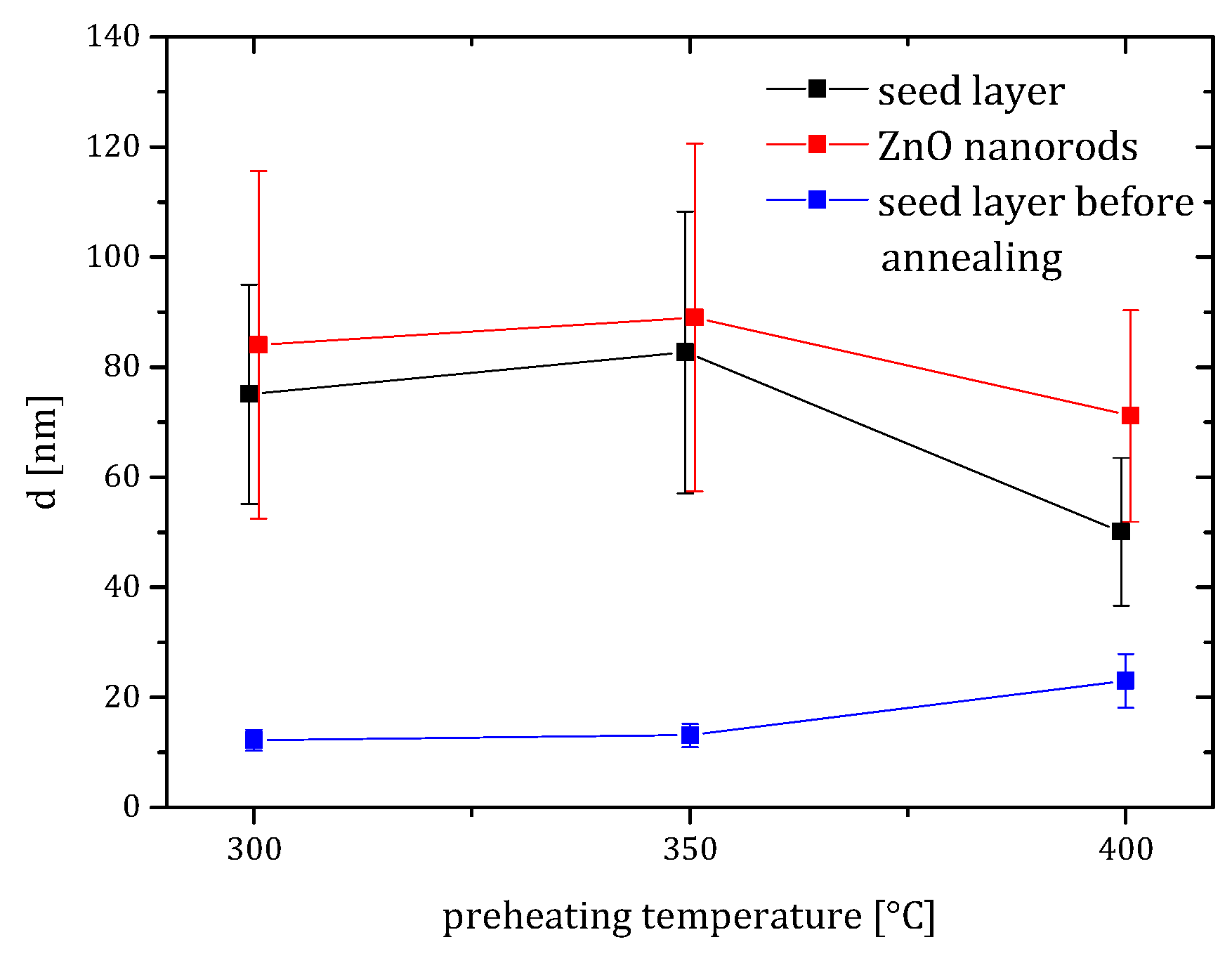
Figure 1. After preheating, the xerogel film deposited by dip coating was transformed into a compact layer consisting of ZnO crystallites with a rounded shape by evaporating the solvent and by decomposing and removing organic compounds. The morphology of the deposited layers was investigated by SEM, from which an average diameter of the crystallites was determined using ImageJ software (
Figure 2), and by AFM, from which the surface roughness was assessed (
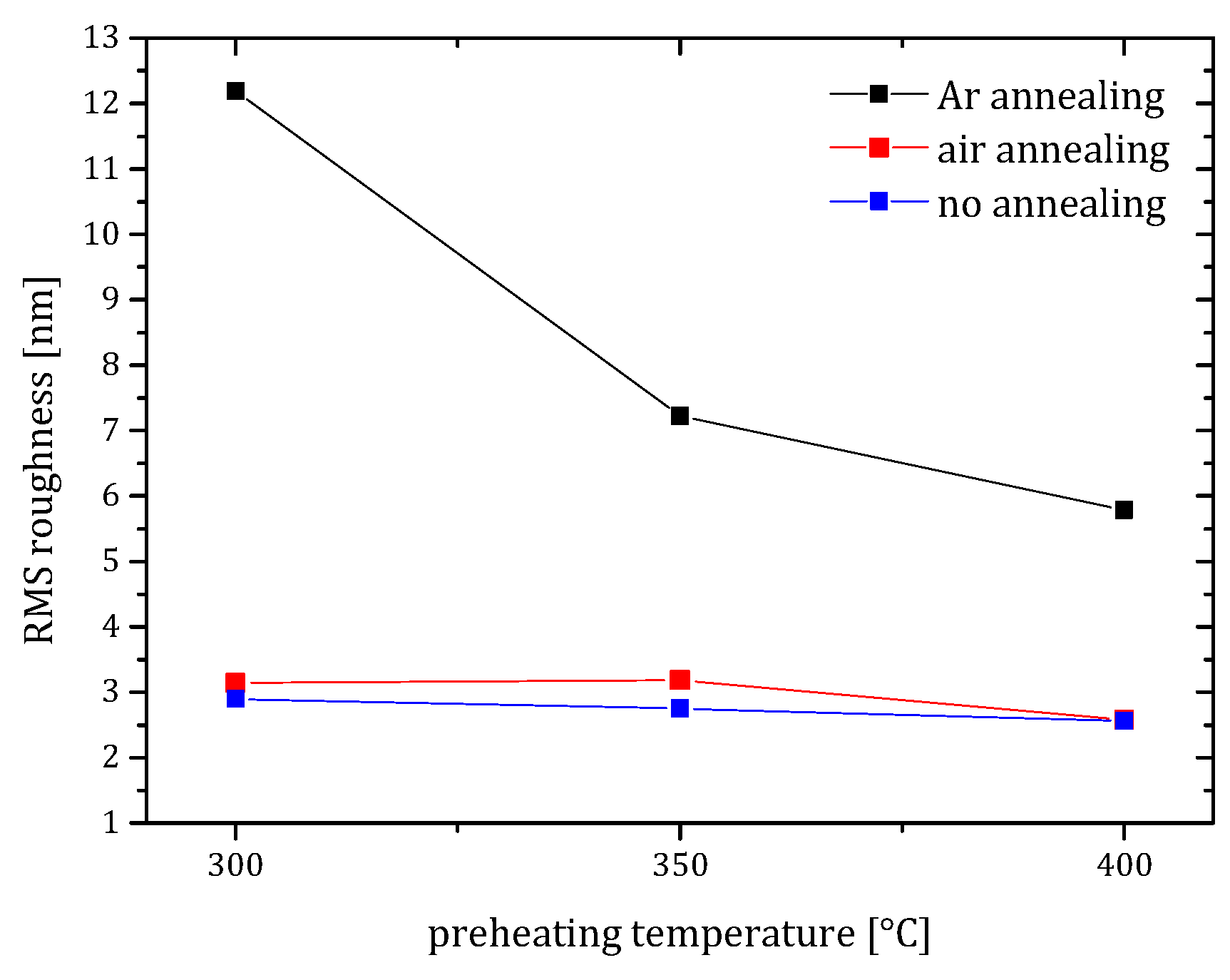
Figure 3).
These measurements clearly show that the diameter of the crystallites moderately increased when the preheating temperature was raised to 400 °C. Moreover, the surface roughness stayed low or even decreased with increasing temperature. Such a high preheating temperature has rarely been reported for the particular precursor–solvent–additive chemical system [
29]; however, it will be further shown that it is highly beneficial for the growth of vertically oriented ZnO nanorods. The high preheating temperature was reported to enhance the decomposition of zinc copolymers in the xerogel film, leaving small pores in the film [
26]. When low preheating temperatures are employed, the copolymers are not decomposed completely and are prone to combustion during the subsequent annealing step. As a result, rough and porous ZnO seed layers form.
During the annealing step, a film made of larger discrete crystallites is formed (
Figure 1Ⅰ). For both annealing atmospheres, the sizes of the seed layer crystallites are systematically larger than the size of the crystallites of the seed layer before annealing (
Figure 2), where the size ratio depends on the preheating temperature. After annealing in the air, the seed layer crystallites preheated at 300 °C and 350 °C had a rounded shape with diameters ranging from 50 to 110 nm with a relatively broad distribution of sizes. Histograms of the size distributions can be seen in the
Supplementary Materials (Figure S1). When the preheating temperature was increased to 400 °C, the diameter decreased and the size distribution was significantly narrowed (
Figure 1Ⅰ). The surface roughness was low at all preheating temperatures with a moderate decrease for the samples preheated to 400 °C (
Figure 3). For the seed layers annealed in the air at 600 °C, a good vertical orientation was achieved for all preheating temperatures. Highly c-axis textured seed layers with a low roughness and porosity promote nucleation on the top free surfaces of the crystallites [
10]. The nanorods then grow epitaxially and adopt a c-plane orientation of the seed layer crystallites, which promotes vertical alignment of the nanorod array (
Figure 1Ⅱ). The size analysis revealed that the diameters of the nanorods were strongly correlated with the sizes of the crystallites of the seed layers. Consequently, the nanorods grown on the seed layers preheated to 400 °C showed a narrow size distribution (
Figure 1Ⅰc and
Figure 2). The nanorods had a moderately larger diameter than the seed layer crystallites. This behavior is related to the not negligible lateral growth rate of the nanorods in nonpolar directions, which can be controlled by the growth parameters, dopants, and surfactants [
4,
13,
38].
After annealing in Ar at 800 °C, irregular and not well-defined shapes of the crystallites were observed by SEM, which did not allow us to carry out the size analysis of the crystallites. Moreover, the overall surface coverage of the seed layer annealed in Ar was lower than the surface coverage of the layers annealed in the air. Large crystalline platelets were observed for the preheating temperature of 400 °C on which vertically oriented nanorods were grown. Lower preheating temperatures resulted in the growth of poorly oriented nanorods
Figure 1Ⅱd,e) related to low texture of the seed layer, its high surface roughness (
Figure 3) and its high porosity (
Figure 1Ⅰd,e). The low texture and the high surface roughness and porosity all contributed to the loss of vertical alignment of the nanorods. Under these conditions, the nanorods not only nucleate on polar c-planes of the crystallites, but also on the semipolar planes, which are not vertically aligned [
39].
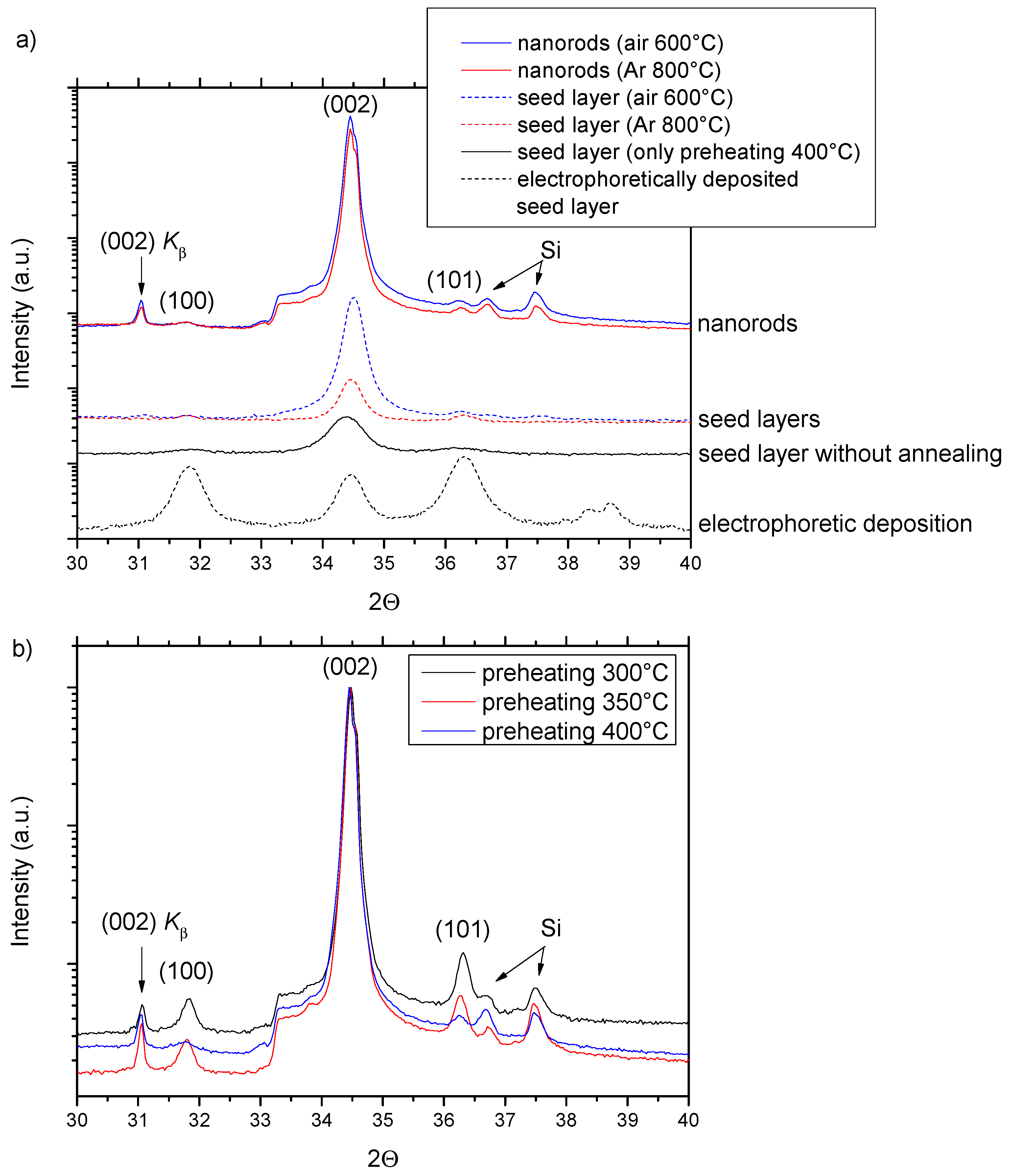
The crystalline analysis by XRD was carried out on both the seed layers and the nanorod arrays grown on the seed layers (
Figure 4). The XRD patterns with the
y-axis in logarithmic scale were analyzed in the range of the angle 2θ between 30° and 40°, where the three main reflections are located for the ZnO wurtzite structure. The complete XRD scans (2θ ≈ 30°–80°) showed reflections corresponding uniquely to ZnO, except for weak reflections from the Si substrate. The complete diffraction patterns are shown in the
Supplementary Materials (Figure S2). First, we discuss the diffraction patterns of the seed layers. For comparison, we show the electrophoretically deposited seed layer with the intensities of the three principal diffraction peaks (100), (002), and (101) corresponding to a random orientation of ZnO nanoparticles. The XRD pattern for the seed layer after preheating at 400 °C was dominated by the (002) reflection, which corresponded to a strong c-axis texture. The same applies to the seed layers after annealing in both air at 600 °C and Ar at 800 °C; however, the full width half maximum of the peak was significantly narrowed. This points to a conclusion that after preheating, the small crystallites are c-axis oriented and that during annealing, the crystallites coalesce into larger ones without losing their orientation. As claimed when the morphology of the seed layers was discussed, the nanorods grow epitaxially and adopt a c-plane orientation of the seed layer crystallites. This translates into the vertical alignment of the nanorods and the corresponding XRD patterns are again dominated by the (002) reflection (
Figure 4a). Similar XRD patterns were measured for the seed layers preheated to 300 °C and to 350 °C and subsequently annealed in the air at 600 °C (
Figure S2). This situation was different for the annealing in Ar at 800 °C (
Figure 4b), where the high preheating temperature of 400 °C was shown to be crucial for the suppression of the (100) and (101) peaks and of the corresponding vertical misalignment.
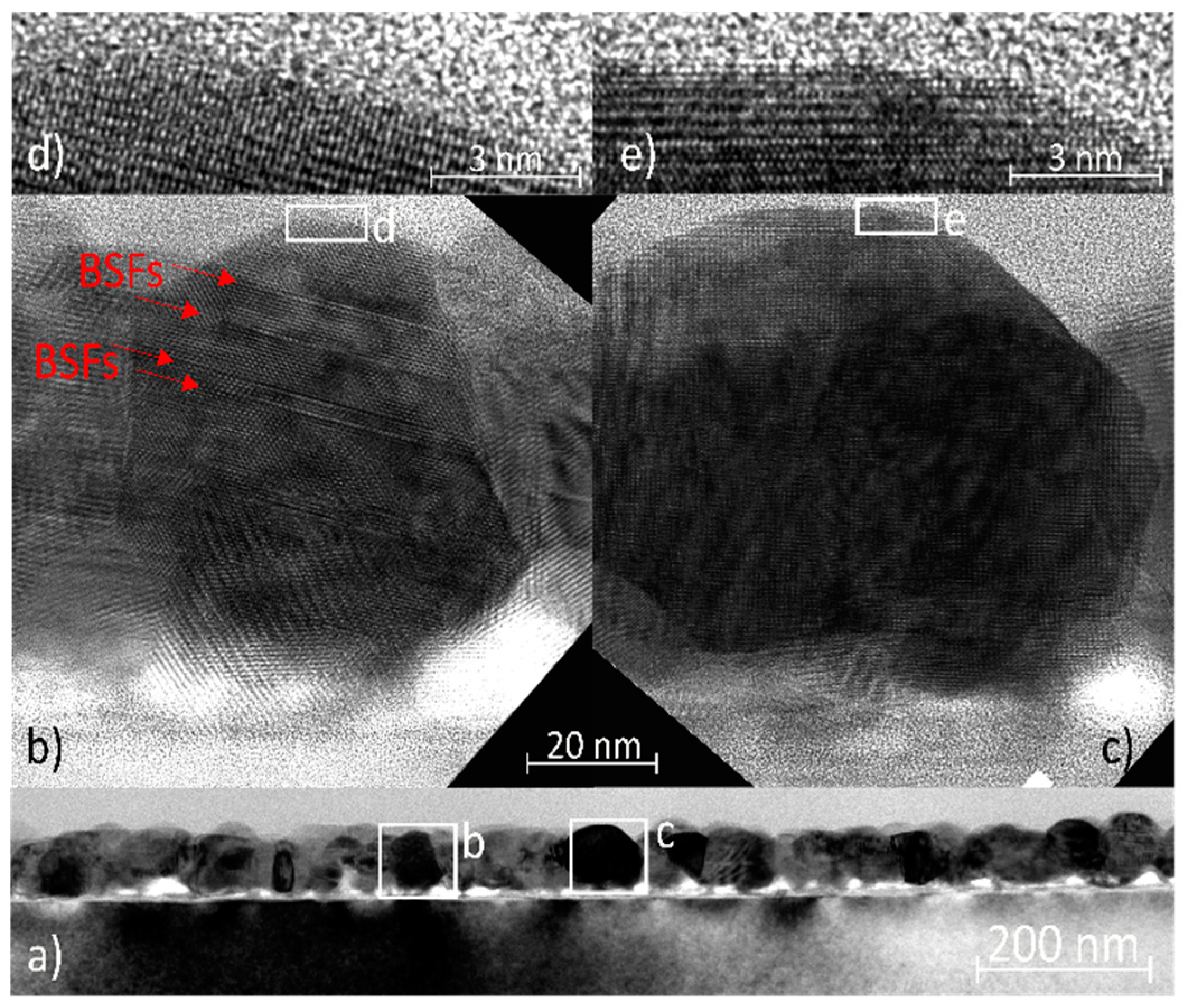
The seed layer preheated at 400 °C and annealed in the air at 600 °C was further investigated by TEM and HRTEM. The low-magnification image shows a seed layer of approximately 100 nm in thickness containing crystallites with diameters ranging from ~40 to 60 nm (
Figure 5a). The seed layer was not fully in contact with the substrate. The gaps at the interface formed as a result of the shape of the crystallites varying from quasi-spherical to columnar. Moreover, this non-uniformity also supports the conclusion from [
40] that the structural role of the substrate is negligible and the ZnO crystallites are attached only by weak bonding to the native amorphous silicon oxide layer, which is approximately 10 nm thick. Two crystallites from the seed layer were chosen for more detailed imagining along the <11−20> zone axis (
Figure 5b,c). Both crystallites were quasi-spherical, terminated with c-planes (0001), and tapered with the side facets composed of semi-polar planes. The crystallite in
Figure 5 contains several basal stacking faults (BSFs), which were not terminated by partial dislocations, but by free surfaces or grain boundaries, as reported in [
41]. The c-plane surfaces can be considered atomically sharp according to HRTEM images (
Figure 5d,e).
To prove the conclusions from other characterization techniques, the seed layers preheated at 400 °C and annealed in the air at 600 °C were further investigated by ACOM-TEM (ASTAR) (
Figure 6). The alignment of the crystallites along the c-axis strongly prevailed, which was confirmed by the red color in
Figure 6b. The ASTAR maps recorded in the basal plane (
Figure 6c) did not show any preferential in-plane orientation.
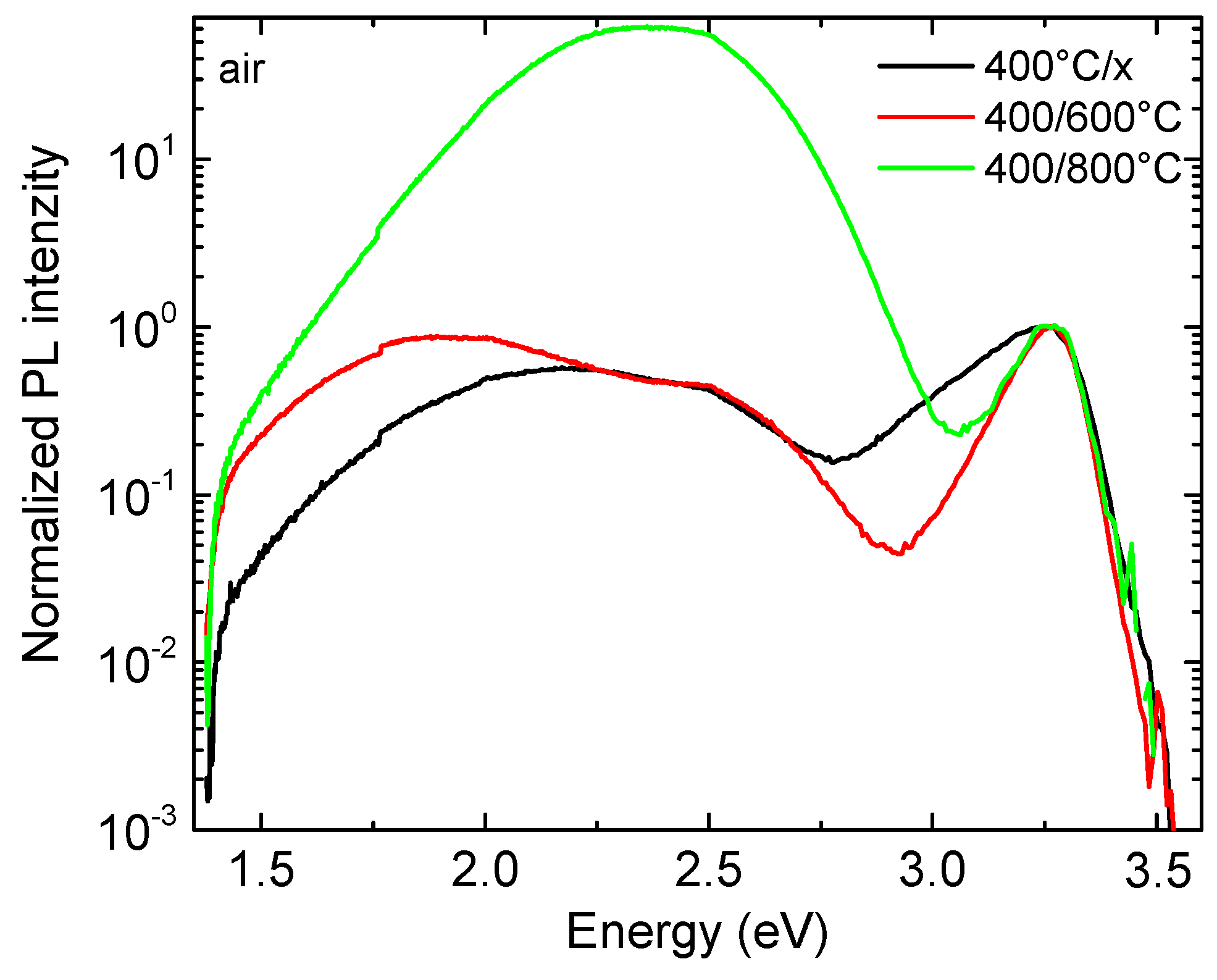
To obtain more information about the nature of defects in the seed layers, they were further investigated by PL spectroscopy. As shown in
Figure 7, the annealing in the air significantly affected the optical properties of the ZnO seed layer. For the seed layers preheated at 400 °C, we observed a broad, asymmetric excitonic emission band (NBE, near-band-edge) with a maximum at 3.26 eV. Such behavior is expected in the case of a large defect concentration. The annealing in the air at 600 °C and 800 °C resulted in the narrowing of the excitonic band, which indicates an improvement in both the optical and structural properties of the seed layer. The qualitative picture of the broad composite band (DLE, deep level emission) in the visible region for different annealing temperature did not change. The DLE was composed of the orange luminescence at ~2.1 eV and of the green luminescence at ~2.47 eV. The origin of the orange emission band is generally attributed to different sources such as ionized oxygen interstitials or Li impurities [
42,
43,
44]. The origin of the green luminescence was assigned to the transitions in which oxygen vacancies, oxygen interstitials, or extrinsic defects such as Cu are involved [
45,
46]. When the temperature increased from 400 to 600 °C, we only observed a moderate increase in the integral DLE intensity, which is typical for the annealing of nanostructured ZnO in air and can be explained by the increase in oxygen related defects such as V
O and O
i [
47]. Further increase of the annealing temperature to 800 °C caused a large increase of the integral DLE intensity, which is related to the degradation of the seed layer due to surface etching. In our previous work, we demonstrated visible degradation of ZnO nanorods after annealing at the temperatures above 600 °C in the air/vacuum, while in an inert atmosphere such behavior was not observed [
48].
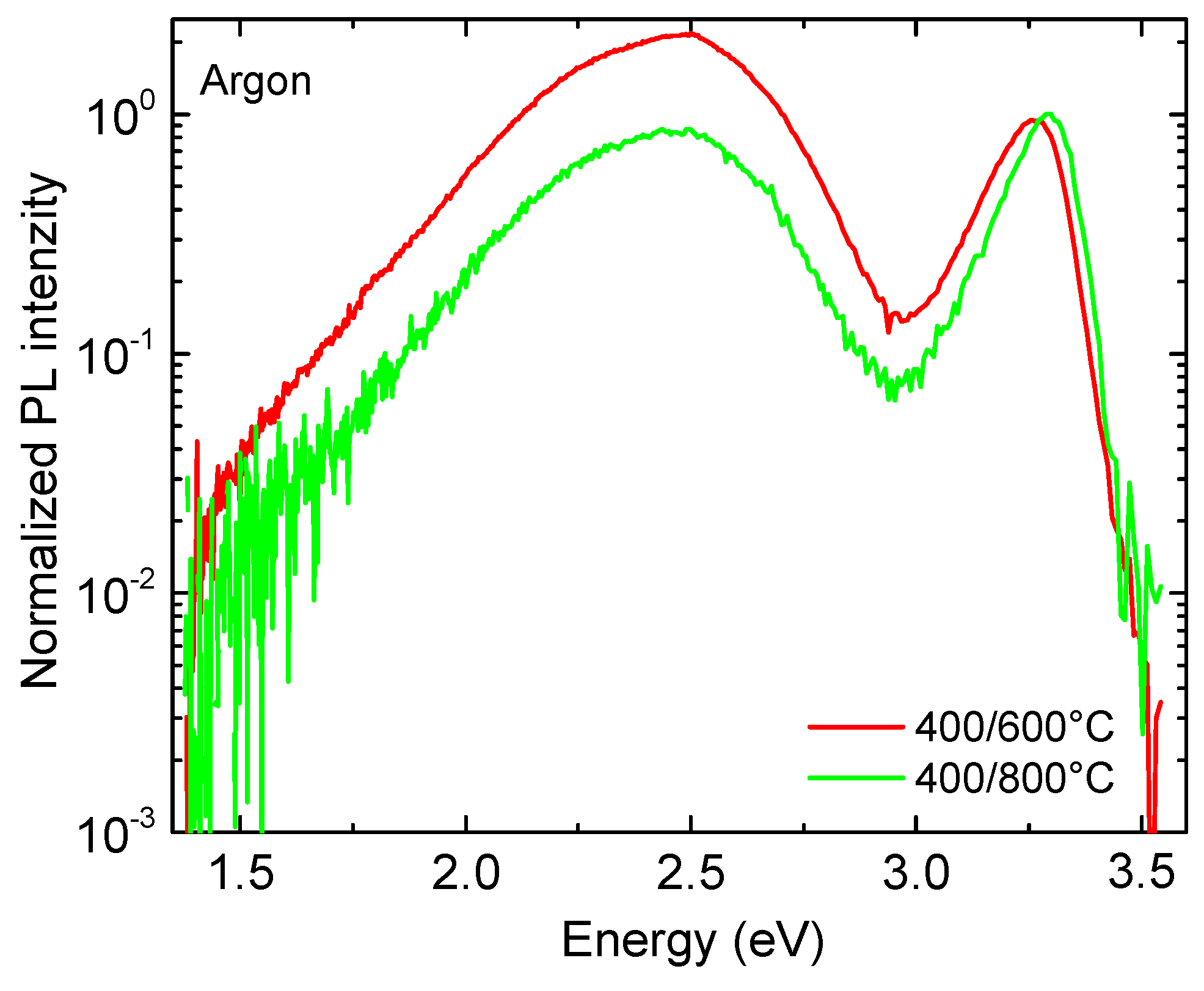
The NBE emission after annealing in argon (
Figure 8) showed a similar behavior to that in air. The FWHM of the DLE peak was significantly lowered with an increase in the annealing temperature. In the DLE spectra, two green bands with the maxima at 2.47 eV and 2.35 eV were observed [
45]. The integral intensity of the DLE substantially decreased and the NBE/DLE intensity ratio increased when the temperature was increased from 600 °C to 800 °C. Such behavior is typical for ZnO, which is annealed in an inert atmosphere [
49]. The increase in the NBE/DLE intensity ratio indicates substantial improvement of the optical and structural properties of the ZnO seed layer.
The PL spectra of the nanorods are shown in the
Supplementary Materials (Figure S3). The PL spectra of other nanorod samples were qualitatively the same, since their optical properties are tightly related to the low growth temperature and the seed layer has a limited impact on the spectra.