Contemporary Apparatus for Single Crystals Growth of Oxide Compounds and Metals by Optical Floating Zone (FZ)
Abstract
:1. Introduction
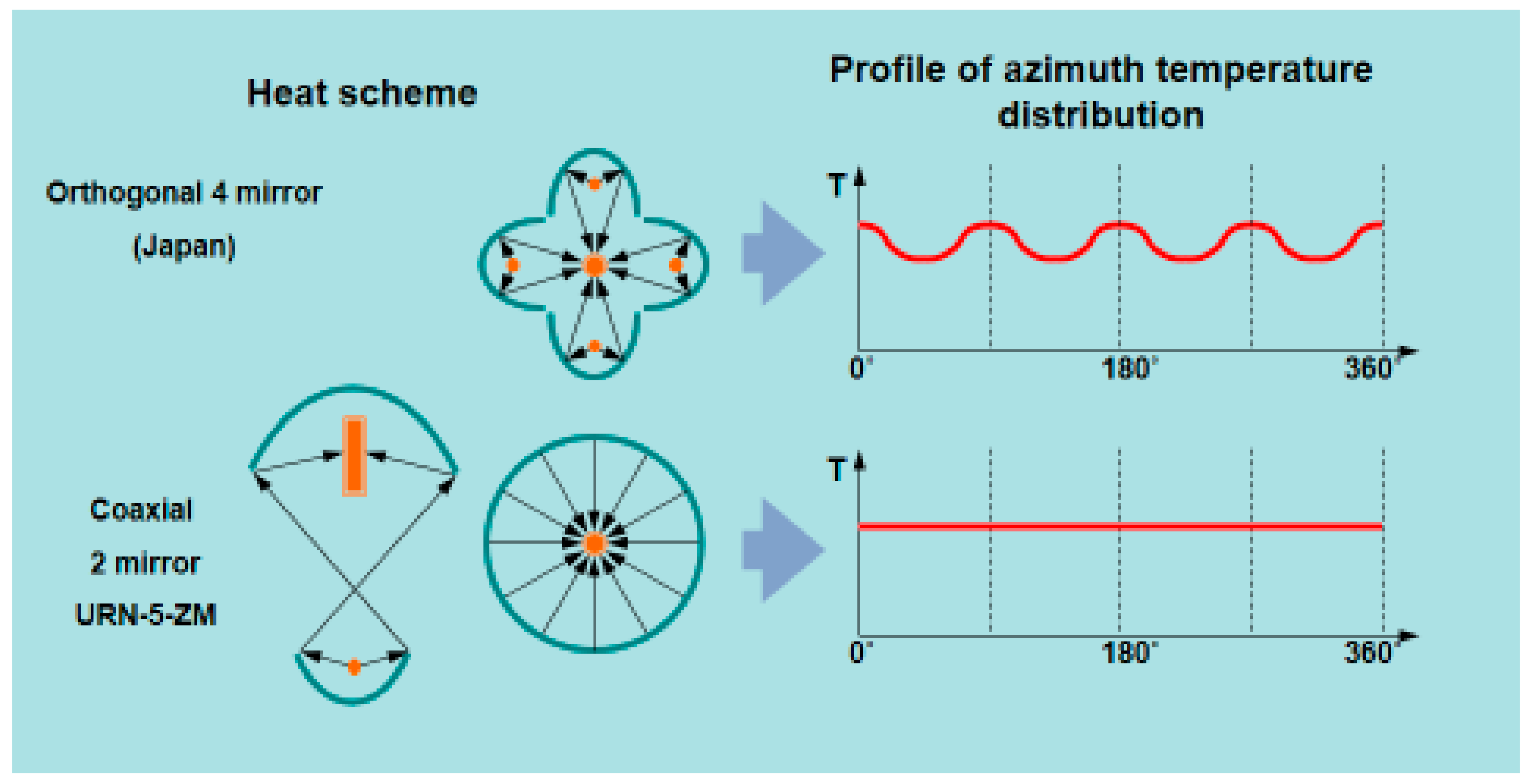
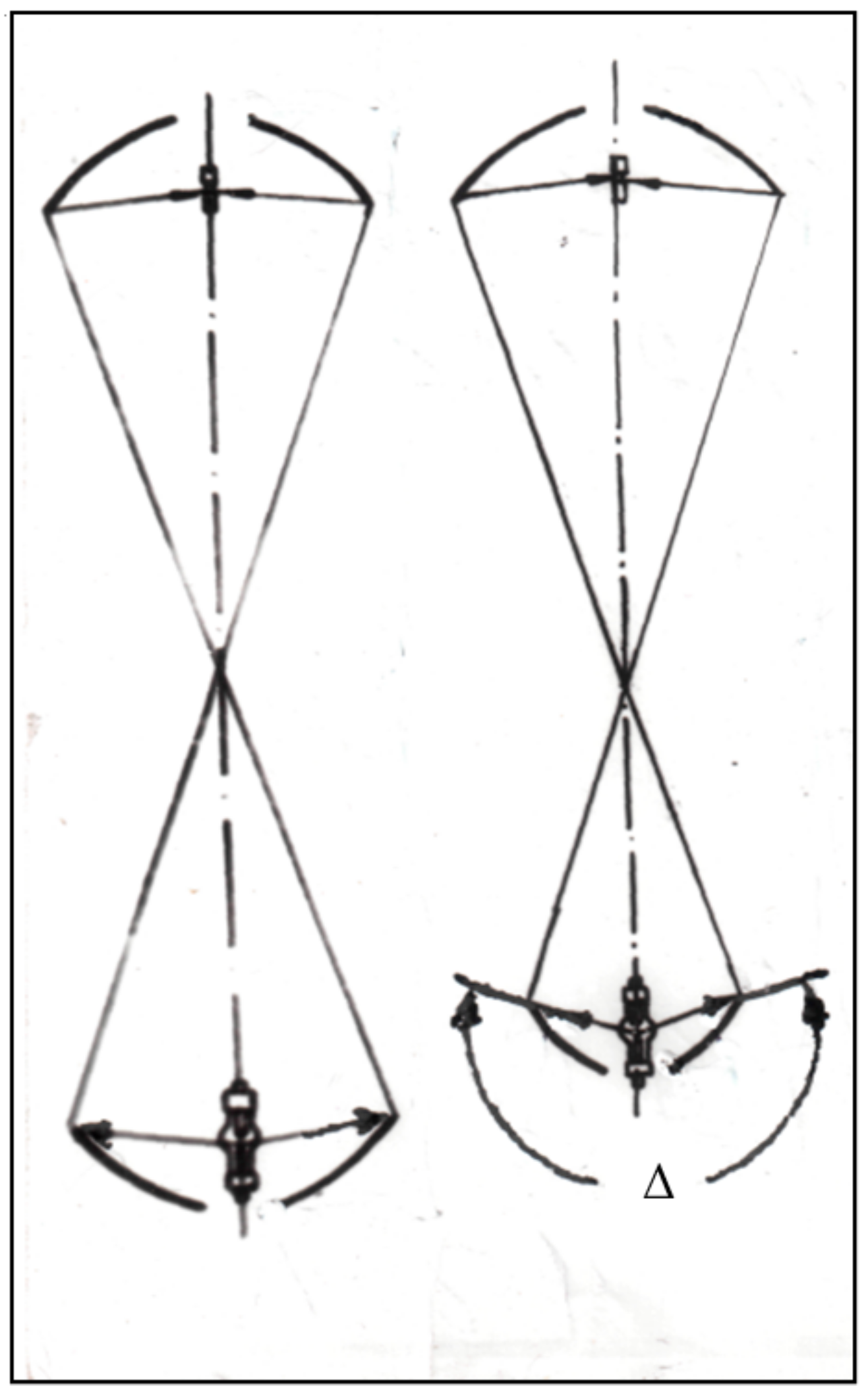
- Change of used mirror geometry,
- New light flux regulation system,
- Use of more precision mechanical systems of moving and rotation of crystals and feed rod,
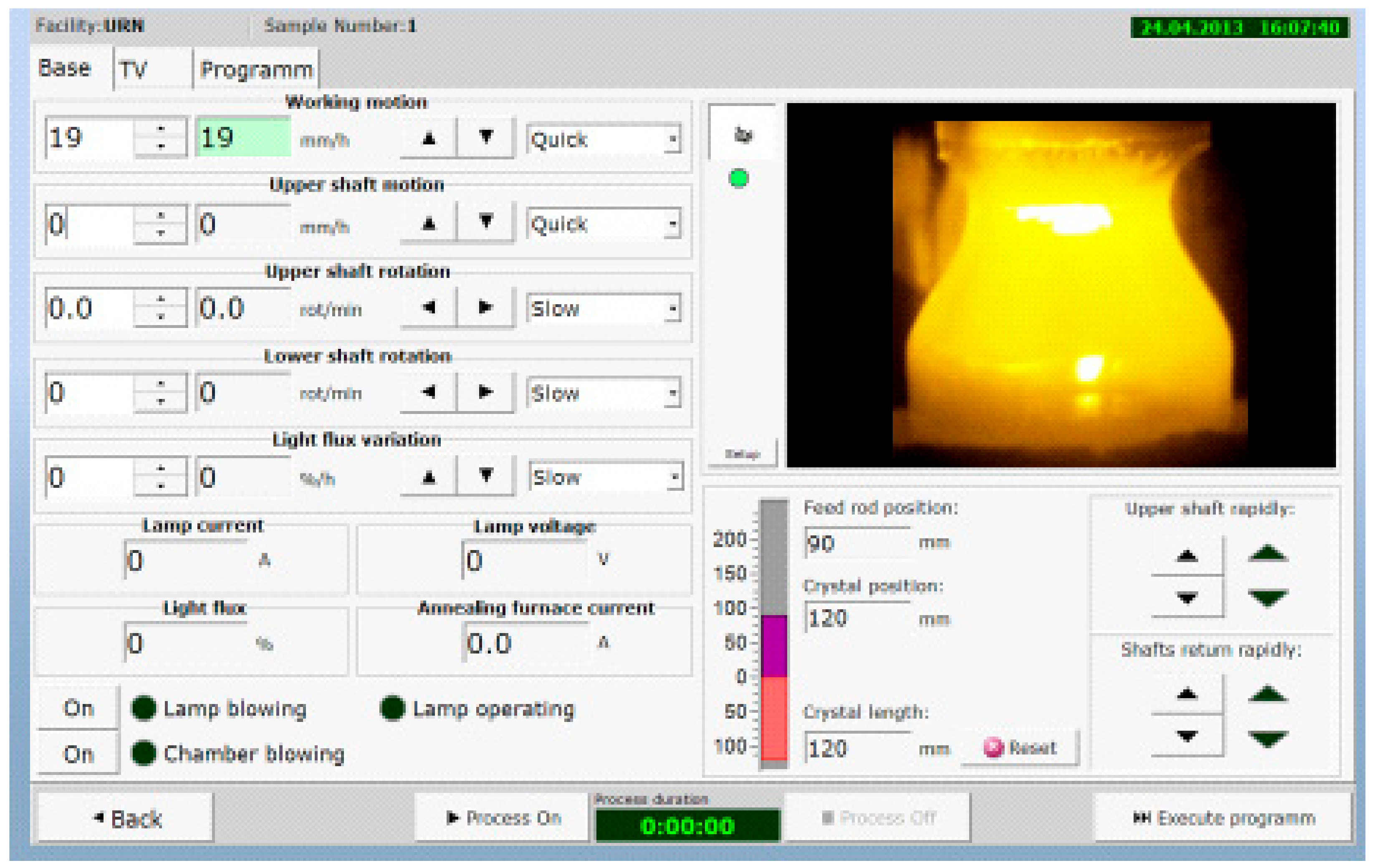
- General use of micro-processing techniques for all electric and electronic systems with PC control and programming,
- Development and application of PC control and stabilization of grown crystal diameters on the base of the TV-image of the FZ recognition processing.
- Use of single-crystalline high-quality sapphire as material for the high-pressure transparent tube of the crystallization chamber.
- -
- More value of light power concentrated in a working focal point as a result of using a lower mirror with bigger scope angle. It leads to increasing of maximal temperature in focus and allows to grow larger size crystals.
- -
- New regulation light flux system without changing the focused power distribution and not disturbing the circular symmetry of the heat field in the focal point.
- -
- Use of micro-processing in all the system increases the long time reliability of the facility.
- -
- The fused quartz was some time ago a unique material for manufacturing of transparent high-pressure tubes of the crystallization chamber. Now the technology of high-quality sapphire crystals is very developed and allows obtaining the tubes of any size. Sapphire have essential advantages against quartz in the durability and strength, and should be estimated as the preferred material for this task.
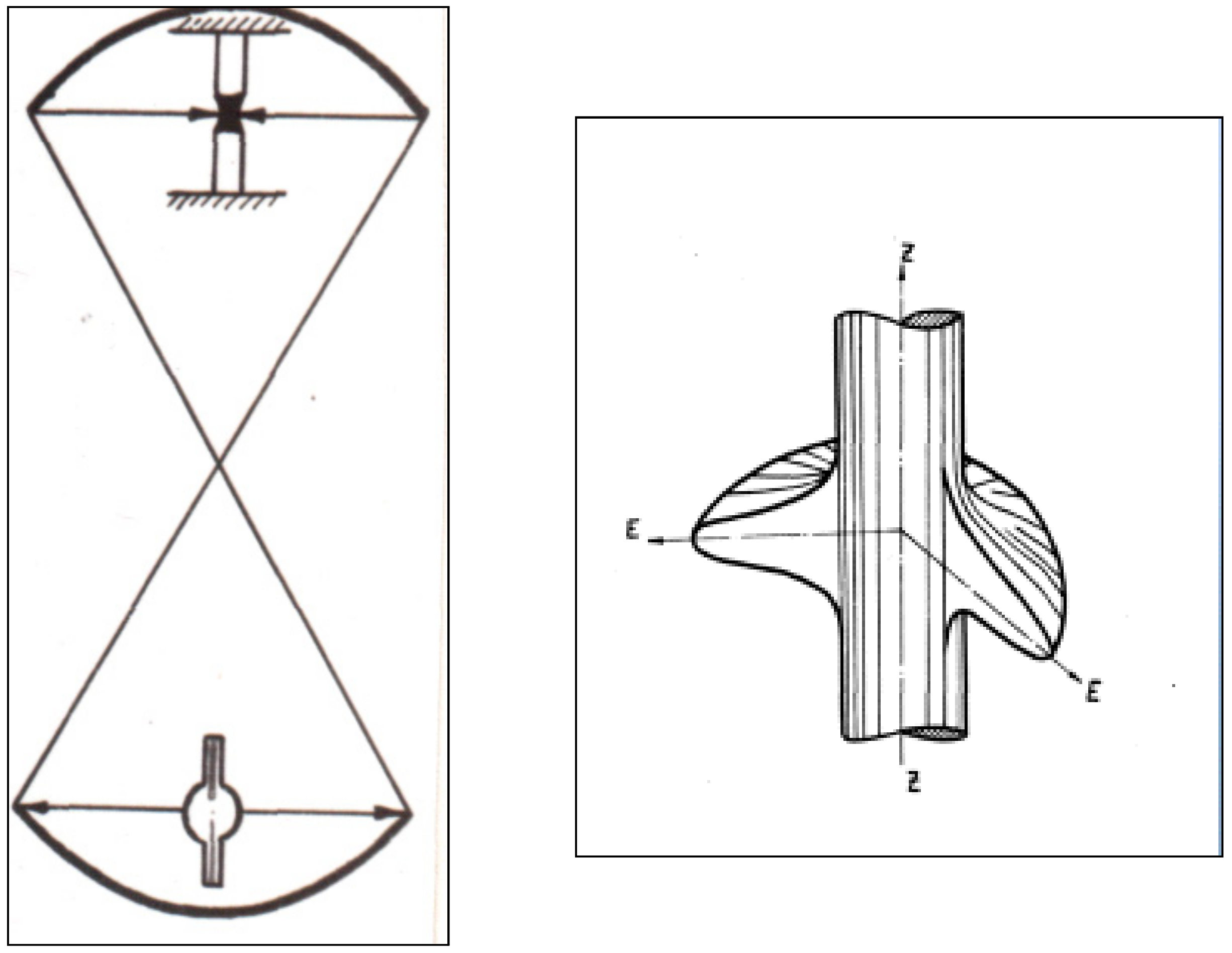
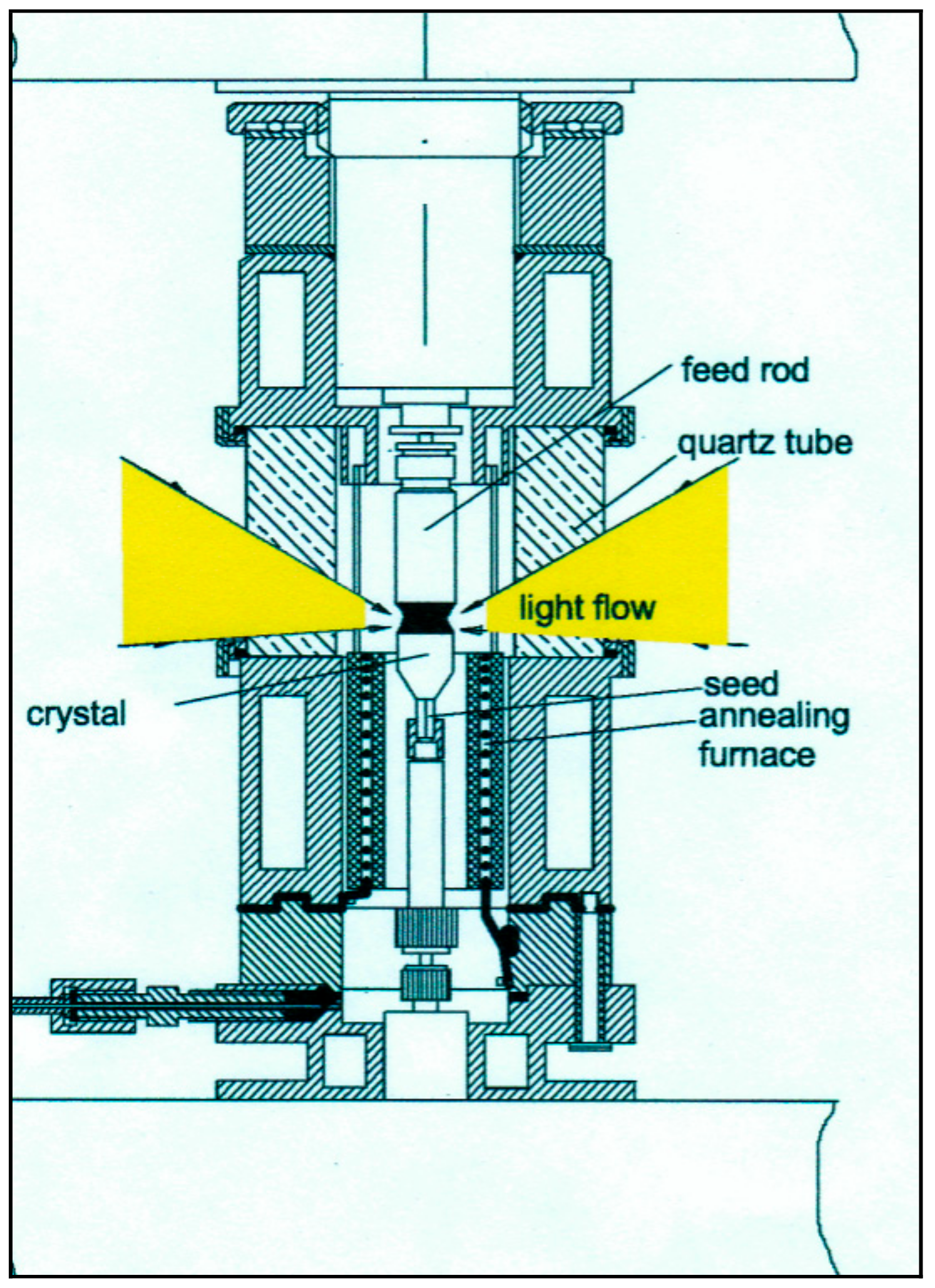
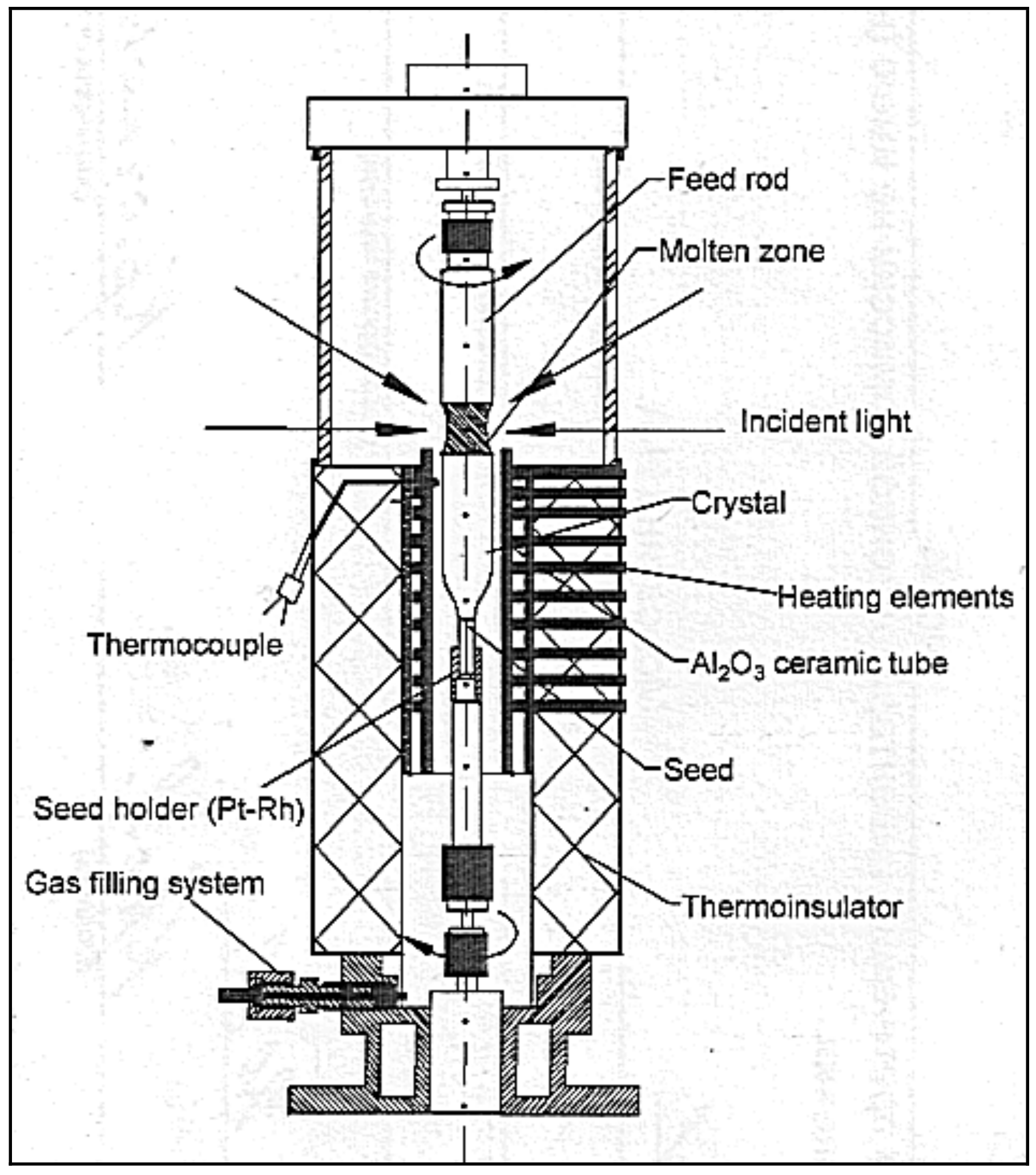
2. Apparatus
- (a)
- The heating zone should be circular uniform, and the crystallization interface should be near to a flat shape.
- (b)
- The gas-phase over the melt during the crystal growth and annealing should be controlled in order to keep the stoichiometric composition and desired valence of the metal ions.
- (c)
- Annealing of the growing crystals at uniform temperatures sufficient to relieve the tenseness should be provided. Such annealing should be controlled during the crystallization process to allow active influence on crystal growth.
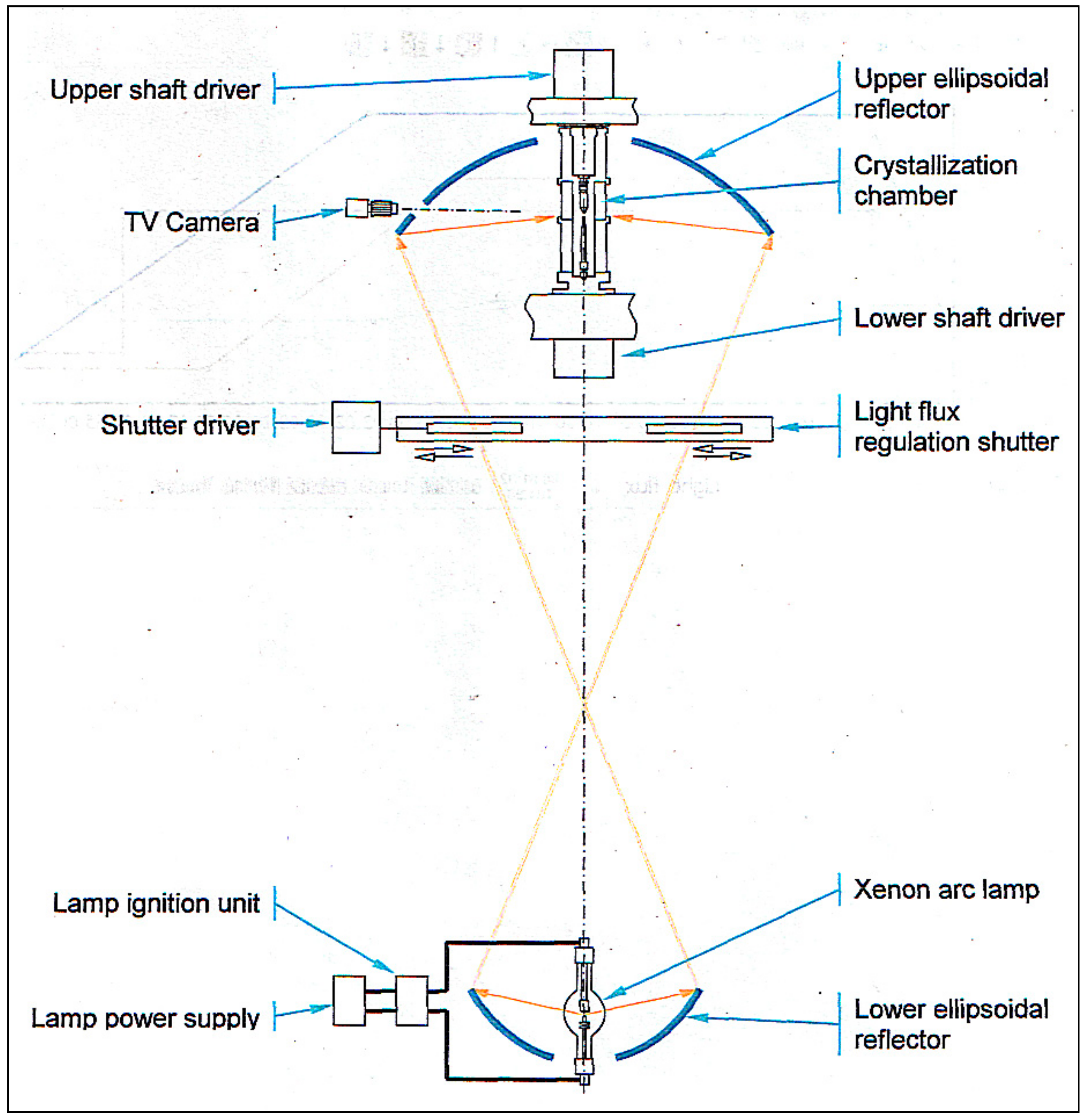
- -
- Xenon high-pressure short arc lamp as the light energy source; electron stabilization of the light flux;
- -
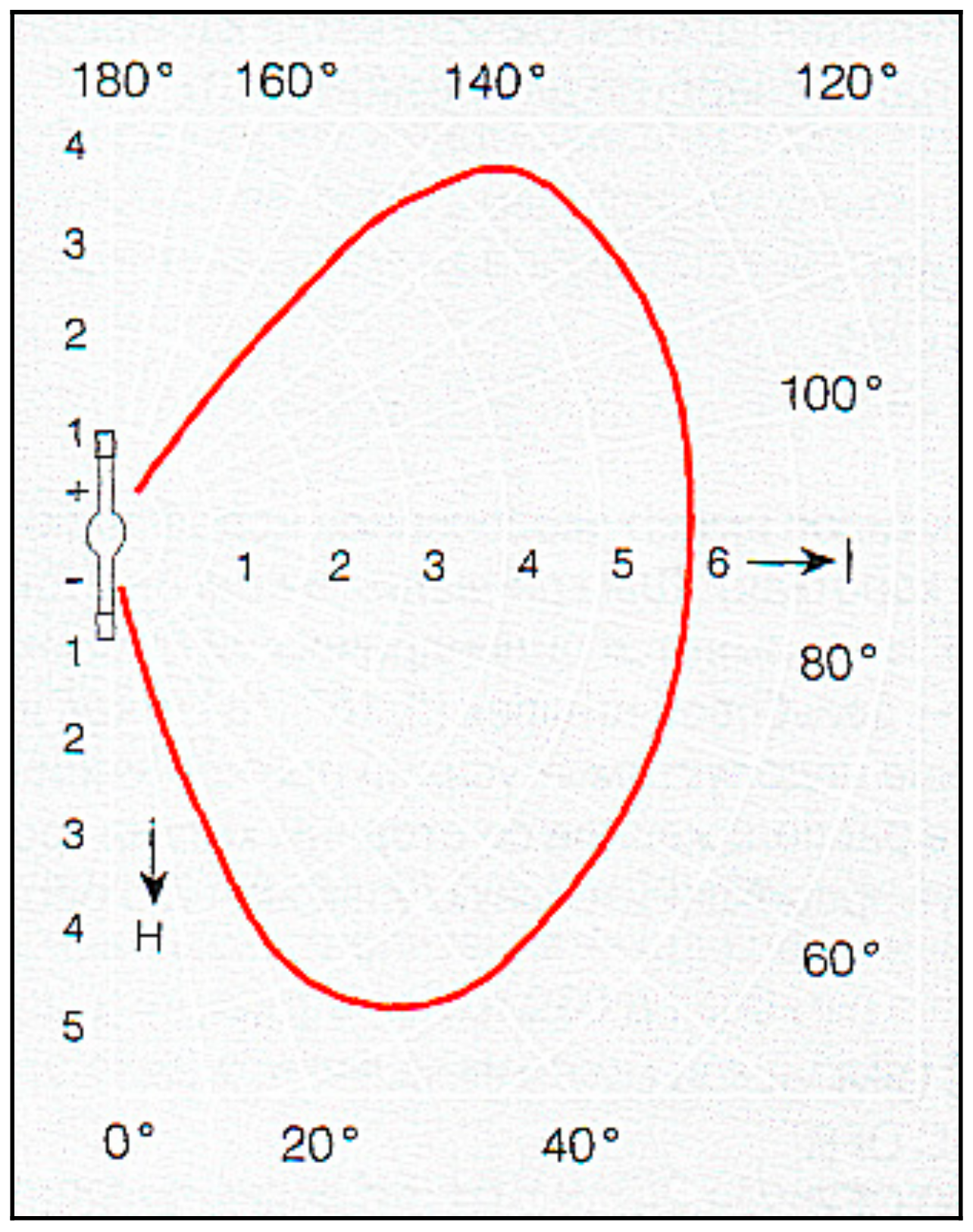
- Employing special ellipsoidal glass reflectors with a scope angle of 97.5°, 600 mm. diam. (upper mirror) and 250°, 360 mm. dia. (lower mirror) (the specific heat load on the reflector surface does not exceed 0.2 W/cm2),
- -
- Smooth control of the light concentrator aperture (and a light quantity on the subject of heating) ranging from zero to maximum.
- -
- The high-pressure crystallization chamber.
- -
- Low pressure and gas flow crystallization chamber.
3. Growth of Single Crystals
3.1. Rotation Effect
3.2. Y3Fe5O12
3.3. Gd3Fe5O12
3.4. BaFe12O19
3.5. (NixCo1−x) Fe2O4
3.6. Ni0.5Zn0.5Fe2O4
3.7. Mn0.5Zn0.5Fe2O4
3.8. RFeO3 (R-Rare Earth Element)
3.9. TiO2
3.10. SrTiO3
3.11. β-Ga2O3
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Poplawsky, R.P.; Tomas, J.E. Floating zone crystals using an arc image furnace. Rev. Sci. Instrum. 1960, 31, 1303. [Google Scholar] [CrossRef]
- Kooy, G.; Gouwenberg, H.J.M. Zone melting of oxides in a carbon arc image furnace. Philips Techn. Rev. 1962, 23. [Google Scholar]
- Halden, F.F.; Sedlacek, R. Verneuil Crystal Growth in the Arc-Image Furnace. Rev. Sci. Instrum. 1963, 34, 622. [Google Scholar] [CrossRef]
- O’Bryan, H.M., Jr.; O’Connor, P.B. Growth of Nd:YAG crystals by floating zone technique. Am. Ce Soc. Bull. 1966, 45, 578. [Google Scholar]
- Marsch, A.; Fairholme, R.; Gill, G. Domain-wall mobility in orthoferrite crystals grown by float-zone technique. IEEE Transact. Magn. 1971, 7, 470–471. [Google Scholar] [CrossRef]
- Field, W.G.; Wagner, R.W. Thermal imaging for single crystal growth and its application to ruby. J. Cryst. Growth 1968, 3, 799–803. [Google Scholar] [CrossRef]
- Medvedev, S.A.; Balbashov, A.M. Application of arc optical furnace for ferrite crystal growth. In Ferrite; 1968; p. 438, (RussMgian). [Google Scholar]
- Okada, T.; Matsumi, K.; Makino, H. Preparation of othoferrite single crystal by the floating zone technique. Nippon Electr. Compagn. Res. Dev. 1970, 19, 102. [Google Scholar]
- Kitazawa, K.; Nagashima, K.; Mizutani, T.; Fueki, K. A new thermal imaging system utilizing a Xe arc lamp and an ellipsoidal mirror for crystallization of refractory oxides. J. Cryst. Growth 1977, 39, 211–215. [Google Scholar] [CrossRef]
- Kimura, S.; Shindo, I. Single crystal growth of YIG by floating zone method. J. Cryst. Growth 1977, 41, 192. [Google Scholar] [CrossRef]
- Tsuiki, H.; Kitazama, K.; Masumoto, K.; Shiroki, K.; Fueki, F. Single crystal growth of pure and Nd-doped Y2O3 by floating zone method with Xe arc lamp imaging furnace. J. Cryst. Growth 1980, 49, 71–76. [Google Scholar] [CrossRef]
- Takei, H.; Kitamura, K. Growth of FeTiO3 (ilmenite) crystals by the floating -zone method. J. Cryst. Growth 1978, 44, 629. [Google Scholar] [CrossRef]
- Eyer, A.; Nitsche, R.; Zimmerman, H. A double-ellipsoid mirror furnace for zone crystallization experiments in spacelab. J. Cryst. Growth 1979, 47, 219–229. [Google Scholar] [CrossRef]
- Shindo, I. Single crystal growth of akermanite (Ca2MgSi2O7) and gehlenite (Ca2Al2SiO4) by the floating zone method. J. Cryst. Growth 1979, 46, 569. [Google Scholar]
- Balbashov, A.M.; Egorov, S.K. Apparatus for growth of single crystals of oxide compounds by floating zone melting with radiation heating. J. Cryst. Growth 1981, 52, 498. [Google Scholar] [CrossRef]
- Rokhlin, G.N. Technical description of Xenon short arc lamp. In Gas Discharge Light Sources; Énergoatomizdat: Moscow, Russia, 1991; p. 691. (In Russian) [Google Scholar]
- Souptel, D.; Behr, G.; Ivanenko, L.; Vinzelberg, H.; Schumann, J. Floating zone growth and characterization of semiconducting Ru2Si3 single crystals. J. Cryst. Growth 2002, 244, 296–304. [Google Scholar] [CrossRef]
- Sampathkumaran, E.V.; Mohapatra, N.; Iyer, K.K.; Cao, C.D.; Loeser, W.; Behr, G. Magnetic anomalies in single crystalline ErPd2Si2. J. Magn. Magn. Mater. 2008, 320, 1549–1552. [Google Scholar] [CrossRef]
- Van Hook, H.J. Phase relation in the ternary system Fe2O3-FeO-YeFeO3. J. Am. Ceram. Soc. 1962, 45, 162–165. [Google Scholar] [CrossRef]
- Van Hook, H.J. Phase equilibrium studies in the system iron oxide Y2O3-Gd2O3. J. Am. Ceram. Soc. 1962, 45, 369–373. [Google Scholar] [CrossRef]
- Van Hook, H.J. Thermal stability of barium ferrite (BaFe12O19). J. Am. Ceram. Soc. 1964, 47, 579–581. [Google Scholar] [CrossRef]
- Nabokin, P.I.; Souptel, D.; Balbashov, A.M. Floating zone crystal growth and properties of SrTiO3. J. Cryst. Growth 2003, 250, 397–404. [Google Scholar] [CrossRef]
- Vocabulary Landolt-Bornstein.








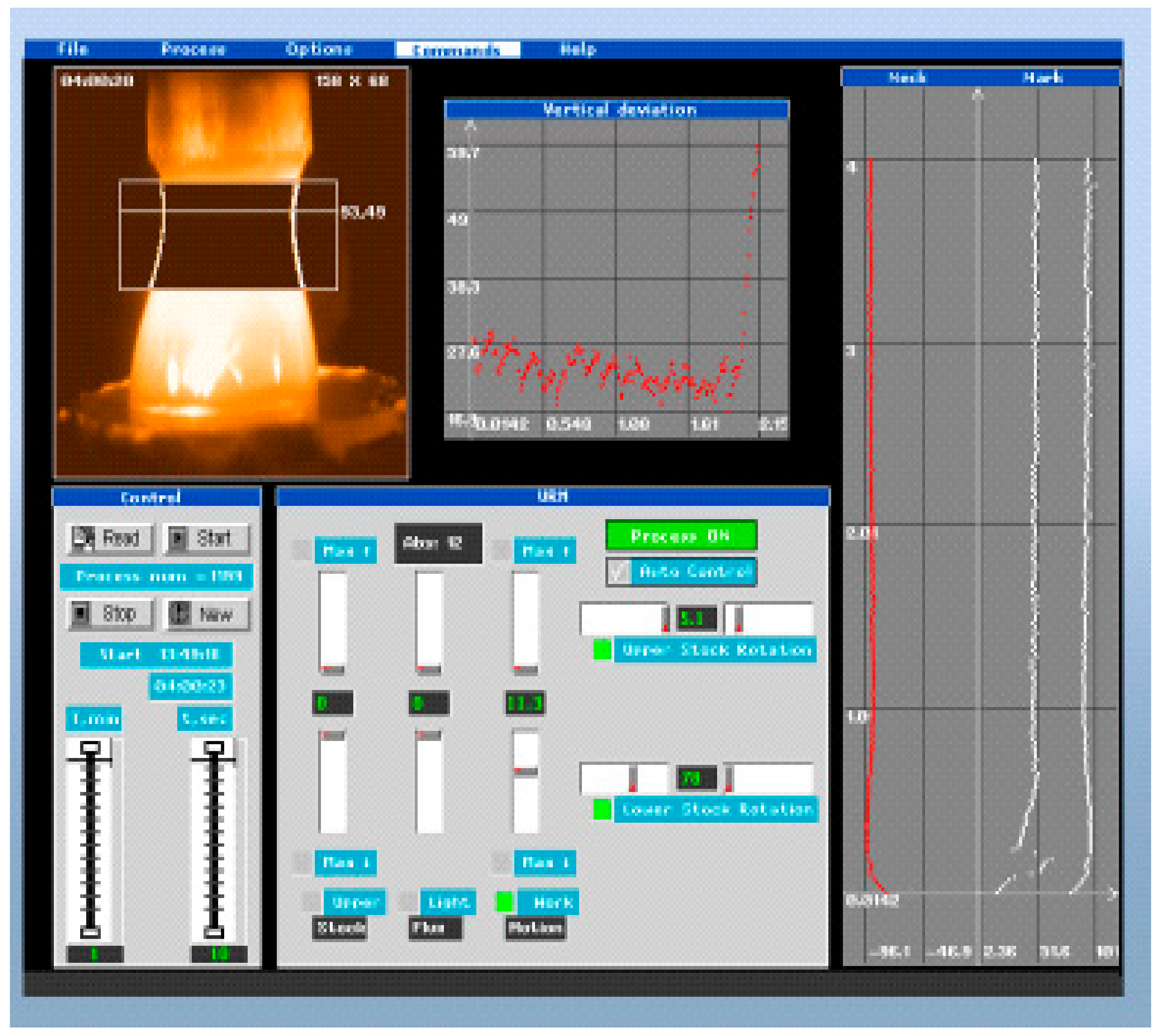


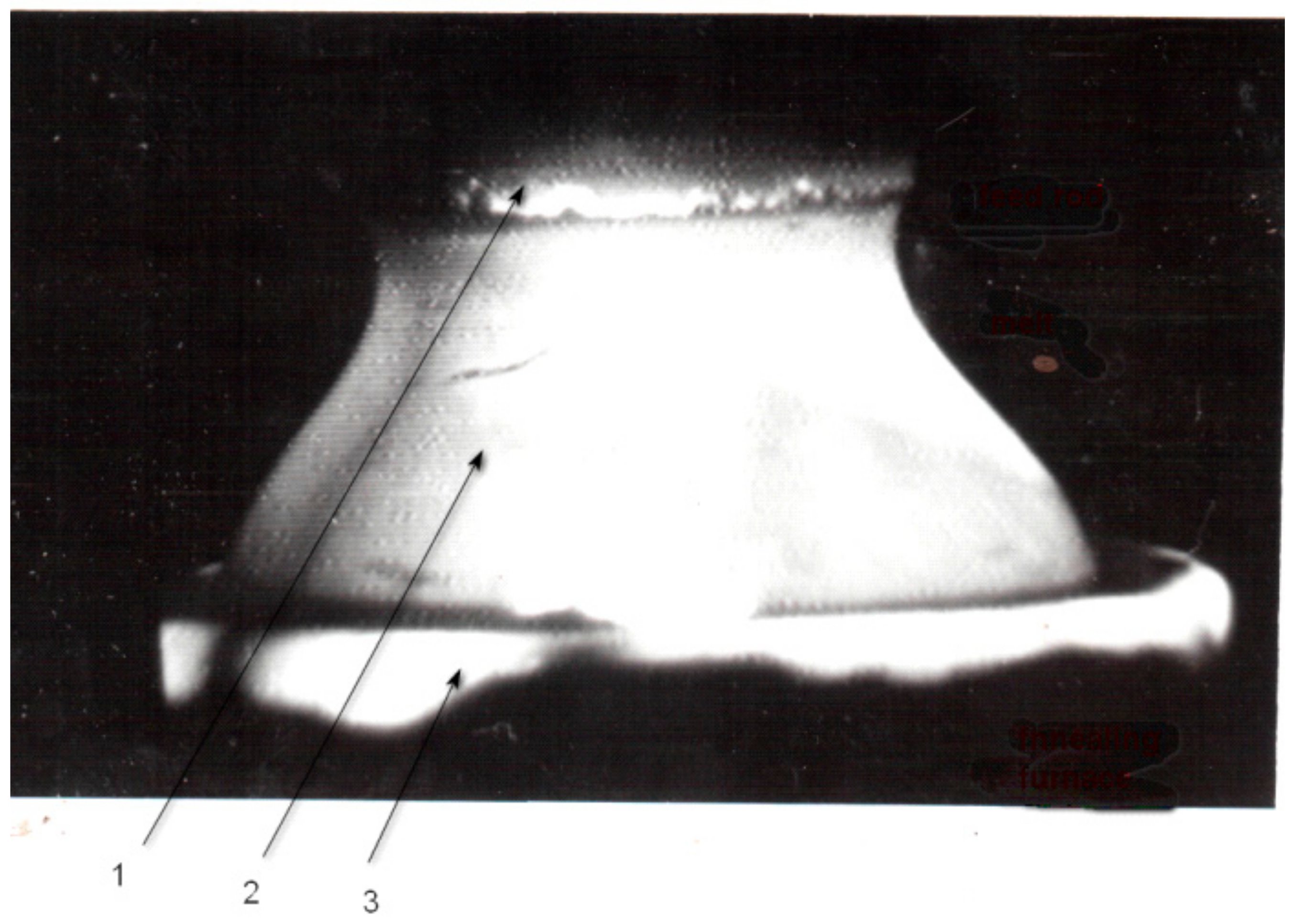



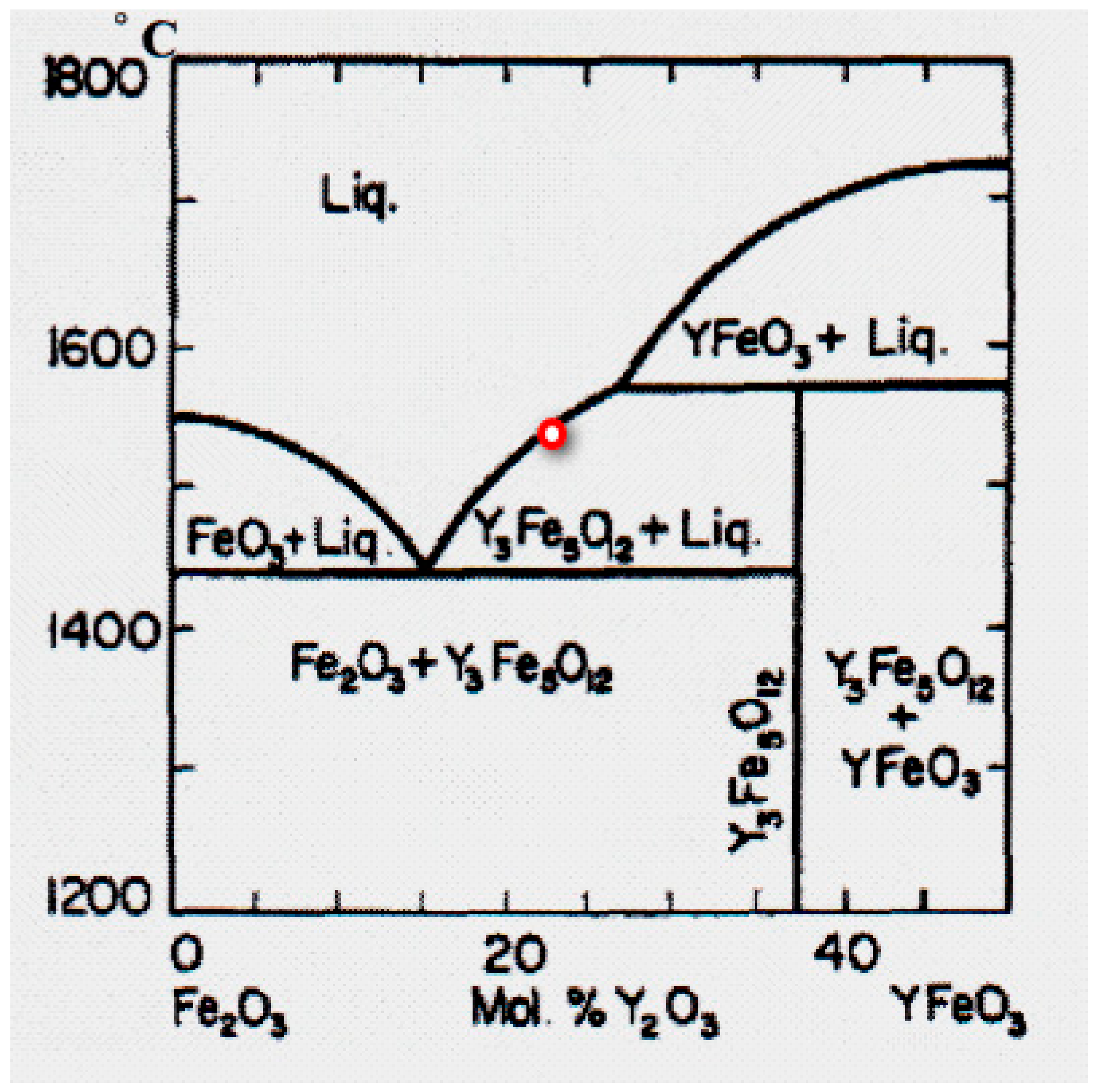
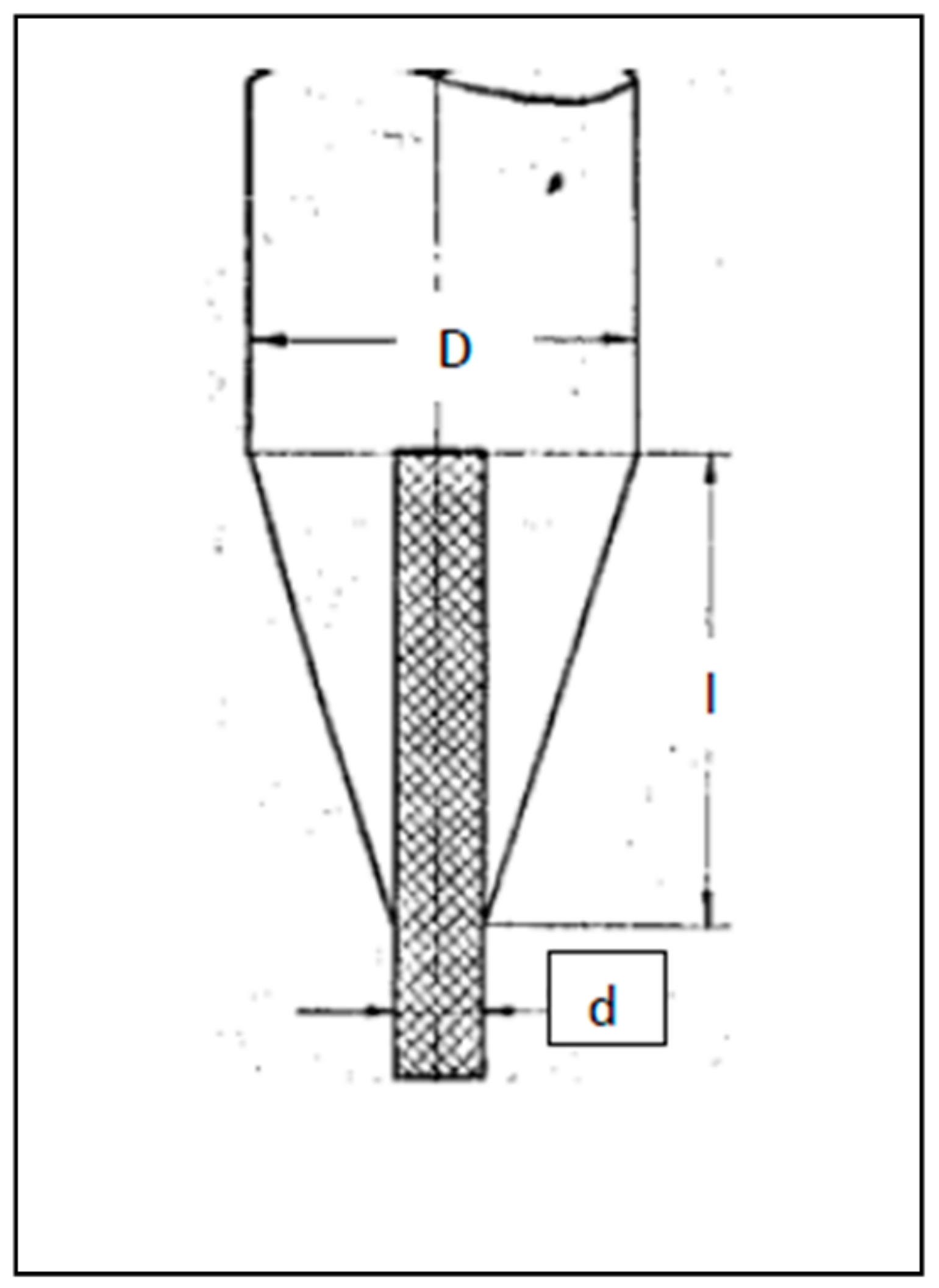



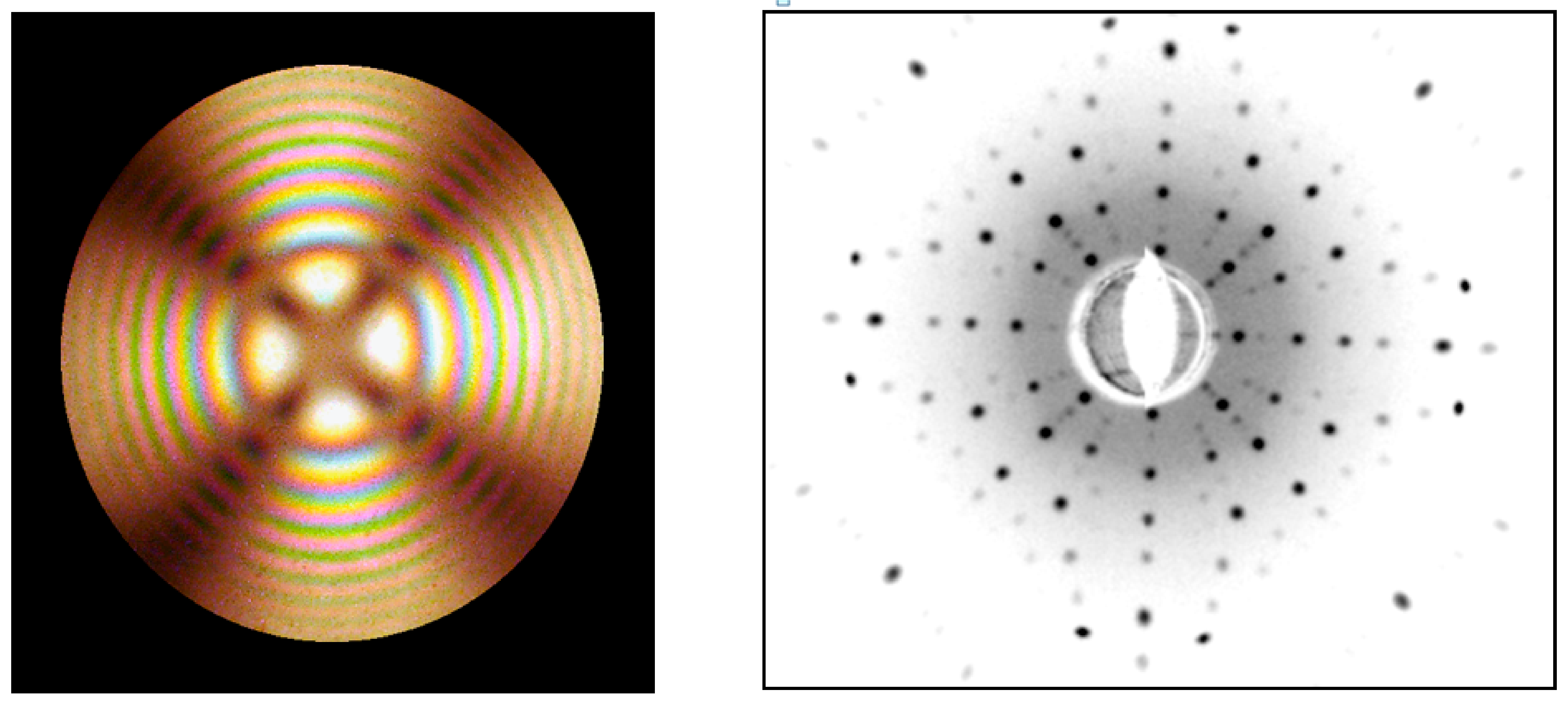
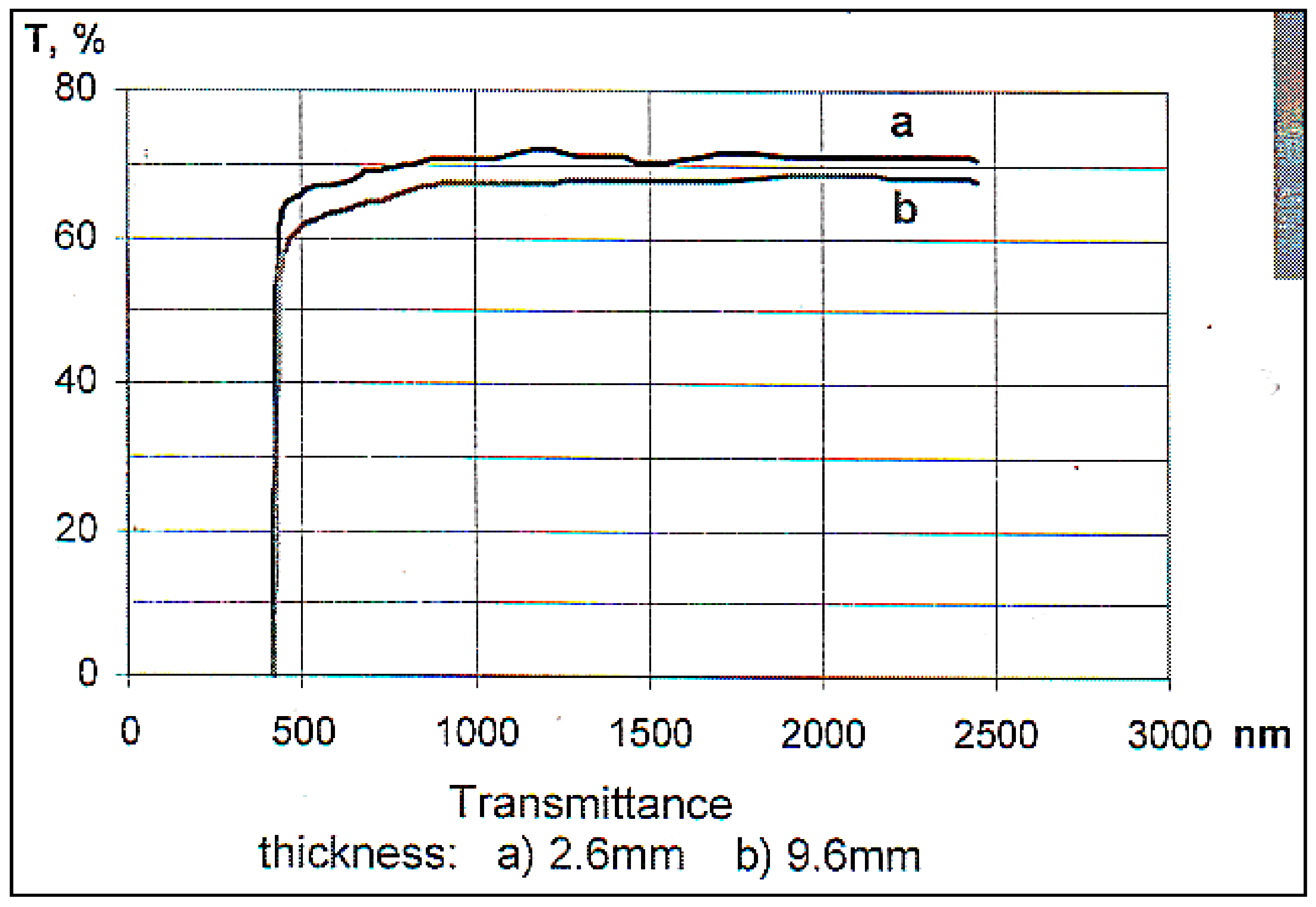
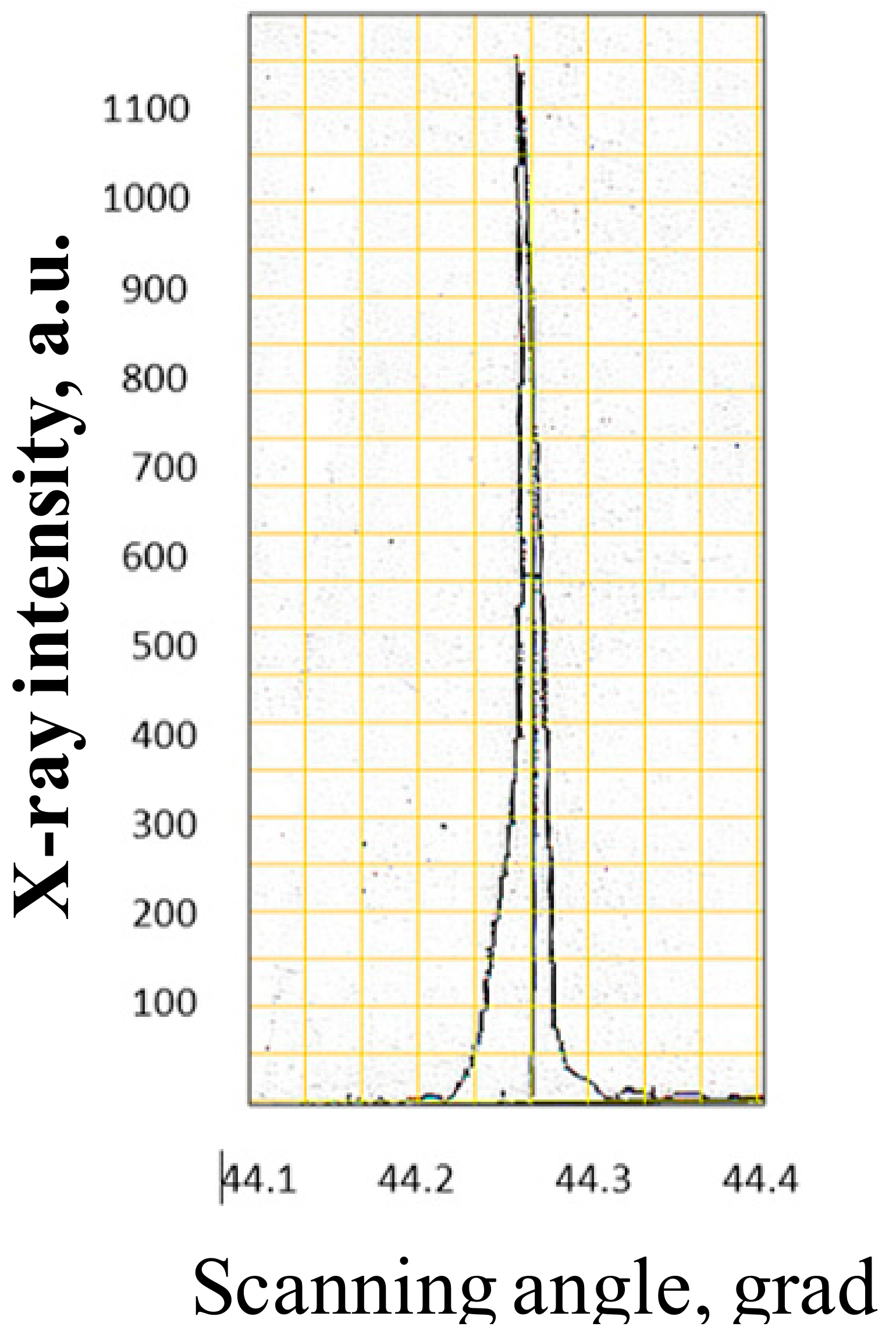

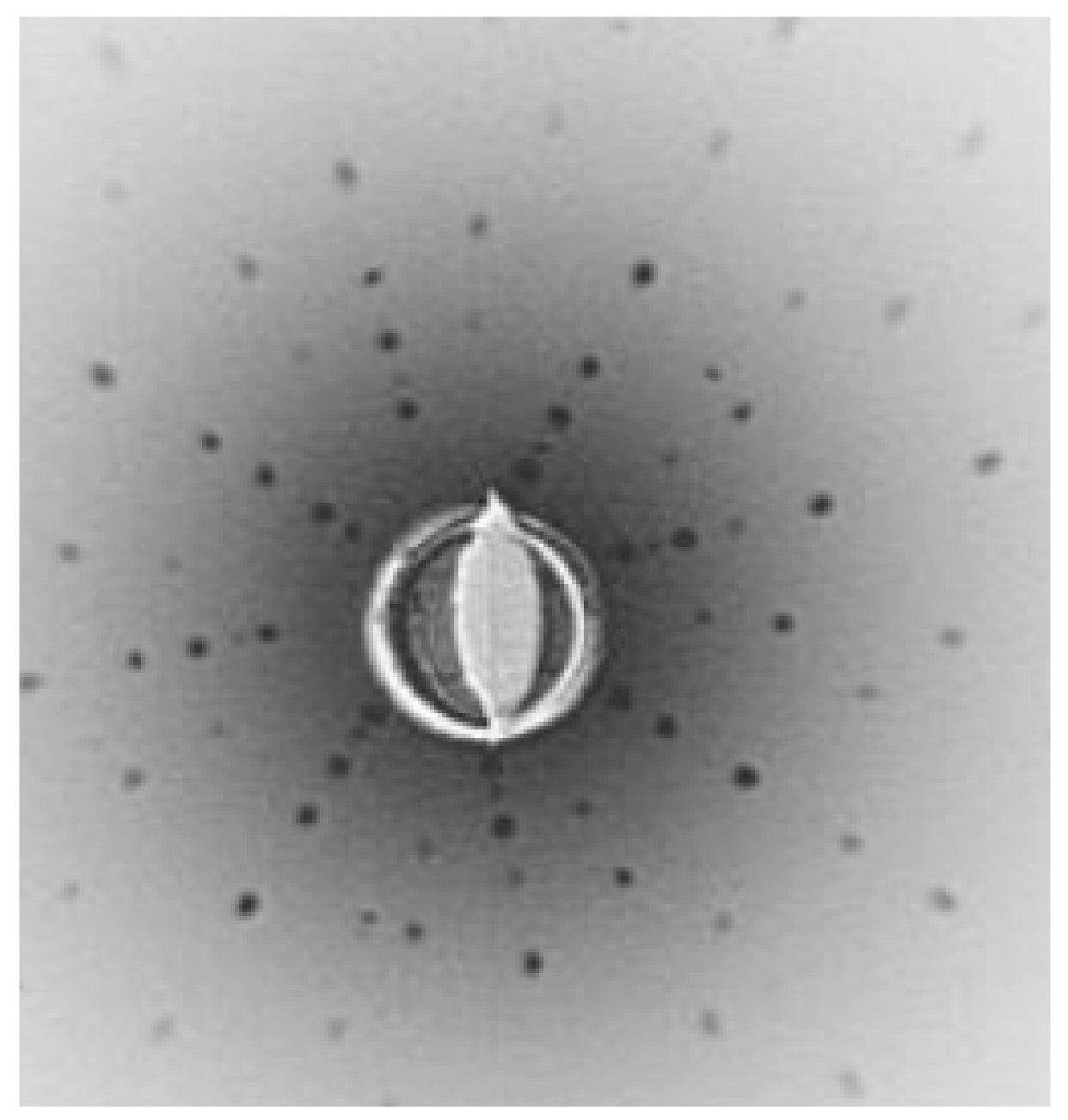

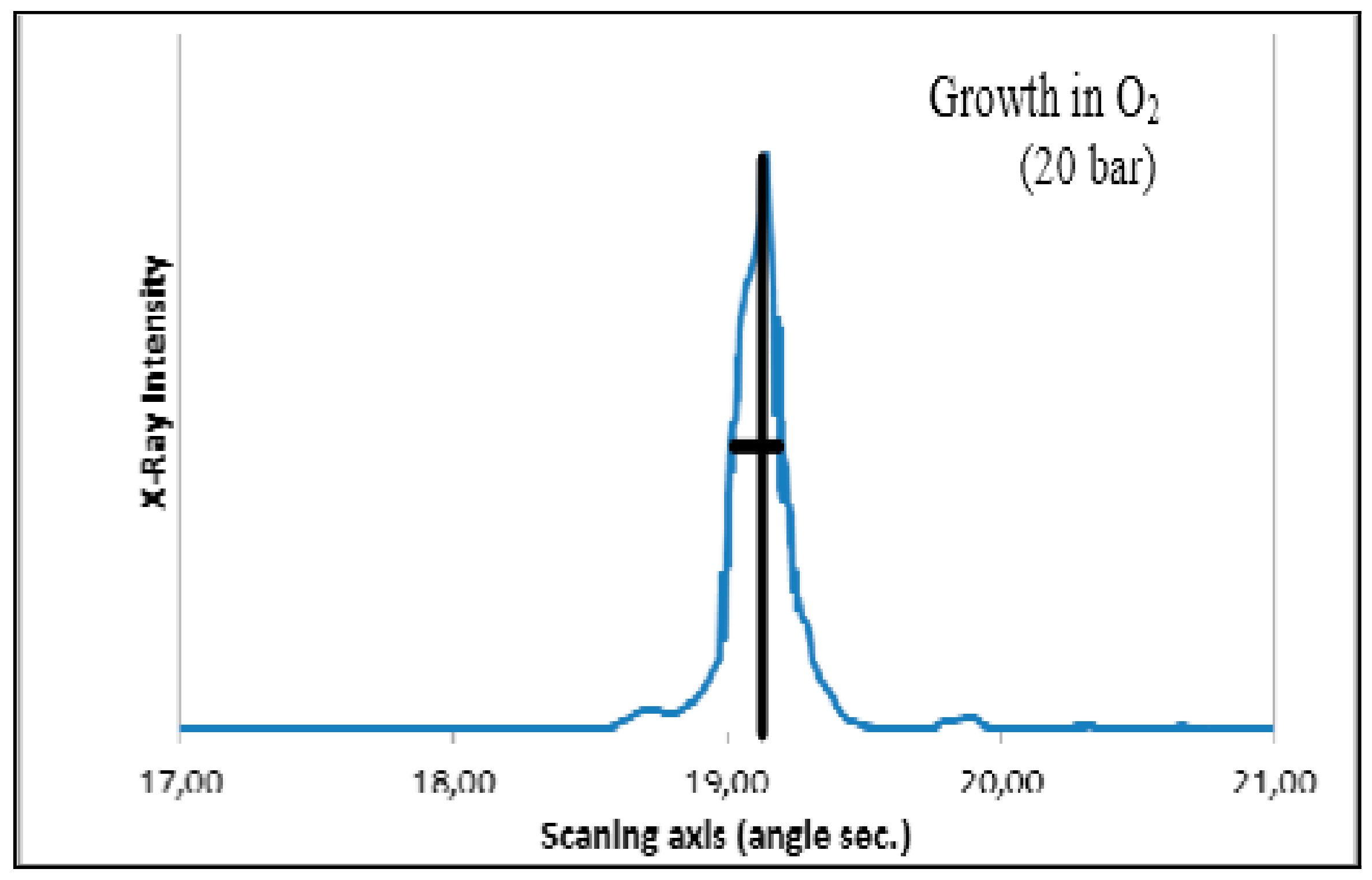
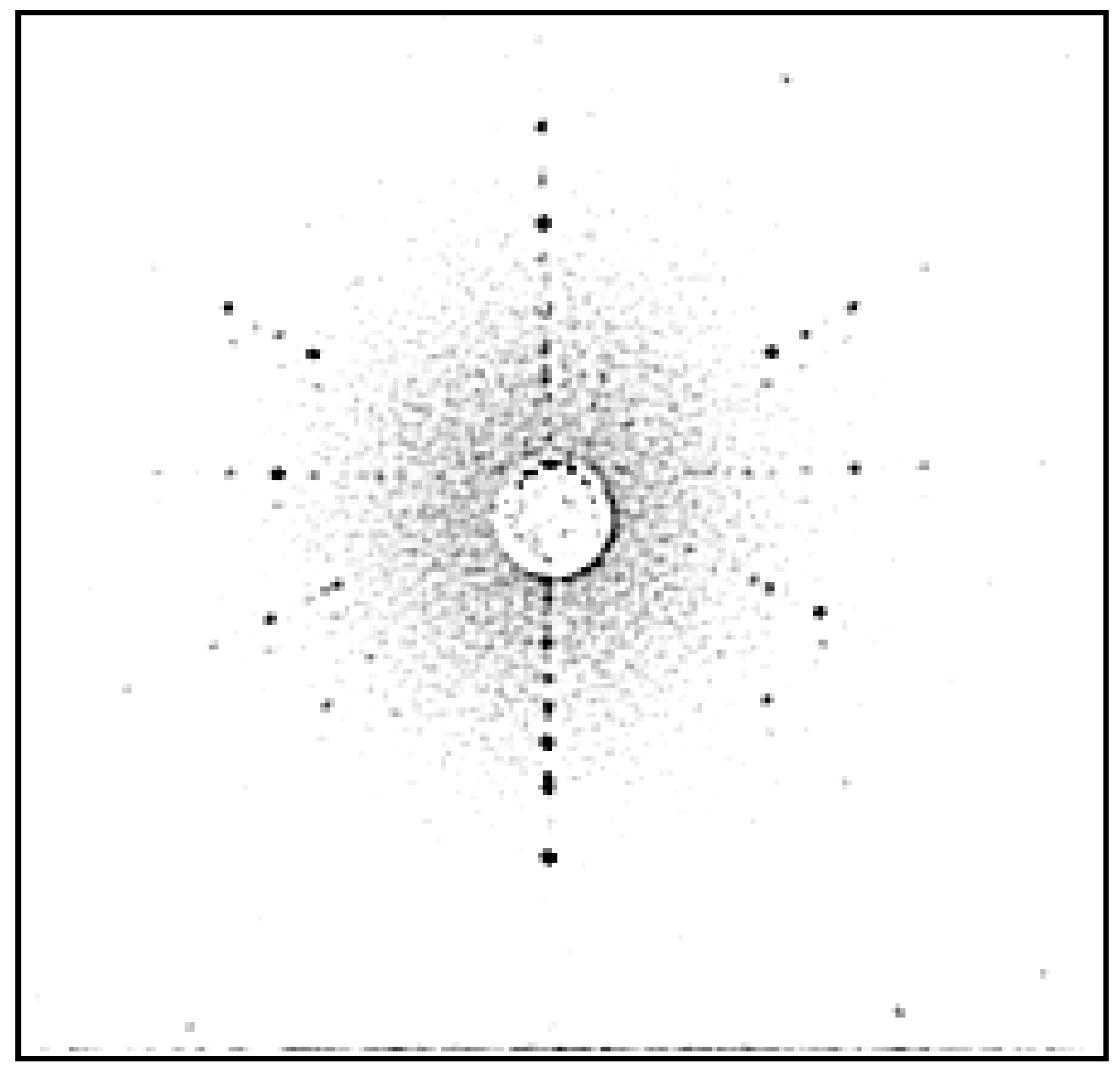
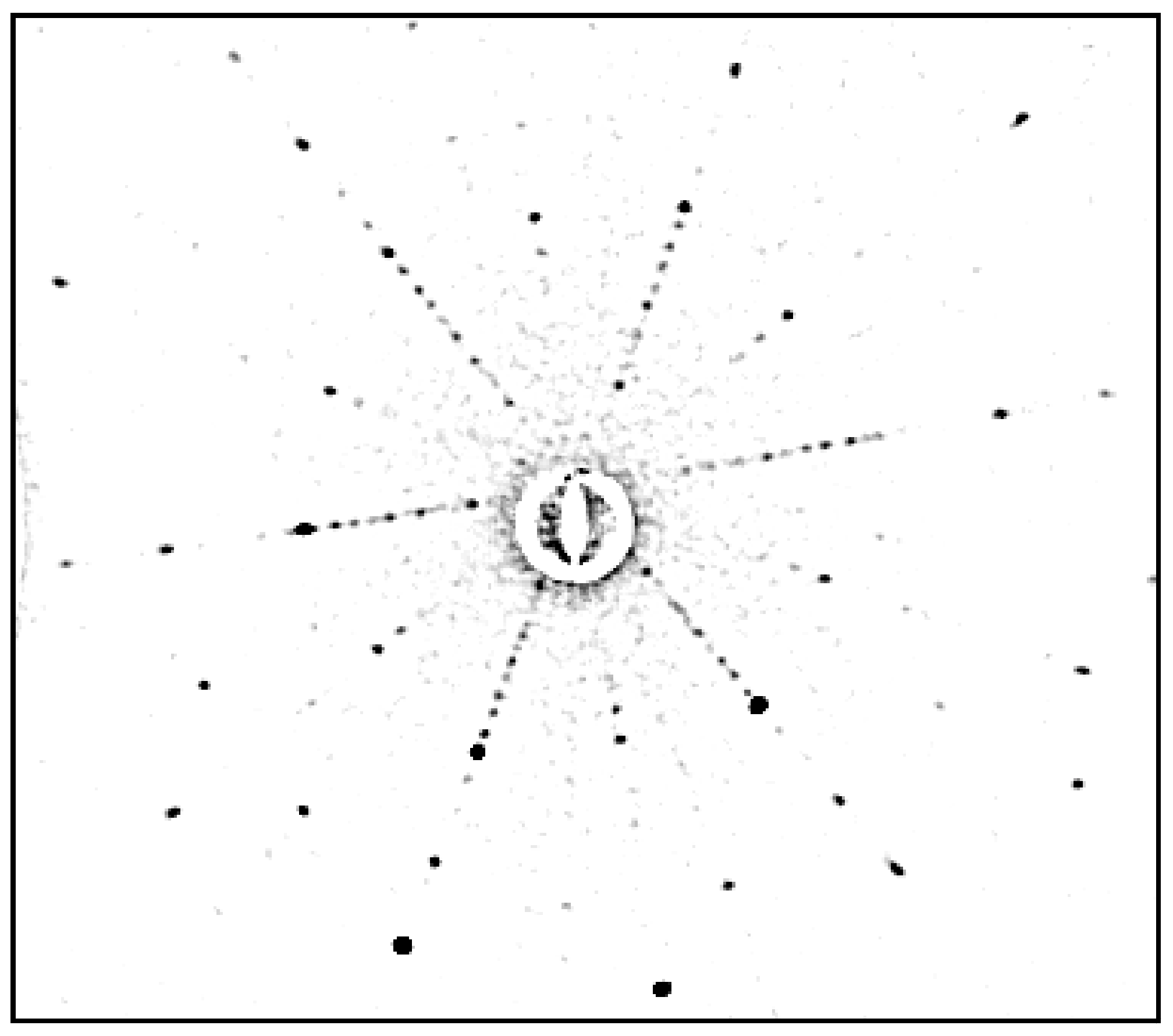


| Composition | Crystal Growth Speed, mm/h | Rotation Crys/Feed Rod r/min. | Gas Environment/Pressure atm. | Crystal Size Ǿ x L, mm | Quality Estimation |
|---|---|---|---|---|---|
| RMnO3 (R-La-Lu) | 8 | 40/5 | Air | 6 × 40 | Laue pattern, cut of oriented samples, powder diffraction. |
| LaSrMnO3 RCaMnO3 (R-La,Sm,Y) | 8 4–6 | 40/5 | Ar flow | 6 × 40 | |
| SmSrMnO3 | 4–6 | 40/5 | Ar, flow | 6 × 40 | |
| TbMnO3 | 4 | Ar, 30 | 6 × 40 |
| Composition | Crystal Growth Speed, mm/h | Rotation Cryst./Feed Rod r/min | Gas Environment/Pressure, atm. | Crystal Size Ǿ × L, mm | Quality Estimation |
|---|---|---|---|---|---|
| Ba(Sr)Fe12O19(M) | 8 | 30/5 | Oxygen, 50 | 6 × 40 | Laue pattern, powder diffraction, cut of oriented samples, torque in the magnetic field. Electrical resistivity measurement. Additional annealing of crystals in oxygen flow, SEM (Figure 38) |
| Ba(Sr)AlxFe12-xO19(M) x = 1.0–6.0 | 6 | 30/5 | Oxygen, 50 | 6 × 40 | |
| Ba(CoTi)xFe12-xO19 (M) x = 1.0–1.8 | 6 | 30/5 | Oxygen, 50 | 6 × 40 | |
| Ba(Sr)ScxFe12-xO19 (M) x = 1.0–1.8 | 8 | 30/5 | Oxygen, 50 | 6 × 40 | |
| Ba(Sr)Co2Fe16O27 (W) | 6 | 30/5 | Oxygen, 50 | 6 × 40 | |
| Ba2Zn2Fe12O22 (Y) | 6 | 30/5 | Oxygen, 50 | 6 × 40 | |
| Ba2Co2Fe12O22 (Y) | 6 | 30/5 | Oxygen, 50 | 6 × 40 | |
| Ba(Sr)2Co2Fe28O45 (X) | 6 | 30/5 | Oxygen, 50 | 6 × 40 | |
| Ba(Sr)3Co4Fe24O41 (Z) | 6 | 30/5 | Oxygen, 50 | 6 × 40 | |
| Ba(Sr)4Co4Fe36O60 (U) | 6 | 30/5 | Oxygen, 50 | 6 × 40 |
| Composition | Crystal Growth Speed, mm/h | Rotation Cryst./feed Rod r/min | Gas Environment/Pressure | Crystal Size Ǿ × L, mm | Quality Estimation |
|---|---|---|---|---|---|
| NiWO4 | 6 | 30/5 | Ar | 6 × 40 | Laue pattern, cut of oriented samples, powder diffraction. (Figure 39) |
| FeWO4 | 6 | 30/5 | Ar | 6 × 40 | |
| CoWO4 | 6 | 30/5 | Air | 6 × 40 | |
| CoxMn1−xWO4 X = 0.05–0.2 | 4 | 305 | Air | 6 × 40 |
| Composition | Crystal Growth Speed, mm/h | Rotation Cryst./Feed Rod r/min | Gas Environment/Pressure | Crystal Size Ǿ × L, mm | Quality Estimation |
|---|---|---|---|---|---|
| Ca2Fe2O5 | 6 | 30/5 | Ar, flow | 8 × 40 | Laue pattern, cut of oriented samples, powder diffraction. |
| Sr2Fe2O5 | 6 | 30/5 or 0/5 | Ar, flow | 8 × 40 | |
| Ca2FeAlO5 | 2 | 30/5 or 0/5 | Ar, flow | 6 × 40 | |
| Ca2FeGaO5 | 4 | 30/5 | Ar, flow | 6 × 40 |
| Composition | Crystal Growth Speed, mm/h | Rotation Cryst./Feed Rod, r/min | Gas Environment/Pressure, atm. | Crystal Size Ǿ × L, mm. | Quality Estimation |
|---|---|---|---|---|---|
| Ba3NbFe3Si2O14 | 4 | 30/5 | Ar flow | 6 × 40 | Laue pattern, powder diffraction, cut of oriented samples ( Figure 40; Figure 41) |
| Ba3TaFe3Si2O14 | 4 | 30/5 | Ar flow | 6 × 40 |
| Composition | Crystal Growth Speed, mm/h | Rotation Cryst./Feed Rod, r/min | Gas Environment/Pressure, atm. | Crystal Size ǾxL, mm | Quality Estimation |
|---|---|---|---|---|---|
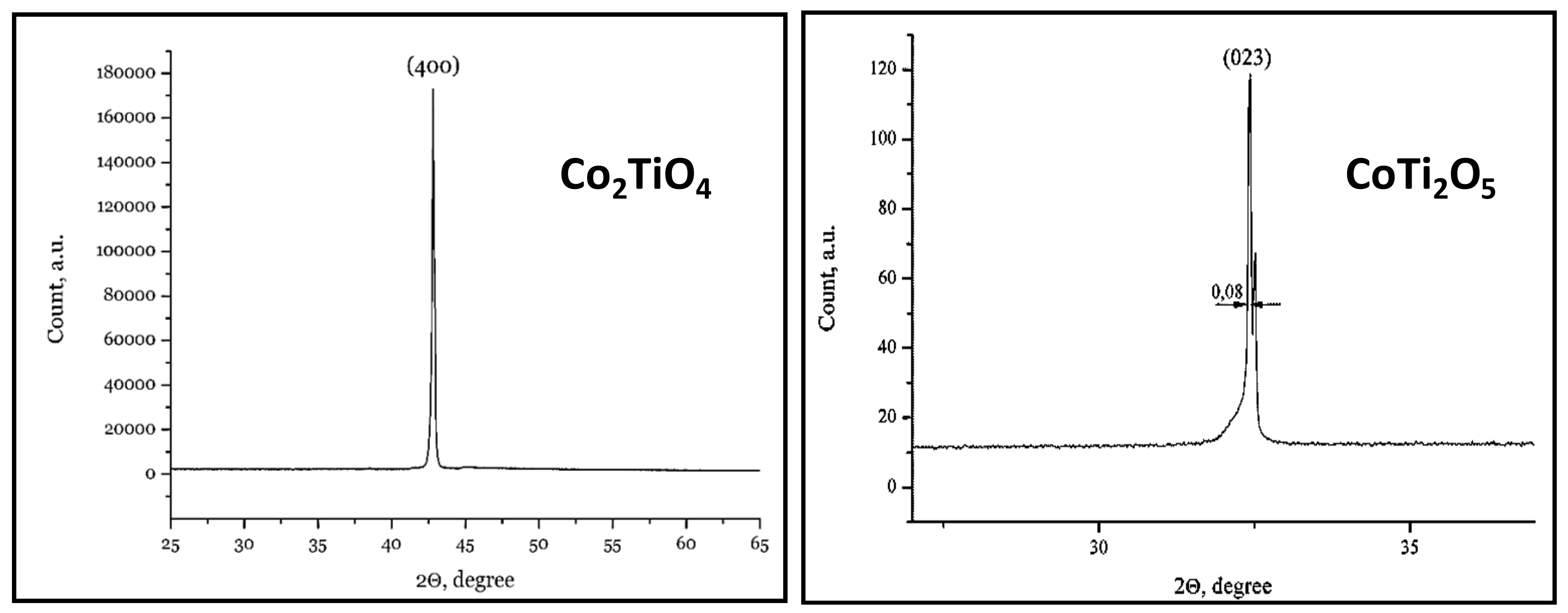
| CoTi2O5 | 8 | 30/5 | Ar flow | 6 × 40 | Laue pattern, powder diffraction, cut of oriented samples (Figure 42) |
| Co2TiO4 | 8 | 30/5 | Ar flow | 6 × 40 | |
| CoTiO3 | 6 | 30/5 | Oxygen, 30 |
| Single crystal | Melting Point, °C | Growth Rate, mm/h; Ambient | Crystal Diameter, mm | Crystal Quality | Photo of Crystal |
|---|---|---|---|---|---|
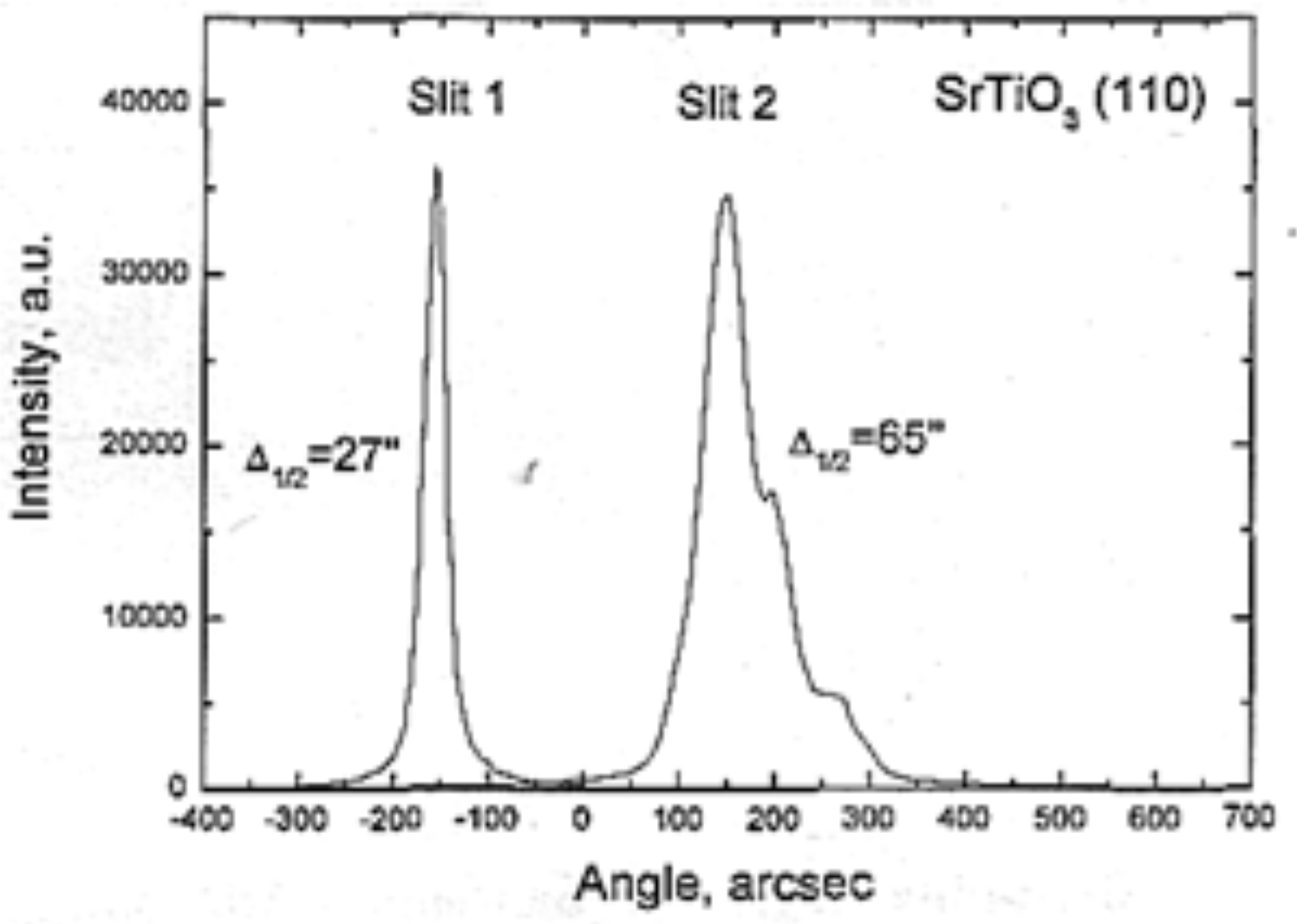
| SrTiO3 | 2030 | 7–10 air | 15–25 | FWHM: Г = 30″ |  |
| TiO2 | 1850 | 10 CO2 | 20–25 | Optical absorption: α = 0.03–0.08 cm−1 (λ = 4.5–0.55 µ) FWHM: Г = 25″ |  |
| α - Fe2O3 | 1600 | 10 50 atm O2 | 10–12 | Optical absorption: α < 1 cm−1 (λ > 1.3 µ) |  |
| Fe3O4 | 1580 | 10 CO2 | 10–12 | Magnetic semiconductor |  |
| Y3Fe5O12 | 1470 | 1–3 25 atm O2 | 5–8 | FMR width: ΔH = 0.4 Oe at 10 GHz Optical absorption: α < 0.1 cm−1 (λ > 1.15 µ) |  |
| SrFe12O19 | 1650 | 6 60 atm O2 | 5–8 | FMR width: ΔH = 18 Oe at 35 GHz |  |
| NiFe2O4 CoFe2O4 (NiZn) Fe2O4 | 1600 | 10 60 atm O2 | 10–12 | Magnetic permeability: µ = 104 G/Oe |  |
| YFeO3 | 1700 | 3–10 air | 15 | FWHM: Г < 4′ Optical absorption: α < 1 cm−1 (λ > 1.2 µ) |  |
| ZrO2 | 2700 | 10–20 air | 10 | Good structure quality |  |
| NaNO3 | 305 | air | 10 | High polarization parameters |  |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
M. Balbashov, A. Contemporary Apparatus for Single Crystals Growth of Oxide Compounds and Metals by Optical Floating Zone (FZ). Crystals 2019, 9, 487. https://doi.org/10.3390/cryst9100487
M. Balbashov A. Contemporary Apparatus for Single Crystals Growth of Oxide Compounds and Metals by Optical Floating Zone (FZ). Crystals. 2019; 9(10):487. https://doi.org/10.3390/cryst9100487
Chicago/Turabian StyleM. Balbashov, Anatoly. 2019. "Contemporary Apparatus for Single Crystals Growth of Oxide Compounds and Metals by Optical Floating Zone (FZ)" Crystals 9, no. 10: 487. https://doi.org/10.3390/cryst9100487
APA StyleM. Balbashov, A. (2019). Contemporary Apparatus for Single Crystals Growth of Oxide Compounds and Metals by Optical Floating Zone (FZ). Crystals, 9(10), 487. https://doi.org/10.3390/cryst9100487




