ZnO as a Functional Material, a Review
Abstract
1. Introduction
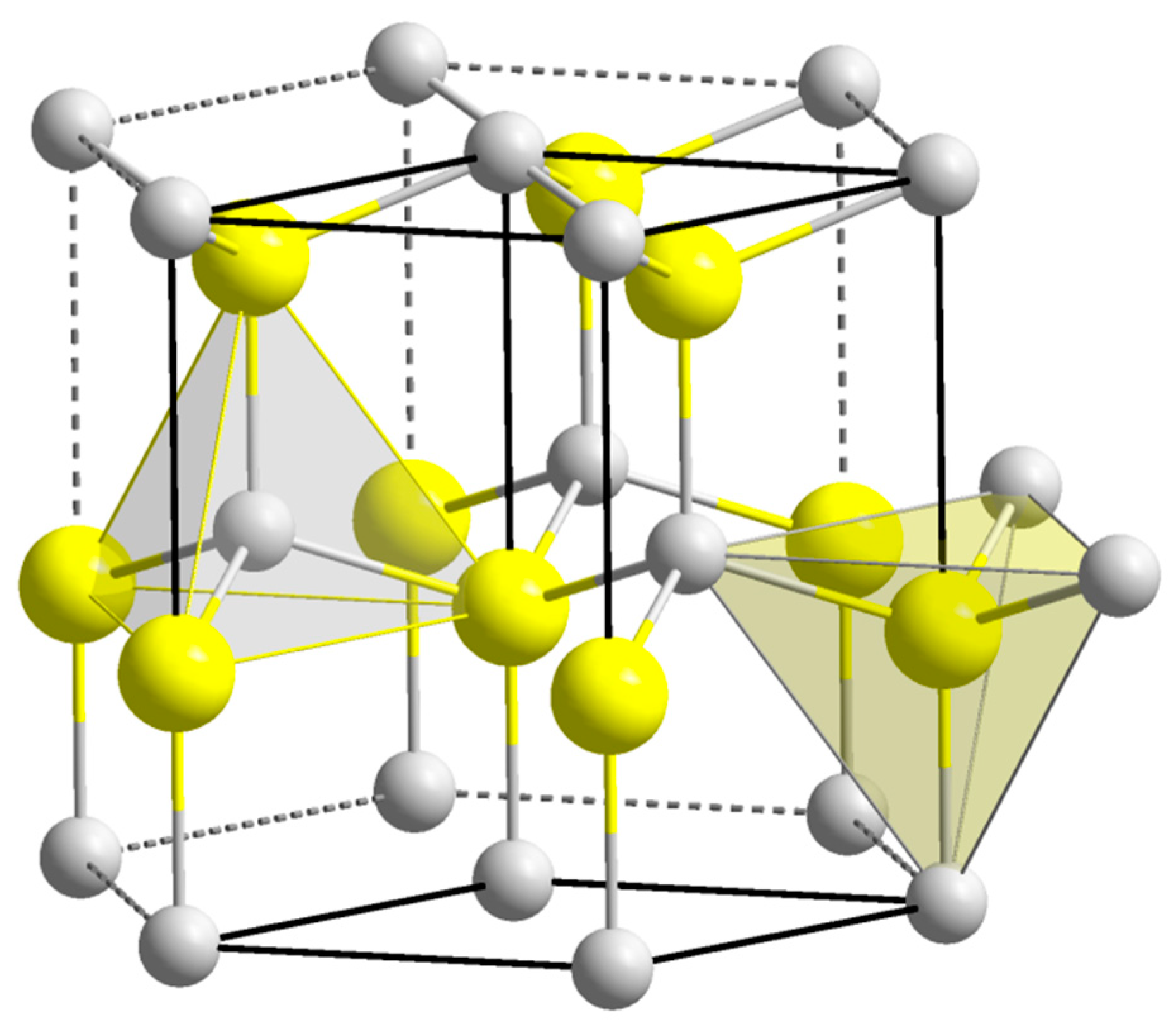
2. General Properties of ZnO
3. Methods of Fabrication
3.1. Bulk Crystals
3.2. Epitaxial Films
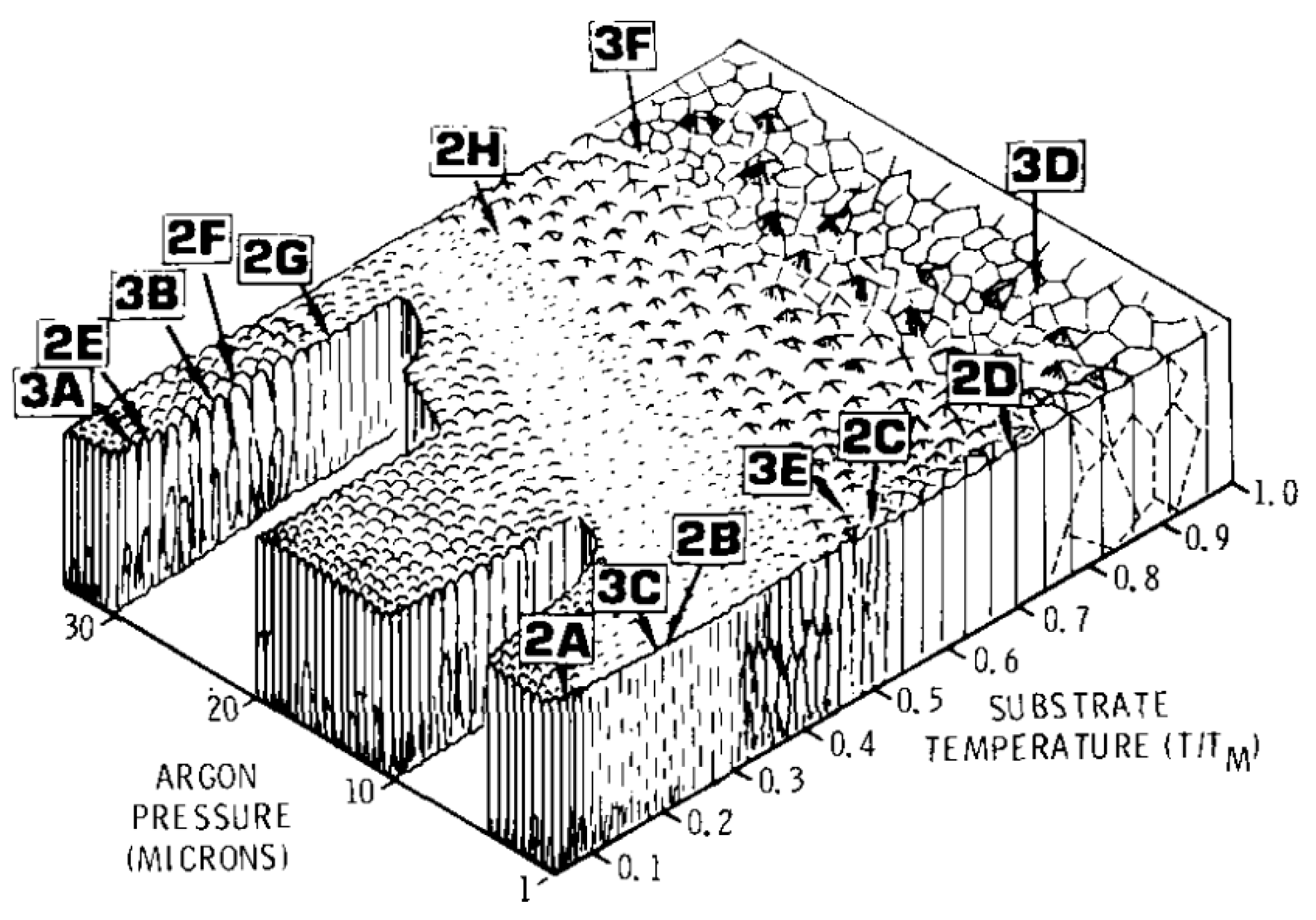
3.3. Thin Films
3.4. Thick Films
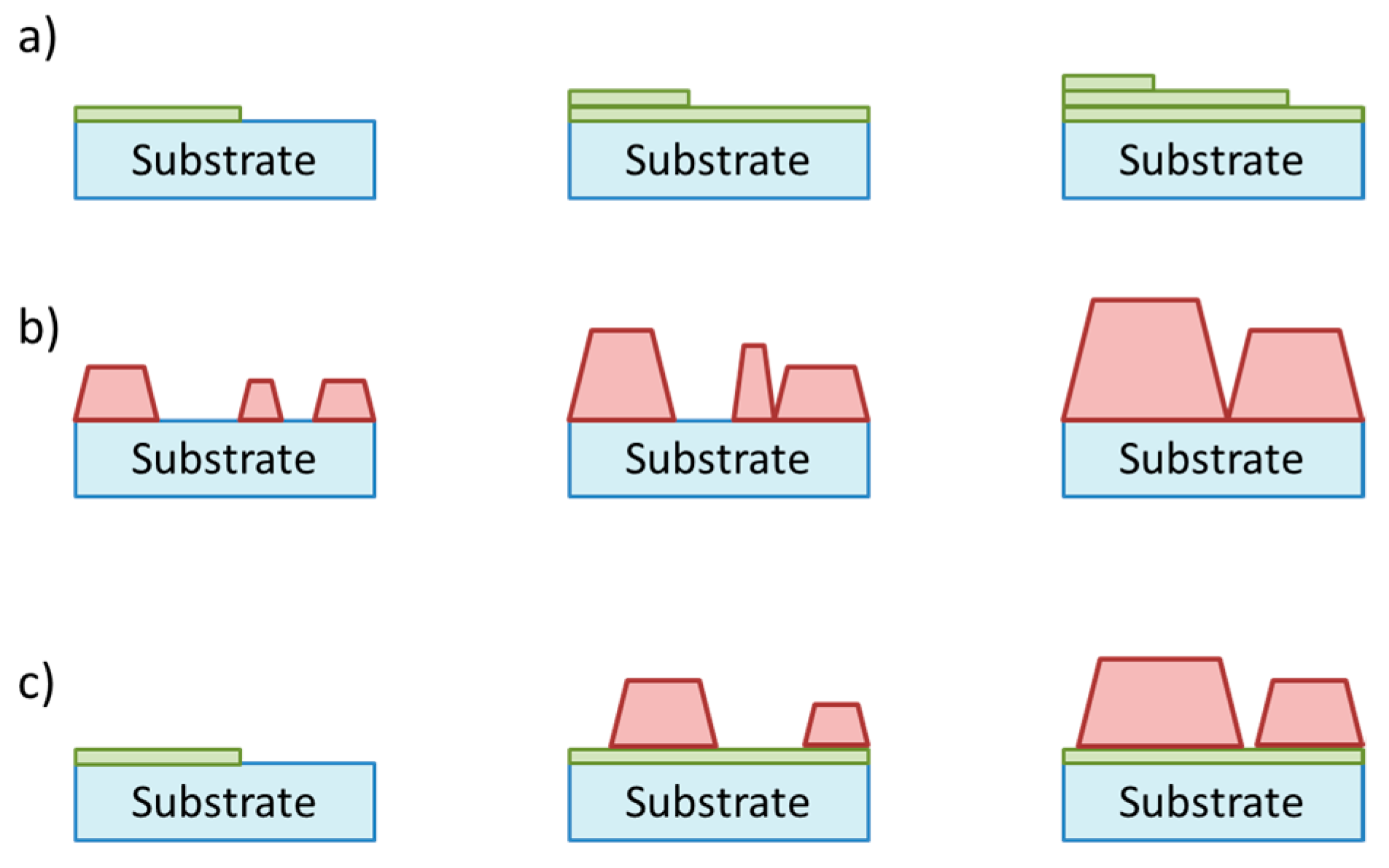
3.5. Nanostructures and Nanoparticles
4. Review of Applications
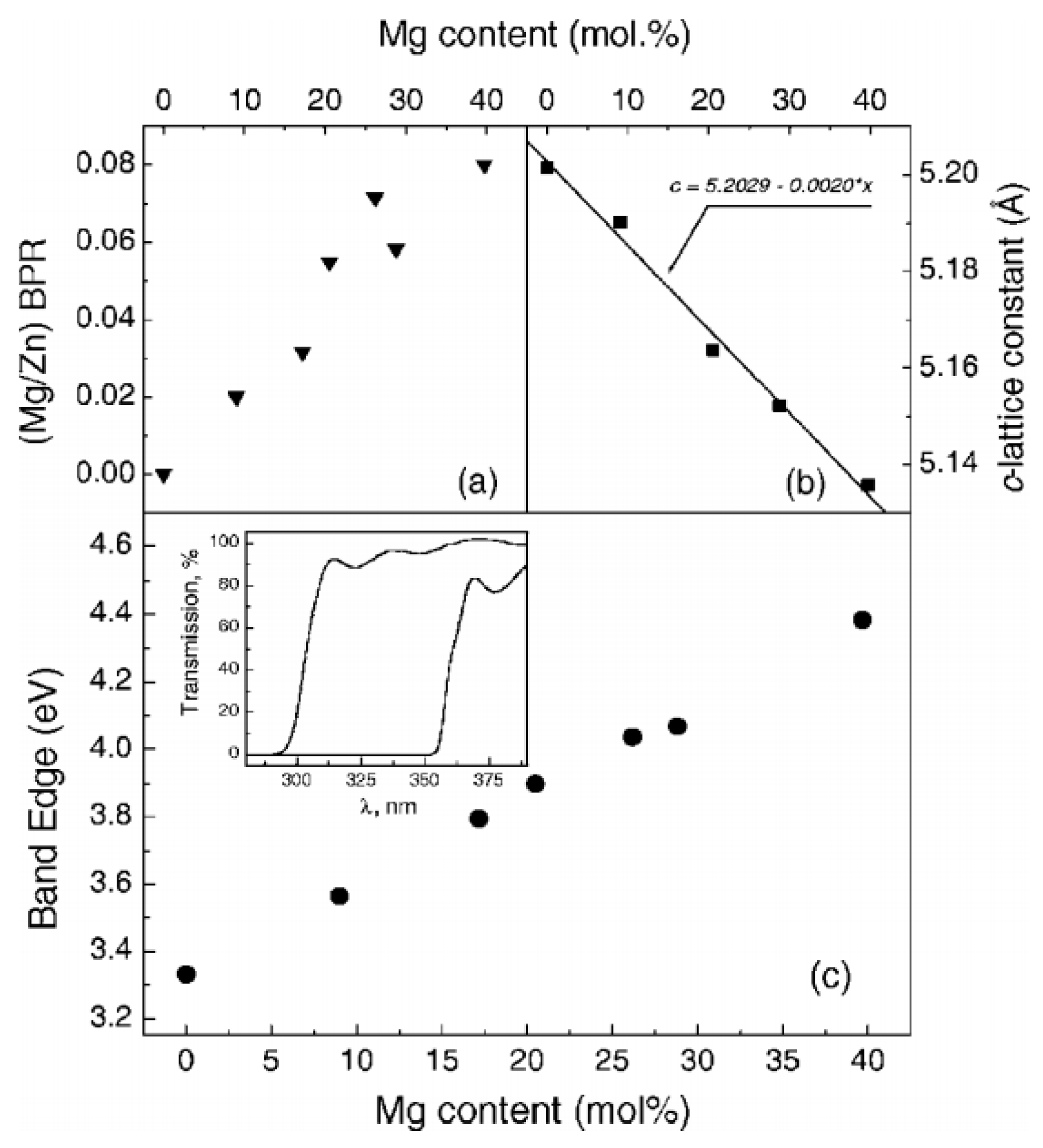
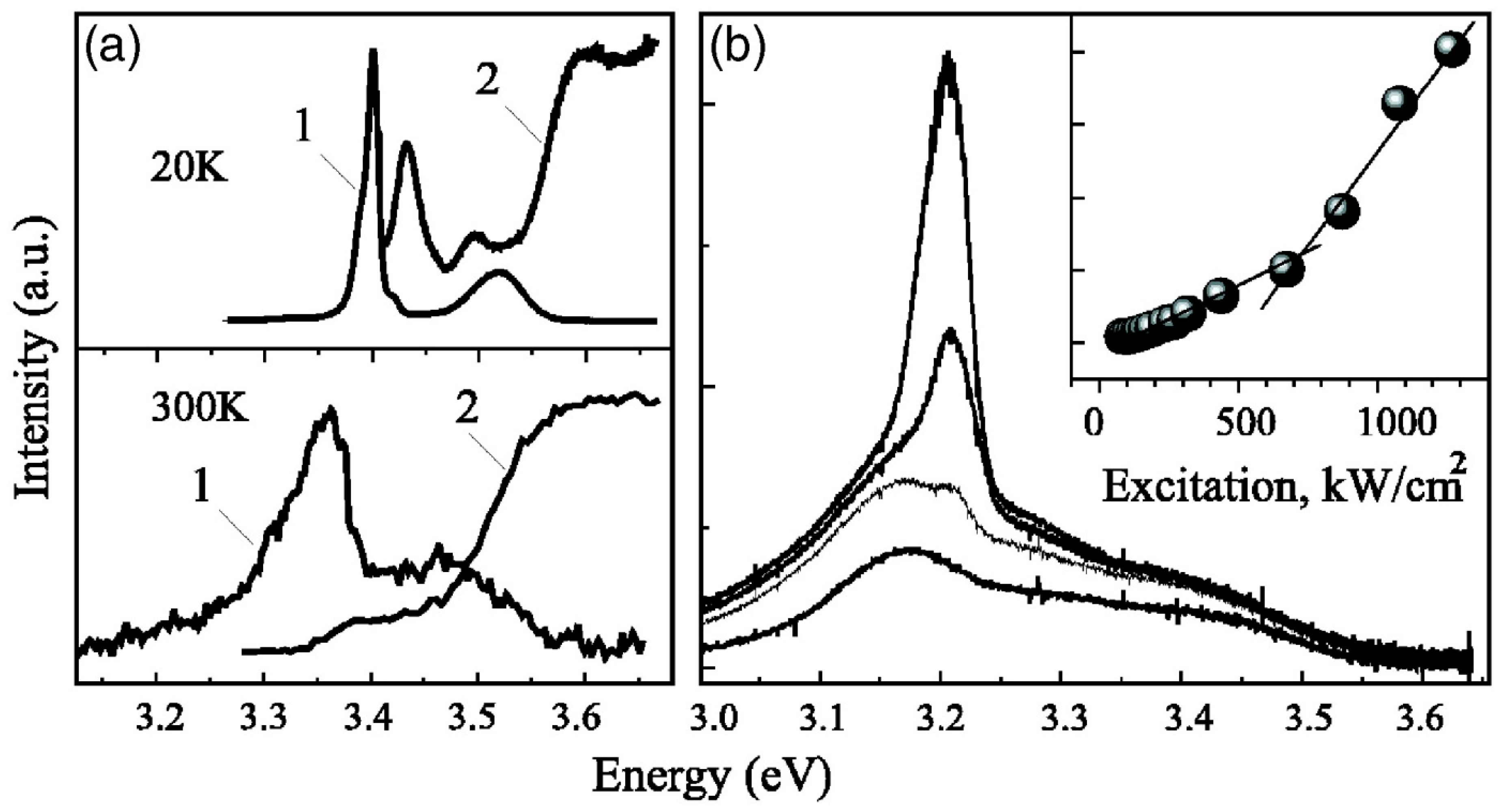
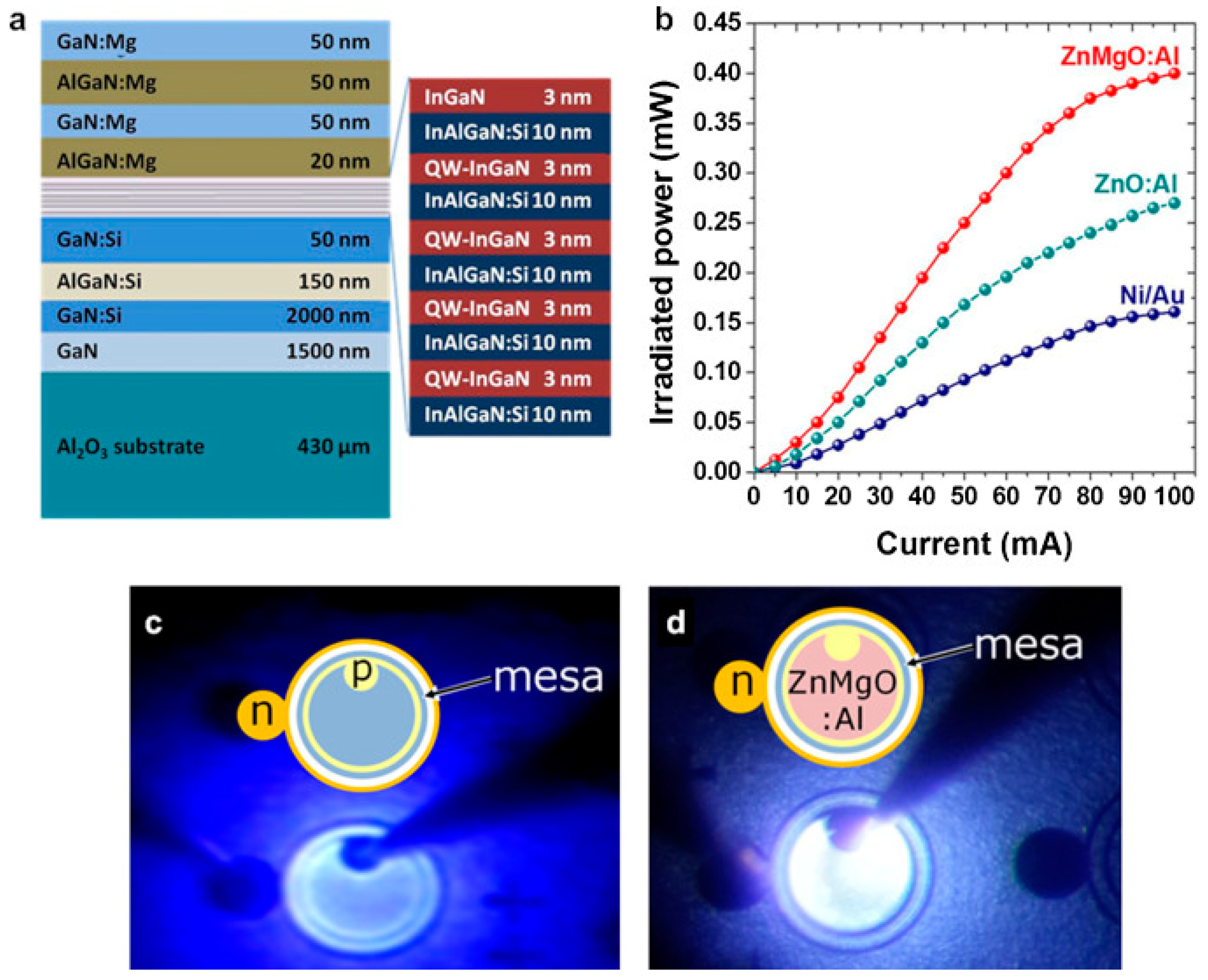
4.1. Epitaxial Structures
4.2. Thin Films
4.3. Thick Films
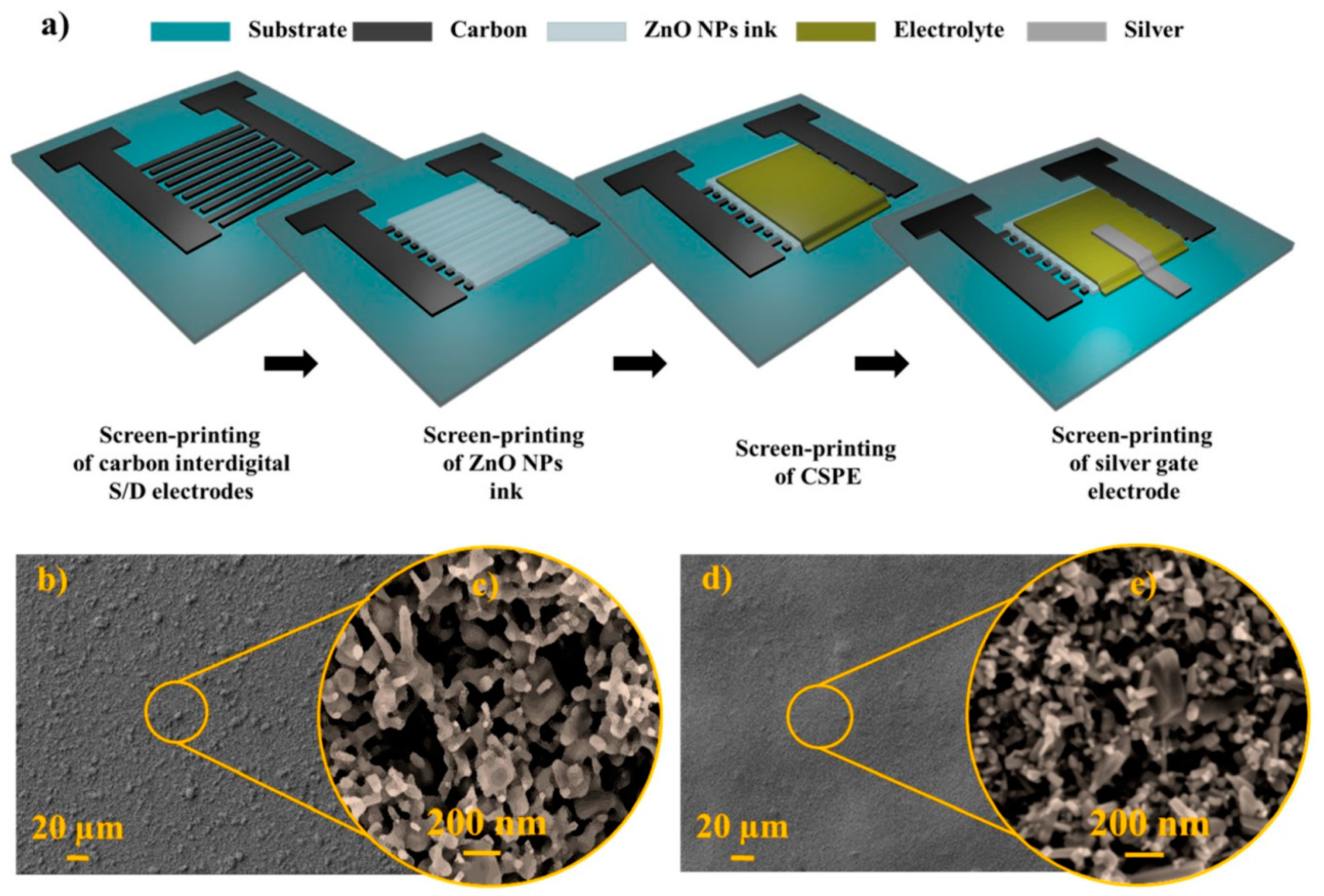
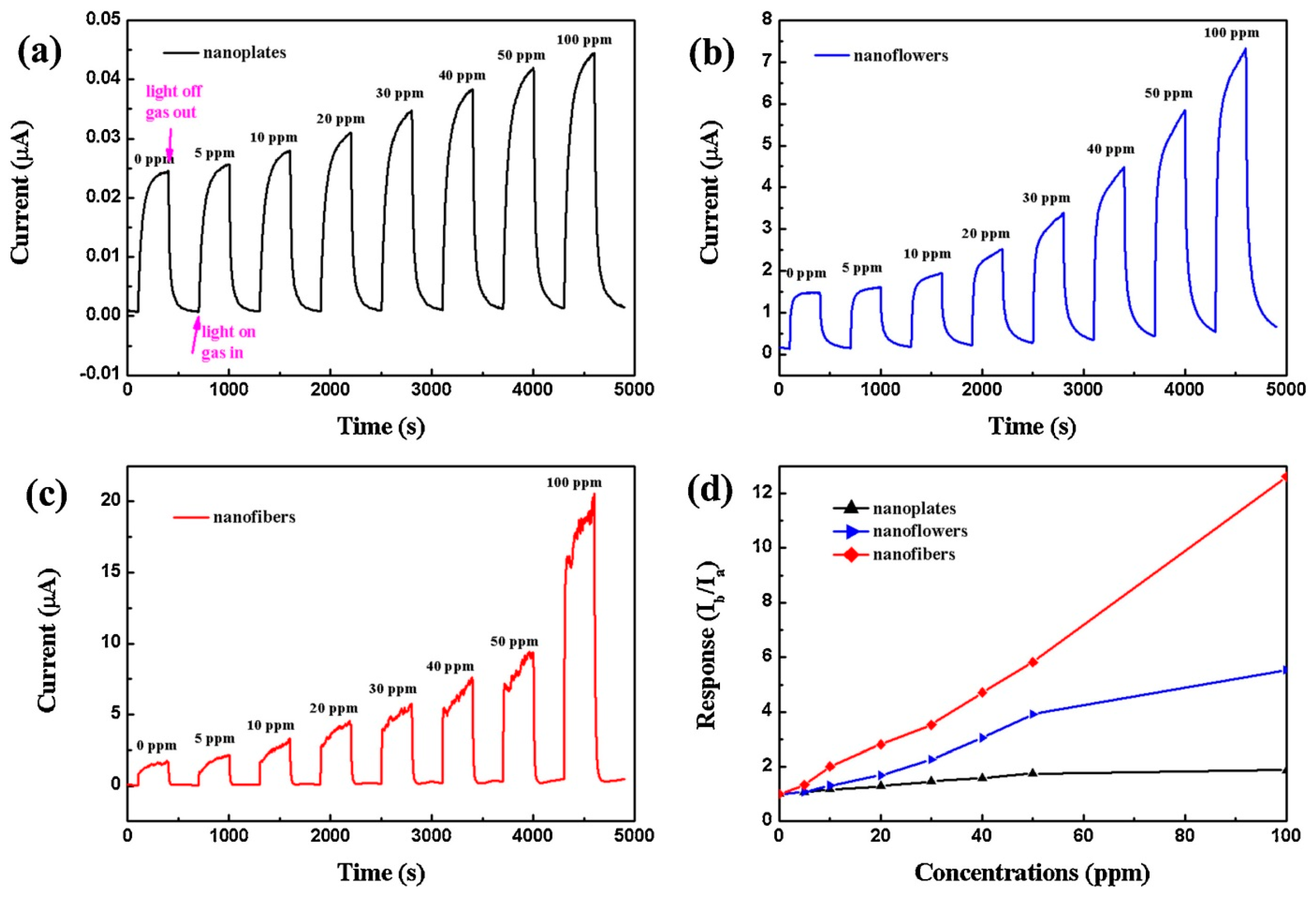
4.4. Nanostructures
5. Unresolved Problems
P-Type Doping Issue in ZnO
6. Summary and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Frederickson, C.J.; Koh, J.-Y.; Bush, A.I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 2005, 6, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Habashi, F. Zinc—The Metal from the East. Bull. Can. Inst. Min. Met. 2001, 94, 71–76. [Google Scholar]
- Biswas, A.K. Rasa–Ratna–Samuccaya and Mineral Processing State–of–Art in the 13th Century AD India. Indian J. Hist. Sci. 1987, 22, 22–46. [Google Scholar]
- Eastaugh, N.; Walsh, V.; Chaplin, T.; Siddall, R. Pigment. Compendium: A Dictionary and Optical Microscopy of Historic Pigments; Elsevier: Burlington, MA, USA, 2008. [Google Scholar]
- Moezzi, A.; McDonagh, A.M.; Cortie, M.B. Zinc oxide particles: Synthesis, properties and applications. Chem. Eng. J. 2012, 185–186, 1–22. [Google Scholar] [CrossRef]
- Brown, H.E. Zinc Oxide Rediscovered; The New Jersey Zinc Company: New York, NY, USA, 1957. [Google Scholar]
- Leong, E.S.P.; Yu, S.F.; Chong, M.K.; Tan, O.K.; Pita, K. Metal-oxide-SiO2 composite ZnO lasers. IEEE Photonics Technol. Lett. 2005, 17, 1815–1817. [Google Scholar] [CrossRef]
- Zhang, X.Q.; Suemune, I.; Kumano, H.; Yao, Z.G.; Huang, S.H. Room temperature ultraviolet lasing action in high-quality ZnO thin films. J. Lumin. 2007, 122, 828–830. [Google Scholar] [CrossRef]
- Chu, S.; Olmedo, M.; Yang, Z.; Kong, J.; Liu, J. Electrically pumped ultraviolet ZnO diode lasers on Si. Appl. Phys. Lett. 2008, 93, 181106. [Google Scholar] [CrossRef]
- Krishnamoorthy, S.; Iliadis, A.A. Properties of High Sensitivity ZnO Surface Acoustic Wave Sensors on SiO2/ (100) Si Substrates. Solid-State Electron. 2008, 52, 1710–1716. [Google Scholar] [CrossRef]
- Kumar, S.; Kim, G.H.; Sreenivas, K.; Tandon, R.P. ZnO based surface acoustic wave ultraviolet photo sensor. J. Electroceram. 2009, 22, 198–202. [Google Scholar] [CrossRef]
- Chivukula, V.; Ciplys, D.; Shur, M.; Dutta, P. ZnO nanoparticle surface acoustic wave UV sensor. Appl. Phys. Lett. 2010, 96, 233512. [Google Scholar] [CrossRef]
- Subramanian, V.; Bakhishev, T.; Redinger, D.; Volkman, S.K. Solution-Processed Zinc Oxide Transistors for Low-Cost Electronics Applications. J. Disp. Technol. 2009, 5, 525–530. [Google Scholar] [CrossRef]
- Dietl, T.; Ohno, H.; Matsukura, F.; Cibert, J.; Ferrand, D. Zener Model Description of Ferromagnetism in Zinc-Blende Magnetic Semiconductors. Science 2000, 287, 1019. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yun, F.; Morkoc, H. Ferromagnetism of ZnO and GaN: A Review. J. Mater. Sci. Mater. Electron. 2005, 16, 555–597. [Google Scholar] [CrossRef]
- Wang, H.T.; Kang, B.S.; Ren, F.; Tien, L.C.; Sadik, P.W.; Norton, D.P.; Pearton, S.J.; Lin, J. Hydrogen-selective sensing at room temperature with ZnO nanorods. Appl. Phys. Lett. 2005, 86, 243503. [Google Scholar] [CrossRef]
- Sadek, A.Z.; Choopun, S.; Wlodarski, W.; Ippolito, S.J.; Kalantar-zadeh, K. Characterization of ZnO Nanobelt-Based Gas Sensor for H2, NO2, and Hydrocarbon Sensing. IEEE Sens. J. 2007, 7, 919–924. [Google Scholar] [CrossRef]
- Phan, D.T.; Chung, G.S. Surface acoustic wave hydrogen sensors based on ZnO nanoparticles incorporated with a Pt catalyst. Sens. Actuators B Chem. 2012, 161, 341–348. [Google Scholar] [CrossRef]
- Han, J.; Fan, F.; Xu, C.; Lin, S.; Wei, M.; Duan, X.; Wang, Z.L. ZnO nanotube-based dye-sensitized solar cell and its application in self-powered devices. Nanotechnology 2010, 21, 405203. [Google Scholar] [CrossRef] [PubMed]
- Martinson, A.B.F.; Elam, J.W.; Hupp, J.T.; Pellin, M.J. ZnO Nanotube Based Dye-Sensitized Solar Cells. Nano Lett. 2007, 7, 2183–2187. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Soltanian, S.; Gholamkhass, B.; Aljaafari, A.; Servati, P. Light-soaking free organic photovoltaic devices with sol–gel deposited ZnO and AZO electron transport layers. RSC Adv. 2018, 8, 36542–36548. [Google Scholar] [CrossRef]
- Morkoç, H.; Özgür, Ü. Zinc Oxide: Fundamentals, Materials and Device Technology, 1st ed.; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009. [Google Scholar]
- Thomas, D.G. The exciton spectrum of zinc oxide. J. Phys. Chem. Solids 1960, 15, 86–96. [Google Scholar] [CrossRef]
- Vigué, F.; Vennéguès, P.; Deparis, C.; Vézian, S.; Laügt, M.; Faurie, J.P. Growth modes and microstructures of ZnO layers deposited by plasma-assisted molecular-beam epitaxy on (0001) sapphire. J. Appl. Phys. 2001, 90, 5115. [Google Scholar] [CrossRef]
- Tsukazaki, A.; Ohtomo, A.; Kita, T.; Ohno, Y.; Ohno, H.; Kawasaki, M. Quantum Hall Effect in Polar Oxide Heterostructures. Science 2007, 315, 1388–1391. [Google Scholar] [CrossRef] [PubMed]
- Tampo, H.; Shibata, H.; Maejima, K.; Yamada, A.; Matsubara, K.; Fons, P.; Kashiwaya, S.; Niki, S.; Chiba, Y.; Wakamatsu, T.; et al. Polarization-induced two-dimensional electron gases in ZnMgO/ZnO heterostructures. Appl. Phys. Lett. 2008, 93, 202104. [Google Scholar] [CrossRef]
- Hwang, H.Y.; Iwasa, Y.; Kawasaki, M.; Keimer, B.; Nagaosa, N.; Tokura, Y. Emergent phenomena at oxide interfaces. Nat. Mater. 2012, 11, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.C.; Wu, C.Y.; Cheng, H.M.; Hsieh, W.F. Band gap engineering and stimulated emission of ZnMgO nanowires. Appl. Phys. Lett. 2006, 89, 013101. [Google Scholar] [CrossRef]
- Ohtomo, A.; Tamura, K.; Kawasaki, M.; Makino, T.; Segawa, Y.; Tang, Z.K.; Wong, L.G.K.; Matsumoto, Y.; Koinuma, H. Room-temperature stimulated emission of excitons in ZnO/(Mg, Zn)O superlattices. Appl. Phys. Lett. 2000, 77, 2204. [Google Scholar] [CrossRef]
- Gruber, T.; Kirchner, C.; Kling, R.; Reuss, F.; Waag, A.; Bertram, F.; Forster, D.; Christen, J.; Schreck, M. Optical and structural analysis of ZnCdO layers grown by metalorganic vapor-phase epitaxy. Appl. Phys. Lett. 2003, 83, 3290. [Google Scholar] [CrossRef]
- Li, S.M.; Kwon, B.J.; Kwack, H.S.; Jin, L.H.; Cho, Y.H.; Park, Y.S.; Han, M.S.; Park, Y.S. Optical transition dynamics in ZnO/ZnMgO multiple quantum well structures with different well widths grown on ZnO substrates. J. Appl. Phys. 2010, 107, 033513. [Google Scholar] [CrossRef]
- Sadofev, S.; Blumstengel, S.; Cui, J.; Puls, J.; Rogaschewski, S.; Schafer, P.; Sadofyev, Y.G.; Henneberger, F. Growth of high-quality ZnMgO epilayers and ZnO/ZnMgO quantum well structures by radical-source molecular-beam epitaxy on sapphire. Appl. Phys. Lett. 2005, 87, 091903. [Google Scholar] [CrossRef]
- Available online: https://commons.wikimedia.org/wiki/File:Wurtzite_polyhedra.png (accessed on 17 August 2019).
- Look, D.C.; Hemsky, J.W.; Sizelove, J.R. Residual Native Shallow Donor in ZnO. Phys. Rev. Lett. 1999, 82, 2552. [Google Scholar] [CrossRef]
- Look, D.C. Recent advances in ZnO materials and devices. Mater. Sci. Eng. B 2001, 80, 383–387. [Google Scholar] [CrossRef]
- Lany, S.; Zunger, A. Anion vacancies as a source of persistent photoconductivity in II-VI and chalcopyrite semiconductors. Phys. Rev. B 2005, 72, 035215. [Google Scholar] [CrossRef]
- Lany, S.; Zunger, A. Dopability, Intrinsic Conductivity, and Nonstoichiometry of Transparent Conducting Oxides. Phys. Rev. Lett. 2007, 98, 045501. [Google Scholar] [CrossRef] [PubMed]
- Janotti, A.; Van de Walle, C.G. New insights into the role of native point defects in ZnO. J. Cryst. Growth 2006, 287, 58–65. [Google Scholar] [CrossRef]
- Janotti, A.; Van de Walle, C.G. Native point defects in ZnO. Phys. Rev. B 2007, 76, 165202. [Google Scholar] [CrossRef]
- Cuscó, R.; Alarcón-Lladó, E.; Ibáñez, J.; Artús, L.; Jiménez, J.; Wang, B.; Callahan, M.J. Temperature dependence of Raman scattering in ZnO. Phys. Rev. B 2007, 75, 165202. [Google Scholar] [CrossRef]
- Kohan, A.F.; Ceder, G.; Morgan, D.; Van de Walle, C.G. First-principles study of native point defects in ZnO. Phys. Rev. B 2000, 61, 15019. [Google Scholar] [CrossRef]
- Hu, J.; Gordon, R.G. Textured aluminum-doped zinc oxide thin films from atmospheric pressure chemical-vapor deposition. J. Appl. Phys. 1992, 71, 880. [Google Scholar] [CrossRef]
- Ko, H.J.; Chen, Y.F.; Hong, S.K.; Wenisch, H.; Yao, T.; Look, D.C. Ga-doped ZnO films grown on GaN templates by plasma-assisted molecular-beam epitaxy. Appl. Phys. Lett. 2000, 77, 3761. [Google Scholar] [CrossRef]
- Hu, J.; Gordon, R.G. Electrical and Optical Properties of Indium Doped Zinc Oxide Films Prepared by Atmospheric Pressure Chemical Vapor Deposition. Mater. Res. Soc. Symp. Proc. 1993, 283, 891. [Google Scholar] [CrossRef]
- Wenas, W.W.; Yamada, A.; Takahashi, K.; Yoshino, M.; Konagai, M. Electrical and optical properties of boron-doped ZnO thin films for solar cells grown by metalorganic chemical vapor deposition. J. Appl. Phys. 1991, 70, 7119. [Google Scholar] [CrossRef]
- Meyer, J.; Görrn, P.; Hamwi, S.; Johannes, H.H.; Riedl, T.; Kowalsky, W. Indium-free transparent organic light emitting diodes with Al doped ZnO electrodes grown by atomic layer and pulsed laser deposition. Appl. Phys. Lett. 2008, 93, 073308. [Google Scholar] [CrossRef]
- Zhang, S.B.; Wei, S.H.; Zunger, A. Intrinsic n-type versus p-type doping asymmetry and the defect physics of ZnO. Phys. Rev. B 2001, 63, 075205. [Google Scholar] [CrossRef]
- Hofmann, D.M.; Hofstaetter, A.; Leiter, F.; Zhou, H.; Henecker, F.; Meyer, B.K.; Orlinskii, S.B.; Schmidt, J.; Baranov, P.G. Hydrogen: A Relevant Shallow Donor in Zinc Oxide. Phys. Rev. Lett. 2002, 88, 045504. [Google Scholar] [CrossRef] [PubMed]
- Pankove, J.I.; Johnson, N.M. Hydrogen in Semiconductors, Semiconductors and Semimetals; Academic Press: Boston, MA, USA, 1991; Volume 34. [Google Scholar]
- Van de Walle, C.G. Hydrogen as a Cause of Doping in Zinc Oxide. Phys. Rev. Lett. 2000, 85, 1012–1015. [Google Scholar] [CrossRef] [PubMed]
- Nemanič, V.; Bogataj, T. Outgassing of thin wall stainless steel chamber. Vacuum 1998, 50, 431–437. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Nemanič, V. An overview of methods to suppress hydrogen outgassing rate from austenitic stainless steel with reference to UHV and EXV. Vacuum 2003, 69, 501–512. [Google Scholar] [CrossRef]
- Čižek, J.; Valenta, J.; Hruška, P.; Melikhova, O.; Procházka, I.; Novotný, M.; Bulíř, J. Origin of green luminescence in hydrothermally grown ZnO single crystals. Appl. Phys. Lett. 2015, 106, 251902. [Google Scholar] [CrossRef]
- Doherty, R.P.; Sun, Y.; Sun, Y.; Warren, J.L.; Fox, N.A.; Cherns, D.; Ashfold, M.N.R. Growth of nanostructured ZnO thin films on sapphire. Appl. Phys. A 2007, 89, 49–55. [Google Scholar] [CrossRef]
- Vayssieres, L. Growth of Arrayed Nanorods and Nanowires of ZnO from Aqueous Solutions. Adv. Mater. 2003, 15, 464–466. [Google Scholar] [CrossRef]
- Kuo, C.L.; Kuo, T.J.; Huang, M.H. Hydrothermal synthesis of ZnO microspheres and hexagonal microrods with sheetlike and platelike nanostructures. J. Phys. Chem. B 2005, 109, 20115–20121. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhu, R.; Liu, X.; Liu, B.; Ramakrishna, S. Facile construction of nanofibrous ZnO photoelectrode for dye-sensitized solar cell applications. Appl. Phys. Lett. 2009, 95, 043304. [Google Scholar] [CrossRef]
- Chen, C.; Lu, Y.; He, H.; Wu, K.; Ye, Z. One-step synthesis of flower-like Au-ZnO microstructures at room temperature and their photocatalytic properties. Appl. Phys. A 2013, 110, 47–53. [Google Scholar] [CrossRef]
- Brayner, R.; Dahoumane, S.A.; Yéprémian, C.; Djediat, C.; Meyer, M.; Couté, A.; Fiévet, F. ZnO Nanoparticles: Synthesis, Characterization, and Ecotoxicological Studies. Langmuir 2010, 26, 6522–6528. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, K.; Mishima, K.; Kato, T.; Irie, K.; Mishima, K. Transparent polymeric hybrid film of ZnO nanoparticle quantum dots and PMMA with high luminescence and tunable emission color. J. Colloid Interface Sci. 2012, 367, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, R.; Chen, Z.; Liu, G.; Zhang, Z.; Jiao, Z. Self-assembly fabrication of 3D flower-like ZnO hierarchical nanostructures and their gas sensing properties. CrystEngComm 2012, 14, 1775–1782. [Google Scholar] [CrossRef]
- Ko, S.H.; Lee, D.; Kang, H.W.; Nam, K.H.; Yeo, J.Y.; Hong, S.J.; Grigoropoulos, C.P.; Sung, H.J. Nanoforest of Hydrothermally Grown Hierarchical ZnO Nanowires for a High Efficiency Dye-Sensitized Solar Cell. Nano Lett. 2011, 11, 666–671. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.L. Novel nanostructures of ZnO for nanoscale photonics, optoelectronics, piezoelectricity, and sensing. Appl. Phys. A 2007, 88, 7–15. [Google Scholar] [CrossRef]
- Li, L.; Pan, S.; Dou, X.; Zhu, Y.; Huang, X.; Yang, Y.; Li, G.; Zhang, L. Direct Electrodeposition of ZnO Nanotube Arrays in Anodic Alumina Membranes. J. Phys. Chem. C 2007, 111, 7288–7291. [Google Scholar] [CrossRef]
- Hu, F.; Chan, K.C.; Yue, T.M.; Surya, C. Dynamic Template Assisted Electrodeposition of Porous ZnO Thin Films Using a Triangular Potential Waveform. J. Phys. Chem. C 2010, 114, 5811–5816. [Google Scholar] [CrossRef]
- Yano, M.; Koike, K.; Ogata, K.; Nogami, T.; Tanabe, S.; Sasa, S. Zinc oxide-based biosensors. Phys. Status Solidi C 2012, 9, 1570–1573. [Google Scholar] [CrossRef]
- Borysiewicz, M.A.; Dynowska, E.; Kolkovsky, V.; Dyczewski, J.; Wielgus, M.; Kamińska, E.; Piotrowska, A. From porous to dense thin ZnO films through reactive DC sputter deposition onto Si (100) substrates. Phys. Status Solidi A 2012, 209, 2463–2469. [Google Scholar] [CrossRef]
- Ohshima, E.; Ogino, H.; Niikura, I.; Maeda, K.; Sato, M.; Ito, M.; Fukuda, T. Growth of the 2-in-size bulk ZnO single crystals by the hydrothermal method. J. Cryst. Growth 2004, 260, 166–170. [Google Scholar] [CrossRef]
- Hirose, M. Growth of Zinc Oxide Single Crystals by Vapor Phase Reaction. Jpn. J. Appl. Phys. 1971, 10, 401. [Google Scholar] [CrossRef]
- Wang, B.; Claflin, B.; Callahan, M.; Fang, Z.Q.; Look, D. Electrical and optical properties of ZnO bulk crystals with and without lithium grown by the hydrothermal technique. Proc. SPIE 2014, 8987, 89871D. [Google Scholar] [CrossRef]
- Johnson, M.A.L.; Fujita, S.; Rowland, W.H.; Hughes, W.C.; Cook, J.W.; Schetzina, J.F. MBE growth and properties of ZnO on sapphire and SiC substrates. J. Electron. Mater. 1996, 25, 855–862. [Google Scholar] [CrossRef]
- Cho, M.Y.; Kim, M.S.; Choi, H.Y.; Jeon, S.M.; Kim, G.S.; Kim, D.Y.; Yim, K.G.; Leem, J.-Y. Effects of Growth Interruption of ZnO Buffer Layers on the Structural and the Optical Properties of ZnO Thin Films Grown by Using PA-MBE. J. Korean Phys. Soc. 2010, 56, 1833–1837. [Google Scholar] [CrossRef]
- Pietrzyk, M.A.; Zielony, E.; Stachowicz, M.; Reszka, A.; Płaczek-Popko, E.; Wierzbicka, A.; Przezdziecka, E.; Droba, A.; Kozanecki, A. Electro-optical characterization of ZnO/ZnMgO structure grown on p-type Si (1 1 1) by PA-MBE method. J. Alloys Compd. 2014, 587, 724–728. [Google Scholar] [CrossRef]
- Kuznetsov, P.; Lusanov, V.; Yakushcheva, G.; Jitov, V.; Zakharov, L.; Kotelyanskii, I.; Kozlovsky, V. MOVPE growth and study of ZnO, ZnMgO epilayers and ZnO/ZnMgO MQW structures. IOP Conf. Ser. Mater. Sci. Eng. 2010, 8, 012040. [Google Scholar] [CrossRef]
- Kaidashev, E.M.; Lorenz, M.; von Wenckstern, H.; Rahm, A.; Semmelhack, H.-C.; Han, K.-H.; Benndorf, G.; Bundesmann, C.; Hochmuth, H.; Grundmann, M. High electron mobility of epitaxial ZnO thin films on c-plane sapphire grown by multistep pulsed-laser deposition. Appl. Phys. Lett. 2003, 82, 3901. [Google Scholar] [CrossRef]
- Nahnas, A.; Kim, H.K. Epitaxial growth of ZnO films on Si substrates using an epitaxial GaN buffer. Appl. Phys. Lett. 2001, 78, 1511. [Google Scholar] [CrossRef]
- Jin, B.J.; Im, S.; Lee, S.Y. Violet and UV luminescence emitted from ZnO thin films grown on sapphire by pulsed laser deposition. Thin Solid Films 2000, 366, 107–110. [Google Scholar] [CrossRef]
- Alfaro Cruz, M.R.; Ceballos-Sanchez, O.; Luévano-Hipólito, E.; Torres-Martínez, L.M. ZnO thin films deposited by RF magnetron sputtering: Effects of the annealing and atmosphere conditions on the photocatalytic hydrogen production. Int. J. Hydrogen Energy 2018, 43, 10301–10310. [Google Scholar] [CrossRef]
- Borysiewicz, M.A.; Pasternak, I.; Dynowska, E.; Jakieła, R.; Kolkovki, V.; Dużyńska, A.; Kamińska, E.; Piotrowska, A. ZnO Thin Films Deposited on Sapphire by High Vacuum High Temperature Sputtering. Acta Phys. Pol. A 2011, 119, 686–688. [Google Scholar] [CrossRef]
- Särkijärvi, S.; Sintonen, S.; Tuomisto, F.; Bosund, M.; Suihkonen, S.; Lipsanen, H. Effect of growth temperature on the epitaxial growth of ZnO on GaN by ALD. J. Cryst. Growth 2014, 398, 18–22. [Google Scholar] [CrossRef]
- Guziewicz, E.; Kowalik, I.A.; Godlewski, M.; Kopalko, K.; Osinniy, V.; Wójcik, A.; Yatsunenko, S.; Łusakowska, E.; Paszkowicz, W.; Guziewicz, M. Extremely low temperature growth of ZnO by atomic layer deposition. J. Appl. Phys. 2008, 103, 033515. [Google Scholar] [CrossRef]
- Minami, T.; Nanoto, H.; Takata, S. Highly Conductive and Transparent Aluminum Doped Zinc Oxide Thin Films Prepared by RF Magnetron Sputtering. Jpn. J. Appl. Phys. 1984, 23, L280. [Google Scholar] [CrossRef]
- Lim, S.J.; Kwon, S.-J.; Kim, H.; Park, J.-S. High performance thin film transistor with low temperature atomic layer deposition nitrogen-doped ZnO. Appl. Phys. Lett. 2008, 91, 183517. [Google Scholar] [CrossRef]
- Kamalasanan, M.N.; Chandra, S. Sol-gel synthesis of ZnO thin films. Thin Solid Films 1996, 288, 112–115. [Google Scholar] [CrossRef]
- Bao, D.; Gu, H.; Kuang, A. Sol–gel-derived c-axis oriented ZnO thin films. Thin Solid Films 1998, 312, 37–39. [Google Scholar] [CrossRef]
- Studenikin, S.A.; Golego, N.; Cocivera, M. Fabrication of green and orange photoluminescent, undoped ZnO films using spray pyrolysis. J. Appl. Phys. 1998, 84, 2287–2294. [Google Scholar] [CrossRef]
- Wu, Y.; Girgis, E.; Ström, V.; Voit, W.; Belova, L.; Rao, K.V. Ultraviolet light sensitive In-doped ZnO thin film field effect transistor printed by inkjet technique. Phys. Status Solidi A 2010, 208, 206–209. [Google Scholar] [CrossRef]
- Manzano, C.V.; Alegre, D.; Caballero-Calero, O.; Alén, B.; Martín-González, M.S. Synthesis and luminescence properties of electrodeposited ZnO films. J. Appl. Phys. 2011, 110, 043538. [Google Scholar] [CrossRef]
- Azeez, O.A.; Sabry, R.S.; Hassan, M.A.M.; Madlul, S.F. Synthesis and characteristics of screen printed ZnO thick films nanostructures grown using different methods. J. Mater. Sci. Mater. Electron. 2015, 26, 4051–4061. [Google Scholar] [CrossRef]
- Ge, C.; Xie, C.; Cai, S. Preparation and gas-sensing properties of Ce-doped ZnO thin-film sensors by dip-coating. Mater. Sci. Eng. B 2007, 137, 53–58. [Google Scholar] [CrossRef]
- Saleh, S.M. ZnO nanospheres based simple hydrothermal route for photocatalytic degradation of azo dye. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 211, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Sahu, K.; Kuriakose, S.; Singh, J.; Satpati, B.; Mohapatra, S. Facile synthesis of ZnO nanoplates and nanoparticle aggregates for highly efficient photocatalytic degradation of organic dyes. J. Phys. Chem. Solids 2018, 121, 186–195. [Google Scholar] [CrossRef]
- Pimentel, A.; Henriques Ferreira, S.; Nunes, D.; Calmeiro, T.; Martins, R.; Fortunato, E. Microwave Synthesized ZnO Nanorod Arrays for UV Sensors: A Seed Layer Annealing Temperature Study. Materials 2016, 9, 299. [Google Scholar] [CrossRef]
- Samadipakchin, P.; Mortaheb, H.R.; Zolfaghari, A. ZnO nanotubes: Preparation and photocatalytic performance evaluation. J. Photochem. Photobiol. A Chem. 2017, 337, 91–99. [Google Scholar] [CrossRef]
- Li, Y.B.; Bando, Y.; Golberg, D. ZnO nanoneedles with tip surface perturbations: Excellent field emitters. Appl. Phys. Lett. 2004, 84, 3603. [Google Scholar] [CrossRef]
- All Abbas, J.M.; Narin, P.; Kutlu, E.; Lisesivdin, S.B.; Ozbay, E. Electronic properties of zigzag ZnO nanoribbons with hydrogen and magnesium passivations. Phys. B Condens. Matter 2019, 556, 12–16. [Google Scholar] [CrossRef]
- Shi, Y.; Bao, S.; Shi, R.; Huang, C.; Amini, A.; Wu, Z.; Zhang, L.; Wang, N.; Cheng, C. Y-shaped ZnO Nanobelts Driven from Twinned Dislocations. Sci. Rep. 2016, 6, 22494. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhou, Y.Z.; Wu, D.Y.; Liu, X.H.; Zhang, R.; Liu, H.; Dong, C.K.; Yang, J.; Kulinich, S.A.; Du, X.W. ZnO nanosheets with atomically thin ZnS overlayers for photocatalytic water splitting. J. Mater. Chem. A 2018, 6, 9057–9063. [Google Scholar] [CrossRef]
- Mahendra, R.; Arianti, M.; Sawitri, D.; Risanti, D.D. Synthesis of Various ZnO Nanotree Morphologies through PEG-Assisted Co-Precipitation Method. Adv. Mater. Res. 2015, 1112, 66–70. [Google Scholar] [CrossRef]
- Zhuo, R.F.; Feng, H.T.; Chen, J.T.; Yan, D.; Feng, J.J.; Li, H.J.; Geng, B.S.; Cheng, S.; Xu, X.Y.; Yan, P.X. Multistep Synthesis, Growth Mechanism, Optical, and Microwave Absorption Properties of ZnO Dendritic Nanostructures. J. Phys. Chem. C 2008, 112, 11767–11775. [Google Scholar] [CrossRef]
- Ren, D.; Li, J.; Bao, Y.; Wu, Z.; He, S.; Wang, A.; Guo, F.; Chen, Y. Low-temperature synthesis of flower-like ZnO microstructures supported on TiO2 thin films as efficient antifungal coatings for bamboo protection under dark conditions. Colloids Surf. A Physicochem. Eng. Asp. 2018, 555, 381–388. [Google Scholar] [CrossRef]
- Leung, Y.H.; Tam, K.H.; Djurišić, A.B.; Xie, M.H.; Chan, W.K.; Lu, D.; Ge, W.K. ZnO nanoshells: Synthesis, structure, and optical properties. J. Cryst. Growth 2005, 283, 134–140. [Google Scholar] [CrossRef]
- Borysiewicz, M.A.; Wzorek, M.; Wojciechowski, T.; Wojtowicz, T.; Kamińska, E.; Piotrowska, A. Photoluminescence of nanocoral ZnO films. J. Lumin. 2014, 147, 367–371. [Google Scholar] [CrossRef]
- Tong, Y.; Liu, Y.; Dong, L.; Zhao, D.; Zhang, J.; Lu, Y.; Shen, D.; Fan, X. Growth of ZnO nanostructures with different morphologies by using hydrothermal technique. J. Phys. Chem. B 2006, 110, 20263–20267. [Google Scholar] [CrossRef]
- Li, P.; Wei, Z.; Wu, T.; Peng, Q.; Li, Y. Au−ZnO Hybrid Nanopyramids and Their Photocatalytic Properties. J. Am. Chem. Soc. 2011, 133, 5660–5663. [Google Scholar] [CrossRef]
- Agrawal, J.; Dixit, T.; Palani, I.A.; Rao, M.S.R.; Singh, V. Fabrication of high responsivity deep UV photo-detector based on Na doped ZnO nanocolumns. J. Phys. D Appl. Phys. 2018, 51, 185106. [Google Scholar] [CrossRef]
- Saleema, N.; Farzaneh, M. Thermal effect on superhydrophobic performance of stearic acid modified ZnO nanotowers. Appl. Surf. Sci. 2008, 254, 2690–2695. [Google Scholar] [CrossRef]
- Fan, D.; Zhang, R.; Li, Y. Synthesis and optical properties of phosphorus-doped ZnO nanocombs. Solid State Commun. 2010, 150, 1911–1914. [Google Scholar] [CrossRef]
- Wang, Z.L. Nanostructures of zinc oxide. Mater. Today 2004, 7, 26–33. [Google Scholar] [CrossRef]
- Gao, P.X.; Wang, Z.L. High-Yield Synthesis of Single-Crystal Nanosprings of ZnO. Small 2005, 1, 945–949. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ram, M.K.; Stefanakos, E.K.; Goswami, Y.D. Synthesis, Characterization, and Applications of ZnO Nanowires. J. Nanomater. 2012, 2012, 624520. [Google Scholar] [CrossRef]
- Li, W.; Wu, X.; Han, N.; Chen, J.; Qian, X.; Deng, Y.; Tang, W.; Chen, Y. MOF-derived hierarchical hollow ZnO nanocages with enhanced low-concentration VOCs gas-sensing performance. Sens. Actuators B Chem. 2016, 225, 158–166. [Google Scholar] [CrossRef]
- Warule, S.S.; Chaudhari, N.S.; Ambekar, J.D.; Kale, B.B.; More, M.A. Hierarchical Nanostructured ZnO with Nanorods Engendered to Nanopencils and Pin-Cushion Cactus with Its Field Emission Study. ACS Appl. Mater. Interfaces 2011, 3, 3454–3462. [Google Scholar] [CrossRef] [PubMed]
- Witkowski, B.S.; Wachnicki, Ł.; Gieraltowska, S.; Sybilski, P.; Kopalko, K.; Stachowicz, M.; Godlewski, M. UV detector based on zinc oxide nanorods obtained by the hydrothermal method. Phys. Status Solidi C 2014, 11, 1447–1451. [Google Scholar] [CrossRef]
- Rai, P.; Kwak, W.K.; Yu, Y.T. Solvothermal Synthesis of ZnO Nanostructures and Their Morphology-Dependent Gas-Sensing Properties. ACS Appl. Mater. Interfaces 2013, 5, 3026–3032. [Google Scholar] [CrossRef]
- Wasly, H.S.; El-Sadek, M.S.A.; Henini, M. Influence of reaction time and synthesis temperature on the physical properties of ZnO nanoparticles synthesized by the hydrothermal method. Appl. Phys. A 2018, 124, 76. [Google Scholar] [CrossRef]
- Widiyastuti, W.; Machmudah, S.; Nurtono, T.; Winardi, S.; Balgis, R.; Ogi, T.; Okuyama, K. Morphology and optical properties of zinc oxide nanoparticles synthesized by solvothermal method. Chem. Eng. Trans. 2017, 56, 955–960. [Google Scholar] [CrossRef]
- Martin, O.; González, V.; Tirado, M.; Comedi, D. Effects of methanol on morphology and photoluminescence in solvothermal grown ZnO powders and ZnO on Si. Mater. Lett. 2019, 251, 41–44. [Google Scholar] [CrossRef]
- Ghorbani, H.R.; Mehr, F.P.; Pazoki, H.; Rahmani, B.M. Synthesis of ZnO Nanoparticles by Precipitation Method. Orient J. Chem 2015, 31, 1219–1221. [Google Scholar] [CrossRef]
- Wojnarowicz, J.; Chudoba, T.; Gierlotka, S.; Łojkowski, W. Effect of Microwave Radiation Power on the Size of Aggregates of ZnO NPs Prepared Using Microwave Solvothermal Synthesis. Nanomaterials 2018, 8, 343. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Kumar, R.V.; Gedanken, A.; Zaban, A. Sonochemical synthesis of crystalline nanoporous zinc oxide spheres and their application in dye-sensitized solar cells. Isr. J. Chem. 2001, 41, 51–54. [Google Scholar] [CrossRef]
- Applerot, G.; Perkas, N.; Amirian, G.; Girshevitz, O.; Gedanken, A. Coating of glass with ZnO via ultrasonic irradiation and a study of its antibacterial properties. Appl. Surf. Sci. 2009, 256 (Suppl. 15), S3–S8. [Google Scholar] [CrossRef]
- Di Mauro, A.; Zimbone, M.; Fragalà, M.E.; Impellizzeri, G. Synthesis of ZnO nanofibers by the electrospinning process. Mater. Sci. Semicond. Process. 2016, 42, 98–101. [Google Scholar] [CrossRef]
- Keong, C.C.; Vivek, Y.S.; Salamatinia, B.; Horri, B.A. Green Synthesis of ZnO Nanoparticles by an Alginate Mediated Ion-Exchange Process and a case study for Photocatalysis of Methylene Blue Dye. J. Phys. Conf. Ser. 2017, 829, 012014. [Google Scholar] [CrossRef]
- Ao, W.; Li, J.; Yang, H.; Zeng, X.; Ma, X. Mechanochemical synthesis of zinc oxide nanocrystalline. Powder Technol. 2006, 168, 148–151. [Google Scholar] [CrossRef]
- Zacour Marinho, J.; da Costa Romeiro, F.; Lemos, S.C.S.; Motta, F.V.; Riccardi, C.; Siu Li, M.; Longo, E.; Lima, R.C. Urea-Based Synthesis of Zinc Oxide Nanostructures at Low Temperature. J. Nanomater. 2012, 2012, 427172. [Google Scholar] [CrossRef]
- Zhu, Y.; Apostoluk, A.; Gautier, P.; Valette, A.; Omar, L.; Cornier, T.; Bluet, J.M.; Masenelli-Varlot, K.; Daniele, S.; Masenelli, B. Intense visible emission from ZnO/PAAX (X = H or Na) nanocomposite synthesized via a simple and scalable sol-gel method. Sci. Rep. 2016, 6, 23557. [Google Scholar] [CrossRef] [PubMed]
- Wong, E.M.; Hoertz, P.G.; Liang, C.J.; Shi, B.M.; Meyer, G.J.; Searson, P.C. Influence of Organic Capping Ligands on the Growth Kinetics of ZnO Nanoparticles. Langmuir 2001, 17, 8362–8367. [Google Scholar] [CrossRef]
- Xiong, H.M.; Liu, D.P.; Xia, Y.Y.; Chen, J.S. Polyether-grafted ZnO Nanoparticles with Tunable and Stable Photoluminescence at Room Temperature. Chem. Mater. 2005, 17, 3062–3064. [Google Scholar] [CrossRef]
- Shaikh, T.; Sharma, A.; Kaur, H. Ultrasonication Assisted Synthesis of 3 Substituted Indoles in Water Using Polymer Grafted ZnO Nanoparticles as Eco-Friendly Catalyst. ChemistrySelect 2019, 4, 245–249. [Google Scholar] [CrossRef]
- Hong, R.Y.; Qian, J.Z.; Cao, J.X. Synthesis and Characterization of PMMA grafted ZnO nanoparticles. Powder Technol. 2006, 163, 160–168. [Google Scholar] [CrossRef]
- Abdolrezapour, F.; Moradi, M. High optical quality long ultrafine ZnO nanowires by low-temperature oxidation of sputtered nanostructured Zn templates. Int. J. Mod. Phys. B 2018, 32, 1850297. [Google Scholar] [CrossRef]
- Huang, M.H.; Wu, Y.; Feick, H.; Tran, N.; Weber, E.; Yang, P. Catalytic Growth of Zinc Oxide Nanowires by Vapor Transport. Adv. Mater. 2001, 13, 113–116. [Google Scholar] [CrossRef]
- Yao, B.D.; Chan, Y.F.; Wang, N. Formation of ZnO nanostructures by a simple way of thermal evaporation. Appl. Phys. Lett. 2002, 81, 757–759. [Google Scholar] [CrossRef]
- Bae, S.Y.; Na, C.W.; Kang, J.H.; Park, J. Comparative Structure and Optical Properties of Ga-, In-, and Sn-Doped ZnO Nanowires Synthesized via Thermal Evaporation. J. Phys. Chem. B 2005, 109, 2526–2531. [Google Scholar] [CrossRef]
- Borysiewicz, M.A.; Gryglas-Borysiewicz, M.; Masłyk, M.; Wojciechowski, T.; Wzorek, M.; Kaczmarski, J.; Wojtowicz, T.; Kamińska, E. Room temperature sputter deposited catalyst-free nanowires with wurtzite/zinc blende ZnO superstructure and their application in electromechanical nanogenerators on polymer and paper substrates. Nanotechnology 2017, 28, 085204. [Google Scholar] [CrossRef] [PubMed]
- Al-Salman, H.S.; Abdullah, M.J. Preparation of ZnO nanostructures by RF-magnetron sputtering on thermally oxidized porous silicon substrate for VOC sensing application. Measurement 2015, 59, 248–257. [Google Scholar] [CrossRef]
- Sundara Venkatesh, P.; Kulandaivel, J. Investigations on the morphological evolution of zinc oxide nanostructures and their optical properties. Cryst. Eng. Comm. 2014, 16, 7426. [Google Scholar] [CrossRef]
- Arroyo-Hernández, M.; Alvaro, R.; Serrano, S.; Luis Costa-Krämer, J. Catalytic growth of ZnO nanostructures by r.f. magnetron sputtering. Nanoscale Res. Lett. 2011, 6, 437. [Google Scholar] [CrossRef]
- Isakov, I.; Panfilova, M.; Sourribes, M.J.L.; Warburton, P.A. Growth of ZnO and ZnMgO nanowires by Au-catalysed molecular-beam epitaxy. Phys. Status Solidi C 2013, 10, 1308. [Google Scholar] [CrossRef]
- Kennedy, O.W.; Coke, M.L.; White, E.R.; Shaffer, M.S.P.; Warburton, P.A. MBE growth and morphology control of ZnO nanobelts with polar axis perpendicular to growth direction. Mater. Lett. 2018, 212, 51–53. [Google Scholar] [CrossRef]
- Sallet, V.; Agouram, S.; Falyouni, F.; Marzouki, A.; Haneche, N.; Sartel, C.; Lusson, A.; Enouz-Vedrenne, S.; Munoz-Sanjose, V.; Galtier, P. Structural characterization of one-dimensional ZnO-based nanostructures grown by MOCVD. Phys. Status Solidi B 2010, 247, 1683. [Google Scholar] [CrossRef]
- Lee, W.; Jeong, M.C.; Myoung, J.M. Catalyst-free growth of ZnO nanowires by metal-organic chemical vapour deposition (MOCVD) and thermal evaporation. Acta Mater. 2004, 52, 3949. [Google Scholar] [CrossRef]
- Szczepanik, A.; Wachnicki, Ł.; Godlewski, M.; Guziewicz, E.; Kopalko, K.; Janik, E.; Łusakowska, E.; Czerwiński, A.; Płuska, M.; Yatsunenko, S.A. ZnO nanostructures by atomic layer deposition method. J. Phys. Conf. Ser. 2009, 146, 012017. [Google Scholar] [CrossRef]
- Fan, G.; Jiao, W.; Yang, L.; Wu, X.; Chen, M.; Gao, R.; Li, Y.; Xie, B.; Liu, J.; Han, M.; et al. Effects of hydriding and ageing of Pd nanoparticles to contact between nanoparticles and quartz and contacts among nanoparticles investigated by the pump-probe technique. Chem. Phys. Lett. 2016, 661, 191. [Google Scholar] [CrossRef]
- Zhao, S.; Yao, C.; Lu, Q.; Song, F.; Wan, J.; Wang, G. Cluster-assembled cobalt doped ZnO nanostructured film prepared by low energy cluster beam deposition. Trans. Nonferrous Met. Soc. China 2009, 19, 1450. [Google Scholar] [CrossRef]
- Tsukazaki, A.; Akasaka, S.; Nakahara, K.; Ohno, Y.; Ohno, H.; Maryenko, D.; Ohtomo, A.; Kawasaki, M. Observation of the fractional quantum Hall effect in an oxide. Nat. Mater. 2010, 9, 889–893. [Google Scholar] [CrossRef] [PubMed]
- Sasa, S.; Maitani, T.; Furuya, Y.; Amano, T.; Koike, K.; Yano, M.; Inoue, M. Microwave performance of ZnO/ZnMgO heterostructure field effect transistors. Phys. Status Solidi A 2011, 208, 449–452. [Google Scholar] [CrossRef]
- Wojtasiak, W.; Góralczyk, M.; Gryglewski, D.; Zając, M.; Kucharski, R.; Prystawko, P.; Piotrowska, A.; Ekielski, M.; Kamińska, E.; Taube, A.; et al. AlGaN/GaN High Electron Mobility Transistors on Semi-Insulating Ammono-GaN Substrates with Regrown Ohmic Contacts. Micromachines 2018, 9, 546. [Google Scholar] [CrossRef] [PubMed]
- Shubina, T.V.; Toropov, A.A.; Lublinskaya, O.G.; Kop’ev, P.S.; Ivanov, S.V.; El-Shaer, A.; Al-Suleiman, M.; Bakin, A.; Waag, A.; Voinilovich, A.; et al. Recombination dynamics and lasing in ZnO/ZnMgO single quantum well structures. Appl. Phys. Lett. 2007, 91, 201104. [Google Scholar] [CrossRef]
- Chou, H.C.; Mazady, A.; Zeller, J.; Manzur, T.; Anwar, M. Room-Temperature Quantum Cascade Laser: ZnO/Zn1−xMgxO Versus GaN/AlxGa1−xN. J. Electron. Mater. 2013, 42, 882–888. [Google Scholar] [CrossRef]
- Yan, X.; Li, W.; Aberle, A.G.; Venkataraj, S. Textured AZO for thin-film Si solar cells: Towards understanding the effect of AZO film thickness on the surface texturing properties. Procedia Eng. 2016, 139, 134–139. [Google Scholar] [CrossRef]
- Dong, H.; Pang, S.; Zhang, Y.; Chen, D.; Zhu, W.; Xi, H.; Chang, J.; Zhang, J.; Zhang, C.; Hao, Y. Improving Electron Extraction Ability and Device Stability of Perovskite Solar Cells Using a Compatible PCBM/AZO Electron Transporting Bilayer. Nanomaterials 2018, 8, 720. [Google Scholar] [CrossRef]
- Kan, Z.; Wang, Z.; Firdaus, Y.; Babics, M.; Alshareef, H.N.; Beaujuge, P.M. Atomic-layer-deposited AZO outperforms ITO in high-efficiency polymer solar cells. J. Mater. Chem. A 2018, 6, 10176–10183. [Google Scholar] [CrossRef]
- Liu, H.; Liu, Y.F.; Xiong, P.P.; Chen, P.; Li, H.Y.; Hou, J.W.; Kang, B.N.; Duan, Y. Aluminum-Doped Zinc Oxide Transparent Electrode Prepared by Atomic Layer Deposition for Organic Light Emitting Devices. IEEE Trans. Nanotechnol. 2017, 16, 634–638. [Google Scholar] [CrossRef]
- Hsu, C.H.; Chen, S.Y.; Chen, W.C.; Chang, C.H.; Chen, C.Y.; Tsai, J.H.; Liu, W.C. Study of a GaN-Based LED with an Al/AZO Composite Transparent Conductive Layer. IEEE Trans. Electron. Devices 2017, 64, 3678–3682. [Google Scholar] [CrossRef]
- Al Eghfeli, M.; Abdul Hadi, S.; El Atab, N.; Nayfeh, A. Demonstration of aluminum doped ZnO as anti-reflection coating. In Proceedings of the 2016 IEEE 43rd Photovoltaic Specialists Conference (PVSC), Portland, OR, USA, 5–10 June 2016; pp. 2765–2769. [Google Scholar] [CrossRef]
- Chowdhury, A.R.; Rao, V.P.; Ghosh, B.; Banerjee, S. Antireflection coating on solar cell using ZnO nanostructure. AIP Conf. Proc. 2012, 1447, 777. [Google Scholar] [CrossRef]
- Grundmann, M.; Frenzel, H.; Lajn, A.; Lorenz, M.; Schein, F.; von Wenckstern, H. Transparent semiconducting oxides: Materials and devices. Phys. Status Solidi A 2010, 207, 1437–1449. [Google Scholar] [CrossRef]
- Faber, H.; Das, S.; Lin, Y.H.; Pliatsikas, N.; Zhao, K.; Kehagias, T.; Dimitrakopulos, G.; Amassian, A.; Patsalas, P.A.; Anthopoulos, T.D. Heterojunction oxide thin-film transistors with unprecedented electron mobility grown from solution. Sci. Adv. 2017, 3, e1602640. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.; Li, T.; Li, Y.; Qiu, J.; Ma, X.; Chen, X.; Hu, X.; Zhang, W. A high power ZnO thin film piezoelectric generator. Appl. Surf. Sci. 2016, 364, 670–675. [Google Scholar] [CrossRef]
- Lai, S.L.; Zakaria, M.R.; Hashim, U.; Foo, K.L.; Haarindra Prasad, R. Fabrication of SAW device by using ZnO thin film as a sensing area. In Proceedings of the 2014 IEEE International Conference on Semiconductor Electronics (ICSE2014), Kuala Lumpur, Malaysia, 27–29 August 2014; pp. 380–383. [Google Scholar] [CrossRef]
- Du, X.; Fu, Y.; Tan, S.C.; Luo, J.; Flewitt, A.; Maeng, S.; Kim, S.H.; Choi, Y.; Lee, D.S.; Park, R.M.; et al. ZnO film based surface acoustic wave micro-pump. J. Phys. Conf. Ser. 2007, 76, 012047. [Google Scholar] [CrossRef]
- Santos, Y.P.; Valença, E.; Machado, R.; Macêdo, M.A. A novel structure ZnO-Fe-ZnO thin film memristor. Mater. Sci. Semicond. Process. 2018, 86, 43–48. [Google Scholar] [CrossRef]
- Kim, K.K.; Kim, H.S.; Hwang, D.K.; Lim, J.H.; Park, S.J. Realization of p-type ZnO thin films via phosphorus doping and thermal activation of the dopant. Appl. Phys. Lett. 2003, 83, 63. [Google Scholar] [CrossRef]
- Snigurenko, D.; Kopalko, K.; Krajewski, T.A.; Jakiela, R.; Guziewicz, E. Nitrogen doped p-type ZnO films and p-n homojunction. Semicond. Sci. Technol. 2015, 30, 015001. [Google Scholar] [CrossRef]
- Bu, I.Y.Y. Sol-gel production of p-type ZnO thin film by using sodium doping. Superlattices Microstruct. 2016, 96, 59–66. [Google Scholar] [CrossRef]
- Li, G.; Wang, H.; Wang, Q.; Zhao, Y.; Wang, Z.; Du, J.; Ma, Y. Structure and properties of Co-doped ZnO films prepared by thermal oxidization under a high magnetic field. Nanoscale Res. Lett. 2015, 10, 112. [Google Scholar] [CrossRef] [PubMed]
- Hadimani, P.L.; Ghosh, S.S.; Sil, A. Preparation of Fe doped ZnO thin films and their structural, magnetic, electrical characterization. Superlattices Microstruct. 2018, 120, 199–208. [Google Scholar] [CrossRef]
- Agarwal, D.C.; Singh, U.B.; Gupta, S.; Singhal, R.; Kulriya, P.K.; Singh, F.; Tripathi, A.; Singh, J.; Joshi, U.S.; Avasthi, D.K. Enhanced room temperature ferromagnetism and green photoluminescence in Cu doped ZnO thin film synthesised by neutral beam sputtering. Sci. Rep. 2019, 9, 6675. [Google Scholar] [CrossRef] [PubMed]
- Levinson, L.; Philipp, H.R. Zinc oxide varistors—A review. Am. Ceram. Soc. Bull. 1986, 65, 639–646. [Google Scholar]
- Wallenhorst, L.; Gurău, L.; Gellerich, A.; Militz, H.; Ohms, G.; Viöla, W. UV-blocking properties of Zn/ZnO coatings on wood deposited by cold plasma spraying at atmospheric pressure. Appl. Surf. Sci. 2018, 434, 1183–1192. [Google Scholar] [CrossRef]
- Carvalho, J.T.; Dubceac, V.; Grey, P.; Cunha, I.; Fortunato, E.; Martins, R.; Clausner, A.; Zschech, E.; Pereira, L. Fully Printed Zinc Oxide Electrolyte-Gated Transistors on Paper. Nanomaterials 2019, 9, 169. [Google Scholar] [CrossRef]
- Grey, P.; Gaspar, D.; Cunha, I.; Barras, R.; Carvalho, J.T.; Ribas, J.R.; Fortunato, E.; Martins, R.; Pereira, L. Handwritten Oxide Electronics on Paper. Adv. Mater. Technol. 2017, 2, 1700009. [Google Scholar] [CrossRef]
- Kim, J.J.; Kim, K.S.; Jung, G.Y. Fabrication of flexible dye-sensitised solar cells with photoanodes composed of periodically aligned single crystalline vertical ZnO NRs by utilising a direct metal transfer method. J. Mater. Chem 2011, 21, 7730–7735. [Google Scholar] [CrossRef]
- Wagh, M.S.; Jain, G.H.; Patil, D.R.; Patil, S.A.; Patil, L.A. Modified zinc oxide thick film resistors as NH3 gas sensor. Sens. Actuators B Chem. 2006, 115, 128–133. [Google Scholar] [CrossRef]
- Neelakantan, N.K.; Weisensee, P.B.; Overcash, J.W.; Torrealba, E.J.; King, W.P.; Suslick, K.S. Spray-on omniphobic ZnO coatings. RSC Adv. 2015, 5, 69243–69250. [Google Scholar] [CrossRef]
- Memarzadeh, K.; Sharili, A.S.; Huang, J.; Rawlinson, S.C.F.; Allaker, R.P. Nanoparticulate zinc oxide as a coating material for orthopedic and dental implants. J. Biomed. Mater. Res. Part A 2015, 103A, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Vogel, E.M.; Nelson, T.J. Novel synthesis and characterization of ZnO-based phosphors. Displays 1986, 7, 78–80. [Google Scholar] [CrossRef]
- Gong, H.; Hu, J.Q.; Wang, J.H.; Ong, C.H.; Zhu, F.R. Nano-crystalline Cu-doped ZnO thin film gas sensor for CO. Sens. Actuators B Chem. 2006, 115, 247–251. [Google Scholar] [CrossRef]
- Kumar, R.; Al-Dossary, O.; Kumar, G.; Umar, A. Zinc Oxide Nanostructures for NO2 Gas Sensor Applications: A Review. Nano-Micro Lett. 2015, 7, 97–120. [Google Scholar] [CrossRef]
- Agarwal, S.; Rai, P.; Navarrete Gatell, E.; Llobet, E.; Güelle, F.; Kumar, M.; Awasthi, K. Gas sensing properties of ZnO nanostructures (flowers/rods) synthesized by hydrothermal method. Sens. Actuators B Chem. 2019, 292, 24–31. [Google Scholar] [CrossRef]
- Ong, C.B.; Ng, L.Y.; Mohammad, A.W. A review of ZnO nanoparticles as solar photocatalysts: Synthesis, mechanisms and applications. Renew. Sustain. Energy Rev. 2018, 81, 536–551. [Google Scholar] [CrossRef]
- Wolska, E.; Kaszewski, J.; Kiełbik, P.; Grzyb, J.; Godlewski, M.M.; Godlewski, M. Rare earth activated ZnO nanoparticles as biomarkers. Opt. Mater. 2014, 36, 1655–1659. [Google Scholar] [CrossRef]
- Premanathan, M.; Karthikeyan, K.; Jeyasubramanian, K.; Manivannan, G. Selective toxicity of ZnO nanoparticles toward Gram-positive bacteria and cancer cells by apoptosis through lipid peroxidation. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lee, J.S.; Kim, D.; Zhu, L. Exploration of zinc oxide nanoparticles as a multitarget and multifunctional anticancer nanomedicine. ACS Appl. Mater. Interfaces 2017, 9, 39971–39984. [Google Scholar] [CrossRef]
- Wang, J.; Gao, S.; Wang, S.; Xu, Z.; Wei, L. Zinc oxide nanoparticles induce toxicity in CAL 27 oral cancer cell lines by activating PINK1/Parkin-mediated mitophagy. Int. J. Nanomed. 2018, 13, 3441–3450. [Google Scholar] [CrossRef]
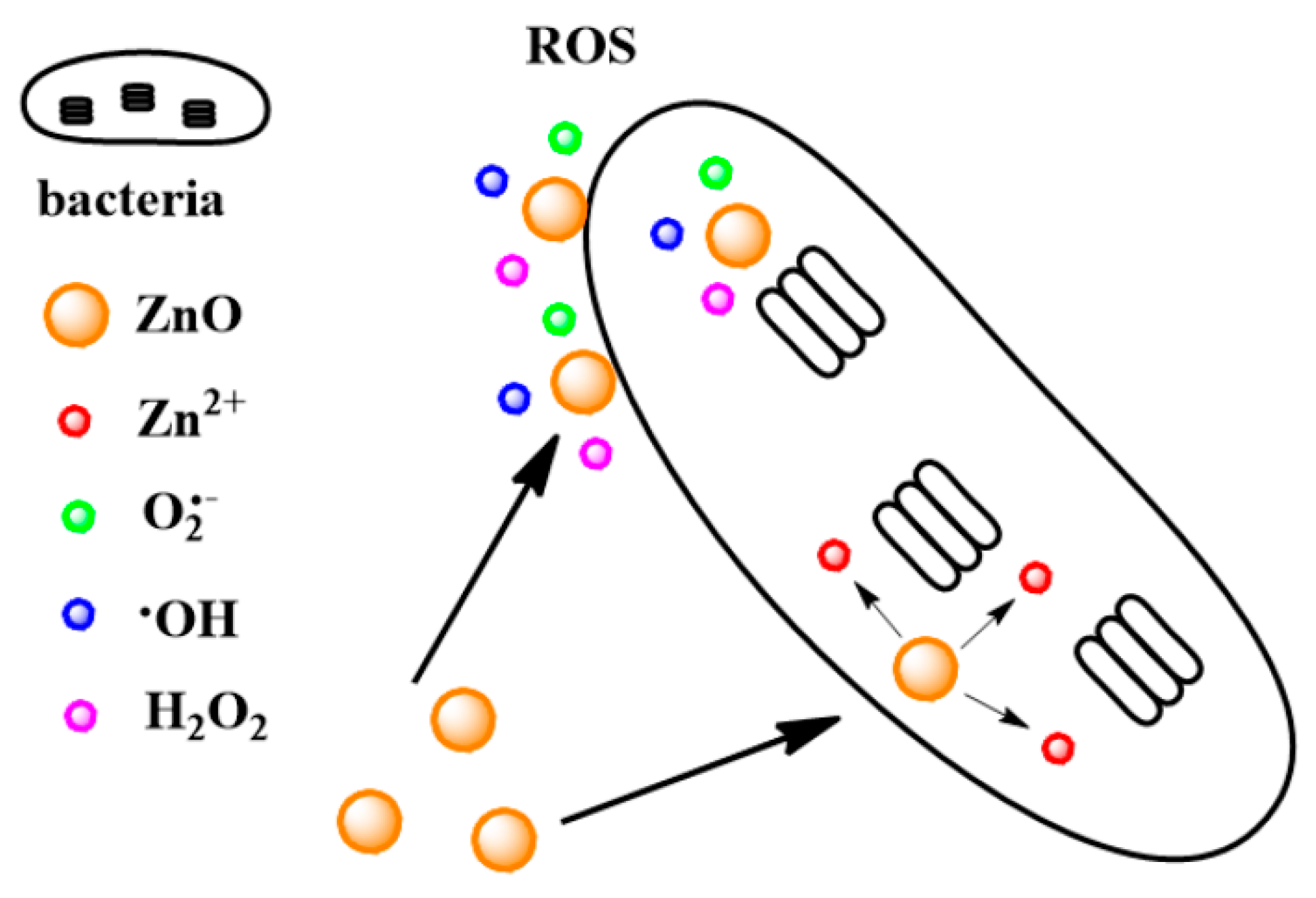
- Jiang, J.; Pi, J.; Cai, J. The Advancing of Zinc Oxide Nanoparticles for Biomedical Applications. Bioinorg. Chem. Appl. 2018, 2018, 1062562. [Google Scholar] [CrossRef] [PubMed]
- Shateri-Khalilabad, M.; Yazdanshenas, M.E. Bifunctionalization of cotton textiles by ZnO nanostructures: Antimicrobial activity and ultraviolet protection. Text. Res. J. 2013, 83, 993–1004. [Google Scholar] [CrossRef]
- Wang, Z.L.; Wu, W.; Falconi, C. Piezotronics and piezo-phototronics with third-generation semiconductors. MRS Bull. 2018, 43, 922–927. [Google Scholar] [CrossRef]
- Bae, J.; Song, M.K.; Park, Y.J.; Kim, J.M.; Liu, M.; Wang, Z.L. Fiber Supercapacitors Made of Nanowire-Fiber Hybrid Structures for Wearable/Flexible Energy Storage. Angew. Chem. Int. Ed. 2011, 50, 1683–1687. [Google Scholar] [CrossRef] [PubMed]
- Borysiewicz, M.A.; Ekielski, M.; Ogorzałek, Z.; Wzorek, M.; Kaczmarski, J.; Wojciechowski, T. Highly transparent supercapacitors based on ZnO/MnO2 nanostructures. Nanoscale 2017, 9, 7577–7587. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, O.; Kiesel, P.; Van de Walle, C.G.; Johnson, N.M.; Nause, J.; Döhler, G.H. Effects of an Electrically Conducting Layer at the Zinc Oxide Surface. Jpn. J. Appl. Phys. 2005, 144, 7271. [Google Scholar] [CrossRef]
- Look, D.C. Quantitative analysis of surface donors in ZnO. Surf. Sci. 2007, 601, 5315–5319. [Google Scholar] [CrossRef]
- Park, G.H.; Zhang, S.B.; Wei, S.H. Origin of p-type doping difficulty in ZnO: The impurity perspective. Phys. Rev. B 2002, 66, 073202. [Google Scholar] [CrossRef]
- Volnianska, O.; Boguslawski, P.; Kaczkowski, J.; Jakubas, P.; Jezierski, A.; Kaminska, E. Theory of doping properties of Ag acceptors in ZnO. Phys. Rev. B 2009, 80, 245212. [Google Scholar] [CrossRef]
- Chai, J.; Mendelsberg, R.J.; Reeves, R.J.; Kennedy, J.; von Wenckstern, H.; Schmidt, M.; Grundmann, M.; Doyle, K.; Myers, T.H.; Durbin, S.M. Identification of a Deep Acceptor Level in ZnO Due to Silver Doping. J. Electron. Mater. 2010, 39, 577–583. [Google Scholar] [CrossRef]
- Yan, Y.; Al-Jassim, M.M.; Wei, S.H. Doping of ZnO by group-IB elements. Appl. Phys. Lett. 2006, 89, 181912. [Google Scholar] [CrossRef]
- Kaminska, E.; Pasternak, I.; Boguslawski, P.; Jezierski, A.; Dynowska, E.; Jakiela, R.; Przezdziecka, E.; Piotrowska, A.; Kossut, J. Group IB acceptors in ZnO: Experiment and theory. AIP Conf. Proc. 2009, 1199, 120–121. [Google Scholar] [CrossRef]
- Lyons, L.; Janotti, A.; Van de Walle, C.G. Why nitrogen cannot lead to p-type conductivity in ZnO. Appl. Phys. Lett. 2009, 95, 252105. [Google Scholar] [CrossRef]
- Meyer, B.K.; Stehr, J.; Hofstaetter, A.; Volbers, N.; Zeuner, A.; Sann, J. On the role of group I elements in ZnO. Appl. Phys. A 2007, 88, 119–123. [Google Scholar] [CrossRef]
- Deng, R.; Yao, B.; Li, Y.F.; Yang, T.; Li, B.H.; Zhang, Z.Z.; Shan, C.X.; Zhang, J.Y.; Shen, D.Z. Influence of oxygen/argon ratio on structural, electrical and optical properties of Ag-doped ZnO thin films. J. Cryst. Growth 2010, 312, 1813–1816. [Google Scholar] [CrossRef]
- Gruzintsev, A.N.; Volkov, V.T.; Khodos, I.I. Photoelectric properties of ZnO films doped with Cu and Ag acceptor impurities. Semiconductors 2003, 37, 259–262. [Google Scholar] [CrossRef]
- Yao, B.; Guan, L.X.; Xing, G.Z.; Zhang, Z.Z.; Li, B.H.; Wei, Z.P.; Wang, X.H.; Cong, C.X.; Xie, Y.P.; Lu, Y.M.; et al. P-type conductivity and stability of nitrogen-doped zinc oxide prepared by magnetron sputtering. J. Lumin. 2007, 122–123, 191–194. [Google Scholar] [CrossRef]
- Lim, J.H.; Kang, C.K.; Kim, K.K.; Park, I.K.; Hwang, D.K.; Park, S.J. UV Electroluminescence Emission from ZnO Light-Emitting Diodes Grown by High-Temperature Radiofrequency Sputtering. Adv. Mater. 2006, 18, 2720–2724. [Google Scholar] [CrossRef]
- Wang, X.H.; Yao, B.; Zhang, Z.Z.; Li, B.H.; Wei, Z.P.; Shen, D.Z.; Lu, Y.M.; Fan, X.W. The mechanism of formation and properties of Li-doped p-type ZnO grown by a two-step heat treatment. Semicond. Sci. Technol. 2006, 21, 494. [Google Scholar] [CrossRef]
- Qin, J.M.; Yao, B.; Yan, Y.; Zhang, J.Y.; Jia, X.P.; Zhang, Z.Z.; Li, B.H.; Shan, C.X.; Shen, D.Z. Formation of stable and reproducible low resistivity and high carrier concentration p-type ZnO doped at high pressure with Sb. Appl. Phys. Lett. 2009, 95, 022101. [Google Scholar] [CrossRef]
- Lin, S.S.; Lu, J.G.; Ye, Z.Z.; He, H.P.; Gu, X.Q.; Chen, L.X.; Huang, J.Y.; Zhao, B.H. P-type behavior in Na-doped ZnO films and ZnO homojunction light-emitting diodes. Solid State Commun. 2008, 148, 25–28. [Google Scholar] [CrossRef]
- Look, D.C.; Reynolds, D.C.; Litton, C.W.; Jones, R.L.; Eason, D.B.; Cantwell, G. Characterization of homoepitaxial p-type ZnO grown by molecular beam epitaxy. Appl. Phys. Lett. 2002, 81, 1830. [Google Scholar] [CrossRef]
- Wang, L.G.; Zunger, A. Cluster-Doping Approach for Wide-Gap Semiconductors: The Case of p-Type ZnO. Phys. Rev. Lett. 2003, 90, 256401. [Google Scholar] [CrossRef] [PubMed]
- Neugebauer, J.; Van de Walle, C.G. Role of hydrogen in doping of GaN. Appl. Phys. Lett. 1996, 68, 1829. [Google Scholar] [CrossRef]
- Chen, L.L.; Lu, J.G.; Ye, Z.Z.; Lin, Y.M.; Zhao, B.H.; Ye, Y.M.; Li, J.S.; Zhu, L.P. P-type behavior in In–N codoped ZnO thin films. Appl. Phys. Lett. 2005, 87, 252106. [Google Scholar] [CrossRef]
- Krtschil, A.; Dadgar, A.; Oleynik, N.; Bläsing, J.; Diez, A.; Krost, A. Local p-type conductivity in zinc oxide dual-doped with nitrogen and arsenic. Appl. Phys. Lett. 2005, 87, 262105. [Google Scholar] [CrossRef]
- Lu, J.G.; Zhang, Y.Z.; Ye, Z.Z.; Zhu, L.P.; Wang, L.; Zhao, B.H.; Liang, Q.L. Low-resistivity, stable p-type ZnO thin films realized using a Li–N dual-acceptor doping method. Appl. Phys. Lett. 2006, 88, 222114. [Google Scholar] [CrossRef]
- Volnianska, O.; Bogusławski, P.; Kaminska, E. Ag and N acceptors in ZnO: An ab initio study of acceptor pairing, doping efficiency, and the role of hydrogen. Phys. Rev. B 2012, 85, 165212. [Google Scholar] [CrossRef]
- Tang, K.; Gu, S.L.; Ye, J.D.; Zhu, S.M.; Zhang, R.; Zheng, Y.D. Recent progress of the native defects and p-type doping of zinc oxide. Chin. Phys. B 2017, 26, 047702. [Google Scholar] [CrossRef]



© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borysiewicz, M.A. ZnO as a Functional Material, a Review. Crystals 2019, 9, 505. https://doi.org/10.3390/cryst9100505
Borysiewicz MA. ZnO as a Functional Material, a Review. Crystals. 2019; 9(10):505. https://doi.org/10.3390/cryst9100505
Chicago/Turabian StyleBorysiewicz, Michał A. 2019. "ZnO as a Functional Material, a Review" Crystals 9, no. 10: 505. https://doi.org/10.3390/cryst9100505
APA StyleBorysiewicz, M. A. (2019). ZnO as a Functional Material, a Review. Crystals, 9(10), 505. https://doi.org/10.3390/cryst9100505




