Synthesis and Crystal Structure of a Zn(II)-Based MOF Bearing Neutral N-Donor Linker and SiF62− Anion
Abstract
:1. Introduction
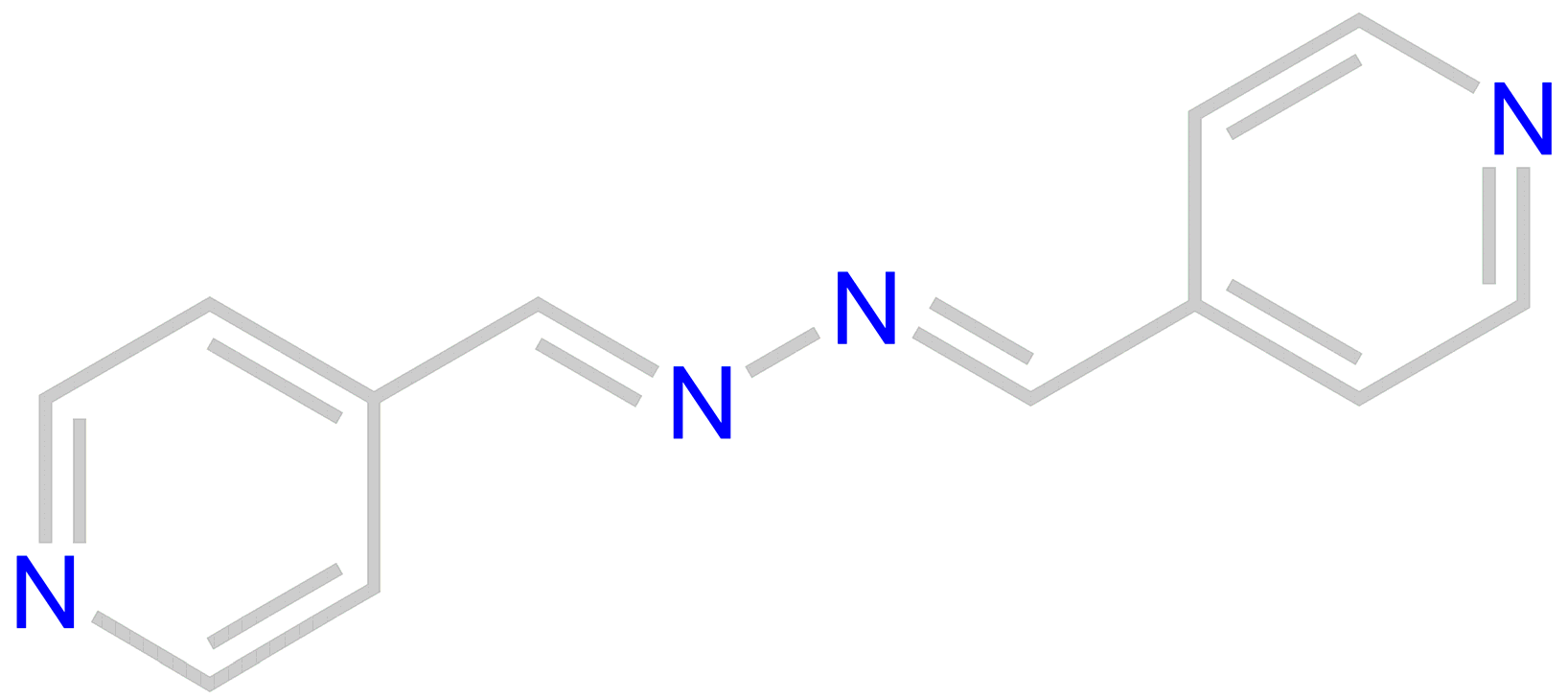
2. Materials and Methods
Experimental Synthesis
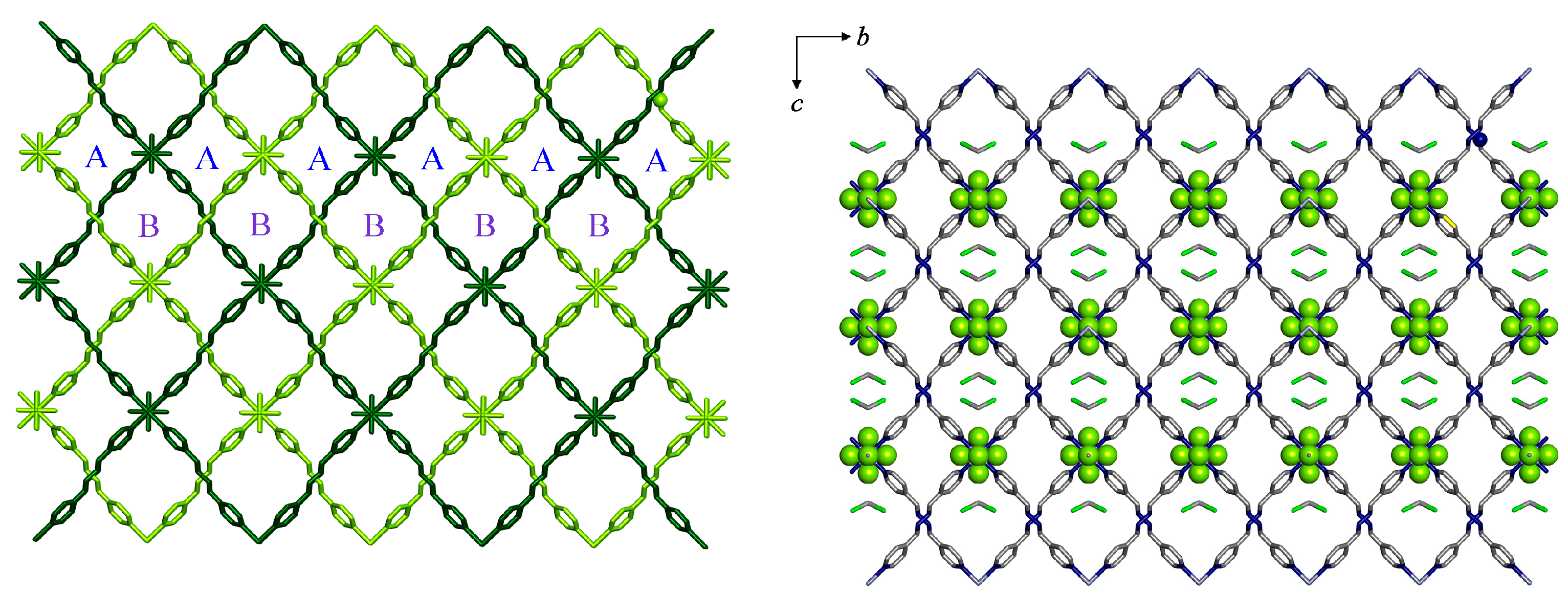
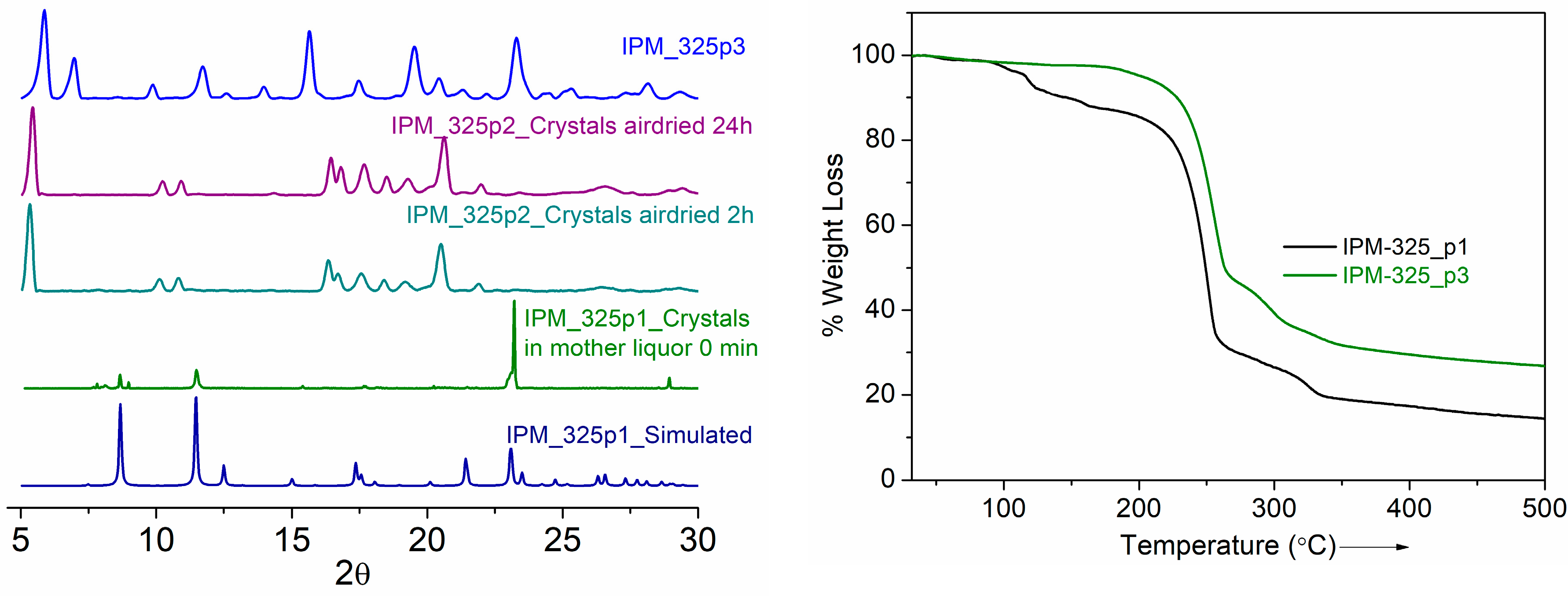
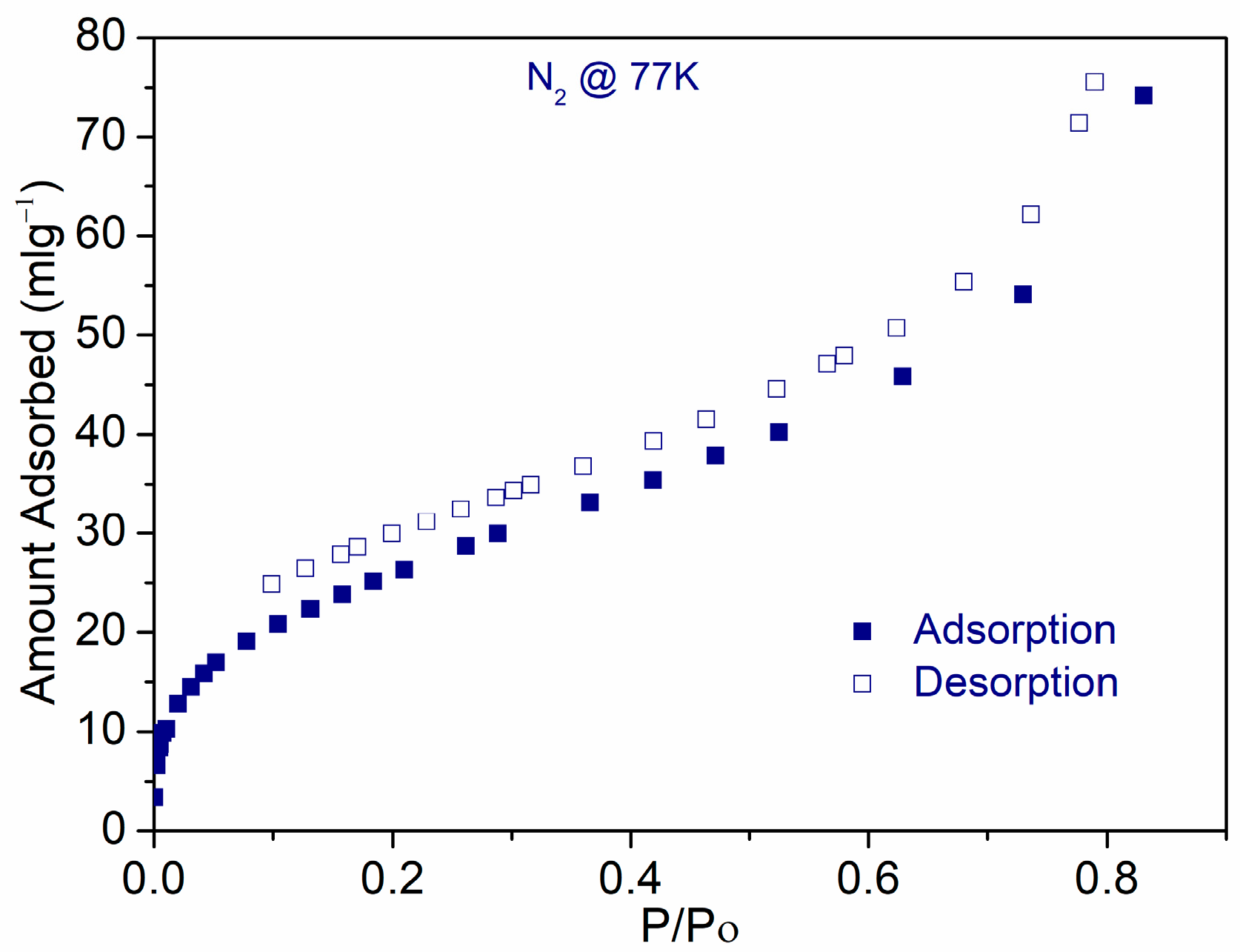
3. Results and Discussions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Furukawa, H.; Cordova, K.E.; O’Keeffe, M.; Yaghi, O.M. The chemistry and Applications of metal-organic frameworks. Science 2013, 341, 974. [Google Scholar] [CrossRef] [PubMed]
- Ouay, B.L.; Kitagawa, S.; Uemura, T. Opening of an accessible microporosity in an otherwise nonporous metal–organic framework by polymeric guests. J. Am. Chem. Soc. 2017, 139, 7886–7892. [Google Scholar] [CrossRef] [PubMed]
- Czaja, A.U.; Trukhan, N.; Muller, U. Industrial applications of metal–organic frameworks. Chem. Soc. Rev. 2009, 38, 1284–1293. [Google Scholar] [CrossRef] [PubMed]
- Lustig, W.P.; Mukherjee, S.; Rudd, N.D.; Desai, A.V.; Li, J.; Ghosh, S.K. Metal–organic frameworks: Functional luminescent and photonic materials for sensing applications. Chem. Soc. Rev. 2017, 46, 3242–3285. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Farha, O.K.; Roberts, J.; Scheidt, K.A.; Nguyen, S.T.; Hupp, J.T. Metal–organic framework as catalysts. Chem. Soc. Rev. 2009, 38, 1450. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Stock, N. Synthesis of metal–organic frameworks (MOFs): Routes to various MOF topologies, morphologies, and composites. Chem. Rev. 2012, 112, 933–969. [Google Scholar]
- Zhou, H.-C.J.; Kitagawa, S. Metal–Organic Frameworks (MOFs). Chem. Soc. Rev. 2014, 43, 5415–5418. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Wei, Z.; Gu, Z.-Y.; Liu, T.-F.; Park, J.; Park, J.; Tian, J.; Zhang, M.; Zhang, Q.; Gentle, T., III; et al. Tuning the structure and function of metal–organic frameworks via linker design. Chem. Soc. Rev. 2014, 43, 5561–5593. [Google Scholar] [CrossRef] [PubMed]
- Lalonde, M.; Bury, W.; Karagiaridi, O.; Brown, Z.; Hupp, J.T.; Farha, O.K. Transmetalation: Routes to metal exchange within metal–organic frameworks. J. Mater. Chem. A 2013, 1, 5453–5468. [Google Scholar] [CrossRef]
- Brozek, C.K.; Dinca, M. Cation exchange at the secondary building units of metal–organic frameworks. Chem. Soc. Rev. 2014, 43, 5456–5467. [Google Scholar] [CrossRef] [PubMed]
- Schoedel, A.; Li, M.; Li, D.; O’Keeffe, M.; Yaghi, O.M. Structures of metal–organic frameworks with rod secondary building units. Chem. Rev. 2016, 116, 12466–12535. [Google Scholar] [CrossRef] [PubMed]
- Rimoldi, M.; Howarth, A.J.; DeStefano, M.R.; Lin, L.; Goswami, S.; Li, P.; Hupp, J.T.; Farha, O.K. Catalytic zirconium/hafnium-based metal–organic frameworks. ACS Catal. 2017, 7, 997–1014. [Google Scholar] [CrossRef]
- Noro, S.-I.; Kitaura, R.; Kondo, M.; Kitagawa, S.; Ishii, T.; Matsuzaka, H.; Yamashita, M. Framework engineering by anions and porous functionalities of Cu(II)/4,4′-bpy coordination polymers. J. Am. Chem. Soc. 2002, 124, 2568–2583. [Google Scholar] [CrossRef] [PubMed]
- Karmakar, A.; Desai, A.V.; Ghosh, S.K. Ionic metal-organic frameworks (iMOFs): Design principles and applications. Coord. Chem. Rev. 2016, 307, 313–341. [Google Scholar] [CrossRef]
- Manna, B.; Desai, A.V.; Ghosh, S.K. Neutral N-donor ligand based flexible metal-organic frameworks. Dalton Trans. 2016, 45, 4060–4072. [Google Scholar] [CrossRef] [PubMed]
- Eddaoudi, M.; Sava, D.F.; Eubank, J.F.; Adil, K.; Guillerm, V. Zeolite-like metal-organic frameworks (ZMOFs): Design, synthesis, and properties. Chem. Soc. Rev. 2015, 44, 228–249. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.; Zaworotko, M.J. Porous solids by design: [Zn(4,4′-bpy)2(SiF6)]n x DMF, a single framework octahedral coordination polymer with large square channels. Angew. Chem. Int. Ed. 1995, 34, 2127. [Google Scholar] [CrossRef]
- Noro, S.-I.; Kitagawa, S.; Kondo, M.; Kenji, S. New methane adsorbent, porous coordination polymer, [Cu(SiF6)(4,4′-bipyridine)2]n. Angew. Chem. Int. Ed. 2000, 39, 2081–2084. [Google Scholar] [CrossRef]
- Shekhah, O.; Belmabkhout, Y.; Chen, Z.; Guillerm, V.; Cairns, A.; Adil, K.; Eddaoudi, M. Made-to-order metal–organic frameworks for trace carbon dioxide removal and air capture. Nat. Commun. 2014, 5, 4228. [Google Scholar] [CrossRef] [PubMed]
- Burd, S.D.; Ma, S.; Perman, J.A.; Sikora, B.J.; Snurr, R.Q.; Thallapally, P.K.; Tian, J.; Wojtas, L.; Zaworotko, M.J. Highly Selective Carbon Dioxide Uptake by [Cu(bpy-n)2(SiF6)] (bpy-1 = 4,4′ -Bipyridine; bpy-2 = 1,2-Bis(4-pyridyl)ethene). J. Am. Chem. Soc. 2012, 134, 3663. [Google Scholar] [CrossRef] [PubMed]
- Nugent, P.; Belmabkhout, Y.; Burd, S.D.; Cairns, A.J.; Luebke, R.; Forrest, K.; Pham, T.; Ma, S.; Space, B.; Wojtas, L.; et al. Porous materials with optimal adsorption thermodynamics and kinetics for CO2 separation. Nature 2013, 495, 80–84. [Google Scholar] [CrossRef] [PubMed]
- O'Nolan, D.; Kumar, A.; Zaworotko, M.J. Water Vapour Sorption in Hybrid Pillared Square Grid Materials. J. Am. Chem. Soc. 2017, 139, 8508–8513. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yang, Q.; Cui, X.; Yang, L.; Bao, Z.; Ren, Q.; Xing, H. Sorting of C4 olefins with interpenetrated hybrid ultramicroporous materials by combining molecular recognition and size-sieving. Angew. Chem. Int. Ed. 2017, 56, 16282–16287. [Google Scholar] [CrossRef] [PubMed]
- Schneemann, A.; Bon, V.; Schwedler, I.; Senkovska, I.; Kaskel, S.; Fischer, R.A. Flexible metal–organic frameworks. Chem. Soc. Rev. 2014, 43, 6062–6096. [Google Scholar] [CrossRef] [PubMed]
- Chang, Z.; Yang, D.-H.; Xu, J.; Hu, T.-L.; Bu, X.-H. Flexible metal–organic frameworks: Recent advances and potential applications. Adv. Mater. 2015, 27, 5432–5441. [Google Scholar] [CrossRef] [PubMed]
- Horike, S.; Shimomura, S.; Kitagawa, S. Soft porous crystals. Nat. Chem. 2009, 1, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Serre, C.; Millange, f.; Thouvenot, C.; Nogues, M.; Marsolier, G.; Louer, D.; Ferey, G. Very large breathing effect in the first nanoporous chromium(iii)-based solids: MIL-53 or CrIII(OH)·{O2C−C6H4−CO2}·{HO2C−C6H4−CO2H}x·H2Oy. J. Am. Chem. Soc. 2002, 124, 13519–13526. [Google Scholar] [CrossRef] [PubMed]
- Hamon, L.; Llewellyn, P.L.; Devic, T.; Ghoufi, A.; Clet, G.; Guillerm, V.; Pirngruber, G.D.; Maurin, G.; Serre, S.; Driver, G.; et al. Co-adsorption and separation of CO2-CH4 mixtures in the highly flexible MIL-53(Cr) MOF. J. Am. Chem. Soc. 2009, 131, 17490–17499. [Google Scholar] [CrossRef] [PubMed]
- SAINT Plus, Version 7.03; Bruker AXS Inc.: Madison, WI, USA, 2004.
- Krause, L.; Herbst-Irmer, R.; Sheldrick, G.M.; Stalke, D. Comparison of Silver and molybdenum microfocus X-ray sources for single crystal structure determination. J. Appl. Cryst. 2015, 48, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Farrugia, L. WinGX, version 1.80.05; University of Glasgow: Glasgow, Scotland, 2009.
- Spek, A.L. PLATON SQUEEZE: A tool for the calculation of the disordered solvent contribution to the calculated structure factors. Acta Cryst. 2015, C71, 9–18. [Google Scholar]
- Bisht, K.K.; Suresh, E. Spontaneous Resolution to Absolute Chiral Induction: Pseudo-Kagome Type Homochiral Zn(II)/Co(II) Coordination Polymers with Achiral Precursors. J. Am. Chem. Soc. 2013, 135, 15690. [Google Scholar]


| Crystal Data | IPM_MOF-325-p1 |
|---|---|
| Formula Crystal | {[Zn(L)2 (SiF6)]·(CH2Cl2)·xG}n Monoclinic |
| CCDC | 1,586,047 |
| a/Å | 7.73 (9) |
| b/Å | 20.36 (3) |
| c/Å | 12.09 (14) |
| α/° | 90 |
| β/° | 102.630 (2) |
| γ/° | 90 |
| Cell volume/Å3 | 1857.6 (4) |
| Space group | C 2/m |
| Z | 2 |
| Dx/Mg m−3 | 1.426 |
| R | 5.4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manna, B.; Sharma, S.; Ghosh, S.K. Synthesis and Crystal Structure of a Zn(II)-Based MOF Bearing Neutral N-Donor Linker and SiF62− Anion. Crystals 2018, 8, 37. https://doi.org/10.3390/cryst8010037
Manna B, Sharma S, Ghosh SK. Synthesis and Crystal Structure of a Zn(II)-Based MOF Bearing Neutral N-Donor Linker and SiF62− Anion. Crystals. 2018; 8(1):37. https://doi.org/10.3390/cryst8010037
Chicago/Turabian StyleManna, Biplab, Shivani Sharma, and Sujit K. Ghosh. 2018. "Synthesis and Crystal Structure of a Zn(II)-Based MOF Bearing Neutral N-Donor Linker and SiF62− Anion" Crystals 8, no. 1: 37. https://doi.org/10.3390/cryst8010037
APA StyleManna, B., Sharma, S., & Ghosh, S. K. (2018). Synthesis and Crystal Structure of a Zn(II)-Based MOF Bearing Neutral N-Donor Linker and SiF62− Anion. Crystals, 8(1), 37. https://doi.org/10.3390/cryst8010037




